Abstract
Pattern recognition receptors (PRRs) play a crucial role in inducing inflammatory responses; they recognize pathogen-associated molecular patterns, damage-associated molecular patterns, and environmental factors. Nucleotide-binding oligomerization domain-leucine-rich repeat-containing receptors (NLRs) are part of the PRR family; they form a large multiple-protein complex called the inflammasome in the cytosol. In mammals, the inflammasome consists of an NLR, used as a sensor molecule, apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) as an adaptor protein, and pro-caspase1 (Casp1). Inflammasome activation induces Casp1 activation, promoting the maturation of proinflammatory cytokines, such as interleukin (IL)-1β and IL-18, and the induction of inflammatory cell death called pyroptosis via gasdermin D cleavage in mammals. Inflammasome activation and pyroptosis in mammals play important roles in protecting the host from pathogen infection. Recently, numerous inflammasome-related genes in teleosts have been identified, and their conservation and/or differentiation between their expression in mammals and teleosts have also been elucidated. In this review, we summarize the current knowledge of the molecular structure and machinery of the inflammasomes and the ASC-spec to induce pyroptosis; moreover, we explore the protective role of the inflammasome against pathogenic infection in teleosts.
1. Introduction
Inflammatory responses play a crucial role in the innate immune system to protect the host body from pathogens or other environmental factors. Pattern recognition systems act as inducers of inflammation [1,2]. For activation of the pattern recognition system, pattern recognition receptors (PRRs) recognize pathogen-associated molecular patterns (PAMPs) (e.g., lipopolysaccharide (LPS) and flagellin), damage-associated molecular patterns (DAMPs) (e.g., high mobility group box 1 (HMGB1), adenosine triphosphate (ATP)), or even environmental factors (e.g., particulate silica [3] and aluminum salt [4]) [1,2,5,6]. In the host cell cytosol, nucleotide-binding oligomerization domain (NOD)-leucine-rich repeats (LRR)-containing receptors (NLRs), part of the PRR family, recognize PAMPs, DAMPs, and environmental factors while forming a multiple-protein complex, called the “inflammasome” [1,7,8,9]. The formation and activation of the inflammasomes induce precursor inflammatory caspases (Casps), such as pro-Casp1, that activate Casp1, which then leads to non-apoptotic cell death, called “pyroptosis” [10,11,12,13]. Moreover, Casp1 induces pyroptosis and triggers precursor inflammatory cytokines to activate interleukin (IL)-1β and IL-18 [13,14,15]. These cytokines are secreted into the extracellular space through the pores formed after pyroptosis; furthermore, inflammation protected the host body from pathogens and environmental factors [14,15,16]. In this review, we discuss the activation of the inflammasome and the induction of pyroptosis in teleosts; moreover, we emphasize the potential of inflammasome activation against pathogens and environmental factors.
2. Inflammasome Activation to Induce Pyroptosis
2.1. Generic Structure of Inflammasome and Mechanisms of Pyroptosis
The inflammasome consists of an NLR (as a sensor protein), an ASC (apoptosis-associated speck-like protein containing a caspase recruitment domain (CARD), also known as pycard or the target of methylation-induced silencing-1 (TMS-1) as an adaptor protein), and pro-Casp1 (an inflammatory caspase) [16,17]. Inflammasome-forming NLRs have an N-terminal effector domain (i.e., pyrin domain (PYD) or CARD) that binds to ASC, a central fish-specific NACHT-associated domain and NACHT domain, and a C-terminal LRR motif that recognizes PAMPs, DAMPs, or environmental factors as ligands [6,9]. Several NLRs recruit the adaptor protein ASC (containing PYD and CARD) and pro-Casp1 to form a multiple-protein complex [16,17,18,19]. After recruitment of ASC and pro-Casp1, pro-Casp1 is self-proteolyzed into its active-form, Casp1 [13]. Casp1 is one of the proteases that cleaves the precursor inflammatory cytokines (i.e., pro-IL-1β and pro-IL-18) into their activated forms IL-1β and IL-18 [13,14,20]. Casp1 also cleaves gasdermin (GSDM) family proteins (i.e., GSDMA, -C, -D, and -E) at the N-terminus of GSDM, which then forms a pore in the cell membrane. Extracellular water then flows into the cell through the GSDM pore; thus, the cell expands. Eventually, the cell membrane is broken, leading to lytic cell death, known as pyroptosis [21,22]; moreover, at this point, cytosolic inflammatory molecules (including IL-1β, IL-18, HMGB1, and ATP) are released into the extracellular space [10,23,24,25]. In mammals, GSDMD induces pyroptosis in a Casp1-dependent manner [26]; however, there is no gsdmd in the fish genome. Current reports demonstrate that GSDME plays a key role in fish pyroptosis, as GSDMD does in mammals [27,28,29]. In zebrafish, CaspA (also known as CaspyA and Caspy) and CaspB (also known as CaspyB and Caspy2) [30], which are orthologs of Casp1, cleave the proinflammatory cytokines and two GSDMEs [27,29].
2.2. The Composition for Inflammasome and Apoptosis-Associated Speck-Like Protein Containing a Caspase Recruitment Domain Formation
2.2.1. Inflammasome Formation
NLRs are components of the inflammasome, and they play a crucial role in recognizing PAMPs, DAMPs, and environmental factors [9]. In mammals, NLRs containing PYD (NLRP1), NLRP3, NLRP12, and CARD (NLRC4) are key sensory molecules in inflammasomes [13,31,32,33,34]. However, in teleosts, only NLRP1 and NLRP3 in zebrafish and NLRP3 in the Japanese flounder have been identified as inflammasome-forming NLRs [27,35,36]. Interestingly, Japanese medaka and zebrafish have many NLRs that belong to NLRC3 and NLRP12 families (67 in Japanese medaka and 43 in zebrafish) in the genome database, although most lack PYD or CARD, which are important in inflammasome formation [37] (Figure 1). In the phylogenetic analysis, Japanese medaka NLRP12 (NLRP12 -3), zebrafish NLRP3, and three zebrafish NLRC3-like (i.e., NLRC3-like 1, NLRC3-4, and NLRC3-7) have PYD, which is divergent from the teleost NLRC3 group (Figure 1). Furthermore, in mammals, NLRC3 consists of only NACHT and LRR. However, mammals also have PYD, DD, FISNA, RING, PRY, and SPRY, in addition to the NACHT and LRR found in the Japanese medaka and NLRC3 and NLRP12 families found in zebrafish, which have branched from the mammalian NLRC3 group.
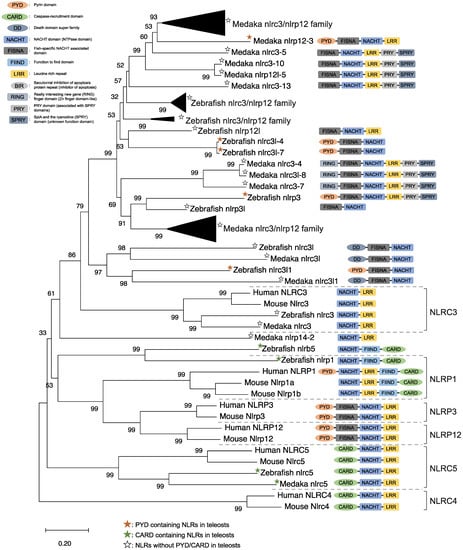
Figure 1.
Phylogenetic relationship of the nucleotide-binding oligomerization domain-leucine-rich repeat-containing receptor family in mammals (human and mouse) and fish (Japanese medaka and zebrafish). The tree was constructed using the MEGAX and a neighbor-joining method with 1000 bootstrap replications. The black triangle shapes show collection of the clusters.
