Abstract
The title compound was prepared by an aza-Michael addition reaction between 1-[1-(4-chlorobenzenesulfonyl)-1H-indole-3-yl]prop-2-en-1-one and 2-piridylpiperazine catalyzed by SiO2. The structural identity of the title compound was proven by elemental analysis and spectroscopic methods (IR, NMR). The compound was assayed in a binding assay at the 5-HT6 receptor, showing poor affinity.
1. Introduction
The serotonin receptor subtype 6 (5-HT6), a metabotropic receptor located exclusively within the central nervous system [1,2,3], is considered a promising target for the treatment of several illnesses such as Alzheimer’s disease, obesity, and major depressive disorder [4,5,6]. A 5-HT6 antagonist pharmacophore has been previously described in the literature [7]. In our efforts to prepare potent and highly selective antagonists targeting 5-HT6 based on this reported pharmacophore [8,9], we performed the synthesis of the title compound 1-[1-(4-chlorobenzenesulfonyl)-1H-indole-3-yl]-3-[4-(pyridin-2-yl)piperazin-1-yl]propan-1-one and evaluated its affinity for the 5-HT6 receptor in a standard binding assay.
2. Results and Discussion
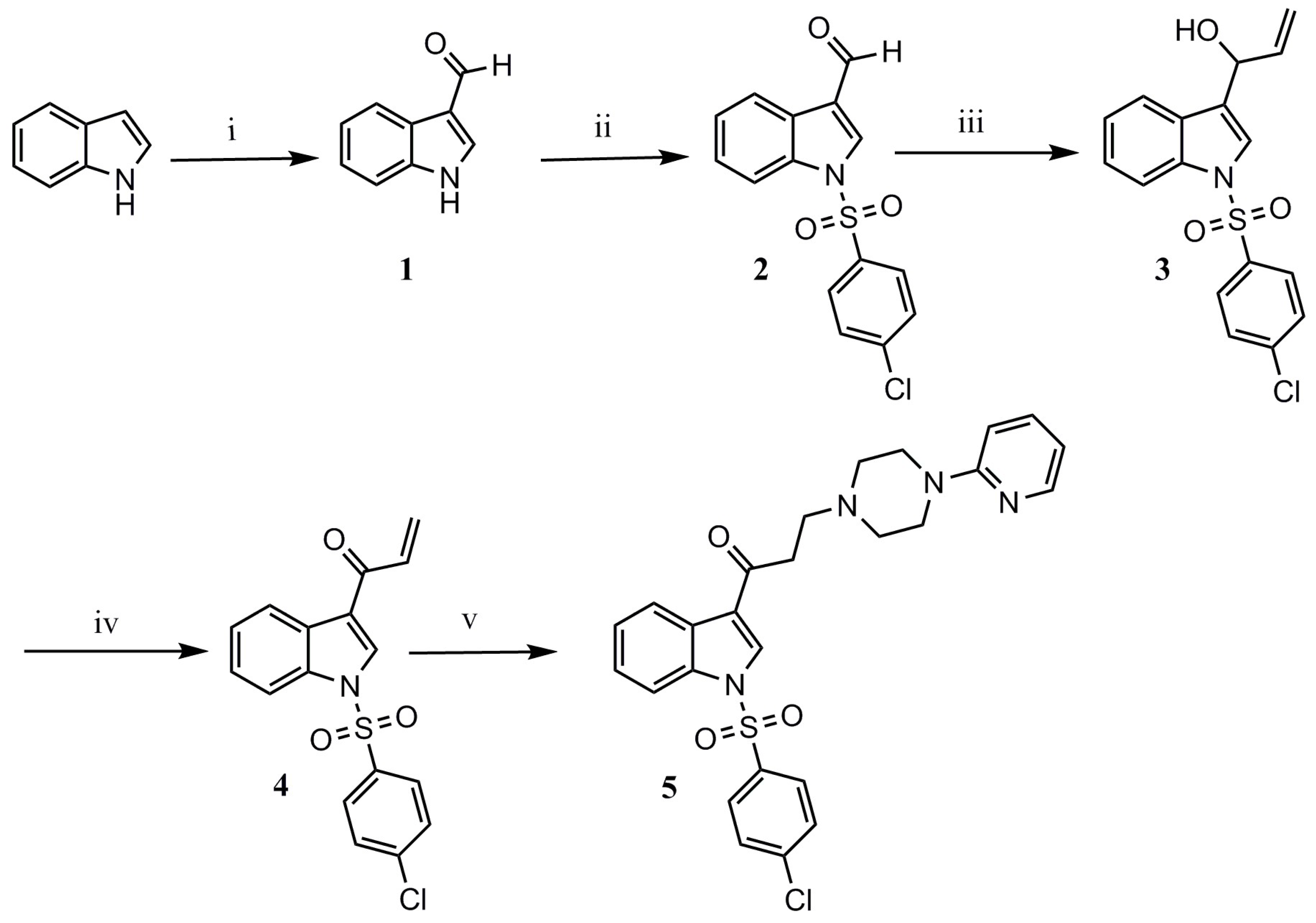
Title compound 5 was synthesized through a series of reactions beginning from commercial indole, and using methods reported in the literature (Scheme 1). First, indole was formylated in C-3 employing the Vilsmeier–Haack synthesis, affording formylindole 1 in excellent yield [10]. Afterwards, N-sulfonylation in basic media gave N-(4-chlorobenzenesulfonyl)-3-formylindole 2 in good yield [8]. A nucleophilic attack on the formyl group with the commercially available Grignard reagent vinylmagnesium bromide led to secondary allylic alcohol 3, which was oxidized to the corresponding α,β-unsaturated ketone 4 employing MnO2/MgSO4 [10]. Synthesis of the title compound involved an aza-Michael addition reaction, with 