Abstract
Human endogenous retroviruses (HERVs) are suggested to be involved in the development of certain diseases, especially cancers. To elucidate the function of HERV-K Env protein in cancers, an HERV-K env gene knockout (KO) in DLD-1 colorectal cancer cell lines was generated using the CRISPR-Cas9 system. Transcriptome analysis of HERV-K env KO cells using next-generation sequencing (NGS) was performed to identify the key genes associated with the function of HERV-K Env protein. The proliferation of HERV-K env KO cells was significantly reduced in in vitro culture as well as in in vivo nude mouse model. Tumorigenic characteristics, including migration, invasion, and tumor colonization, were also significantly reduced in HERV-K env KO cells. Whereas, they were enhanced in HERV-K env over-expressing DLD-1 cells. The expression of nuclear protein-1 (NUPR1), an ER-stress response factor that plays an important role in cell proliferation, migration, and reactive oxygen species (ROS) generation in cancer cells, significantly reduced in HERV-K env KO cells. ROS levels and ROS-related gene expression was also significantly reduced in HERV-K env KO cells. Cells transfected with NUPR1 siRNA (small interfering RNA) exhibited the same phenotype as HERV-K env KO cells. These results suggest that the HERV-K env gene affects tumorigenic characteristics, including cell proliferation, migration, and tumor colonization through NUPR1 related pathway.
1. Introduction
The vertebrate genome contains an endogenous retrovirus that inherited parts millions of years ago. Although approximately 8% of human chromosomal DNA consists of sequences derived from human endogenous retrovirus (HERV) fragments, most HERVs are currently inactive and non-infectious due to recombination, deletions, and mutations after insertion into the host genome [1,2].
Among the various HERVs, the well-known HERV-K (HML-2) group, comprises numerous human-specific proviruses that are transcriptionally active in the brain [3,4,5], encoding various former retroviral proteins and retrovirus-like particles [6,7]. HERV-K elements have been reported to be transcribed and expressed as proteins in certain diseases, including cancer. HERV-K has been reported to be highly related to cancer in various reports. HERV-K RNA levels are significantly upregulated in breast cancer, prostate cancer, melanoma, and ovarian cancer [8,9,10,11,12,13]. HERV-K hypomethylation in ovarian clear cell carcinoma has been reported to be associated with a poor prognosis and platinum resistance [14]. Additionally, HERV-K has also been suggested as a potential biomarker and immunotherapeutic target in cancer [15]. We previously reported that the expression of HERV-K Env protein was higher in various cancers than in normal tissues [16].
Colorectal cancer is the second most predicted cancer in 2020. Unlike other cancers, colorectal cancer has almost the same inventory ratio between male and female. In particular, it is a cancer that is gradually increasing as we move toward an aging society [17]. We found that the expression of HERV-K Env protein was specifically high in colorectal cancer compared with surrounding normal tissue. However, the role of HERV-K Env protein in colorectal cancer has not yet been reported.
Although it is clear that HERV-K elements are closely related to cancer, there are few studies on its mechanism of action. A recent study showed that shRNA mediated downregulation of HERV-K env RNA in pancreatic cancer cells showed decreased cell proliferation and tumor growth through the RAS-ERK-RSK pathway [18]. In this study, the HERV-K env gene in DLD-1 colorectal cancer was knocked out using the CRISPR-Cas9 system. RNA seq analysis revealed that NUPR-1, a new target of the HERV-K env gene, is linked with tumorigenic characteristics modulated by HERV-K env gene.
2. Results
2.1. Knockout of HERV-K env Gene in Human Colorectal Cancer Cell Lines
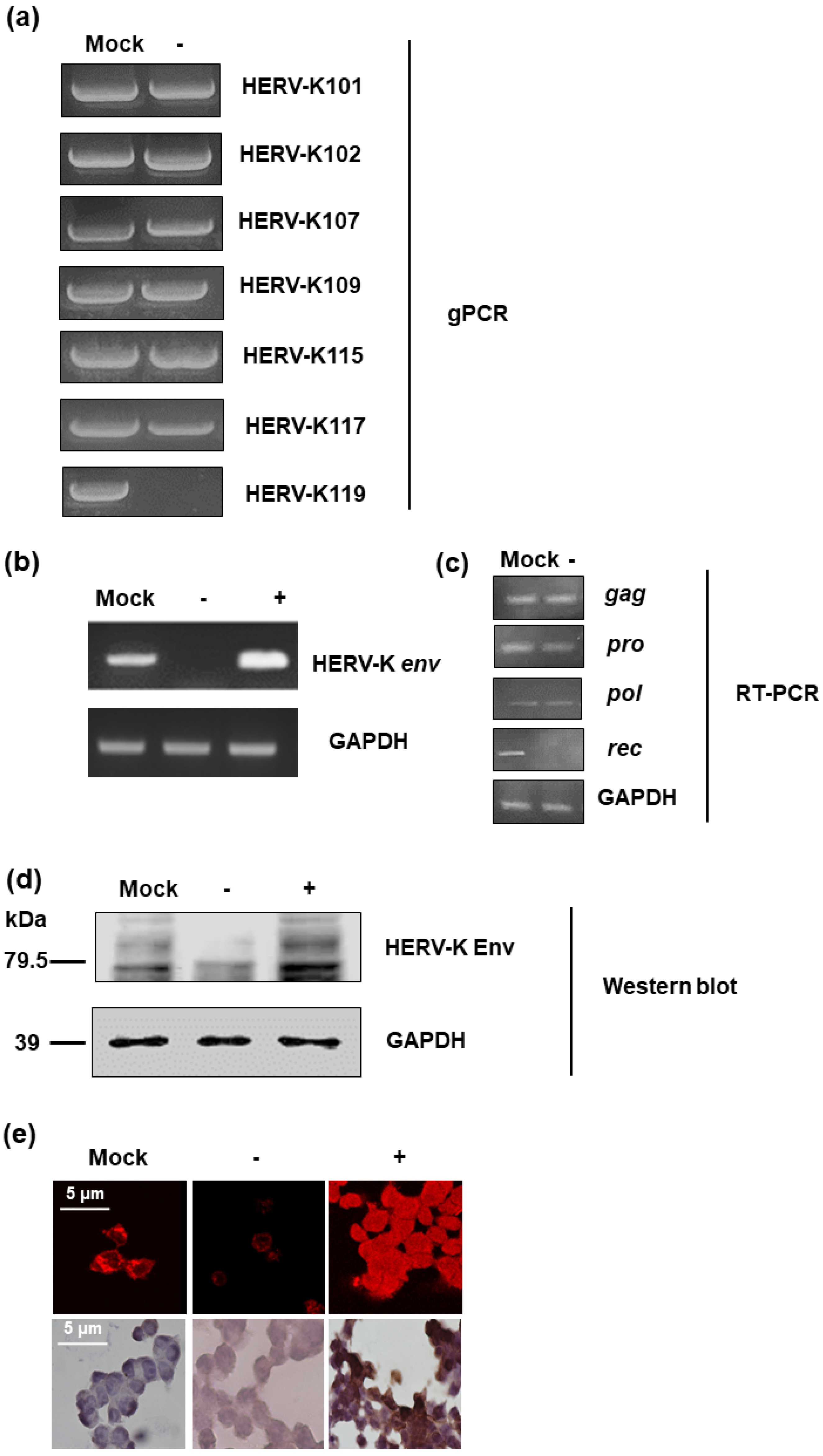
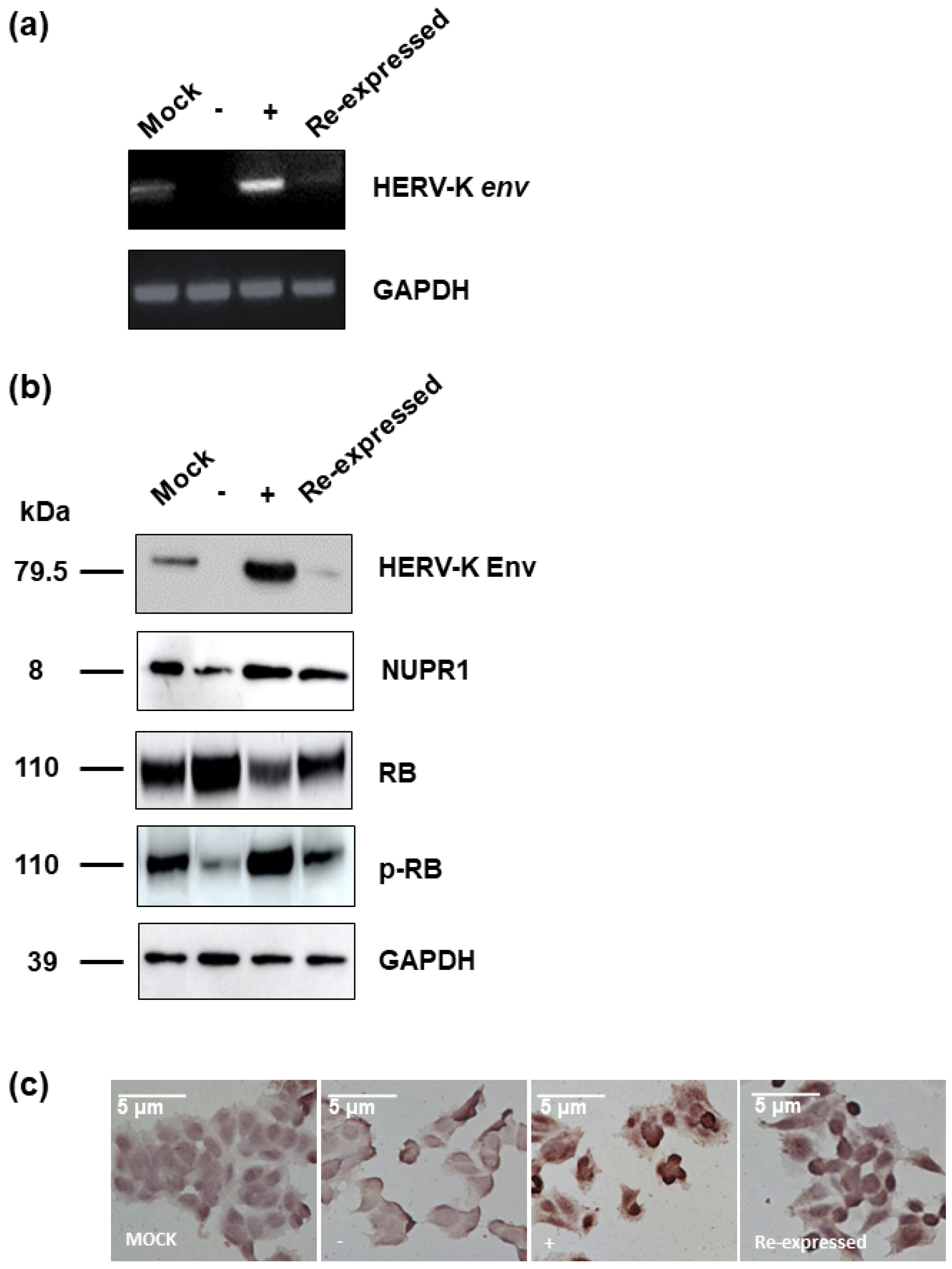
In order to identify the function of the HERV-K env gene in tumorigenic characteristics in colorectal cancer, HERV-K env knockout (KO) DLD-1 colorectal cancer cell lines were generated using the CRISPR-Cas9 gene editing system. Guide RNA was designed with the K119 region of the HERV-K env gene, which codes the most functional Env protein and selected HERV-K env KO stable clones. Genomic polymerase chain reaction (PCR) (Figure 1) showed that the HERV-K env gene in K119 was completely removed, whereas other derivatives at different loci remained. The detail information of HERV-K env genes in different loci was described at Table 1. Although only HERV-K119 region was deleted, the RT-PCR for HERV-K env gene showed that the RNA level of HERV-K env was dramatically reduced (Figure 1b). However, the expression of other HERV-K elements including gag, pro, and pol was not changed. The expression of rec which is very close to env was significantly reduce (Figure 1c). An HERV-K env overexpression system was also constructed to analyze the function by over-expression of HERV-K env or re-expressed of HERV-K env gene in HERV-K env KO cells. As shown in Figure 1b, HERV-K env gene overexpression was confirmed at RNA level. HERV-K Env protein expression was also analyzed by Western blot and immunostaining. As shown in Figure 1d, HERV-K Env protein levels were reduced in HERV-K env KO cells, whereas increased in HERV-K env over-expressing cells. Immunofluorescence (IF) and immunohistochemistry (IHC) also showed that HERV-K Env protein expression levels decreased in HERV-K env KO cells and increased in HERV-K env over-expressing cells. (Figure 1e).