In mice, NLRC3 inhibits TLR signaling by suppressing the signaling adaptor of TRAF6 alongside the transcription factor NF-κB [38]. However, NLRP3, which is clustered with both the zebrafish and Japanese medaka NLRC3 and NLRP12 families (Figure 1), activates the inflammatory response as an inflammasome in zebrafish. Additionally, the Japanese flounder NLRC3 involved extracellular ATP-mediated inflammatory responses [39], while Nile tilapia NLRC3 induced NF-κB activity in mammalian cells [40]. Therefore, both the NLRC3 and NLRP12 families in teleosts, which have branched from mammalian NLRC3 (Figure 1), may undergo a variety of unique evolutionary paths and have different functions from mammalian NLRCs. Furthermore, some NLRC3, NLRP12, and NLRP3 in the Japanese medaka and zebrafish conserve PRY and SPRY domains (also known as B30.2 domain), whereas these domains are not conserved in mammalian NLRs [37] (Figure 1). In zebrafish NLRP3, the B30.2 domain did not influence CaspA or CaspB activities; however, both PYD and NACHT influenced CaspA or CaspB activities [27]. According to the domain structures and phylogenetic analysis, there could be uniquely evolved inflammasome-forming NLRs with similar functions in mammals.
ASC is an adaptor protein of the inflammasome, which is recruited after NLRs recognize ligands (Figure 2). The ASC has two functional domains, PYD and CARD, and has been identified in many fish species, including zebrafish and Japanese medaka [41,42,43,44,45,46,47]. ASC binds to NLRPs via PYD–PYD interactions in mammals [48]. Moreover, in zebrafish and Japanese flounder, NLRP3 and ASC are co-localized in the cytosol, and they are detected as small spot signals under a microscope [27,36]. Furthermore, deletion of PYD in the ASC is not co-localized with NLRP3, and CARD deletion does not influence co-localization in either zebrafish or Japanese flounder [27,36]. Therefore, NLRP3 may bind to ASC through PYD–PYD interactions in teleosts and mammals. NLRP1 has two functional domains in humans: the N-terminal PYD and C-terminal CARD domains, which are important in inflammasome formation (Figure 2). However, zebrafish NLRP1 only has a C-terminal CARD, similar to mice [31,35] (Figure 1). The N-terminal PYD of NLRP1 has autolytic activity in function to find domain (FIIND), and it inhibits the formation of the NLRP1 inflammasome in humans [31,32]. As a result, the important inflammasome-forming domain in humans, like in mice, is CARD. In mammals, NLRP1 can recruit ASC pro-Casp1 via CARD–CARD interactions [31] (Figure 2). In contrast, zebrafish NLRP1 recruits only ASC via the CARD–CARD interaction; subsequently, the ASC recruits pro-CaspA or pro-CaspB [35] (Figure 2). The difference in the NLRP1 recruitment molecules between mammals and zebrafish is thought to be related to the structure of Casp1. The differences in the structure of the aforementioned Casp1 proteins are described in the following section. In zebrafish, NLRP1 is co-localized with ASC during spot formation in the cytosol. However, the PYD-deleted ASC is not entirely co-localized with NLRP3 and oligomerizes during filament formation. In contrast, the CARD-deleted ASC does not show any co-localization with NLRP1 in zebrafish [35]. Consequently, zebrafish NLRP1 may bind to ASC via a CARD–CARD interaction (Figure 2). In a recent study, there were three replicated ASCs in Japanese medaka, all of which had PYD and CARD [42]. It is unclear whether these ASCs also bind to NLRP family members, which needs to be investigated in the future.
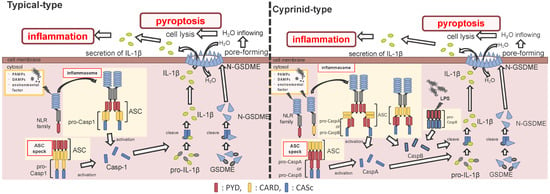
Figure 2.
Differences between the cyprinids and other fish species in their inflammasome activation and pyroptosis pathways. In the most fish, the nucleotide-binding oligomerization domain-leucine-rich repeat-containing receptor (NLR) family recognizes the ligands, and bind to apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) and pro-Caspase1 (Casp1) via pyrin domain (PYD)-PYD and caspase recruitment domain (CARD)-CARD interactions, thus forming the inflammasome. However, in cyprinids, ligands recognize the NLR family and construct two types of inflammasomes: in the first, the NLR family binds to ASC and pro-CaspA/B via CARD–CARD and PYD–PYD interactions, respectively. The NLR family interacts with ASC via the PYD–PYD interaction, and the ASC oligomerization occurs via CARD–CARD (called ASC core). Then, the pro-CaspA/B binds to the ASC core via the PYD–PYD interaction. After activation, the Casp1/A/B undergoes self-proteolysis, and the Casp1 matures interleukin-1β, inducing pyroptosis through gasdermin-E cleavage in all fish, including cyprinids. In addition, the cyprinid’s CaspB works as a non-canonical inflammasome activator, similar to mammalian Casp4/5/11. The pro-CaspB directly recognizes liposaccharides and thus activates by itself.
After the interaction between NLRs and ASC, ASC then recruits pro-Casp1, and together, they form the inflammasome (Figure 2). In all vertebrates, excluding cyprinid fish, pro-Casp1 has two functional domains, CARD and caspase consensus (CASc) (Table 1). Pro-Casp1 is self-proteolyzed into the active-form Casp1 after it binds with ASC, mutually catalyzing other pro-Casp1s [49,50]. Japanese flounder pro-Casp1 was also activated by NLRP3 [36]. In contrast, cyprinids pro-CaspA and pro-CaspyB possess PYD and CASc domains instead of CARD; moreover, zebrafish pro-CaspA and-B are co-localized with NLRP1, NLRP3, and ASC in the spot formation [27,35]. Therefore, in teleosts, pro-Casp1 is activated via NLRP1 or NLRP3 inflammasome formation.

Table 1.
Structures of typical and cyprinid-types pro-Caspase 1 in vertebrates.
To determine the similarity between ASC-binding domains in the vertebrate Casp1 families, the CARD domains of typical Casp1s were compared to the PYD domain in the cyprinid-types (Figure S1). In the alignment and WebLogo analyses, 41 amino acid residues (Met1, Ala2, Asp3, Lys7, Leu9, Arg13, Phe16, Val20, Ile25, Leu28, Leu29, Asp30, Leu32, Leu33, Glu34, Val37, Leu38, Asn39, Glu42, Glu44, Glu50, Asn51, Asp56, Ala58, Arg59, Leu61, Ile62, Asp63, Val65, Lys68, Gly69, Ala72, Ile77, Asp84, Leu87, Leu91, Gly92, Leu93, Thr95, His96, and Ile97) in the general teleost CARD well conserved to those of tetrapods; however, only 8 amino acid residues (Ile25, Leu28, Val37, Asp56, Gly70, Thr93, His94, and Ile95) in the cyprinid PYDs are conserved within the tetrapod CARDs. Furthermore, the 30th Asp (D) residue, related to the interaction of the CARD part of the ASC [49], is conserved in both tetrapod and general teleost CARDs, whereas this conservation does not exist in the cyprinid PYD (Figure S1). Consequently, the ASC-binding domain in the cyprinids showed a unique structure compared to those of the other vertebrates.
2.2.2. Apoptosis-Associated Speck-Like Protein Containing a Caspase Recruitment Domain Formation
ASC is not only an inflammasome adaptor molecule; it also forms a speck, called ASC speck (also known as ASC pyroptosome), which is an oligomerized ASC that can also activate Casp1 [51,52,53,54]. In mammals, LPS or monosodium urete (MSU) induces ASC speck formation, then the ASC speck recruits and activates Casp1, induces IL-1β maturation, and triggers pyroptosis [52]. In zebrafish, ASC speck formation was induced by ASC overexpression or via the induction of inflammation with CuSO4 stimulation in vivo, after which the endogenous ASC recruits to the overexpressing ASC to form the speck [55]. Moreover, ASC overexpression induced speck formation in goldfish and turbot [43,47]. During overexpression of ASC, the PYD-deleted ASC demonstrated filament formation, and the CARD-deleted ASC diffused (not co-localized) into the cytosol of zebrafish and turbot [41,47]. In accordance with these results, the PYD domain of ASC could be the key to ASC speck formation in teleosts. The zebrafish ASC speck was co-localized with CaspA [41], suggesting that PYD-ASC interacts with PYD-CaspA (Figure 2).
