Muscle Proteomic Profile before and after Enzyme Replacement Therapy in Late-Onset Pompe Disease
Abstract
1. Introduction
2. Results
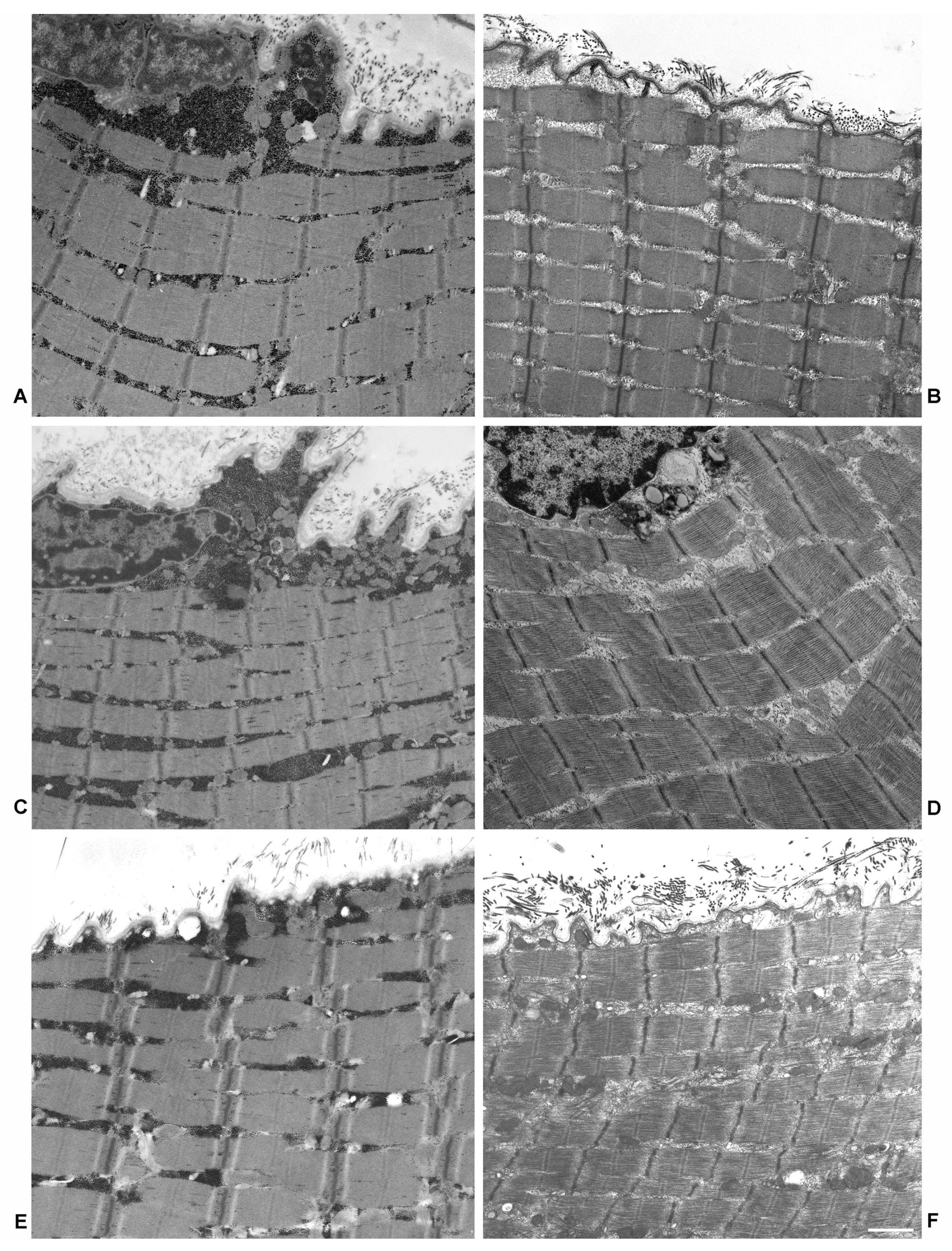
2.1. Electron Microscopy
2.2. Comparative Proteomic Analysis
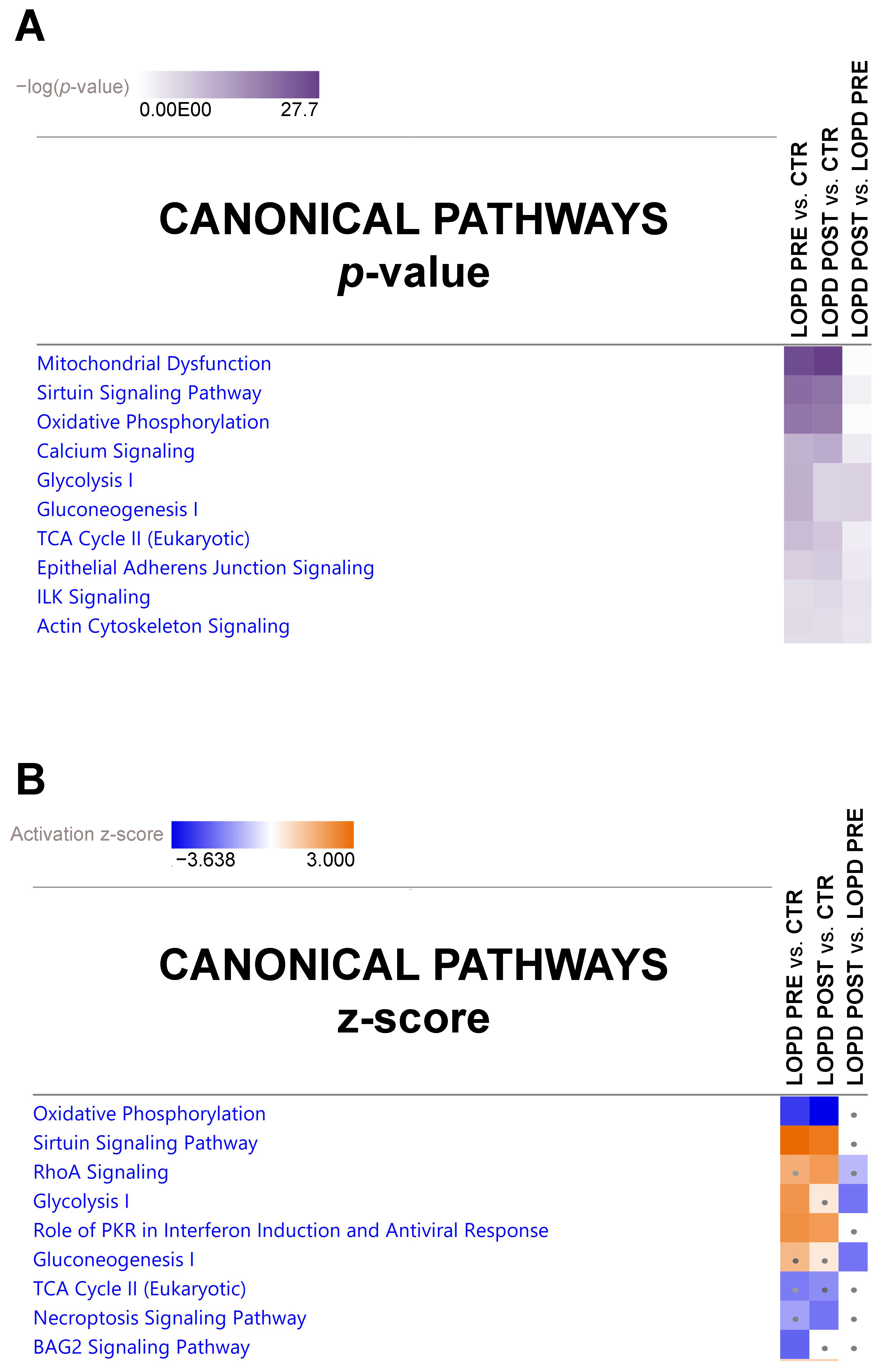
2.3. Pathway Analysis
2.3.1. Canonical Pathways
2.3.2. Diseases and Bio Functions
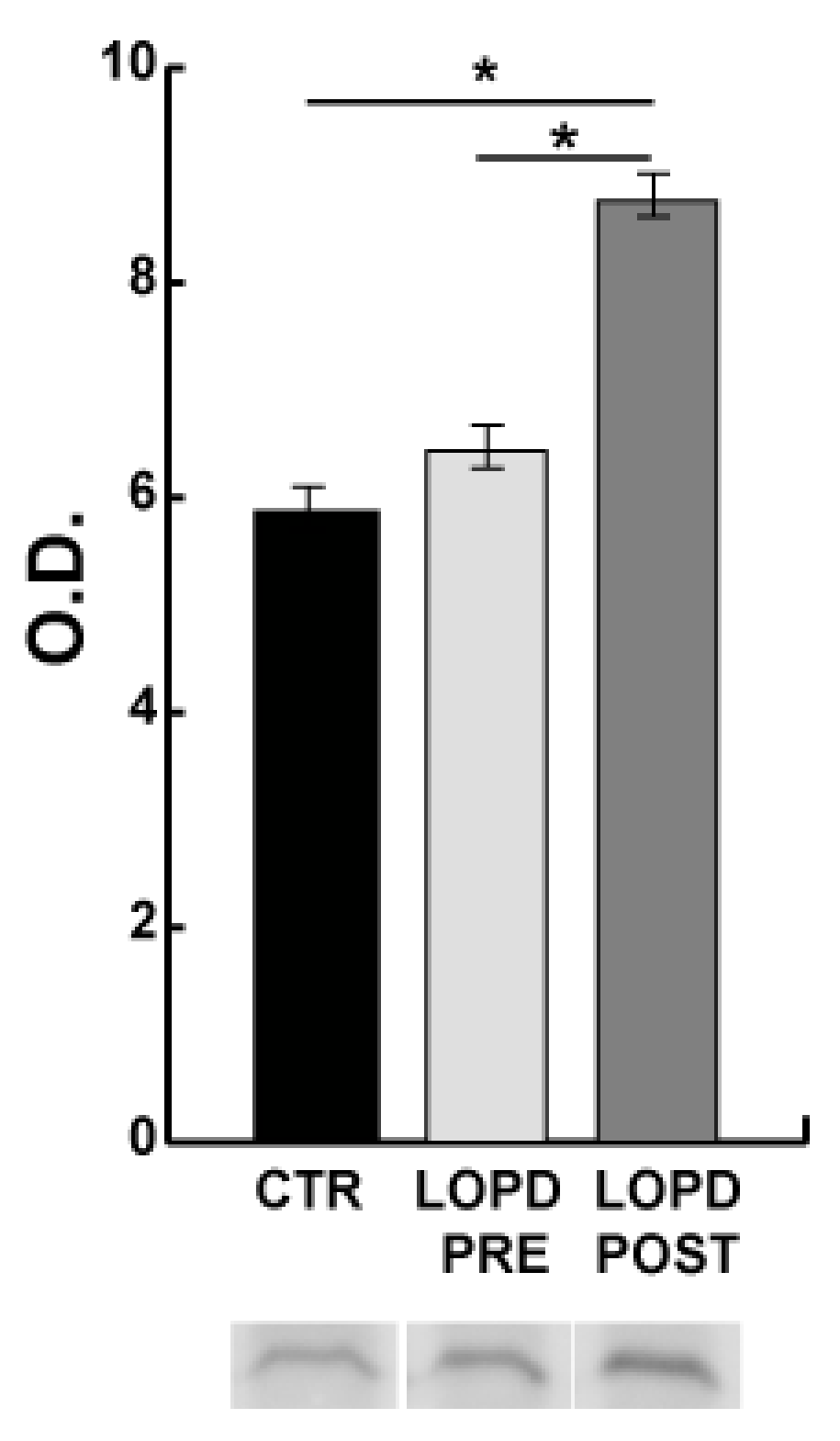
2.4. ER Stress
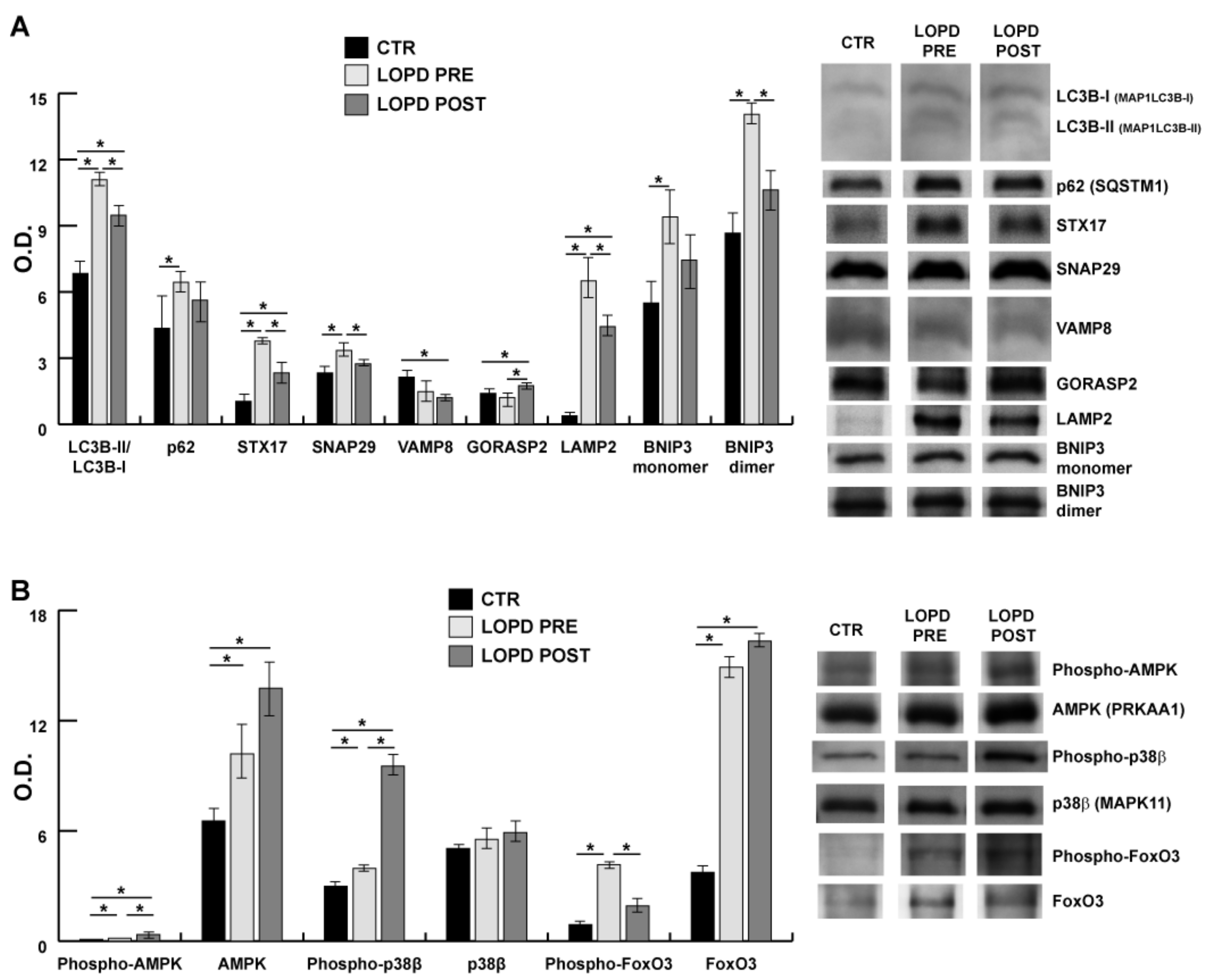
2.5. Lysosomal Autophagy and Apoptosis
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Patients
4.3. Electron Microscopy
4.4. Proteomic Analysis
4.4.1. Protein Extraction
4.4.2. Two-Dimensional Difference in Gel Electrophoresis
4.4.3. Label-Free Liquid Chromatography with Tandem Mass Spectrometry
4.4.4. Ingenuity Pathway Analysis
4.5. Immunoblotting
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kroos, M.; Hoogeveen-Westerveld, M.; Van Der Ploeg, A.; Reuser, A.J. The genotype-phenotype correlation in Pompe disease. Am. J. Med. Genet. Part C Semin. Med. Genet. 2012, 160C, 59–68. [Google Scholar] [CrossRef]
- van der Ploeg, A.T.; Reuser, A.J. Pompe’s Disease. Lancet 2008, 372, 1342–1353. [Google Scholar] [CrossRef]
- De Vries, J.M.; van der Beek, N.A.M.E.; Hop, W.C.J.; Karstens, F.P.J.; Wokke, J.H.; de Visser, M.; van Engelen, B.G.M.; Kuks, J.B.M.; van der Kooi, A.J.; Notermans, N.C. Clinical Features and Predictors for Disease Natural Progression in Adults with Pompe Disease: A Nationwide Prospective Observational Study. Orphanet J. Rare Dis. 2012, 7, 88. [Google Scholar]
- Hagemans, M.; Winkel, L.P.; Hop, W.C.; Reuser, A.J.; Van Doorn, P.A.; Van Der Ploeg, A.T. Disease severity in children and adults with Pompe disease related to age and disease duration. Neurology 2005, 64, 2139–2141. [Google Scholar] [CrossRef]
- Harlaar, L.; Hogrel, J.-Y.; Perniconi, B.; Kruijshaar, M.E.; Rizopoulos, D.; Taouagh, N.; Canal, A.; Brusse, E.; Van Doorn, P.A.; Van Der Ploeg, A.T.; et al. Large variation in effects during 10 years of enzyme therapy in adults with Pompe disease. Neurology 2019, 93, e1756–e1767. [Google Scholar] [CrossRef] [PubMed]
- Gungor, D.; Kruijshaar, M.E.; Plug, I.; D’Agostino, R.B.; Hagemans, M.L.C.; van Doorn, P.A.; Reuser, A.J.J.; van der Ploeg, A.T. Impact of Enzyme Replacement Therapy on Survival in Adults with Pompe Disease: Results from a Prospective International Observational Study. Orphanet J. Rare Dis. 2013, 8, 49. [Google Scholar] [CrossRef]
