Effects of Land Use Changes on the Plant Community Characteristics in the Wetlands of the Semi-Arid Regions
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Area
2.2. Experimental Methods
2.3. Data Analyses
3. Results
3.1. Species Composition Characteristics of Plant Communities in Wetlands
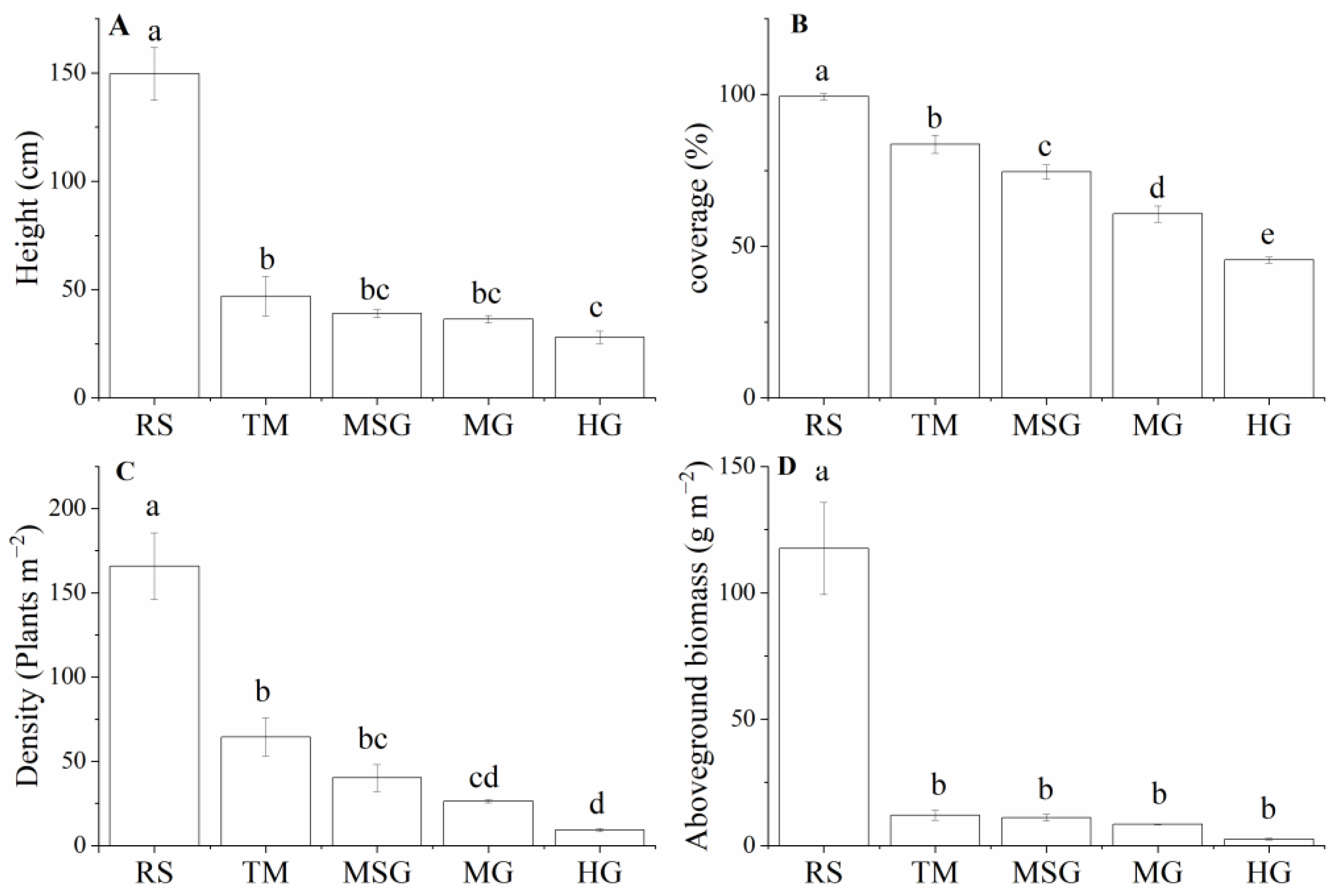
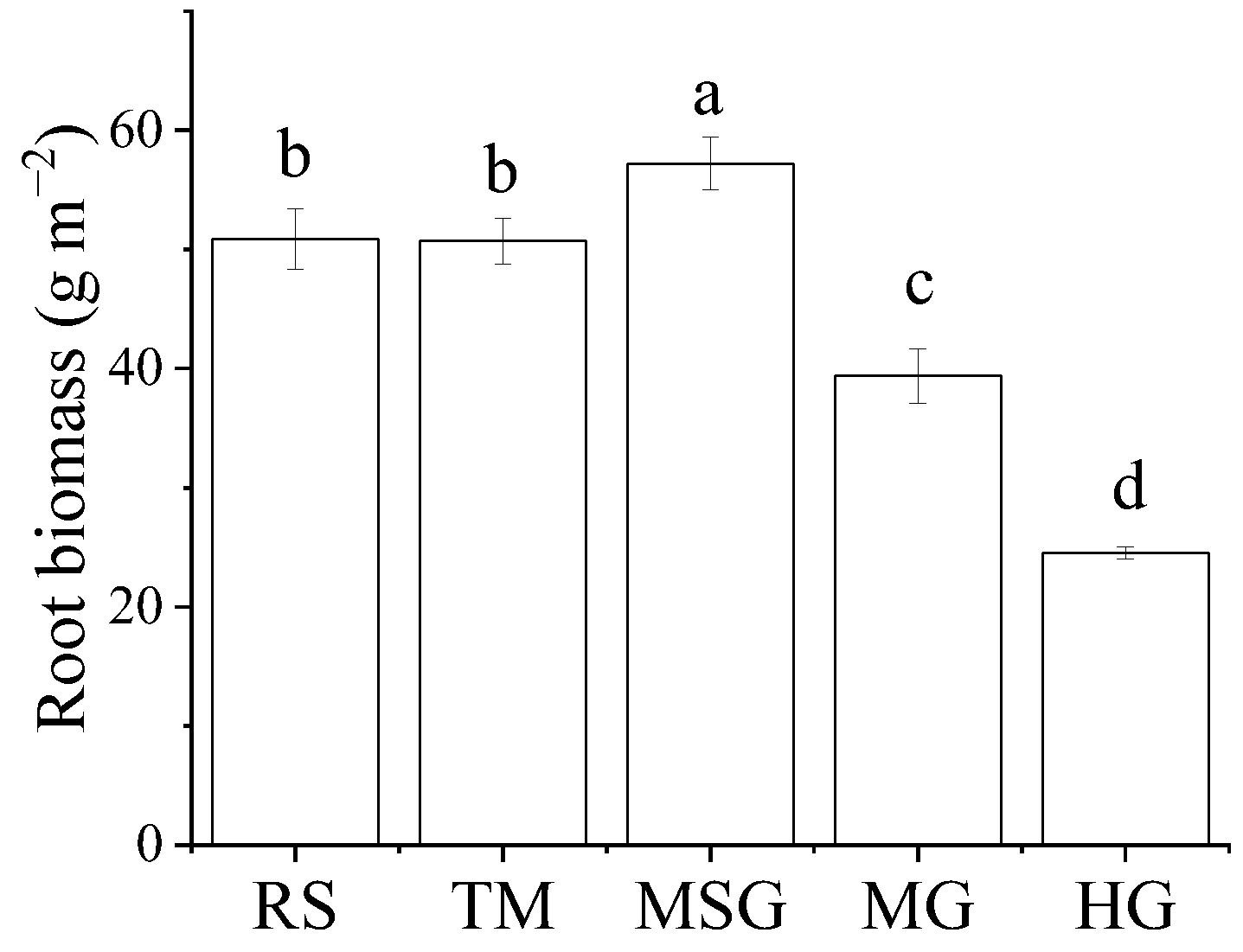
3.2. Variations of Plant Community Characteristics in Wetlands under Different Land Uses
3.3. Plant Diversity in Wetlands under Different Land Uses
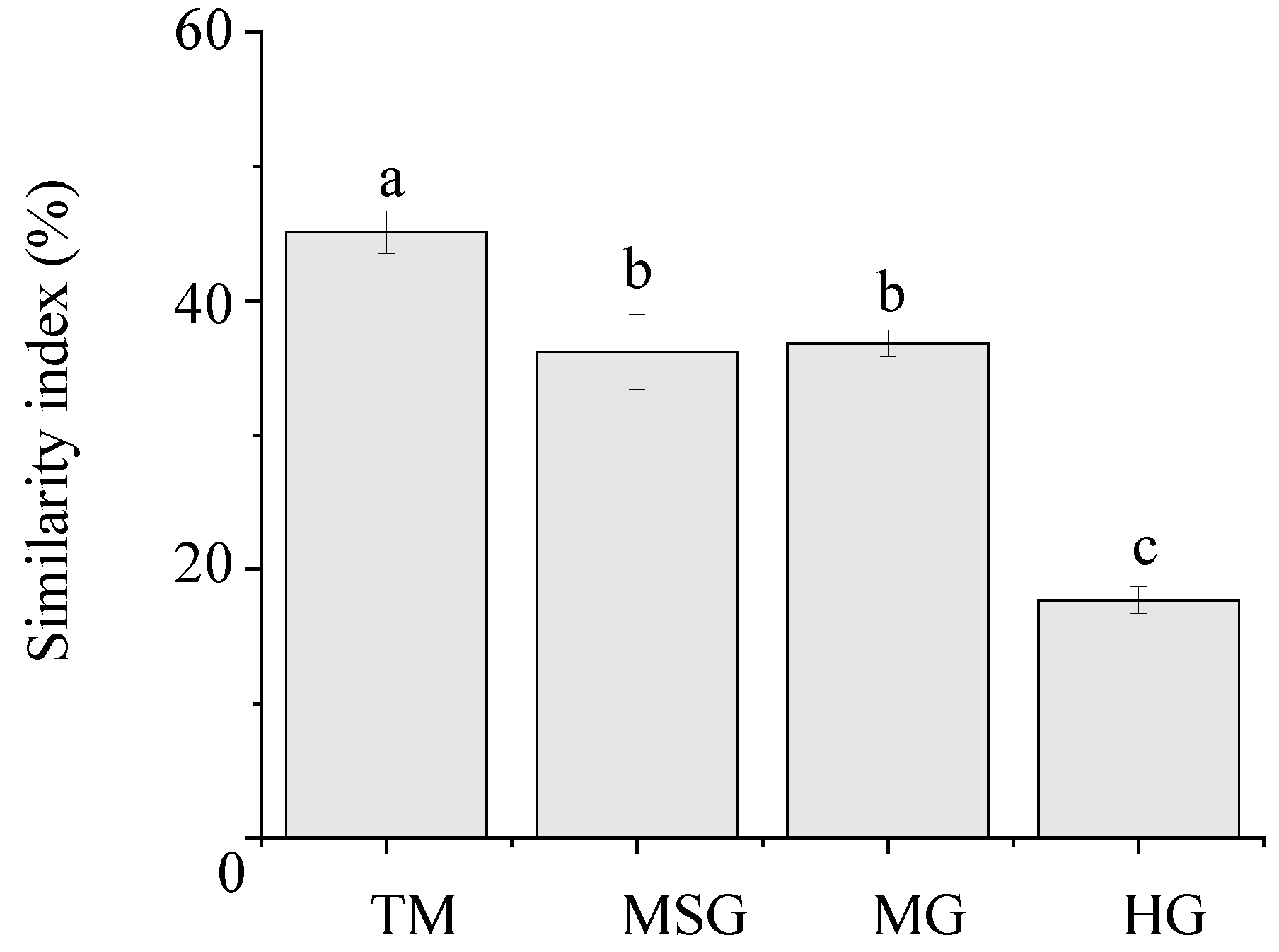
3.4. Similarity of Plant Communities to Reference System
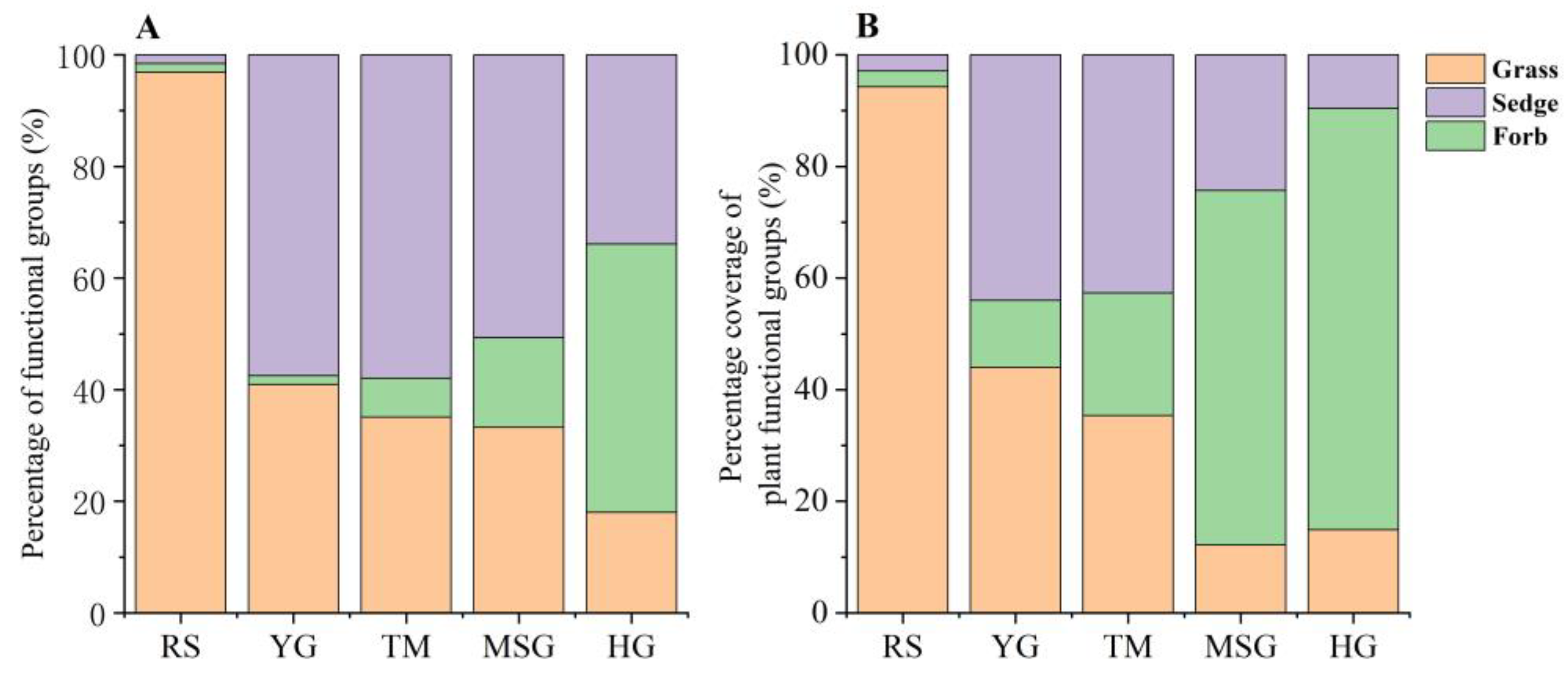
3.5. Composition Characteristics of Three Plant Functional Groups
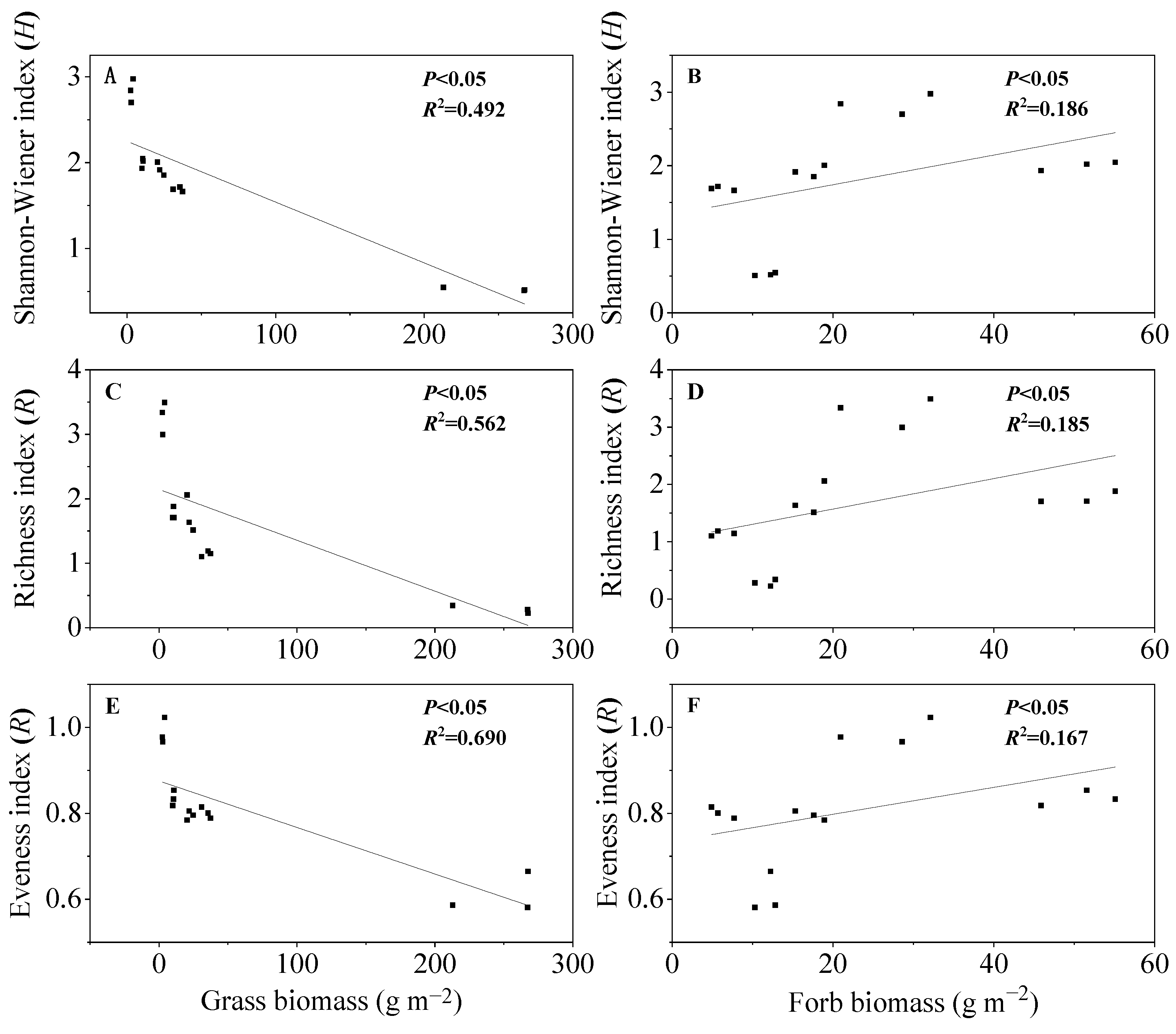
3.6. Relationships between Plant Functional Group Biomass and Species Diversity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Adams, C.R.; Hovick, S.M.; Anderson, N.O.; Kettenring, K.M. We can better manage ecosystems by connecting solutions to constraints: Learning from wetland plant invasions. Front. Environ. Sci. 2021, 9, 715350. [Google Scholar] [CrossRef]
- Åhlén, I.; Thorslund, J.; Hambäck, P.; Destouni, G.; Jarsjö, J. Wetland position in the landscape: Impact on water storage and flood buffering. Ecohydrology 2022, 15, 2458. [Google Scholar] [CrossRef]
- Villa, J.A.; Mitsch, W.J. Carbon sequestration in different wetland plant communities in the Big Cypress Swamp region of southwest Florida. Int. J. Biodivers. Sci. Ecosyst. Serv. Manag. 2014, 11, 17–28. [Google Scholar] [CrossRef]
- Bannister, J.W.; Clairmont, L.K.; Stevens, K.J.; Slawson, R.M. Exposure to elevated nutrient load results in structural and functional changes to microbial communities associated with riparian wetland plants Phalaris arundinaceae and Veronica anagallis-aquatica. Rhizosphere 2021, 18, 100350. [Google Scholar] [CrossRef]
- Brunhoferova, H.; Venditti, S.; Schlienz, M.; Hansen, J. Removal of 27 micropollutants by selected wetland macrophytes in hydroponic conditions. Chemosphere 2021, 281, 130980. [Google Scholar] [CrossRef]