2.3. The Mechanism of Pyroptosis via Caspase 1
2.3.1. Induction of Pyroptosis through Caspase 1
Pro-Casp1 has two functional domains, an ASC-binding domain (CARD or PYD) and a caspase consensus (CASc) domain (Table 1). The CASc domain consists of p10 and p20 subunits, which are cleaved by self-proteolysis [56]. Then, the p10 and p20 subunits construct heterotetramers and play a role in converting pro-IL-1β, pro-IL18, or GSDMD to IL-1β, IL-18, or N-terminal GSDMD in mammals [22,57]. In seabass, pro-Casp1 can self-proteolyze into the active-form Casp1 (p10 and p20) while leaving a p24 fragment [58]. During PAMP and Aeromonas hydrophila stimulation, p10 and p20 subunits have been found in rainbow trout [59]. These results indicate that pro-Casp1 is self-proteolyzed into the active-form Casp1, consisting of the p10 and p20 subunits in teleosts and mammals.
2.3.2. Induction of Pyroptosis in Fish
The N-terminus of GSDME is key in the induction of pyroptosis via its capabilities in pore formation on the cell membrane [60] (Figure 2). In the tongue sole, the CARD-deleted pro-Casp1 could cleave GSDME; thus, the N-terminus of GSDME induced pyroptosis through pore formation on the cell membrane [28]. In zebrafish, there are two types of GSDMEs (i.e., GSDMEa and GSDMEb), with a stronger induced pyroptotic cell death via GSDMEb-induced CaspB activation [27,29]. The mature forms of CaspB, which are associated with GSDME cleavage, occur in the presence of both p10 and p20 subunits [27]. In this case, the N-terminus of GSDME forms a pore on the cell membrane, which induces pyroptosis, and both the p10 and p20 subunits in the activated Casp1 are necessary for cleavage of GSDME in teleosts. In humans, Casp3 cleaves GSDME and induces pyroptosis [60]. Interestingly, in the WebLogo analysis, there is a consensus motif sequence “DMPD” at the Casp3 cleavage site of the mammalian GSDME, whereas teleosts have a different consensus motif sequence, “FEVD”, at the Casp1 cleavage site [28]. Casp1, Casp3, and Casp7 were recently shown to cleave GSDME in the tongue sole [28]. However, additional investigations for other factors that cleave GSDME and induce pyroptosis in teleosts are needed in the future.
2.3.3. Induction of Pyroptosis via Non-Canonical Inflammasome Activation in Cyprinids
In mammals, Casp4/5/11 belong to the Casp1 family, and they directly recognize cytosolic LPS and activate it into mature forms to induce pyroptosis through GSDMD cleavage [54,60,61]. This inflammasome-independent induction of pyroptosis via Casp activation is called non-canonical inflammasome activation [61]. There are no existing orthologs of the Casp4/5/11 in most fish. However, CaspB in cyprinids shows activity similar to that of mammalian Casp5 [35,62]. The zebrafish CaspB shares the highest homology with that of human Casp5, and it also has a specific enzyme activity of the Casp5 substrate [30]. Besides, CaspB does not interact with the ASC speck [35]. During hemolysin-overexpressing Edwardsiella piscicida infection, Casp5 activity-dependent pyroptotic cell death was observed in zebrafish fibroblast cells [62]. The cells stimulated with LPS and cholera toxin B subunit (CTB), which transports LPS into the cell cytosol, also showed Casp5 activity and pyroptotic cell death [62]. Furthermore, the PYD domain of CaspB directly binds to LPS, and CaspB forms oligomers and induces pyroptosis via GSDMEb cleavage during hemolysin-overexpressing E. piscicida or cytosolic LPS stimulation [62] (Figure 2). In contrast, oligomerization and activation of CaspB did not result in the overexpression of CaspB alone [35]. Therefore, hemolysin from bacterial or cytosolic LPS stimulation may be necessary for non-canonical inflammasome activation in cyprinids.
3. Inflammasome-Related Gene Expression and Its Activation by Stimuli
An inflammasome activation model using a stimulant is important for understanding the inflammasome activation mechanism in detail. In mammals, NLRP3 inflammasome activation requires two steps. The first step is promoting the expression of NLRP3 and other inflammasome-related genes (called the priming step), and the second step is the formation and activation of the NLRP3 inflammasome [63]. A commonly used method of NLRP3 inflammasome priming is LPS stimulation, which recognizes TLR4 and promotes NF-κB-mediated gene expression of NLRP3 and inflammasome-related genes [63]. Moreover, NLRP3 needs to be phosphorylated by JNK1, located downstream of TLR signals for priming [63]. Several fish species, including Asian seabass, Japanese flounder, Japanese pufferfish, miiuy croaker, and rainbow trout, demonstrated that the NLR family or inflammasome-related genes were upregulated by LPS stimulation (Table 2) [39,46,64,65,66,67,68]. However, TLR4 does not exist in most fish, with it only located in cyprinid fish; moreover, TLR does not activate NF-κB via LPS stimulation in cyprinids [69,70]. Additionally, it is still unclear whether NLR family phosphorylation in fish is caused by LPS stimulation. Therefore, it is necessary to explore a suitable stimulant for inflammasome priming in fish. However, Pam3CSk4, a TLR2 ligand, is already used as a non-canonical inflammasome priming reagent in zebrafish [62]. In zebrafish, Pam3CSk4-stimulated TLR2 signaling was similar to that in mammals [71]. However, NLR family phosphorylation mechanisms are still unknown. Other than these stimulants, PAMPs (i.e., Vibrio anguillarum DNA [72], MDP [72], poly(I:C) [39,46,65,66,67,72], zymosan [39,46], flagellin [73], and peptidoglycan (PGN) [67,73]), and environmental factors (i.e., particulate silica [74] and cadmium [75]) may be used as stimulants for inflammasome priming in fish (Table 2). Thereafter, inflammasome activation in fish is indexed by the formation of the ASC speck, activation of Casp1, cleavage of IL-1β or GSDM, and the induction of pyroptosis (i.e., propidium iodide (PI) staining and measurement of LDH release from the cytosol), similar to that in mammals. Moreover, nigericin is the most common inflammasome activation inducer in mammals and induces inflammasome activation indices [76]. Nigericin is a Streptomyces hygroscopicus-derived antibiotic that acts as a potassium ionophore. Potassium efflux occurs in the cell, and this efflux activates the NLRP3 inflammasome in mammals [76]. In Japanese flounder, nigericin promotes Casp1 activity and IL-1β cleavage [36]. In several species of fish, nigericin induces the expression of inflammasome-related gene expression [42,43,68] (Table 2). Therefore, nigericin may induce the expression of inflammasome-related genes as stimulants in other fish species. Furthermore, extracellular ATP acts as an inflammasome activation inducer via the P2X7 receptor (P2X7R), a plasma membrane potassium ion channel, inducing potassium efflux in the cytosol [77,78,79,80]. P2X7R has been identified in several fish species, including Japanese flounder [81]. Here, nigericin and extracellular ATP promote Casp1 activation, IL-1β cleavage, and the induction of pyroptosis [65]. Moreover, Casp1 is activated by extracellular ATP in the orange-spotted grouper [44]. These results suggest that inflammasome activation may be promoted by potassium efflux in fish as well as in mammals via nigericin or extracellular ATP factors. The inflammasome-related genes were upregulated with extracellular ATP stimulation in several species of fish [39,44,46] (Table 2). Thus, extracellular ATP may promote inflammasome activation as well as priming in fish. However, it is unclear whether potassium efflux occurs with nigericin or extracellular ATP stimulation, and which receptors sense this efflux in fish remains unclear. Therefore, it is necessary to further understand the relationship between the inflammasomes and potassium efflux in fish.

Table 2.
Inflammasome priming and activation by pathogenic infection or stimulation in fish.