- Toscano, A.; Schoser, B. Enzyme replacement therapy in late-onset Pompe disease: A systematic literature review. J. Neurol. 2013, 260, 951–959. [Google Scholar] [CrossRef] [PubMed]
- Schoser, B.; Stewart, A.; Kanters, S.; Hamed, A.; Jansen, J.; Chan, K.; Karamouzian, M.; Toscano, A. Survival and long-term outcomes in late-onset Pompe disease following alglucosidase alfa treatment: A systematic review and meta-analysis. J. Neurol. 2016, 264, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, T.; Ahearn, M.; Roberts, A.; Mattaliano, R.J.; Zaal, K.; Ralston, E.; Plotz, P.H.; Raben, N. Autophagy and Mistargeting of Therapeutic Enzyme in Skeletal Muscle in Pompe Disease. Mol. Ther. 2006, 14, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Lieberman, A.P.; Puertollano, R.; Raben, N.; Slaugenhaupt, S.; Walkley, S.U.; Ballabio, A. Autophagy in lysosomal storage disorders. Autophagy 2012, 8, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Spampanato, C.; Feeney, E.; Li, L.; Cardone, M.; Lim, J.-A.; Annunziata, F.; Zare, H.; Polishchuk, R.; Puertollano, R.; Parenti, G.; et al. Transcription factor EB (TFEB) is a new therapeutic target for Pompe disease. EMBO Mol. Med. 2013, 5, 691–706. [Google Scholar] [CrossRef]
- Gatto, F.; Rossi, B.; Tarallo, A.; Polishchuk, E.; Polishchuk, R.; Carrella, A.; Nusco, E.; Alvino, F.G.; Iacobellis, F.; De Leonibus, E.; et al. AAV-mediated transcription factor EB (TFEB) gene delivery ameliorates muscle pathology and function in the murine model of Pompe Disease. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Zhang, H.; Franco, L.M.; Brown, T.; Bird, A.; Schneider, A.; Koeberl, D.D. Correction of glycogen storage disease type II by an adeno-associated virus vector containing a muscle-specific promoter. Mol. Ther. 2005, 11, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Byrne, B.J.; Falk, D.J.; Pacak, C.A.; Nayak, S.; Herzog, R.W.; Elder, M.E.; Collins, S.W.; Conlon, T.J.; Clement, N.; Cleaver, B.D.; et al. Pompe disease gene therapy. Hum. Mol. Genet. 2011, 20, R61–R68. [Google Scholar] [CrossRef] [PubMed]
- Raben, N.; Schreiner, C.; Baum, R.; Takikita, S.; Xu, S.; Xie, T.; Myerowitz, R.; Komatsu, M.; Van Der Meulen, J.H.; Nagaraju, K.; et al. Suppression of autophagy permits successful enzyme replacement therapy in a lysosomal storage disorder—murine Pompe disease. Autophagy 2010, 6, 1078–1089. [Google Scholar] [CrossRef]
