Suppression of Hypoxia-Inducible Factor 1α by Low-Molecular-Weight Heparin Mitigates Ventilation-Induced Diaphragm Dysfunction in a Murine Endotoxemia Model
Abstract
1. Introduction
2. Results
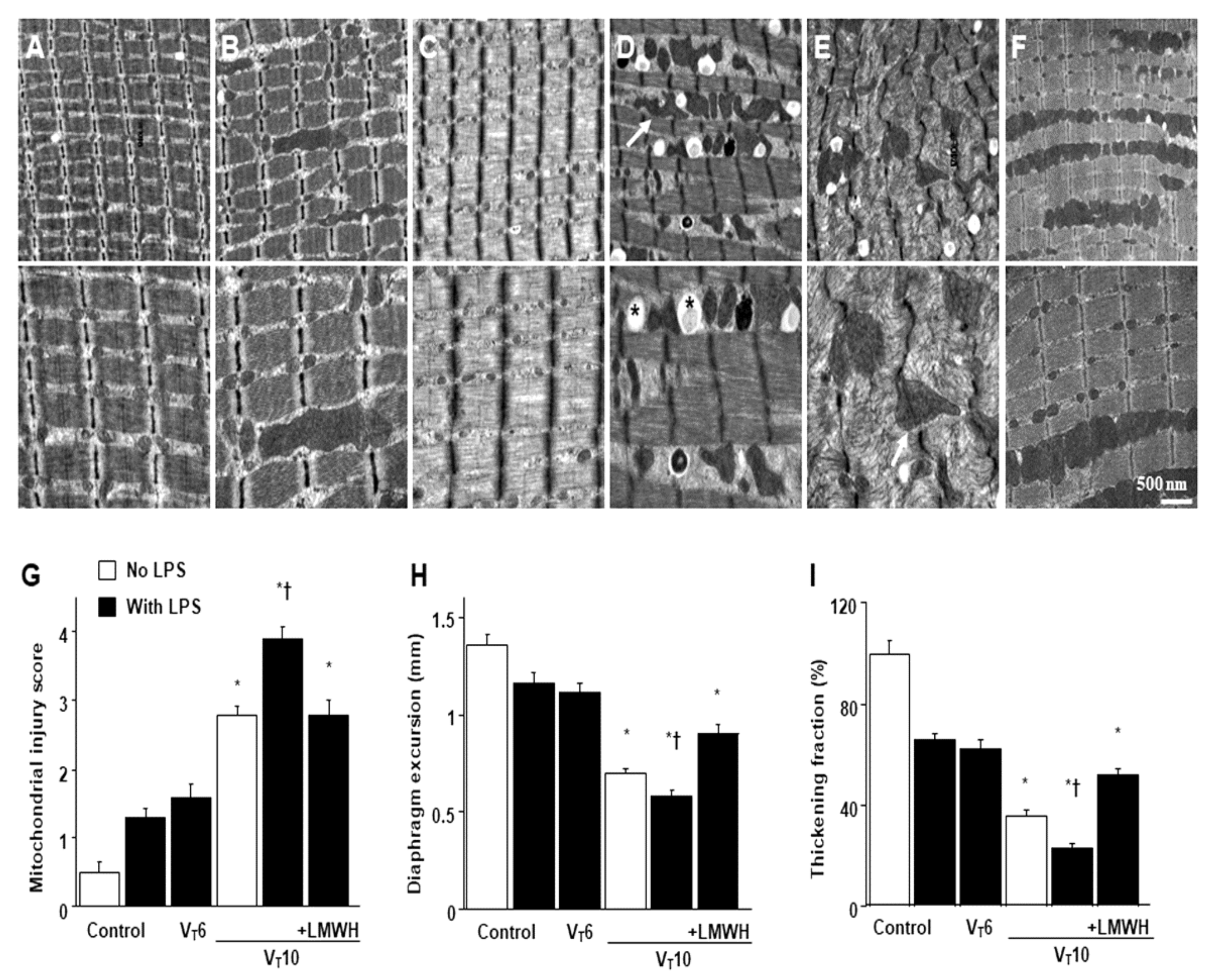
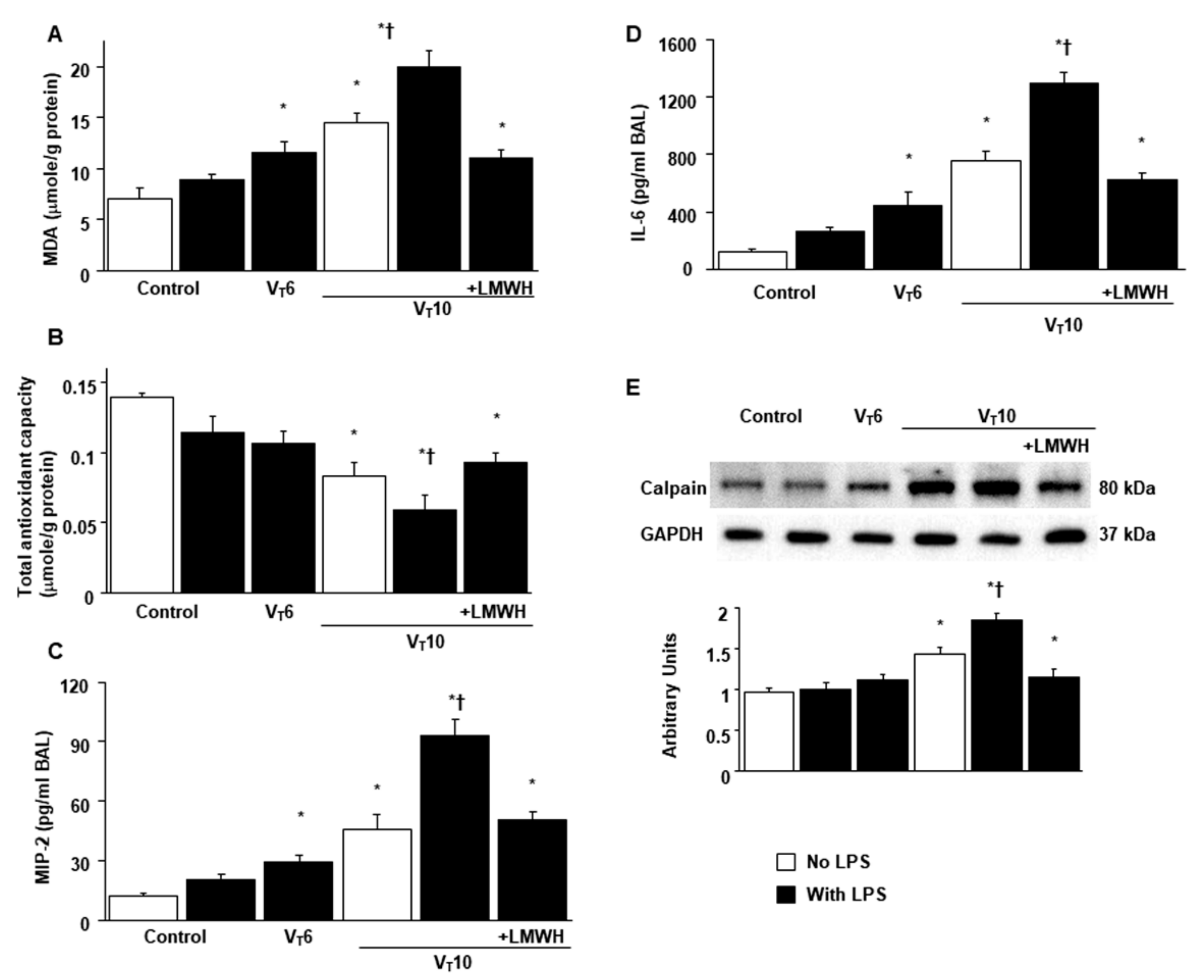
2.1. Reduction of Endotoxin-Enhanced MV-Induced VIDD, Diaphragmatic Oxygen Radicals, and Inflammatory Cytokines by Enoxaparin
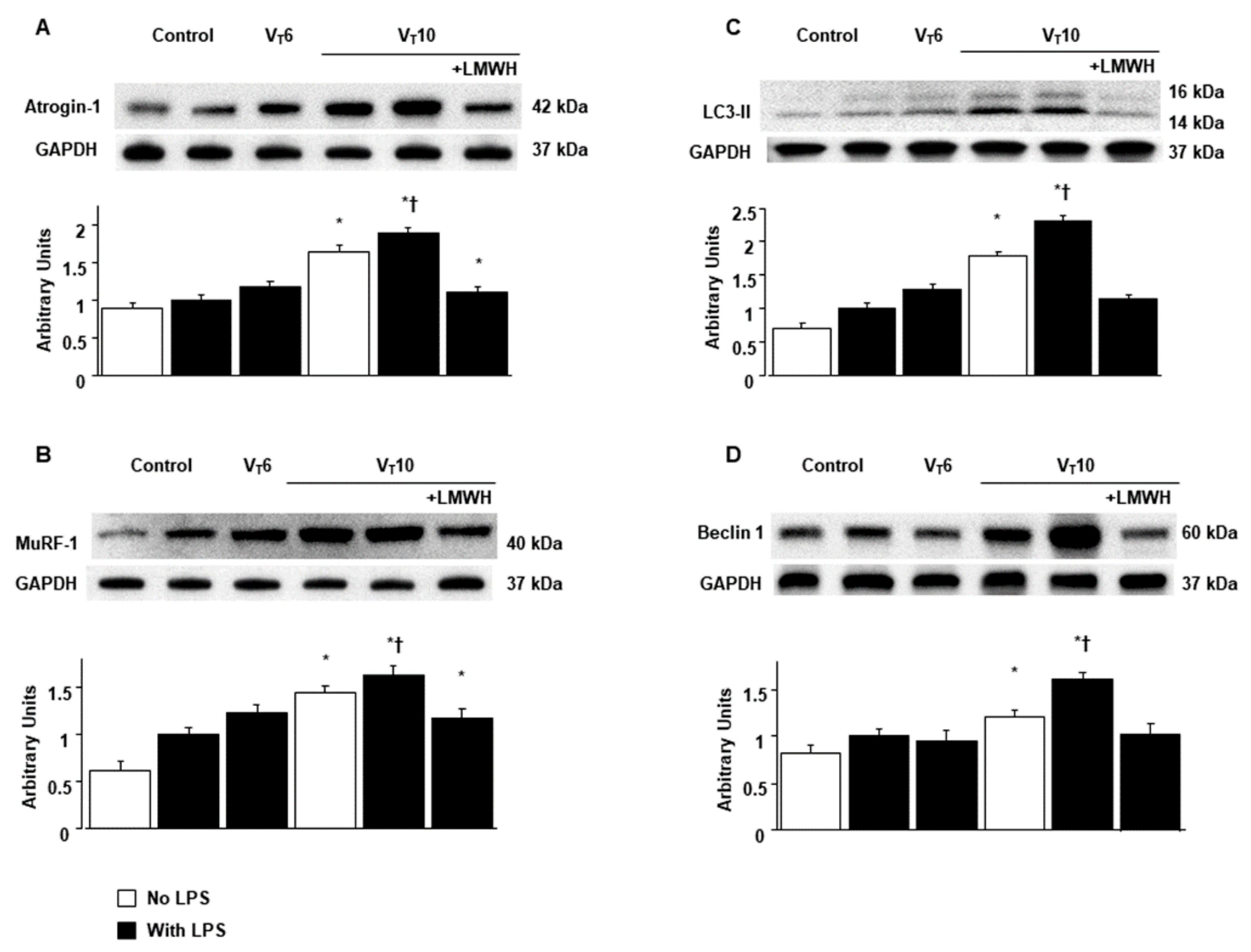
2.2. Suppression of Endotoxin-Augmented MV-Induced Diaphragmatic Calpain, Atrogin-1, and Murf-1 Expression and Autophagy by Enoxaparin
2.3. Inhibition of Endotoxin-Stimulated MV-Induced Diaphragmatic HIF-1α mRNA and HIF-1α Protein Expression by Enoxaparin
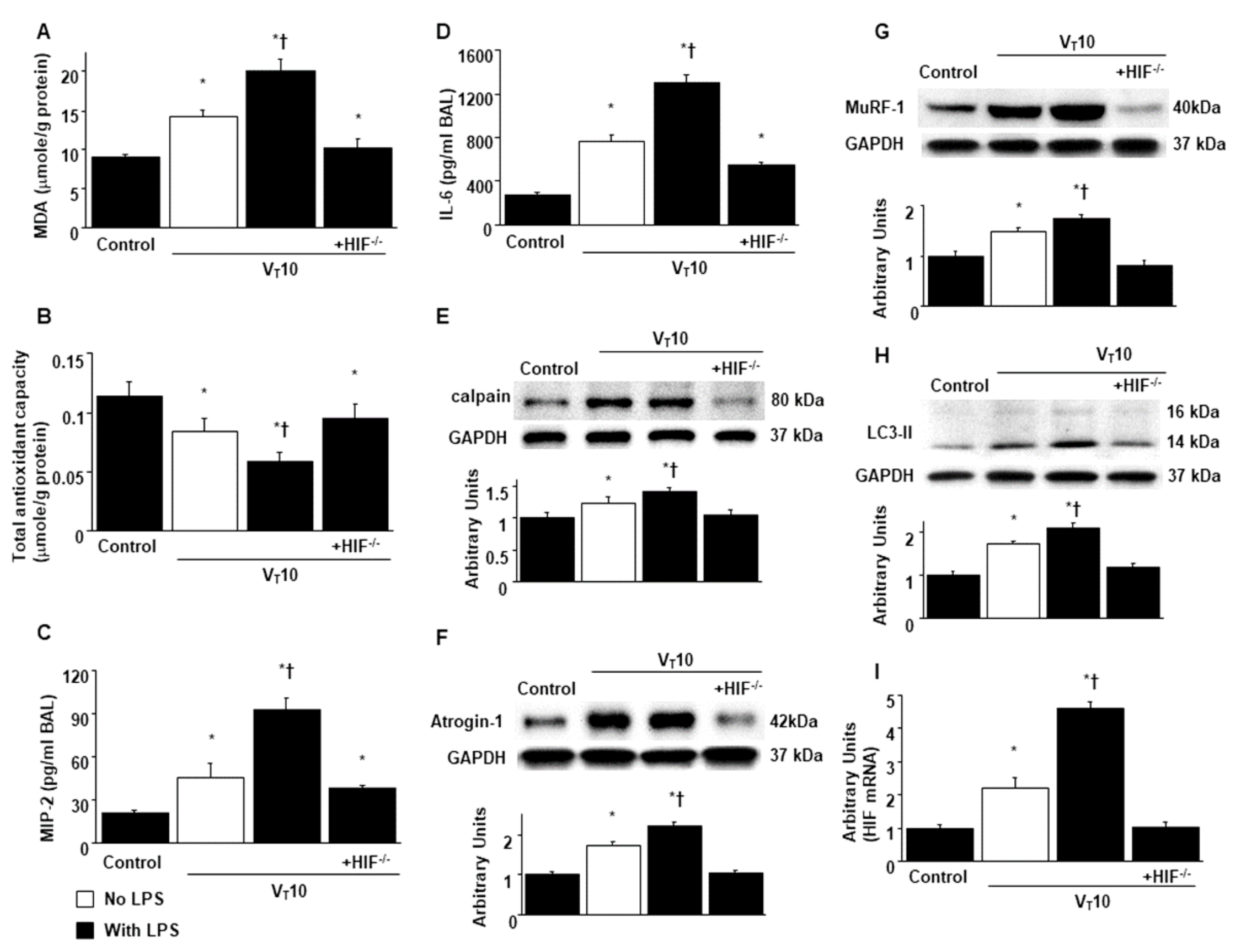
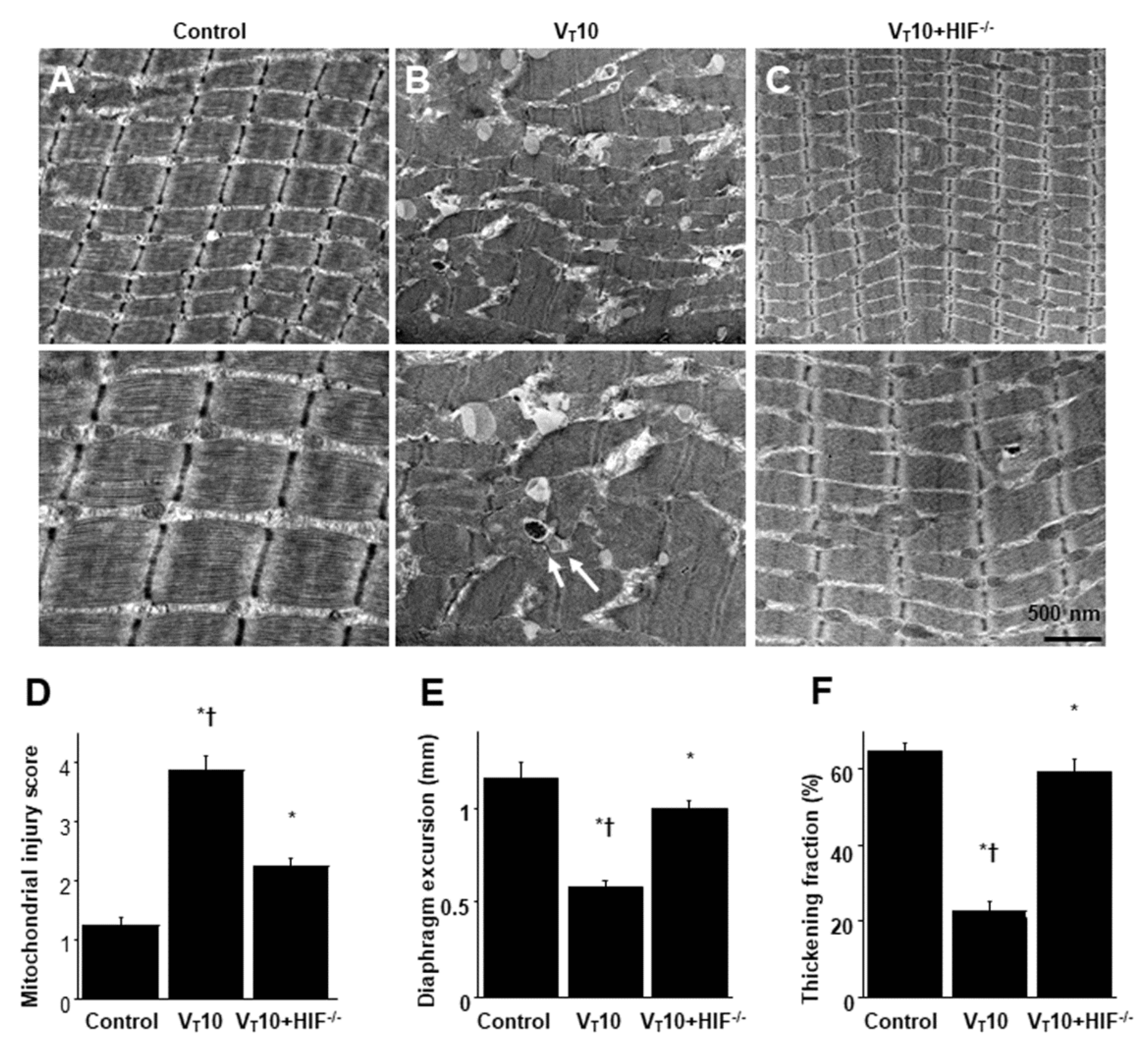
2.4. Reduction of Endotoxin-Enhanced VIDD in HIF-1α–Deficient Mice
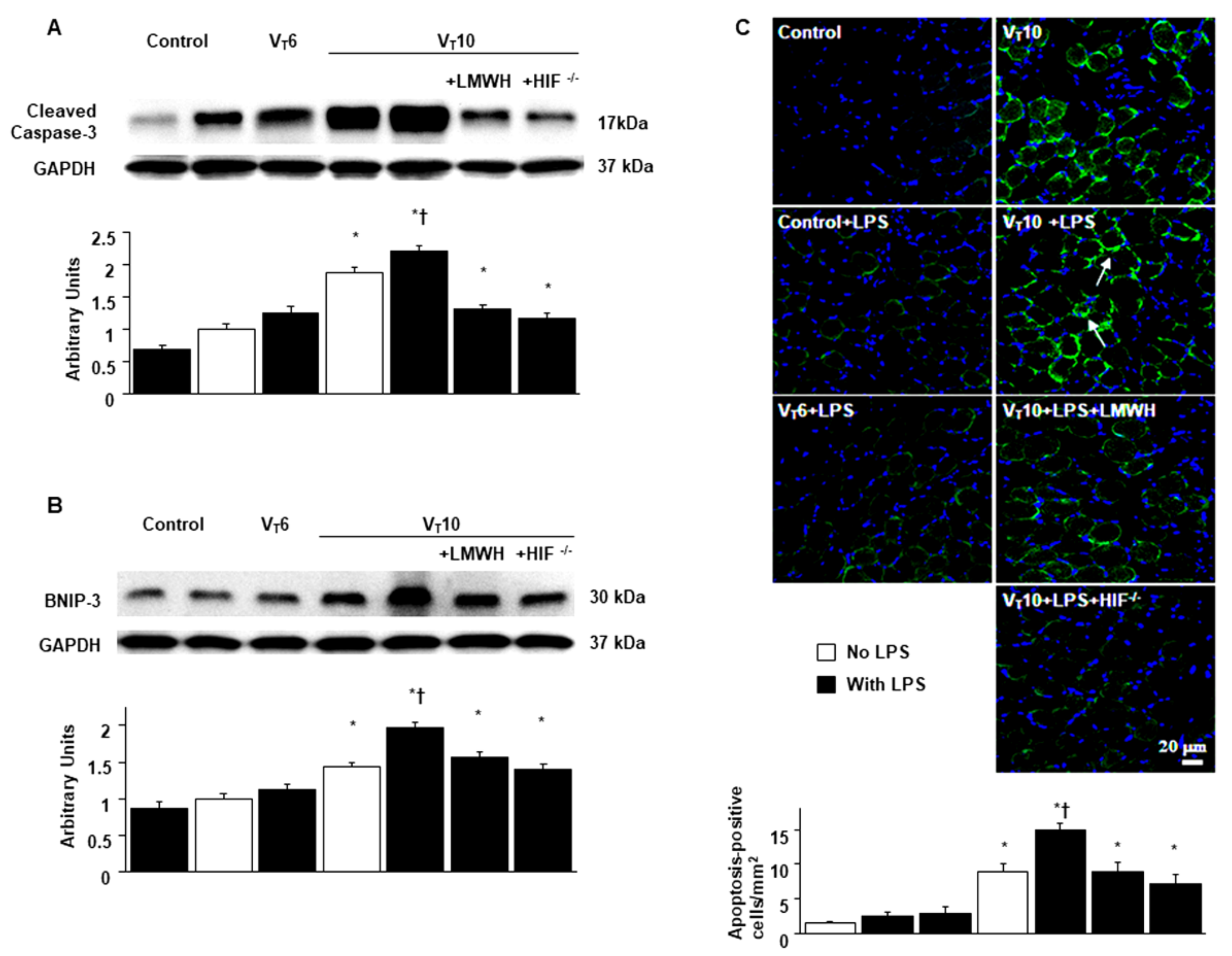
2.5. Suppression of Endotoxin-Augmented MV-Induced Diaphragmatic Expression of Caspase-3 and BNIP-3 and Epithelial Apoptosis by Enoxaparin in HIF-1α-Deficient Mice
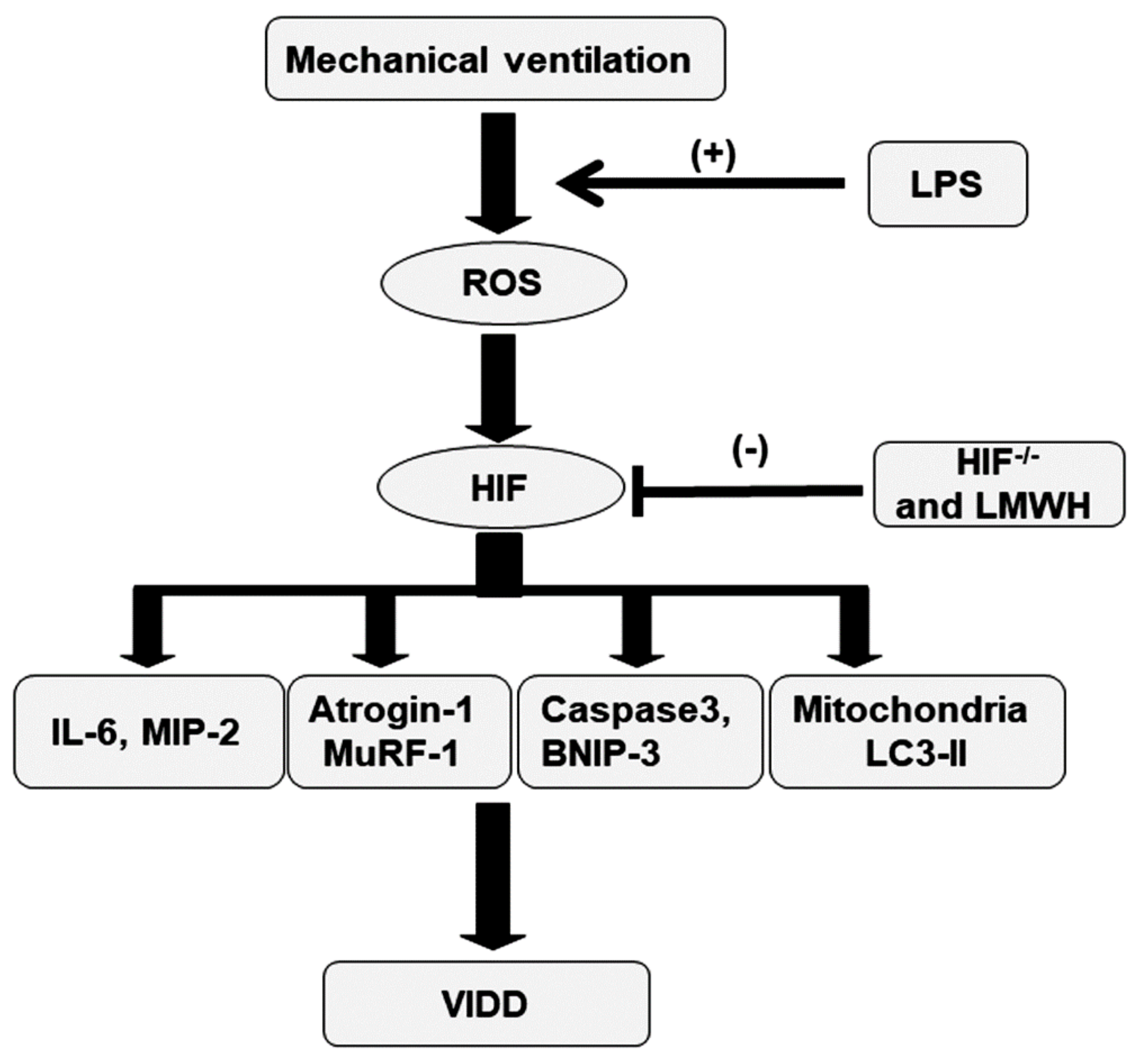
3. Discussion
4. Materials and Methods
4.1. Experimental Animals
4.2. Experimental Groups
4.3. Lipopolysaccharide Administration
4.4. Enoxaparin Administration
4.5. Measurement of Diaphragm Excursion and Thickness
4.6. Transmission Electron Microscopy
4.7. Immunoblot Analysis
4.8. Real-Time Polymerase Chain Reaction
4.9. Analysis of Data
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Dehne, N.; Brüne, B. HIF-1 in the inflammatory microenvironment. Exp. Cell Res. 2009, 315, 1791–1797. [Google Scholar] [CrossRef]
- Peyssonnaux, C.; Cejudo-Martin, P.; Doedens, A.; Zinkernagel, A.S.; Johnson, R.S.; Nizet, V. Cutting Edge: Essential Role of Hypoxia Inducible Factor-1α in Development of Lipopolysaccharide-Induced Sepsis. J. Immunol. 2007, 178, 7516–7519. [Google Scholar] [CrossRef]
- Maes, K.; Stamiris, A.; Thomas, D.; Cielen, N.; Smuder, A.; Powers, S.K.; Leite, F.D.S.; Hermans, G.; Decramer, M.; Hussain, S.N.; et al. Effects of Controlled Mechanical Ventilation on Sepsis-Induced Diaphragm Dysfunction in Rats. Crit. Care Med. 2014, 42, e772–e782. [Google Scholar] [CrossRef]
