The Use of Photodynamic Therapy for Head, Neck, and Brain Diseases
Abstract
1. Introduction
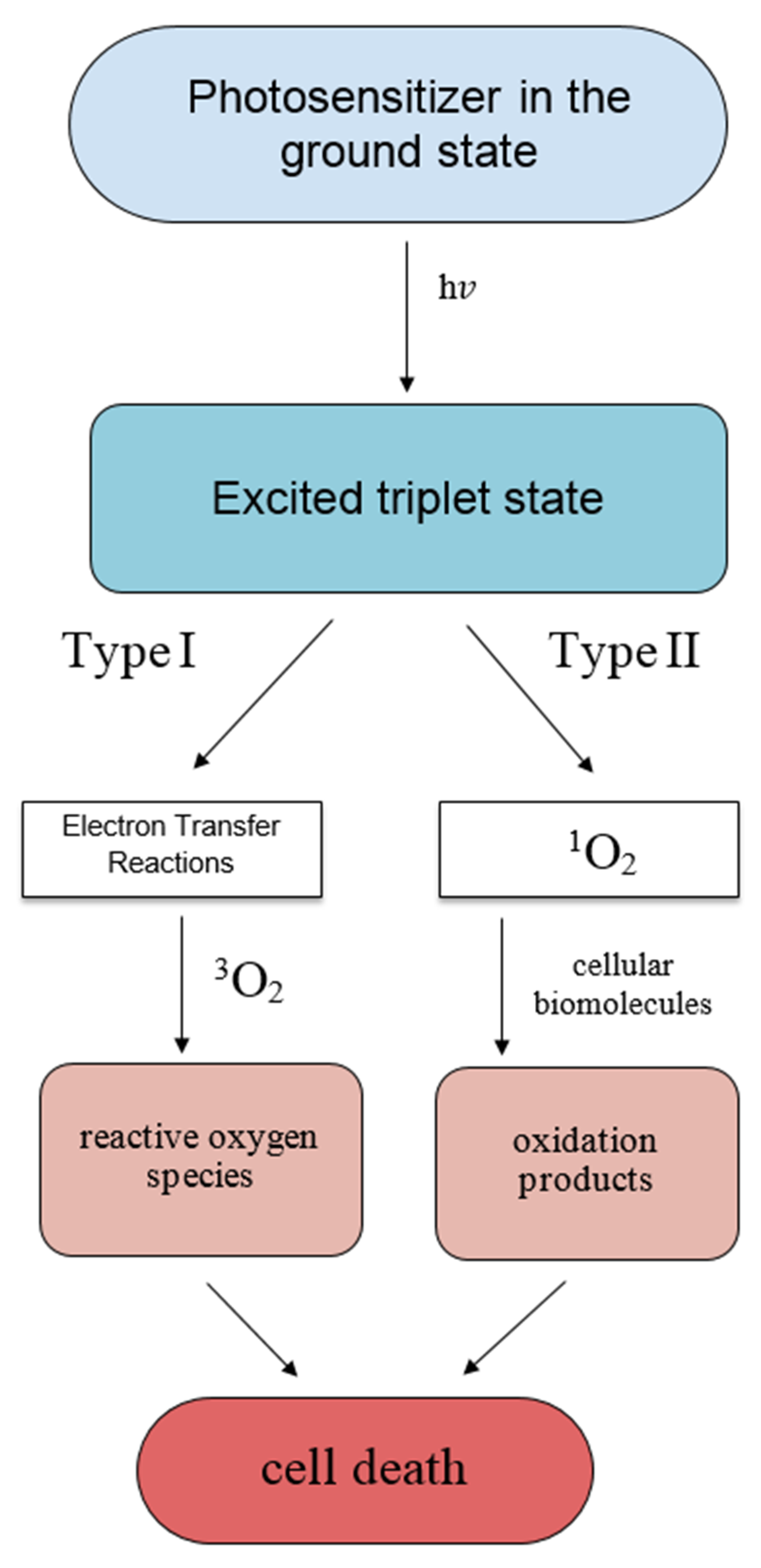
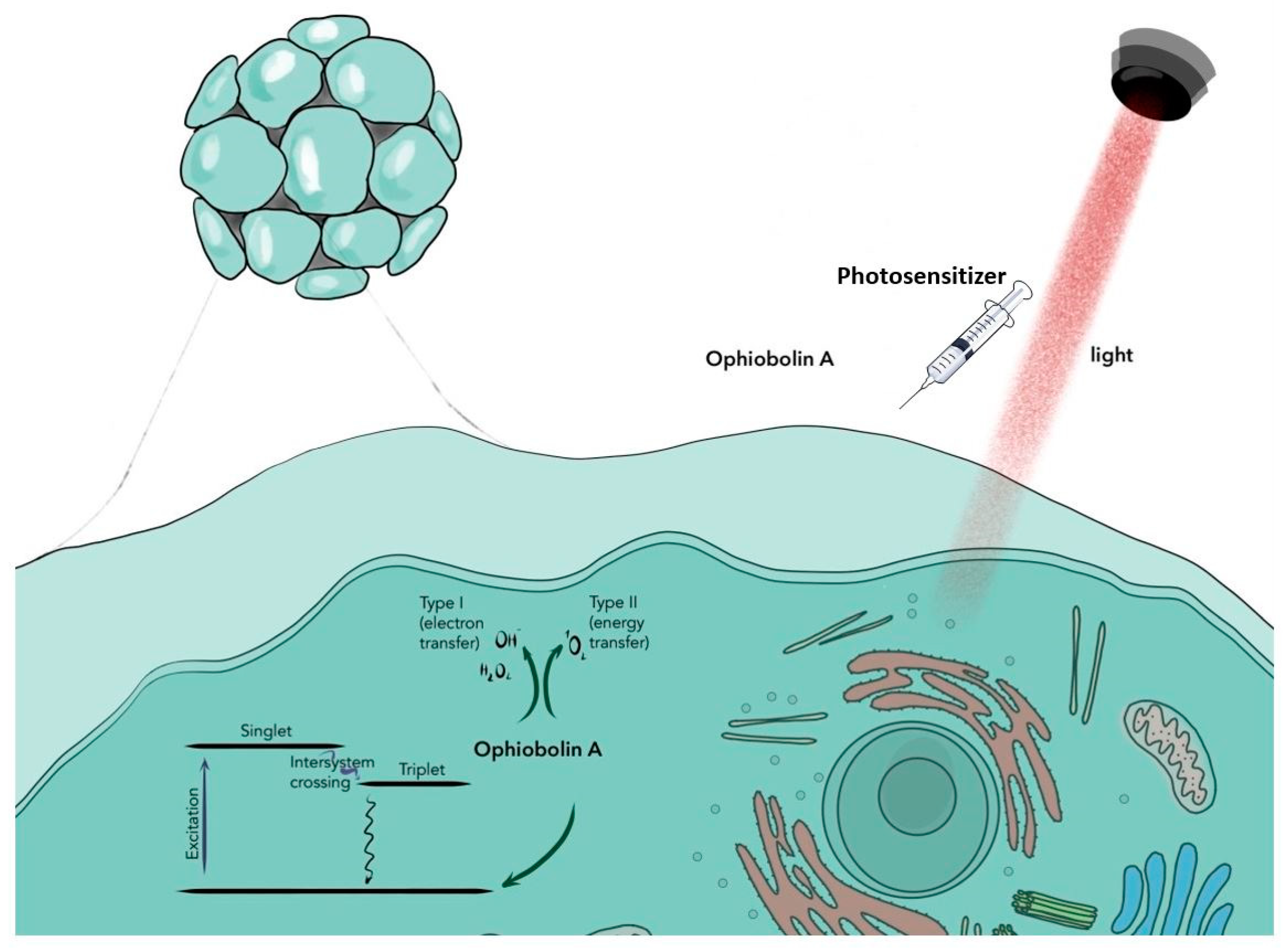
General Principles of Photodynamic Therapy
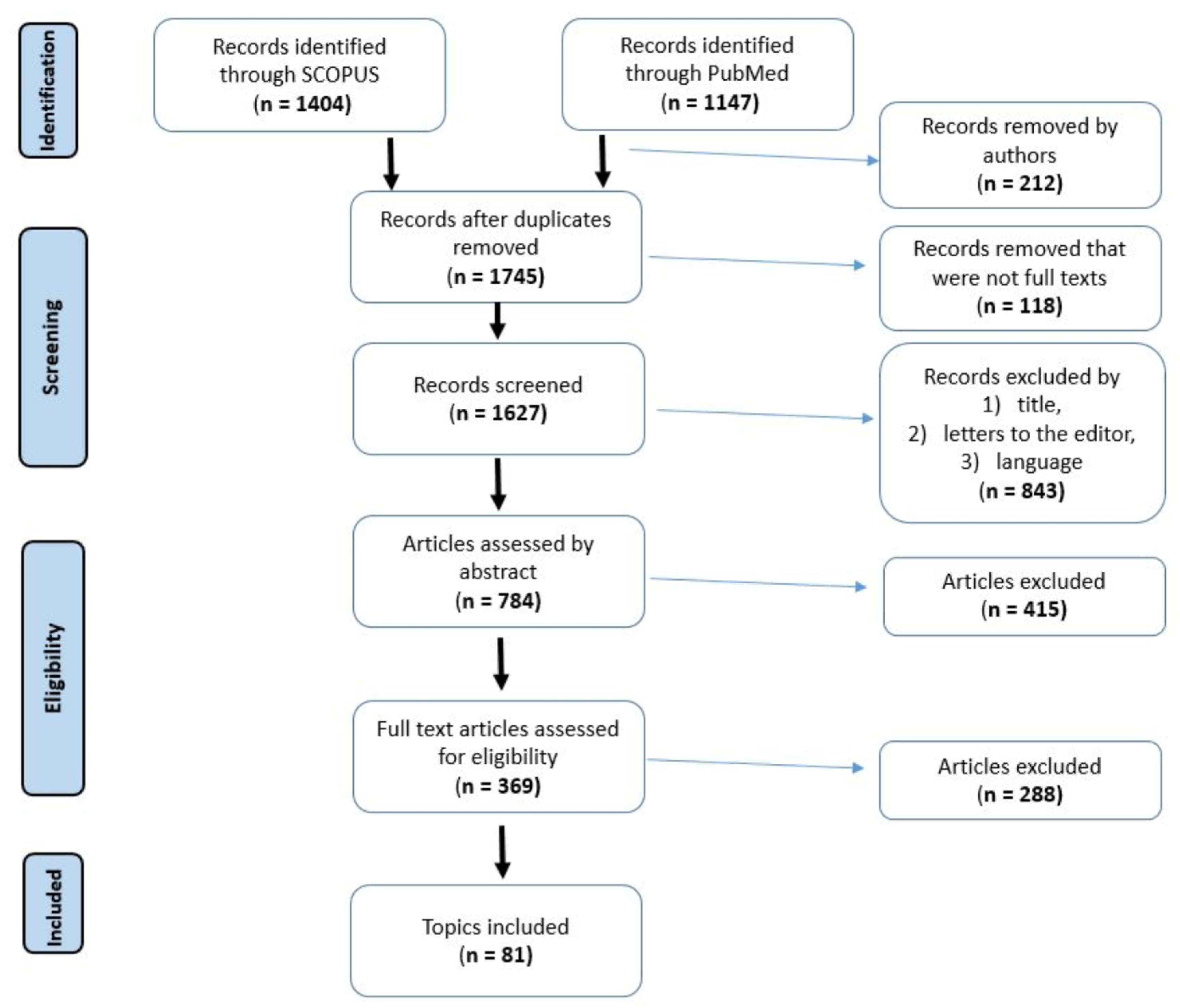
2. Methodology
3. A Review of the Literature
3.1. Brain Diseases
3.2. Head, Neck, and Oral Diseases
4. Discussion
Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Margalit, D.N.; Salz, T.; Venchiarutti, R.; Milley, K.; McNamara, M.; Chima, S.; Wong, J.; Druce, P.; Nekhlyudov, L. Interventions for head and neck cancer survivors: Systematic review. Head Neck 2022, 44, 2579–2599. [Google Scholar] [CrossRef] [PubMed]
- Wild, C.P.; Espina, C.; Bauld, L.; Bonanni, B.; Brenner, H.; Brown, K.; Dillner, J.; Forman, D.; Kampman, E.; Nilbert, M.; et al. Cancer Prevention Europe. Mol. Oncol. 2019, 13, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef]
- Cohen, E.; LaMonte, S.J.; Erb, N.L.; Beckman, K.L.; Sadeghi, N.; Hutcheson, K.; Stubblefield, M.D.; Abbott, D.M.; Fisher, P.S.; Stein, K.D.; et al. American Cancer Society Head and Neck Cancer Survivorship Care Guideline. CA A Cancer J. Clin. 2016, 66, 203–239, Erratum in CA Cancer J. Clin. 2016, 66, 351. [Google Scholar] [CrossRef] [PubMed]
- Rettig, E.M.; D’souza, G. Epidemiology of Head and Neck Cancer. Surg. Oncol. Clin. North Am. 2015, 24, 379–396. [Google Scholar] [CrossRef]
- Fried, J.; Etter, J.L.; Stenzel, A.E.; Joseph, J.M.; Cannioto, R.; Danziger, I.R.; Moysich, K.B. Physical inactivity and head and neck cancer mortality. Head Neck 2020, 42, 2516–2523. [Google Scholar] [CrossRef] [PubMed]
- Mathew, A.; Tirkey, A.J.; Li, H.; Steffen, A.; Lockwood, M.B.; Patil, C.L.; Doorenbos, A.Z. Symptom Clusters in Head and Neck Cancer: A Systematic Review and Conceptual Model. Semin. Oncol. Nurs. 2021, 37, 151215. [Google Scholar] [CrossRef] [PubMed]
- Perkins, A.; Liu, G. Primary Brain Tumors in Adults: Diagnosis and Treatment. Am. Fam. Physician 2016, 93, 211–217. [Google Scholar]
- Miller, K.D.; Ostrom, Q.T.; Kruchko, C.; Patil, N.; Tihan, T.; Cioffi, G.; Fuchs, H.E.; Waite, K.A.; Jemal, A.; Siegel, R.L.; et al. Brain and other central nervous system tumor statistics, 2021. CA A Cancer J. Clin. 2021, 71, 381–406. [Google Scholar] [CrossRef]
- Ailuno, G.; Balboni, A.; Caviglioli, G.; Lai, F.; Barbieri, F.; Dellacasagrande, I.; Florio, T.; Baldassari, S. Boron Vehiculating Nanosystems for Neutron Capture Therapy in Cancer Treatment. Cells 2022, 11, 4029. [Google Scholar] [CrossRef] [PubMed]