- Colmer, T.D.; Pedersen, O. Underwater photosynthesis and respiration in leaves of submerged wetland plants: Gas films improve CO2 and O2 exchange. New Phytol. 2007, 177, 918–926. [Google Scholar] [CrossRef]
- Yao, S.Y.; Chen, C.; Chen, Q.W.; Cui, Z.; Zhang, J.Y.; Zeng, Y.H. Impact of short-term hydrological components on landscape pattern of waterbird habitat in floodplain wetlands. Water Resour. Res. 2022, 58, 031822. [Google Scholar] [CrossRef]
- Carney, K.M.; Sydeman, W.J. A review of human disturbance effects on nesting colonial waterbirds. Waterbirds 1999, 2222, 68–79. [Google Scholar] [CrossRef]
- Goodwillie, C.; McCoy, M.W.; Peralta, A.L. Long-term nutrient enrichment, mowing, and ditch drainage interact in the dynamics of a wetland plant community. Ecosphere 2020, 11, e03252. [Google Scholar] [CrossRef]
- Nagaraja, B.C.; Somashekar, R.K.; Bunty, R.M. Tree species diversity and composition in logged and unlogged rainforest of Kudremukh National Park, South India. J. Environ. Biol. 2005, 26, 627–634. [Google Scholar]
- He, Y.Z.; Huang, W.D.; Zhao, X.; Lv, P.; Wang, H.H. Review on the impact of climate changes on plant diversity. J. Desert Res. 2021, 41, 59–66. [Google Scholar]
- Möls, T.; Vellak, K.; Vellak, A.; Ingerpuu, N. Global gradients in moss and vascular plant diversity. Biodivers. Conserv. 2013, 22, 1537–1551. [Google Scholar] [CrossRef]
- Gu, C.; Chen, W.J.; Du, Y.F.; Wang, Y.T.; Zhao, T.Q.; Zhao, M.L. Stocking rates affect the resource allocation pattern of Artemisia frigidain the inner mongolia desert steppe. Acta Ecol. Sin. 2017, 37, 2237–2243. (In Chinese) [Google Scholar]
- Tahmasebi, P.; Khedri, H.A.; Ebrahimi, A.A. Effect of livestock grazing on reproductive characteristics of plant communities in steppe rangelands. Iran. J. Range Desert Res. 2013, 20, 345–357. [Google Scholar]
- Diaz, S.; Cabido, M. Plant functional types and ecosystem function in relation to global change. J. Veg. Sci. 1997, 8, 463–474. [Google Scholar] [CrossRef]