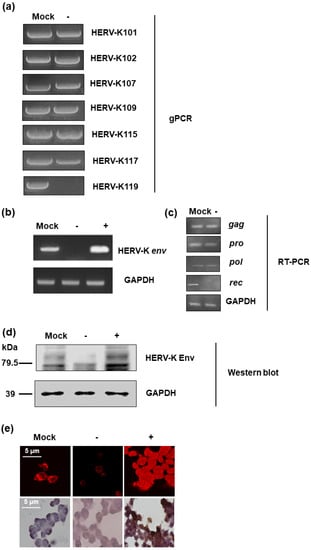
Figure 1.
Knockout of Human endogenous retroviruse (HERV)-K env gene in DLD-1 colorectal cancer cells. (a) Knockout of HERV-K env gene in HERV-K119 region. Genomic polymerase chain reaction (PCR) was performed for specific regions of HERV-K env derivatives. (b) Expression of HERV-K env RNA in HERV-K env knockout (KO) and over-expressing DLD-1 colorectal cancer cells. RT-PCR performed for general region of HERV-K env gene. (c) Expression of HERV-K119 gag, pro, pol, and rec genes in DLD-1 colorectal cancer cells (d) Expression of HERV-K Env protein in HERV-K env KO and over-expressing DLD-1 colorectal cancer cells. Western blot was performed to analyze the protein level of HERV-K Env. (e) Immunofluorescence and immunohistochemical staining of the HERV-K Env expression in colorectal cancer cells. HERV-K env gene in HERV-K119 region was completely deleted and the expression of RNA and protein level of HERV-K env gene was significantly reduced. The expression of glucose 6 phosphate dehydrogenase (GAPDH) was served as a loading control. (−: KO, +: over-expression).

Table 1.
Position and PCR primer information of HERV-K variants.
2.2. HERV-K env KO Reduced Tumorigenic Characteristics Including Proliferation, Invasion, Migration, and Tumor Colonization in DLD-1 Colorectal Cancer Cells
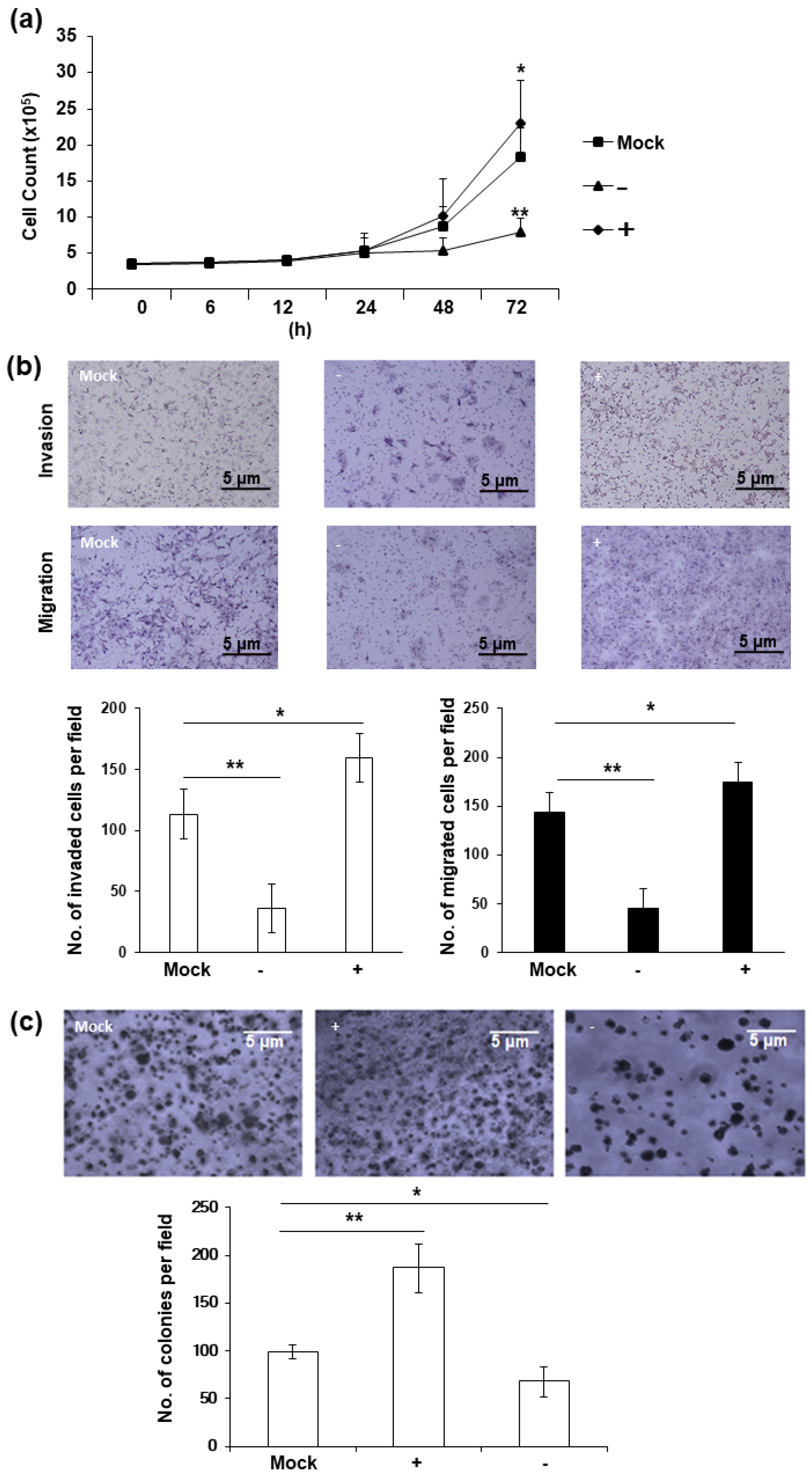
Phenotypical changes were observed in HERV-K env KO DLD-1 colorectal cancer cells. A significant reduction in cell proliferation is the most prominent characteristic of HERV-K env KO cells. As shown in Figure 2a, the cell growth was significantly reduced in HERV-K env KO cells, whereas increased in HERV-K env over-expressing cells compared with Mock cells. Cell invasion and migration were also significantly reduced in HERV-K env KO cells, but increased in HERV-K env over-expressing cells compared with Mock cells (Figure 2b). Additionally, tumor colonization on soft agar also showed that the number of tumor colonies increased in HERV-K env over-expressing cells and decreased in HERV-K env KO cells (Figure 2c). These results suggest that HERV-K env plays a critical role in tumorigenic characteristics in DLD-1 colorectal cancer cells.
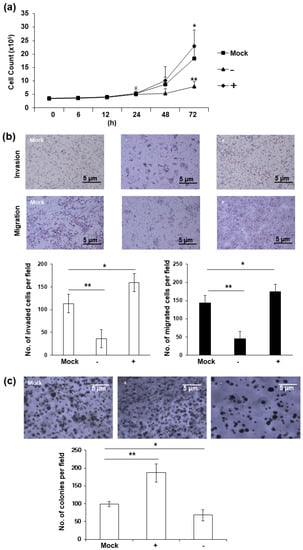
Figure 2.
HERV-K env KO reduced tumorigenic characteristics including proliferation, invasion, migration, and tumor colonization in DLD-1 colorectal cancer cells. (a) Cell proliferation of HERV-K env KO and over-expressing DLD-1 colorectal cancer cell. Cell proliferation was significantly reduced in HERV-K KO group and increased in over-expression groups of DLD-1 colorectal cancer cells. (b) Invasion and migration of HERV-K env KO and over-expressing DLD-1 colorectal cancer cell. Invasion and migration were significantly reduced in HERV-K KO group and increased in over-expression groups of DLD-1 colorectal cancer cells. (c) Soft-agar colony forming assay. Colony formation ability on soft agar was significantly reduced in HERV-K KO group and increased in over-expression groups of DLD-1 colorectal cancer cells. * p < 0.05, ** p < 0.01. (−: KO, +: over-expression).