2-pyridylpiperazine. This reaction was initially attempted using microwave irradiation [10]; however, yields were poor, and degradation products were observed. We therefore attempted a reflux reaction in acetonitrile, employing SiO2 as a catalyst [11]. These conditions afforded the desired product 5, which was obtained as a crude product and then purified by gravity column chromatography in 89% yield. Spectroscopic data confirmed the structure of the product.
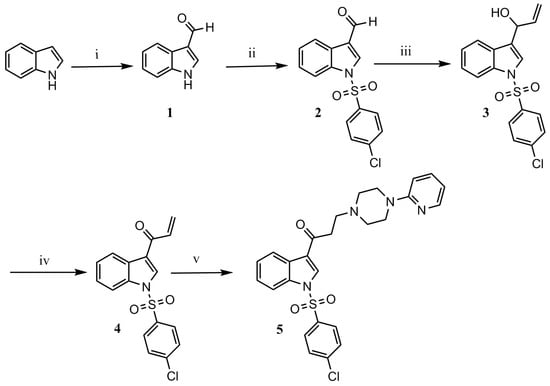
Scheme 1.
Synthesis of 1-[1-(4-chlorobenzenesulfonyl)-1H-indol-3-yl]-3-[4-(pyridin-2-yl)piperazin-1-yl]propan-1-one. Reagents and conditions: (i) POCl3, DMF, 0 °C, 30 min; (ii) CH2Cl2, Et3N, DMAP, 4-chlorobenzenesulfonyl chloride; (iii) vinylmagnesium bromide, anhydrous THF, r.t., 8–10 h; (iv) anhydrous CH2Cl2, MnO2, anhydrous MgSO4; (v) SiO2, CH3CN, 2-pyridylepiperazine, reflux, 12–20 h.
In the context of our interest to produce highly active antagonists towards the 5-HT6 receptor, 5 was tested in a standard radioligand competition binding assay, using membranes of HEK-293 cells expressing a recombinant human 5-HT6 receptor, as previously described [8,12]. The product was assayed as a free base at eight concentrations, in triplicate, to obtain the dose–response curve, determine the IC50 value and calculate the Ki through the Cheng-Prusoff equation [13]. Using this approach, it was determined that the Ki was 1.33 μM, and that the IC50 had a magnitude of 1.38 μM, which is regarded as a poor affinity for the receptor (for the dose–response curve, see Figure S16).
3. Materials and Methods
3.1. Materials
Reagents were purchased from commercial suppliers, specifically Merck (Darmstadt, Germany) and Sigma-Aldrich (St. Louis, MO, USA), and used without further purification. Solvents were purchased from commercial suppliers and were purified by distillation prior to their use.
