Abstract
Metabolic syndrome (MetS) forms a cluster of metabolic dysregulations including insulin resistance, atherogenic dyslipidemia, central obesity, and hypertension. The pathogenesis of MetS encompasses multiple genetic and acquired entities that fall under the umbrella of insulin resistance and chronic low-grade inflammation. If left untreated, MetS is significantly associated with an increased risk of developing diabetes and cardiovascular diseases (CVDs). Given that CVDs constitute by far the leading cause of morbidity and mortality worldwide, it has become essential to investigate the role played by MetS in this context to reduce the heavy burden of the disease. As such, and while MetS relatively constitutes a novel clinical entity, the extent of research about the disease has been exponentially growing in the past few decades. However, many aspects of this clinical entity are still not completely understood, and many questions remain unanswered to date. In this review, we provide a historical background and highlight the epidemiology of MetS. We also discuss the current and latest knowledge about the histopathology and pathophysiology of the disease. Finally, we summarize the most recent updates about the management and the prevention of this clinical syndrome.
1. Introduction
1.1. History
In 1988, Reaven raised the possibility that insulin resistance (IR) is not only involved in the etiology of type 2 diabetes mellitus (T2DM) but also that of cardiovascular disease (CVD) [1]. Reaven remarked that IR frequently presents in conjunction with a set of abnormalities and described them as syndrome X. The qualifier “metabolic” was added to Reaven’s syndrome X to differentiate it from the pre-existing syndrome X in cardiology [2]. In sum, metabolic syndrome X is a risk factor for cardiovascular diseases (CVDs) even without concomitant T2DM and it includes IR, hyperinsulinemia, dysglycemia, dyslipidemia, and hypertension. The disorders are, respectively, assessed using six indices to make the diagnosis of metabolic syndrome (MetS): waist circumference, fasting glucose levels, triglyceride levels, high-density lipoprotein (HDL) levels, cholesterol levels, and blood pressure (Table 1) [2,3].

Table 1.
Evolution of metabolic syndrome diagnostic definitions throughout the years.
Notice that blood insulin is not part of the five criteria because measuring this parameter for large-scale screening is cumbersome in clinical practice. Waist circumference is employed instead, as it was later proven to strongly correlate with IR [9]. Total body fat did not initially take part in the characterization of Reaven’s MetS as he found many cases showing a reverse association between total body fat and IR. Subgroup analyses later revealed that body fat distribution was the missing element in the equation explaining heterogeneity in results [10]. “Apple-shaped” bodies demonstrate central or abdominal visceral obesity and therefore carry a greater risk of developing IR than individuals with “pear-shaped” bodies with subcutaneous fat accumulation [11,12]. However, waist circumference alone remains inconclusive of abdominal adiposity and must be interpreted along with body mass index (BMI) in order to promptly evaluate for the high-risk abdominal obesity [9]. In addition, excess visceral adipose tissue is often accompanied by fat infiltration of hepatocytes, a condition known as non-alcoholic fatty liver. The latter is a dangerous phenotype of the disease, possibly progressing to cirrhosis or liver cancer [13].
Not a disease per se, but a term that serves as an umbrella of risk factors for individuals to be at an increased risk of disease, MetS has appeared for the first time on PubMed in 1940. Alberti et al. described a global definition of MetS in 2009 where three abnormal findings would qualify a person for MetS. Risk factors such as raised blood pressure, dyslipidemia (raised triglycerides and lowered high-density lipoprotein cholesterol), raised fasting glucose, and central obesity were proposed by the International Diabetes Federation and the American Heart Association/National Heart, Lung, and Blood Institute [14].
A single set of cut points was used for all components except waist circumference, for which national or regional cut points were utilized. Several studies later on have shown that waist-to-height ratio was a better screening tool than waist circumference [14,15].
1.2. Epidemiology
According to the Center of Disease Control and Prevention (CDC), the United States witnessed a 35% increase in MetS prevalence since the appearance of the term in the 1980s till 2012 [16]. The incidence of MetS matches that of obesity and T2DM. About 85% of T2DM patients also have MetS and thus are at higher risk of CVDs [14]. As such, in 2017, about 12.2% of the USA adult population had T2DM. Around one-quarter of them were unaware of their disease. Unsurprisingly, the MetS prevalence was three times higher, marking about one-third of the American adult population [4]. Fortunately, the National Health and Nutrition Examination Survey (NHANES) released recent data demonstrating declining numbers of the disease with 24% in men and 22% in women [17].
2. Pathophysiology
2.1. Established Ideas
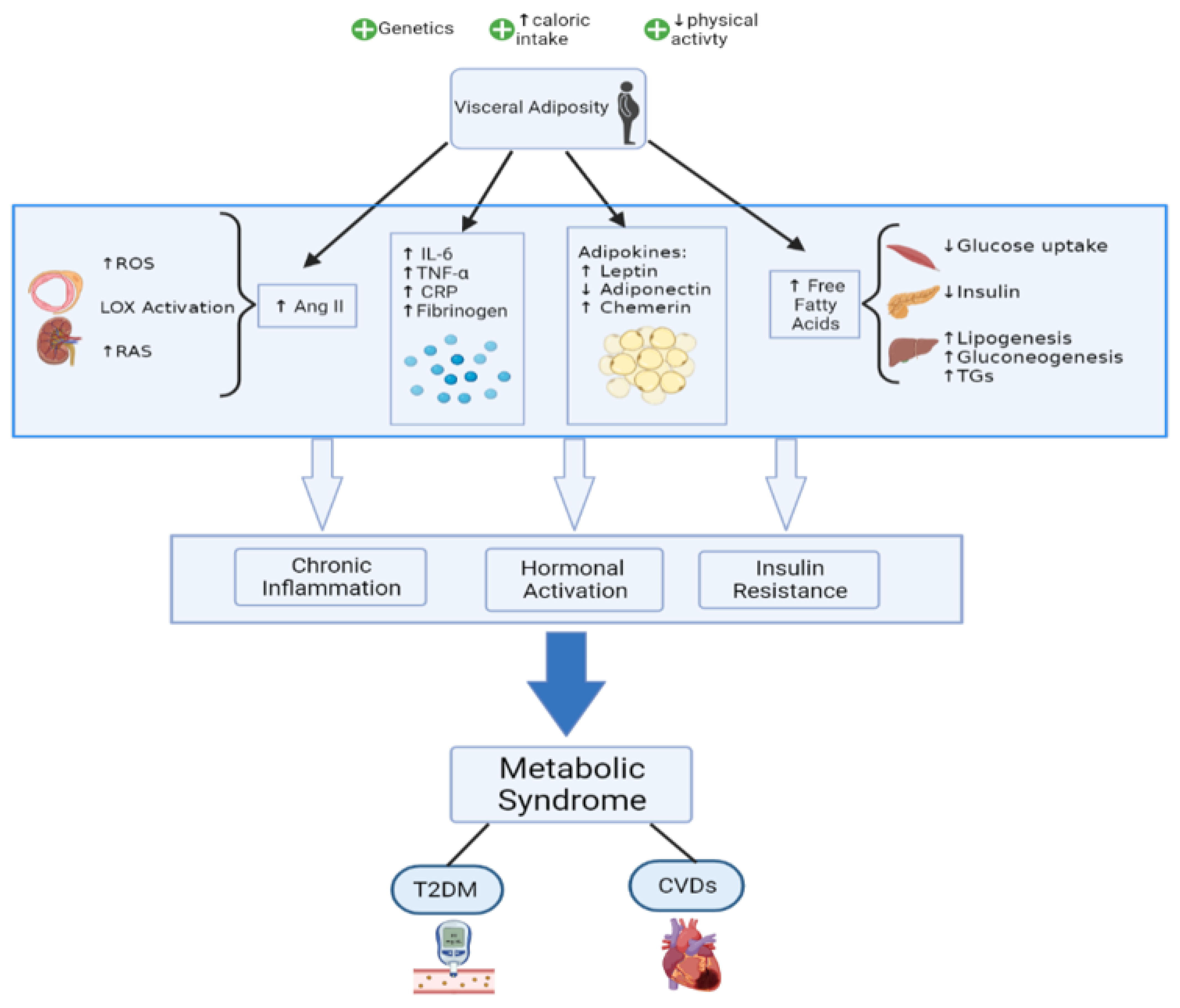
The pathophysiology of the MetS encompasses several complex mechanisms that are yet to be fully elucidated. It is still debated as to whether the different elements of MetS form by themselves distinct pathologies or fall under a common, broader pathogenic process. In addition to genetic and epigenetic factors [18], some lifestyle and environmental such as overeating and lack of physical activity have been identified as major contributors to the development of MetS. A causative role can be given to high caloric intake since visceral adiposity has been shown to be an important trigger that activates most of the pathways of MetS [19,20]. Among the proposed mechanisms, insulin resistance, chronic inflammation, and neurohormonal activation seem to be essential players in the progression of MetS and its subsequent transition to CVDs and T2DM (Figure 1).
Figure 1.
Mechanisms highlighting MetS pathophysiology.
2.1.1. Insulin Resistance
Insulin, a peptide hormone secreted by the pancreatic beta cells in response to high blood glucose, exerts its anabolic effects by inhibiting lipolysis and hepatic gluconeogenesis, while increasing glucose uptake in liver, muscles, and adipose tissues. When insulin resistance develops in fat tissues, insulin-mediated inhibition of lipolysis is impaired. The resulting increase in circulating free fatty acids (FFAs) in turn worsens insulin resistance by causing alterations in the insulin signaling cascade in different organs, thus creating a vicious cycle [21,22]. In muscles, FFAs affect insulin receptor substrate (IRS-1)-associated PI3K activity, leading to decreased GLUT-4 translocation to the surface and hence reduced glucose uptake [22]. In parallel, FFAs act on the liver to promote gluconeogenesis and lipogenesis. The net result is a hyperinsulinemic state to maintain normal glucose levels. However, the compensation eventually fails, leading to a decrease in insulin levels, which is further exacerbated by the lipotoxic effect of FFAs on beta cells of the pancreas [21,23]. It is essential to note that visceral lipolysis increases the supply of FFAs directly to the liver via the splanchnic circulation, thus making visceral fat deposits more important contributors to insulin resistance than subcutaneous fat [24].
In addition, high concentrations of FFAs increase cholesterol esters and triglyceride (TG) synthesis and subsequently the production of very low-density lipoproteins (VLDLs) rich in TGs. These in turn activate cholesterol ester transfer protein (CETP), which promotes the TGs transfer from VLDL to HDL, increasing HDL clearance and decreasing its concentrations [25]. Moreover, triglyceride-rich LDL, formed after exchange for LDL cholesterol ester, becomes hydrolyzed by lipoprotein or hepatic lipase, leading to cholesterol-depleted small dense LDL particles (Sd-LDL) [26]. All these alterations in lipoproteins concentrations constitute the hallmark of the atherogenic dyslipidemia caused by insulin resistance in MetS.
Another contribution of insulin resistance to MetS is the development of hypertension caused partly by loss of insulin’s vasodilatory effect and by FFA-induced vasoconstriction due to reactive oxygen species production and subsequent scavenging of nitric oxide [27]. Other mechanisms involve increased sympathetic stimulation and renin-induced sodium reabsorption in the kidneys [28]. Moreover, insulin resistance leads to higher serum viscosity, creates a prothrombotic state, and increases pro-inflammatory cytokine release from the adipose tissue, all of which play an important role in increasing the risks of CVDs and T2DM [29].
The best way to diagnose insulin resistance was known to be euglycemic hyperinsulinemic; however, many simple, cost effective and minimally invasive technics were established to assess a good predictive accuracy of this condition [30,31,32,33]. Utilizing these technics, simply using the fasting blood glucose and insulin levels, homeostatic model assessment–insulin resistance (HOMA-IR) index and quantitative insulin sensitivity check index (QUICKI) can be calculated. According to Hrebicek et al. (2002) and Singh and Saxena (2010), these two indices have been shown to be valuable and very helpful tools in evaluating insulin resistance in epidemiological studies and clinical practices.
This table demonstrates the area under the curve (AUC) of insulin resistance indices and their optimal cutoff points for diagnosis of MetS by sex. According to the results of Motamed et al. [34], the optimal cutoff points for HOMA-IR in the diagnosis of MetS in men was 2.0 (YI = 0.312) and in women was 2.5 (YI = 0.255). The optimal cutoff points for diagnosis of MetS for QUICKI was 0.343 (YI = 0.315) in men and 0.331 (YI = 0.264) in women. The AUC curves of insulin resistance indices and their optimal cutoff points for diagnosis of MetS can be found in Table 2.

Table 2.
HOMA-IR and QUICKI cut off value.
2.1.2. Adipose Tissue, an Endocrine Organ
Aside from being a thermoregulator and lipid storage facility, the recently discovered endocrine function of the adipose tissue provides additional mechanistic understandings to the development of MetS [35]. The various adipokines released include hormones (e.g., leptin, adiponectin), peptides (e.g., angiotensinogen, apelin, resistin, and plasminogen activator inhibitor (PAI)-1), and inflammatory cytokines (e.g., interleukin (IL)-6, tumor necrosis factor α (TNFα), visfatin, omentin, and chemerin), all of which play a major role in the pathophysiology of insulin resistance and MetS [36] (Table 2). Among the hormones released, leptin levels were shown to be directly proportional to obesity and body fat levels [37]. When body energy stores are adequate, leptin suppresses food intake and stimulates energy expenditure while also controlling glucose homeostasis and insulin sensitivity [38]. However, the failure of high leptin levels to correct the metabolic imbalance seen in obesity has given rise to the concept of “leptin resistance”, in which tissues have decreased sensitivity to leptin [39]. Moreover, leptin is known to promote a proinflammatory immune response as it was shown to activate the Th1 pathway and reverse the starvation-induced immunosuppression [40]. Since higher leptin levels correlate with increased cardiovascular risk and inflammation, leptin is suggested to be an important factor linking obesity, MetS, and CVDs [41].
In contrast, adiponectin’s effects counter those of leptin as it was established as an anti-atherogenic, anti-inflammatory, and anti-diabetic adipokine [42,43]. These properties emanate from the fact that adiponectin affects the nuclear factor kappa-light-chain enhancer of activated B cells (NF-kB) inflammatory pathway [44], increases insulin sensitivity [45], inhibits vascular smooth muscle cell (VSMC) proliferation, and stabilizes plaque formation [46]. Several studies have shown decreased adiponectin levels in patients with coronary heart disease [47,48], diabetes [49], and hypertension [50] compared to control subjects, thus conferring a protective role for adiponectin against the development and progression of insulin resistance, high blood pressure, and CVDs. In fact, individuals with reported genetic hypoadiponectinemia caused by a missense mutation demonstrate higher tendency to develop MetS [51].
In addition to the dysregulation of leptin and adiponectin levels in MetS, chemerin, a well characterized chemoattractant abundantly produced by adipocytes, has recently gained attention as a potential MetS biomarker after its suggested involvement in inflammation, glucose metabolism, adipogenesis, and angiogenesis in animal models and cell culture studies [52,53]. In humans, several small cohort studies revealed a significant increase in circulating chemerin levels in individuals fulfilling MetS criteria [54,55,56], and this significance persisted even after adjusting for waist circumference or BMI, since obesity itself is known to increase chemerin levels [57]. Although Chu et al. [58] report that high chemerin with low adiponectin increase the risk of MetS with an odds ratio of 5.8, a recent report by Shafer-Eggleton et al. [59] suggests that while the chemerin/HDL-C ratio can be a superior biomarker and better predictor of nascent MetS than hsCRP, the chemerin/adiponectin ratio is not significant after adjustment for waist circumference [59]. Larger studies need to confirm these findings and establish the true relationship between chemerin levels and MetS.
Another important neurohormonal pathway in the development of MetS is the involvement of the renin–angiotensinogen system (RAS). As mentioned earlier, the adipose tissue produces the peptide angiotensin II (Ang II) after activation of angiotensin-converting enzyme. Plasma Ang II levels were shown to be increased in obesity and insulin resistance [60]. The peptide exerts its pathogenic effects through activation of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, which increases the production of reactive oxygen species (ROS) [61,62]. ROS has multiple pleiotropic effects, including endothelial injury, expression of NF-kB, platelet aggregation, oxidation of LDL, and expression of lipoprotein receptor-1 (LOX-1) on VSMCs and endothelium. Together, RAS, LOX-1, and ROS form a positive feedback loop and induce a vicious cycle of endothelial dysfunction, inflammation, and fibroblast proliferation, leading to the progression of dyslipidemia, T2DM, hypertension, vasculopathies, and CVDs [63,64,65].
2.1.3. Chronic Inflammation
The various pathogenic pathways contributing to the development of MetS culminate in a pro-inflammatory state that explains the elevation in various inflammatory markers such as IL-6, C-reactive protein (CRP), and TNFα seen in individuals with MetS [66] (Table 3). As mentioned earlier, insulin resistance and obesity-induced systemic oxidant stress activates downstream inflammatory cascades, leading to tissue fibrosis, atherogenesis, and subsequently CVDs [67].

Table 3.
Important inflammatory biomarkers and adipokine dysregulation in MetS. ↑ signifies increase. ↓ signifies decrease.
IL-6 is one of the cytokines released by both macrophages and adipocytes [68], and its levels were shown to be increased with insulin resistance and obesity. In fact, IL-6 is known to regulate fat and glucose metabolism, mediating insulin resistance by way of different complex mechanisms [69].
This cytokine acts on various tissues, leading to the metabolic effects of obesity. In the liver, IL-6 increases the production of acute phase reactants including CRP. High levels of CRP were demonstrated in several studies to have the strongest correlation with cardiac events, T2DM, and MetS [70]. IL-6 also favors a prothrombotic state by increasing fibrinogen levels, another acute phase reactant [71]. Moreover, IL-6 targets other tissues such as VSMCs and endothelial cells to promote the expression of vascular cell adhesion molecules (VCAMs) and local RAS pathway activation, leading to vascular wall atherosclerosis, inflammation, and dysfunction [72].
TNFα is another cytokine produced within the adipose tissue, mainly from the local macrophages, and its production also varies proportionally with adipose tissue mass and correlates with insulin resistance, both being major features of MetS [68,73]. TNFα exerts its pathogenic effects by impairing insulin signaling in adipocytes and hepatocytes through serine phosphorylation and inactivation of insulin receptors and downstream signaling molecules, resulting in diminished metabolic effects of insulin [74]. TNFα also contributes to insulin resistance by inducing hepatic lipolysis, thus increasing FFA levels in the circulation [75].
Other players contributing to the chronic inflammatory state seen in MetS are the innate immune system receptors such as Toll-like receptors (TLRs). TLRs are involved in pathogen recognition and modulate the innate immune response by activating downstream inflammatory signaling pathways that lead to the release of various cytokines (TNFα, IL-6, IL-1β, and monocyte chemoattractant protein-1 (MCP-1)) [76]. The immune response is initiated after TLR recognition of ligands such as pathogen-associated molecular patterns (PAMPs) emanating from pathogens (e.g., LPS) and damage-associated molecular patterns (DAMPs) derived from damaged inflamed host tissues. Among the DAMPs, many endogenous ligands such as saturated fatty acids (SFAs), modified LDLs, advanced glycation end-products, extracellular matrix degradation products, and heat shock proteins, are recognized by TLRs, especially TLR2 and TLR4, activating a pro-inflammatory cascade [76,77].
Several animal and human studies have highlighted the role of TLRs in MetS. In mouse models with diet induced MetS, TLR4 expression and activity was shown to be increased. In addition, protection from high-fat diet-induced insulin resistance was conferred to TLR4 KO mice with lesser tissue inflammation observed [78]. Similarly, when compared to wild-type mice, TLR2 deficient models exhibited diminished insulin resistance, adiposity, macrophage infiltration, and cytokine expression in adipose tissue [79].
In humans, a rise in mRNA levels and a significant increase in expression of both TLR2 and TLR4 on the surface of monocytes has been demonstrated in subjects with MetS, even after accounting for waist circumference [80]. Similar findings were observed by Hardy et al. [81] in adolescents with MetS when compared to BMI-matched controls. This increase in innate immune receptor activation has been correlated with an increasing level of both endogenous (fatty acids, Ox-LDL, lipopolysaccharide-binding protein, etc.) and exogenous (LPS) activators in patients with MetS [80,82]. The latter finding of higher endotoxin (LPS) levels in MetS is intriguing as it supports the hypothesis of gut microbiota perturbation in MetS, resulting in increased gut permeability [83] and higher levels of LPS, the classical ligand of TLR4 [80].