- Picard, M.; Liang, F.; Hussain, S.N.; Goldberg, P.; Danialou, G.; Chaturvedi, R.; Matecki, S.; Samir, J.; Rosiers, C.D.; Karpati, G.; et al. Mitochondrial Dysfunction and Lipid Accumulation in The Human Diaphragm During Mechanical Ventilation. Am. J. Respir. Crit. Care Med. 2012, 186, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Huang, Y.; Xu, H.; Hu, R.; Li, Q.-F. Inhibition of hypoxia inducible factor-1α ameliorates lung injury induced by trauma and hemorrhagic shock in rats. Acta Pharmacol. Sin. 2012, 33, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. A compendium of proteins that interact with HIF-1α. Exp. Cell Res. 2017, 356, 128–135. [Google Scholar] [CrossRef]
- Petrof, B.J. Diaphragm Weakness in the Critically Ill. Chest 2018, 154, 1395–1403. [Google Scholar] [CrossRef]
- Demoule, A.; Molinari, N.; Jung, B.; Prodanovic, H.; Chanques, G.; Matecki, S.; Mayaux, J.; Similowski, T.; Jaber, S. Patterns of diaphragm function in critically ill patients receiving prolonged mechanical ventilation: A prospective longitudinal study. Ann. Intensiv. Care 2016, 6, 1–8. [Google Scholar] [CrossRef]
- Jung, B.; Moury, P.H.; Mahul, M.; de Jong, A.; Galia, F.; Prades, A.; Albaladejo, P.; Chanques, G.; Molinari, N.; Jaber, S. Diaphragmatic dysfunction in patients with ICU-acquired weakness and its impact on extubation failure. Intensiv. Care Med. 2016, 42, 853–861. [Google Scholar] [CrossRef]
- Jaber, S.; Petrof, B.J.; Jung, B.; Chanques, G.; Berthet, J.-P.; Rabuel, C.; Bouyabrine, H.; Courouble, P.; Koechlin-Ramonatxo, C.; Sebbane, M.; et al. Rapidly Progressive Diaphragmatic Weakness and Injury during Mechanical Ventilation in Humans. Am. J. Respir. Crit. Care Med. 2011, 183, 364–371. [Google Scholar] [CrossRef]
- Textoris, J.; Beaufils, N.; Quintana, G.; Ben-Lassoued, A.; Zieleskiewicz, L.; Wiramus, S.; Blasco, V.; Lesavre, N.; Martin, C.; Gabert, J.; et al. Hypoxia-inducible factor (HIF1α) gene expression in human shock states. Crit. Care 2012, 16, R120. [Google Scholar] [CrossRef]
- Supinski, G.S.; Callahan, L.A. Diaphragm weakness in mechanically ventilated critically ill patients. Crit. Care 2013, 17, R120. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Shrager, J.B. The Signaling Network Resulting in Ventilator-induced Diaphragm Dysfunction. Am. J. Respir. Cell Mol. Biol. 2018, 59, 417–427. [Google Scholar] [CrossRef]