3.2. Instrumentation
Melting points were determined on a Stuart Scientific SMP30 apparatus (Bibby Scientific Limited, Stone, UK), employing open-glass capillaries. Infrared spectra were recorded on a BRUKER Vector 22 spectrometer using KBr pellets. NMR spectra were recorded on a Bruker Avance III HD 400 (Billerica, MA, USA) at 400 MHz for 1H and 100 MHz for 13C-NMR spectra were recorded in CDCl3, using the solvent signal as a reference. The chemical shifts are expressed in ppm (δ scale) downfield from tetramethylsilane (TMS) and coupling constants values (J) are given in Hertz. The following multiplicity abbreviations were utilized: singlet (s), broad signal (bs); doublet (d); doublet of doublets (dd); multiplet (m). Elemental analyses were performed on a FISONS EA 1108 CHNS-O elemental analyzer (Thermo Scientific, Waltham, MA, USA). Radioligand binding studies were performed by Scottish Biomedical Drug Discovery.
3.3. Synthesis
3.3.1. 3-Formylindole (1)
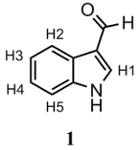
POCl3 (0.8 mL, 1.34 g, 8.74 mmol) was added dropwise over DMF (5 mL, 4.72 g, 64.58 mmol) in a round-bottomed flask at 0 °C. Afterwards, indole (1 g, 8.54 mmol) was dissolved in another round-bottomed flask containing DMF (5 mL). The POCl3 solution was slowly added to the indole solution. The mixture was stirred at 0 °C for 30 min, after which the contents were poured over a water–ice mixture and the pH was regulated to 12. A pale yellow precipitate was formed, which was filtered and dried in a stove. This product was used in subsequent syntheses without further purification. Yield: 95%. m.p.: 189.1–192.5 °C (lit.: 193–195 °C [14]). 1H-NMR (400 MHz, DMSO-d6): δ (ppm) 12.18 (s, 1H; NH); 9.99 (s, 1H; CHO); 8.31 (s, 1H; H1); 8.17 (d, J = 7.2 Hz; 1H; H2); 7.56 (d, J = 7.6 Hz; 1H; H5); 7.23–7.31 (m, 2H; H3, H4). 13C-NMR (101 MHz, DMSO-d6): δ 185.43; 138.86; 137.55; 124.63; 123.93; 122.60; 121.32; 118.68; 112.90. IR (cm−1): 3169 (NH); 2932 (CHAr); 1634 (CHO). Anal. calculated for C9H7NO (%): C: 74.47; H: 4.86; N: 9.65. Anal. found for C9H7NO (%): C: 74.02; H: 4.79; N: 9.92.
3.3.2. 1-(4-Chlorobenzenesulfonyl)-3-formylindole (2)
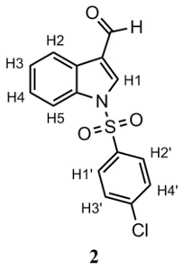
Triethylamine (Et3N; 0.44 mL, 0.318 g, 3.14 mmol) and 4-dimethylaminopyridine (DMAP; 0.048 g, 0.31 mmol) were added over a stirred solution of 1 (0.46 g, 3.14 mmol) in CH2Cl2 (30 mL). After 10 min, p-chlorobenzenesulfonyl chloride (0.796 g, 3.77 mmol) is added. Reaction progress was monitored by TLC until the starting materials had disappeared. At this point, the reaction was quenched by adding a 1 M HCl solution. The organic layer was extracted with EtOAc (3 × 20 mL) and dried over anhydrous Na2SO4. Solvent was removed under a vacuum, affording a crude product that was purified by gravity column chromatography, employing silica gel as adsorbent and CH2Cl2 as eluent. This purification affords a white crystalline solid. Yield: 76%. m.p.: 152.8–155.1 °C (lit.: 152–154 °C [15]). 1H-NMR (400 MHz, CDCl3): δ 10.09 (s, 1H; CHO); 8.26 (d, J = 7.3 Hz; 1H; H2); 8.21 (s, 1H; H1); 7.92 (d, J = 8.2 Hz; 1H; H5); 7.89 (d, J = 8.7 Hz; 2H; H1’, H2’); 7.45 (d, J = 8.7 Hz; 2H; H3’, H4’); 7.35–7.42 (m, 2H; H3, H4). 13C-NMR (101 MHz, CDCl3): δ 185.69; 142.06; 136.34; 136.09; 135.54; 130.50; 128.96; 126.98; 126.76; 125.75; 123.20; 123.18; 113.52. IR (cm−1): 3137 (CHhet); 3086 (CHar); 1664 (C=O); 1380 (SO2); 1182 (SO2). Anal. calculated for C15H10ClNO3S (%): C: 56.34; H: 3.15; N: 4.38; S: 10.03 Anal. found for C15H10ClNO3S (%): C: 56.38; H: 3.20; N: 4.94; S: 10.16.