- Kohler, L.; Puertollano, R.; Raben, N. Pompe Disease: From Basic Science to Therapy. Neurotherapeutics 2018, 15, 928–942. [Google Scholar] [CrossRef]
- Dasouki, M.; Jawdat, O.; Almadhoun, O.; Pasnoor, M.; McVey, A.L.; Abuzinadah, A.; Herbelin, L.; Barohn, R.J.; Dimachkie, M.M. Pompe Disease: Literature Review and Case Series. Neurol. Clin. 2014, 32, 751–776. [Google Scholar] [CrossRef] [PubMed]
- Colella, P.; Mingozzi, F. Gene Therapy for Pompe Disease: The Time is now. Hum. Gene Ther. 2019, 30, 1245–1262. [Google Scholar] [CrossRef] [PubMed]
- Sancak, Y.; Bar-Peled, L.; Zoncu, R.; Markhard, A.L.; Nada, S.; Sabatini, D.M. Ragulator-Rag Complex Targets mTORC1 to the Lysosomal Surface and Is Necessary for Its Activation by Amino Acids. Cell 2010, 141, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Tan, S.-H.; Nicolas, V.; Bauvy, C.; Yang, N.-D.; Zhang, J.; Xue, Y.; Codogno, P.; Shen, H.-M. Activation of lysosomal function in the course of autophagy via mTORC1 suppression and autophagosome-lysosome fusion. Cell Res. 2013, 23, 508–523. [Google Scholar] [CrossRef]
- Raben, N.; Baum, R.; Schreiner, C.; Takikita, S.; Mizushima, N.; Ralston, E.; Plotz, P.H. When more is less: Excess and deficiency of autophagy coexist in skeletal muscle in Pompe disease. Autophagy 2009, 5, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Picton, C.; Klee, C.B.; Cohen, P. The regulation of muscle phosphorylase kinase by calcium ions, calmodulin and troponin-C. Cell Calcium 1981, 2, 281–294. [Google Scholar] [CrossRef]
- Howlett, R.A.; Parolin, M.L.; Dyck, D.J.; Hultman, E.; Jones, N.L.; Heigenhauser, G.J.F.; Spriet, L.L. Regulation of skeletal muscle glycogen phosphorylase and PDH at varying exercise power outputs. Am. J. Physiol. Integr. Comp. Physiol. 1998, 275, R418–R425. [Google Scholar] [CrossRef] [PubMed]
- Wisselaar, H.A.; Kroos, M.A.; Hermans, M.M.; Van Beeumen, J.; Reuser, A.J. Structural and functional changes of lysosomal acid alpha-glucosidase during intracellular transport and maturation. J. Biol. Chem. 1993, 268, 2223–2231. [Google Scholar] [CrossRef]
- Thurberg, B.L.; Maloney, C.L.; Vaccaro, C.; Afonso, K.; Tsai, A.C.-H.; Bossen, E.; Kishnani, P.S.; O’Callaghan, M. Characterization of pre- and post-treatment pathology after enzyme replacement therapy for pompe disease. Lab. Investig. 2006, 86, 1208–1220. [Google Scholar] [CrossRef]