Thus, a thorough understanding of TLRs involvement in MetS would help establish a new selective therapeutic target which can alleviate the burden of the chronic inflammation present in MetS.
2.2. Novel Studies
The complex interplay of environmental factors, lifestyle, and genetic/epigenetic factors in the pathophysiology of MetS has led to the emergence of novel studies that evaluate new perspectives in the early diagnosis, classification of new biomarkers, and discovery of potential targets for therapeutic interventions.
2.2.1. Fetuin-A
Fetuin-A, also referred to as α2-Heremans–Schmid glycoprotein (AHSG), is a protein with pleiotropic metabolic effects secreted by the liver. In addition to being a hepatokine, fetuin-A has recently been described as a potential adipokine since its expression and secretion levels have been also shown to be increased in visceral adipose tissue of obese animal models [84] and humans with MetS [85]. The association of its circulating levels with MetS is currently gaining importance in the literature as a novel biomarker and possible risk factor for the development of MetS [86]. A recently published meta-analysis of 14 eligible studies showed that circulating fetuin-A levels are significantly higher in patients with MetS compared to controls, with a possible trend towards an increase in the risk of MetS with an increase in the circulating fetuin-A concentrations [87]. One mechanistic approach is the contribution of fetuin-A to insulin resistance, as demonstrated in several animal [88,89] and human [90] studies. In fact, fetuin-A inhibits the tyrosine kinase activity of the insulin receptor as well as IRS-1 phosphorylation and downstream molecules in the PI3k/akt pathway [88]. These effects were reversed in fetuin-A knockout mice, which exhibited enhanced insulin sensitivity, glucose clearance, and less serum FFAs and triglycerides [91]. Furthermore, fetuin-A mediates macrophage migration and infiltration into the adipose tissue by way of chemo-attractants, thus inducing inflammatory cytokine release and subsequently contributing to the development of MetS as discussed earlier [92].
In terms of regulation, the elevated concentrations of FFAs seen in patients with MetS induce the binding of the inflammatory protein NF-κB to fetuin-A promoter, hence increasing circulating fetuin-A mRNA expression, protein synthesis, and secretion [93]. In contrast, adiponectin has recently been shown to have an inhibitory effect on the expression of fetuin-A, which can partly explain the increased circulating fetuin-A concentrations in hypoadiponectinemia seen with the MetS [94].
Aside from being supported by experimental and clinical studies, the link between circulating fetuin-A and MetS has also been proposed by genetic studies. The human fetuin-A or the AHSG gene is located on chromosome 3q27, which has been identified as T2DM susceptibility locus [95] and has been mapped as a quantitative trait locus for MetS [96]. Moreover, single-nucleotide polymorphisms (SNPs) of the AHSG gene and their relationship to MetS features such as BMI have been reported in several studies [97]. Taken together, it appears that the association between circulating fetuin-A and MetS is facilitated at least partially by genetics.
2.2.2. Mitochondrial Dysfunction and PGC-1α in MetS
Aside from its essential role in ATP production, mitochondria are a major source of ROS generation due to the leak of few high energy electrons from the electron transport chain (ETC) and their direct reaction with oxygen [98]. However, mitochondria are equipped with a very effective antioxidant mechanism that activates superoxide dismutase and other enzymes to scavenge ROS produced either locally or by other organelles like the peroxisomes [99,100]. Thus, mitochondrial dysfunction can lead to ROS overproduction causing cell injury, the hallmark “oxidative stress” that contributes to the development of many disease processes including MetS [101].
In turn, several components of the MetS can be responsible for the excessive ROS production that overwhelms mitochondrial antioxidant capacity and leads to its dysfunction. On one hand, the adipocyte nutrient excess seen in individuals with MetS result in a compensatory upregulation of fatty acid oxidation that increases NADH and FADH2 load from the tricarboxylic acid cycle, providing more electron supply to the ETC. Spilling of some of these high-energy electrons off the chain produces excessive ROS and subsequently exhausts and damages the mitochondrial antioxidant defense mechanisms [102,103]. Moreover, the excess FFAs in adipocytes activate NADPH oxidase, which increases ROS generation [104].
On the other hand, insulin resistance may also contribute to the decline in mitochondrial function. Insulin-resistant cells exhibit increased susceptibility to oxidative stress and reduced oxidative phosphorylation and energy production [105].
In addition, the decreased energy requirements due to the sedentary lifestyle seen in MetS reduces the levels of peroxisome proliferator-activated receptor gamma coactivator −1 α (PGC-1α) and thus decreases mitochondrial biogenesis. PGC-1α is described as an essential transcriptional activator and master regulator of mitochondrial biogenesis and function, particularly oxidative phosphorylation, and ROS detoxification [106]. Mitochondrial biogenesis pathways are activated in response to high-energy requirements translated by increased AMP:ADP/ATP and NAD+:NADH ratios, which are mediated by AMP-activated protein kinase (AMPK) and Sirtuin-1 pathways, respectively [107]. Increased expression of PGC-1α in energy-demanding tissues leads to the activation of the transcription factors estrogen related receptors (ERRs) and nuclear respiratory factors 1 and 2 (NRF-1 and NRF-2), which in turn induce the mitochondrial transcription factor A (TFAM) [108]. TFAM, along with the mitochondrial transcription factor B2 (TFB2M), activate the transcription of mitochondrial genes, leading to increased mitochondrial mass [109]. In addition, PGC-1α enhances fatty acid oxidation as it co-activates peroxisome proliferator-activated receptor α and δ (PPARα and PPARδ), responsible for the expression of mitochondrial fatty acid oxidation genes [110]. PGC-1α also increases the expression of antioxidant enzymes in the mitochondria, such as catalase, manganese superoxide dismutase, and heme oxygenase, leading to less oxidative stress and less mitochondrial dysfunction [111,112].
Therefore, any dysregulation in PGC-1α activity may alter the metabolic function of tissues, leading to the development of various metabolic diseases. In fact, SNPs in the human PGC-1α gene was demonstrated to be associated with diabetes, obesity, and hypertension, major components of the MetS [113]. Research interest in PGC-1α and its correlation with MetS is substantially growing since PGC-1α can be a potential therapeutic target against MetS [114,115] as it connects mitochondrial metabolism with redox homeostasis that is found to be dysfunctional in metabolic diseases.
As mentioned above, NADPH oxidase plays an essential role in the generation of ROS. It has been shown that the activation of the NADPH oxidase system increases the production of ROS, contributing to the pathogenesis of atherosclerosis, diabetes, and cardiovascular diseases [116]. Jialal et al. examined the difference in the expression of two subunits of NADPH oxidase (p22 phox and p47 phox) between participants with and without MetS and concluded that both subunits were significantly increased in participants with MetS as compared to controls [117]. While previous studies have shown an increase in the p22 phox subunit only, this is the first study to show an increase in both subunits [117,118]. This is likely because this study recruited patients with nascent MetS, and participants with diabetes mellitus or on statin therapy were excluded [117,118].
2.2.3. Circulatory MicroRNAs
MicroRNAs (miRNAs) are small evolutionarily conserved noncoding RNAs with various biological functions, mainly negatively regulating post-transcriptional gene expression [119]. In mammals, miRNAs control around 60% of all protein-coding genes [120]. Circulatory microRNAs (c-miRNAs) have gained enormous research interest because of the underscored significance of their expression profiles and regulatory functions in many diseases, as well as their putative use as biomarkers for diagnosis, progression, and disease prognosis [121,122]. The role of c-miRNAs in metabolism regulation and cardio-metabolic disorder development has been recently studied in both animal and human models. In fact, a dysregulation in the c-miRNA expression profile is demonstrated to be linked to different components of the MetS [123,124]. For example, miR-17-5p and miR-519d are related to obesity and lipogenesis [125,126], miR-375 controls insulin secretion from the pancreas [127], whereas the let-7 family affect insulin sensitivity and regulate various players in glucose metabolism [128]. Moreover, a differential miRNA expression between sexes has been reported, and this might help explain sex differences attributed to the progression to MetS and its complications [129,130].
A recent study by Ramzan et al. analyzed participants’ plasma expression of 26 miRNAs selected on the basis of evidence from multiple studies as being essential in the regulation of main components of cardio-metabolic diseases. The authors characterized different expression of panels and examined their relationship with early stage MetS before the progression to T2DM or CVDs [131]. Among these 26 miRNAs, 16 miRNAs were sex-specific, and 10 miRNAs were found to have altered circulating levels in participants diagnosed as having MetS, compared to healthy controls. Two of them, miR-17-5p and miR-15a-5p, were found to be the strongest predictors of MetS presence compared to healthy controls, as their expression panel was decreased in individuals with MetS, independent of sex [131]. Following putative target gene analysis [132], the two c-miRNAs had a robust regulatory communication with cellular pathways (fatty acid metabolism, AMPK, wnt, insulin, and TGF-β signaling) involved in the development of cardiometabolic diseases [133]. In addition to regulating insulin secretion and pancreatic β-cell proliferation and adaptation to metabolic stress [134], miR-17-5p was reported by Ramzan et al. to have a negative association with BMI and waist circumference, which was in concordance to previous studies [125], thus confirming the potential correlation between miR-17-5p and central adiposity, an important feature of MetS. Similarly, the pancreatic-specific miR-15a-5p was previously identified to interact with various signaling pathways such as insulin secretion and glucose metabolism [135], pancreatic inflammation [136], angiogenesis, and endothelial dysfunction [137]. The downregulation of miR-15a-5p in MetS was suggested in the study by Ramzan et al. to be mostly associated with increased total circulating TGs and visceral fat in both men and women [131]. As for the differential expression of miRNAs based on sex, this study featured a uniform downregulation in the let-7 miRNA family (let-7a-5p, miR-7c-5p, miR-7d-5p, and miR-7e-5p) in MetS men when compared to healthy controls [131]. This difference was not evident in women with MetS, which makes the let-7 family a sex-specific biomarker for MetS in men and hence can be further studied to delineate the sex differences attributed to the development and progression of MetS.
It is important to note that the miRNA expression panel modification is not only associated with the disease itself, but also shown to be induced by dietary interventions [138]. In a recent randomized clinical trial, Marsetti et al. analyzed the expression panel of miRNAs of white blood cells (WBCs) after administering each group of MetS participants two hypocaloric diets (traditional Mediterranean diet (MD) and the American Heart Association (AHA) dietary recommendations) for 8 weeks [139]. MD was previously suggested to reduce proinflammatory markers together with inducing weight loss [140], and similarly the AHA dietary recommendations were proven to promote health benefits, lower body fat, and ameliorate some features of the MetS [141]. In their trial, Marsetti et al. reported a significant change in the expression of a total of 49 miRNAs after 8 weeks of the weight loss dietary interventions. In particular, the most relevant changes were the downregulation of miR-214 and miR-190 expression panel and the upregulation of miR-410 and miR-637 expression panel. Moreover, miR-2115, miR-587, and miR-96 showed differential expressions after 8 weeks between the MD and AHA recommended diet [139]. Interestingly, the study highlights an association between miRNA expression modifications with biochemical and anthropometric parameters such as BMI and leptin (particularly with miR-410) and VCAM-1 (particularly with miR-587). This relationship suggests that these miRNAs might be directly or indirectly involved in inducing the effects of hypocaloric diets with different macronutrient composition, which subsequently regulate metabolic disease progression. Hence, in addition to being easy-to-measure disease biomarkers [142], miRNA expression panels can be potentially used as nutritional biomarkers to monitor response to dietary interventions.
2.2.4. IDEFICS/I.Family Study and the Underlying Genetic Predisposition for MetS Risk
Since the prevalence of MetS in children and young adult is on the rise [143,144], understanding the genetics behind MetS and their effect on the MetS network are essential for early detection of people with high genetic risk of disease development throughout their life. This provides a critical insight into the etiology of the disease and might encourage individuals at high risk to adopt lifestyle changes to reduce this risk. Nagrani et al. recently studied the link between common genetic variants and repeated MetS score of children and adolescents enrolled in the IDEFICS/I.Family cohort study with meta-analysis [145]. The IDEFICS (Identification and Prevention of Dietary- and Lifestyle-Induced Health Effects in Children and Infants) is a large multicenter population-based European cohort study that aims to explore the causes and prevent the development of diet and lifestyle-related diseases in children and adolescents with a special focus on overweight and obesity [146]. Baseline measures and survey of 16,224 children aged 2–9 years were collected with a repeated follow up after two years. A third wave of follow-up and measurements is done after six years by the I.Family study, creating a longitudinal database for the young population and their families [147].
Various studies reported common genetic loci associated with MetS at a single time-point using case–control or cross-sectional designs [148,149]. However, Nagrani et al. were the first to explore the longitudinal association of 350 pre-selected genetic loci with a MetS score calculated continuously throughout the transition from childhood to adolescence in the IDEFICS/I.Family cohort of children. Their results were confirmed with meta-analysis of identified SNPs [145]. A significant association affecting the repeated MetS score is seen in five SNPs of the fat mass and obesity associated (FTO) gene on chromosomal region 16q12., the strongest of which was with the variant rs80540136. This is supported by other studies, whereby variations of the FTO genes have been reported as contributors to childhood and adult obesity [150], as well as T2DM [151], both being features of the MetS. The nuclear protein coded by the FTO gene belongs to the superfamily of non-heme iron and 2-oxoglutarate-dependent oxygenases and is mainly expressed in the hypothalamus where it plays a fundamental role in regulation of food intake and energy homeostasis [152]. Its expression in the liver has recently gained attention and found to modify gene expression implicated in lipid and glucose metabolism [153].
In conclusion, we can infer that gene of obesity and lipid metabolism drive the genetic predisposition to MetS development, and some variants contribute to a more severe MetS phenotype (higher MetS score) than others. Accordingly, devising polygenic risk scores for MetS that include the associated genetic variants might be valuable for identifying high-risk groups in children and subsequently adults, thus allowing for prevention and earlier targeted interventions.
2.2.5. GPCRs Gene Expression Affected by MetS Etiology
As discussed previously, MetS development consists of an interplay between genetic, epigenetic, and environmental factors. Interestingly, gene expression of certain proteins has been recently shown to vary according to the MetS etiology. For example, Romero-Nava et al. highlight the differential genetic expression of two types of G protein-coupled receptors (GPCRs) caused by environmental and genetic etiologies of MetS by comparing gene expression in Wistar rats fed with 70% fructose for 9 weeks, serving as a diet-induced MetS model, and Zucker obese rats serving as the genetic model [154]. The studied receptors belong to the orphan receptors, a new class of GPCRs whose endogenous ligand was initially unknown. Orphan receptors’ expression had been demonstrated to change in different pathologies, such as GPR22 and GPR162 in diabetes [155], and GPR3, GPR6, and GPR12 in dyslipidemia [156].
As for their association with MetS, Romero-Nava et al. evaluated the gene expression of GPR21 and GPR82 in the heart, liver, kidney, brain, and aorta in both the diet rat models and genetic models in comparison with the controls. In fact, GPR21 has been suggested to play a role in affecting body weight, insulin sensitivity, and glucose tolerance as GPR21 KO mice have improved insulin response and are protected from obesity [157]. In the two MetS models (diet and genetic-induced) of Romero-Nava et al., GPR21 had different pattern of expression in different tissues. In the liver, a decrease in expression was reported in fructose-treated rats, contrarily to the increase seen in Zucker obese rats. This difference might have been due to the participation of this receptor in glucose metabolism in the liver and the fact that fructose-fed rats are overloaded with carbohydrates. No difference in expression was noted in the brain; however, the two models share the same pattern of decreased expression in the kidney, aorta, and the heart compared to controls. This hints that the GPR21 receptor could be involved in hypertension seen in MetS independently of its etiology since these three tissues are important for blood pressure control [154]. GPR21 mediates its signaling mechanism by Gq protein activation [158], which is known to form the second messengers diacylglycerol (DAG) and inositol 1,4,5-triphosphate (IP3) responsible for increasing intracellular calcium and vasoconstriction at the vascular level, promoting cardiac hypertrophy at the cardiac level and controlling glomerular filtration rate in the kidneys [159,160]. We postulate that the decrease GPR21 expression in the aorta, heart, and kidneys of the diet and genetic MetS models is a compensation towards lowering the high blood pressure observed in MetS.
GPR82 is another orphan receptor involved in metabolism, as GPR82-deficient mice were shown to decrease body weight, fat content, and TGs levels and to increase glucose tolerance and insulin sensitivity, all of which are associated with influences on food intake rather than modified respiratory and metabolic rates [161]. Although GPR82 receptors also mediate their effect through coupling to Gq proteins, their expression and effects vary according to tissue type and depend on whether the MetS model is diet or genetically induced [154]. For example, in the fructose-treated rats, their expression was decreased in the brain, kidney, and liver; increased in the heart; and not changed in the aorta. However, obese Zucker rats had an increased expression of GPR82 in the liver and brain but a decreased expression in the heart, aorta, and kidney.
The described results highlight that the differential expression of GPR21 and GPR82 receptors in various tissues is not only associated with the MetS but also depends on the etiology of the syndrome, thus being selective candidates for new therapeutic targets of metabolic diseases depending on their cause.
2.2.6. Parental MetS Epigenetic Effects on Offspring
Although the pathophysiology and the consequences of MetS on the patients have been extensively studied in the literature, little is known about the impact of maternal and/or paternal MetS on disease risk modification in offspring. Interestingly, it has been postulated that parental epigenetic alterations could be transferred to the next generation and thus reprogram the hepatic lipid metabolism of the offspring, contributing in particular to the development of non-alcoholic fatty liver disease (NAFLD) [162,163].
Animal models are being used to test this hypothesis linking parental MetS to NAFLD priming. For example, De Jesus et al. used a liver-specific insulin receptor knockout (LIKRO) mouse model that manifests three hallmarks of MetS: dyslipidemia, hyperglycemia, and insulin resistance [164,165]. Their results demonstrate that maternal and paternal MetS affect hepatic methylation status of members of the TGF-β superfamily, particularly the neuronal regeneration-related protein (NREP) and growth differentiation factor 15 (GDF15), which in turn alter the expression of many genes regulating hepatic lipid metabolism [166]. In fact, the offspring of either maternal or paternal LIKRO mice showed remarkable exacerbation in hepatic steatosis after administration of a high-fat diet [166].
One mechanistic explanation to NAFLD priming by De Jesus et al. suggests that downregulation of NREP increases protein expression of ATP-citrate lyase (ACLY) and 3-hydroxy-3-methylglutaryl-CoA reductase (HMGCR) [166], two enzymes involved in cholesterol and fatty acids biosynthesis [167]. This ACLY and HMGCR activation was demonstrated to be Akt (protein kinase B)-dependent in response to TGF-β signaling. In addition to previous studies that report that NREP exerts its action through inhibiting TGF-β1 and TGF-β2 receptor levels and decreasing TGF-β autoinduction by binding to the latency-associated protein (LAP) of TGF-β1 and TGF-β2 [168], we can infer that NREP decreases HMGCR and ACLY levels by altering Akt signaling via the noncanonical TGF-β pathway.
De Jesus et al. extrapolated their results to human subjects by reporting decreased hepatic NREP expression in patients with NAFLD, a significant negative correlation between NREP and ACLY hepatic mRNA levels, and an important association between low-serum NREP concentrations with steatosis grade and NAFL activity score [166].
Altogether, the results of this novel study highlight the contribution of parental MetS to the epigenome of the next generation by widespread DNA methylation modification and shed light on potential therapeutic targets to prevent disease progression and its subsequent complications in the clinic.
2.2.7. Metabolomics
The role of various metabolites in the pathogenesis of MetS has also been recently assessed [169]. In fact, biogenic amines that are usually found in red meats, including choline, L-carnitine, and trimethylamine-N-oxide, were shown to be associated with an adverse cardiometabolic profile and insulin resistance [169,170,171,172]. In addition, several amino acids, including alanine, glutamate, glutamine, aspartate and asparagine, arginine, histidine, methionine, cysteine, and lysine have been shown to play a role in the pathogenesis of MetS [169]. For example, branch chain amino acids and alanine have been shown to be involved in insulin resistance development [173]. In addition, together with aromatic amines (phenylalanine, tryptophan, tyrosine, and phospholipids), branch chain amino acids can even serve as biomarkers to predict the start of MetS in patients who have not yet developed diabetes [174,175,176]. On the other side, histidine and lysine were shown to have antioxidant properties, decreasing the inflammatory burden and oxidative stress [169,177,178].