- Burian, J.; Marek, M.; Rataj, J.; Flibor, S. Report on the first patient group of the phase I BNCT trial at the LVR-15 reactor. Dev. Neurosci. 2004, 1259, 27–32. [Google Scholar] [CrossRef]
- Yamamoto, T.; Matsumura, A.; Nakai, K.; Shibata, Y.; Endo, K.; Sakurai, F.; Kishi, T.; Kumada, H.; Yamamoto, K.; Torii, Y. Current clinical results of the Tsukuba BNCT trial. Appl. Radiat. Isot. 2004, 61, 1089–1093. [Google Scholar] [CrossRef]
- Bown, S.G. Photodynamic therapy for photochemists. Philos. Trans. A Math Phys. Eng. Sci. 2013, 371, 20120371. [Google Scholar] [CrossRef]
- Webber, J.; Herman, M.; Kessel, D.; Fromm, D. Current Concepts in Gastrointestinal Photodynamic Therapy. Ann. Surg. 1999, 230, 12–23. [Google Scholar] [CrossRef]
- Biel, M.A. Photodynamic Therapy and the Treatment of Neoplastic Diseases of the Larynx. Laryngoscope 1994, 104, 399–403. [Google Scholar] [CrossRef]
- Donovan, L.; Welford, S.M.; Haaga, J.; LaManna, J.; Strohl, K.P. Hypoxia—Implications for pharmaceutical developments. Sleep Breath. 2010, 14, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.L.; Ribeiro, V. Drug Metabolism and Transport Under Hypoxia. Curr. Drug Metab. 2013, 14, 969–975. [Google Scholar] [CrossRef] [PubMed]
- Allison, R.; Mang, T.; Hewson, G.; Snider, W.; Dougherty, D. Photodynamic therapy for chest wall progression from breast carcinoma is an underutilized treatment modality. Cancer 2001, 91, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xie, Y.; Li, J.; Peng, Z.-H.; Sheinin, Y.; Zhou, J.; Oupický, D. Tumor-Penetrating Nanoparticles for Enhanced Anticancer Activity of Combined Photodynamic and Hypoxia-Activated Therapy. ACS Nano 2017, 11, 2227–2238, Erratum in ACS Nano 2019, 13, 4855. [Google Scholar] [CrossRef]
- Peng, Q.; Nesland, J.M. Effects of Photodynamic Therapy on Tumor Stroma. Ultrastruct. Pathol. 2004, 28, 333–340s. [Google Scholar] [CrossRef]
- Hodgkinson, N.; Kruger, C.A.; Abrahamse, H. Targeted photodynamic therapy as potential treatment modality for the eradication of colon cancer and colon cancer stem cells. Tumor Biol. 2017, 39, 1010428317734691. [Google Scholar] [CrossRef]
- Chidambaram, M.; Manavalan, R.; Kathiresan, K. Nanotherapeutics to overcome conven-tional cancer chemotherapy limitations. J. Pharm. Pharm. Sci. 2011, 14, 67–77. [Google Scholar] [CrossRef]
- Chenthamara, D.; Subramaniam, S.; Ramakrishnan, S.G.; Krishnaswamy, S.; Essa, M.M.; Lin, F.H.; Qoronfleh, M.W. Therapeutic efficacy of nanoparticles and routes of administration. Biomater. Res. 2019, 23, 20. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, T.J.; Gomer, C.J.; Henderson, B.W.; Jori, G.; Kessel, D.; Korbelik, M.; Moan, J.; Peng, Q. Photodynamic Therapy. J. Natl. Cancer Inst. 1998, 90, 889–905. [Google Scholar] [CrossRef]