- Moriggi, M.; Vasso, M.; Fania, C.; Capitanio, D.; Bonifacio, G.; Salanova, M.; Blottner, D.; Rittweger, J.; Felsenberg, D.; Cerretelli, P.; et al. Long term bed rest with and without vibration exercise countermeasures: Effects on human muscle protein dysregulation. Proteomics 2010, 10, 3756–3774. [Google Scholar] [CrossRef] [PubMed]
- Salanova, M.; Gelfi, C.; Moriggi, M.; Vasso, M.; Viganò, A.; Minafra, L.; Bonifacio, G.; Schiffl, G.; Gutsmann, M.; Felsenberg, D.; et al. Disuse deterioration of human skeletal muscle challenged by resistive exercise superimposed with vibration: Evidence from structural and proteomic analysis. FASEB J. 2014, 28, 4748–4763. [Google Scholar] [CrossRef] [PubMed]
- Rittweger, J.; Albracht, K.; Fluck, M.; Ruoss, S.; Brocca, L.; Longa, E.; Moriggi, M.; Seynnes, O.; Di Giulio, I.; Tenori, L.; et al. Erratum: Author Correction: Sarcolab Pilot Study into Skeletal Muscle’s Adaptation to Longterm Spaceflight. NPJ Microgravity 2018, 4, 23. [Google Scholar] [CrossRef] [PubMed]
- Capitanio, D.; Fania, C.; Torretta, E.; Viganò, A.; Moriggi, M.; Bravatà, V.; Caretti, A.; Levett, D.Z.H.; Grocott, M.P.W.; Samaja, M.; et al. TCA cycle rewiring fosters metabolic adaptation to oxygen restriction in skeletal muscle from rodents and humans. Sci. Rep. 2017, 7, 9723. [Google Scholar] [CrossRef]
- Celegato, B.; Capitanio, D.; Pescatori, M.; Romualdi, C.; Pacchioni, B.; Cagnin, S.; Vigano, A.; Colantoni, L.; Begum, S.; Ricci, E.; et al. Parallel Protein and Transcript Profiles of Fshd Patient Muscles Correlate to the D4z4 Arrangement and Reveal a Common Impairment of Slow to Fast Fibre Differentiation and a General Deregulation of My-od-Dependent Genes. Proteomics 2006, 6, 5303–5321. [Google Scholar] [CrossRef] [PubMed]
- De Palma, S.; Capitanio, D.; Vasso, M.; Braghetta, P.; Scotton, C.; Bonaldo, P.; Lochmüller, H.; Muntoni, F.; Ferlini, A.; Gelfi, C. Muscle Proteomics Reveals Novel Insights into the Pathophysiological Mechanisms of Collagen VI Myopathies. J. Proteome Res. 2014, 13, 5022–5030. [Google Scholar] [CrossRef]
- Chiarelli, N.; Zoppi, N.; Ritelli, M.; Venturini, M.; Capitanio, D.; Gelfi, C.; Colombi, M. Biological insights in the pathogenesis of hypermobile Ehlers-Danlos syndrome from proteome profiling of patients’ dermal myofibroblasts. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2021, 1867, 166051. [Google Scholar] [CrossRef]