4. Inflammasome Activation during Pathogenic Infection
Inflammasome activation helps to restrict pathogen replication, including in bacteria and viruses [19]. In mice, NLRP1B inflammasome activation plays a crucial role in the host’s defense against infection with the Gram-positive bacteria Bacillus anthracis [90] and the intracellular parasite Toxoplasma gondii [31]. The NLRP3 inflammasome is activated by a wide range of pathogens, including Gram-positive and Gram-negative bacteria (e.g., Staphylococcus aureus [91], Mycobacterium tuberculosis [92,93,94], E. tarda [95,96,97], Listeria monocytogenes [98]) [99], RNA and DNA viruses (e.g., influenza virus, adenovirus [100], SARS-CoV-2 [101]) [102], fungi (e.g., Candida albicans [103]), and parasites (e.g., Leishmania amazonensis [104]) in mammals. The relationship between the inflammasomes and E. piscicida (also known as E. tarda in mammals) and Aeromonas sp. infection have been previously investigated in mammals and teleosts.
Edwardsiella piscicida is an intracellular Gram-negative bacterium that causes edwardsiellosis [105]. In mammals, E. piscicida infection alone does not cause an inflammasome priming step. However, zebrafish nlrp1 and nlrp3 gene expression was induced by E. tarda [27,35], and the expression of the nlrp3 gene in Japanese flounder was upregulated during E. piscicida infection [27] (Table 2). Therefore, it is inferred that E. piscicida infection causes the inflammasome priming step in teleosts, unlike in mammals. The type III secretion system (T3SS) of E. piscicida induced both ASC and Casp1-dependent NLRP3 and NLRC4 inflammasome activation (i.e., Casp1 activation, IL-1β secretion, and pyroptosis) in mouse bone marrow-derived macrophages (BMDMs). However, the LPS-priming step was necessary for activation [95,97]. The E. piscicida virulence effector trxlp also activated the NLRC4 inflammasome in mouse BMDMs and the mouse macrophage-like cell line J774A.1 [96]. However, the E. piscicida type VI secretion system (T6SS) effector protein (EvpP) suppressed NLRP3 inflammasome activation via ASC oligomerization by inhibiting calcium ion-dependent JNK (c-Jun N-terminal kinase) signals in the J774A.1 cells [97]. Furthermore, EvpP inhibited NLRC4-mediated bacterial clearance in the mouse spleen and liver [97]. Additionally, the T3SS of E. piscicida promoted intracellular invasion and pyroptosis, and it suppressed bacterial replication in human epithelial cells, such as the HeLa cell line [106]. In Japanese flounder head kidney macrophages, E. piscicida T3SS promoted Casp1 activity, IL-1β maturation, and pyroptosis induction [36]. Conversely, EvpP suppressed inflammasome activation by inhibiting JNK signaling in zebrafish larvae; thus, E. piscicida T6SS contributed to the colonization of zebrafish larvae [84,106]. Moreover, the mortality rate during wild type E. piscicida infection is higher than those of either T3SS or T6SS deficient strains [106]. In zebrafish liver epithelial or fibroblast cells, hemolysin (EthA+)-overexpressing E. piscicida 0909I strains induced CaspB-dependent pyroptotic cell death [62]. Thus, E. piscicida virulence factors, especially T3SS, T6SS, and hemolysin, are related to fish canonical and non-canonical inflammasome activation. In teleosts, E. piscicida pathogenicity depends on both T3SS and T6SS. Furthermore, due to the relationship between the inflammasome and E. piscicida infection, it has been considered that the intracellular bacteria can proliferate intracellularly, employing a mechanism to suppress inflammasome activation and pyroptosis, such as in the case of E. piscicida T6SS. Clarifying the relationship between the fish inflammasome and E. piscicida T3SS and T6SS may lead to the future suppression of edwardsiellosis in aquaculture.
Aeromonas spp., such as A. hydrophila and A. veronii, are Gram-negative bacteria that cause gastroenteritis in mammals and act as opportunistic infectious diseases in fish. In mouse BMDMs, A. veronii aerolysin, a type of Aeromonas sp. hemolysin, and T3SS induced NLRP3-dependent Casp1 activation and pyroptosis [107]. Furthermore, A. hydrophila T3SS induced ASC-dependent NLRP3 and NLRC4 inflammasome activation (i.e., Casp1 activation, IL-1β cleavage, and pyroptosis) in BMDMs [108]. In teleosts, the gene expression of the striped murrel casp1 and Japanese medaka asc1 was upregulated during A. hydrophila infection [42,88]. In rainbow trout, Casp1 activity was promoted during LPS or zymosan priming after A. hydrophila infection [59]. In a recent study, pyroptotic-like cell death was promoted by A. hydrophila infection in Japanese medaka kidney cells; however, cell death was suppressed in ASC knockout (KO) medaka kidney cells [109]. Moreover, A. hydrophila burden was observed in the ASC-KO medaka compared to the wild type [109]. These results suggest that the inflammasome is probably primed and activated during A. hydrophila infection and inflammasome activity plays an important role in eliminating A. hydrophila in teleosts. Other than these pathogens, as well as bacterial infection (i.e., V. anguillarum [72], Bacillus subtilis [83], E. ictaluri [110], V. alginolyticus [67], S. aureus [67], Lactobacillus paracasei spp. paracasei [68], Aphanomyces invadans [88], Streptococcus iniae [88], and S. agalactiae [40]), viral infection (rhabdovirus spring viremia of carp virus (SVCV) [21]) can also induce the expression of inflammasome-related genes, Moreover, Salmonella enterica sv. Typhimurium [82] or Francisella noatunensis [85] promoted Casp1/CaspA activity in teleosts (Table 2).
5. Conclusions
In the last decade, the structure of inflammasomes, as well as the mechanism and role of inflammasome activation and pyroptosis during pathogenic infections, have been elucidated in mammals. Inflammasome and pyroptosis-related genes, such as the NLR family members (e.g., nlrp1, nlrp3), asc, casp1, and gsdme, have been identified in numerous fish species, and the functions of the molecules they encode are becoming clear. However, there are very few studies on inflammasome activation via these molecules in teleosts. This is because there may be multiple molecules with different structures or activities, depending on the fish species selected. It is also different among small fish model organisms, such as the differences between zebrafish and Japanese medaka (Figure 2). In particular, the structure of pro-Casp-1, which is the inflammasome effector molecule, differs between most fish and cyprinids. Furthermore, there is non-canonical inflammasome activation in the cyprinid, whereas there are no reports of this occurring in other fish species. Therefore, it is necessary to proceed with research, keeping in mind that it depends on the fish species. During bacterial infection, inflammasome activation may restrict pathogen replication in teleosts and mammals. A better understanding of the relationship between inflammasomes and pathogens in teleosts will lead to the development of new immunostimulants to suppress aquaculture diseases.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/ijms22094389/s1, Figure S1: Comparison of the caspase recruitment domain (CARD) and pyrin domain (PYD) of the pro-Caspase1 (Casp1) amino acid sequences.
