Keywords:
pentanedione, nitro arene 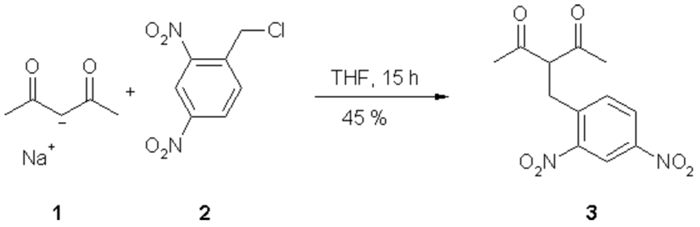
Substituted pentanediones are widely used as ligands for metal complexes.1 We report the synthesis and characterization of a 3-substituted pentanedione with an electron poor dinitrobenzyl substituent. The compound is sensitive and decomposes readily in the presence of ammonia.
To a solution of sodium acetyl acetonate (1.22 g, 10 mmol) (1) in THF (200 ml) were added 2,4-dinitrobenzylchloride (2.16 g, 10 mmol) (2) in 25 ml of THF at 0 °C. The reaction mixture was heated to reflux 1.5 h and kept at room temperature overnight. The solution was diluted with Et2O (100 ml), acidified with aqueous HCl (2 M), washed three times with H2O, dried over MgSO4 and the solvent was removed in vacuo. The crude product was purified by column chromatography on silica gel [petrol ether (60/70)/Et2O 3:1] to yield 1.28 g (45 %) of 3 (Rf = 0.35); yellow-orange solid, mp. 118 - 120 °C.
IR (KBr): 3117 cm-1, 1700, 1604, 1539, 1350, 1174, 734.
UV/Vis (CH3CN): lmax (log e) = 192 nm (4.248), 200 (4.153), 250 (4.177).
1H NMR (400 MHz, CDCl3): Enol form: d = 2.04 (s, 6H, CH3), 4.08 (s, 2H, benzyl-CH2), 7.54 (d, 3J = 8.7 Hz, 1H, arene-H), 8.42 (dd, 3J = 8.7 Hz, 4J = 2.4 Hz, 1H, arene-H), 8.84 (m, 1H, arene-H), 16.96 (s, 1H, enol-H). Keto form: d = 2.27 (s, 6H, CH3), 3.49 (d, 3J = 6.7 Hz, 2H, benzyl-CH2), 4.20 (t, 3J = 6.7 Hz, 1H, CH), 7.74 (d, 3J = 8.5 Hz, 1H, arene-H), 8.36 (dd, 3J = 8.5 Hz, 4J = 2.4 Hz, 1H, arene-H), 8.84 (m, 1H, arene-H).
13C NMR (100 MHz, CDCl3): d = 23.16 (+), 30.04 (+), 30.17 (-), 30.89 (-), 67.14 (+), 105.00 (Cquat), 120.51 (+), 120.57 (+), 127.22 (+), 127.40 (+), 130.30 (+), 135.32 (+), 140.60 (Cquat), 141.66 (Cquat), 192.31 (Cquat), 201.84 (Cquat).
MS (70 eV), m/z (%): 237 (2) [M+-CO-CH3], 43 (100) [CO-CH3].
EA Calculated (%): C 51.43 H 4.32 N 10.00
found (%): C 51.26 H 4.31 N 9.96.
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References and Notes
- Murakami, Y.; Nakamura, K.; Uchida, H.; Kanaoka, Y. Inorg. Chim. Acta 1968, 2(2), 133–8.
- Sample availability: available form the authors.
© 2003 MDPI. All rights reserved.