- Davies, R.J.P.; Mackay, D.A.; Whalen, M.A.; Smyth, A.K. Vegetation and seed banks of arid ephemeral gilgai wetlands subject to contrasting grazing regimes. J. Arid. Environ. 2018, 154, 51–60. [Google Scholar] [CrossRef]
- Zhao, C.Z.; Li, G.D.; Li, Q.; Zhou, D.W. Effects of mowing frequency on biomass allocation and yield of Leymus chinensis. Rangel. Ecol. Manag. 2022, 83, 102–111. [Google Scholar] [CrossRef]
- Zheng, Y.C.; Wang, X.C.; Ge, Y.; Mawuli, D.; Zhao, Y.Q.; Xiong, J.Q. Effects of annual harvesting on plants growth and nutrients removal in surface-flow constructed wetlands in northwestern China. Ecol. Eng. 2015, 83, 268–275. [Google Scholar] [CrossRef]
- Ford, H.; Garbutt, A.; Ladd, C.; Malarkey, J.; Skov, M.W. Soil stabilization linked to plant diversity and environmental context in coastal wetlands. J. Veg. Sci. Off. Organ Int. Assoc. Veg. Sci. 2016, 27, 259–268. [Google Scholar] [CrossRef]
- Fu, H.; Yuan, G.; Ge, D.; Li, W.; Jeppesen, E. Cascading effects of elevation, soil moisture and soil nutrients on plant traits and ecosystem multi-functioning in Poyang Lake wetland, China. Aquat. Sci. 2020, 82, 34. [Google Scholar] [CrossRef]
- Grime, J.P. Competitive exclusion in herbaceous vegetation. Nature 1973, 242, 344–347. [Google Scholar] [CrossRef]
- Wu, G.L.; Du, G.Z.; Liu, Z.H.; Simon, T. Effect of fencing and grazing on a Kobresia-dominated meadow in the Qinghai-Tibetan Plateau. Plant Soil 2009, 319, 115–126. [Google Scholar] [CrossRef]
- Masasi, A.; Gobolo, A.; Rwiza, I. Impacts of human activities on biodiversity of the Simiyu wetland, Tanzania. Afr. J. Trop. Hydrobiol. Fish. 2018, 16, 108–114. [Google Scholar]
- Niu, K.C.; Philippe, C.; Francesco, D.B.; Nicholas, M.; Du, G.Z.; Sun, S.C. Fertilization decreases species diversity but increases functional diversity: A three-year experiment in a Tibetan alpine meadow. Agric. Ecosyst. Environ. 2014, 182, 106–112. [Google Scholar] [CrossRef]
- Zelnik, I.; Čarni, A. Plant species diversity and composition of wet grasslands in relation to environmental factors. Biodivers. Conserv. 2013, 22, 2179–2192. [Google Scholar] [CrossRef]