- Longley, D.B.; Johnston, P.G. Molecular mechanisms of drug resistance. J. Pathol. 2005, 205, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Klotz, L.-O.; Kröncke, K.-D.; Sies, H. Singlet oxygen-induced signaling effects in mammalian cells. Photochem. Photobiol. Sci. 2003, 2, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Robertson, C.A.; Evans, D.H.; Abrahamse, H. Photodynamic therapy (PDT): A short review on cellular mechanisms and cancer research applications for PDT. J. Photochem. Photobiol. B 2009, 96, 1–8. [Google Scholar] [CrossRef]
- Mroz, P.; Yaroslavsky, A.; Kharkwal, G.B.; Hamblin, M.R. Cell Death Pathways in Photodynamic Therapy of Cancer. Cancers 2011, 3, 2516–2539. [Google Scholar] [CrossRef]
- Szliszka, E.; Czuba, Z.P.; Kawczyk-Krupka, A.; Sieron-Stoltny, K.; Sieron, A.; Krol, W. Chlorin-based photodynamic therapy enhances the effect of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) in bladder cancer cells. Med. Sci. Monit. 2012, 18, BR47–BR53. [Google Scholar] [CrossRef]
- Sibata, C.H.; Colussi, V.C.; Oleinick, N.L.; Kinsella, T.J. Photodynamic therapy: A new concept in medical treatment. Braz. J. Med. Biol. Res. 2000, 33, 869–880. [Google Scholar] [CrossRef] [PubMed]
- de Faria, C.M.G.; Inada, N.M.; Kurachi, C.; Bagnato, V.S. Determination of the threshold dose distribution in photodynamic action from in vitro experiments. J. Photochem. Photobiol. B Biol. 2016, 162, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Liu, P.; Tan, Y.; Zhang, Z.; Kang, M.; Wang, D.; Tang, B.Z. Type I Photosensitizers Based on Aggregation-Induced Emission: A Rising Star in Photodynamic Therapy. Biosensors 2022, 12, 722. [Google Scholar] [CrossRef] [PubMed]
- Maharjan, P.S.; Bhattarai, H.K. Singlet Oxygen, Photodynamic Therapy, and Mechanisms of Cancer Cell Death. J. Oncol. 2022, 2022, 7211485. [Google Scholar] [CrossRef] [PubMed]
- Baptista, M.S.; Cadet, J.; Di Mascio, P.; Ghogare, A.A.; Greer, A.; Hamblin, M.R.; Lorente, C.; Nunez, S.C.; Ribeiro, M.S.; Thomas, A.H.; et al. Type I and Type II Photosensitized Oxidation Reactions: Guidelines and Mechanistic Pathways. Photochem. Photobiol. 2017, 93, 912–919. [Google Scholar] [CrossRef]
- Barr, H.; Dix, A.J.; Kendall, C.; Stone, N. Review article: The potential role for photodynamic therapy in the management of upper gastrointestinal disease. Aliment. Pharmacol. Ther. 2001, 15, 311–321. [Google Scholar] [CrossRef]
