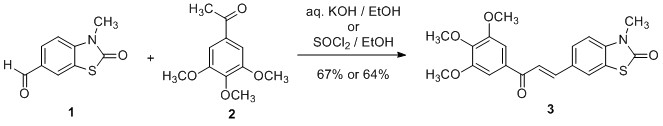
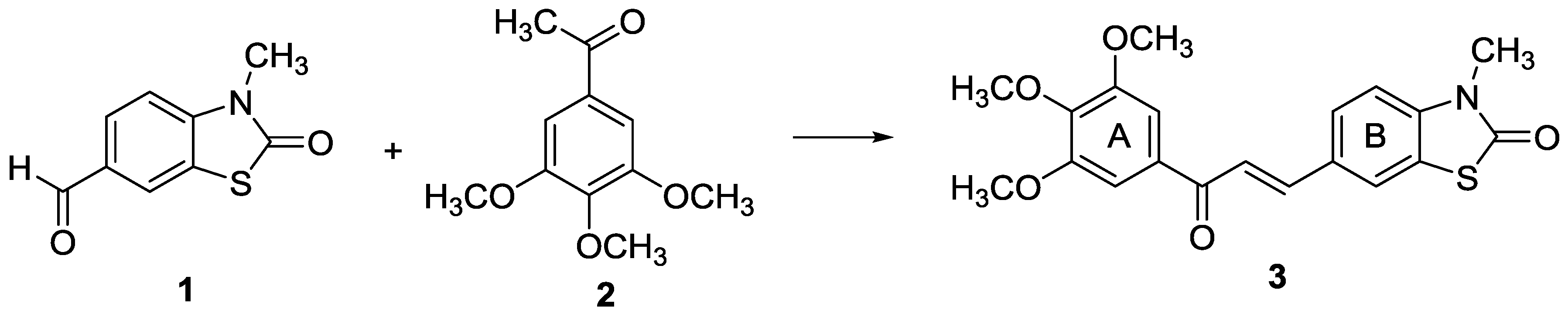
(E)-3-Methyl-6-(3-oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)-2(3H)-benzothiazolone
Abstract
:1. Introduction
2. Results
3. Experimental Section
3.1. General Information
3.2. Synthesis of (E)-3-Methyl-6-(3-oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)-2(3H)-benzothiazolone (3)
3.2.1. Base-catalyzed Aldol Condensation:
3.2.2. Acid-catalyzed Aldol Condensation:
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Karthikeyan, C.; Moorthy, N.; Ramasamy, S.; Vanamc, U.; Manivannan, E.; Karunagaran, D.; Trivedi, P. Advances in Chalcones with Anticancer Activities. Recent Pat. Anti-Cancer Drug Discov. 2015, 10, 97–115. [Google Scholar] [CrossRef]
- Sinha, S.; Medhi, B.; Sehgal, R. Chalcones as an Emerging Lead Molecule for Antimalarial Therapy: A Review. J. Modern Med. Chem. 2013, 1, 64–77. [Google Scholar]
- Dimmock, J.; Elias, D.; Beazely, M.; Kandepu, N. Bioactivities of chalcones. Curr. Med. Chem. 1999, 6, 1125–1149. [Google Scholar] [PubMed]
- Dhar, D.N. The Chemistry of Chalcones and related Compounds; John Wiley & Sons: New York, NY, USA, 1981. [Google Scholar]
- Miquel, J.F. Isomere cis-trans des styryl-cetones – para et meta-hydroxy-chalcones. Bull. Soc. Chim. Fr. 1961, 1369–1376. [Google Scholar]
- Petrov, O.; Ivanova, Y.; Gerova, M. SOCl2/EtOH: Catalytic system for synthesis of chalcones. Catal. Commun. 2008, 9, 315–316. [Google Scholar] [CrossRef]
- Ivanova, Y.; Momekov, G.; Kalcheva, V.; Petrov, O. Synthesis of chalcones condensed with an 1,3-azole ring using SOCl2/EtOH catalytic systhem. C. R. Acad. Bulg. Sci. 2008, 61, 41–48. [Google Scholar]
- Ivanova, Y.; Momekov, G.; Petrov, O. New heterocyclic chalcones. Part 6: Synthesis and cytotoxic activities of 5- or 6-(3-aryl-2-propenoyl)-2(3H)-benzoxazolones. Heterocycl. Commun. 2013, 19, 23–28. [Google Scholar] [CrossRef]
- Ivanova, Y.; Petrov, O.; Gerova, M.; Momekov, G. Synthetic chalcones of 2(3H)-benzothiazolones with potential cytotoxic activity. C. R. Acad. Bulg. Sci. 2007, 60, 642–650. [Google Scholar]
- Ivanova, Y.; Momekov, G.; Petrov, O. Synthesis of novel substituted 1,3-diarylpropenone derivatives and their in vitro cytotoxic activity. Lett. Drug Des. Discov. 2009, 6, 353–357. [Google Scholar] [CrossRef]
- Petrov, O.; Ivanova, Y.; Momekov, G.; Kalcheva, V. New synthetic chalcones: Cytotoxic Mannich bases of 6-(4-chlorocinnamoyl)-2(3H)-benzoxazolone. Lett. Drug Des. Discov. 2008, 5, 358–362. [Google Scholar] [CrossRef]
- Ivanova, Y.; Momekov, G.; Petrov, O.; Karaivanova, M.; Kalcheva, V. Cytotoxic Mannich bases of 6-(3-aryl-2-propenoyl)-2(3H)-benzoxazolones. Eur. J. Med. Chem. 2007, 42, 1382–1387. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivanova, Y.; Gerova, M.; Chanev, C.; Petrov, O. (E)-3-Methyl-6-(3-oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)-2(3H)-benzothiazolone. Molbank 2016, 2016, M907. https://doi.org/10.3390/M907
Ivanova Y, Gerova M, Chanev C, Petrov O. (E)-3-Methyl-6-(3-oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)-2(3H)-benzothiazolone. Molbank. 2016; 2016(3):M907. https://doi.org/10.3390/M907
Chicago/Turabian StyleIvanova, Yordanka, Mariana Gerova, Christo Chanev, and Ognyan Petrov. 2016. "(E)-3-Methyl-6-(3-oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)-2(3H)-benzothiazolone" Molbank 2016, no. 3: M907. https://doi.org/10.3390/M907
APA StyleIvanova, Y., Gerova, M., Chanev, C., & Petrov, O. (2016). (E)-3-Methyl-6-(3-oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)-2(3H)-benzothiazolone. Molbank, 2016(3), M907. https://doi.org/10.3390/M907