Radiofrequency Ablation of Indeterminate Thyroid Nodules: The First North American Comparative Analysis
Abstract
1. Introduction
2. Results
2.1. Study Population
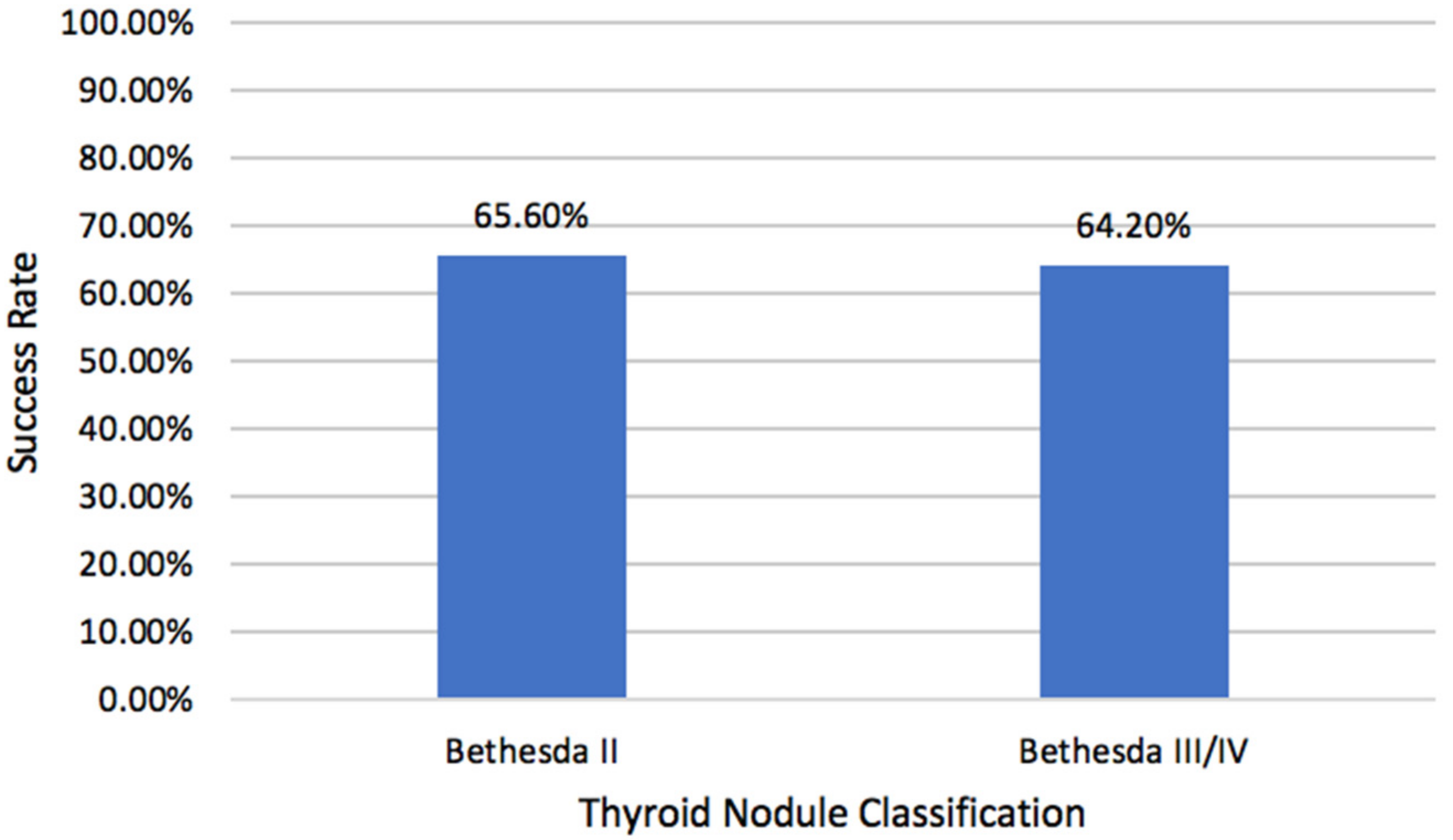
2.2. Volume Reduction Rates
2.3. Complications
2.4. Nodular Regrowth
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Recruited Cohort
4.3. Definitions
4.4. RFA Procedure
4.5. Postoperative Evaluation
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dean, D.S.; Gharib, H. Fine-Needle Aspiration Biopsy of the Thyroid Gland. Endotext Internet 2015, 118, 282–289. [Google Scholar]
- Bongiovanni, M.; Spitale, A.; Faquin, W.C.; Mazzucchelli, L.; Baloch, Z.W. The Bethesda System for Reporting Thyroid Cytopathology: A Meta-Analysis. Acta Cytol. 2012, 56, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Angell, T.E.; Vyas, C.M.; Barletta, J.A.; Cibas, E.S.; Cho, N.L.; Doherty, G.M.; Gawande, A.A.; Howitt, B.E.; Krane, J.F.; Marqusee, E. Reasons Associated with Total Thyroidectomy as Initial Surgical Management of an Indeterminate Thyroid Nodule. Ann. Surg. Oncol. 2018, 25, 1410–1417. [Google Scholar] [CrossRef]
- Almquist, M.; Muth, A. Surgical Management of Cytologically Indeterminate Thyroid Nodules. Gland Surg. 2019, 8, S105. [Google Scholar] [CrossRef] [PubMed]
- Weiss, A.; Parina, R.P.; Tang, J.A.; Brumund, K.T.; Chang, D.C.; Bouvet, M. Outcomes of Thyroidectomy from a Large California State Database. Am. J. Surg. 2015, 210, 1170–1177. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Baek, J.H.; Lim, H.K.; Ahn, H.S.; Baek, S.M.; Choi, Y.J.; Choi, Y.J.; Chung, S.R.; Ha, E.J.; Hahn, S.Y. 2017 Thyroid Radiofrequency Ablation Guideline: Korean Society of Thyroid Radiology. Korean J. Radiol. 2018, 19, 632–655. [Google Scholar] [CrossRef]
- Gharib, H.; Hegedüs, L.; Pacella, C.M.; Baek, J.H.; Papini, E. Nonsurgical, Image-Guided, Minimally Invasive Therapy for Thyroid Nodules. J. Clin. Endocrinol. Metab. 2013, 98, 3949–3957. [Google Scholar] [CrossRef]
- Papini, E.; Pacella, C.M.; Hegedus, L. Diagnosis of Endocrine Disease: Thyroid Ultrasound (US) and US-Assisted Procedures: From the Shadows into an Array of Applications. Eur. J. Endocrinol. 2014, 170, R133–R146. [Google Scholar] [CrossRef]
- Park, K.W.; Shin, J.H.; Han, B.-K.; Ko, E.Y.; Chung, J.H. Inoperable Symptomatic Recurrent Thyroid Cancers: Preliminary Result of Radiofrequency Ablation. Ann. Surg. Oncol. 2011, 18, 2564–2568. [Google Scholar] [CrossRef]
- Baek, J.H.; Kim, Y.S.; Sung, J.Y.; Choi, H.; Lee, J.H. Locoregional Control of Metastatic Well-Differentiated Thyroid Cancer by Ultrasound-Guided Radiofrequency Ablation. Am. J. Roentgenol. 2011, 197, W331–W336. [Google Scholar] [CrossRef] [PubMed]