- Bogdanovski, D.A.; di Fazio, L.T.; Bogdanovski, A.K.; Csóka, B.; Jordan, G.B.; Paul, E.R.; Antonioli, L.; Pilip, S.A.; Nemeth, Z.H. Hypoxia-inducible-factor-1 in trauma and critical care. J. Crit. Care 2017, 42, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Richard, D.E.; Berra, E.; Pouyssegur, J. Nonhypoxic pathway mediates the induction of hypoxia-inducible factor 1alpha in vascular smooth muscle cells. J. Biol. Chem. 2000, 275, 26765–26771. [Google Scholar] [CrossRef]
- Jung, Y.; Isaacs, J.S.; Lee, S.; Trepel, J.; Liu, Z.-G.; Neckers, L. Hypoxia-inducible factor induction by tumour necrosis factor in normoxic cells requires receptor-interacting protein-dependent nuclear factor kappaB activation. Biochem. J. 2003, 370, 1011–1017. [Google Scholar] [CrossRef]
- Bruells, C.S.; Maes, K.; Rossaint, R.; Thomas, D.; Cielen, N.; Bleilevens, C.; Bergs, I.; Loetscher, U.; Dreier, A.; Gayan-Ramirez, G.; et al. Prolonged Mechanical Ventilation Alters the Expression Pattern of Angio-neogenetic Factors in a Pre-Clinical Rat Model. PLoS ONE 2013, 8, e70524. [Google Scholar] [CrossRef]
- Santos, S.A.D.; Andrade, D.R.J. HIF-1alpha and infectious diseases: A new frontier for the development of new therapies. Rev. Inst. Med. Trop. Sao Paulo 2017, 59, e92. [Google Scholar] [CrossRef]
- Epstein, A.C.; Gleadle, J.M.; McNeill, L.A.; Hewitson, K.S.; O’Rourke, J.; Mole, D.R.; Mukherji, M.; Metzen, E.; Wilson, M.I.; Dhanda, A.; et al. elegans EGL-9 and Mammalian Homologs Define a Family of Dioxygenases that Regulate HIF by Prolyl Hydroxylation. Cell 2001, 107, 43–54. [Google Scholar] [CrossRef]
- Hirota, K. Involvement of Hypoxia-Inducible Factors in the Dysregulation of Oxygen Homeostasis in Sepsis. Cardiovasc. Hematol. Disord. Targets 2015, 15, 29–40. [Google Scholar] [CrossRef]
- Pisani, D.F.; Dechesne, C.A. Skeletal Muscle HIF-1α Expression Is Dependent on Muscle Fiber Type. J. Gen. Physiol. 2005, 126, 173–178. [Google Scholar] [CrossRef]
- Picard, M.; Azuelos, I.; Jung, B.; Giordano, C.; Matecki, S.; Hussain, S.N.A.; White, K.; Li, T.; Liang, F.; Benedetti, A.; et al. Mechanical ventilation triggers abnormal mitochondrial dynamics and morphology in the diaphragm. J. Appl. Physiol. 2015, 118, 1161–1171. [Google Scholar] [CrossRef]
- Kavazis, A.N.; Talbert, E.E.; Smuder, A.J.; Hudson, M.B.; Nelson, W.B.; Powers, S.K. Mechanical ventilation induces diaphragmatic mitochondrial dysfunction and increased oxidant production. Free. Radic. Biol. Med. 2009, 46, 842–850. [Google Scholar] [CrossRef]
- Cornet, A.D.; Smit, E.G.; Beishuizen, A.; Groeneveld, A.B. The role of heparin and allied compounds in the treatment of sepsis. Thromb. Haemost. 2007, 98, 579–586. [Google Scholar] [CrossRef]