2.3. MetS and Mice Studies
Mouse models can be used to study the pathophysiology by which obesity can lead to the development of IR. Elevations in abdominal circumference and IR have been seen in diet-induced and genetic mouse models of MetS. Many of the mouse models most recognized in Kennedy et al.’s study of obesity arose from spontaneous mutations. Among these are leptin-deficient (Lepob/ob), leptin receptor-deficient (LepRdb/db), and lethal yellow agouti (Ay/a) mice (Table 4) [179]. The abbreviations Lepob/ob and LepRdb/db will be used as they bring in elements of their traditional phenotypic, and their more recently discovered genetic, designations. Although their major phenotype is obesity, these mice also exhibit IR and some degree of dyslipidemia; thus, they can be used as models of MetS. The tub and fat mutations are other spontaneous monogenic forms of obesity seen in mice; however, these mice are less well characterized and are not as commonly used for studies of MetS. Two genetic knockout models of obesity were discussed, the melanocortin 4 receptor (MC4-R) and melanocortin 3 receptor (MC3-R) knockout mice.

Table 4.
Latest studies revolving around MetS in mice. ↑ signifies increase. ↓ signifies decrease.
3. Management
3.1. Mediterranean Diet
Long known to be associated with low risk of cardiovascular disease, the adherence to the Mediterranean diet has also been shown to decrease the risk of MetS [194,195]. It has been shown to have a protective role on every component of the MetS pentad [194]. The antioxidant and anti-inflammatory properties of the different Mediterranean diet components, including olive oil, fish, cereals, vegetables, and fruits, likely constitute the most likely explanation of those findings [196,197,198,199]. A recent metanalysis of 50 prospective studies and randomized control trials showed that the greater the adherence to the Mediterranean diet, the greater the reversion of MetS and its components [200].
3.2. Nutraceuticals
Recent research is assessing the role of nutraceuticals in the management of MetS. Several plant extracts, spices, herbs, and essential oil extracts have apparent benefit in the management of patients with MetS. Although some benefits have been documented, these agents are still under investigation and cannot be considered as an alternative for pharmacotherapy. However, the dependence on nutraceuticals—that are graciously available and with minimal complications—in the management of MetS may be a promising field in the development of novel therapies [201].
Table 5 illustrates the nutraceuticals that have been studied and have proven some benefits in MetS.

Table 5.
Role of nutraceuticals in MetS. ↑ signifies increase. ↓ signifies decrease.
3.3. Butyrate
Human gastrointestinal microbiota have been broadly explored because of their role in both maintaining gut health and disease causation. Dietary fiber and resistant starch are not completely hydrolyzed by host enzymes in the small intestine; instead, they are broken down by the large intestinal microbiota. The principal fermentation products resulting from the breakdown of these fibers are the short-chain fatty acids (SCFAs) acetate, propionate, and butyrate. SCFAs are an important fuel for intestinal epithelial cells, and the main energy source is butyrate, followed by propionate and then acetate (butyrate > propionate > acetate). In general, approximately 10% of the daily energy requirement in adults on a westernized diet originates from the colon-derived SCFAs.
Recently, butyrate has been reported to improve lipid profile, glycemia, body weight, composition, and insulin sensitivity in animal models of MetS. In vitro analyses tested the effect of butyrate on adipose tissue, intestinal cells, skeletal muscle, pancreatic islets, hepatocytes, and blood vessels, emphasizing the role of genes and pathways interplay in its beneficial effects.
A link between obesity and the components of the human microbiota has been described. Nevertheless, these conclusions depend on experimental studies completed using relatively small and distinct groups of volunteers or model animals. Moreover, microbiota components and nutrient intake regulate the total amount and relative quantity of butyrate [216].
A copious and phylogenetically distinct group of bacteria in the colon produce butyrate; the main species are Faecalibacterium prausnitzii, Eubacterium rectale, and Roseburia intestinalis. These bacteria are reduced in diabetic and pre-diabetic subjects [217]. Microbiota taken from lean subjects show increased comparative levels of bacteria that produce butyrate and after transferring them to subjects with MetS, the insulin sensitivity is improved [218].
Butyrate mechanisms of action are diverse, and several of these include epigenetic regulation of gene expression by inhibiting histone deacetylase [219].
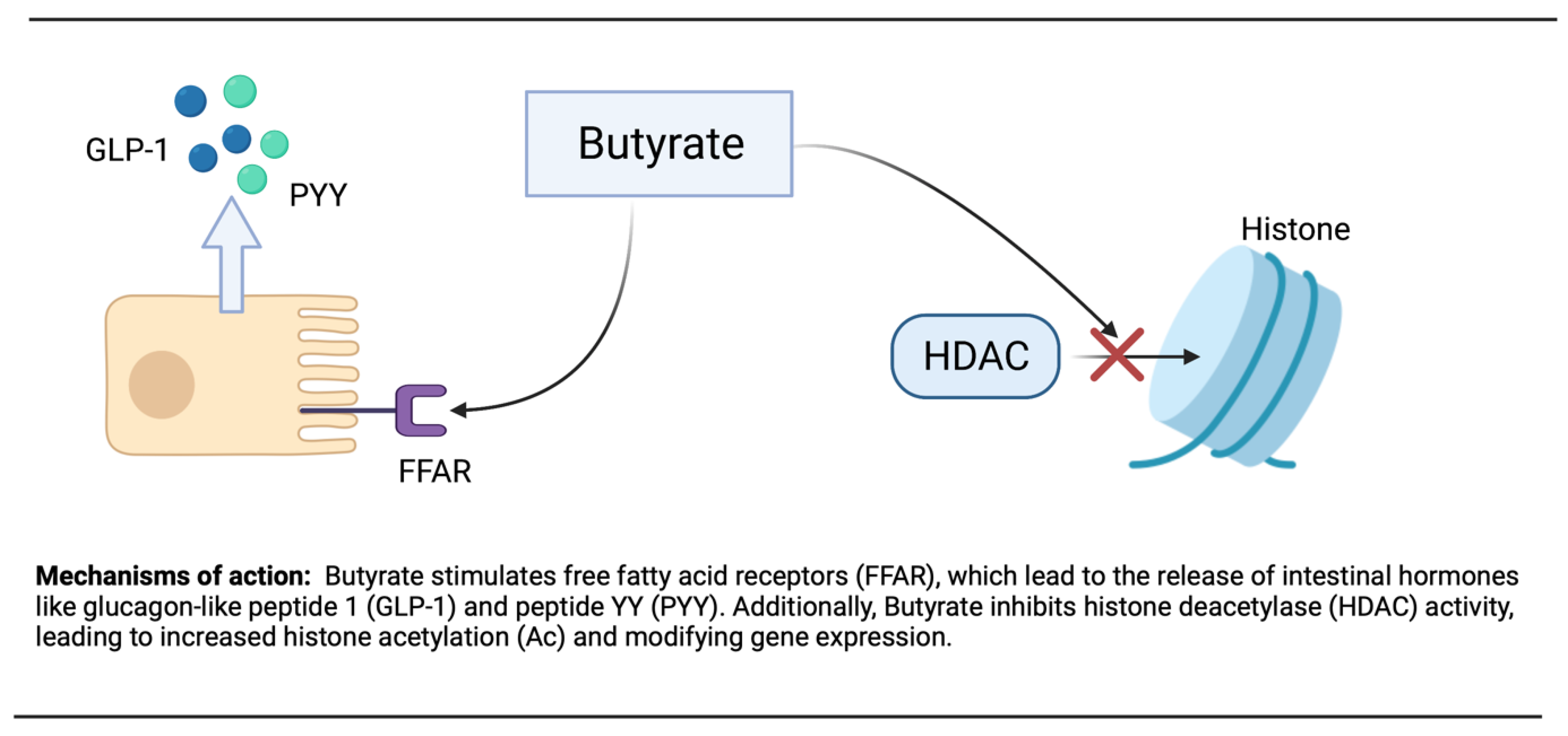
In epigenetics, histone N-terminal tail acetylation is believed to improve gene transcription. This adjustment of histone acetylation and deacetylation by environmental factors, such as dietary compounds, may decrease diseases and maintain good health. Additionally, the inhibition of histone deacetylase (HDAC) and the activation of AMPK by butyrate stimulate PGC-1α activity can occur [220]. By binding and activating the G protein-coupled free fatty acid receptors (FFAR), FFAR2 and FFAR3 in the intestinal enteroendocrine cells, butyrate is believed to play a major role in metabolism and appetite suppression [221].
Remarkably, butyrate supplementation or high levels of fibers in the diet, as well as increase gut bacteria, have been shown to prevent or attenuate obesity and insulin resistance [222]. By stimulating FFARs in the intestine, butyrate may reduce appetite and weight. The activation of FFARs stimulates the release of glucagon-like peptide 1 (GLP-1) and peptide YY (PYY) (Figure 2). GLP-1 will increase insulin secretion and inhibit glucagon secretion, whereas PYY will reduce appetite and slow gastric emptying [223]. Additionally, butyrate decreases the secretion of ghrelin, the hunger hormone. In animals given high-fat diets (HFD), butyrate supplementation decreased weight gain by reducing increases in adiposity. Matheus et al. [224] showed that butyrate did not modify weight, adiposity, glycemia, or insulinemia in mice on a non-HDF, whereas notably these paramters are ameliorated in mice on an HFD, indicating that butyrate improves the unfavorable effects of an HFD. The beneficial effects seen with butyrate supplementation can also be observed with fermentable fibers and resistant starches [225].
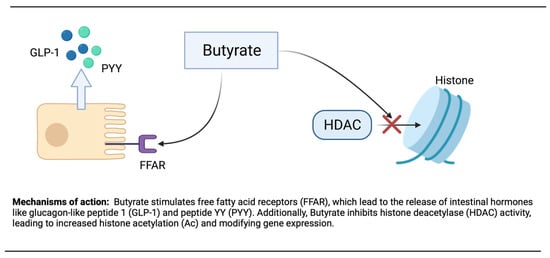
Figure 2.
Mechanism of action of butyrate.
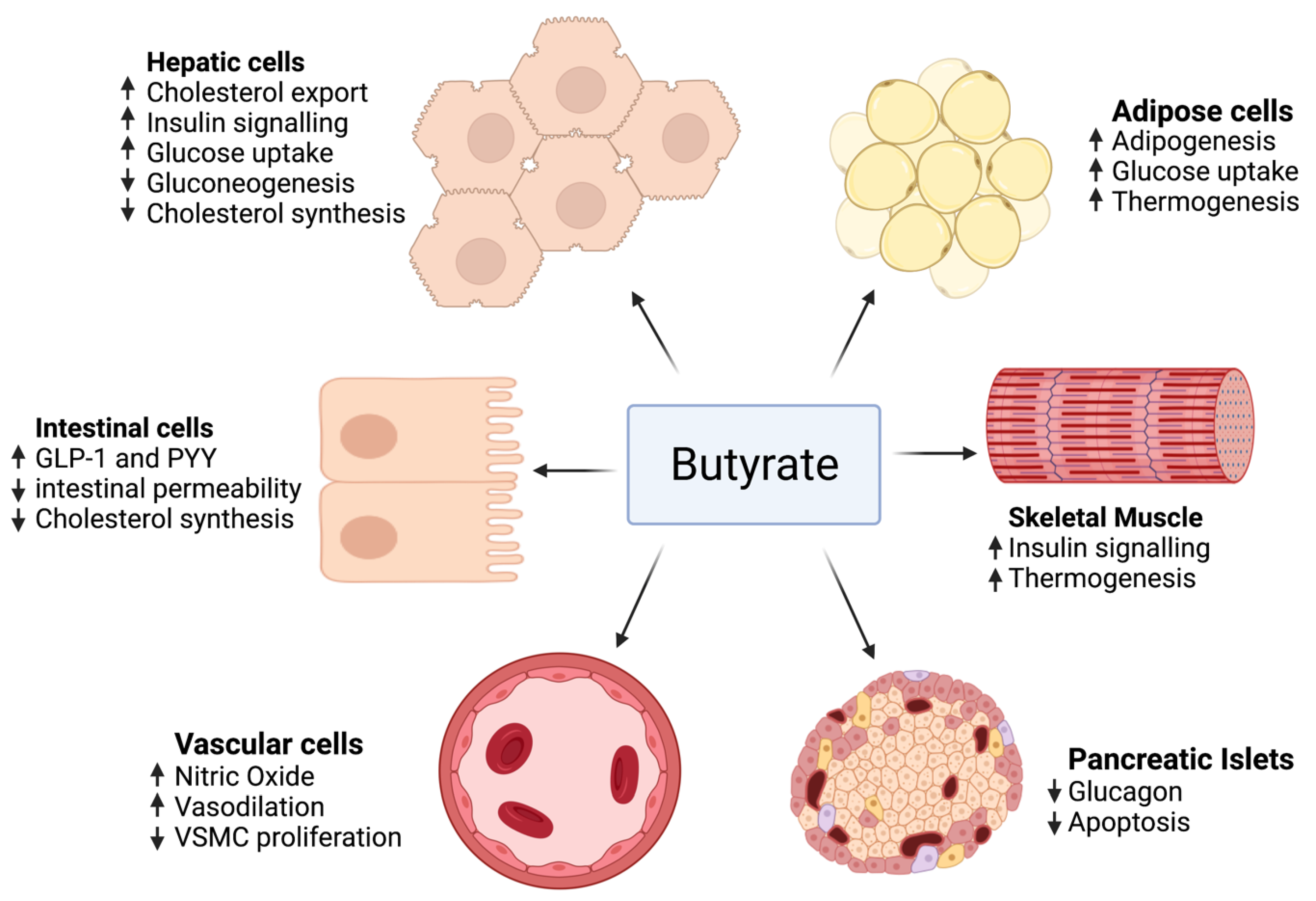
As mentioned previously, the impact of butyrate on intestinal cells, adipose tissue, skeletal muscle, blood vessels, hepatocytes, and pancreatic islets has been studied by accentuating the role of different genes and pathways.
The effect of butyrate on adipose tissue has been described in several studies, revealing that butyrate activates adipogenesis by several mechanisms, mainly by increasing the expression of genes that play major roles in adipocyte differentiation [226]. Butyrate markedly stimulated the expression of SREBP-1c’s mRNA, a major regulator for adipogenesis and fatty acid de novo synthesis. Moreover, the expression of the adipocyte’s differentiation markers, PPARγ, C/EBPα, and C/EBPβ, were significantly upregulated in adipocytes treated with butyrate compared to the control group. Increased adipocytes lead to more lipids being sequestered, thus more adipogenesis reduces circulating fatty acids and hence protects different organs from lipotoxicity and ensuing metabolic disorders such as insulin resistance [227].
As for cholesterol metabolism, several animal studies have revealed a decrease in total serum cholesterol level when given butyrate. This reduction in total serum cholesterol may be due to modifications in dietary cholesterol uptake, cholesterol import, and export from cells or cholesterol biosynthesis. These three processes are proposed in in vitro studies. In a study conducted by Marcil et al. [228], butyrate was found to reduce cholesterol synthesis in Caco-2 enterocytes, and this process is related to a decrease in the activity of 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase (HMGCR), a rate-limiting step in the cholesterol biosynthesis. Additionally, a study of Caco-2 cells described the role of butyrate in reducing the absorption of cholesterol by intestinal cells and decreasing the expression of a major gene in intestinal cholesterol absorption [229]. Moreover, the gene expression of ABCG5 and ABCG8, implicated in moving cholesterol from intestinal cells into the intestinal lumen and therefore away from the circulation, was enhanced. These outcomes were not seen in cells treated with propionate or acetate, signifying this is not a conventional effect of SCFAs.
In diabetic rats, Khan and Jena [230] observed lower HBA1c and plasma glucose with unchanged plasma insulin when given butyrate, thus showing that butyrate decreases insulin resistance. Moreover, butyrate may improve insulin sensitivity by promoting adipogenesis. This implies that butyrate can act in a similar way to thiazolidinediones, improving adipogenesis through PPARγ stimulation. An increase in expression of the glucose transporter GLUT4 and hence an increase glucose uptake was observed in porcine adipocytes treated with butyrate [226]. Finally, a study made by Chriett et al. indicated that butyrate increased histone acetylation near the IRS1 promoter, thus amplifying its expression [231].
Protecting the pancreatic β cell function is also a major role of butyrate. Sodium butyrate given to a rat model of type 1 diabetes decreased the destruction of pancreatic islets caused by streptozotocin (STZ) injection [232].
In terms of endothelial function, hypertension and vascular function were improved after the supplementation of butyrate in a human clinical trial [233,234]. Several mechanisms responsible for blood pressure reduction were hypothesized, including vasodilation, increased nitric oxide production, decreased proliferation of vascular smooth muscle cells, and the renin–angiotensin system.
Whether results from animal studies can apply to humans is open to question because of butyrate’s poor systemic availability and rapid clearance from the circulation. Studies have shown that the amount of butyrate in the gastrointestinal tract that reaches the peripheral circulation is minimal [233,234]. In fact, it is the main reason why many butyric prodrugs and derivatives have been developed. As an example, Pivanex (pivaloyloxymethyl butyrate, Prodrug AN9) has considerably higher cellular uptake than butyrate because of its lipophilicity and is active at lesser concentrations [235]. Nevertheless, several side effects were reported in a phase I trial involving nausea; vomiting; fatigue; visual disturbance; and, captivatingly, hyperglycemia [236].
However, a study on non-human primates has shown increase uptake in specific tissues such as the pancreas and the liver. Thus, oral administration of butyrate may yield positive effects on lipid and glucose metabolism by acting in the liver and pancreas [237].
For the moment, most human clinical trials have focused on the role of butyrate in intestinal disorders administered rectally or orally, and several trials showed improvement in diverticulitis [238], Crohn’s disease [239], ulcerative colitis [240], and travelers’ diarrhea [241]. No major side effects were described, and it was well tolerated. The unique problem was that the orally administered butyrate led to foul-smelling belching [240].
On the other hand, no sufficient human clinical trials studied butyrate benefits in MetS. Only two small trials were conducted. One was a randomized placebo-controlled clinical trial that studied the outcome of 100 mg sodium butyrate given six times daily for 45 days in type 2 diabetics [233], finding that there was no improvement in fasting insulin, fasting blood glucose, cholesterol, or triglyceride levels; however, an increase in GLP-1 and a reduction in diastolic blood pressure were observed. The second trial used a small sample size and was not placebo-controlled. It studied the effect of 4 g of oral sodium butyrate given daily for 4 weeks. The outcome was an improvement of hepatic and peripheral insulin sensitivity in lean males rather than obese males [242]. Figure 3 summarizes the role of butyrate in MetS.
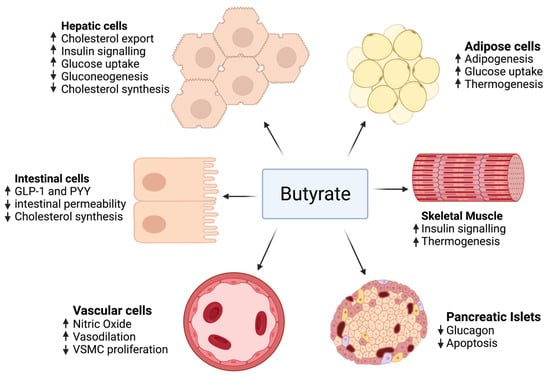
Figure 3.
The effect of butyrate on adipose tissue, intestinal cells, skeletal muscle, pancreatic islets, hepatocytes, and blood vessels.
3.4. Probiotics
Nowadays, there is evidence that gastrointestinal dysbiosis, which is the modification of the gastrointestinal microbiome, can participate in insulin resistance development [243]. Viewed in this way, many studies have revealed the undesirable effects of low or atypical gastrointestinal bacterial load on dyslipidemia, inflammatory markers, adiposity, and insulin resistance [244]. Additionally, one of the mechanisms was exposure to the bacterial LPS, which is the main cause of metabolic endotoxemia linked to MetS [245]. Moreover, studies have shown that compared to normal-weight people, obese individuals have a reduced ratio of Bacteroidetes/Firmicutes [246].