- Al-Waili, N.S.; Butler, G.J.; Beale, J.; Hamilton, R.W.; Lee, B.Y.; Lucas, P. Hyperbaric oxygen and malignancies: A potential role in radiotherapy, chemotherapy, tumor surgery and phototherapy. Med. Sci. Monit. 2005, 11, RA279-289. [Google Scholar]
- Alvarez-Lorenzo, C.; Bromberg, L.; Concheiro, A. Light-sensitive Intelligent Drug Delivery Systems. Photochem. Photobiol. 2009, 85, 848–860. [Google Scholar] [CrossRef]
- Correia, J.H.; Rodrigues, J.A.; Pimenta, S.; Dong, T.; Yang, Z. Photodynamic Therapy Review: Principles, Photosensitizers, Applications, and Future Directions. Pharmaceutics. 2021, 13, 1332. [Google Scholar] [CrossRef]
- Wyss, P.; Schwarz, V.; Dobler-Girdziunaite, D.; Hornung, R.; Walt, H.; Degen, A.; Fehr, M. Photodynamic therapy of locoregional breast cancer recurrences using a chlorin-type photosensitizer. Int. J. Cancer 2001, 93, 720–724. [Google Scholar] [CrossRef]
- Lu, J.; Choi, E.; Tamanoi, F.; Zink, J.I. Light-Activated Nanoimpeller-Controlled Drug Release in Cancer Cells. Small 2008, 4, 421–426. [Google Scholar] [CrossRef]
- Shishkova, N.; Kuznetsova, O.; Berezov, T. Photodynamic Therapy for Gynecological Diseases and Breast Cancer. Cancer Biol. Med. 2012, 9, 9–17. [Google Scholar] [CrossRef]
- Swartling, J.; Axelsson, J.; Ahlgren, G.; Kalkner, K.M.; Nilsson, S.; Svanberg, S.; Svanberg, K.; Andersson-Engels, S. System for interstitial photodynamic therapy with online dosimetry: First clinical experiences of prostate cancer. J. Biomed. Opt. 2010, 15, 058003. [Google Scholar] [CrossRef]
- Yavlovich, A.; Smith, B.; Gupta, K.; Blumenthal, R.; Puri, A. Light-sensitive lipid-based nanoparticles for drug delivery: Design principles and future considerations for biological applications. Mol. Membr. Biol. 2010, 27, 364–381. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, T. Photodynamic Therapy Expands Its Horizons. J. Natl. Cancer Inst. 1997, 89, 112–114. [Google Scholar] [CrossRef] [PubMed]
- Lou, P.-J.; Jäger, H.R.; Jones, L.; Theodossy, T.; Bown, S.G.; Hopper, C. Interstitial photodynamic therapy as salvage treatment for recurrent head and neck cancer. Br. J. Cancer 2004, 91, 441–446. [Google Scholar] [CrossRef]
- Wang, L.; Yang, H.; Li, B. Photodynamic therapy for prostate cancer: A systematic review and meta-analysis. Prostate Int. 2018, 7, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Hudson, R.; Carcenac, M.; Smith, K.; Madden, L.; Clarke, O.J.; Pèlegrin, A.; Greenman, J.; Boyle, R.W. The development and characterisation of porphyrin isothiocyanate–monoclonal antibody conjugates for photoimmunotherapy. Br. J. Cancer 2005, 92, 1442–1449. [Google Scholar] [CrossRef]