- Zarychanski, R.; Abou-Setta, A.; Kanji, S.; Turgeon, A.; Kumar, A.; Houston, D.; Rimmer, E.; Houston, B.; McIntyre, L.; Fox-Robichaud, A.; et al. Efficacy and safety of heparin in patients with sepsis: A systematic review and meta-analysis. Crit. Care 2015, 19, P123. [Google Scholar] [CrossRef]
- Fan, Y.; Jiang, M.; Gong, D.; Zou, C. Efficacy and safety of low-molecular-weight heparin in patients with sepsis: A meta-analysis of randomized controlled trials. Sci. Rep. 2016, 6, 25984. [Google Scholar] [CrossRef]
- Li, L.-F.; Huang, C.-C.; Lin, H.-C.; Tsai, Y.-H.; A Quinn, D.; Liao, S.-K. Unfractionated heparin and enoxaparin reduce high-stretch ventilation augmented lung injury: A prospective, controlled animal experiment. Crit. Care 2009, 13, R108. [Google Scholar] [CrossRef]
- Li, L.F.; Liu, Y.Y.; Chen, N.H.; Chen, Y.H.; Huang, C.C.; Kao, K.C.; Chang, C.H.; Chuang, L.P.; Chiu, L.C. Attenuation of ventilation-induced diaphragm dysfunction through toll-like receptor 4 and nuclear factor-kappaB in a murine endotoxemia model. Lab. Invest. 2018, 98, 1170–1183. [Google Scholar] [CrossRef]
- Supinski, G.S.; Morris, P.E.; Dhar, S.; Callahan, L.A. Diaphragm Dysfunction in Critical Illness. Chest 2018, 153, 1040–1051. [Google Scholar] [CrossRef]
- Kim, W.-Y.; Park, S.H.; Huh, J.W.; Hong, S.-B.; Koh, Y.; Lim, C.-M.; Kim, W.-Y. Effect of theophylline on ventilator-induced diaphragmatic dysfunction. J. Crit. Care 2016, 33, 145–150. [Google Scholar] [CrossRef]
- Eckle, T.; Brodsky, K.S.; Bonney, M.; Packard, T.A.; Han, J.; Borchers, C.H.; Mariani, T.J.; Kominsky, D.J.; Mittelbronn, M.; Eltzschig, M.H.K. HIF1A Reduces Acute Lung Injury by Optimizing Carbohydrate Metabolism in the Alveolar Epithelium. PLoS Biol. 2013, 11, e1001665. [Google Scholar] [CrossRef]
- Mofarrahi, M.; Sigala, I.; Guo, Y.; Godin, R.; Davis, E.C.; Petrof, B.; Sandri, M.; Burelle, Y.; Husain, S.N.A. Autophagy and Skeletal Muscles in Sepsis. PLoS ONE 2012, 7, e47265. [Google Scholar] [CrossRef]
- Smuder, A.J.; Sollanek, K.J.; Nelson, W.B.; Min, K.; Talbert, E.E.; Kavazis, A.N.; Hudson, M.B.; Sandri, M.; Szeto, H.H.; Powers, S.K. Crosstalk between autophagy and oxidative stress regulates proteolysis in the diaphragm during mechanical ventilation. Free. Radic. Biol. Med. 2018, 115, 179–190. [Google Scholar] [CrossRef]