In terms of therapy, several studies have evaluated the beneficial effects of probiotics in obese and MetS patients (Table 6). Analysis of the data shows that the inclusion of probiotics to the treatment of patients with MetS may improve the current medical treatment but would not affect all of the clinical characteristics of the MetS and related inflammatory biomarkers. Although we cannot deduce that probiotics exert beneficial effects, they may have some positive outcomes that are marginal compared to drug therapy and a healthy lifestyle, and are likely dose- and strain-specific. In addition, the duration of the interventions may have not been long enough to prove the decrease in MetS associated complications. Accordingly, better RCTs were needed in humans with MetS able to fully enlighten if probiotics can be truly used as a coadjuvant therapy for this pathology.

Table 6.
Association between metabolic syndrome and probiotics.
Nine clinical trials are highlighted in the following section. Six of them were performed in both men and women, whereas three of them were performed in postmenopausal women. The dose used and the duration of the intervention varied from 108 cells/mL to 1.5 × 1011 colony-forming units (CFU)/g, and from 3 to 12 weeks, respectively. The main results for primary and secondary outcomes, specific strains used, and characteristics of the studies are described in the table below (Table 6) [245,246,247,248,249,250,251,252,253].
Interestingly, one specific strain (Lactobacillus reuteri V3401) has gained attention in the literature and has shown encouraging results in animal studies. Thus, a randomized, placebo-controlled, crossover study was conducted on 53 adults with MetS [255], and the participants were divided among two groups: one group received the probiotic Lactobacillus reuteri V3401 for 12 weeks concurrently with hypocaloric diet and physical activity and the other group received a placebo, also with a healthy lifestyle recommendation. No differences were observed regarding clinical characteristics of MetS in the group treated with Lactobacillus reutri V3401; however, changes in inflammatory biomarkers, anthropometric measurements, and microbiome composition were reported: reduction in IL-6 and VCAM-1 levels, improvement in anthropometric parameters, and increased proportion of Verrucomicrobia phylum [255].
Results regarding the utility of probiotics in the treatment of MetS have been contradictory. These conflicting results are probably due to the diverse doses, strains, and study designs [247]. Hence, more studies are needed to confirm the beneficial effects of probiotics and further explore their mechanism of action.
3.5. Coconut Oil Effect on Asymmetric Dimethylarginine (ADMA)
Diet has been shown to be one of the most crucial factors that contribute to MetS. Additionally, dietary fats are well known to affect lipoprotein and triglyceride levels [256]. Therefore, many studies have evaluated the effect of dietary fat on MetS. Some of them suggested that reducing dietary fat had a beneficial effect [257]. However, several other studies indicated that the type of fats that an individual consumes is more essential than the quantity of fat for the treatment of MetS [258].
Saturated and trans fatty acids are generally considered to have negative effects on cardiovascular risk, and recent studies have advised reducing their consumption to no more than 10% of total calories daily [259]. Conversely, medium-chain fatty acid (MCFAs) and SCFA consumption showed a beneficial effect on MetS [260].
Lately, several studies have evaluated the beneficial effect of coconut oil as an antithrombotic, anti-inflammatory, and hypolipidemic agent [261]. Coconut oil has an elevated quantity of MCFAs—it is made of 8% caprylic acid, 7% capric acid, 8% myristic acid, 2% stearic acid, 6% oleic acid, 8% palmitic acid, 2% linoleic acid, and 49% lauric acid [262]. Although 85% of coconut oil is composed of saturated fatty acid (SFA), the major SFA, lauric acid, has been shown to have positive health outcomes in CVD risk profile and body composition when compared with other SFAs such as palmitic acid, which exists mainly in palm oil, butter, and animal fat [263]. Additionally, when compared with refined coconut oil, virgin coconut oil (VCO) has a more antioxidant effect because of the high proportion of phenolic content. Consequently, by decreasing oxidative stress, it has further benefits in preventing cardiovascular dysfunction [261].
ADMA, which is a product of methylated protein metabolism in the body, plays a major role in endothelial dysfunction (ED) by inhibiting NO synthase [264]. Many consider endothelial dysfunction (ED) as part of the MetS because it is an early stage of atherosclerosis [265]. Moreover, elevated serum levels of ADMA in MetS have been described [265,266]. Consequently, ADMA can be a target to prevent adverse cardiovascular outcomes in MetS. Several studies have shown that consumption of high-fat meals is associated with an increased level of ADMA [266]. Yet, little is known about the effect of the composition of dietary fat on ADMA blood levels.
The latest review emphasized on the need for more studies on the benefits of VCO on dyslipidemia and CVDs risk factors. Thus, in a study conducted by Parinaz Nikooei et al., 30 mL of VCO was given for 4 weeks, replacing the daily used oil. Its effect on MetS components as well as on serum ADMA levels were investigated and showed that consuming 30 mL of VCO for 4 weeks instead of the habitual daily oil decreased serum fasting blood glucose and triglyceride and increased HDL-C, total cholesterol, LDL-C, and ADMA. However, the ratio of LDL-C/HDL-C did not change. Additionally, there were no beneficial effects on waist circumference and blood pressure. Further studies are needed to cautiously recommend VCO, specifically in people at risk for CVDs [267].
3.6. Curcumin
Curcumin, a polyphenolic compound, is an orange-yellow pigment that is derived from turmeric [268]. Its beneficial effects have been studied widely. It has an anti-oxidative effect, as well as anti-inflammatory properties, and increases nitric oxide production [269]. These effects may help improve arterial stiffness caused by inflammation and increased luminal pressure. A study conducted by Alidadi et al. revealed that consuming 500 mg of curcumin every day for 12 weeks has beneficial effects on arterial stiffness and can control the weight of individuals with MetS [270]. After 12 weeks of curcumin administration, there was a statistically significant decrease in mean body weight in the control compared to the placebo group; however, body composition and other anthropometric parameters revealed no major changes. Curcumin supplementation showed no effect on vascular stiffness and hemodynamic parameters at the baseline of the study. However, after 12 weeks of curcumin supplementation, a significant decrease in PWV was observed, with amelioration of aortic stiffness in MetS patients. Even after adjusting for confounding factors, including gender, age, change in physical activity, and energy intake by regression, the daily consumption of curcumin significantly lowered aortic PWV, relative to placebo [270]. Moreover, in terms of diabetes and dyslipidemia, fasting blood glucose did not improve in the group treated with curcumin, and lipid profiles remained unchanged. However, preclinical studies have shown that curcumin reduces serum cholesterol levels by upregulation of hepatic LDL receptor expression and inhibition of LDL oxidation [271]. Gene expression involved in cholesterol biosynthesis is also affected by curcumin, as well as cholesterol excretion by increasing bile acid secretion [271]. In addition, it is believed that curcumin contributes to the downregulation of major lipogenesis and adipogenesis transcription factors such as PPARγ [272]. Thus, to explain the discrepancy in results, it has been postulated that to affect lipid profiles and fasting plasma glucose in individuals with MetS, a higher dose or higher efficacy form of curcumin should be taken [273].
Preliminary evidence in a preclinical analysis completed by Fleenor et al. proved that dietary curcumin supplementation reduces age-related large elastic artery stiffness by restoration of nitric oxide bioavailability, reduction of oxidative stress, and regulation of collagen I and advanced glycation end products buildup in the arterial wall [274].
3.7. Statins
The role of the proinflammatory and prothrombotic state in MetS has been investigated by several studies. Statins have been proven to reduce high-sensitivity C-reactive protein (hs-CRP) blood levels, confirming their anti-inflammatory effect.
In a study conducted by Gladys P. Velarde et al., women with MetS were administered high-intensity atorvastatin for 3 months, and several markers were reviewed, such as lipogenesis, inflammation, and thrombogenesis [275]. Of note, women in the study were not affected by cardiovascular disease or diabetes and had minor 10 year ASCVD risk. Results showed that atorvastatin led to the reduction of total cholesterol, LDL-C, and Apo-B levels as well as the Apo-A1/Apo-B ratio. However, no effect was shown on Apo-A1, human leptin levels, and lipogenic markers. Compared to placebo, there was no meaningful impact of atorvastatin on myeloperoxidase and plasminogen activator inhibitor-1 levels, and no major effect on fasting blood glucose (FBG) and microalbumin level. Moreover, no conclusion was made regarding platelet aggregation in response to adenosine diphosphate and collagen by treatment.
Adding up to the expected effect of atorvastatin on lipid profile, a significant reduction in Apo-B levels and Apo-B/Apo-A1 ratio and increase VCAM-1 levels was detected. No significant effect on Apo-A1, human leptin, hs-CRP, ICAM-1 was noticed. However, CRP levels were monitored in a different study and appeared to be decreased after statin therapy, independently of LDL cholesterol [276,277].
Interestingly, in a study conducted by de Oliveira et al. in 2014, simvastatin alone, pioglitazone alone, as well as their combination were shown to decrease the epicardial fat and the inflammatory markers in patients with coronary artery disease and MetS [278].
3.8. Anti-Hyperglycemic Agents
In addition to pioglitazone, mentioned above, dipeptidyl peptidase-4 (DPP-4) inhibitors have also been studied in patients with MetS. Multiple studies have suggested that the DPP-4/incretins axis has an influence on the cardiovascular system [279,280]. In addition, DPP-4 expression was shown to positively correlate with the extent of central obesity, visceral adiposity, and inflammation [281,282]. Sitagliptin, one of the DPP-4 inhibitors, has been shown to decrease MetS, obesity-induced adipose tissue inflammation, and fatty liver by regulating the adenosine monophosphate-activated protein kinase and adiponectin levels in obese mice [283]. Moreover, sitagliptin has been shown to have a positive influence on hyperglycemia-induced vascular changes in an endothelium-dependent manner [284]. Finally, sitagliptin has been shown to decrease visceral adiposity as well as the maximal response to oral glucose tolerance test in women with polycystic ovarian syndrome. Glucagon-like peptide 1 (GLP-1) receptor agonists have become popular and more widely used in the past couple of years. Rizzo et al. recently highlighted the importance of utilizing GLP-1 receptor agonist and their effect on various cardiometabolic markers and overall cardiovascular risk [285]. Even though the exact mechanisms remain largely unknown, the favorable effects of liraglutide, a GLP-1 receptor agonist, appear to be driven by direct anti-atherosclerotic action and by decreasing plaque formation and progression [286].
The Diabetes Prevention Program (DPP) addresses in the post hoc analyses of their clinical trial the possibility of modifying MetS prevalence estimates after treatment with either lifestyle (exercise-induced weight loss + diet) or metformin [287]. They reported the incidence of new MetS cases and the resolution of existing MetS cases in treatment groups compared with placebo in participants (n = 3234) of the DPP trial after 3.2 years of follow-up. However, since the impaired glucose tolerance (IGT) was a primary inclusion criterion in the trial, 53% of the participants met the criteria of MetS at baseline. Compared to placebo, the incidence of MetS was decreased by 41% in the lifestyle group and by 17% in the metformin group [287]. Among those who fulfilled MetS criteria at baseline, prevalence at 3 years varied significantly by treatment group: 38% of the lifestyle group, 23% of the metformin group, and 18% of the placebo group no longer had the syndrome [287]. Whether these effects apply to non-IGT population remains unknown. Several efforts have been put in pace to answer this question [288].
4. Conclusions
A cluster of components or risk factors are associated with an increased risk of CVDs and T2DM, and MetS has a prevalence that has been on the rise across all ages. While it is not clear if MetS can be treated in and of itself, ongoing research seems to be unlocking different targets along the disease’s pathway. Concentrating therapeutic efforts on treating the excess adiposity and insulin resistance associated with the MetS may provide the most overall success in attaining these goals. Much has been answered in the past few years, but much remains to be unraveled. The associations of MetS with other comorbidities such as NAFLD, sleep disorders, reproductive tract disorders, and microvascular disease are examples of areas yet to be fully determined.
Author Contributions
Conceptualization, M.B.Z. (Maroun Bou Zerdan) and Y.B. resources, H.I.A.; data curation, S.A. writing—original draft preparation, all authors; writing—review and editing, all authors; visualization, L.A. and G.F.; supervision, H.I.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ACLY | ATP-citrate lyase |
| ADMA | asymmetric dimethylarginine |
| AHA | American Heart Association |
| AHSG | α2-Heremans–Schmid glycoprotein |
| ALT | alanine aminotransferase |
| AMPK | AMP-activated protein kinase |
| Ang II | angiotensin II |
| Apo-A1 | apolipoprotein A1 |
| Apo-B | apolipoprotein B |
| ASCVD | atherosclerotic cardiovascular disease |
| AST | aspartate aminotransferase |
| AUC | area under the curve |
| BMI | body mass index |
| CDC | Center of Disease Control and Prevention |
| CETP | cholesterol ester transfer protein |
| c-miRNAs | circulatory microRNAs |
| CRP | C-reactive protein |
| DAG | diacylglycerol |
| DAMPs | damage-associated molecular patterns |
| ED | endothelial dysfunction |
| ERRs | estrogen-related receptors |
| ETC | electron transport chain |
| FA | fatty acids |
| FADH2 | reduced flavin adenine dinucleotide |
| FFAs | free fatty acids |
| FFAR | free fatty acid receptors |
| FTO | fat mass and obesity-associated |
| GDF15 | growth differentiation factor 15 |
| GPCRs | G protein-coupled receptors |
| GLUT4 | glucose transporter |
| GLP-1 | glucagon-like peptide 1 |
| HDAC | histone deacetylase |
| HDL | high-density lipoprotein |
| HFD | high-fat diets |
| HMG-CoA | 3-hydroxy-3-methyl-glutaryl-coenzyme A |
| HMGCR | HMG-CoA reductase |
| HOMA-IR | homeostatic model assessment-insulin resistance |
| hs-CRP | high-sensitivity C-reactive protein |
| ICAM-1 | intercellular adhesion molecule 1 |
| IDEFICS | Identification and Prevention of Dietary- and Lifestyle-Induced Health Effects in Children and Infants |
| IGT | impaired glucose tolerance |
| IL-6 | interleukin 6 |
| IP3 | inositol 1,4,5-triphosphate |
| IRS-1 | insulin receptor substrate-1 |
| KO | knockout |
| LDL-C | low-density lipoprotein |
| LAP | latency-associated protein |
| LIKRO | liver-specific insulin receptor knockout |
| LOX-1 | lipoprotein receptor-1 |
| LPS | lipopolysaccharides |
| MCP-1 | monocyte chemoattractant protein-1 |
| MD | Mediterranean diet |
| MetS | metabolic syndrome |
| MCFAs | medium-chain fatty acids |
| miRNAs | microRNAs |
| NHANES | National Health and Nutrition Examination Survey |
| NADH | nicotinamide adenine dinucleotide |
| NADPH oxidase | nicotinamide adenine dinucleotide phosphate oxidase |
| NAFLD | non-alcoholic fatty liver disease |
| NF-kB | nuclear factor kappa-light-chain enhancer of activated B cells |
| NO | nitric oxide |
| NREP | neuronal regeneration–related protein |
| NRF-1 and NRF-2 | nuclear respiratory factors 1 and 2 |
| PAI-1 | plasminogen activator inhibitor 1 |
| PAMPs | pathogen-associated molecular patterns |
| PGC-1α | peroxisome proliferator-activated receptor-γ coactivator 1α |
| PI3K | phosphoinositide 3-kinase |
| PPARα and PPARδ | peroxisome proliferator-activated receptor α and δ |
| PPARγ | peroxisome proliferator-activated receptor-γ |
| PPY | peptide YY |
| PWV | pulse wave velocity |
| QUICKI | quantitative insulin sensitivity check index |
| RAS | renin–angiotensinogen system |
| ROS | reactive oxygen species |
| SCFAs | short-chain fatty acids |
| Sd-LDL | small dense LDL |
| SFA | saturated fatty acid |
| SNPs | single nucleotide polymorphisms |
| SREBP-1 | sterol regulatory element-binding protein 1 |
| STZ | streptozotocin |
| T2DM | type II diabetes mellitus |
| TFAM | mitochondrial transcription factor A |
| TFB2M | mitochondrial transcription factor B2 |
| TGs | triglycerides |
| Th1 | T helper type 1 |
| TNF α | tumor necrosis factor α |
| TLRs | Toll-like receptors |
| VCAM-1 | vascular cell adhesion molecule 1 |
| VLDLs | very low-density lipoproteins |
| VSMCs | vascular smooth muscle cells |
| VCO | virgin coconut oil |
| WBCs | white blood cells |
References