- Gaberščik, A.; Krek, J.L.; Zelnik, I. Habitat diversity along a hydrological gradient in a complex wetland results in high plant species diversity. Ecol. Eng. 2018, 118, 84–92. [Google Scholar] [CrossRef]
- Smith, M.D.; Knapp, A.K. Dominant species maintain ecosystem function with non-random species loss. Ecol. Lett. 2003, 6, 509–517. [Google Scholar] [CrossRef]
- Wei, J.; Gao, J.; Wang, N.; Liu, Y.; Wang, Y.W.; Bai, Z.H.; Zhuang, X.L.; Zhuang, G.Q. Differences in soil microbial response to anthropogenic disturbances in Sanjiang and Momoge Wetlands, China. FEMS Microbiol. Ecol. 2019, 95, fiz110. [Google Scholar] [CrossRef]
- Liu, Q.; Ma, H.W.; Tian, H.; An, Z.X. Analysis on the distribution change and influencing factors of the wetlands in Songnen Plain. Geol. Resour. 2010, 19, 76–80. (In Chinese) [Google Scholar]
- An, Y.; Song, T.J.; Zhang, Y.; Tong, S.Z.; Liu, B. Optimum water depth for restoration of Bolboschoenus planiculmis in wetlands in semi-arid regions. Hydrobiologia 2021, 849, 13–28. [Google Scholar] [CrossRef]
- Wassens, S.; Ning, N.; Hardwick, L.; Bino, G.; Maguire, J. Long-term changes in freshwater aquatic plant communities following extreme drought. Hydrobiologia 2017, 799, 233–247. [Google Scholar] [CrossRef]
- Editorial Committee of Flora of China. Flora of China; Science Press: Beijing, China, 2004. [Google Scholar]
- An, Y.; Gao, Y.; Tong, S.Z.; Lu, X.G.; Wang, X.; Wang, G.; Liu, X.H.; Zhang, D.J. Variations in vegetative characteristics of Deyeuxia angustifolia wetlands following natural restoration in the Sanjiang Plain, China. Ecol. Eng. 2018, 112, 34–40. [Google Scholar] [CrossRef]
- Hillhouse, H.L.; Tunnell, S.J.; Stubbendieck, J. Spring grazing impacts on the vegetation of reed canarygrass-invaded wetlands. Rangel. Ecol. Manag. 2010, 63, 581–587. [Google Scholar] [CrossRef]
- BiróM, M.Z.; Öllerer, K.; Lengyel, A.; Ulicsni, V.; Szabados, K.; Kiš, A.; Perić, R.; Demeter, L.; Babai, D. Conservation and herding co-benefit from traditional extensive wetland grazing. Agric. Ecosyst. Environ. 2020, 300, 106983. [Google Scholar] [CrossRef]