- Van Dijk, S.P.; Coerts, H.I.; Gunput, S.T.; Van Velsen, E.F.; Medici, M.; Moelker, A.; Peeters, R.P.; Verhoef, C.; Van Ginhoven, T.M. Assessment of Radiofrequency Ablation for Papillary Microcarcinoma of the Thyroid: A Systematic Review and Meta-Analysis. JAMA Otolaryngol. Neck Surg. 2022, 148, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.M.; Sung, J.Y.; Baek, J.H.; Na, D.G.; Kim, J.; Yoo, H.; Lee, D.; Whan Choi, D. Radiofrequency Ablation of Small Follicular Neoplasms: Initial Clinical Outcomes. Int. J. Hyperth. 2017, 33, 931–937. [Google Scholar] [CrossRef]
- Mansur, A.; Garg, T.; Shrigiriwar, A.; Etezadi, V.; Georgiades, C.; Habibollahi, P.; Huber, T.C.; Camacho, J.C.; Nour, S.G.; Sag, A.A. Image-Guided Percutaneous Ablation for Primary and Metastatic Tumors. Diagnostics 2022, 12, 1300. [Google Scholar] [CrossRef] [PubMed]
- Bisceglia, A.; Rossetto, R.; Garberoglio, S.; Franzin, A.; Cerato, A.; Maletta, F.; Papotti, M.G.; Ghigo, E.; Pagano, L.; Maccario, M. Predictor Analysis in Radiofrequency Ablation of Benign Thyroid Nodules: A Single Center Experience. Front. Endocrinol. 2021, 12, 638880. [Google Scholar] [CrossRef]
- Ahn, H.S.; Kim, S.J.; Park, S.H.; Seo, M. Radiofrequency Ablation of Benign Thyroid Nodules: Evaluation of the Treatment Efficacy Using Ultrasonography. Ultrasonography 2016, 35, 244. [Google Scholar] [CrossRef] [PubMed]
- Vuong, N.L.; Dinh, L.Q.; Bang, H.T.; Thuy, T.T.M.; Bac, N.H.; Vy, T.T. Radiofrequency Ablation for Benign Thyroid Nodules: 1-Year Follow-up in 184 Patients. World J. Surg. 2019, 43, 2447–2453. [Google Scholar] [CrossRef]
- Cibas, E.S.; Ali, S.Z. The 2017 Bethesda System for Reporting Thyroid Cytopathology. Thyroid 2017, 27, 1341–1346. [Google Scholar] [CrossRef]
- Ho, A.S.; Sarti, E.E.; Jain, K.S.; Wang, H.; Nixon, I.J.; Shaha, A.R.; Shah, J.P.; Kraus, D.H.; Ghossein, R.; Fish, S.A. Malignancy Rate in Thyroid Nodules Classified as Bethesda Category III (AUS/FLUS). Thyroid 2014, 24, 832–839. [Google Scholar] [CrossRef]
- Gharib, H.; Papini, E.; Paschke, R.; Duick, D.S.; Valcavi, R.; Hegedüs, L.; Vitti, P. American Association of Clinical Endocrinologists, Associazione Medici Endocrinologi, and European Thyroid Association Medical Guidelines for Clinical Practice for the Diagnosis and Management of Thyroid Nodules: Executive Summary of Recommendations. J. Endocrinol. Investig. 2010, 33, 287–291. [Google Scholar] [CrossRef]