Author Contributions
Conceptualization, N.M., T.K., M.S., J.-i.H.; visualization, N.M., J.-i.H.; investigation and resources, N.M., J.-i.H.; data curation, validation, and formal analysis, N.M., J.-i.H.; project administration, N.M., M.S., J.-i.H.; supervision, J.-i.H.; writing—original draft, N.M., T.K., J.-i.H.; writing—review and editing, T.K., M.S., J.-i.H.; funding acquisition, N.M., M.S., J.-i.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by a Grant-in-Aid for Scientific Research (A) and (B) (KAKENHI Grant numbers 17H01486 and 17H03863) from the Japan Society for the Promotion of Science (JSPS), Japan. This work was also supported by a Grant-in-Aid for JSPS Research Fellows (grant number 20J14958) from JSPS, Japan.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Publicly available datasets were analyzed in this study. This data can be found here: https://www.ncbi.nlm.nih.gov (accessed on 21 April 2021).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
References
- Kawai, T.; Akira, S. The roles of TLRs, RLRs and NLRs in pathogen recognition. Int. Immunol. 2009, 21, 317–337. [Google Scholar] [CrossRef]
- Takeuchi, O.; Akira, S. Pattern recognition receptors and inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef]
- Franchi, L.; Eigenbrod, T.; Núñez, G. Cutting Edge: TNF-α mediates sensitization to ATP and silica via the NLRP3 inflammasome in the absence of microbial stimulation. J. Immunol. 2009, 183, 792–796. [Google Scholar] [CrossRef]
- Franchi, L.; Núñez, G. The Nlrp3 inflammasome is critical for aluminium hydroxide-mediated IL-1beta secretion but dispensable for adjuvant activity. Eur. J. Immunol. 2008, 38, 2085–2089. [Google Scholar] [CrossRef]
- Brubaker, S.W.; Bonham, K.S.; Zanoni, I.; Kagan, J.C. Innate immune pattern recognition: A cell biological perspective. Annu. Rev. Immunol. 2015, 33, 257–290. [Google Scholar] [CrossRef]
- Sahoo, B.R. Structure of fish toll-like receptors (TLR) and NOD-like receptors (NLR). Int. J. Biol. Macromol. 2020, 161, 1602–1617. [Google Scholar] [CrossRef] [PubMed]
- Becker, C.E.; O’Neill, L.A.J. Inflammasomes in inflammatory disorders: The role of TLRs and their interactions with NLRs. Semin. Immunopathol. 2007, 29, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.K.; Wen, H.; Ting, J.P.-Y. The inflammasome NLRs in immunity, inflammation, and associated diseases. Annu. Rev. Immunol. 2011, 29, 707–835. [Google Scholar] [CrossRef] [PubMed]
- Netea, M.G.; Van de Veerdonk, F.L.; Kullberg, B.J.; Van der Meer, J.W.M.; Joosten, L.A.B. The role of NLRs and TLRs in the activation of the inflammasome. Expert Opin. Biol. Ther. 2008, 8, 1867–1872. [Google Scholar] [CrossRef] [PubMed]
- Bergsbaken, T.; Fink, S.L.; Cookson, B.T. Pyroptosis: Host cell death and inflammation. Nat. Rev. Microbiol. 2009, 7, 99–109. [Google Scholar] [CrossRef]
- Vande Walle, L.; Lamkanfi, M. Pyroptosis. Curr. Biol. 2016, 26, R568–R572. [Google Scholar] [CrossRef]
- Fink, S.L.; Cookson, B.T. Apoptosis, pyroptosis, and necrosis: Mechanistic description of dead and dying eukaryotic cells. Infect. Immun. 2005, 73, 1907–1916. [Google Scholar] [CrossRef] [PubMed]
- Franchi, L.; Eigenbrod, T.; Muñoz-Planillo, R.; Nuñez, G. The inflammasome: A caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nat. Immunol. 2009, 10, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, M.; Ceballos-Olvera, I.; Del Barrio, L.; Re, F. Role of the inflammasome, IL-1β, and IL-18 in bacterial infections. Sci. World J. 2011, 11, 2037–2050. [Google Scholar] [CrossRef] [PubMed]
- Netea, M.G.; Nold-Petry, C.A.; Nold, M.F.; Joosten, L.A.B.; Opitz, B.; Van Der Meer, J.H.M.; Van De Veerdonk, F.L.; Ferwerda, G.; Heinhuis, B.; Devesa, I.; et al. Differential requirement for the activation of the inflammasome for processing and release of IL-1β in monocytes and macrophages. Blood 2009, 113, 2324–2335. [Google Scholar] [CrossRef]
- Mariathasan, S.; Monack, D.M. Inflammasome adaptors and sensors: Intracellular regulators of infection and inflammation. Nat. Rev. Immunol. 2007, 7, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.; Wu, H. Structural mechanisms of inflammasome assembly. FEBS J. 2015, 282, 435–444. [Google Scholar] [CrossRef]
- Guo, H.; Callaway, J.B.; Ting, J.P. Inflammasomes: Mechanism of action, role in disease, and therapeutics. Nat. Med. 2015, 21, 677–687. [Google Scholar] [CrossRef]
- Ta, A.; Vanaja, S.K. Inflammasome activation and evasion by bacterial pathogens. Curr. Opin. Immunol. 2021, 68, 125–133. [Google Scholar] [CrossRef]
- Miller, L.S.; Pietras, E.M.; Uricchio, L.H.; Hirano, K.; Rao, S.; Lin, H.; O’Connell, R.M.; Iwakura, Y.; Cheung, A.L.; Cheng, G.; et al. Inflammasome-mediated production of IL-1beta is required for neutrophil recruitment against Staphylococcus aureus in vivo. J. Immunol. 2007, 179, 6933–6942. [Google Scholar] [CrossRef]
- Varela, M.; Romero, A.; Dios, S.; van der Vaart, M.; Figueras, A.; Meijer, A.H.; Novoa, B. Cellular visualization of macrophage pyroptosis and interleukin-1β release in a viral hemorrhagic infection in zebrafish larvae. J. Virol. 2014, 88, 12026–12040. [Google Scholar] [CrossRef]
- Shi, J.; Zhao, Y.; Wang, K.; Shi, X.; Wang, Y.; Huang, H.; Zhuang, Y.; Cai, T.; Wang, F.; Shao, F. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 2015, 526, 660–665. [Google Scholar] [CrossRef]
- Lopez-Castejon, G.; Brough, D. Understanding the mechanism of IL-1β secretion. Cytokine Growth Factor Rev. 2011, 22, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Ceballos-Olvera, I.; Sahoo, M.; Miller, M.A.; del Barrio, L.; Re, F. Inflammasome-dependent pyroptosis and IL-18 protect against Burkholderia pseudomallei lung infection while IL-1β is deleterious. PLoS Pathog. 2011, 7. [Google Scholar] [CrossRef] [PubMed]
- Willingham, S.B.; Allen, I.C.; Bergstralh, D.T.; Brickey, W.J.; Huang, M.T.H.; Taxman, D.J.; Duncan, J.A.; Ting, J.P.Y. NLRP3 (NALP3, cryopyrin) facilitates in vivo caspase-1 activation, necrosis, and HMGB1 release via inflammasome-dependent and -independent pathways. J. Immunol. 2009, 183, 2008–2015. [Google Scholar] [CrossRef]
- Vince, J.E.; Silke, J. The intersection of cell death and inflammasome activation. Cell. Mol. Life Sci. 2016, 73, 2349–2367. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Wang, Y.Y.; Shao, T.; Fan, D.D.; Lin, A.F.; Xiang, L.X.; Shao, J.Z. The zebrafish NLRP3 inflammasome has functional roles in ASC-dependent interleukin-1β maturation and gasdermin E-mediated pyroptosis. J. Biol. Chem. 2020, 295, 1120–1141. [Google Scholar] [CrossRef]
- Jiang, S.; Gu, H.; Zhao, Y.; Sun, L. Teleost gasdermin E is cleaved by caspase 1, 3, and 7 and induces pyroptosis. J. Immunol. 2019, 203, 1369–1382. [Google Scholar] [CrossRef]
- Wang, Z.; Gu, Z.; Hou, Q.; Chen, W.; Mu, D.; Zhang, Y.; Liu, Q.; Liu, Z.; Yang, D. Zebrafish GSDMEb cleavage-gated pyroptosis drives septic acute kidney injury in vivo. J. Immunol. 2020, 204, 1929–1942. [Google Scholar] [CrossRef]