3.3.3. 1-[1-(4-Chlorobenzenesulfonyl)-1H-indole-3-yl]-prop-2-en-1-ol (3)
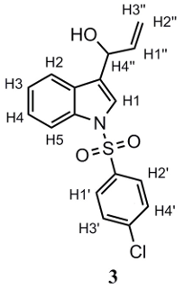
2 was added to a round-bottomed flask, which was purged with N2. Afterwards, anhydrous THF and vinylmagnesium bromide were added. The contents were stirred for 8–10 h, after which the reaction was quenched adding a saturated NH4Cl solution. The product was extracted with CH2Cl2 (3 × 20 mL) and dried over Na2SO4. Solvent was removed under vacuum, affording an orange-colored oil, which was purified by gravity column chromatography, employing silica gel as adsorbent and CH2Cl2 as eluent. The purified product corresponds to a yellow oil. Yield: 83%. 1H-NMR (400 MHz, CDCl3): δ 7.94 (d, J = 8.3 Hz; 1H; H2); 7.79 (d, J = 8,6 Hz; 2H; H1’, H2’); 7.62 (d, J = 7.8 Hz; 1H; H5); 7.50 (s, 1H; H1); 7.30–7.36 (m, 3H; H3, H3’, H4’); 7.23 (t, J = 7.5 Hz; 1H; H4); 6.08–6.12 (m, 1H; H1’’); 5.40–5.44 (m, 2H; H3’’, H4’’); 5.27 (d, Jcis = 10.2 Hz; 1H; H2’’); 2.25 (bs, 1H; OH). 13C-NMR (101 MHz, CDCl3): δ 140.97; 138.89; 136.82; 135.87; 130.04; 129.39; 128.61; 125.58; 125.26; 123.97; 123.45; 121.19; 116.90; 113.99; 69.23. IR (cm−1): 3441 (OH); 3128 (CHAr), 3055 (CH2), 1363 (asymmetric R-SO2-R); 1173 (symmetric R-SO2-R). Anal. calculated for C21H17NO3S (%): C: 69.40; H: 4.71; N: 3.85; S, 8.82. Anal. found for C21H17NO3S (%): C: 69.22; H: 5.07; N: 3.56; S: 8.93.
3.3.4. 1-[1-(4-Chlorobenzenesulfonyl)-1H-indole-3-yl]-prop-2-en-1-one (4)
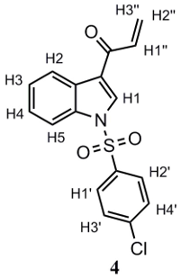
A solution of 3 (0.382 g, 1.10 mmol) in anhydrous CH2Cl2 (30 mL) was prepared. Afterwards, γ-MnO2 (1.91 g) and anhydrous MgSO4 (0.127 g, 1.10 mmol) were added. The mixture was stirred vigorously while monitoring the reaction progress by TLC. Once oxidation was complete, MnO2 was removed by filtration, and the solvent was removed under a vacuum. The resulting crude product was purified by gravity column chromatography, employing silica gel as adsorbent and CH2Cl2 as eluent, affording a pale yellow amorphous solid. Yield: 51%. m.p.: 137.7–140.1 °C. 1H-NMR (400 MHz, CDCl3): δ 8.42 (d, J = 6.7 Hz; 1H; H2); 8.26 (s, 1H; H1); 7.94 (d, J = 7.3 Hz, 1H; H5); 7.88 (d, J = 8.4 Hz; 2H; H1’, H2’); 7.45 (d, J = 8.5 Hz; 2H; H3’, H4’), 7.38–7.42 (m, 2H; H3, H4); 7.07 (dd, Jtrans = 17.0 and Jcis = 10,5 Hz; 1H; H1’’); 6.51 (d, Jtrans = 17.0 Hz, 1H; H3’’); 5.90 (d, Jcis = 10.5 Hz; 1H; H2’’). 13C-NMR (101 MHz, CDCl3): δ 185.77; 141.89; 136.25; 135.33; 133.43; 132.20; 130.42; 129.22; 128.88; 128.41; 126.64; 125.57; 123.88; 122.27; 113.37. IR (cm−1): 3122–3122 (CHAr), 3054 (=CH2), 1665 (C=O), 1603 (C=C), 1373 (asymmetric R-SO2-R); 1174 (symmetric R-SO2-R). Anal. calculated for C17H12ClNO3S (%): C: 59.05; H: 3.50; N: 4.05; S: 9.27. Anal. found for C17H12ClNO3S (%): C: 58.89; H: 3.67; N: 4.59; S: 9.60.