- Faid, V.; Michalski, J.-C.; Morelle, W. A mass spectrometric strategy for profiling glycoproteinoses, Pompe disease, and sialic acid storage diseases. Proteom. Clin. Appl. 2008, 2, 528–542. [Google Scholar] [CrossRef] [PubMed]
- Sidorina, A.; Catesini, G.; Mortera, S.L.; Marzano, V.; Putignani, L.; Boenzi, S.; Taurisano, R.; Garibaldi, M.; Deodato, F.; Dionisi-Vici, C. Combined proteomic and lipidomic studies in Pompe disease allow a better disease mechanism understanding. J. Inherit. Metab. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Myers, M.J.; Shepherd, D.L.; Durr, A.J.; Stanton, D.S.; Mohamed, J.S.; Hollander, J.M.; Alway, S.E. The role of SIRT1 in skeletal muscle function and repair of older mice. J. Cachex- Sarcopenia Muscle 2019, 10, 929–949. [Google Scholar] [CrossRef]
- Fu, R.; Deng, Q.; Zhang, H.; Hu, X.; Li, Y.; Liu, Y.; Hu, J.; Luo, Q.; Zhang, Y.; Jiang, X.; et al. A novel autophagy inhibitor berbamine blocks SNARE-mediated autophagosome-lysosome fusion through upregulation of BNIP3. Cell Death Dis. 2018, 9, 1–15. [Google Scholar] [CrossRef]
- Conley, K.E.; Amara, C.E.; Jubrias, S.A.; Marcinek, D.J. Mitochondrial Function, Fibre Types and Ageing: New Insights from Human Muscle in Vivo. Exp. Physiol. 2007, 92, 333–339. [Google Scholar] [CrossRef]
- Conley, K.E.; Marcinek, D.J.; Villarin, J. Mitochondrial dysfunction and age. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 688–692. [Google Scholar] [CrossRef] [PubMed]
- Ait-Aissa, K.; Blaszak, S.C.; Beutner, G.; Tsaih, S.-W.; Morgan, G.; Santos, J.H.; Flister, M.J.; Joyce, D.L.; Camara, A.K.S.; Gutterman, D.D.; et al. Mitochondrial Oxidative Phosphorylation Defect in the Heart of Subjects with Coronary Artery Disease. Sci. Rep. 2019, 9, 7623. [Google Scholar] [CrossRef]
- Sánchez-González, C.; Nuevo-Tapioles, C.; Martín, J.C.H.; Pereira, M.P.; Sanz, S.S.; De Molina, A.R.; Cuezva, J.M.; Formentini, L. Dysfunctional oxidative phosphorylation shunts branched-chain amino acid catabolism onto lipogenesis in skeletal muscle. EMBO J. 2020, 39, e103812. [Google Scholar] [CrossRef] [PubMed]
- Arndt, V.; Daniel, C.; Nastainczyk, W.; Alberti, S.; Höhfeld, J. BAG-2 Acts as an Inhibitor of the Chaperone-associated Ubiquitin Ligase CHIP. Mol. Biol. Cell 2005, 16, 5891–5900. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Guo, J.; Zheng, Q.; Zhang, H. BAG2 structure, function and involvement in disease. Cell. Mol. Biol. Lett. 2016, 21, 18. [Google Scholar] [CrossRef]