- Demoule, A.; Jung, B.; Prodanovic, H.; Molinari, N.; Chanques, G.; Coirault, C.; Matecki, S.; Duguet, A.; Similowski, T.; Jaber, S. Diaphragm Dysfunction on Admission to the Intensive Care Unit. Prevalence, Risk Factors, and Prognostic Impact—A Prospective Study. Am. J. Respir. Crit. Care Med. 2013, 188, 213–219. [Google Scholar] [CrossRef]
- Azuelos, I.; Jung, B.; Picard, M.; Liang, F.; Li, T.; Lemaire, C.; Giordano, C.; Hussain, S.; Petrof, B.J. Relationship between Autophagy and Ventilator-induced Diaphragmatic Dysfunction. Anesthesiology 2015, 122, 1349–1361. [Google Scholar] [CrossRef]
- Davis, R.T.; Bruells, C.S.; Stabley, J.N.; McCullough, D.J.; Powers, S.K.; Behnke, B.J. Mechanical ventilation reduces rat diaphragm blood flow and impairs oxygen delivery and uptake. Crit. Care Med. 2012, 40, 2858–2866. [Google Scholar] [CrossRef] [PubMed]
- Haddad, J.J. Science review: Redox and oxygen-sensitive transcription factors in the regulation of oxidant-mediated lung injury: Role for hypoxia-inducible factor-1α. Crit. Care 2002, 7, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Li, L.-F.; Liu, Y.-Y.; Lin, S.-W.; Chang, C.-H.; Chen, N.-H.; Hung, C.-Y.; Lee, C.-S. Low-Molecular-Weight Heparin Reduces Ventilation-Induced Lung Injury through Hypoxia Inducible Factor-1α in a Murine Endotoxemia Model. Int. J. Mol. Sci. 2020, 21, 3097. [Google Scholar] [CrossRef]
- Shalova, I.N.; Lim, J.Y.; Chittezhath, M.; Zinkernagel, A.S.; Beasley, F.; Hernández-Jiménez, E.; Toledano, V.; Cubillos-Zapata, C.; Rapisarda, A.; Chen, J.; et al. Human Monocytes Undergo Functional Re-programming during Sepsis Mediated by Hypoxia-Inducible Factor-1α. Immunity 2015, 42, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Horn, A.G.; Davis, R.T.; Baumfalk, D.R.; Kunkel, O.N.; Bruells, C.S.; McCullough, D.J.; Opoku-Acheampong, A.B.; Poole, D.C.; Behnke, B.J. Impaired diaphragm resistance vessel vasodilation with prolonged mechanical ventilation. J. Appl. Physiol. 2019, 127, 423–431. [Google Scholar] [CrossRef]
- Ning, F.; Wang, X.; Shang, L.; Wang, T.; Lv, C.; Qi, Z.; Wu, D. Low molecular weight heparin may prevent acute lung injury induced by sepsis in rats. Gene 2015, 557, 88–91. [Google Scholar] [CrossRef]
- Luan, Z.-G.; Naranpurev, M.; Ma, X.-C. Treatment of Low Molecular Weight Heparin Inhibits Systemic Inflammation and Prevents Endotoxin-Induced Acute Lung Injury in Rats. Inflammation 2014, 37, 924–932. [Google Scholar] [CrossRef] [PubMed]