- Narumi, A.; Tsuji, T.; Shinohara, K.; Yamazaki, H.; Kikuchi, M.; Kawaguchi, S.; Mae, T.; Ikeda, A.; Sakai, Y.; Kataoka, H.; et al. Maltotriose-conjugation to a fluorinated chlorin derivative generating a PDT photosensitizer with improved water-solubility. Org. Biomol. Chem. 2016, 14, 3608–3613. [Google Scholar] [CrossRef]
- Nishie, H.; Kataoka, H.; Yano, S.; Kikuchi, J.-I.; Hayashi, N.; Narumi, A.; Nomoto, A.; Kubota, E.; Joh, T. A next-generation bifunctional photosensitizer with improved water-solubility for photodynamic therapy and diagnosis. Oncotarget 2016, 7, 74259–74268. [Google Scholar] [CrossRef]
- Pröll, S.; Wilhelm, B.; Robert, B.; Scheer, H. Myoglobin with modified tetrapyrrole chromophores: Binding specificity and photochemistry. Biochim. Biophys. Acta 2006, 1757, 750–763. [Google Scholar] [CrossRef]
- Abrahamse, H.; Hamblin, M.R. New photosensitizers for photodynamic therapy. Biochem. J. 2016, 473, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Baskaran, R.; Lee, J.; Yang, S.-G. Clinical development of photodynamic agents and therapeutic applications. Biomater. Res. 2018, 22, 25. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guide-line for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Zilidis, G.; Aziz, F.; Telara, S.; Eljamel, M.S. Fluorescence image-guided surgery and repetitive Photodynamic Therapy in brain metastatic malignant melanoma. Photodiagn. Photodyn. Ther. 2008, 5, 264–266. [Google Scholar] [CrossRef] [PubMed]
- Stylli, S.S.; Kaye, A.H.; MacGregor, L.; Howes, M.; Rajendra, P. Photodynamic therapy of high grade glioma–long term survival. J. Clin. Neurosci. 2005, 12, 389–398. [Google Scholar] [CrossRef]
- Kim, I.Y.; Kwon, M.; Choi, M.-K.; Lee, D.; Lee, D.M.; Seo, M.J.; Choi, K.S. Ophiobolin A kills human glioblastoma cells by inducing endoplasmic reticulum stress via disruption of thiol proteostasis. Oncotarget 2017, 8, 106740–106752. [Google Scholar] [CrossRef]
- Masi, M.; Dasari, R.; Evidente, A.; Mathieu, V.; Kornienko, A. Chemistry and biology of ophiobolin A and its congeners. Bioorg. Med. Chem. Lett. 2019, 29, 859–869. [Google Scholar] [CrossRef]
- Akimoto, J.; Haraoka, J.; Aizawa, K. Preliminary clinical report on safety and efficacy of photodynamic therapy using talaporfin sodium for malignant gliomas. Photodiagn. Photodyn. Ther. 2012, 9, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Marks, P.V.; Belchetz, P.E.; Saxena, A.; Igbaseimokumo, U.; Thomson, S.; Nelson, M.; Stringer, M.R.; Holroyd, J.A.; Brown, S.B. Effect of photodynamic therapy on recurrent pituitary adenomas: Clinical phase I/II trial–an early report. Br. J. Neurosurg. 2000, 14, 317–325. [Google Scholar] [CrossRef]