- Hanus, J.; Anderson, C.; Wang, S. RPE necroptosis in response to oxidative stress and in AMD. Ageing Res. Rev. 2015, 24, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Meena, N.K.; Ralston, E.; Raben, N.; Puertollano, R. Enzyme Replacement Therapy Can Reverse Pathogenic Cascade in Pompe Disease. Mol. Ther. Methods Clin. Dev. 2020, 18, 199–214. [Google Scholar] [CrossRef]
- Lim, J.-A.; Sun, B.; Puertollano, R.; Raben, N. Therapeutic Benefit of Autophagy Modulation in Pompe Disease. Mol. Ther. 2018, 26, 1783–1796. [Google Scholar] [CrossRef]
- Cai, Y.; Arikkath, J.; Yang, L.; Guo, M.-L.; Periyasamy, P.; Buch, S. Interplay of endoplasmic reticulum stress and autophagy in neurodegenerative disorders. Autophagy 2016, 12, 225–244. [Google Scholar] [CrossRef] [PubMed]
- Reynolds-Peterson, C.E.; Zhao, N.; Xu, J.; Serman, T.M.; Xu, J.; Selleck, S.B. Heparan sulfate proteoglycans regulate autophagy in Drosophila. Autophagy 2017, 13, 1262–1279. [Google Scholar] [CrossRef] [PubMed]
- Reynolds-Peterson, C.; Xu, J.; Zhao, N.; Cruse, C.; Yonel, B.; Trasorras, C.; Toyoda, H.; Kinoshita-Toyoda, A.; Dobson, J.; Schultheis, N.; et al. Heparan Sulfate Structure Affects Autophagy, Lifespan, Responses to Oxidative Stress, and Cell Degeneration in Drosophila parkin Mutants. G3 Genes Genomes Genet. 2020, 10, 129–141. [Google Scholar] [CrossRef]
- Fiorenza, M.T.; Moro, E.; Erickson, R.P. The pathogenesis of lysosomal storage disorders: Beyond the engorgement of lysosomes to abnormal development and neuroinflammation. Hum. Mol. Genet. 2018, 27, R119–R129. [Google Scholar] [CrossRef]
- Burton, T.R.; Gibson, S.B. The role of Bcl-2 family member BNIP3 in cell death and disease: NIPping at the heels of cell death. Cell Death Differ. 2009, 16, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Dávila, D.; Connolly, N.M.C.; Bonner, H.; Weisová, P.; Düssmann, H.; Concannon, C.G.; Huber, H.J.; Prehn, J.H.M. Two-step activation of FOXO3 by AMPK generates a coherent feed-forward loop determining excitotoxic cell fate. Cell Death Differ. 2012, 19, 1677–1688. [Google Scholar] [CrossRef]
- Picca, A.; Lezza, A.M.S.; Leeuwenburgh, C.; Pesce, V.; Calvani, R.; Bossola, M.; Manes-Gravina, E.; Landi, F.; Bernabei, R.; Marzetti, E. Circulating Mitochondrial DNA at the Crossroads of Mitochondrial Dysfunction and Inflammation During Aging and Muscle Wasting Disorders. Rejuvenation Res. 2018, 21, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Hall, A. Rho GTPases and the Actin Cytoskeleton. Science 1998, 279, 509–514. [Google Scholar] [CrossRef]