- Ren, G.H.; Deng, B.; Shang, Z.H.; Hou, Y.; Long, R.J. Plant communities and soil variations along a successional gradient in an alpine wetland on the Qinghai-Tibetan Plateau. Ecol. Eng. 2013, 61, 110–116. [Google Scholar] [CrossRef]
- Seimandi, G.; Mesa, L.; María, L.S.Z.; Saigo, M.; Gutiérrez, H. Effect of rotational grazing management on vegetation of floodplain wetlands. Wetl. Ecol. Manag. 2021, 29, 565–580. [Google Scholar]
- Poorter, H.; Niklas, K.J.; Reich, P.B.; Oleksyn, J.; Poot, P.; Mommer, L. Biomass allocation to leaves, stems and roots: Meta-analyses of interspecific variation and environmental control. New Phytol. 2012, 193, 30–50. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Gong, J.R.; Wang, B.; Li, X.B.; Ding, Y.; Yang, B.; Zhu, C.C.; Liu, M.; Zhang, W. Regrowth strategies of Leymus chinensis in response to different grazing intensities. Ecol. Appl. 2020, 30, e02113. [Google Scholar] [CrossRef] [PubMed]
- Loreau, M.; Naeem, S.; Inchausti, P.; Bengtsson, J.; Grime, J.P.; Hector, A.; Hooper, D.U.; Huston, M.A.; Raffaelli, D.; Schmid, B.; et al. Biodiversity and ecosystem functioning: Current knowledge and future challenges. Science 2001, 294, 804–808. [Google Scholar] [CrossRef]
- Hautier, Y.; Tilman, D.; Isbell, F.; Seabloom, E.W.; Borer, E.T.; Reich, P.B. Anthropogenic environmental changes affect ecosystem stability via biodiversity. Science 2015, 348, 336–340. [Google Scholar] [CrossRef]
- Zotos, A.; Kosma, C.; Triantafyllidis, V.; Kakabouki, I.; Kehayias, G.; Roussis, I.; Mavroeidis, A.; Tataridas, A.; Bilalis, D. Plant species diversity of the wet meadows under natural and anthropogenic interventions: The case of the Lakes Amvrakia and Ozeros (W. Greece). Not. Bot. Horti Agrobot. Cluj-Napoca 2021, 49, 1–21. [Google Scholar] [CrossRef]
- May, R.M. Will a large complex system be stable? Nature 1972, 238, 413–414. [Google Scholar] [CrossRef] [PubMed]