- Igbaseimokumo, U. Quantification of in vivo Photofrin uptake by human pituitary adenoma tissue. J. Neurosurg. 2004, 101, 272–277. [Google Scholar] [CrossRef]
- Stepp, H.; Beck, T.; Pongratz, T.; Meinel, T.; Kreth, F.-W.; Tonn, J.C.; Stummer, W. ALA and Malignant Glioma: Fluorescence-Guided Resection and Photodynamic Treatment. J. Environ. Pathol. Toxicol. Oncol. 2007, 26, 157–164. [Google Scholar] [CrossRef]
- Muragaki, Y.; Akimoto, J.; Maruyama, T.; Iseki, H.; Ikuta, S.; Nitta, M.; Maebayashi, K.; Saito, T.; Okada, Y.; Kaneko, S.; et al. Phase II clinical study on intraoperative photodynamic therapy with talaporfin sodium and semiconductor laser in patients with malignant brain tumors. J. Neurosurg. 2013, 119, 845–852. [Google Scholar] [CrossRef]
- Muller, P.J.; Wilson, B.C. Photodynamic Therapy for Malignant Newly Diagnosed Supratentorial Gliomas. J. Clin. Laser Med. Surg. 1996, 14, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Biel, M.A. Photodynamic Therapy and the Treatment of Head and Neck Cancers. J. Clin. Laser Med. Surg. 1996, 14, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Tobita, T.; Ohba, S.; Uehara, M.; Asahina, I. Treatment outcome of Photofrin-based photodynamic therapy for T1 and T2 oral squamous cell carcinoma and dysplasia. Photodiagn. Photodyn. Ther. 2013, 10, 229–235. [Google Scholar] [CrossRef]
- Jerjes, W.; Upile, T.; Hamdoon, Z.; Mosse, C.A.; Akram, S.; Hopper, C. Photodynamic therapy outcome for oral dysplasia. Lasers Surg. Med. 2011, 43, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Jerjes, W.; Upile, T.; Hamdoon, Z.; Alexander Mosse, C.; Morcos, M.; Hopper, C. Photodynamic therapy outcome for T1/T2 N0 oral squamous cell carcinoma. Lasers Surg. Med. 2011, 43, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Wanitphakdeedecha, R.; Tavechodperathum, N.; Tantrapornpong, P.; Suphatsathienkul, P.; Techapichetvanich, T.; Eimpunth, S.; Manuskiatti, W. Acne treatment efficacy of intense pulsed light photodynamic therapy with topical licochalcone A, l -carnitine, and decanediol: A spilt-face, double-blind, randomized controlled trial. J. Cosmet. Dermatol. 2020, 19, 78–87. [Google Scholar] [CrossRef]
- Stoker, S.; Indrasari, S.; Herdini, C.; Hariwiyanto, B.; Karakullukcu, B.; Dhamiyati, W.; Widayati, K.; Romdhoni, A.; Fles, R.; Haryana, S.; et al. Photodynamic therapy as salvage therapy for patients with nasopharyngeal carcinoma experiencing local failures following definitive radiotherapy. Photodiagn. Photodyn. Ther. 2015, 12, 519–525. [Google Scholar] [CrossRef]