2.3. HERV-K env KO Reduced Tumor Growth in the In Vivo Xenograft Models
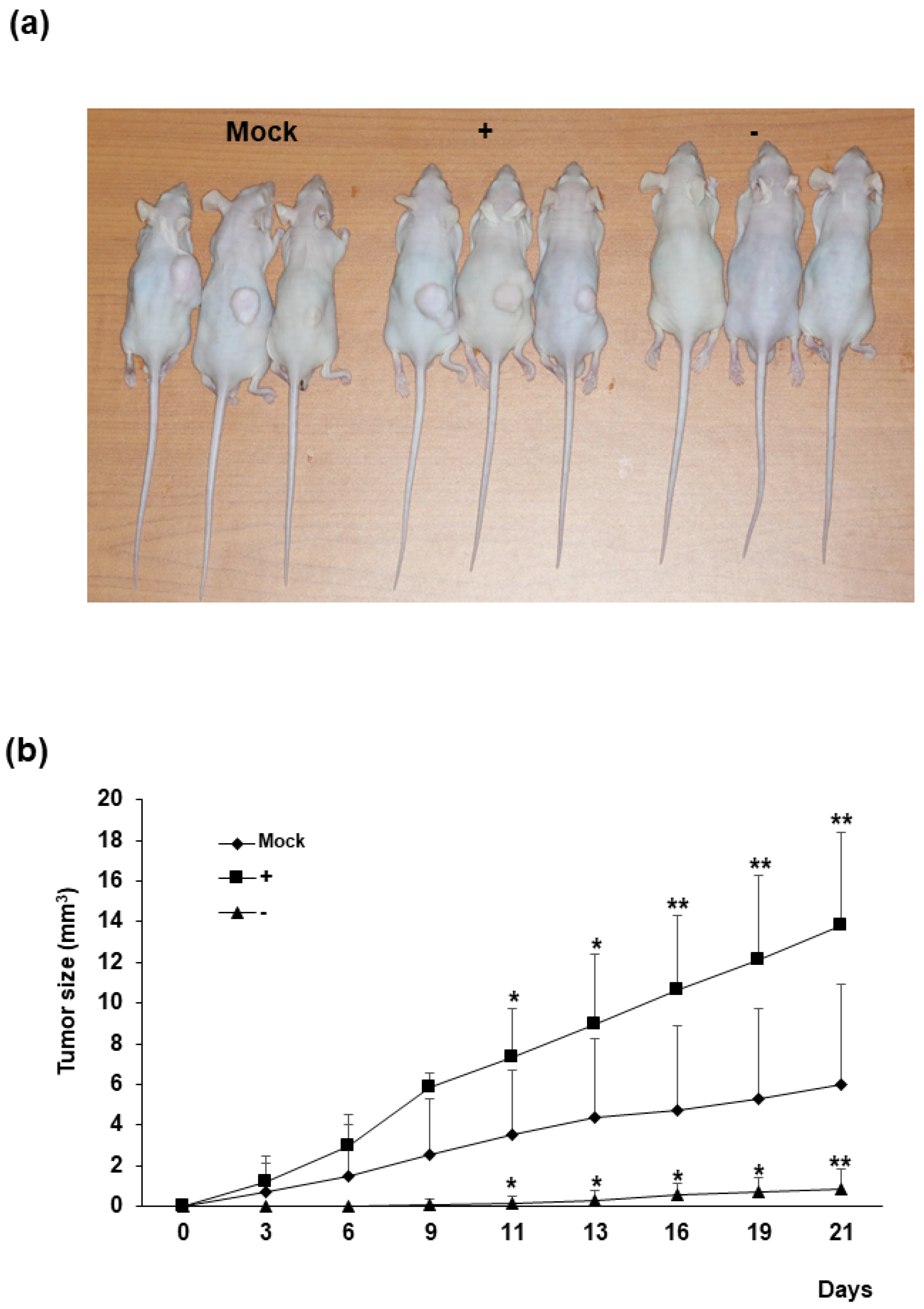
Human colorectal cancer cell xenografts were generated in nude mice by injection of Mock, HERV-K env KO, and HERV-K env over-expressing DLD-1 colorectal cancer cells (Figure 3). Each group of cells was injected into five nude mice subcutaneously, and tumor growth was monitored visually for 3 weeks. In the control group where Mock cells were injected, tumors were formed in three mice, whereas in mice injected with HERV-K env over-expressing cells, tumors formed in all five mice. In the group where HERV-K env KO cells were injected, only one mouse was observed to form tumors. Comparing the mean size of total tumors, tumor growth was significantly reduced or the tumor was not formed in the HERV-K env KO injected group, being significantly increased in the HERV-K env over-expressing cells injected group. These results showed that tumor growth is significantly related to HERV-K env genes in vivo.
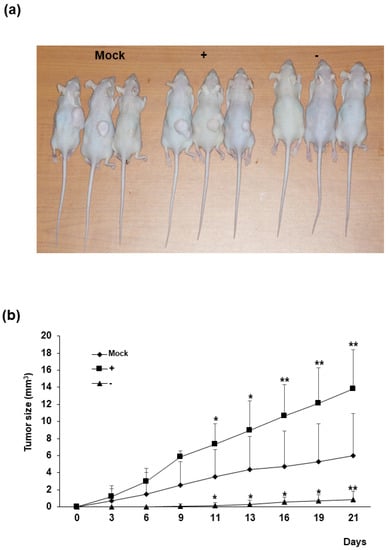
Figure 3.
The effect of HERV-K env on in vivo tumor growth. (a) Subcutaneous implantation of Mock, HERV-K env KO and over-expressing DLD-1 colorectal cancer cells in nude mice led to tumor formation. (b) Tumors from sacrificed mice. Growth curve of xenograft tumors volume of Mock, HERV-K env KO and over-expressing DLD-1 colorectal cancer cells. Tumor size was significantly reduced in HERV-K env KO group and increased in over-expression groups of DLD-1 colorectal cancer cells. p-value was determined by comparing tumor size in Mock group with HERV-K env + or − group. * p < 0.05, ** p < 0.01. (−: KO, +: over-expression).
2.4. Next-Generation Sequencing (NGS) for Analysis of Gene Expression Profile
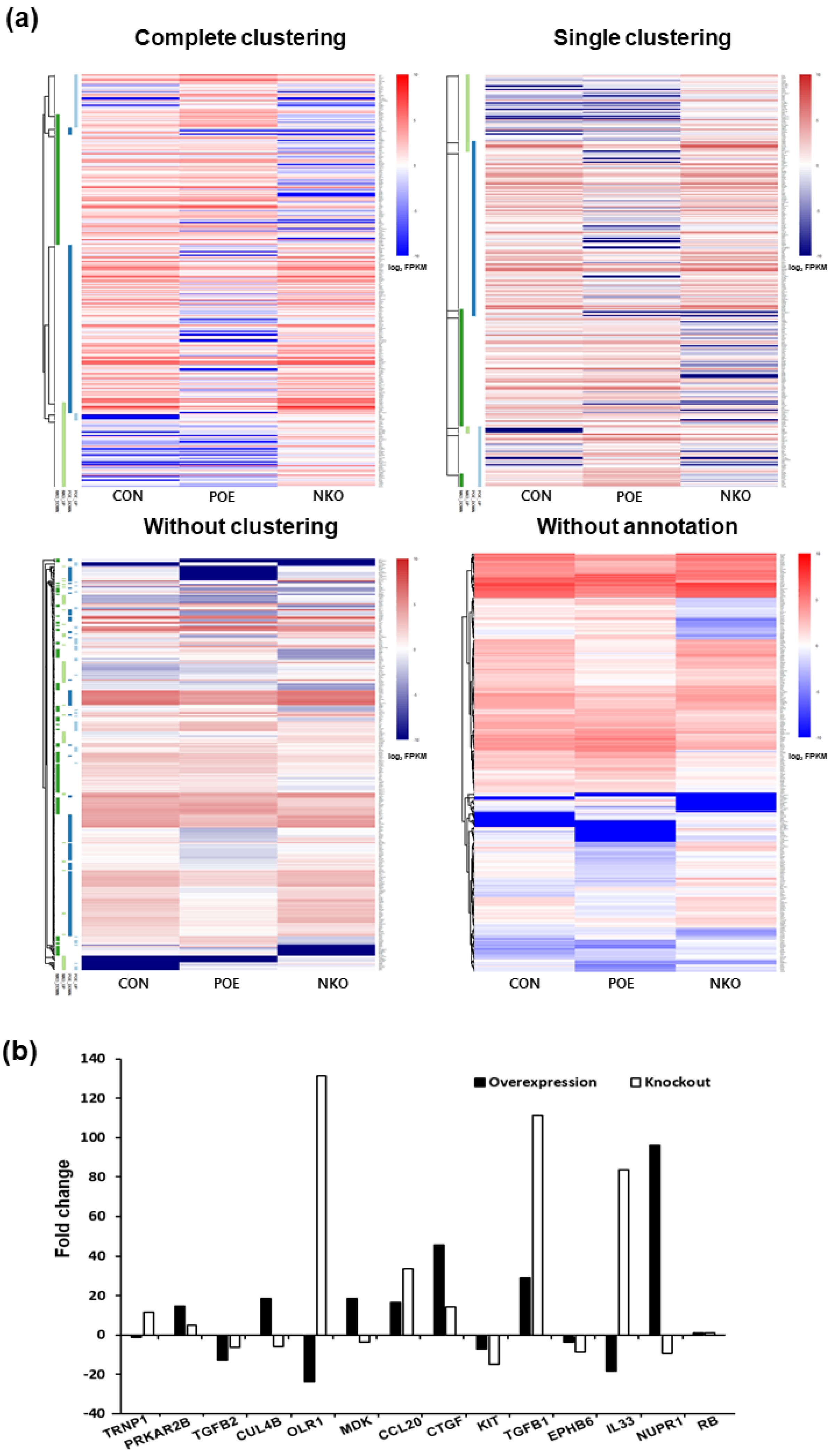
In order to identify the functional mechanism of the HERV-K env gene in tumorigenic characteristics, the transcriptome of HERV-K env KO and over-expressing DLD-1 colorectal cancer cells were analyzed and compared with Mock cells through RNA sequencing (Table 2). The heatmap of gene expression changes is shown in Figure 4a for the env KO group (108 downregulated and 70 upregulated: top panel) and env overexpression group (145 down-regulated and 50 up-regulated: bottom panel). The expression fold changes (FC) of HERV-K env KO and over-expressing DLD-1 colorectal cancer cells were compared with Mock cells using the ExDEGA program (Figure 4b). To find a meaningful target of the HERV-K env gene, protein expression levels of target genes in HERV-K env KO, over-expressing, and KO re-expression transfected HERV-K env overexpression vector in KO DLD-1 colorectal cancer cells were analyzed (Figure 5a). As shown in Figure 5b, nupr1, rb were changed in a meaningful way at the protein level. Expression of NUPR1 protein was significantly reduced in HERV-K env KO cells and increased in HERV-K env over-expressing cells. The reduced level of NUPR1 in HERV-K env KO cells was re-expressed when transfected with HERV-K env overexpression vector. On the other hand, RB protein levels were increased in HERV-K env KO cells and decreased in HERV-K env over-expressing cells. The increased levels of these proteins were re-expression in HERV-K env KO re-expressed cells (Figure 5b). Among these genes, the nupr1 gene was of main focus, as one of the most prominent targets to be related to the function of the HERV-K env gene according to results of NGS analysis and bioinformatics analysis with phenotypical change of HERV-K env KO. This is due to nupr1 being suggested as an upstream regulator of other genes. The expression of the nupr1 gene was 0.174-fold lower in HERV-K env KO and increased 1.793 in over-expressing cells (Table 3). The protein expression level of the nupr1 gene was also confirmed by immunohistochemical analysis (Figure 5c).