- Reaven, G.M. Banting Lecture 1988. Role of insulin resistance in human disease. Nutrition 1997, 13, 65. [Google Scholar] [CrossRef]
- Cheng, T.O. Cardiac syndrome X versus metabolic syndrome X. Int. J. Cardiol. 2006, 119, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [PubMed]
- Saklayen, M.G. The global epidemic of the metabolic syndrome. Curr. Hypertens. Rep. 2018, 20, 1–8. [Google Scholar] [CrossRef]
- Eckel, R.H.; Alberti, K.G.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2010, 375, 181–183. [Google Scholar] [CrossRef]
- McCracken, E.; Monaghan, M.; Sreenivasan, S. Pathophysiology of the metabolic syndrome. Clin. Dermatol. 2018, 36, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (adult treatment panel III). JAMA 2001, 285, 2486–2497. [Google Scholar] [CrossRef]
- Balkau, B.; Charles, M.-A. Comment on the provisional report from the WHO consultation. European Group for the Study of Insulin Resistance (EGIR). Diabet. Med. A J. Br. Diabet. Assoc. 1999, 16, 442–443. [Google Scholar]
- Ross, R.; Neeland, I.J.; Yamashita, S.; Shai, I.; Seidell, J.; Magni, P.; Santos, R.D.; Arsenault, B.; Cuevas, A.; Hu, F.B.; et al. Waist circumference as a vital sign in clinical practice: A Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nat. Rev. Endocrinol. 2020, 16, 177–189. [Google Scholar] [CrossRef]
- Neeland, I.J.; Poirier, P.; Després, J.-P. Cardiovascular and metabolic heterogeneity of obesity: Clinical challenges and implications for management. Circulation 2018, 137, 1391–1406. [Google Scholar] [CrossRef]
- Neeland, I.J.; Ross, R.; Després, J.-P.; Matsuzawa, Y.; Yamashita, S.; Shai, I.; Seidell, J.; Magni, P.; Santos, R.D.; Arsenault, B. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: A position statement. Lancet Diabetes Endocrinol. 2019, 7, 715–725. [Google Scholar] [CrossRef]
- Nauli, A.M.; Matin, S. Why do men accumulate abdominal visceral fat? Front. Physiol. 2019, 10, 1486. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fox, C.S.; Hickson, D.; Bidulescu, A.; Carr, J.J.; Taylor, H.A. Fatty liver, abdominal visceral fat, and cardiometabolic risk factors: The Jackson Heart Study. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 2715–2722. [Google Scholar] [CrossRef]
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.-C.; James, W.P.T.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the metabolic syndrome: A joint interim statement of the international diabetes federation task force on epidemiology and prevention; national heart, lung, and blood institute; American heart association; world heart federation; international atherosclerosis society; and international association for the study of obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [PubMed]
- Ashwell, M.; Gunn, P.; Gibson, S. Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: Systematic review and meta-analysis. Obes. Rev. 2012, 13, 275–286. [Google Scholar] [CrossRef]
- Moore, J.X.; Chaudhary, N.; Akinyemiju, T. Peer reviewed: Metabolic syndrome prevalence by race/ethnicity and sex in the United States, National Health and Nutrition Examination Survey, 1988–2012. Prev. Chronic Dis. 2017, 14, E24. [Google Scholar] [CrossRef]
- Swarup, S.; Goyal, A.; Grigorova, Y.; Zeltser, R. Metabolic Syndrome; StatPearls: Treasure Island, FL, USA, 2020. [Google Scholar]
- Fathi Dizaji, B. The investigations of genetic determinants of the metabolic syndrome. Diabetes Metab. Syndr. 2018, 12, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Matsuzawa, Y.; Funahashi, T.; Nakamura, T. The concept of metabolic syndrome: Contribution of visceral fat accumulation and its molecular mechanism. J. Atheroscler. Thromb. 2011, 18, 629–639. [Google Scholar] [CrossRef]
- Pekgor, S.; Duran, C.; Berberoglu, U.; Eryilmaz, M.A. The Role of Visceral Adiposity Index Levels in Predicting the Presence of Metabolic Syndrome and Insulin Resistance in Overweight and Obese Patients. Metab. Syndr. Relat. Disord. 2019, 17, 296–302. [Google Scholar] [CrossRef]
- Boden, G.; Shulman, G.I. Free fatty acids in obesity and type 2 diabetes: Defining their role in the development of insulin resistance and beta-cell dysfunction. Eur. J. Clin. Investig. 2002, 32, 14–23. [Google Scholar] [CrossRef]
- Griffin, M.E.; Marcucci, M.J.; Cline, G.W.; Bell, K.; Barucci, N.; Lee, D.; Goodyear, L.J.; Kraegen, E.W.; White, M.F.; Shulman, G.I. Free fatty acid-induced insulin resistance is associated with activation of protein kinase C theta and alterations in the insulin signaling cascade. Diabetes 1999, 48, 1270–1274. [Google Scholar] [CrossRef] [PubMed]
- Unger, R.H.; Zhou, Y.T. Lipotoxicity of beta-cells in obesity and in other causes of fatty acid spillover. Diabetes 2001, 50 (Suppl. 1), S118–S121. [Google Scholar] [CrossRef]
- Patel, P.; Abate, N. Body fat distribution and insulin resistance. Nutrients 2013, 5, 2019–2027. [Google Scholar] [CrossRef]
- Murakami, T.; Michelagnoli, S.; Longhi, R.; Gianfranceschi, G.; Pazzucconi, F.; Calabresi, L.; Sirtori, C.R.; Franceschini, G. Triglycerides are major determinants of cholesterol esterification/transfer and HDL remodeling in human plasma. Arter. Thromb. Vasc. Biol. 1995, 15, 1819–1828. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, S.; Gavish, D.; Oschry, Y.; Fainaru, M.; Deckelbaum, R.J. Abnormalities in very low, low and high density lipoproteins in hypertriglyceridemia. Reversal toward normal with bezafibrate treatment. J. Clin. Investig. 1984, 74, 470–482. [Google Scholar] [CrossRef] [PubMed]
- Tripathy, D.; Mohanty, P.; Dhindsa, S.; Syed, T.; Ghanim, H.; Aljada, A.; Dandona, P. Elevation of free fatty acids induces inflammation and impairs vascular reactivity in healthy subjects. Diabetes 2003, 52, 2882–2887. [Google Scholar] [CrossRef]
- Esler, M.; Rumantir, M.; Wiesner, G.; Kaye, D.; Hastings, J.; Lambert, G. Sympathetic nervous system and insulin resistance: From obesity to diabetes. Am. J. Hypertens. 2001, 14, 304S–309S. [Google Scholar] [CrossRef]
- Juhan-Vague, I.; Alessi, M.C.; Mavri, A.; Morange, P.E. Plasminogen activator inhibitor-1, inflammation, obesity, insulin resistance and vascular risk. J. Thromb. Haemost. 2003, 1, 1575–1579. [Google Scholar] [CrossRef]
- Duncan, M.; Singh, B.; Wise, P.H.; Carter, G.; Alaghband-Zadeh, J. A simple measure of insulin resistance. Lancet (Lond. Engl.) 1995, 346, 120–121. [Google Scholar] [CrossRef]
- Hřebíček, J.i.; Janout, V.r.; Malinčíková, J.; Horáková, D.; Čízžek, L.k. Detection of insulin resistance by simple quantitative insulin sensitivity check index QUICKI for epidemiological assessment and prevention. J. Clin. Endocrinol. Metab. 2002, 87, 144–147. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.; Rudenski, A.; Naylor, B.; Treacher, D.; Turner, R. Homeostasis model assessment: Insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Mondal, N.; Singh, V.; Saxena, V. Determining the interaction between groundwater and saline water through groundwater major ions chemistry. J. Hydrol. 2010, 388, 100–111. [Google Scholar] [CrossRef]
- Motamed, N.; Miresmail, S.J.H.; Rabiee, B.; Keyvani, H.; Farahani, B.; Maadi, M.; Zamani, F. Optimal cutoff points for HOMA-IR and QUICKI in the diagnosis of metabolic syndrome and non-alcoholic fatty liver disease: A population based study. J. Diabetes Complicat. 2016, 30, 269–274. [Google Scholar] [CrossRef]
- Mohamed-Ali, V.; Pinkney, J.H.; Coppack, S.W. Adipose tissue as an endocrine and paracrine organ. Int. J. Obes. Relat. Metab. Disord. 1998, 22, 1145–1158. [Google Scholar] [CrossRef]
- Trayhurn, P.; Wood, I.S. Adipokines: Inflammation and the pleiotropic role of white adipose tissue. Br. J. Nutr. 2004, 92, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L.; et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef]
- Berglund, E.D.; Vianna, C.R.; Donato, J., Jr.; Kim, M.H.; Chuang, J.C.; Lee, C.E.; Lauzon, D.A.; Lin, P.; Brule, L.J.; Scott, M.M.; et al. Direct leptin action on POMC neurons regulates glucose homeostasis and hepatic insulin sensitivity in mice. J. Clin. Investig. 2012, 122, 1000–1009. [Google Scholar] [CrossRef] [PubMed]
- Obradovic, M.; Sudar-Milovanovic, E.; Soskic, S.; Essack, M.; Arya, S.; Stewart, A.J.; Gojobori, T.; Isenovic, E.R. Leptin and Obesity: Role and Clinical Implication. Front. Endocrinol. (Lausanne) 2021, 12, 585887. [Google Scholar] [CrossRef] [PubMed]
- Lord, G.M.; Matarese, G.; Howard, J.K.; Baker, R.J.; Bloom, S.R.; Lechler, R.I. Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature 1998, 394, 897–901. [Google Scholar] [CrossRef]
- Patel, S.B.; Reams, G.P.; Spear, R.M.; Freeman, R.H.; Villarreal, D. Leptin: Linking obesity, the metabolic syndrome, and cardiovascular disease. Curr. Hypertens. Rep. 2008, 10, 131–137. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Waki, H.; Imai, Y.; Shimozawa, N.; Hioki, K.; Uchida, S.; Ito, Y.; Takakuwa, K.; Matsui, J.; et al. Globular adiponectin protected ob/ob mice from diabetes and ApoE-deficient mice from atherosclerosis. J. Biol. Chem. 2003, 278, 2461–2468. [Google Scholar] [CrossRef] [PubMed]
- Esteve, E.; Ricart, W.; Fernandez-Real, J.M. Adipocytokines and insulin resistance: The possible role of lipocalin-2, retinol binding protein-4, and adiponectin. Diabetes Care 2009, 32, S362–S367. [Google Scholar] [CrossRef] [PubMed]
- Wulster-Radcliffe, M.C.; Ajuwon, K.M.; Wang, J.; Christian, J.A.; Spurlock, M.E. Adiponectin differentially regulates cytokines in porcine macrophages. BioChem Biophys. Res. Commun. 2004, 316, 924–929. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Waki, H.; Terauchi, Y.; Kubota, N.; Hara, K.; Mori, Y.; Ide, T.; Murakami, K.; Tsuboyama-Kasaoka, N.; et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 2001, 7, 941–946. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Kihara, S.; Arita, Y.; Nishida, M.; Matsuyama, A.; Okamoto, Y.; Ishigami, M.; Kuriyama, H.; Kishida, K.; Nishizawa, H.; et al. Adipocyte-derived plasma protein, adiponectin, suppresses lipid accumulation and class A scavenger receptor expression in human monocyte-derived macrophages. Circulation 2001, 103, 1057–1063. [Google Scholar] [CrossRef]
- Ouchi, N.; Kihara, S.; Arita, Y.; Maeda, K.; Kuriyama, H.; Okamoto, Y.; Hotta, K.; Nishida, M.; Takahashi, M.; Nakamura, T.; et al. Novel modulator for endothelial adhesion molecules: Adipocyte-derived plasma protein adiponectin. Circulation 1999, 100, 2473–2476. [Google Scholar] [CrossRef]
- Pischon, T.; Girman, C.J.; Hotamisligil, G.S.; Rifai, N.; Hu, F.B.; Rimm, E.B. Plasma adiponectin levels and risk of myocardial infarction in men. JAMA 2004, 291, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Hotta, K.; Funahashi, T.; Arita, Y.; Takahashi, M.; Matsuda, M.; Okamoto, Y.; Iwahashi, H.; Kuriyama, H.; Ouchi, N.; Maeda, K.; et al. Plasma concentrations of a novel, adipose-specific protein, adiponectin, in type 2 diabetic patients. Arter. Thromb. Vasc. Biol. 2000, 20, 1595–1599. [Google Scholar] [CrossRef]
- Chow, W.S.; Cheung, B.M.; Tso, A.W.; Xu, A.; Wat, N.M.; Fong, C.H.; Ong, L.H.; Tam, S.; Tan, K.C.; Janus, E.D.; et al. Hypoadiponectinemia as a predictor for the development of hypertension: A 5-year prospective study. Hypertension 2007, 49, 1455–1461. [Google Scholar] [CrossRef]
- Kondo, H.; Shimomura, I.; Matsukawa, Y.; Kumada, M.; Takahashi, M.; Matsuda, M.; Ouchi, N.; Kihara, S.; Kawamoto, T.; Sumitsuji, S.; et al. Association of adiponectin mutation with type 2 diabetes: A candidate gene for the insulin resistance syndrome. Diabetes 2002, 51, 2325–2328. [Google Scholar] [CrossRef]
- Buechler, C.; Feder, S.; Haberl, E.M.; Aslanidis, C. Chemerin Isoforms and Activity in Obesity. Int. J. Mol. Sci. 2019, 20, 1128. [Google Scholar] [CrossRef] [PubMed]
- Helfer, G.; Wu, Q.F. Chemerin: A multifaceted adipokine involved in metabolic disorders. J. Endocrinol. 2018, 238, R79–R94. [Google Scholar] [CrossRef]
- Jialal, I.; Devaraj, S.; Kaur, H.; Adams-Huet, B.; Bremer, A.A. Increased chemerin and decreased omentin-1 in both adipose tissue and plasma in nascent metabolic syndrome. J. Clin. Endocrinol. Metab. 2013, 98, E514–E517. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yuan, G.Y.; Wang, X.Z.; Jia, J.; Di, L.L.; Yang, L.; Chen, X.; Qian, F.F.; Chen, J.J. Plasma chemerin level in metabolic syndrome. Genet. Mol. Res. 2013, 12, 5986–5991. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Ji, W.; Zhang, Y. Elevated serum chemerin levels are associated with the presence of coronary artery disease in patients with metabolic syndrome. Intern. Med. 2011, 50, 1093–1097. [Google Scholar] [CrossRef]
- Bozaoglu, K.; Bolton, K.; McMillan, J.; Zimmet, P.; Jowett, J.; Collier, G.; Walder, K.; Segal, D. Chemerin is a novel adipokine associated with obesity and metabolic syndrome. Endocrinology 2007, 148, 4687–4694. [Google Scholar] [CrossRef]
- Chu, S.H.; Lee, M.K.; Ahn, K.Y.; Im, J.A.; Park, M.S.; Lee, D.C.; Jeon, J.Y.; Lee, J.W. Chemerin and adiponectin contribute reciprocally to metabolic syndrome. PLoS ONE 2012, 7, e34710. [Google Scholar] [CrossRef]
- Shafer-Eggleton, J.; Adams-Huet, B.; Jialal, I. Chemerin Ratios to HDL-cholesterol and Adiponectin as Biomarkers of Metabolic Syndrome. Endocr. Res. 2020, 45, 241–245. [Google Scholar] [CrossRef]
- Saiki, A.; Ohira, M.; Endo, K.; Koide, N.; Oyama, T.; Murano, T.; Watanabe, H.; Miyashita, Y.; Shirai, K. Circulating angiotensin II is associated with body fat accumulation and insulin resistance in obese subjects with type 2 diabetes mellitus. Metabolism 2009, 58, 708–713. [Google Scholar] [CrossRef]
- Rajagopalan, S.; Kurz, S.; Munzel, T.; Tarpey, M.; Freeman, B.A.; Griendling, K.K.; Harrison, D.G. Angiotensin II-mediated hypertension in the rat increases vascular superoxide production via membrane NADH/NADPH oxidase activation. Contribution to alterations of vasomotor tone. J. Clin. Investig. 1996, 97, 1916–1923. [Google Scholar] [CrossRef]
- Lassegue, B.; Sorescu, D.; Szocs, K.; Yin, Q.; Akers, M.; Zhang, Y.; Grant, S.L.; Lambeth, J.D.; Griendling, K.K. Novel gp91(phox) homologues in vascular smooth muscle cells: Nox1 mediates angiotensin II-induced superoxide formation and redox-sensitive signaling pathways. Circ. Res. 2001, 88, 888–894. [Google Scholar] [CrossRef]
- Li, D.Y.; Zhang, Y.C.; Philips, M.I.; Sawamura, T.; Mehta, J.L. Upregulation of endothelial receptor for oxidized low-density lipoprotein (LOX-1) in cultured human coronary artery endothelial cells by angiotensin II type 1 receptor activation. Circ. Res. 1999, 84, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Zafari, A.M.; Ushio-Fukai, M.; Akers, M.; Yin, Q.; Shah, A.; Harrison, D.G.; Taylor, W.R.; Griendling, K.K. Role of NADH/NADPH oxidase-derived H2O2 in angiotensin II-induced vascular hypertrophy. Hypertension 1998, 32, 488–495. [Google Scholar] [CrossRef]
- Mehta, P.K.; Griendling, K.K. Angiotensin II cell signaling: Physiological and pathological effects in the cardiovascular system. Am. J. Physiol. Cell Physiol. 2007, 292, C82–C97. [Google Scholar] [CrossRef]
- Kopp, H.P.; Kopp, C.W.; Festa, A.; Krzyzanowska, K.; Kriwanek, S.; Minar, E.; Roka, R.; Schernthaner, G. Impact of weight loss on inflammatory proteins and their association with the insulin resistance syndrome in morbidly obese patients. Arter. Thromb. Vasc. Biol. 2003, 23, 1042–1047. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Kanemaki, T.; Kitade, H.; Kaibori, M.; Sakitani, K.; Hiramatsu, Y.; Kamiyama, Y.; Ito, S.; Okumura, T. Interleukin 1beta and interleukin 6, but not tumor necrosis factor alpha, inhibit insulin-stimulated glycogen synthesis in rat hepatocytes. Hepatology 1998, 27, 1296–1303. [Google Scholar] [CrossRef]
- Ridker, P.M.; Hennekens, C.H.; Buring, J.E.; Rifai, N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N. Engl. J. Med. 2000, 342, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Burstein, S.A.; Peng, J.; Friese, P.; Wolf, R.F.; Harrison, P.; Downs, T.; Hamilton, K.; Comp, P.; Dale, G.L. Cytokine-induced alteration of platelet and hemostatic function. Stem Cells 1996, 14, 154–162. [Google Scholar] [CrossRef]
- Wassmann, S.; Stumpf, M.; Strehlow, K.; Schmid, A.; Schieffer, B.; Bohm, M.; Nickenig, G. Interleukin-6 induces oxidative stress and endothelial dysfunction by overexpression of the angiotensin II type 1 receptor. Circ. Res. 2004, 94, 534–541. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Arner, P.; Caro, J.F.; Atkinson, R.L.; Spiegelman, B.M. Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J. Clin. Investig. 1995, 95, 2409–2415. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Murray, D.L.; Choy, L.N.; Spiegelman, B.M. Tumor necrosis factor alpha inhibits signaling from the insulin receptor. Proc. Natl. Acad. Sci. USA 1994, 91, 4854–4858. [Google Scholar] [CrossRef]
- Zhang, H.H.; Halbleib, M.; Ahmad, F.; Manganiello, V.C.; Greenberg, A.S. Tumor necrosis factor-alpha stimulates lipolysis in differentiated human adipocytes through activation of extracellular signal-related kinase and elevation of intracellular cAMP. Diabetes 2002, 51, 2929–2935. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. The role of pattern-recognition receptors in innate immunity: Update on Toll-like receptors. Nat. Immunol. 2010, 11, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Akira, S.; Takeda, K.; Kaisho, T. Toll-like receptors: Critical proteins linking innate and acquired immunity. Nat. Immunol. 2001, 2, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Kokoeva, M.V.; Inouye, K.; Tzameli, I.; Yin, H.; Flier, J.S. TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Investig. 2006, 116, 3015–3025. [Google Scholar] [CrossRef] [PubMed]
- Himes, R.W.; Smith, C.W. Tlr2 is critical for diet-induced metabolic syndrome in a murine model. FASEB J. 2010, 24, 731–739. [Google Scholar] [CrossRef]