- Che, Y.; Jin, S.; Shi, C.; Wang, L.; Zhang, X.; Li, Y.; Baek, J. Treatment of Benign Thyroid Nodules: Comparison of Surgery with Radiofrequency Ablation. Am. J. Neuroradiol. 2015, 36, 1321–1325. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Luo, Y.; Tang, J.; Yang, M.; Li, J.; Zhang, Y.; Zhang, M. Ultrasound-Guided Radiofrequency Ablation for Papillary Thyroid Microcarcinoma: A Retrospective Analysis of 198 Patients. Int. J. Hyperth. 2020, 37, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Kandil, E.; Omar, M.; Aboueisha, M.; Attia, A.S.; Ali, K.M.; RF, A.A.; Issa, P.P.; Wolfe, S.; Omari, S.; Buti, Y. Efficacy and Safety of Radiofrequency Ablation of Thyroid Nodules: A Multi-Institutional Prospective Cohort Study. Ann. Surg. 2022, 276, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-C.; Tung, Y.-C.; Chang, Y.-H.; Luo, S.-D.; Chiang, P.-L.; Huang, S.C.; Chen, W.-C.; Chou, C.-K.; Su, Y.-Y.; Chen, W.-C. Radiofrequency Ablation for Treatment of Thyroid Follicular Neoplasm with Low SUV in PET/CT Study. Int. J. Hyperth. 2021, 38, 963–969. [Google Scholar] [CrossRef]
- Yoshida, Y.; Horiuchi, K.; Okamoto, T. Patients’ View on the Management of Papillary Thyroid Microcarcinoma: Active Surveillance or Surgery. Thyroid 2020, 30, 681–687. [Google Scholar] [PubMed]
- Dobrinja, C.; Bernardi, S.; Fabris, B.; Eramo, R.; Makovac, P.; Bazzocchi, G.; Piscopello, L.; Barro, E.; de Manzini, N.; Bonazza, D. Surgical and Pathological Changes after Radiofrequency Ablation of Thyroid Nodules. Int. J. Endocrinol. 2015, 2015, 576576. [Google Scholar] [CrossRef]
- Yan, L.; Luo, Y.; Xie, F.; Zhang, M.; Xiao, J. Residual Vital Ratio: Predicting Regrowth after Radiofrequency Ablation for Benign Thyroid Nodules. Int. J. Hyperth. 2020, 37, 1139–1148. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, S.; Giudici, F.; Cesareo, R.; Antonelli, G.; Cavallaro, M.; Deandrea, M.; Giusti, M.; Mormile, A.; Negro, R.; Palermo, A. Five-Year Results of Radiofrequency and Laser Ablation of Benign Thyroid Nodules: A Multicenter Study from the Italian Minimally Invasive Treatments of the Thyroid Group. Thyroid 2020, 30, 1759–1770. [Google Scholar] [CrossRef]
- Bergenfelz, A.; Kanngiesser, V.; Zielke, A.; Nies, C.; Rothmund, M. Conventional Bilateral Cervical Exploration versus Open Minimally Invasive Parathyroidectomy under Local Anaesthesia for Primary Hyperparathyroidism. J. Br. Surg. 2005, 92, 190–197. [Google Scholar] [CrossRef]
- Baek, J.H.; Lee, J.H.; Sung, J.Y.; Bae, J.-I.; Kim, K.T.; Sim, J.; Baek, S.M.; Kim, Y.; Shin, J.H.; Park, J.S. Complications Encountered in the Treatment of Benign Thyroid Nodules with US-Guided Radiofrequency Ablation: A Multicenter Study. Radiology 2012, 262, 335–342. [Google Scholar] [CrossRef]