Table 2.
Summary of statistics for RNA sequencing data.
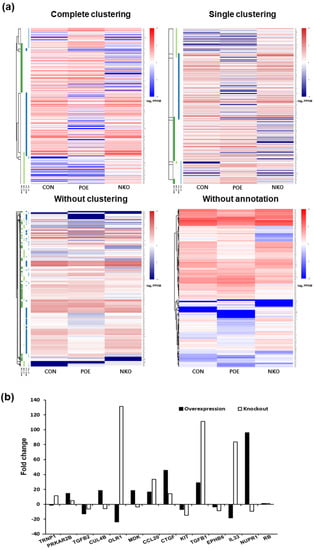
Figure 4.
Transcriptome analysis of Mock, HERV-K env KO, and HERV-K env over-expressing DLD-1 colorectal cancer cells. (a) Heatmap of gene expression changes of DLD-1 cells. Differentially expressed genes were classified based on the log2 ratio of expression in Mock to HERV-K env KO or over-expression. The number of up- and down-regulated genes identified from the three comparison groups (CON vs. POE, CON vs. NKO, and POE vs. NKO). Red: upregulation, Blue: downregulation. CON: Control, POE: overexpressed, NKO: knockout. (b) Fold changes of HERV-K env KO and over-expressing compared with Mock DLD-1 colorectal cancer cells. (−: KO, +: over-expression).
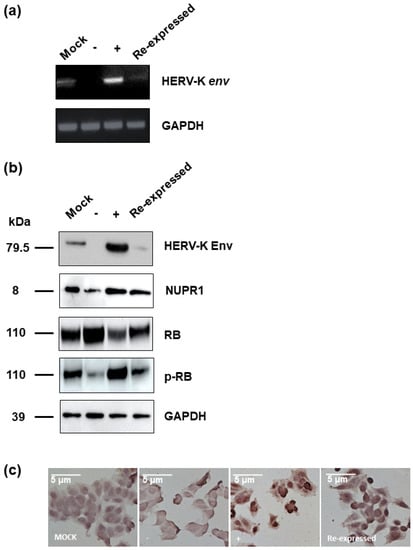
Figure 5.
Protein expression of various target genes regulate by HERV-K env gene. (a) RNA expression of HERV-K env gene in HERV-K env KO, over-expressing, and HERV-K env re-expressed (Re-expressed) DLD-1 colorectal cancer cells. (b) Protein expression of various target genes regulated by HERV-K env gene. The expression of RB protein was significantly increased in HERV-K env KO groups of DLD-1 colorectal cancer cells. (c) Immunohistochemical analysis of NUPR1 protein in HERV-K env KO, over-expressing, and HERV-K env re- expressed DLD-1 colorectal cancer cells. NUPR1 protein level was reduced in HERV-K env KO groups. (−: KO, +: over-expression).

Table 3.
Differential gene expression in nuclear protein-1 (NUPR1) by HERV-K KO and overexpression cells.
2.5. The Effect of NUPR1 on DLD-1 Colorectal Cancer Cells
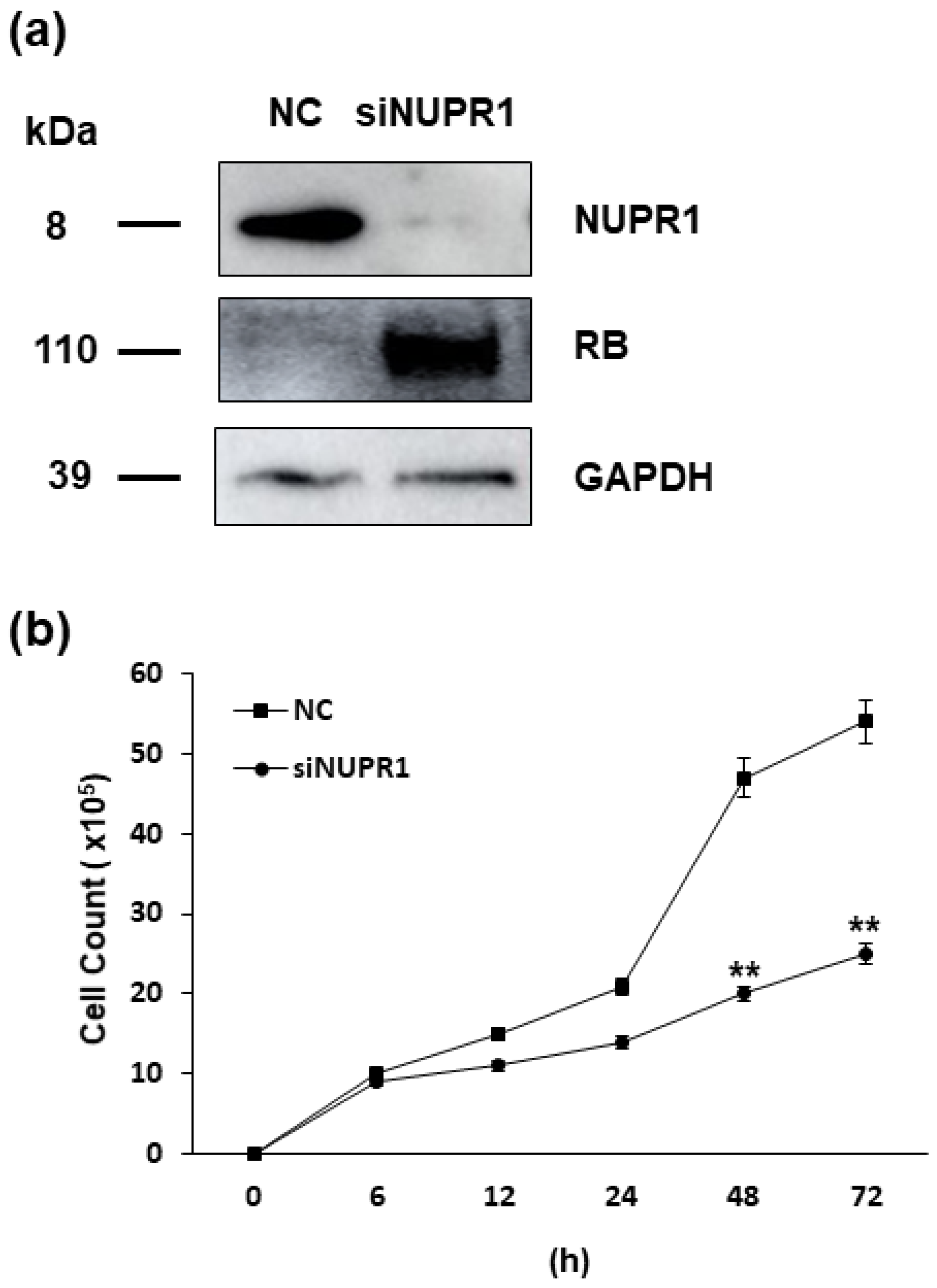
As the nupr1 gene is suggested to be the one of most important targets of HERV-K env gene, its effect on DLD-1 colorectal cancer cells was analyzed. We treated small interfering RNA (siRNA) of nupr1 gene and confirmed the reduced expression of NUPR1 protein level by Western blot. The expression of RB protein was significantly induced when silencing the nupr1 gene, a result consistent with that of HERV-K env gene KO cells. However, the expression of P53 protein was not changed in DLD-1 cells treated with siRNA of nupr1 gene (Figure 6a). Cell proliferation, which is the most prominent characteristics of HERV-K env gene KO cells, was significantly reduced by treatment of siRNA of nupr1 gene, with these results suggesting that reduced level of nupr1 gene by HERV-K env KO induces the RB protein expression to inhibit cell growth in DLD-1 colorectal cancer cells (Figure 6b).
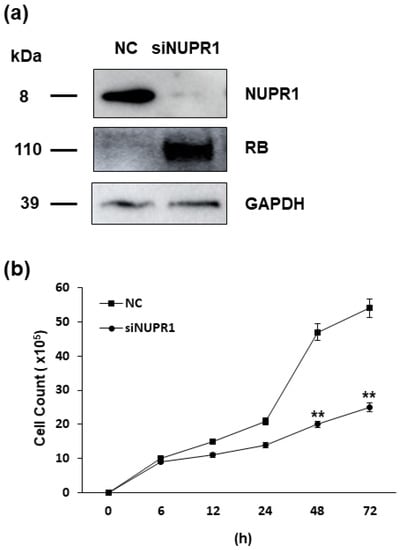
Figure 6.
Effect of NUPR1 on DLD-1 colorectal cancer cells. (a) Expression of target genes in the DLD-1 colorectal cancer cells treated with siRNA of nupr1 gene. Western blot analysis shows that the expression of NUPR1 protein was markedly reduced, with RB protein levels markedly induced by silencing of nupr1 gene respectively. (b) The effect of nupr1 gene silencing on cell proliferation of DLD-1 colorectal cancer cells. Cell proliferation rate was significantly reduced by treatment with siRNA of nupr1 gene in DLD-1 colorectal cancer cells. ** p < 0.01.