- Levi, M.; van der Poll, T. Inflammation and coagulation. Crit. Care Med. 2010, 38 (Suppl. 2), 414–422. [Google Scholar] [CrossRef]
- Li, J.; Li, Y.; Yang, B.; Wang, H.; Li, L. Low-molecular-weight heparin treatment for acute lung injury acute respiratory distress syndrome a meta-analysis of randomized controlled trials.pdf. Int. J. Clin. Exp. 2018, 11, 414–422. [Google Scholar]
- Lemos, A.C.B.; Santo, D.A.D.E.; Salvetti, M.C.; Gilio, R.N.; Agra, L.B.; Pazin-Filho, A.; Miranda, C.H. Therapeutic versus prophylactic anticoagulation for severe COVID-19: A randomized phase II clinical trial (HESACOVID). Thromb. Res. 2020, 196, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Guo, Z.Y.; Gao, X.H.; Bian, Q.; Jia, M.; Lai, X.L.; Wang, T.Y.; Bian, X.L.; Wang, H.Y. Low Molecular Weight Heparin (LMWH) Improves Peritoneal Function and Inhibits Peritoneal Fibrosis Possibly through Suppression of HIF-1α, VEGF and TGF-β1. PLoS ONE 2015, 10, e0118481. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guo, R.-F.; Ward, P.A. Role of Oxidants in Lung Injury During Sepsis. Antioxid. Redox Signal. 2007, 9, 1991–2002. [Google Scholar] [CrossRef]
- Kim, W.-Y.; Lim, C.-M. Ventilator-Induced Diaphragmatic Dysfunction: Diagnosis and Role of Pharmacological Agents. Respir. Care 2017, 62, 1485–1491. [Google Scholar] [CrossRef] [PubMed]
- Elshazly, M.I.; Kamel, K.M.; Elkorashy, R.I.; Ismail, M.S.; Ismail, J.H.; Assal, H.H. Role of Bedside Ultrasonography in Assessment of Diaphragm Function as a Predictor of Success of Weaning in Mechanically Ventilated Patients. Tuberc. Respir. Dis. 2020, 83, 295–302. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, H.; Xiaoyin, M.; Yang, G.; Hu, Y.; Xie, K.; Yu, Y. Autophagy Activation Improves Lung Injury and Inflammation in Sepsis. Inflammation 2019, 42, 426–439. [Google Scholar] [CrossRef]
- Vishnupriya, S.; Dharshini, L.C.P.; Sakthivel, K.M.; Rasmi, R.R. Autophagy markers as mediators of lung injury-implication for therapeutic intervention. Life Sci. 2020, 260, 118308. [Google Scholar] [CrossRef]
- Hotchkiss, J.R.; Blanch, L.; Murias, G.; Adams, A.B.; Olson, D.A.; Wangensteen, O.D.; Leo, P.H.; Marini, J.J. Effects of Decreased Respiratory Frequency on Ventilator-induced Lung Injury. Am. J. Respir. Crit. Care Med. 2000, 161, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Pickford, F.; Masliah, E.; Britschgi, M.; Lucin, K.; Narasimhan, R.; Jaeger, P.A.; Small, S.; Spencer, B.; Rockenstein, E.; Levine, B.; et al. The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid beta accumulation in mice. J. Clin. Invest. 2008, 118, 2190–2199. [Google Scholar]