- Tousignant, M.; Pellerin, S.; Brisson, J. The relative impact of human disturbances on the vegetation of a large wetland complex. Wetlands 2010, 30, 333–344. [Google Scholar] [CrossRef]
- Liu, T.T.; Wang, X.F.; Yuan, X.Z.; Gong, X.J.; Hou, C.L. Species composition and diversity of riparian vegetation in the river network of Xinjin county, Chengdu. Chin. J. Hydroecol. 2021, 42, 24–33. (In Chinese) [Google Scholar]
- Steinaker, D.F.; Jobbágy, E.G.; Martini, J.P.; Arroyo, D.N.; Pacheco, J.L.; Marchesini, V.A. Vegetation composition and structure changes following roller-chopping deforestation in central Argentina woodlands. J. Arid. Environ. 2016, 133, 19–24. [Google Scholar] [CrossRef]
- Zhang, Y.; Su, J.H.; Jing, G.H.; Cheng, J.M. Forbs dominate plant nutrient resorption of plant community along a 34-year grazing exclusion gradient in a semiarid grassland. Ecol. Eng. 2022, 175, 106497. [Google Scholar] [CrossRef]
- Díaz, S.; Lavorel, S.; Mcintyre, S.; Falczuk, V.; Casanoves, F.; Milchunas, D.G.; Skarpe, C.; Rusch, G.; Sternberg, M.; Noy-Meir, I. Plant trait responses to grazing-a global synthesis. Glob. Chang. Biol. 2007, 13, 313–341. [Google Scholar] [CrossRef]
- Moinardeau, C.; Mesléard, F.; Ramone, H.; Dutoit, T. Short-term effects on diversity and biomass on grasslands from artificial dykes under grazing and mowing treatments. Environ. Conserv. 2018, 46, 132–139. [Google Scholar] [CrossRef]
- Golluscio, R.A.; Martínez, G.G.; Cavagnaro, F.P. How does grazing affect soil water availability in the Patagonian steppe? J. Arid. Environ. 2022, 205, 104800. [Google Scholar] [CrossRef]
- Quezada, I.M.; Gianoli, E. Counteractive biomass allocation responses to drought and damage in the perennial herb Convolvulus demissus. Austral Ecol. 2010, 35, 544–548. [Google Scholar] [CrossRef]
- Ye, R.H.; Liu, G.F.; Chang, H.; Shan, Y.M.; Mu, L.; Wen, C.; Te, R.; Wu, N.T.; Shi, L.; Liu, Y.H.; et al. Response of plant traits of Stipa breviflora to grazing intensity and fluctuation in annual precipitation in a desert steppe, northern China. Glob. Ecol. Conserv. 2020, 24, e01237. [Google Scholar] [CrossRef]