- Schalch, M.S.; Costa, A.C.N.; de Souza, R.P.; Guerra, F.L.B.; Guerreiro, R.; Cicco, R.D. Radiofrequency Ablation of Thyroid Nodules: Prospective Cost-Effectiveness Analysis in Comparison to Conventional Thyroidectomy. Arch. Endocrinol. Metab. 2021, 65, 752–757. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yin, J.; Hu, C.; Ye, Q.; Wang, P.; Huang, P. Comparison of Ultrasound Guided Percutaneous Radiofrequency Ablation and Open Thyroidectomy in the Treatment of Low-Risk Papillary Thyroid Microcarcinoma: A Propensity Score Matching Study. Clin. Hemorheol. Microcirc. 2021, 80, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cibas, E.S.; Ali, S.Z. The Bethesda System for Reporting Thyroid Cytopathology. Thyroid 2009, 19, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- Mauri, G.; Pacella, C.M.; Papini, E.; Solbiati, L.; Goldberg, S.N.; Ahmed, M.; Sconfienza, L.M. Image-Guided Thyroid Ablation: Proposal for Standardization of Terminology and Reporting Criteria. Thyroid 2019, 29, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, H.; Santhanam, P.; Russell, J.O. Radiofrequency Ablation and Thyroid Nodules: Updated Systematic Review. Endocrine 2021, 72, 619–632. [Google Scholar] [CrossRef] [PubMed]
- Hadedeya, D.; Attia, A.S.; Shihabi, A.N.; Omar, M.; Shama, M.; Kandil, E. Technique and Procedural Aspects of Radiofrequency Ablation of Thyroid Nodules. Curr. Otorhinolaryngol. Rep. 2021, 9, 200–206. [Google Scholar] [CrossRef]
- Baek, J.H.; Jeong, H.J.; Kim, Y.S.; Kwak, M.S.; Lee, D. Radiofrequency Ablation for an Autonomously Functioning Thyroid Nodule. Thyroid 2008, 18, 675–676. [Google Scholar] [CrossRef]

| Characteristics | Levels | Total | Bethesda III and IV Nodules | Bethesda II Nodules | p-Value |
|---|---|---|---|---|---|
| Number | 178 | 53 | 125 | ||
| Demographic data | |||||
| Age | Median (IQR) | 64 (53–69) | 63 (56–68) | 65 (52.5–70) | 0.49 |
| <55 years | 48 (27) | 12 (22.6) | 36 (28.8) | 0.39 | |
| ≥55 years | 130 (73) | 41 (77.4) | 89 (71.2) | ||
| Gender | Female | 128 (71.9) | 30 (56.6) | 98 (78.4) | 0.003 |
| Male | 50 (28.1) | 23 (43.4) | 27 (21.6) | ||
| Race | African American | 100 (56.2) | 28 (52.8) | 72 (57.6) | 0.82 |
| White | 71 (39.9) | 23 (43.4) | 48 (38.4) | ||
| BMI | Median (IQR) | 30 (26.7–34.3) | 30.5 (27.5–34.5) | 29.8 (26.4–34.3) | 0.38 |
| Baseline sonographic features | |||||
| Nodule maximum diameter | Median (IQR) | 2.4 (1.5–4) | 2.5 (1.5–4.3) | 2.3 (1.4–3.7) | 0.47 |
| Baseline Volume | Median (IQR) | 1.8 (1.1–2.9) | 1.9 (1.1–3.2) | 1.7 (1.1–2.7) | 0.45 |