- Wang, X.; Du, Z.; Li, L.; Shi, M.; Yu, Y. Beclin 1 and p62 expression in non-small cell lung cancer: Relation with malignant behaviors and clinical outcome. Int. J. Clin. Exp. Pathol. 2015, 8, 10644–10652. [Google Scholar]
- Burton, T.R.; Gibson, S.B. The role of Bcl-2 family member BNIP3 in cell death and disease: NIPping at the heels of cell death. Cell Death Differ. 2009, 16, 515–523. [Google Scholar] [CrossRef]
- Regula, K.M.; Ens, K.; Kirshenbaum, L.A. Inducible Expression of BNIP3 Provokes Mitochondrial Defects and Hypoxia-Mediated Cell Death of Ventricular Myocytes. Circ. Res. 2002, 91, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Boussuges, A.; Gole, Y.; Blanc, P. Diaphragmatic motion studied by m-mode ultrasonography: Methods, reproducibility, and normal values. Chest 2009, 135, 391–400. [Google Scholar] [CrossRef]
- Goligher, E.C.; Laghi, F.; Detsky, M.E.; Farias, P.; Murray, A.; Brace, D.; Brochard, L.J.; Bolz, S.S.; Rubenfeld, G.D.; Kavanagh, B.P.; et al. Measuring diaphragm thickness with ultrasound in mechanically ventilated patients: Feasibility, reproducibility and validity. Intensive Care Med. 2015, 41, 734. [Google Scholar] [CrossRef] [PubMed]
- Lerolle, N.; Guérot, E.; Dimassi, S.; Zegdi, R.; Faisy, C.; Fagon, J.-Y.; Diehl, J.-L. Ultrasonographic Diagnostic Criterion for Severe Diaphragmatic Dysfunction After Cardiac Surgery. Chest 2009, 135, 401–407. [Google Scholar] [CrossRef]
- Summerhill, E.M.; Abu El-Sameed, Y.; Glidden, T.J.; McCool, F.D. Monitoring Recovery from Diaphragm Paralysis with Ultrasound. Chest 2008, 133, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.N.A.; Cornachione, A.S.; Guichon, C.; Al Khunaizi, A.; Leite, F.D.S.; Petrof, B.J.; Mofarrahi, M.; Moroz, N.; de Varennes, B.; Goldberg, P.; et al. Prolonged controlled mechanical ventilation in humans triggers myofibrillar contractile dysfunction and myofilament protein loss in the diaphragm. Thorax 2016, 71, 436–445. [Google Scholar] [CrossRef]
- Dang, E.V.; Barbi, J.; Yang, H.Y.; Jinasena, D.; Yu, H.; Zheng, Y.; Bordman, Z.; Fu, J.; Kim, Y.; Yen, H.R.; et al. Control of T(H)17/T(reg) balance by hypoxia-inducible factor 1. Cell 2011, 146, 772–784. [Google Scholar] [CrossRef]