- Wang, H.; Ding, B.; Liu, C.; Ma, X.; Deschamps, S.; Roe, B.; Lengyel, P. The Increase in Levels of Interferon-Inducible Proteins p202a and p202b and RNA-Dependent Protein Kinase (PKR) During Myoblast Differentiation Is Due to Transactivation by MyoD: Their Tissue Distribution in Uninfected Mice Does Not Depend on Interferons. J. Interf. Cytokine Res. 2002, 22, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Schaaf, G.J.; van Gestel, T.J.; Brusse, E.; Verdijk, R.M.; de Coo, I.F.; van Doorn, P.A.; van der Ploeg, A.T.; Pijnappel, W.W. Lack of Robust Satellite Cell Activation and Muscle Regeneration During the Progression of Pompe Disease. Acta Neuropathol Commun. 2015, 3, 65. [Google Scholar] [CrossRef]
- Raben, N.; Takikita, S.; Pittis, M.G.; Bembi, B.; Marie, S.K.; Roberts, A.; Page, L.; Kishnani, P.S.; Schoser, B.G.; Chien, Y.H.; et al. Deconstructing Pompe Disease by Analyzing Single Muscle Fibers: To See a World in a Grain of Sand. Autophagy 2007, 3, 546–552. [Google Scholar] [CrossRef]
- Lange, S.; Pinotsis, N.; Agarkova, I.; Ehler, E. The M-band: The underestimated part of the sarcomere. Biochim. Biophys. Acta (BBA) Bioenerg. 2020, 1867, 118440. [Google Scholar] [CrossRef]
- Kontrogianni-Konstantopoulos, A.; Bloch, R.J. Obscurin: A multitasking muscle giant. J. Muscle Res. Cell Motil. 2006, 26, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Kontrogianni-Konstantopoulos, A.; Catino, D.H.; Strong, J.C.; Sutter, S.; Borisov, A.B.; Pumplin, D.W.; Russell, M.W.; Bloch, R.J. Obscurin modulates the assembly and organization of sarcomeres and the sarcoplasmic reticulum. FASEB J. 2006, 20, 2102–2111. [Google Scholar] [CrossRef]
- Fernando, R.; Drescher, C.; Nowotny, K.; Grune, T.; Castro, J.P. Impaired proteostasis during skeletal muscle aging. Free. Radic. Biol. Med. 2019, 132, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Zhong, Q. A tethering coherent protein in autophagosome maturation. Autophagy 2012, 8, 985–986. [Google Scholar] [CrossRef][Green Version]