3.3.5. 1-[1-(4-Chlorobenzenesulfonyl)-1H-indole-3-yl]-3-[4-(pyridin-2-yl)piperazin-1-yl]propan-1-one (5)
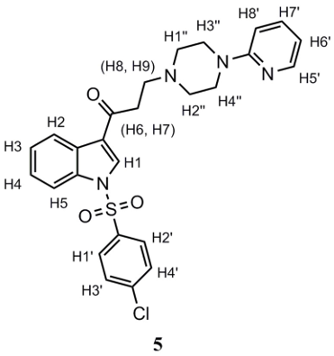
4 (0.185 g, 0.53 mmol), 2-pyridylpiperazine (0.105 g, 0.64 mmol) and a catalytic amount of silica gel (SiO2, 0.01–0.1 g), were dissolved in CH3CN (40 mL). The mixture was refluxed for 12 to 20 h, and the reaction progress was monitored by TLC. The product, once purified by gravity column chromatography employing silica gel as adsorbent and EtOAc as eluent, corresponds to an amorphous white solid. Yield: 89%. m.p.: 154.9–158.9 °C; 1H-NMR (400 MHz, CDCl3): δ 8.34 (d, J = 7.3 Hz; 1H; H2); 8.26 (s, 1H; H1); 8.20 (bs, 1H; H5’); 7.92 (d, J = 7.5 Hz; 1H; H5); 7.87 (d, J = 8.1 Hz; 2H; H1’, H2’); 7.46 (m; 3H; H3’, H4’,H7’); 7.38 (m; 2H; H3, H4); 6.65 (m; 2H; H6’, H8’); 3.56 (bs, 4H; H3”, H4”); 3.15 (t, J = 7.0 Hz, 2H; H6, H7); 2.91 (t, J = 7.0 Hz; 2H; H8, H9); 2.64 (bs, 4H; H1”, H2”). 13C-NMR (101 MHz, CDCl3): δ 195.01; 159.88; 148.38; 141.89; 137.86; 136.29; 135.27; 131.99; 130.43; 128.85; 128.05; 126.46; 125.55; 123.71; 122.19; 113.79; 113.36; 107.49; 53.59; 53.50; 45.61; 38.31. IR (cm−1): 3141–3065 (CHAr), 1664 (C=O), 1382 (R-SO2-R), 1174 (R-SO2-R). Anal. calculated for C26H25ClN4O3S (%): C: 61.35; H: 4.95; N: 11.01; S: 6.30. Anal. found for C26H25ClN4O3S (%): C: 61.83; H: 5.04; N: 10.56; S: 6.05.
3.4. Radioligand Binding Studies
Affinity of the title compound for 5-HT6 receptors was evaluated using HEK-293 cells expressing human 5-HT6R, employing the iodinated specific radioligand [125I]-SB-258585 (4-iodo-N-[4-methoxy-3-(4-methyl-piperazin-1-yl)-phenyl]-benzenesulfonamide); Kd = 1.3 nM; 2200 Ci/mmol). Competitive inhibition assays were performed according to standard procedures, briefly detailed below.