2.6. The Effect of HERV-K env KO on Reactive Oxygen Species (ROS) Generation and Protein Expression Levels Involved in Apoptosis, Autophagy, and ER Stress
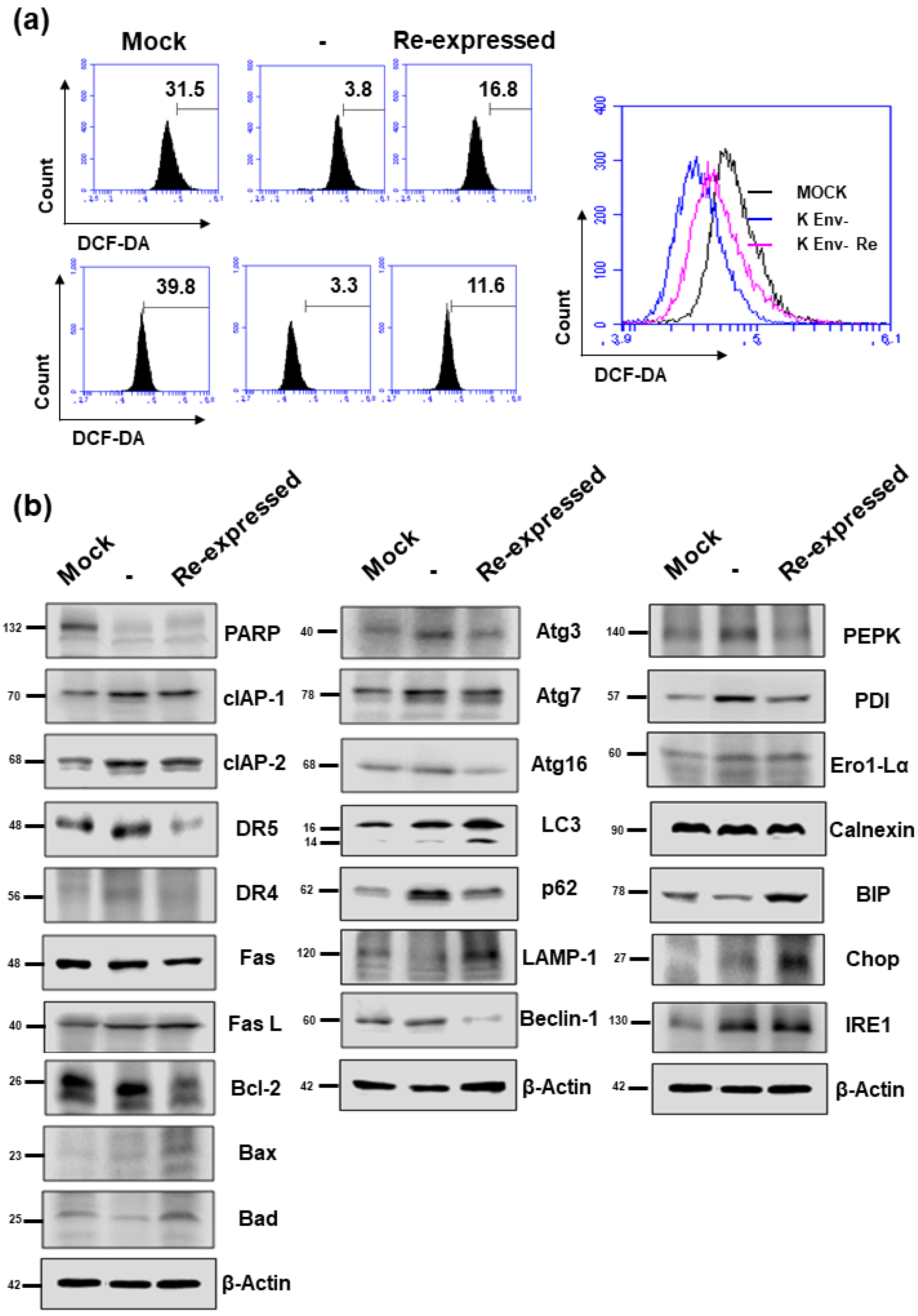
As NUPR1 protein has been reported to be involved in ROS generation, apoptosis, autophagy, and ER stress, the effect of HERV-K env KO on these functions in DLD-1 colorectal cancer cells were analyzed. As shown in Figure 7a, ROS levels were markedly decreased in HERV-K env KO cells but re-expression in HERV-K env KO re-expressed cells. Expression levels of proteins of apoptosis marker genes (PARP, clAP-1, clAP-2, DR5, DR4 Fas Fas L, Bcl-2, Bax, and Bad), autophagy marker genes (LAMP-1 and Atg3, Atg7, Atg16, Beclin-1, p62, and LC3), and ER-stress marker genes (PEPK, PDI, Ero1-Lα, Calnexin, BIP, Chop, and IRE1) were analyzed by Western blot (Figure 7b). In the case of proteins involved in apoptosis, the protein expression levels changed as follows: PARP expression levels were reduced in HERV-K env KO cells, whereas CIAP-1 and CIAP-2 protein were increased in HERV-K env KO cells. However, these proteins did not re-expression in HERV-K env KO re-expressed cells. DR4 and DR5 protein levels were increased in HERV-K env KO cells. The expression of both proteins was re-expression in HERV-K env KO re-expressed cells (Figure 7b). Protein expression levels of LAMP-1 were reduced in HERV-K env KO cells, whereas p62 protein was increased in HERV-K env KO cells. Both protein levels were re-expression in HERV-K env KO re-expressed cells (Figure 7b). CHOP was increased only in HERV-K env KO re-expressed cells (Figure 7b). Among the proteins involved in ER stress, PEPK and PDI were increased in HERV-K env KO cells and re-expression in HERV-K env KO re-expressed cells, whereas IRE1 was increased in HERV-K env KO cells but not in HERV-K env KO re-expressed cells.
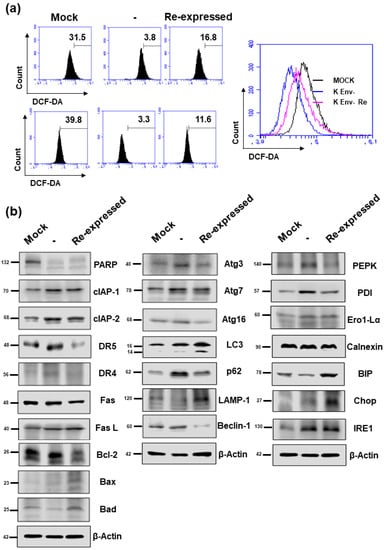
Figure 7.
The effect of HERV-K env KO on reactive oxygen species (ROS) generation and protein expression levels involved in apoptosis, autophagy, and ER stress. (a) Flow cytometric analyses of ROS level in Mock, HERV-K env KO, and HERV-K env KO re-expressed cells. (b) Protein expression levels of apoptosis, autophagy, and ER-stress markers by Western blot analysis.
2.7. Confirmation of the Effect of HERV-K env KO on HCT116 Colorectal Cancer
In order to confirm the effect of HERV-K env KO on other colorectal cancer cell lines, we generated HERV-K env KO HCT116 cells. As shown in Figure S2, HERV-K env gene in K119 region was completely deleted and the RNA expression of HERV-K env was significantly reduced. Other HERV-K elements including gag, pro, and pol were not changed but rec was significantly reduced at RNA level. The protein level of HERV-K Env was also reduced and other proteins affected by HERV-K Env KO were changed by the same pattern with DLD-1 cells. These data suggest that HERV-K Env KO specifically affect the protein expression of NUPR1 and RB to regulate cell proliferation in colorectal cancer cells.
3. Discussion
Previous studies have reported that HERV-K Env protein expression is specifically increased in colorectal cancer compared to surrounding normal tissues [16]. These results suggest that the HERV-K Env protein may play an important role in cancer progression. The increase in HERV-K Env protein in cancer tissues compared to normal tissues can be considered from two aspects. The first is the possibility that HERV-K Env expression increased as several genes locked by the de-methylation of cancer tissues were actively expressed during the process of cancer. The other possibility is that the HERV-K Env protein may play an important role in the process of cancer. In order to clarify the role of HERV-K Env in the carcinogenesis process, HERV-K env KO cells were generated using the CRISPR-Cas9 system to clarify the role of HERV-K Env in the carcinogenesis process. The role of HERV-K Env in the process of cancer was then investigated.
In a previous study to find the functions of HERV-K env in cancer progression, shRNA was used to reduce the expression of HERV-K genes in breast cancer and pancreatic cancer [18,19]. These two studies revealed that the expression of HERV-K env plays an important role in carcinogenesis, including cell proliferation, invasion, and migration. However, the possibility of different target genes and functions of HERV-K env for different types of cancer remains a need for further studies. The present study identified nupr1 as a new target gene by the HERV-K env KO system in colorectal cancer cells. This study also showed that HERV-K env KO significantly reduced cell proliferation, tumor growth in vivo, cell migration, invasion, and tumor colonization. Additionally, nupr1 was first identified as the key target gene by transcriptome analysis of HERV-K env KO and over-expression.
NUPR1, known as p8, was first described in pancreatic acinar cells of rats in a study evaluating molecular changes caused by acute pancreatitis [20]. Although intrinsic functions of NUPR1 are still debated, it is clear that it acts as an essential component during stress cell responses, protecting cells from genotoxic or oxidative damage (Taieb 2005, Clark et al. 2008, Hamidi et al. 2012, and BarbosaSampaio et al. 2013). NUPR1 is also involved in the onset of ER stress, and its role in the context remains largely unexplored [21,22]. NUPR1 has been studied as a new target for various cancers since its discovery as it affects cell growth, migration, and invasion [23]. Knockdown of the nupr1 gene could inhibit cell proliferation, migration, and cell growth in glioblastoma cells through ERK1/2, p38 MAPK and caspase-3 [24]. Down-regulation of the nupr1 gene reduces genes functionally involved in cell growth and proliferation in liver cancer cell lines [23]. However, NUPR1 has multiple functions and may be a double-edged knife with the ability to suppress tumors and promote tumor development in a variety of cancers [25].