- Jialal, I.; Huet, B.A.; Kaur, H.; Chien, A.; Devaraj, S. Increased toll-like receptor activity in patients with metabolic syndrome. Diabetes Care 2012, 35, 900–904. [Google Scholar] [CrossRef]
- Hardy, O.T.; Kim, A.; Ciccarelli, C.; Hayman, L.L.; Wiecha, J. Increased Toll-like receptor (TLR) mRNA expression in monocytes is a feature of metabolic syndrome in adolescents. Pediatr. Obes. 2013, 8, e19–e23. [Google Scholar] [CrossRef]
- Lim, P.S.; Chang, Y.K.; Wu, T.K. Serum Lipopolysaccharide-Binding Protein is Associated with Chronic Inflammation and Metabolic Syndrome in Hemodialysis Patients. Blood Purif. 2019, 47, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Leber, B.; Tripolt, N.J.; Blattl, D.; Eder, M.; Wascher, T.C.; Pieber, T.R.; Stauber, R.; Sourij, H.; Oettl, K.; Stadlbauer, V. The influence of probiotic supplementation on gut permeability in patients with metabolic syndrome: An open label, randomized pilot study. Eur. J. Clin. Nutr. 2012, 66, 1110–1115. [Google Scholar] [CrossRef] [PubMed]
- Perez-Sotelo, D.; Roca-Rivada, A.; Larrosa-Garcia, M.; Castelao, C.; Baamonde, I.; Baltar, J.; Crujeiras, A.B.; Seoane, L.M.; Casanueva, F.F.; Pardo, M. Visceral and subcutaneous adipose tissue express and secrete functional alpha2hsglycoprotein (fetuin a) especially in obesity. Endocrine 2017, 55, 435–446. [Google Scholar] [CrossRef]
- Jialal, I.; Devaraj, S.; Bettaieb, A.; Haj, F.; Adams-Huet, B. Increased adipose tissue secretion of Fetuin-A, lipopolysaccharide-binding protein and high-mobility group box protein 1 in metabolic syndrome. Atherosclerosis 2015, 241, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Ix, J.H.; Shlipak, M.G.; Brandenburg, V.M.; Ali, S.; Ketteler, M.; Whooley, M.A. Association between human fetuin-A and the metabolic syndrome: Data from the Heart and Soul Study. Circulation 2006, 113, 1760–1767. [Google Scholar] [CrossRef]
- Pan, X.; Wen, S.W.; Bestman, P.L.; Kaminga, A.C.; Acheampong, K.; Liu, A. Fetuin-A in Metabolic syndrome: A systematic review and meta-analysis. PLoS ONE 2020, 15, e0229776. [Google Scholar] [CrossRef]
- Kalabay, L.; Chavin, K.; Lebreton, J.P.; Robinson, K.A.; Buse, M.G.; Arnaud, P. Human recombinant alpha 2-HS glycoprotein is produced in insect cells as a full length inhibitor of the insulin receptor tyrosine kinase. Horm. Metab. Res. 1998, 30, 1–6. [Google Scholar] [CrossRef]
- Auberger, P.; Falquerho, L.; Contreres, J.O.; Pages, G.; Le Cam, G.; Rossi, B.; Le Cam, A. Characterization of a natural inhibitor of the insulin receptor tyrosine kinase: cDNA cloning, purification, and anti-mitogenic activity. Cell 1989, 58, 631–640. [Google Scholar] [CrossRef]
- Srinivas, P.R.; Wagner, A.S.; Reddy, L.V.; Deutsch, D.D.; Leon, M.A.; Goustin, A.S.; Grunberger, G. Serum alpha 2-HS-glycoprotein is an inhibitor of the human insulin receptor at the tyrosine kinase level. Mol. Endocrinol. 1993, 7, 1445–1455. [Google Scholar] [CrossRef]
- Mathews, S.T.; Singh, G.P.; Ranalletta, M.; Cintron, V.J.; Qiang, X.; Goustin, A.S.; Jen, K.L.; Charron, M.J.; Jahnen-Dechent, W.; Grunberger, G. Improved insulin sensitivity and resistance to weight gain in mice null for the Ahsg gene. Diabetes 2002, 51, 2450–2458. [Google Scholar] [CrossRef]
- Siegel-Axel, D.I.; Ullrich, S.; Stefan, N.; Rittig, K.; Gerst, F.; Klingler, C.; Schmidt, U.; Schreiner, B.; Randrianarisoa, E.; Schaller, H.E.; et al. Fetuin-A influences vascular cell growth and production of proinflammatory and angiogenic proteins by human perivascular fat cells. Diabetologia 2014, 57, 1057–1066. [Google Scholar] [CrossRef]
- Dasgupta, S.; Bhattacharya, S.; Biswas, A.; Majumdar, S.S.; Mukhopadhyay, S.; Ray, S.; Bhattacharya, S. NF-kappaB mediates lipid-induced fetuin-A expression in hepatocytes that impairs adipocyte function effecting insulin resistance. BioChem. J. 2010, 429, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Stefan, N.; Sun, Q.; Fritsche, A.; Machann, J.; Schick, F.; Gerst, F.; Jeppesen, C.; Joost, H.G.; Hu, F.B.; Boeing, H.; et al. Impact of the adipokine adiponectin and the hepatokine fetuin-A on the development of type 2 diabetes: Prospective cohort- and cross-sectional phenotyping studies. PLoS ONE 2014, 9, e92238. [Google Scholar] [CrossRef] [PubMed]
- Vionnet, N.; Hani, E.H.; Dupont, S.; Gallina, S.; Francke, S.; Dotte, S.; De Matos, F.; Durand, E.; Lepretre, F.; Lecoeur, C.; et al. Genomewide search for type 2 diabetes-susceptibility genes in French whites: Evidence for a novel susceptibility locus for early-onset diabetes on chromosome 3q27-qter and independent replication of a type 2-diabetes locus on chromosome 1q21-q24. Am. J. Hum. Genet. 2000, 67, 1470–1480. [Google Scholar] [CrossRef]
- Kissebah, A.H.; Sonnenberg, G.E.; Myklebust, J.; Goldstein, M.; Broman, K.; James, R.G.; Marks, J.A.; Krakower, G.R.; Jacob, H.J.; Weber, J.; et al. Quantitative trait loci on chromosomes 3 and 17 influence phenotypes of the metabolic syndrome. Proc. Natl. Acad. Sci. USA 2000, 97, 14478–14483. [Google Scholar] [CrossRef] [PubMed]
- Thakkinstian, A.; Chailurkit, L.; Warodomwichit, D.; Ratanachaiwong, W.; Yamwong, S.; Chanprasertyothin, S.; Attia, J.; Sritara, P.; Ongphiphadhanakul, B. Causal relationship between body mass index and fetuin-A level in the asian population: A bidirectional Mendelian randomization study. Clin. Endocrinol. (Oxf) 2014, 81, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Brookes, P.S. Mitochondrial H(+) leak and ROS generation: An odd couple. Free Radic. Biol. Med. 2005, 38, 12–23. [Google Scholar] [CrossRef]
- Wang, B.; Van Veldhoven, P.P.; Brees, C.; Rubio, N.; Nordgren, M.; Apanasets, O.; Kunze, M.; Baes, M.; Agostinis, P.; Fransen, M. Mitochondria are targets for peroxisome-derived oxidative stress in cultured mammalian cells. Free Radic. Biol. Med. 2013, 65, 882–894. [Google Scholar] [CrossRef]
- Andreyev, A.Y.; Kushnareva, Y.E.; Starkov, A.A. Mitochondrial metabolism of reactive oxygen species. Biochemistry (Moscow) 2005, 70, 200–214. [Google Scholar] [CrossRef]
- Bhatti, J.S.; Bhatti, G.K.; Reddy, P.H. Mitochondrial dysfunction and oxidative stress in metabolic disorders—A step towards mitochondria based therapeutic strategies. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1066–1077. [Google Scholar] [CrossRef]
- Masschelin, P.M.; Cox, A.R.; Chernis, N.; Hartig, S.M. The Impact of Oxidative Stress on Adipose Tissue Energy Balance. Front. Physiol. 2019, 10, 1638. [Google Scholar] [CrossRef] [PubMed]
- Kowaltowski, A.J.; Vercesi, A.E. Mitochondrial damage induced by conditions of oxidative stress. Free Radic. Biol. Med. 1999, 26, 463–471. [Google Scholar] [CrossRef]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M.; Yamada, Y.; Nakajima, Y.; Nakayama, O.; Makishima, M.; Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Investig. 2004, 114, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Burkart, A.M.; Tan, K.; Warren, L.; Iovino, S.; Hughes, K.J.; Kahn, C.R.; Patti, M.E. Insulin Resistance in Human iPS Cells Reduces Mitochondrial Size and Function. Sci. Rep. 2016, 6, 22788. [Google Scholar] [CrossRef]
- Handschin, C.; Spiegelman, B.M. Peroxisome proliferator-activated receptor gamma coactivator 1 coactivators, energy homeostasis, and metabolism. Endocr. Rev. 2006, 27, 728–735. [Google Scholar] [CrossRef]
- Canto, C.; Auwerx, J. PGC-1alpha, SIRT1 and AMPK, an energy sensing network that controls energy expenditure. Curr. Opin. Lipidol. 2009, 20, 98–105. [Google Scholar] [CrossRef]
- Scarpulla, R.C.; Vega, R.B.; Kelly, D.P. Transcriptional integration of mitochondrial biogenesis. Trends Endocrinol. Metab. 2012, 23, 459–466. [Google Scholar] [CrossRef]
- Barshad, G.; Marom, S.; Cohen, T.; Mishmar, D. Mitochondrial DNA Transcription and Its Regulation: An Evolutionary Perspective. Trends Genet. 2018, 34, 682–692. [Google Scholar] [CrossRef]
- Vega, R.B.; Huss, J.M.; Kelly, D.P. The coactivator PGC-1 cooperates with peroxisome proliferator-activated receptor alpha in transcriptional control of nuclear genes encoding mitochondrial fatty acid oxidation enzymes. Mol. Cell Biol. 2000, 20, 1868–1876. [Google Scholar] [CrossRef]
- Olmos, Y.; Valle, I.; Borniquel, S.; Tierrez, A.; Soria, E.; Lamas, S.; Monsalve, M. Mutual dependence of Foxo3a and PGC-1alpha in the induction of oxidative stress genes. J. Biol. Chem. 2009, 284, 14476–14484. [Google Scholar] [CrossRef]
- Valle, I.; Alvarez-Barrientos, A.; Arza, E.; Lamas, S.; Monsalve, M. PGC-1alpha regulates the mitochondrial antioxidant defense system in vascular endothelial cells. Cardiovasc. Res. 2005, 66, 562–573. [Google Scholar] [CrossRef]
- Vandenbeek, R.; Khan, N.P.; Estall, J.L. Linking Metabolic Disease With the PGC-1alpha Gly482Ser Polymorphism. Endocrinology 2018, 159, 853–865. [Google Scholar] [CrossRef]
- Komen, J.C.; Thorburn, D.R. Turn up the power—Pharmacological activation of mitochondrial biogenesis in mouse models. Br. J. Pharmacol. 2014, 171, 1818–1836. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Boss, O. Targeting PGC-1 alpha to control energy homeostasis. Expert Opin. Targets 2007, 11, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Fortuño, A.; San José, G.; Moreno, M.U.; Beloqui, O.; Díez, J.; Zalba, G. Phagocytic NADPH oxidase overactivity underlies oxidative stress in metabolic syndrome. Diabetes 2006, 55, 209–215. [Google Scholar] [CrossRef]
- Jialal, I.; Devaraj, S.; Adams-Huet, B.; Chen, X.; Kaur, H. Increased cellular and circulating biomarkers of oxidative stress in nascent metabolic syndrome. J. Clin. Endocrinol. Metab. 2012, 97, E1844–E1850. [Google Scholar] [CrossRef]
- Avogaro, A.; Pagnin, E.; Calò, L. Monocyte NADPH oxidase subunit p22phox and inducible hemeoxygenase-1 gene expressions are increased in type II diabetic patients: Relationship with oxidative stress. J. Clin. Endocrinol. Metab. 2003, 88, 1753–1759. [Google Scholar] [CrossRef][Green Version]
- Friedman, R.C.; Farh, K.K.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef]
- Selbach, M.; Schwanhausser, B.; Thierfelder, N.; Fang, Z.; Khanin, R.; Rajewsky, N. Widespread changes in protein synthesis induced by microRNAs. Nature 2008, 455, 58–63. [Google Scholar] [CrossRef]
- Li, M.; Zhang, J. Circulating MicroRNAs: Potential and Emerging Biomarkers for Diagnosis of Cardiovascular and Cerebrovascular Diseases. BioMed Res. Int. 2015, 2015, 730535. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Song, Y.; Yao, L.; Song, G.; Teng, C. Circulating microRNAs: Promising Biomarkers Involved in Several Cancers and Other Diseases. DNA Cell Biol. 2017, 36, 77–94. [Google Scholar] [CrossRef] [PubMed]
- Deiuliis, J.A. MicroRNAs as regulators of metabolic disease: Pathophysiologic significance and emerging role as biomarkers and therapeutics. Int. J. Obes. 2016, 40, 88–101. [Google Scholar] [CrossRef]
- Calderari, S.; Diawara, M.R.; Garaud, A.; Gauguier, D. Biological roles of microRNAs in the control of insulin secretion and action. Physiol. Genom. 2017, 49, 1–10. [Google Scholar] [CrossRef]
- Heneghan, H.M.; Miller, N.; McAnena, O.J.; O’Brien, T.; Kerin, M.J. Differential miRNA expression in omental adipose tissue and in the circulation of obese patients identifies novel metabolic biomarkers. J. Clin. Endocrinol. Metab. 2011, 96, E846–E850. [Google Scholar] [CrossRef]
- Martinelli, R.; Nardelli, C.; Pilone, V.; Buonomo, T.; Liguori, R.; Castano, I.; Buono, P.; Masone, S.; Persico, G.; Forestieri, P.; et al. miR-519d overexpression is associated with human obesity. Obesity (Silver Spring) 2010, 18, 2170–2176. [Google Scholar] [CrossRef] [PubMed]
- Poy, M.N.; Eliasson, L.; Krutzfeldt, J.; Kuwajima, S.; Ma, X.; Macdonald, P.E.; Pfeffer, S.; Tuschl, T.; Rajewsky, N.; Rorsman, P.; et al. A pancreatic islet-specific microRNA regulates insulin secretion. Nature 2004, 432, 226–230. [Google Scholar] [CrossRef]
- Frost, R.J.; Olson, E.N. Control of glucose homeostasis and insulin sensitivity by the Let-7 family of microRNAs. Proc. Natl. Acad. Sci. USA 2011, 108, 21075–21080. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.T.; Tsai, P.C.; Liao, Y.C.; Hsu, C.Y.; Juo, S.H. Circulating microRNAs have a sex-specific association with metabolic syndrome. J. BioMed Sci. 2013, 20, 72. [Google Scholar] [CrossRef]
- Sharma, S.; Eghbali, M. Influence of sex differences on microRNA gene regulation in disease. Biol. Sex Differ. 2014, 5, 3. [Google Scholar] [CrossRef]
- Ramzan, F.; D’Souza, R.F.; Durainayagam, B.R.; Milan, A.M.; Markworth, J.F.; Miranda-Soberanis, V.; Sequeira, I.R.; Roy, N.C.; Poppitt, S.D.; Mitchell, C.J.; et al. Circulatory miRNA biomarkers of metabolic syndrome. Acta Diabetol. 2020, 57, 203–214. [Google Scholar] [CrossRef]
- Backes, C.; Kehl, T.; Stockel, D.; Fehlmann, T.; Schneider, L.; Meese, E.; Lenhof, H.P.; Keller, A. miRPathDB: A new dictionary on microRNAs and target pathways. Nucleic. Acids Res. 2017, 45, D90–D96. [Google Scholar] [CrossRef]
- Sliwinska, A.; Kasinska, M.A.; Drzewoski, J. MicroRNAs and metabolic disorders—Where are we heading? Arch. Med. Sci. 2017, 13, 885–896. [Google Scholar] [CrossRef]
- Chen, Y.; Tian, L.; Wan, S.; Xie, Y.; Chen, X.; Ji, X.; Zhao, Q.; Wang, C.; Zhang, K.; Hock, J.M.; et al. MicroRNA-17-92 cluster regulates pancreatic beta-cell proliferation and adaptation. Mol. Cell Endocrinol. 2016, 437, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.L.; Jiang, B.G.; Li, W.T.; Zou, J.J.; Shi, Y.Q.; Liu, Z.M. MicroRNA-15a positively regulates insulin synthesis by inhibiting uncoupling protein-2 expression. Diabetes Res. Clin. Pract. 2011, 91, 94–100. [Google Scholar] [CrossRef]
- Kamalden, T.A.; Macgregor-Das, A.M.; Kannan, S.M.; Dunkerly-Eyring, B.; Khaliddin, N.; Xu, Z.; Fusco, A.P.; Yazib, S.A.; Chow, R.C.; Duh, E.J.; et al. Exosomal MicroRNA-15a Transfer from the Pancreas Augments Diabetic Complications by Inducing Oxidative Stress. Antioxid. Redox Signal. 2017, 27, 913–930. [Google Scholar] [CrossRef]
- Rawal, S.; Munasinghe, P.E.; Nagesh, P.T.; Lew, J.K.S.; Jones, G.T.; Williams, M.J.A.; Davis, P.; Bunton, D.; Galvin, I.F.; Manning, P.; et al. Down-regulation of miR-15a/b accelerates fibrotic remodelling in the Type 2 diabetic human and mouse heart. Clin. Sci. (Lond.) 2017, 131, 847–863. [Google Scholar] [CrossRef] [PubMed]
- Banikazemi, Z.; Haji, H.A.; Mohammadi, M.; Taheripak, G.; Iranifar, E.; Poursadeghiyan, M.; Moridikia, A.; Rashidi, B.; Taghizadeh, M.; Mirzaei, H. Diet and cancer prevention: Dietary compounds, dietary MicroRNAs, and dietary exosomes. J. Cell BioChem 2018, 119, 185–196. [Google Scholar] [CrossRef]
- Marsetti, P.S.; Milagro, F.I.; Zulet, M.A.; Martinez, J.A.; Lorente-Cebrian, S. Changes in miRNA expression with two weight-loss dietary strategies in a population with metabolic syndrome. Nutrition 2021, 83, 111085. [Google Scholar] [CrossRef] [PubMed]
- Hermsdorff, H.H.; Zulet, M.A.; Abete, I.; Martinez, J.A. Discriminated benefits of a Mediterranean dietary pattern within a hypocaloric diet program on plasma RBP4 concentrations and other inflammatory markers in obese subjects. Endocrine 2009, 36, 445–451. [Google Scholar] [CrossRef]
- Van Horn, L.; Carson, J.A.; Appel, L.J.; Burke, L.E.; Economos, C.; Karmally, W.; Lancaster, K.; Lichtenstein, A.H.; Johnson, R.K.; Thomas, R.J.; et al. Recommended Dietary Pattern to Achieve Adherence to the American Heart Association/American College of Cardiology (AHA/ACC) Guidelines: A Scientific Statement From the American Heart Association. Circulation 2016, 134, e505–e529. [Google Scholar] [CrossRef]
- Rome, S. Use of miRNAs in biofluids as biomarkers in dietary and lifestyle intervention studies. Genes Nutr. 2015, 10, 483. [Google Scholar] [CrossRef]
- Cook, S.; Weitzman, M.; Auinger, P.; Nguyen, M.; Dietz, W.H. Prevalence of a metabolic syndrome phenotype in adolescents: Findings from the third National Health and Nutrition Examination Survey, 1988–1994. Arch. Pediatr. Adolesc. Med. 2003, 157, 821–827. [Google Scholar] [CrossRef]
- Reinehr, T.; de Sousa, G.; Toschke, A.M.; Andler, W. Comparison of metabolic syndrome prevalence using eight different definitions: A critical approach. Arch. Dis. Child. 2007, 92, 1067–1072. [Google Scholar] [CrossRef]
- Nagrani, R.; Foraita, R.; Gianfagna, F.; Iacoviello, L.; Marild, S.; Michels, N.; Molnar, D.; Moreno, L.; Russo, P.; Veidebaum, T.; et al. Common genetic variation in obesity, lipid transfer genes and risk of Metabolic Syndrome: Results from IDEFICS/I.Family study and meta-analysis. Sci. Rep. 2020, 10, 7189. [Google Scholar] [CrossRef]
- Ahrens, W.; Bammann, K.; Siani, A.; Buchecker, K.; De Henauw, S.; Iacoviello, L.; Hebestreit, A.; Krogh, V.; Lissner, L.; Marild, S.; et al. The IDEFICS cohort: Design, characteristics and participation in the baseline survey. Int. J. Obes. 2011, 35, S3–S15. [Google Scholar] [CrossRef]
- Ahrens, W.; Siani, A.; Adan, R.; De Henauw, S.; Eiben, G.; Gwozdz, W.; Hebestreit, A.; Hunsberger, M.; Kaprio, J.; Krogh, V.; et al. Cohort Profile: The transition from childhood to adolescence in European children-how I. Family extends the IDEFICS cohort. Int. J. Epidemiol. 2017, 46, 1394–1395j. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhang, D.; Zhou, D.; Li, Z.; Li, Z.; Fang, L.; Yang, M.; Shan, Z.; Li, H.; Chen, J.; et al. Susceptibility loci for metabolic syndrome and metabolic components identified in Han Chinese: A multi-stage genome-wide association study. J. Cell Mol. Med. 2017, 21, 1106–1116. [Google Scholar] [CrossRef] [PubMed]
- Khella, M.S.; Hamdy, N.M.; Amin, A.I.; El-Mesallamy, H.O. The (FTO) gene polymorphism is associated with metabolic syndrome risk in Egyptian females: A case- control study. BMC Med. Genet. 2017, 18, 101. [Google Scholar] [CrossRef] [PubMed]