- Harris, T.J.; Grosso, J.F.; Yen, H.-R.; Xin, H.; Kortylewski, M.; Albesiano, E.; Hipkiss, E.L.; Getnet, D.; Goldberg, M.V.; Maris, C.H.; et al. Cutting Edge: An In Vivo Requirement for STAT3 Signaling in TH17 Development and TH17-Dependent Autoimmunity. J. Immunol. 2007, 179, 4313–4317. [Google Scholar] [CrossRef]
- Crouser, E.D.; Julian, M.W.; Blaho, D.V.; Pfeiffer, D.R. Endotoxin-induced mitochondrial damage correlates with impaired respiratory activity. Crit. Care Med. 2002, 30, 276–284. [Google Scholar] [CrossRef]
- Liu, Y.-Y.; Lee, C.-H.; Dedaj, R.; Zhao, H.; Mrabat, H.; Shiedlin, A.; Syrkina, O.; Huang, P.-M.; Garg, H.G.; A Hales, C.; et al. High molecular weight hyaluronan: A possible new treatment for sepsis-induced lung injury—A preclinical study in mechanically ventilated rats. Crit. Care 2008, 12, R102. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.-F.; Yu, C.-C.; Huang, H.-Y.; Wu, H.-P.; Chu, C.-M.; Huang, C.-Y.; Liu, P.-C.; Liu, Y.-Y. Suppression of Hypoxia-Inducible Factor 1α by Low-Molecular-Weight Heparin Mitigates Ventilation-Induced Diaphragm Dysfunction in a Murine Endotoxemia Model. Int. J. Mol. Sci. 2021, 22, 1702. https://doi.org/10.3390/ijms22041702
Li L-F, Yu C-C, Huang H-Y, Wu H-P, Chu C-M, Huang C-Y, Liu P-C, Liu Y-Y. Suppression of Hypoxia-Inducible Factor 1α by Low-Molecular-Weight Heparin Mitigates Ventilation-Induced Diaphragm Dysfunction in a Murine Endotoxemia Model. International Journal of Molecular Sciences. 2021; 22(4):1702. https://doi.org/10.3390/ijms22041702
Chicago/Turabian StyleLi, Li-Fu, Chung-Chieh Yu, Hung-Yu Huang, Huang-Pin Wu, Chien-Ming Chu, Chih-Yu Huang, Ping-Chi Liu, and Yung-Yang Liu. 2021. "Suppression of Hypoxia-Inducible Factor 1α by Low-Molecular-Weight Heparin Mitigates Ventilation-Induced Diaphragm Dysfunction in a Murine Endotoxemia Model" International Journal of Molecular Sciences 22, no. 4: 1702. https://doi.org/10.3390/ijms22041702
APA StyleLi, L.-F., Yu, C.-C., Huang, H.-Y., Wu, H.-P., Chu, C.-M., Huang, C.-Y., Liu, P.-C., & Liu, Y.-Y. (2021). Suppression of Hypoxia-Inducible Factor 1α by Low-Molecular-Weight Heparin Mitigates Ventilation-Induced Diaphragm Dysfunction in a Murine Endotoxemia Model. International Journal of Molecular Sciences, 22(4), 1702. https://doi.org/10.3390/ijms22041702