- Hooper, D.U.; Adair, E.C.; Cardinale, B.J.; Byrnes, J.E.K.; Hungate, B.A.; Matulich, K.L.; Gonzalez, A.; Duffy, J.E.; Gamfeldt, L.; O’Connor, M. A global synthesis reveals biodiversity loss as a major driver of ecosystem change. Nature 2012, 486, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt-Römermann, M.; Römermann, C.; Sperlich, S.; Schmidt, W. Explaining grassland biomass—The contribution of climate, species and functional diversity depends on fertilization and mowing frequency. J. Appl. Ecol. 2011, 48, 1088–1097. [Google Scholar] [CrossRef]
- Busch, V.; Klaus, V.H.; Penone, C.; Schäfer, D.; Boch, S.; Prati, D.; Müller, J.; Socher, S.A.; Niinemets, Ü.; Peñuelas, J. Nutrient stoichiometry and land use rather than species richness determine plant functional diversity. Ecol. Evol. 2018, 8, 601–616. [Google Scholar] [CrossRef]
- Fidalski, J.; Bordin, I.; Alves, S.J.; de Cesare Barbosa, G.M. Grazing heights, stocking rate, soil structure, and water infiltration in a crop-livestock integration. Semin. Cienc. Agrar. 2021, 42, 123–135. [Google Scholar] [CrossRef]
- Ungar, I. Are biotic factors significant in influencing the distribution of halophytes in saline habitats? Bot. Rev. 1998, 64, 176–199. [Google Scholar] [CrossRef]
- Yan, R.R.; Zhang, Y.; Xin, X.P.; Wei, Z.J.; Wu, R.Q.; Guo, M.L. Effects of mowing disturbance on plant functional groups and diversity of leymus chinensis steppe. Sci. Agric. Sin. 2020, 53, 2573–2583. (In Chinese) [Google Scholar]
- Helm, N.; Essl, F.; Mirtl, M.; Dirnböck, T. Multiple environmental changes drive forest floor vegetation in a temperate mountain forest. Ecol. Evol. 2017, 7, 2155–2168. [Google Scholar] [CrossRef]
- Poggio, S.L.; Ghersa, C.M. Species richness and evenness as a function of biomass in arable plant communities. Weed Res. 2011, 51, 241–249. [Google Scholar] [CrossRef]
- Zhao, L.P.; Cui, Z.; Yu, L.; Chang, X.F.; Wu, G.L. Grazing exclusion erodes the forbs functional group without altering offspring recruitment composition in a typical steppe. Land Degrad. Dev. 2020, 31, 710–720. [Google Scholar] [CrossRef]