Fractions of 45 μL of diluted 5-HT6 membrane preparation were incubated at 27 °C for 180 min with 25 5-HT6R L of [125I]-SB-258585 (0.2 nM) and 25 5-HT6R L of WGA PVT SPA beads (4 mg/mL), in the presence of increasing concentrations (10−11 to 10−4 M) of the competing drug (5 μL) or DMSO, in a final volume of 100 μL of assay buffer (50 mM Tris, 120 mM NaCl, pH 7.4). Non-specific binding was determined by radioligand binding in the presence of a saturating concentration of 100 μM of clozapine. Binding of [125I]-SB-258585 to 5-HT6 receptors directly correlates to an increase in signal that was read on a Perkin Elmer Topcount NXT HTS (PerkinElmer, Waltham, MA, USA). Compounds were tested at eight concentrations, in triplicate. Clozapine was used as an internal standard for comparison. Data generated were analyzed using GraphPad Prism version 7.0 (GraphPad Software Inc., La Jolla, CA, USA). A linear regression line of data points was plotted, from which the concentration of the competing ligand that displaces 50% of the specific binding of the radioligand (IC50 value) was determined and the Ki value was calculated based upon the Cheng–Prusoff equation, Ki = IC50/(1 + L/Kd), where L is the concentration of free radioligand used in the assay and Kd is the dissociation constant of the radioligand for the receptor.
4. Conclusions
In this work, we report the synthesis of 1-[1-(4-chlorobenzenesulfonyl)-1H-indole-3-yl]-3-[4-(pyridin-2-yl)piperazin-1-yl]propan-1-one by a route starting from commercial indole. The final step of this route, which leads to the title compound, involved an aza-Michael addition reaction between 1-[1-(4-chlorobenzenesulfonyl)-1H-indole-3-yl]prop-2-en-1-one and 2-piridylpiperazine. Use of SiO2 as a catalyst afforded the desired final product in excellent yield. Both the title compound and its precursors had their structural identity proven employing spectroscopic methods, and a complete physical characterization was provided for all of them. In the context of our efforts to produce potent and selective novel 5-HT6 receptor antagonists, we measured the title compound’s affinity for this receptor employing a standard radioligand binding assay. Unfortunately, the results of said assay revealed that the title compound displays a poor affinity for the receptor. Nevertheless, this study highlights an interesting synthetic route to the preparation of arylsulfonylindolepropanones, thus providing orientation for the design of future ligands acting as 5-HT6 receptor antagonists.
Supplementary Materials
The following are available online, Figure S1: 1H-NMR spectrum of 1; Figure S2: 13C-NMR spectrum of 1; Figure S3: 1H-NMR spectrum of 2; Figure S4: 13C-NMR spectrum of 2; Figure S5: 1H-NMR spectrum of 3; Figure S6: 13C-NMR spectrum of 3; Figure S7: 1H-NMR spectrum of 4; Figure S8: 13C-NMR spectrum of 4; Figure S9: 1H-NMR spectrum of 5; Figure S10: 13C-NMR spectrum of 5; Figure S11: IR spectrum of 1; Figure S12: IR spectrum of 2; Figure S13: IR spectrum of 3; Figure S14: IR spectrum of 4; Figure S15: IR spectrum of 5; Figure S16: Dose-response curve for the 5-HT6 receptor of 5.
Acknowledgments
This work was supported by FONDECYT No. 11121418 project.
Author Contributions
B.D.: Literature research, writing of manuscript; G.R.-G.: Synthesis planning, IR and NMR interpretation, proofreading of manuscript; S.A.: Experimental synthetic work, literature research, IR and NMR interpretation.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| 5-HT | serotonin |
| 5-HT6R | serotonin receptor subtype 6 |
| Anal. | elemental analysis |
| IC50 | half maximal inhibitory concentration |
| IR | infrared spectroscopy |
| Ki | inhibition constant |
| NMR | nuclear magnetic resonance spectroscopy |
| TLC | thin layer chromatography |
| TMS | tetramethylsilane |
References