| Composition | Solid | 5 (2.8) | 3 (5.7) | 2 (1.6) | 0.31 |
| Cystic | 157 (88.2) | 46 (86.8) | 111 (88.8) | ||
| Mixed | 16 (9) | 4 (7.5) | 12 (9.6) | ||
| Echogenicity | Hypoechoic | 33 (21.3) | 11 (23.4) | 22 (20.4) | 0.74 |
| Isoechoic | 121 (78.1) | 36 (76.6) | 85 (78.7) | ||
| Hyperechoic | 1 (0.6) | 0 (0) | 1 (0.9) | ||
| Vascularity | Grade 0 | 11 (7.9) | 1 (2.3) | 10 (10.3) | 0.054 |
| Grade 1 | 46 (32.9) | 11 (25.6) | 35 (36.1) | ||
| Grade 2 | 53 (37.9) | 23 (53.5) | 30 (30.9) | ||
| Grade 3 | 30 (21.4) | 8 (18.6) | 22 (22.7) | ||
| Elastography | Soft | 7 (5.3) | 4 (10) | 3 (3.2) | 0.27 |
| Mixed | 96 (72.2) | 27 (67.5) | 69 (74.2) | ||
| Stiff | 30 (22.6) | 9 (22.5) | 21 (22.6) | ||
| Calcifications | No Calcifications | 91 (58.7) | 29 (61.7) | 62 (57.4) | 0.25 |
| Microcalcifications | 51 (32.9) | 12 (25.5) | 39 (36.1) | ||
| Macrocalcifications | 13 (8.4) | 6 (12.8) | 7 (6.5) | ||
| Laboratory data | |||||
| Baseline TSH uIU/mL | Median (IQR) | 1.3 (0.7–1.9) | 1.3 (0.8–1.9) | 1.3 (0.7–2) | 0.67 |
| Post procedural TSH uIU/mL | Median (IQR) | 1.2 (0.7–1.8) | 1.1 (0.7–1.8) | 1.2 (0.8–1.8) | 0.58 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Issa, P.P.; Omar, M.; Issa, C.P.; Buti, Y.; Hussein, M.; Aboueisha, M.; Abdelhady, A.; Shama, M.; Lee, G.S.; Toraih, E.; et al. Radiofrequency Ablation of Indeterminate Thyroid Nodules: The First North American Comparative Analysis. Int. J. Mol. Sci. 2022, 23, 11493. https://doi.org/10.3390/ijms231911493
Issa PP, Omar M, Issa CP, Buti Y, Hussein M, Aboueisha M, Abdelhady A, Shama M, Lee GS, Toraih E, et al. Radiofrequency Ablation of Indeterminate Thyroid Nodules: The First North American Comparative Analysis. International Journal of Molecular Sciences. 2022; 23(19):11493. https://doi.org/10.3390/ijms231911493
Chicago/Turabian StyleIssa, Peter P., Mahmoud Omar, Chad P. Issa, Yusef Buti, Mohammad Hussein, Mohamed Aboueisha, Ali Abdelhady, Mohamed Shama, Grace S. Lee, Eman Toraih, and et al. 2022. "Radiofrequency Ablation of Indeterminate Thyroid Nodules: The First North American Comparative Analysis" International Journal of Molecular Sciences 23, no. 19: 11493. https://doi.org/10.3390/ijms231911493
APA StyleIssa, P. P., Omar, M., Issa, C. P., Buti, Y., Hussein, M., Aboueisha, M., Abdelhady, A., Shama, M., Lee, G. S., Toraih, E., & Kandil, E. (2022). Radiofrequency Ablation of Indeterminate Thyroid Nodules: The First North American Comparative Analysis. International Journal of Molecular Sciences, 23(19), 11493. https://doi.org/10.3390/ijms231911493