- Dina, C.; Meyre, D.; Gallina, S.; Durand, E.; Korner, A.; Jacobson, P.; Carlsson, L.M.; Kiess, W.; Vatin, V.; Lecoeur, C.; et al. Variation in FTO contributes to childhood obesity and severe adult obesity. Nat. Genet. 2007, 39, 724–726. [Google Scholar] [CrossRef]
- Zeggini, E.; Weedon, M.N.; Lindgren, C.M.; Frayling, T.M.; Elliott, K.S.; Lango, H.; Timpson, N.J.; Perry, J.R.; Rayner, N.W.; Freathy, R.M.; et al. Replication of genome-wide association signals in UK samples reveals risk loci for type 2 diabetes. Science 2007, 316, 1336–1341. [Google Scholar] [CrossRef]
- Gerken, T.; Girard, C.A.; Tung, Y.C.; Webby, C.J.; Saudek, V.; Hewitson, K.S.; Yeo, G.S.; McDonough, M.A.; Cunliffe, S.; McNeill, L.A.; et al. The obesity-associated FTO gene encodes a 2-oxoglutarate-dependent nucleic acid demethylase. Science 2007, 318, 1469–1472. [Google Scholar] [CrossRef]
- Mizuno, T.M. Fat Mass and Obesity Associated (FTO) Gene and Hepatic Glucose and Lipid Metabolism. Nutrients 2018, 10, 1600. [Google Scholar] [CrossRef] [PubMed]
- Romero-Nava, R.; Garcia, N.; Aguayo-Ceron, K.A.; Sanchez Munoz, F.; Huang, F.; Hong, E.; Villafana, S. Modifications in GPR21 and GPR82 genes expression as a consequence of metabolic syndrome etiology. J. Recept. Signal Transduct. Res. 2021, 41, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Hernandez, A.; Sanchez-Munoz, F.; Rodriguez, J.; Calderon-Zamora, L.; Romero-Nava, R.; Huang, F.; Hong, E.; Villafana, S. Expression of orphan receptors GPR22 and GPR162 in streptozotocin-induced diabetic rats. J. Recept. Signal Transduct. Res. 2015, 35, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Uhlenbrock, K.; Gassenhuber, H.; Kostenis, E. Sphingosine 1-phosphate is a ligand of the human gpr3, gpr6 and gpr12 family of constitutively active G protein-coupled receptors. Cell Signal. 2002, 14, 941–953. [Google Scholar] [CrossRef]
- Osborn, O.; Oh, D.Y.; McNelis, J.; Sanchez-Alavez, M.; Talukdar, S.; Lu, M.; Li, P.; Thiede, L.; Morinaga, H.; Kim, J.J.; et al. G protein-coupled receptor 21 deletion improves insulin sensitivity in diet-induced obese mice. J. Clin. Investig. 2012, 122, 2444–2453. [Google Scholar] [CrossRef]
- Bresnick, J.N.; Skynner, H.A.; Chapman, K.L.; Jack, A.D.; Zamiara, E.; Negulescu, P.; Beaumont, K.; Patel, S.; McAllister, G. Identification of signal transduction pathways used by orphan g protein-coupled receptors. Assay Drug Dev. Technol. 2003, 1, 239–249. [Google Scholar] [CrossRef]
- Molkentin, J.D.; Dorn, G.W., 2nd. Cytoplasmic signaling pathways that regulate cardiac hypertrophy. Annu. Rev. Physiol. 2001, 63, 391–426. [Google Scholar] [CrossRef]
- Edwards, R.M.; Trizna, W.; Kinter, L.B. Renal microvascular effects of vasopressin and vasopressin antagonists. Am. J. Physiol. 1989, 256, F274–F278. [Google Scholar] [CrossRef]
- Engel, K.M.; Schrock, K.; Teupser, D.; Holdt, L.M.; Tonjes, A.; Kern, M.; Dietrich, K.; Kovacs, P.; Krugel, U.; Scheidt, H.A.; et al. Reduced food intake and body weight in mice deficient for the G protein-coupled receptor GPR82. PLoS ONE 2011, 6, e29400. [Google Scholar] [CrossRef]
- Lee, J.H.; Friso, S.; Choi, S.W. Epigenetic mechanisms underlying the link between non-alcoholic fatty liver diseases and nutrition. Nutrients 2014, 6, 3303–3325. [Google Scholar] [CrossRef] [PubMed]
- Del Campo, J.A.; Gallego-Duran, R.; Gallego, P.; Grande, L. Genetic and Epigenetic Regulation in Nonalcoholic Fatty Liver Disease (NAFLD). Int. J. Mol. Sci. 2018, 19, 911. [Google Scholar] [CrossRef] [PubMed]
- Michael, M.D.; Kulkarni, R.N.; Postic, C.; Previs, S.F.; Shulman, G.I.; Magnuson, M.A.; Kahn, C.R. Loss of insulin signaling in hepatocytes leads to severe insulin resistance and progressive hepatic dysfunction. Mol. Cell 2000, 6, 87–97. [Google Scholar] [CrossRef]
- Cohen, S.E.; Kokkotou, E.; Biddinger, S.B.; Kondo, T.; Gebhardt, R.; Kratzsch, J.; Mantzoros, C.S.; Kahn, C.R. High circulating leptin receptors with normal leptin sensitivity in liver-specific insulin receptor knock-out (LIRKO) mice. J. Biol. Chem. 2007, 282, 23672–23678. [Google Scholar] [CrossRef]
- De Jesus, D.F.; Orime, K.; Kaminska, D.; Kimura, T.; Basile, G.; Wang, C.H.; Haertle, L.; Riemens, R.; Brown, N.K.; Hu, J.; et al. Parental metabolic syndrome epigenetically reprograms offspring hepatic lipid metabolism in mice. J. Clin. Investig. 2020, 130, 2391–2407. [Google Scholar] [CrossRef]
- Wang, Q.; Jiang, L.; Wang, J.; Li, S.; Yu, Y.; You, J.; Zeng, R.; Gao, X.; Rui, L.; Li, W.; et al. Abrogation of hepatic ATP-citrate lyase protects against fatty liver and ameliorates hyperglycemia in leptin receptor-deficient mice. Hepatology 2009, 49, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
- Paliwal, S.; Shi, J.; Dhru, U.; Zhou, Y.; Schuger, L. P311 binds to the latency associated protein and downregulates the expression of TGF-beta1 and TGF-beta2. BioChem Biophys Res. Commun. 2004, 315, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Lent-Schochet, D.; McLaughlin, M.; Ramakrishnan, N.; Jialal, I. Exploratory metabolomics of metabolic syndrome: A status report. World J. Diabetes 2019, 10, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Yamada, J.; Tomiyama, H.; Yambe, M.; Koji, Y.; Motobe, K.; Shiina, K.; Yamamoto, Y.; Yamashina, A. Elevated serum levels of alanine aminotransferase and gamma glutamyltransferase are markers of inflammation and oxidative stress independent of the metabolic syndrome. Atherosclerosis 2006, 189, 198–205. [Google Scholar] [CrossRef]
- Velasquez, M.T.; Ramezani, A.; Manal, A.; Raj, D.S. Trimethylamine N-Oxide: The Good, the Bad and the Unknown. Toxins 2016, 8, 326. [Google Scholar] [CrossRef]
- Tang, W.H.; Hazen, S.L. Microbiome, trimethylamine N-oxide, and cardiometabolic disease. Transl. Res. 2017, 179, 108–115. [Google Scholar] [CrossRef]
- Hart, L.M.; Vogelzangs, N.; Mook-Kanamori, D.O.; Brahimaj, A.; Nano, J.; van der Heijden, A.; Willems van Dijk, K.; Slieker, R.C.; Steyerberg, E.W.; Ikram, M.A.; et al. Blood Metabolomic Measures Associate With Present and Future Glycemic Control in Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2018, 103, 4569–4579. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.H. Emerging perspectives on essential amino acid metabolism in obesity and the insulin-resistant state. Adv. Nutr. 2011, 2, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Zheng, X.; Ma, X.; Bao, Y.; Ni, Y.; Hu, C.; Rajani, C.; Huang, F.; Zhao, A.; Jia, W.; et al. Tryptophan Predicts the Risk for Future Type 2 Diabetes. PLoS ONE 2016, 11, e0162192. [Google Scholar] [CrossRef] [PubMed]
- Oxenkrug, G.; van der Hart, M.; Summergrad, P. Elevated anthranilic acid plasma concentrations in type 1 but not type 2 diabetes mellitus. Integr. Mol. Med. 2015, 2, 365–368. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Feng, R.N.; Niu, Y.C.; Sun, X.W.; Li, Q.; Zhao, C.; Wang, C.; Guo, F.C.; Sun, C.H.; Li, Y. Histidine supplementation improves insulin resistance through suppressed inflammation in obese women with the metabolic syndrome: A randomised controlled trial. Diabetologia 2013, 56, 985–994. [Google Scholar] [CrossRef]
- Iida, M.; Harada, S.; Kurihara, A.; Fukai, K.; Kuwabara, K.; Sugiyama, D.; Takeuchi, A.; Okamura, T.; Akiyama, M.; Nishiwaki, Y.; et al. Profiling of plasma metabolites in postmenopausal women with metabolic syndrome. Menopause 2016, 23, 749–758. [Google Scholar] [CrossRef]
- Kennedy, A.J.; Ellacott, K.L.; King, V.L.; Hasty, A.H. Mouse models of the metabolic syndrome. Dis. Model. Mech. 2010, 3, 156–166. [Google Scholar] [CrossRef]
- Ingalls, A.M.; Dickie, M.M.; Shell, G. Obese, a new mutation in the house mouse. J. Hered. 1950, 41, 317–318. [Google Scholar] [CrossRef]
- Hummel, K.P.; Dickie, M.M.; Coleman, D.L. Diabetes, a new mutafton in the mouse. Science 1966, 153, 1127–1128. [Google Scholar] [CrossRef]
- Dickie, M.M. Mutations at the agouti locus in the mouse. J. Hered. 1969, 60, 20–25. [Google Scholar] [CrossRef]
- Huszar, D.; Lynch, C.A.; Fairchild-Huntress, V.; Dunmore, J.H.; Fang, Q.; Berkemeier, L.R.; Gu, W.; Kesterson, R.A.; Boston, B.A.; Cone, R.D.; et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 1997, 88, 131–141. [Google Scholar] [CrossRef]
- Ishibashi, S.; Brown, M.S.; Goldstein, J.L.; Gerard, R.D.; Hammer, R.E.; Herz, J. Hypercholesterolemia in low density lipoprotein receptor knockout mice and its reversal by adenovirus-mediated gene delivery. J. Clin. Investig. 1993, 92, 883–893. [Google Scholar] [CrossRef]
- Hasty, A.H.; Shimano, H.; Osuga, J.-I.; Namatame, I.; Takahashi, A.; Yahagi, N.; Perrey, S.; Iizuka, Y.; Tamura, Y.; Amemiya-Kudo, M.; et al. Severe hypercholesterolemia, hypertriglyceridemia, and atherosclerosis in mice lacking both leptin and the low density lipoprotein receptor. J. Biol. Chem. 2001, 276, 37402–37408. [Google Scholar] [CrossRef]
- Plump, A.S.; Smith, J.D.; Hayek, T.; Aalto-Setälä, K.; Walsh, A.; Verstuyft, J.G.; Rubin, E.M.; Breslow, J.L. Severe hypercholesterolemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell 1992, 71, 343–353. [Google Scholar]
- Gruen, M.L.; Plummer, M.R.; Zhang, W.; Posey, K.A.; Linton, M.; Fazio, S.; Hasty, A.H. Persistence of high density lipoprotein particles in obese mice lacking apolipoprotein AI. J. Lipid Res. 2005, 46, 2007–2014. [Google Scholar] [CrossRef]
- Coenen, K.R.; Hasty, A.H. Obesity potentiates development of fatty liver and insulin resistance, but not atherosclerosis, in high-fat diet-fed agouti LDLR-deficient mice. Am. J. Physiol.-Endocrinol. Metab. 2007, 293, E492–E499. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chang, G.-R.; Chiu, Y.-S.; Wu, Y.-Y.; Chen, W.-Y.; Liao, J.-W.; Chao, T.-H.; Mao, F.C. Rapamycin protects against high fat diet–induced obesity in C57BL/6J mice. J. Pharmacol. Sci. 2009, 109, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Aizawa-Abe, M.; Ogawa, Y.; Masuzaki, H.; Ebihara, K.; Satoh, N.; Iwai, H.; Matsuoka, N.; Hayashi, T.; Hosoda, K.; Inoue, G.; et al. Pathophysiological role of leptin in obesity-related hypertension. J. Clin. Investig. 2000, 105, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, D.J.; McCormick, J.; Helmering, J.; Kim, K.W.; Wang, M.; Fordstrom, P.; Kaufman, S.A.; Lindberg, R.A.; Véniant, M.M. Generation and characterization of two novel mouse models exhibiting the phenotypes of the metabolic syndrome: Apob48−/− Lepob/ob mice devoid of ApoE or Ldlr. Am. J. Physiol.-Endocrinol. Metab. 2008, 294, E496–E505. [Google Scholar] [CrossRef][Green Version]
- King, V.L.; Hatch, N.W.; Chan, H.W.; De Beer, M.C.; De Beer, F.C.; Tannock, L.R. A murine model of obesity with accelerated atherosclerosis. Obesity 2010, 18, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.J.; McLemore, G.R., Jr.; Hendrix, S.T. Insulin resistance and obesity in a mouse model of systemic lupus erythematosus. Hypertension 2006, 48, 988–993. [Google Scholar] [CrossRef]
- Kastorini, C.M.; Milionis, H.J.; Esposito, K.; Giugliano, D.; Goudevenos, J.A.; Panagiotakos, D.B. The effect of Mediterranean diet on metabolic syndrome and its components: A meta-analysis of 50 studies and 534,906 individuals. J. Am. Coll. Cardiol. 2011, 57, 1299–1313. [Google Scholar] [CrossRef]
- Esposito, K.; Kastorini, C.M.; Panagiotakos, D.B.; Giugliano, D. Mediterranean diet and metabolic syndrome: An updated systematic review. Rev. Endocr. Metab. Disord. 2013, 14, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Ciotola, M.; Giugliano, D. Mediterranean diet and the metabolic syndrome. Mol. Nutr. Food Res. 2007, 51, 1268–1274. [Google Scholar] [CrossRef]
- Dai, J.; Jones, D.P.; Goldberg, J.; Ziegler, T.R.; Bostick, R.M.; Wilson, P.W.; Manatunga, A.K.; Shallenberger, L.; Jones, L.; Vaccarino, V. Association between adherence to the Mediterranean diet and oxidative stress. Am. J. Clin. Nutr. 2008, 88, 1364–1370. [Google Scholar] [PubMed]
- Pitsavos, C.; Panagiotakos, D.B.; Tzima, N.; Chrysohoou, C.; Economou, M.; Zampelas, A.; Stefanadis, C. Adherence to the Mediterranean diet is associated with total antioxidant capacity in healthy adults: The ATTICA study. Am. J. Clin. Nutr. 2005, 82, 694–699. [Google Scholar] [CrossRef]
- Giugliano, D.; Esposito, K. Mediterranean diet and metabolic diseases. Curr. Opin. Lipidol. 2008, 19, 63–68. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Guasch-Ferré, M.; Lee, C.-H.; Estruch, R.; Clish, C.B.; Ros, E. Protective effects of the Mediterranean diet on type 2 diabetes and metabolic syndrome. J. Nutr. 2015, 146, 920S–927S. [Google Scholar] [CrossRef]
- Rochlani, Y.; Pothineni, N.V.; Kovelamudi, S.; Mehta, J.L. Metabolic syndrome: Pathophysiology, management, and modulation by natural compounds. Adv. Cardiovasc. Dis. 2017, 11, 215–225. [Google Scholar] [CrossRef]
- Aggarwal, B.B. Targeting inflammation-induced obesity and metabolic diseases by curcumin and other nutraceuticals. Annu. Rev. Nutr. 2010, 30, 173–199. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Fu, Y.; Chen, A. Activation of peroxisome proliferator-activated receptor-gamma contributes to the inhibitory effects of curcumin on rat hepatic stellate cell growth. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 285, G20–G30. [Google Scholar] [CrossRef] [PubMed]
- Reinhart, K.M.; Talati, R.; White, C.M.; Coleman, C.I. The impact of garlic on lipid parameters: A systematic review and meta-analysis. Nutr. Res. Rev. 2009, 22, 39–48. [Google Scholar] [CrossRef]
- Yang, J.; Yin, J.; Gao, H.; Xu, L.; Wang, Y.; Xu, L.; Li, M. Berberine improves insulin sensitivity by inhibiting fat store and adjusting adipokines profile in human preadipocytes and metabolic syndrome patients. Evid. Based Complement Altern. Med. 2012, 2012, 363845. [Google Scholar] [CrossRef]
- Pérez-Rubio, K.G.; González-Ortiz, M.; Martínez-Abundis, E.; Robles-Cervantes, J.A.; Espinel-Bermúdez, M.C. Effect of berberine administration on metabolic syndrome, insulin sensitivity, and insulin secretion. Metab. Syndr. Relat. Disord. 2013, 11, 366–369. [Google Scholar] [CrossRef]
- Bhat, M.; Kothiwale, S.K.; Tirmale, A.R.; Bhargava, S.Y.; Joshi, B.N. Antidiabetic Properties of Azardiracta indica and Bougainvillea spectabilis: In Vivo Studies in Murine Diabetes Model. Evid. Based Complement Altern. Med. 2011, 2011, 561625. [Google Scholar] [CrossRef]
- Mollace, V.; Ragusa, S.; Sacco, I.; Muscoli, C.; Sculco, F.; Visalli, V.; Palma, E.; Muscoli, S.; Mondello, L.; Dugo, P.; et al. The protective effect of bergamot oil extract on lecitine-like oxyLDL receptor-1 expression in balloon injury-related neointima formation. J. Cardiovasc. Pharm. 2008, 13, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Bremer, A.A. Resveratrol use in metabolic syndrome. Metab. Syndr. Relat. Disord. 2014, 12, 493–495. [Google Scholar] [CrossRef]
- Méndez-del Villar, M.; González-Ortiz, M.; Martínez-Abundis, E.; Pérez-Rubio, K.G.; Lizárraga-Valdez, R. Effect of resveratrol administration on metabolic syndrome, insulin sensitivity, and insulin secretion. Metab. Syndr. Relat. Disord. 2014, 12, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Rivera, L.; Morón, R.; Sánchez, M.; Zarzuelo, A.; Galisteo, M. Quercetin ameliorates metabolic syndrome and improves the inflammatory status in obese Zucker rats. Obesity (Silver Spring) 2008, 16, 2081–2087. [Google Scholar] [CrossRef]
- Gillies, P.J.; Bhatia, S.K.; Belcher, L.A.; Hannon, D.B.; Thompson, J.T.; Vanden Heuvel, J.P. Regulation of inflammatory and lipid metabolism genes by eicosapentaenoic acid-rich oil. J. Lipid Res. 2012, 53, 1679–1689. [Google Scholar] [CrossRef]
- Wu, L.; Juurlink, B.H. The impaired glutathione system and its up-regulation by sulforaphane in vascular smooth muscle cells from spontaneously hypertensive rats. J. Hypertens. 2001, 19, 1819–1825. [Google Scholar] [CrossRef]
- Castellino, G.; Nikolic, D.; Magán-Fernández, A.; Malfa, G.A.; Chianetta, R.; Patti, A.M.; Amato, A.; Montalto, G.; Toth, P.P.; Banach, M.; et al. Altilix® Supplement Containing Chlorogenic Acid and Luteolin Improved Hepatic and Cardiometabolic Parameters in Subjects with Metabolic Syndrome: A 6 Month Randomized, Double-Blind, Placebo-Controlled Study. Nutrients 2019, 11, 2580. [Google Scholar] [CrossRef]
- Fogacci, F.; Banach, M.; Mikhailidis, D.P.; Bruckert, E.; Toth, P.P.; Watts, G.F.; Reiner, Ž.; Mancini, J.; Rizzo, M.; Mitchenko, O.; et al. Safety of red yeast rice supplementation: A systematic review and meta-analysis of randomized controlled trials. Pharm. Res. 2019, 143, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Roy, C.C.; Kien, C.L.; Bouthillier, L.; Levy, E. Short-chain fatty acids: Ready for prime time? Nutr. Clin. Pract. 2006, 21, 351–366. [Google Scholar] [CrossRef]
- Zhang, X.; Shen, D.; Fang, Z.; Jie, Z.; Qiu, X.; Zhang, C.; Chen, Y.; Ji, L. Human gut microbiota changes reveal the progression of glucose intolerance. PLoS ONE 2013, 8, e71108. [Google Scholar]
- Vrieze, A.; Van Nood, E.; Holleman, F.; Salojärvi, J.; Kootte, R.S.; Bartelsman, J.F.; Dallinga–Thie, G.M.; Ackermans, M.T.; Serlie, M.J.; Oozeer, R.; et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology 2012, 143, 913–916.e7. [Google Scholar] [CrossRef] [PubMed]
- Canani, R.B.; Di Costanzo, M.; Leone, L.; Pedata, M.; Meli, R.; Calignano, A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. WJG 2011, 17, 1519. [Google Scholar] [CrossRef]
- Gao, Q.; Zhang, L. Ultraviolet-B-induced oxidative stress and antioxidant defense system responses in ascorbate-deficient vtc1 mutants of Arabidopsis. J. Plant Physiol. 2008, 165, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Hara, T.; Kimura, I.; Inoue, D.; Ichimura, A.; Hirasawa, A. Free fatty acid receptors and their role in regulation of energy metabolism. Rev. Physiol. Biochem. Pharmacol. 2013, 164, 77–116. [Google Scholar]