- Kriegenburg, F.; Ungermann, C.; Reggiori, F. Coordination of Autophagosome–Lysosome Fusion by Atg8 Family Members. Curr. Biol. 2018, 28, R512–R518. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Shi, Y.; Liu, J.; Su, H.; Huang, J.; Zhang, Y.; Peng, C.; Zhou, T.; Sun, Q.; Wan, W.; et al. Acetylation of STX17 (syntaxin 17) controls autophagosome maturation. Autophagy 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mammucari, C.; Schiaffino, S.; Sandri, M. Downstream of Akt: FoxO3 and mTOR in the regulation of autophagy in skeletal muscle. Autophagy 2008, 4, 524–526. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.; Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 2008, 26, 1367–1372. [Google Scholar] [CrossRef]
- Cox, J.; Hein, M.Y.; Luber, C.A.; Paron, I.; Nagaraj, N.; Mann, M. Accurate Proteome-wide Label-free Quantification by Delayed Normalization and Maximal Peptide Ratio Extraction, Termed MaxLFQ. Mol. Cell. Proteom. 2014, 13, 2513–2526. [Google Scholar] [CrossRef] [PubMed]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.; Green, J.; Pollard, J., Jr.; Tugendreich, S. Causal Analysis Approaches in Ingenuity Pathway Analysis. Bioinformatics 2014, 30, 523–530. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moriggi, M.; Capitanio, D.; Torretta, E.; Barbacini, P.; Bragato, C.; Sartori, P.; Moggio, M.; Maggi, L.; Mora, M.; Gelfi, C. Muscle Proteomic Profile before and after Enzyme Replacement Therapy in Late-Onset Pompe Disease. Int. J. Mol. Sci. 2021, 22, 2850. https://doi.org/10.3390/ijms22062850
Moriggi M, Capitanio D, Torretta E, Barbacini P, Bragato C, Sartori P, Moggio M, Maggi L, Mora M, Gelfi C. Muscle Proteomic Profile before and after Enzyme Replacement Therapy in Late-Onset Pompe Disease. International Journal of Molecular Sciences. 2021; 22(6):2850. https://doi.org/10.3390/ijms22062850
Chicago/Turabian StyleMoriggi, Manuela, Daniele Capitanio, Enrica Torretta, Pietro Barbacini, Cinzia Bragato, Patrizia Sartori, Maurizio Moggio, Lorenzo Maggi, Marina Mora, and Cecilia Gelfi. 2021. "Muscle Proteomic Profile before and after Enzyme Replacement Therapy in Late-Onset Pompe Disease" International Journal of Molecular Sciences 22, no. 6: 2850. https://doi.org/10.3390/ijms22062850
APA StyleMoriggi, M., Capitanio, D., Torretta, E., Barbacini, P., Bragato, C., Sartori, P., Moggio, M., Maggi, L., Mora, M., & Gelfi, C. (2021). Muscle Proteomic Profile before and after Enzyme Replacement Therapy in Late-Onset Pompe Disease. International Journal of Molecular Sciences, 22(6), 2850. https://doi.org/10.3390/ijms22062850