- Jerjes, W.; Upile, T.; Radhi, H.; Hopper, C. Photodynamic therapy and end-stage tongue base cancer: Short communication. Head Neck Oncol. 2011, 3, 49. [Google Scholar] [CrossRef]
- Kübler, A.; de Carpentier, J.; Hopper, C.; Leonard, A.; Putnam, G. Treatment of squamous cell carcinoma of the lip using Foscan-mediated Photodynamic Therapy. Int. J. Oral Maxillofac. Surg. 2001, 30, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Jansen, M.H.E.; Kessels, J.P.H.M.; Nelemans, P.J.; Kouloubis, N.; Arits, A.H.M.M.; van Pelt, H.P.A.; Quaedvlieg, P.J.F.; Essers, B.A.B.; Steijlen, P.M.; Kelleners-Smeets, N.W.J.; et al. Randomized Trial of Four Treatment Approaches for Actinic Keratosis. New Engl. J. Med. 2019, 380, 935–946. [Google Scholar] [CrossRef] [PubMed]
- Buinauskaite, E.; Maciulaitis, R.; Buinauskiene, J.; Valiukeviciene, S. Topical photodynamic therapy of actinic keratoses with 5-aminolevulinic acid: Randomized controlled trial with six months follow-up. J. Dermatol. Treat. 2013, 25, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Alfouzan, A.F. Radiation therapy in head and neck cancer. Saudi Med. J. 2021, 42, 247–254. [Google Scholar] [CrossRef]
- Schirrmacher, V. From chemotherapy to biological therapy: A review of novel concepts to reduce the side effects of systemic cancer treatment (Review). Int. J. Oncol. 2019, 54, 407–419. [Google Scholar] [CrossRef]
- Secchi, V.; Cova, F.; Villa, I.; Babin, V.; Nikl, M.; Campione, M.; Monguzzi, A. Energy Partitioning in Multicomponent Nanoscintillators for Enhanced Localized Radiotherapy. ACS Appl. Mater. Interfaces 2023, 15, 24693–24700. [Google Scholar] [CrossRef]
- Li, M.; Luo, Z.; Zhao, Y. Self-Assembled Hybrid Nanostructures: Versatile Multifunctional Nanoplatforms for Cancer Diagnosis and Therapy. Chem. Mater. 2018, 30, 25–53. [Google Scholar] [CrossRef]
- Crapanzano, R.; Secchi, V.; Villa, I. Co-Adjuvant Nanoparticles for Radiotherapy Treatments of Oncological Diseases. Appl. Sci. 2021, 11, 7073. [Google Scholar] [CrossRef]
- Secchi, V.; Monguzzi, A.; Villa, I. Design Principles of Hybrid Nanomaterials for Radiotherapy Enhanced by Photodynamic Therapy. Int. J. Mol. Sci. 2022, 23, 8736. [Google Scholar] [CrossRef]
- Shrestha, S.; Wu, J.; Sah, B.; Vanasse, A.; Cooper, L.N.; Ma, L.; Li, G.; Zheng, H.; Chen, W.; Antosh, M.P. X-ray induced photodynamic therapy with copper-cysteamine nanoparticles in mice tumors. Proc. Natl. Acad. Sci. USA 2019, 116, 16823–16828. [Google Scholar] [CrossRef] [PubMed]