| Wetlands | Species | Important Value (IV) |
|---|---|---|
| RS | C. angustifolia | 0.80 ± 0.05 a |
| P. persicaria | 0.15 ± 0.02 b | |
| C. appendiculata | 0.08 ± 0.01 b | |
| TM | C. appendiculata | 0.45 ± 0.01 a |
| C. angustifolia | 0.31 ± 0.01 b | |
| A. selengensis | 0.07 ± 0.01 c | |
| MSG | C. humida | 0.45 ± 0.03 a |
| C. angustifolia | 0.20 ± 0.01 b | |
| P. persicaria | 0.06 ± 0.01 c | |
| MG | A.selengensis | 0.30 ± 0.02 a |
| C. appendiculata | 0.25 ± 0.09 a | |
| C. angustifolia | 0.20 ± 0.05 a | |
| HG | A. selengensis | 0.38 ± 0.02 a |
| C. humida | 0.20 ± 0.01 b | |
| C. angustifolia | 0.16 ± 0.04 b |
| Wetlands | Diversity Index (H) | Richness Index (R) | Evenness Index (E) |
|---|---|---|---|
| RS | 0.52 ± 0.02 d | 0.28 ± 0.05 d | 0.61 ± 0.01 d |
| TM | 1.69 ± 0.02 c | 1.15 ± 0.04 c | 0.80 ± 0.01 c |
| MSG | 1.92 ± 0.06 b | 1.74 ± 0.23 b | 0.79 ± 0.23 c |
| MG | 2.00 ± 0.05 b | 1.77 ± 0.08 b | 0.83 ± 0.01 b |
| HG | 2.84 ± 0.11 a | 3.27 ± 0.21 a | 0.99 ± 0.02 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Wang, X.; An, Y.; Song, T.; Tong, S.; Wang, X. Effects of Land Use Changes on the Plant Community Characteristics in the Wetlands of the Semi-Arid Regions. Diversity 2022, 14, 1049. https://doi.org/10.3390/d14121049
Wang L, Wang X, An Y, Song T, Tong S, Wang X. Effects of Land Use Changes on the Plant Community Characteristics in the Wetlands of the Semi-Arid Regions. Diversity. 2022; 14(12):1049. https://doi.org/10.3390/d14121049
Chicago/Turabian StyleWang, Le, Xiaodong Wang, Yu An, Tiejun Song, Shouzheng Tong, and Xuan Wang. 2022. "Effects of Land Use Changes on the Plant Community Characteristics in the Wetlands of the Semi-Arid Regions" Diversity 14, no. 12: 1049. https://doi.org/10.3390/d14121049
APA StyleWang, L., Wang, X., An, Y., Song, T., Tong, S., & Wang, X. (2022). Effects of Land Use Changes on the Plant Community Characteristics in the Wetlands of the Semi-Arid Regions. Diversity, 14(12), 1049. https://doi.org/10.3390/d14121049