- Masumoto, J.; Zhou, W.; Chen, F.F.; Su, F.; Kuwada, J.Y.; Hidaka, E.; Katsuyama, T.; Sagara, J.; Taniguchi, S.; Ngo-Hazelett, P.; et al. Caspy, a zebrafish caspase, activated by ASC oligomerization is required for pharyngeal arch development. J. Biol. Chem. 2003, 278, 4268–4276. [Google Scholar] [CrossRef]
- Chavarría-Smith, J.; Vance, R.E. The NLRP1 inflammasomes. Immunol. Rev. 2015, 265, 22–34. [Google Scholar] [CrossRef]
- Schroder, K.; Tschopp, J. The Inflammasomes. Cell 2010, 140, 821–832. [Google Scholar] [CrossRef] [PubMed]
- Cassel, S.L.; Joly, S.; Sutterwala, F.S. The NLRP3 inflammasome: A sensor of immune danger signals. Semin. Immunol. 2009, 21, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Tuladhar, S.; Kanneganti, T.D. NLRP12 in innate immunity and inflammation. Mol. Asp. Med. 2020, 76, 100887. [Google Scholar] [CrossRef]
- Li, J.; Gao, K.; Shao, T.; Fan, D.; Hu, C.; Sun, C.; Dong, W.; Lin, A.; Xiang, L.; Shao, J. Characterization of an NLRP1 inflammasome from zebrafish reveals a unique sequential activation mechanism underlying inflammatory caspases in ancient vertebrates. J. Immunol. 2018, 201, 1946–1966. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Ding, S.; Tan, J.; Yang, D.; Zhang, Y.; Liu, Q. Characterization of the Japanese flounder NLRP3 inflammasome in restricting Edwardsiella piscicida colonization in vivo. Fish Shellfish Immunol. 2020, 103, 169–180. [Google Scholar] [CrossRef]
- Chang, M.X.; Xiong, F.; Wu, X.M.; Hu, Y.W. The expanding and function of NLRC3 or NLRC3-like in teleost fish: Recent advances and novel insights. Dev. Comp. Immunol. 2021, 114, 103859. [Google Scholar] [CrossRef]
- Schneider, M.; Zimmermann, A.G.; Roberts, R.A.; Zhang, L.; Swanson, K.V.; Wen, H.; Davis, B.K.; Allen, I.C.; Holl, E.K.; Ye, Z.; et al. The innate immune sensor NLRC3 attenuates toll-like receptor signaling via modification of the signaling adaptor TRAF6 and transcription factor NF-κB. Nat. Immunol. 2012, 13, 823–831. [Google Scholar] [CrossRef]
- Li, S.; Chen, X.; Hao, G.; Geng, X.; Zhan, W.; Sun, J. Identification and characterization of a novel NOD-like receptor family CARD domain containing 3 gene in response to extracellular ATP stimulation and its role in regulating LPS-induced innate immune response in Japanese flounder (Paralichthys olivaceus). Fish Shellfish Immunol. 2016, 50, 79–90. [Google Scholar] [CrossRef]
- Gao, F.-Y.; Pang, J.-C.; Lu, M.-X.; Yang, X.-L.; Zhu, H.-P.; Ke, X.-L.; Liu, Z.-G.; Cao, J.-M.; Wang, M. Molecular characterization, expression and functional analysis of NOD1, NOD2 and NLRC3 in Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2018, 73, 207–219. [Google Scholar] [CrossRef]
- Li, Y.; Huang, Y.; Cao, X.; Yin, X.; Jin, X.; Liu, S.; Jiang, J.; Jiang, W.; Xiao, T.S.; Zhou, R.; et al. Functional and structural characterization of zebrafish ASC. FEBS J. 2018, 285, 2691–2707. [Google Scholar] [CrossRef]
- Morimoto, N.; Okamura, Y.; Kono, T.; Sakai, M.; Hikima, J. Characterization and expression analysis of tandemly-replicated asc genes in the Japanese medaka, Oryzias latipes. Dev. Comp. Immunol. 2020, 115, 103894. [Google Scholar] [CrossRef]
- Xie, J.; Belosevic, M. Functional characterization of apoptosis-associated speck-like protein (ASC) of the goldfish (Carassius auratus L.). Dev. Comp. Immunol. 2016, 65, 201–210. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Z.; Li, C.; Zhang, Y.; Wang, L.; Wei, J.; Qin, Q. Characterization of orange-spotted grouper (Epinephelus coioides) ASC and caspase-1 involved in extracellular ATP-mediated immune signaling in fish. Fish Shellfish Immunol. 2020, 97, 58–71. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, J.; Lao, H.; Yin, Z.; He, W.; Weng, S.; Yu, X.; Chan, S.M.; He, J. Molecular cloning and expression analysis of the ASC gene from mandarin fish and its regulation of NF-κB activation. Dev. Comp. Immunol. 2008, 32, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Chen, X.; Peng, W.; Hao, G.; Geng, X.; Zhan, W.; Sun, J. Cloning and characterization of apoptosis-associated speck-like protein containing a CARD domain (ASC) gene from Japanese flounder Paralichthys olivaceus. Fish Shellfish Immunol. 2016, 54, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Tan, J.; Wang, Z.; Zhang, Y.; Liu, Q.; Yang, D. Characterization of the inflammasome component SmASC in turbot (Scophthalmus maximus). Fish Shellfish Immunol. 2020, 100, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Moriya, M.; Taniguchi, S.; Wu, P.; Liepinsh, E.; Otting, G.; Sagara, J. Role of charged and hydrophobic residues in the oligomerization of the pyrin domain of ASC. Biochemistry 2005, 44, 575–583. [Google Scholar] [CrossRef]
- Kersse, K.; Lamkanfi, M.; Bertrand, M.J.M.; Berghe, T.V.; Vandenabeele, P. Interaction patches of procaspase-1 caspase recruitment domains (CARDs) are differently involved in procaspase-1 activation and receptor-interacting protein 2 (RIP2)-dependent nuclear factor κB signaling. J. Biol. Chem. 2011, 286, 35874–35882. [Google Scholar] [CrossRef] [PubMed]
- Srinivasula, S.M.; Poyet, J.L.; Razmara, M.; Datta, P.; Zhang, Z.; Alnemri, E.S. The pyrin-CARD protein ASC is an activating adaptor for caspase-1. J. Biol. Chem. 2002, 277, 21119–21122. [Google Scholar] [CrossRef]
- Hoss, F.; Rodriguez-Alcazar, J.F.; Latz, E. Assembly and regulation of ASC specks. Cell. Mol. Life Sci. 2017, 74, 1211–1229. [Google Scholar] [CrossRef] [PubMed]
- Hara, H.; Tsuchiya, K.; Kawamura, I.; Fang, R.; Hernandez-Cuellar, E.; Shen, Y.; Mizuguchi, J.; Schweighoffer, E.; Tybulewicz, V.; Mitsuyama, M. Phosphorylation of the adaptor ASC acts as a molecular switch that controls the formation of speck-like aggregates and inflammasome activity. Nat. Immunol. 2013, 14, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- De Alba, E. Structure, interactions and self-assembly of ASC-dependent inflammasomes. Arch. Biochem. Biophys. 2019, 670, 15–31. [Google Scholar] [CrossRef]
- Broz, P.; Dixit, V.M. Inflammasomes: Mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 2016, 16, 407–420. [Google Scholar] [CrossRef]
- Kuri, P.; Schieber, N.L.; Thumberger, T.; Wittbrodt, J.; Schwab, Y.; Leptin, M. Dynamics of ASC speck formation during skin inflammatory responses in vivo. bioRxiv 2017, 216. [Google Scholar] [CrossRef]
- Broz, P.; von Moltke, J.; Jones, J.W.; Vance, R.E.; Monack, D.M. Differential requirement for caspase-1 autoproteolysis in pathogen-induced cell death and cytokine processing. Cell Host Microbe 2010, 8, 471–483. [Google Scholar] [CrossRef]
- Keller, M.; Rüegg, A.; Werner, S.; Beer, H.D. Active caspase-1 is a regulator of unconventional protein secretion. Cell 2008, 132, 818–831. [Google Scholar] [CrossRef] [PubMed]
- Reis, M.I.R.; do Vale, A.; Pereira, P.J.B.; Azevedo, J.E.; dos Santos, N.M.S. Caspase-1 and IL-1β processing in a teleost fish. PLoS ONE 2012, 7, e50450. [Google Scholar] [CrossRef]