| Type of Photosensitizer | Activation Wavelength | Characteristics |
|---|---|---|
| Hematoporphyrin derivatives | 620–650 nm | Absorb the light wave effectively at the certain wavelength |
| Phenothiazine (including toluidine blue and methylene blue) | 620–700 nm | Have appropriate energy at the triplet state to provide sufficient energy at the transfer to the ground state |
| Cyanine | 600–805 nm | Possess appropriate quantum yield |
| Phytotherapeutic agents | 550–700 nm | Possess long lifetime at the triplet state |
| Phthalocyanine | 660–700 nm | Have appropriate and high photostability |
| Photosensitizer | Wavelength [nm] | Application |
|---|---|---|
| Ameluz® (Biofrontera, Inc., Wakefield, MA, USA)/Levulan® | 635 | Mild to moderate actinic keratosis |
| Metvix®/Metvixia® | 570–670 | Non-hyperkeratotic actinic keratosis and basal cell carcinoma |
| Foscan® | 652 | Advanced head and neck cancer |
| Laserphyrin® (Meiji Seika Pharma, Tokyo, Japan) | 664 | Early centrally located lung cancer |
| Visudyne® (Novartis, Basel, Switzerland) | 690 | Age-related macular degeneration |
| Redaporfin® (Luzitin SA, Coimbra, Portugal) | 749 | Biliary tract cancer |
| Fotolon | 665 | Nasopharyngeal, sarcoma |
| Radachlorin | 662 | skin cancer |
| Photochlor | 664 | Head and neck cancer |
| Tookad® | 762 | Prostate cancer |
| Antrin | 732 | Coronary artery disease |
| Photrex | 664 | AMD |
| Talaporfin | 664 | Colorectal neoplasms, Liver metastasis |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domka, W.; Bartusik-Aebisher, D.; Mytych, W.; Dynarowicz, K.; Aebisher, D. The Use of Photodynamic Therapy for Head, Neck, and Brain Diseases. Int. J. Mol. Sci. 2023, 24, 11867. https://doi.org/10.3390/ijms241411867
Domka W, Bartusik-Aebisher D, Mytych W, Dynarowicz K, Aebisher D. The Use of Photodynamic Therapy for Head, Neck, and Brain Diseases. International Journal of Molecular Sciences. 2023; 24(14):11867. https://doi.org/10.3390/ijms241411867
Chicago/Turabian StyleDomka, Wojciech, Dorota Bartusik-Aebisher, Wiktoria Mytych, Klaudia Dynarowicz, and David Aebisher. 2023. "The Use of Photodynamic Therapy for Head, Neck, and Brain Diseases" International Journal of Molecular Sciences 24, no. 14: 11867. https://doi.org/10.3390/ijms241411867
APA StyleDomka, W., Bartusik-Aebisher, D., Mytych, W., Dynarowicz, K., & Aebisher, D. (2023). The Use of Photodynamic Therapy for Head, Neck, and Brain Diseases. International Journal of Molecular Sciences, 24(14), 11867. https://doi.org/10.3390/ijms241411867