As a result of transcriptome analysis, nupr1 was first reported as one of the most critical targets of the HERV-K env gene. When the nupr1 gene was downregulated by siRNA treatment, cell proliferation was also significantly downregulated in DLD-1 cells. Furthermore, RB protein expression, a tumor suppressor, was also upregulated in nupr1 silencing cells, which coincided with the results of HERV-K env KO cells. These results suggest that reduced NUPR1 levels in HERV-K env KO cells activated RB levels to inhibit cell proliferation in DLD-1 colorectal cancer cells. The protein expression level of RB and phosphor-RB was significantly changed by HERV-K env KO cells but not changed at RNA level. These data suggest that HERV-K env KO and down-regulation of NUPR1 may regulate the protein stability of RB protein. The functions of NUPR1 have been reported to be highly related to ROS generation, apoptosis induced by autophagy, and ER stress [26,27]. Therefore, the ROS-NUPR1 pathway was analyzed via ROS levels and related protein levels in HERV-K env KO cells. As a result, ROS levels decreased in HERV-K env KO cells compared to that in Mock cells. The reduced level of ROS in HERV-K env KO cells was re-expression in HERV-K env re-expressed cells. These results suggest that ROS levels were critically affected by HERV-K env KO. Moreover, it was identified that the target proteins of NUPR1 were critically changed in HERV-K env KO and re-expressed cells. These results suggest that HERV-K env related NUPR1 may activation the ROS-NUPR1 pathway in DLD-1 colorectal cancer cells.
4. Materials and Methods
4.1. Cell Culture and Transfection
The human colorectal cancer cell lines DLD-1 and HCT116 were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). DLD-1 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (FBS) (Invitrogen, Carlsbad, CA, USA), 1% penicillin, and L-glutamine (Thermo Fisher Scientific, Rockford, IL, USA). HCT116 cells were cultured in RPMI 1640 (Thermo Fisher Scientific, Rockford, IL, USA) containing 10% fetal bovine serum (FBS) (Invitrogen, Carlsbad, CA, USA), 1% penicillin, and L-glutamine (Thermo Fisher Scientific, Rockford, IL, USA). All cell lines were maintained at 37 °C in a humidified atmosphere containing 5% CO2 and 90% humidity.
Transfection of plasmids or siRNA was carried out using the Lipofectamine 2000 reagent (Thermo Fisher Scientific, Rockford, IL, USA) according to the protocol instructions. Briefly, cells were trypsinized, counted, and seeded in plates the day before transfection to ensure about 80% cell confluence on the day of transfection. Transfection efficiency was monitored by RT-PCR.
4.2. Generation of Knockout Cell Line with CRISPR-Cas9
Guide RNA sequences for CRISPR/Cas9 were designed using the CRISPR design provided by Toolgen, the special company for CRISPR-Cas9 technology (Toolgen, Seoul, Rep Korea). Insert oligonucleotides for HERV-K119 Env gRNA #1, #2, and #3 were 5′-CTGTCATTTGGATGGGAGACAGG-3′/5′-GTTTCCAGTTACAGTGTGACTGG-3′and 5′-TATTCCAGTCACACTGTAACTGG-3′, respectively. The HERV-K env guide RNA targets chr12:58,721,197–58,722,612 (-) of HERV-K119 env gene. The detail information of guide RNAs were shown in Figure S1. The complementary oligonucleotides for guide RNAs (gRNAs) were annealed and cloned into CRISPR/Cas9-Puro vector and pRGEN-Cas9-CMV/T7-Hygro-EGFP (Toolgen, Seoul, Korea). DLD-1 and HCT116 cells transfected with CRISPR/Cas9+gRNA #1, #2, and #3. At 18 h after transfection, cells were treated with 100 μg/mL hygromycin for two days. After two weeks, colonies were isolated with the cloning cylinders for further investigations including RT-PCR, genomic PCR, and Western blot.
4.3. Plasmid Construct for Over-Expression
The coding sequence of the HERV-K env gene was analyzed using the NCBI (https://www.ncbi.nlm.nih.gov/, accessed on 24 February 2021) and UCSC Genome Browser Database (https://genome.ucsc.edu, accessed on 24 February 2021) for PCR. Amplified HERV-K env gene was inserted into pcDNA3.1 (+) expression vector (Invitrogen, Carlsbad, CA, USA) and full sequences were confirmed by sequencing.
4.4. RT-PCR and Genomic PCR
Total RNA was isolated from cells using Trizol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. cDNA was synthesized using a PCR Mix (Bioneer, Seoul, Rep Korea) to measure the HERV-K element gene expressions. Primer sequences for HERV-K element genes are as follows: HERV-K env sense, 5′-CAC AAC TAA AGA AGC TGA CG-3′; HERV-K env antisense, 5′-CAT AGG CCC AGT TGG TAT AG-3′; HERV-K gag sense, 5′-GAG AGC CTC CCA CAG TTG AG-3′; HERV-K gag antisense, 5′-TTT GCC AGA ATC TCC CAA TC-3′, HERV-K pro sense, 5′-TGG CCT AAA CAA AAG GCT GT-3′; HERV-K pro antisense, 5′-CGA CCC CAC AGA TTA AGA GG-3′; HERV-K pol sense, 5′-TTG AGC CTT CGT TCT CAC CT-3′; HERV-K pol antisense, 5′-CTG CCA GAG GGA TGG TAA AA-3′; HERV-K rec sense, 5′-ATC GAG CAC CGT TGA CTC ACA AGA-3′; HERV-K rec antisense, 5′-GGT ACA CCT GCA GAC ACC ATT GAT-3′ [28]. GAPDH was used as a control (sense primer, 5′-CAA TGA CCC CTT CAT TGA CC-3′; antisense primer, 5′-GAC AAG CTT CCC GTT CTC AG-3′. RT-PCR cycling conditions were 94 °C for 2 min to activate DNA polymerase, followed by 40 cycles of 94 °C for 1 min, 60 °C for 1 min, 72 °C for 1 min, and 72 °C for 10 min for post-elongation. Products were analyzed on a 2% agarose gel and photographed under LED light. Genomic PCR was performed with chromosomal DNA of cells and PCR primers of each location of HERV-K env genes were described in Table 1.
4.5. Western Blot Analysis
Western blot analysis was conducted as previously described [29]. Briefly, 100 μg of protein extract was prepared using PRO-PREP™ Protein Extraction Solution (Intron Biotechnology, Kyunggi, Korea), and separated by electrophoresis on a Novex 4%–12% Bis-Tris gel (Invitrogen, Carlsbad, CA, USA). Protein concentrations were determined by the bicinchoninic acid protein assay system (Pierce, Rockford, IL, USA), with equal amounts of each sample separated by electrophoresis on Novex 4–12% Bis-Tris gels. Equal protein loading was confirmed by Coomassie blue staining of duplicate gels after electrophoresis. The gels were incubated in blotting buffer containing 1 × NuPAGE® Bis-Tris transfer buffer (Invitrogen, Carlsbad, CA, USA) and 20% methanol for 30 min at room temperature. Proteins were transferred to a nitrocellulose membrane (Invitrogen, Carlsbad, CA, USA) by electrotransfer. The membrane was pre-incubated for 2 h in Tris-buffered saline (TBS) containing 5% skim milk and 0.05% Tween 20 (TBS-T). The membrane was incubated overnight at 4 °C in TBS-T plus each antibody. The dilution and information of antibodies are as follows: HERV-K Env (1:2000 dilution, Austral Biologicals, San Ramon, CA, USA); NUPR1 (1:1000 dilution) [30]; RB (1:1000 dilution, Santa Cruz Biotechnology, Santa Cruz, CA, USA); p-RB (1:1000 dilution, Cell signaling, Danvers, MA, USA); PARP-1, clAP-1, clAP-2, DR5, DR4, Fas, Fas L, Bcl-2, Bax, Bad (1:1000 dilution, Santa Cruz Biotechnology); LAMP-1 (1:1000 dilution, proteintech, Rosemont, IL, USA); atg3, atg7, atg16,Beclin-1,p62, PEPK, PDI, Ero1-Lα, Calnexin, BIP, Chop, IRE1 (1:1000 dilution, Cell signaling); LC3 (1:2000 dilution, Cell signaling); β-Actin (1: dilution, bioworld, Dublin, OH, USA); glucose 6 phosphate dehydrogenase (GAPDH) (1:5000 dilution, R&D Systems, Minneapolis, MN, USA). The membranes were washed five more times with TBS-T, and immunoreactive proteins were detected using the enhanced chemiluminescence detection kit (Thermo Fisher Scientific, Rockford, IL, USA).
4.6. Invasion and Migration Assays
In vitro migration and invasion assays were performed as previously described [31]. Briefly, transwell chambers containing membranes with an 8-μm pore size (Invitrogen, Carlsbad, CA, USA) were used for both assays. For the migration assay, 600 μL of conditioned medium obtained by culturing DLD-1 cells for 18 h with serum-free DMEM was placed into the lower chambers of each well. DLD-1 cells (1 × 104 cells) were resuspended in 100 μL of serum-free DMEM and placed in the upper chambers of each well. The chambers were incubated for 18 h at 37 °C, and the cells in the lower chambers were fixed with methanol and stained with hematoxylin and eosin (Invitrogen) for counting. The invasion assay was performed in a similar fashion, except that the upper surface of the transwell filter was coated with 20 µL of 0.5 mg/mL Matrigel (BD Biosciences, Bedford, MA, USA) before the cells were added to the upper chambers. All experiments were repeated at least three times, with each data point measured in triplicate. The mean values and 95% confidence intervals were calculated.
4.7. Soft-Agar Colonogenic Assay
For the colony-formation assay, trypsin-treated cells were suspended in medium containing DMEM medium with 10% fetal bovine serum, antibiotics, and 3 mL of 0.35% noble agar (Difco, Sparks, MD, USA). Cells (1 × 106 cells/well) were plated onto a solidified medium containing 3 mL of 0.7% noble agar in a 60-mm dish. The dishes were incubated at 37 °C with 5% CO2, and fresh medium was added every 3–4 days. Cells have been cultured for 25 days before staining with 0.05% crystal violet. Forming colonies (>100 μm in diameter) were counted using microscopy.
4.8. Cell Viability Assay
For the cell viability assay, cells (1 × 104 cells per well) were plated in complete medium in a 6-well plate and incubated for 72 h. Cells were then harvested, and cell proliferation rates were measured by counting viable cells using the trypan blue dye exclusion method.