- Rojas, V.; Camus-Guerra, H.; Guzmán, F.; Mercado, L. Pro-inflammatory caspase-1 activation during the immune response in cells from rainbow trout Oncorhynchus mykiss (Walbaum 1792) challenged with pathogen-associated molecular patterns. J. Fish Dis. 2015, 38, 993–1003. [Google Scholar] [CrossRef]
- Tang, L.; Lu, C.; Zheng, G.; Burgering, B.M.T. Emerging insights on the role of gasdermins in infection and inflammatory diseases. Clin. Transl. Immunol. 2020, 9, e1186. [Google Scholar] [CrossRef]
- Winsor, N.; Krustev, C.; Bruce, J.; Philpott, D.J.; Girardin, S.E. Canonical and non-canonical inflammasomes in intestinal epithelial cells. Cell. Microbiol. 2019, 21, e13079. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Zheng, X.; Chen, S.; Wang, Z.; Xu, W.; Tan, J.; Hu, T.; Hou, M.; Wang, W.; Gu, Z.; et al. Sensing of cytosolic LPS through caspy2 pyrin domain mediates non-canonical inflammasome activation in zebrafish. Nat. Commun. 2018, 9, 3052. [Google Scholar] [CrossRef] [PubMed]
- Song, N.; Liu, Z.S.; Xue, W.; Bai, Z.F.; Wang, Q.Y.; Dai, J.; Liu, X.; Huang, Y.J.; Cai, H.; Zhan, X.Y.; et al. NLRP3 phosphorylation is an essential priming event for inflammasome activation. Mol. Cell 2017, 68, 185–197.e6. [Google Scholar] [CrossRef] [PubMed]
- Unajak, S.; Santos, M.D.; Hikima, J.; Jung, T.S.S.; Kondo, H.; Hirono, I.; Aoki, T. Molecular characterization, expression and functional analysis of a nuclear oligomerization domain proteins subfamily C (NLRC) in Japanese flounder (Paralichthys olivaceus). Fish Shellfish Immunol. 2011, 31, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Peng, W.; Li, J.; Hao, G.; Geng, X.; Sun, J. Characterization of Japanese flounder (Paralichthys olivaceus) caspase1 involved in extracellular ATP-mediated immune signaling in fish. Fish Shellfish Immunol. 2017, 67, 536–545. [Google Scholar] [CrossRef]
- Álvarez, C.A.; Ramírez-Cepeda, F.; Santana, P.; Torres, E.; Cortés, J.; Guzmán, F.; Schmitt, P.; Mercado, L. Insights into the diversity of NOD-like receptors: Identification and expression analysis of NLRC3, NLRC5 and NLRX1 in rainbow trout. Mol. Immunol. 2017, 87, 102–113. [Google Scholar] [CrossRef]
- Paria, A.; Deepika, A.; Sreedharan, K.; Makesh, M.; Chaudhari, A.; Purushothaman, C.S.; Thirunavukkarasu, A.R.; Rajendran, K.V. Identification of Nod like receptor C3 (NLRC3) in Asian seabass, Lates calcarifer: Characterisation, ontogeny and expression analysis after experimental infection and ligand stimulation. Fish Shellfish Immunol. 2016, 55, 602–612. [Google Scholar] [CrossRef]
- Biswas, G.; Bilen, S.; Kono, T.; Sakai, M.; Hikima, J. Inflammatory immune response by lipopolysaccharide-responsive nucleotide binding oligomerization domain (NOD)-like receptors in the Japanese pufferfish (Takifugu rubripes). Dev. Comp. Immunol. 2016, 55, 21–31. [Google Scholar] [CrossRef]
- Meijer, A.H.; Gabby Krens, S.F.; Medina Rodriguez, I.A.; He, S.; Bitter, W.; Snaar-Jagalska, B.E.; Spaink, H.P. Expression analysis of the toll-like receptor and TIR domain adaptor families of zebrafish. Mol. Immunol. 2004, 40, 773–783. [Google Scholar] [CrossRef]
- Sepulcre, M.P.; Alcaraz-Pérez, F.; López-Muñoz, A.; Roca, F.J.; Meseguer, J.; Cayuela, M.L.; Mulero, V. Evolution of lipopolysaccharide (LPS) recognition and signaling: Fish TLR4 does not recognize LPS and negatively regulates NF-kappaB activation. J. Immunol. 2009, 182, 1836–1845. [Google Scholar] [CrossRef]
- Yang, S.; Marín-Juez, R.; Meijer, A.H.; Spaink, H.P. Common and specific downstream signaling targets controlled by Tlr2 and Tlr5 innate immune signaling in zebrafish. BMC Genom. 2015, 16, 547. [Google Scholar] [CrossRef]
- López-Castejón, G.; Sepulcre, M.P.; Mulero, I.; Pelegrín, P.; Meseguer, J.; Mulero, V. Molecular and functional characterization of gilthead seabream Sparus aurata caspase-1: The first identification of an inflammatory caspase in fish. Mol. Immunol. 2008, 45, 49–57. [Google Scholar] [CrossRef]
- Li, T.; Shan, S.; Wang, L.; Yang, G.; Zhu, J. Identification of a fish-specific NOD-like receptor subfamily C (NLRC) gene from common carp (Cyprinus carpio L.): Characterization, ontogeny and expression analysis in response to immune stimulation. Fish Shellfish Immunol. 2018, 82, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, T.; Biswas, G.; Kono, T.; Sakai, M.; Hikima, J. Immune responses in the Japanese pufferfish (Takifugu rubripes) head kidney cells stimulated with particulate silica. Fish Shellfish Immunol. 2016, 49, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, Q.; Yin, H.; Li, S. Cadmium exposure induces pyroptosis of lymphocytes in carp pronephros and spleens by activating NLRP3. Ecotoxicol. Environ. Saf. 2020, 202, 110903. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Planillo, R.; Kuffa, P.; Martínez-Colón, G.; Smith, B.L.; Rajendiran, T.M.; Núñez, G. K+ efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate Matter. Immunity 2013, 38, 1142–1153. [Google Scholar] [CrossRef]
- Franchi, L.; Kanneganti, T.D.; Dubyak, G.R.; Núñez, G. Differential requirement of P2X7 receptor and intracellular K+ for caspase-1 activation induced by intracellular and extracellular bacteria. J. Biol. Chem. 2007, 282, 18810–18818. [Google Scholar] [CrossRef] [PubMed]
- Hewinson, J.; Moore, S.F.; Glover, C.; Watts, A.G.; MacKenzie, A.B. A key role for redox signaling in rapid P2X 7 receptor-induced IL-1 beta processing in human monocytes. J. Immunol. 2008, 180, 8410–8420. [Google Scholar] [CrossRef]
- Lister, M.F.; Sharkey, J.; Sawatzky, D.A.; Hodgkiss, J.P.; Davidson, D.J.; Rossi, A.G.; Finlayson, K. The role of the purinergic P2X7 receptor in inflammation. J. Inflamm. 2007, 4, 5. [Google Scholar] [CrossRef]
- Pelegrin, P.; Barroso-Gutierrez, C.; Surprenant, A. P2X 7 receptor differentially couples to distinct release pathways for IL-1β in mouse macrophage. J. Immunol. 2008, 180, 7147–7157. [Google Scholar] [CrossRef]
- Li, S.; Li, X.; Coddou, C.; Geng, X.; Wei, J.; Sun, J. Molecular characterization and expression analysis of ATP-gated P2X7 receptor involved in Japanese flounder (Paralichthys olivaceus) innate immune response. PLoS ONE 2014, 9, e96625. [Google Scholar] [CrossRef] [PubMed]
- Angosto, D.; López-Castejó, G.; López-Muñ, A.; Sepulcre, M.P.; Arizcun, M.; Meseguer, J.; Mulero, V. Evolution of inflammasome functions in vertebrates: Inflammasome and caspase-1 trigger fish macrophage cell death but are dispensable for the processing of IL-1β. Innate Immun. 2012, 18, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Cerezuela, R.; Meseguer, J.; Esteban, M.Á. Effects of dietary inulin, Bacillus subtilis and microalgae on intestinal gene expression in gilthead seabream (Sparus aurata L.). Fish Shellfish Immunol. 2013, 34, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Yang, D.; Wang, Z.; Zheng, X.; Zhang, Y.; Liu, Q. EvpP inhibits neutrophils recruitment via Jnk-caspy inflammasome signaling in vivo. Fish Shellfish Immunol. 2019, 92, 851–860. [Google Scholar] [CrossRef] [PubMed]