- Kohen, R.; Metcalf, M.A.; Khan, N.; Druck, T.; Huebner, K.; Lachowicz, J.E.; Meltzer, H.Y.; Sibley, D.R.; Roth, B.L.; Hamblin, M.W. Cloning, characterization, and chromosomal localization of a human 5-HT6 serotonin receptor. J. Neurochem. 1996, 66, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Woolley, M.L.; Marsden, C.A.; Fone, K.C. 5-HT6 receptors. Curr. Drug Targets CNS Neurol. Disord. 2004, 3, 59–79. [Google Scholar] [CrossRef] [PubMed]
- Monsma, F.J., Jr.; Shen, Y.; Ward, R.P.; Hamblin, M.W.; Sibley, D.R. Cloning and expression of a novel serotonin receptor with high affinity for tricyclic psychotropic drugs. Mol. Pharmacol. 1993, 43, 320–327. [Google Scholar] [PubMed]
- Heal, D.J.; Smith, S.L.; Fisas, A.; Codony, X.; Buschmann, H. Selective 5-HT6 receptor ligands: Progress in the development of a novel pharmacological approach to the treatment of obesity and related metabolic disorders. Pharmacol. Ther. 2008, 117, 207–231. [Google Scholar] [CrossRef] [PubMed]
- Fisas, A.; Codony, X.; Romero, G.; Dordal, A.; Giraldo, J.; Merce, R.; Holenz, J.; Vrang, N.; Sorensen, R.V.; Heal, D.; et al. Chronic 5-HT6 receptor modulation by e-6837 induces hypophagia and sustained weight loss in diet-induced obese rats. Br. J. Pharmacol. 2006, 148, 973–983. [Google Scholar] [CrossRef] [PubMed]
- Upton, N.; Chuang, T.T.; Hunter, A.J.; Virley, D.J. 5-HT6 receptor antagonists as novel cognitive enhancing agents for alzheimer’s disease. Neurotherapeutics 2008, 5, 458–469. [Google Scholar] [CrossRef] [PubMed]
- López-Rodríguez, M.L.; Benhamú, B.; de la Fuente, T.; Sanz, A.; Pardo, L.; Campillo, M. A three-dimensional pharmacophore model for 5-hydroxytryptamine6 (5-HT6) receptor antagonists. J. Med. Chem. 2005, 48, 4216–4219. [Google Scholar] [CrossRef] [PubMed]
- Vera, G.; Lagos, C.F.; Almendras, S.; Hebel, D.; Flores, F.; Valle-Corvalán, G.; Pessoa-Mahana, C.; Mella-Raipán, J.; Montecinos, R.; Recabarren-Gajardo, G. Extended N-arylsulfonylindoles as 5-HT6 receptor antagonists: Design, synthesis & biological evaluation. Molecules 2016, 21, 1070. [Google Scholar]
- Mella, J.; Villegas, F.; Morales-Verdejo, C.; Lagos, C.F.; Recabarren-Gajardo, G. Structure-activity relationships studies on weakly basic n-arylsulfonylindoles with an antagonistic profile in the 5-HT6 receptor. J. Mol. Struct. 2017, 1139, 362–370. [Google Scholar] [CrossRef]
- Pessoa-Mahana, H.; Recabarren-Gajardo, G.; Temer, J.F.; Zapata-Torres, G.; Pessoa-Mahana, C.D.; Barria, C.S.; Araya-Maturana, R. Synthesis, docking studies and biological evaluation of benzo[b]thiophen-2-yl-3-(4-arylpiperazin-1-yl)-propan-1-one derivatives on 5-HT1A serotonin receptors. Molecules 2012, 17, 1388–1407. [Google Scholar] [CrossRef] [PubMed]
- You, L.S.; Feng, S.; An, R.; Wang, X.H.; Bai, D.L. Silica gel accelerated aza-michael addition of amines to α,β-unsaturated amides. Tetrahedron Lett. 2008, 49, 5147–5149. [Google Scholar] [CrossRef]
- Hirst, W.D.; Minton, J.A.; Bromidge, S.M.; Moss, S.F.; Latter, A.J.; Riley, G.; Routledge, C.; Middlemiss, D.N.; Price, G.W. Characterization of [125I]-SB-258585 binding to human recombinant and native 5-HT6 receptors in rat, pig and human brain tissue. Br. J. Pharmacol. 2000, 130, 1597–1605. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Prusoff, W.H. Relationship between the inhibition constant (K1) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem. Pharmacol. 1973, 22, 3099–3108. [Google Scholar] [PubMed]
- Tyson, F.T.; Shaw, J.T. A new approach to 3-indolecarboxaldehyde. J. Am. Chem. Soc. 1952, 74, 2273–2274. [Google Scholar] [CrossRef]
- Aggarwal, R.; Benedetti, F.; Berti, F.; Buchini, S.; Colombatti, A.; Dinon, F.; Galasso, V.; Norbedo, S. A catalytic antibody programmed for torsional activation of amide bond hydrolysis. Chem.-Eur. J. 2003, 9, 3132–3142. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).