4.9. Immunofluorescence (IF) and Immunohistochemistry (IHC)
For immunofluorescence analysis, the assay was performed as described previously [32]. Cells cultured chamber slides were incubated in 10% normal serum in PBS for 1 h to block nonspecific antibodies. Slides were then incubated with an antibody against HERV-K Env (1:2000 dilution, Austral Biologicals) overnight at 4 °C. After primary antibody incubation, slides were washed three times in PBS for five minutes and incubated with secondary antibodies, including Alexa Fluor 546 anti-mouse antibody (Invitrogen, Carlsbad, CA, USA) for 1 h. Specimen epifluorescence was determined with the use of a confocal laser-scanning microscope (LSM510 META, ZEISS, Jena, Germany). Confocal images were analyzed using AlphaEase FC image analysis software (Alpha Innotech, San Leandro, CA, USA). For immunohistochemistry, the color response was developed with Dako’s EnVision System (DAKO, Carpinteria, CA, USA), and hematoxylin was used as a counterstain.
4.10. Tumor Xenograft Assays
For tumor xenograft assay, male immunodeficient athymic nude mice, 5-weeks of age (NCI, Frederick, MD, USA), were used. Cells (1 × 105 cells/100 μL) were injected subcutaneously and tumor diameters were measured every 3 days for 3 weeks post-injection using digital calipers. The animal experiment was approved by Kosin University College of Medicine Institutional Animal Care and Use Committee: KUCMIACUC (KMAP-17-25).
4.11. RNA-Seq Data Analysis
RNA was isolated from each group of cells including Mock, HERV-K env KO, and HERV-K env over-expression cells, and next generation sequencing (NGS) for transcriptome analysis was performed by clustering analysis of the DEGs based on the log2 FPKM values and a heat map was generated using Pheatmap software (v1.0.8, available at http://cran.r-project.org/web/packages/pheatmap/index.html, accessed on 24 February 2021) with hierarchical clustering method (complete) functions. We selected genes that showed more than two expressional differences among each group. Genes with statistically significant differences in expression were identified using the ExDEGA program (Ebiogen, Seoul, Korea).
4.12. Flow Cytometry Analysis for Measurement Generation of ROS
The production of intracellular ROS was evaluated using 2,7-dichlorofluorescein diacetate (DCF-DA; Sigma-Aldrich Chemical Co., St. Louis, MO, USA). Cells were incubated with 10 μM DCF-DA in a dark room at 37 °C for 30 min. Cells were then washed with phosphate buffered saline (PBS) and the amount of ROS produced was recorded using a flow cytometer (BD Biosciences, San Jose, CA, USA). At least 10,000 cells were analyzed from each sample.
4.13. si RNA of NUPR1
NUPR1 siRNA (siNUPR1) and its negative control oligonucleotide (siNC) purchased from GenePharma, Co, Ltd. (Shanghai, China). The sequences of nupr1-specific siRNA is as follows: r(GGAGGACCCAGGACAGGAU)dTdT [30].
4.14. Statistical Analysis
A two-tailed p-value < 0.05 was considered to indicate statistical significance. Statistical significance of differences among the groups was determined using a two-tailed Student t-test.
5. Conclusions
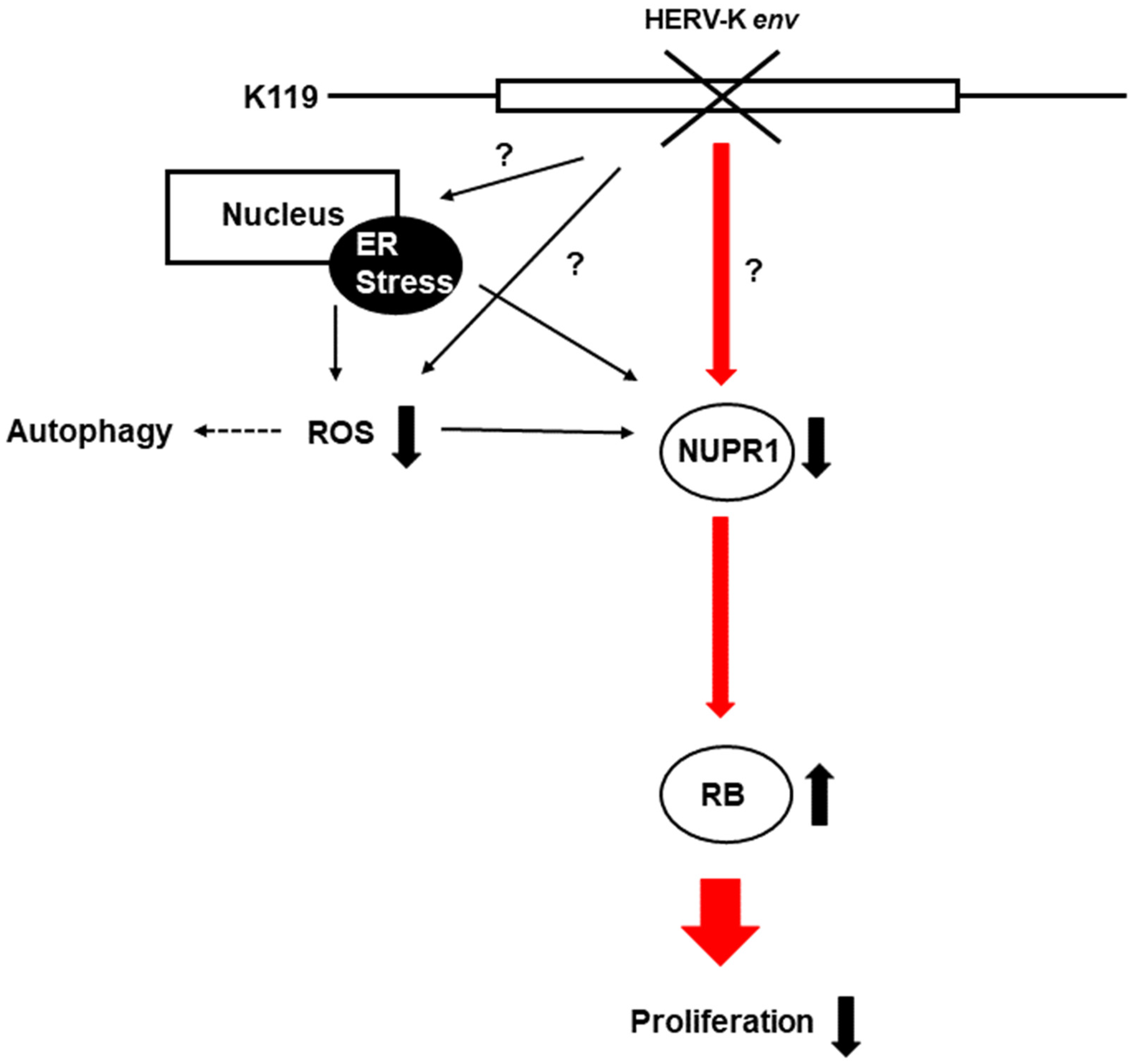
In conclusion, HERV-K env KO reduced Nupr1 levels affect cell proliferation by inducing RB1 proteins, and is also involved in ROS generation and regulation of autophagic cell death-related proteins (Figure 8).
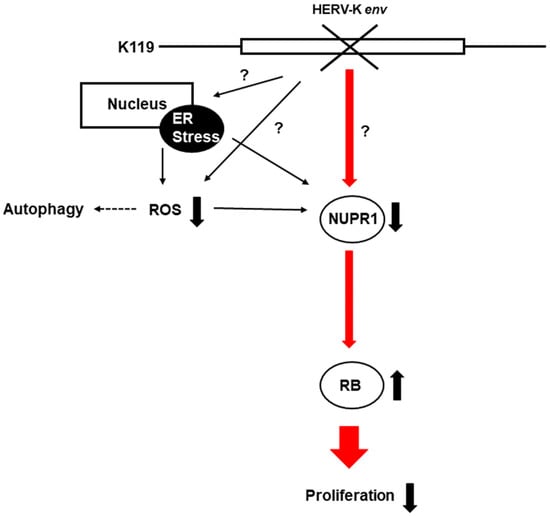
Figure 8.
A schematic diagram on the pathways related to the HERV-K env KO. HERV-K env KO reduced the protein level of NUPR1, and its signaling activates the RB tumor suppressor gene to reduce the cell proliferation of DLD-1 colorectal cancer. (−: KO, +: over-expression).
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/ijms22083941/s1, Figure S1: Selection of three guide RNAs for targeting of HERV-K Env knockout system, Figure S2: Knockout of HERV-K env gene in HCT116 colorectal cells.
Author Contributions
Conceptualization, E.-J.K., H.-S.K., and H.-J.C.; investigation, E.-J.K.; writing—original draft preparation, E.-J.K.; writing—review and editing, M.-S.O., Y.-H.C., J.L.I., S.M., K.H., S.M., K.H., H.-S.K., and H.-J.C.; funding acquisition, H.-J.C. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by a National Research Foundation Grant funded by the Korean Government (NRF-2016R1D1A3B01007444).
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of Kosin University College of Medicine Institutional Animal Care and Use committee: KUCMIACUC (KMAP-17-25).