- Vojtech, L.N.; Scharping, N.; Woodson, J.C.; Hansen, J.D. Roles of inflammatory caspases during processing of zebrafish interleukin-1β in Francisella noatunensis infection. Infect. Immun. 2012, 80, 2878–2885. [Google Scholar] [CrossRef]
- Li, J.; Kong, L.; Gao, Y.; Wu, C.; Xu, T. Characterization of NLR-A subfamily members in miiuy croaker and comparative genomics revealed NLRX1 underwent duplication and lose in actinopterygii. Fish Shellfish Immunol. 2015, 47, 397–406. [Google Scholar] [CrossRef]
- Li, J.; Chu, Q.; Xu, T. A genome-wide survey of expansive NLR-C subfamily in miiuy croaker and characterization of the NLR-B30.2 genes. Dev. Comp. Immunol. 2016, 61, 116–125. [Google Scholar] [CrossRef]
- Kumaresan, V.; Ravichandran, G.; Nizam, F.; Dhayanithi, N.B.; Arasu, M.V.; Al-Dhabi, N.A.; Harikrishnan, R.; Arockiaraj, J. Multifunctional murrel caspase 1, 2, 3, 8 and 9: Conservation, uniqueness and their pathogen-induced expression pattern. Fish Shellfish Immunol. 2016, 49, 493–504. [Google Scholar] [CrossRef]
- Davis, J.M.; Clay, H.; Lewis, J.L.; Ghori, N.; Herbomel, P.; Ramakrishnan, L.; Masumoto, J.; Zhou, W.; Chen, F.F.; Su, F.; et al. Molecular characterization reveals involvement of four caspases in the antibacterial immunity of tongue sole (Cynoglossus semilaevis). Fish Shellfish Immunol. 2016, 7, 116–125. [Google Scholar] [CrossRef]
- Terra, J.K.; Cote, C.K.; France, B.; Jenkins, A.L.; Bozue, J.A.; Welkos, S.L.; LeVine, S.M.; Bradley, K.A. Cutting Edge: Resistance to Bacillus anthracis infection mediated by a lethal toxin sensitive allele of Nalp1b/Nlrp1b. J. Immunol. 2010, 184, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Kebaier, C.; Chamberland, R.R.; Allen, I.C.; Gao, X.; Broglie, P.M.; Hall, J.D.; Jania, C.; Doerschuk, C.M.; Tilley, S.L.; Duncan, J.A. Staphylococcus aureus α-hemolysin mediates virulence in a murine model of severe pneumonia through activation of the NLRP3 inflammasome. J. Infect. Dis. 2012, 205, 807–817. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Zhao, S.; Gao, X.; Wang, R.; Liu, J.; Zhou, X.; Zhou, Y. The roles of inflammasomes in host defense against Mycobacterium tuberculosis. Pathogens 2021, 10, 120. [Google Scholar] [CrossRef] [PubMed]
- McElvania Tekippe, E.; Allen, I.C.; Hulseberg, P.D.; Sullivan, J.T.; McCann, J.R.; Sandor, M.; Braunstein, M.; Ting, J.P.Y. Granuloma formation and host defense in chronic Mycobacterium tuberculosis infection requires PYCARD/ASC but not NLRP3 or caspase-1. PLoS ONE 2010, 5, e12320. [Google Scholar] [CrossRef]
- Kleinnijenhuis, J.; Joosten, L.A.B.; van de Veerdonk, F.L.; Savage, N.; van Crevel, R.; Kullberg, B.J.; van der Ven, A.; Ottenhfoff, T.H.M.; Dinarello, C.A.C.A.; van der Meer, J.W.M.; et al. Transcriptional and inflammasome-mediated pathways for the induction of IL-1β production by Mycobacterium tuberculosis. Eur. J. Immunol. 2009, 39, 1914–1922. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.X.; Lu, J.F.; Rolhion, N.; Holden, D.W.; Nie, P.; Zhou, Y.; Yu, X.J. Edwardsiella tarda-induced cytotoxicity depends on its type III secretion system and flagellin. Infect. Immun. 2014, 82, 3436–3445. [Google Scholar] [CrossRef]
- Xu, W.; Gu, Z.; Zhang, L.; Zhang, Y.; Liu, Q.; Yang, D. Edwardsiella piscicida virulence effector trxlp promotes the NLRC4 inflammasome activation during infection. Microb. Pathog. 2018, 123, 496–504. [Google Scholar] [CrossRef]
- Chen, H.; Yang, D.; Han, F.; Tan, J.; Zhang, L.; Xiao, J.; Zhang, Y.; Liu, Q. The bacterial T6SS effector EvpP prevents NLRP3 inflammasome activation by inhibiting the Ca2+-dependent MAPK-Jnk pathway. Cell Host Microbe 2017, 21, 47–58. [Google Scholar] [CrossRef]
- Kaphingst, K.A.; Persky, S.; Lachance, C. Listeria monocytogenes is sensed by NLRP3 and AIM2 inflammasome. Eur. J. Immunol. 2010, 40, 1545–1551. [Google Scholar] [CrossRef]
- Vladimer, G.I.; Marty-Rolx, R.; Ghosh, S.; Wang, D.; Lien, E. Inflammasomes and host defenses against bacterial infections. Curr. Opin. Microbiol. 2013, 16, 23–31. [Google Scholar] [CrossRef]
- Barlan, A.U.; Danthi, P.; Wiethoff, C.M. Lysosomal localization and mechanism of membrane penetration influence nonenveloped virus activation of the NLRP3 inflammasome. Virology 2011, 412, 306–314. [Google Scholar] [CrossRef]
- Rodrigues, T.S.; de Sá, K.S.G.; Ishimoto, A.Y.; Becerra, A.; Oliveira, S.; Almeida, L.; Gonçalves, A.V.; Perucello, D.B.; Andrade, W.A.; Castro, R.; et al. Inflammasomes are activated in response to SARS-CoV-2 infection and are associated with COVID-19 severity in patients. J. Exp. Med. 2020, 218, e20201707. [Google Scholar] [CrossRef]
- Lupfer, C.; Kanneganti, T.-D. The expanding role of NLRs in antiviral immunity. Immunol. Rev. 2013, 255, 13–24. [Google Scholar] [CrossRef]
- Lowes, D.J.; Hevener, K.E.; Peters, B.M. Second-generation antidiabetic sulfonylureas inhibit Candida albicans and candidalysin-mediated activation of the NLRP3 inflammasome. Antimicrob. Agents Chemother. 2020, 64, e01777-19. [Google Scholar] [CrossRef]
- De Carvalho, R.V.H.; Silva, A.L.N.; Santos, L.L.; Andrade, W.A.; de Sá, K.S.G.; Zamboni, D.S. Macrophage priming is dispensable for NLRP3 inflammasome activation and restriction of Leishmania amazonensis replication. J. Leukoc. Biol. 2019, 106, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, B.R.; Sahoo, P.K. Edwardsiellosis in fish: A brief review. J. Biosci. 2007, 32, 1331–1344. [Google Scholar] [CrossRef]
- Hu, T.; Chen, R.; Zhang, L.; Wang, Z.; Yang, D.; Zhang, Y.; Liu, X.; Liu, Q. Balanced role of T3SS and T6SS in contribution to the full virulence of Edwardsiella piscicida. Fish Shellfish Immunol. 2019, 93, 871–878. [Google Scholar] [CrossRef]
- McCoy, A.J.; Koizumi, Y.; Higa, N.; Suzuki, T. Differential regulation of caspase-1 activation via NLRP3/NLRC4 inflammasomes mediated by aerolysin and type III secretion system during Aeromonas veronii infection. J. Immunol. 2010, 185, 7077–7084. [Google Scholar] [CrossRef]
- McCoy, A.J.; Koizumi, Y.; Toma, C.; Higa, N.; Dixit, V.; Taniguchi, S.; Tschopp, J.; Suzuki, T. Cytotoxins of the human pathogen Aeromonas hydrophila trigger, via the NLRP3 inflammasome, caspase-1 activation in macrophages. Eur. J. Immunol. 2010, 40, 2797–2803. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, N.; Okamura, Y.; Maekawa, S.; Wang, H.; Aoki, T.; Kono, T.; Sakai, M.; Hikima, J. ASC-deficiency impairs host defense against Aeromonas hydrophila infection in Japanese medaka, Oryzias latipes. Fish Shellfish Immunol. 2020, 105, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Sha, Z.; Abernathy, J.W.; Wang, S.; Li, P.; Kucuktas, H.; Liu, H.; Peatman, E.; Liu, Z. NOD-like subfamily of the nucleotide-binding domain and leucine-rich repeat containing family receptors and their expression in channel catfish. Dev. Comp. Immunol. 2009, 33, 991–999. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).