- Gao, Z.; Yin, J.; Zhang, J.; Ward, R.E.; Martin, R.J.; Lefevre, M.; Cefalu, W.T.; Ye, J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 2009, 58, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Steinert, R.E.; Feinle-Bisset, C.; Asarian, L.; Horowitz, M.; Beglinger, C.; Geary, N. Ghrelin, CCK, GLP-1, and PYY (3–36): Secretory controls and physiological roles in eating and glycemia in health, obesity, and after RYGB. Physiol. Rev. 2017, 97, 411–463. [Google Scholar] [CrossRef]
- Matheus, V.; Monteiro, L.; Oliveira, R.; Maschio, D.; Collares-Buzato, C. Butyrate reduces high-fat diet-induced metabolic alterations, hepatic steatosis and pancreatic beta cell and intestinal barrier dysfunctions in prediabetic mice. Exp. Biol. Med. 2017, 242, 1214–1226. [Google Scholar] [CrossRef] [PubMed]
- Keenan, M.J.; Zhou, J.; McCutcheon, K.L.; Raggio, A.M.; Bateman, H.G.; Todd, E.; Jones, C.K.; Tulley, R.T.; Melton, S.; Martin, R.J. Effects of resistant starch, a non-digestible fermentable fiber, on reducing body fat. Obesity 2006, 14, 1523–1534. [Google Scholar] [CrossRef]
- Yan, H.; Ajuwon, K.M. Mechanism of butyrate stimulation of triglyceride storage and adipokine expression during adipogenic differentiation of porcine stromovascular cells. PLoS ONE 2015, 10, e0145940. [Google Scholar]
- Hafidi, M.E.; Buelna-Chontal, M.; Sánchez-Muñoz, F.; Carbó, R. Adipogenesis: A necessary but harmful strategy. Int. J. Mol. Sci. 2019, 20, 3657. [Google Scholar] [CrossRef]
- Marcil, V.; Delvin, E.; Garofalo, C.; Levy, E. Butyrate impairs lipid transport by inhibiting microsomal triglyceride transfer protein in Caco-2 cells. J. Nutr. 2003, 133, 2180–2183. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xu, C.; Huang, R.; Song, J.; Li, D.; Xia, M. Butyrate from pectin fermentation inhibits intestinal cholesterol absorption and attenuates atherosclerosis in apolipoprotein E-deficient mice. J. Nutr. Biochem. 2018, 56, 175–182. [Google Scholar] [CrossRef]
- Khan, S.; Jena, G. Sodium butyrate reduces insulin-resistance, fat accumulation and dyslipidemia in type-2 diabetic rat: A comparative study with metformin. Chem. Biol. Interact. 2016, 254, 124–134. [Google Scholar] [CrossRef]
- Chriett, S.; Zerzaihi, O.; Vidal, H.; Pirola, L. The histone deacetylase inhibitor sodium butyrate improves insulin signalling in palmitate-induced insulin resistance in L6 rat muscle cells through epigenetically-mediated up-regulation of Irs1. Mol. Cell. Endocrinol. 2017, 439, 224–232. [Google Scholar] [CrossRef]
- Khan, S.; Jena, G. Protective role of sodium butyrate, a HDAC inhibitor on beta-cell proliferation, function and glucose homeostasis through modulation of p38/ERK MAPK and apoptotic pathways: Study in juvenile diabetic rat. Chem. Biol. Interact. 2014, 213, 1–12. [Google Scholar] [CrossRef]
- Roshanravan, N.; Mahdavi, R.; Alizadeh, E.; Jafarabadi, M.A.; Hedayati, M.; Ghavami, A.; Alipour, S.; Alamdari, N.M.; Barati, M.; Ostadrahimi, A. Effect of butyrate and inulin supplementation on glycemic status, lipid profile and glucagon-like peptide 1 level in patients with type 2 diabetes: A randomized double-blind, placebo-controlled trial. Horm. Metab. Res. 2017, 49, 886–891. [Google Scholar] [CrossRef] [PubMed]
- Cook, S.; Sellin, J. Short chain fatty acids in health and disease. Aliment. Pharmacol. Ther. 1998, 12, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Rephaeli, A.; Rabizadeh, E.; Aviram, A.; Shaklai, M.; Ruse, M.; Nudelman, A. Derivatives of butyric acid as potential anti-neoplastic agents. Int. J. Cancer 1991, 49, 66–72. [Google Scholar] [CrossRef]
- Patnaik, A.; Rowinsky, E.K.; Villalona, M.A.; Hammond, L.A.; Britten, C.D.; Siu, L.L.; Goetz, A.; Felton, S.A.; Burton, S.; Valone, F.H.; et al. A phase I study of pivaloyloxymethyl butyrate, a prodrug of the differentiating agent butyric acid, in patients with advanced solid malignancies. Clin. Cancer Res. 2002, 8, 2142–2148. [Google Scholar] [PubMed]
- Kim, S.W.; Hooker, J.M.; Otto, N.; Win, K.; Muench, L.; Shea, C.; Carter, P.; King, P.; Reid, A.E.; Volkow, N.D.; et al. Whole-body pharmacokinetics of HDAC inhibitor drugs, butyric acid, valproic acid and 4-phenylbutyric acid measured with carbon-11 labeled analogs by PET. Nucl. Med. Biol. 2013, 40, 912–918. [Google Scholar] [CrossRef]
- Krokowicz, L.; Stojcev, Z.; Kaczmarek, B.F.; Kociemba, W.; Kaczmarek, E.; Walkowiak, J.; Krokowicz, P.; Drews, M.; Banasiewicz, T. Microencapsulated sodium butyrate administered to patients with diverticulosis decreases incidence of diverticulitis—a prospective randomized study. Int. J. Color. Dis. 2014, 29, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, A.D.; Morera, R.; Ciccocioppo, R.; Cazzola, P.; Gotti, S.; Tinozzi, F.; Tinozzi, S.; Corazza, G. Oral butyrate for mildly to moderately active Crohn’s disease. Aliment. Pharmacol. Ther. 2005, 22, 789–794. [Google Scholar] [CrossRef]
- Vernia, P.; Monteleone, G.; Grandinetti, G.; Villotti, G.; Di Giulio, E.; Frieri, G.; Marcheggiano, A.; Pallone, F.; Caprilli, R.; Torsoli, A. Combined oral sodium butyrate and mesalazine treatment compared to oral mesalazine alone in ulcerative colitis. Dig. Dis. Sci. 2000, 45, 976–981. [Google Scholar] [CrossRef] [PubMed]
- Krokowicz, L.; Kaczmarek, B.F.; Krokowicz, P.; Stojcev, Z.; Mackiewicz, J.; Walkowiak, J.; Drews, M.; Banasiewicz, T. Sodium butyrate and short chain fatty acids in prevention of travellers’ diarrhoea: A randomized prospective study. Travel Med. Infect. Dis. 2014, 12, 183–188. [Google Scholar] [CrossRef]
- Bouter, K.; Bakker, G.; Levin, E.; Hartstra, A.; Kootte, R.; Udayappan, S.; Katiraei, S.; Bahler, L.; Gilijamse, P.; Tremaroli, V.; et al. Differential metabolic effects of oral butyrate treatment in lean versus metabolic syndrome subjects. Clin. Transl. Gastroenterol. 2018, 9, e155. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Ding, H.; Wang, T.; Hooper, L.V.; Koh, G.Y.; Nagy, A.; Semenkovich, C.F.; Gordon, J.I. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl. Acad. Sci. USA 2004, 101, 15718–15723. [Google Scholar] [CrossRef] [PubMed]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.-M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet–induced obesity and diabetes in mice. Diabetes 2008, 57, 1470–1481. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A core gut microbiome in obese and lean twins. Nature 2009, 457, 480–484. [Google Scholar] [CrossRef]
- Barreto, F.M.; Simão, A.N.C.; Morimoto, H.K.; Lozovoy, M.A.B.; Dichi, I.; da Silva Miglioranza, L.H. Beneficial effects of Lactobacillus plantarum on glycemia and homocysteine levels in postmenopausal women with metabolic syndrome. Nutrition 2014, 30, 939–942. [Google Scholar] [CrossRef] [PubMed]
- Stadlbauer, V.; Leber, B.; Lemesch, S.; Trajanoski, S.; Bashir, M.; Horvath, A.; Tawdrous, M.; Stojakovic, T.; Fauler, G.; Fickert, P.; et al. Lactobacillus casei Shirota supplementation does not restore gut microbiota composition and gut barrier in metabolic syndrome: A randomized pilot study. PLoS ONE 2015, 10, e0141399. [Google Scholar] [CrossRef] [PubMed]
- Tripolt, N.; Leber, B.; Blattl, D.; Eder, M.; Wonisch, W.; Scharnagl, H.; Stojakovic, T.; Obermayer-Pietsch, B.; Wascher, T.; Pieber, T. Effect of supplementation with Lactobacillus casei Shirota on insulin sensitivity, β-cell function, and markers of endothelial function and inflammation in subjects with metabolic syndrome—A pilot study. J. Dairy Sci. 2013, 96, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Szulińska, M.; Łoniewski, I.; Skrypnik, K.; Sobieska, M.; Korybalska, K.; Suliburska, J.; Bogdański, P. Multispecies probiotic supplementation favorably affects vascular function and reduces arterial stiffness in obese postmenopausal women—A 12-week placebo-controlled and randomized clinical study. Nutrients 2018, 10, 1672. [Google Scholar] [CrossRef] [PubMed]
- Tenorio-Jiménez, C.; Martínez-Ramírez, M.J.; Gil, Á. and Gómez-Llorente, C. Effects of Probiotics on Metabolic Syndrome: A Systematic Review of Randomized Clinical Trials. Nutrients 2020, 12, 124. [Google Scholar] [CrossRef] [PubMed]
- Wallace, C.J.; Milev, R.V. The efficacy, safety, and tolerability of probiotics on depression: Clinical results from an open-label pilot study. Front. Psychiatry 2021, 12, 132. [Google Scholar] [CrossRef]
- Szulińska, M.; Łoniewski, I.; Van Hemert, S.; Sobieska, M.; Bogdański, P. Dose-dependent effects of multispecies probiotic supplementation on the lipopolysaccharide (LPS) level and cardiometabolic profile in obese postmenopausal women: A 12-week randomized clinical trial. Nutrients 2018, 10, 773. [Google Scholar] [CrossRef]
- Sahhaf Ebrahimi, F.; Homayouni Rad, A.; Mosen, M.; Abbasalizadeh, F.; Tabrizi, A.; Khalili, L. Effect of L. acidophilus and B. lactis on blood glucose in women with gestational diabetes mellitus: A randomized placebo-controlled trial. Diabetol. Metab. Syndr. 2019, 11, 75–77. [Google Scholar] [CrossRef]
- Tenorio-Jimenez, C.; Martinez-Ramirez, M.J.; Del Castillo-Codes, I.; Arraiza-Irigoyen, C.; Tercero-Lozano, M.; Camacho, J.; Chueca, N.; Garcia, F.; Olza, J.; Plaza-Diaz, J.; et al. Lactobacillus reuteri V3401 Reduces Inflammatory Biomarkers and Modifies the Gastrointestinal Microbiome in Adults with Metabolic Syndrome: The PROSIR Study. Nutrients 2019, 11, 1761. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P. Metabolic syndrome—role of dietary fat type and quantity. Nutrients 2019, 11, 1438. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Hwang, K.M.; Cheon, S.Y.; Lee, H.J.; Yoon, H.S. Associations of obesity and dyslipidemia with intake of sodium, fat, and sugar among Koreans: A qualitative systematic review. Clin. Nutr. Res. 2016, 5, 290–304. [Google Scholar] [CrossRef] [PubMed]
- Hoyas, I.; Leon-Sanz, M. Nutritional challenges in metabolic syndrome. J. Clin. Med. 2019, 8, 1301. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Lichtenstein, A.H.; Wu, J.H.; Appel, L.J.; Creager, M.A.; Kris-Etherton, P.M.; Miller, M.; Rimm, E.B.; Rudel, L.L.; Robinson, J.G.; et al. Dietary fats and cardiovascular disease: A presidential advisory from the American Heart Association. Circulation 2017, 136, e1–e23. [Google Scholar] [CrossRef] [PubMed]
- Unger, A.L.; Torres-Gonzalez, M.; Kraft, J. Dairy fat consumption and the risk of metabolic syndrome: An examination of the saturated fatty acids in dairy. Nutrients 2019, 11, 2200. [Google Scholar] [CrossRef] [PubMed]
- Babu, A.S.; Veluswamy, S.K.; Arena, R.; Guazzi, M.; Lavie, C.J. Virgin coconut oil and its potential cardioprotective effects. Postgrad. Med. 2014, 126, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Boateng, L.; Ansong, R.; Owusu, W.; Steiner-Asiedu, M. Coconut oil and palm oil’s role in nutrition, health and national development: A review. Ghana Med. J. 2016, 50, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Eyres, L.; Eyres, M.F.; Chisholm, A.; Brown, R.C. Coconut oil consumption and cardiovascular risk factors in humans. Nutr. Rev. 2016, 74, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Sibal, L.; Agarwal, S.; Home, P.; Boger, R. The role of asymmetric dimethylarginine (ADMA) in endothelial dysfunction and cardiovascular disease. Curr. Cardiol. Rev. 2010, 6, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Palomo, I.; Contreras, A.; Alarcón, L.M.; Leiva, E.; Guzmán, L.; Mujica, V.; Icaza, G.; Díaz, N.; González, D.R.; Moore-Carrasco, R. Elevated concentration of asymmetric dimethylarginine (ADMA) in individuals with metabolic syndrome. Nitric. Oxide 2011, 24, 224–228. [Google Scholar] [CrossRef]
- Engeli, S.; Tsikas, D.; Lehmann, A.; Böhnke, J.; Haas, V.; Strauß, A.; Janke, J.; Gorzelniak, K.; Luft, F.; Jordan, J. Influence of dietary fat ingestion on asymmetrical dimethylarginine in lean and obese human subjects. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Nikooei, P.; Hosseinzadeh-Attar, M.J.; Asghari, S.; Norouzy, A.; Yaseri, M.; Vasheghani-Farahani, A. Effects of virgin coconut oil consumption on metabolic syndrome components and asymmetric dimethylarginine: A randomized controlled clinical trial. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, I.; Biswas, K.; Bandyopadhyay, U.; Banerjee, R.K. Turmeric and curcumin: Biological actions and medicinal applications. Curr. Sci. 2004, 44–53. [Google Scholar]
- Hasanzadeh, S.; Read, M.I.; Bland, A.R.; Majeed, M.; Jamialahmadi, T.; Sahebkar, A. Curcumin: An inflammasome silencer. Pharmacol. Res. 2020, 159, 104921. [Google Scholar] [CrossRef] [PubMed]
- Alidadi, M.; Sahebkar, A.; Eslami, S.; Vakilian, F.; Jarahi, L.; Alinezhad-Namaghi, M.; Arabi, S.M.; Vakili, S.; Tohidinezhad, F.; Nikooiyan, Y.; et al. The Effect of Curcumin Supplementation on Pulse Wave Velocity in Patients with Metabolic Syndrome: A Randomized, Double-Blind, Placebo-Controlled Trial. Adv. Exp. Med. Biol. 2021, 1308, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Panahi, Y.; Ahmadi, Y.; Teymouri, M.; Johnston, T.P.; Sahebkar, A. Curcumin as a potential candidate for treating hyperlipidemia: A review of cellular and metabolic mechanisms. J. Cell Physiol. 2018, 233, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Ejaz, A.; Wu, D.; Kwan, P.; Meydani, M. Curcumin inhibits adipogenesis in 3T3-L1 adipocytes and angiogenesis and obesity in C57/BL mice. J. Nutr. 2009, 139, 919–925. [Google Scholar] [CrossRef]
- Giglio, R.V.; Patti, A.M.; Cicero, A.; Lippi, G.; Rizzo, M.; Toth, P.P.; Banach, M. Polyphenols: Potential Use in the Prevention and Treatment of Cardiovascular Diseases. Curr Pharm Des. 2018, 24, 239–258. [Google Scholar] [CrossRef]
- Fleenor, B.S.; Sindler, A.L.; Marvi, N.K.; Howell, K.L.; Zigler, M.L.; Yoshizawa, M.; Seals, D.R. Curcumin ameliorates arterial dysfunction and oxidative stress with aging. Exp. Gerontol. 2013, 48, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Velarde, G.P.; Choudhary, N.; Bravo-Jaimes, K.; Smotherman, C.; Sherazi, S.; Kraemer, D.F. Effect of atorvastatin on lipogenic, inflammatory and thrombogenic markers in women with the metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Rifai, N.; Pfeffer, M.A.; Sacks, F.M.; Moye, L.A.; Goldman, S.; Flaker, G.C.; Braunwald, E. Inflammation, pravastatin, and the risk of coronary events after myocardial infarction in patients with average cholesterol levels. Circulation 1998, 98, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Cannon, C.P.; Morrow, D.; Rifai, N.; Rose, L.M.; McCabe, C.H.; Pfeffer, M.A.; Braunwald, E. C-reactive protein levels and outcomes after statin therapy. N. Engl. J. Med. 2005, 352, 20–28. [Google Scholar] [CrossRef]
- Grosso, A.F.; de Oliveira, S.F.; Higuchi Mde, L.; Favarato, D.; Dallan, L.A.; da Luz, P.L. Synergistic anti-inflammatory effect: Simvastatin and pioglitazone reduce inflammatory markers of plasma and epicardial adipose tissue of coronary patients with metabolic syndrome. Diabetol. Metab. Syndr. 2014, 6, 47. [Google Scholar] [CrossRef]
- Shah, Z.; Pineda, C.; Kampfrath, T.; Maiseyeu, A.; Ying, Z.; Racoma, I.; Deiuliis, J.; Xu, X.; Sun, Q.; Moffatt-Bruce, S.; et al. Acute DPP-4 inhibition modulates vascular tone through GLP-1 independent pathways. Vascul. Pharmacol. 2011, 55, 2–9. [Google Scholar] [CrossRef]
- Ishii, M.; Shibata, R.; Kondo, K.; Kambara, T.; Shimizu, Y.; Tanigawa, T.; Bando, Y.K.; Nishimura, M.; Ouchi, N.; Murohara, T. Vildagliptin stimulates endothelial cell network formation and ischemia-induced revascularization via an endothelial nitric-oxide synthase-dependent mechanism. J. Biol. Chem. 2014, 289, 27235–27245. [Google Scholar] [CrossRef]
- Lamers, D.; Famulla, S.; Wronkowitz, N.; Hartwig, S.; Lehr, S.; Ouwens, D.M.; Eckardt, K.; Kaufman, J.M.; Ryden, M.; Müller, S.; et al. Dipeptidyl peptidase 4 is a novel adipokine potentially linking obesity to the metabolic syndrome. Diabetes 2011, 60, 1917–1925. [Google Scholar] [CrossRef]
- Sell, H.; Blüher, M.; Klöting, N.; Schlich, R.; Willems, M.; Ruppe, F.; Knoefel, W.T.; Dietrich, A.; Fielding, B.A.; Arner, P.; et al. Adipose dipeptidyl peptidase-4 and obesity: Correlation with insulin resistance and depot-specific release from adipose tissue in vivo and in vitro. Diabetes Care 2013, 36, 4083–4090. [Google Scholar] [CrossRef]
- Prakash, S.; Rai, U.; Kosuru, R.; Tiwari, V.; Singh, S. Amelioration of diet-induced metabolic syndrome and fatty liver with sitagliptin via regulation of adipose tissue inflammation and hepatic Adiponectin/AMPK levels in mice. Biochimie 2020, 168, 198–209. [Google Scholar] [CrossRef]
- Cicek, F.A.; Tokcaer-Keskin, Z.; Ozcinar, E.; Bozkus, Y.; Akcali, K.C.; Turan, B. Di-peptidyl peptidase-4 inhibitor sitagliptin protects vascular function in metabolic syndrome: Possible role of epigenetic regulation. Mol. Biol. Rep. 2014, 41, 4853–4863. [Google Scholar] [CrossRef]
- Rizzo, M.; Nikolic, D.; Patti, A.M.; Mannina, C.; Montalto, G.; McAdams, B.S.; Rizvi, A.A.; Cosentino, F. GLP-1 receptor agonists and reduction of cardiometabolic risk: Potential underlying mechanisms. Biochim. Biophys Acta Mol. Basis Dis. 2018, 1864, 2814–2821. [Google Scholar] [CrossRef] [PubMed]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Meier, J.J. GLP-1 receptor agonists in the treatment of type 2 diabetes—State-of-the-art. Mol Metab. 2021, 46, 101102. [Google Scholar] [CrossRef]
- Orchard, T.J.; Temprosa, M.; Goldberg, R.; Haffner, S.; Ratner, R.; Marcovina, S.; Fowler, S.; Diabetes Prevention Program Research Group. The effect of metformin and intensive lifestyle intervention on the metabolic syndrome: The Diabetes Prevention Program randomized trial. Ann. Intern. Med. 2005, 142, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, A.A.; Stoian, A.P.; Rizzo, M. Metabolic Syndrome: From Molecular Mechanisms to Novel Therapies. Int. J. Mol. Sci. 2021, 22, 10038. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).