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| HERV | human endogenous retrovirus |
| HERV-K | HML-2 |
| Env | envelope |
| ROS | reactive oxygen species |
| NUPR1 | nuclear protein-1 |
| KO | knockout |
| KD | knockdown |
| NGS | next generation sequencing |
| FC | fold change |
| CRC | colorectal cancer |
| siRNA | Small interfering RNA |
References
- Belshaw, R.; Pereira, V.; Katzourakis, A.; Talbot, G.; Paces, J.; Burt, A.; Tristem, M. Long-term reinfection of the human genome by endogenous retroviruses. Proc. Natl. Acad. Sci. USA 2004, 101, 4894–4899. [Google Scholar] [CrossRef] [PubMed]
- Belshaw, R.; Dawson, A.L.; Woolven-Allen, J.; Redding, J.; Burt, A.; Tristem, M. Genomewide Screening Reveals High Levels of Insertional Polymorphism in the Human Endogenous Retrovirus Family HERV-K(HML2): Implications for Present-Day Activity. J. Virol. 2005, 79, 12507–12514. [Google Scholar] [CrossRef]
- Hohn, O.; Hanke, K.; Bannert, N. HERV-K(HML-2), the Best Preserved Family of HERVs: Endogenization, Expression, and Implications in Health and Disease. Front. Oncol. 2013, 3, 246. [Google Scholar] [CrossRef] [PubMed]
- Flockerzi, A.; Ruggieri, A.; Frank, O.; Sauter, M.; Maldener, E.; Kopper, B.; Wullich, B.; Seifarth, W.; Muller-Lantzsch, N.; Leib-Mosch, C.; et al. Expression patterns of transcribed human endogenous retrovirus HERV-K(HML-2) loci in human tissues and the need for a HERV Transcriptome Project. BMC Genom. 2008, 9, 354. [Google Scholar] [CrossRef]
- Hanke, K.; Hohn, O.; Bannert, N. HERV-K(HML-2), a seemingly silent subtenant—But still waters run deep. Apmis 2016, 124, 67–87. [Google Scholar] [CrossRef]
- Boller, K.; Schonfeld, K.; Lischer, S.; Fischer, N.; Hoffmann, A.; Kurth, R.; Tonjes, R.R. Human endogenous retrovirus HERV-K113 is capable of producing intact viral particles. J. Gen. Virol. 2008, 89, 567–572. [Google Scholar] [CrossRef]
- Ruprecht, K.; Ferreira, H.; Flockerzi, A.; Wahl, S.; Sauter, M.; Mayer, J.; Mueller-Lantzsch, N. Human Endogenous Retrovirus Family HERV-K(HML-2) RNA Transcripts Are Selectively Packaged into Retroviral Particles Produced by the Human Germ Cell Tumor Line Tera-1 and Originate Mainly from a Provirus on Chromosome 22q11.21. J. Virol. 2008, 82, 10008–10016. [Google Scholar] [CrossRef] [PubMed]
- Wang-Johanning, F.; Rycaj, K.; Plummer, J.B.; Li, M.; Yin, B.; Frerich, K.; Garza, J.G.; Shen, J.; Lin, K.; Yan, P.; et al. Immunotherapeutic Potential of Anti-Human Endogenous Retrovirus-K Envelope Protein Antibodies in Targeting Breast Tumors. J. Natl. Cancer Inst. 2012, 104, 189–210. [Google Scholar] [CrossRef]
- Wang-Johanning, F.; Li, M.; Esteva, F.J.; Hess, K.R.; Yin, B.; Rycaj, K.; Plummer, J.B.; Garza, J.G.; Ambs, S.; Johanning, G.L. Human endogenous retrovirus type K antibodies and mRNA as serum biomarkers of early-stage breast cancer. Int. J. Cancer 2014, 134, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Rycaj, K.; Geng, S.; Li, M.; Plummer, J.B.; Yin, B.; Liu, H.; Xu, X.; Zhang, Y.; Yan, Y.; et al. Expression of Human Endogenous Retrovirus Type K Envelope Protein is a Novel Candidate Prognostic Marker for Human Breast Cancer. Genes Cancer 2011, 2, 914–922. [Google Scholar] [CrossRef]
- Ishida, T.; Obata, Y.; Ohara, N.; Matsushita, H.; Sato, S.; Uenaka, A.; Saika, T.; Miyamura, T.; Chayama, K.; Nakamura, Y.; et al. Identification of the HERV-K gag antigen in prostate cancer by SEREX using autologous patient serum and its immunogenicity. Cancer Immun. 2008, 8, 15. [Google Scholar] [PubMed]
- Hahn, S.; Ugurel, S.; Hanschmann, K.M.; Strobel, H.; Tondera, C.; Schadendorf, D.; Lower, J.; Lower, R. Serological Response to Human Endogenous Retrovirus K in Melanoma Patients Correlates with Survival Probability. AIDS Res. Hum. Retrovir. 2008, 24, 717–723. [Google Scholar] [CrossRef] [PubMed]
- Wang-Johanning, F.; Liu, J.; Rycaj, K.; Huang, M.; Tsai, K.; Rosen, D.G.; Chen, D.T.; Lu, D.W.; Barnhart, K.F.; Johanning, G.L. Expression of multiple human endogenous retrovirus surface envelope proteins in ovarian cancer. Int. J. Cancer 2007, 120, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Iramaneerat, K.; Rattanatunyong, P.; Khemapech, N.; Triratanachat, S.; Mutirangura, A. HERV-K Hypomethylation in Ovarian Clear Cell Carcinoma is Associated with a Poor Prognosis and Platinum Resistance. Int. J. Gyn. Cancer 2011, 21, 51–57. [Google Scholar] [CrossRef]
- Curty, G.; Marston, J.L.; Rougvie, M.D.M.; Leal, F.E.; Nixon, D.F.; Soares, M.A. Human Endogenous Retrovirus K in Cancer: A Potential Biomarker and Immunotherapeutic Target. Viruses 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Jo, J.O.; Kang, Y.J.; Ock, M.S.; Song, K.S.; Jeong, M.J.; Jeong, S.J.; Choi, Y.H.; Ko, E.J.; Leem, S.H.; Kim, S.; et al. Expression profiles of HERV-K Env protein in normal and cancerous tissues. Genes Genom. 2016, 98, 91–107. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Radvanyi, L.; Yin, B.; Rycaj, K.; Li, J.; Chivukula, R.; Lin, K.; Lu, Y.; Shen, J.; Chang, D.Z.; et al. Downregulation of Human Endogenous Retrovirus Type K (HERV-K) Viral env RNA in Pancreatic Cancer Cells Decreases Cell Proliferation and Tumor Growth. Clin. Cancer Res. 2017, 23, 5892–5911. [Google Scholar] [CrossRef]
- Zhou, F.; Li, M.; Wei, Y.; Lin, K.; Lu, Y.; Shen, J.; Johanning, G.L.; Wang-Johanning, F. Activation of HERV-K Env protein is essential for tumorigenesis and metastasis of breast cancer cells. Oncotarget 2016, 7, 84093–84117. [Google Scholar] [CrossRef] [PubMed]
- Mallo, G.V.; Fiedler, F.; Calvo, E.L.; Ortiz, E.M.; Vasseur, S.; Keim, V.; Morisset, J.; Iovanna, J.L. loning and Expression of the Rat p8 cDNA, a New Gene Activated in Pancreas during the Acute Phase of Pancreatitis, Pancreatic Development, and Regeneration, and Which Promotes Cellular Growth. J. Biol. Chem. 1997, 272, 32360–32369. [Google Scholar] [CrossRef]
- Santofimia-Castano, P.; Lan, W.; Bintz, J.; Gayet, O.; Carrier, A.; Lomberk, G.; Neira, J.L.; Gonzalez, A.; Urrutia, R.; Soubeyran, P.; et al. Inactivation of NUPR1 promotes cell death by coupling ER-stress responses with necrosis. Sci. Rep. 2018, 8, 16999. [Google Scholar] [CrossRef]
- Cai, D.; Huang, E.; Luo, B.; Yang, Y.; Zhang, F.; Liu, C.; Lin, Z.; Xie, W.B.; Wang, H. Nupr1/Chop signal axis is involved in mitochondrion-related endothelial cell apoptosis induced by methamphetamine. Cell Death Dis. 2016, 7, e2161. [Google Scholar] [CrossRef]
- Emma, M.R.; Iovanna, J.L.; Bachvarov, D.; Puleio, R.; Loria, G.R.; Augello, G.; Candido, S.; Libra, M.; Gulino, A.; Cancila, V.; et al. NUPR1, a new target in liver cancer: Implication in controlling cell growth, migration, invasion and sorafenib resistance. Cell Death Dis. 2016, 7, e2269. [Google Scholar] [CrossRef]
- Li, J.; Ren, S.; Liu, Y.; Lian, Z.; Dong, B.; Yao, Y.; Xu, Y. Knockdown of NUPR1 inhibits the proliferation of glioblastoma cells via ERK1/2, p38 MAPK and caspase-3. J. Neuro Oncol. 2017, 132, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Cano, C.E.; Hamidi, T.; Sandi, M.J.; Iovanna, J.L. Nupr1: The Swiss-knife of cancer. J. Cell. Physiol. 2011, 226, 1439–1443. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, T.; Cano, C.E.; Grasso, D.; Garcia, M.N.; Sandi, M.J.; Calvo, E.L.; Dagorn, J.C.; Lomberk, G.; Urrutia, R.; Goruppi, S.; et al. Nupr1-Aurora Kinase A Pathway Provides Protection against Metabolic Stress-Mediated Autophagic-Associated Cell Death. Clin. Cancer Res. 2012, 18, 5234–5246. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Zeng, W.; Ouyang, W.; Xu, Q.; Chen, J.; Wang, B.; Zhang, X. Quercetin induced NUPR1-dependent autophagic cell death by disturbing reactive oxygen species homeostasis in osteosarcoma cells. J. Clin. Biochem. Nutr. 2020, 67, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Tavakolian, S.; Goudarzi, H.; Faghihloo, E. Evaluating the expression level of HERV-K env, np9, rec and gag in breast tissue. Infect. Agents Cancer 2019, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Jo, J.O.; Kim, S.R.; Bae, M.K.; Kang, Y.J.; Ock, M.S.; Kleinman, H.K.; Cha, H.J. Thymosin beta4 induces the expression of vascular endothelial growth factor (VEGF) in a hypoxia-inducible factor (HIF)-1alpha-dependent manner. Biochim. Biophys. Acta 2010, 1803, 1244–1251. [Google Scholar] [CrossRef] [PubMed]
- Grasso, D.; Garcia, M.N.; Hamidi, T.; Cano, C.; Calvo, E.; Lomberk, G.; Urrutia, R.; Iovanna, J.L. Genetic inactivation of the pancreatitis-inducible gene Nupr1 impairs PanIN formation by modulating Kras(G12D)-induced senescence. Cell Death Differ. 2014, 21, 1633–1641. [Google Scholar] [CrossRef] [PubMed]
- Cha, H.J.; Jeong, M.J.; Kleinman, H.K. Role of Thymosin β4 in Tumor Metastasis and Angiogenesis. J. Natl. Cancer Inst. 2003, 95, 1674–1680. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Jo, J.O.; Ock, M.S.; Chang, H.K.; Baek, K.W.; Lee, J.R.; Choi, Y.H.; Kim, W.J.; Leem, S.H.; Kim, H.S.; et al. Human ERV3-1 env protein expression in various human tissues and tumours. J. Clin. Pathol. 2013. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).