Metal–Curcumin Complexes in Therapeutics: An Approach to Enhance Pharmacological Effects of Curcumin
Abstract
:1. Introduction
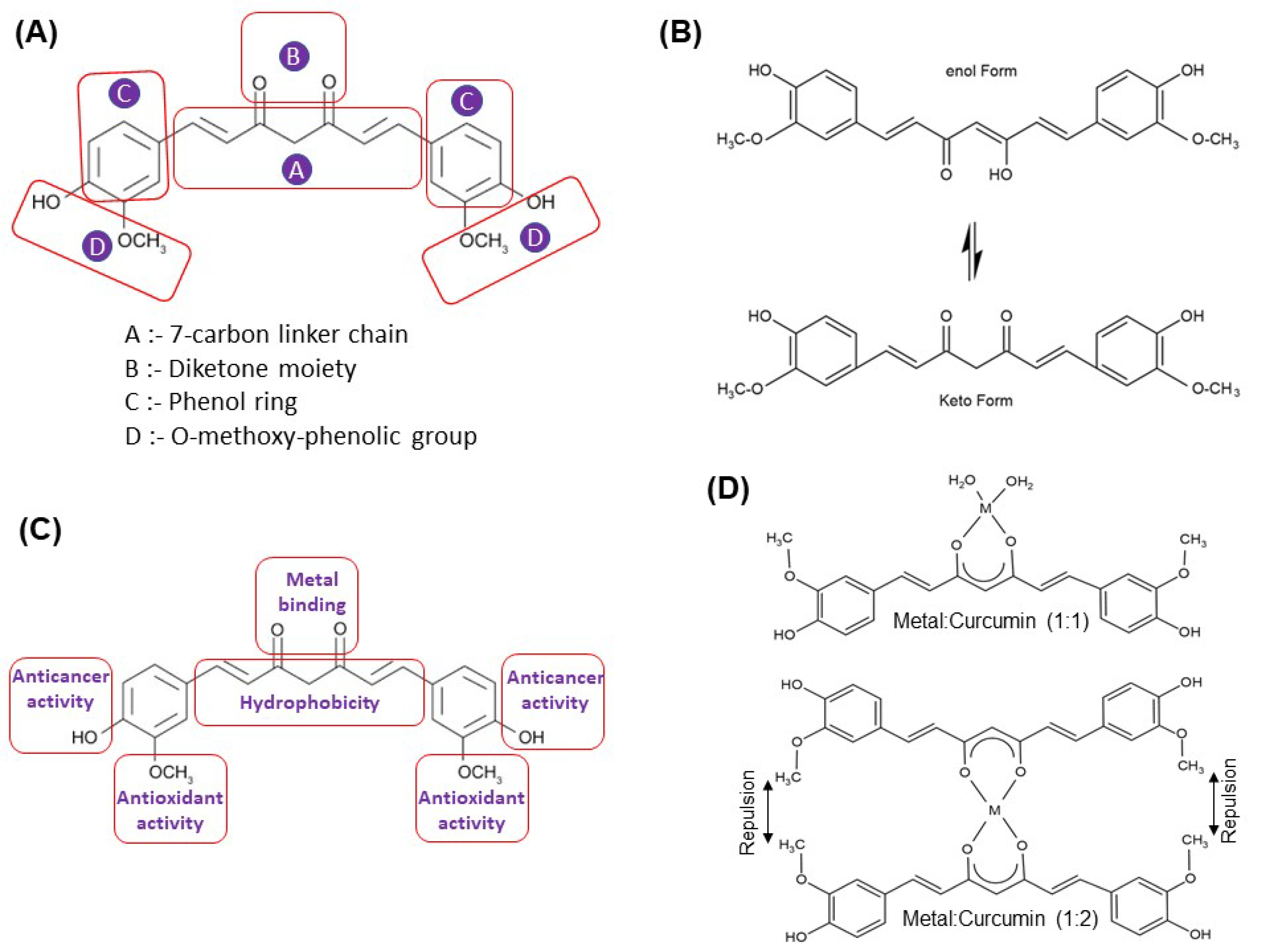
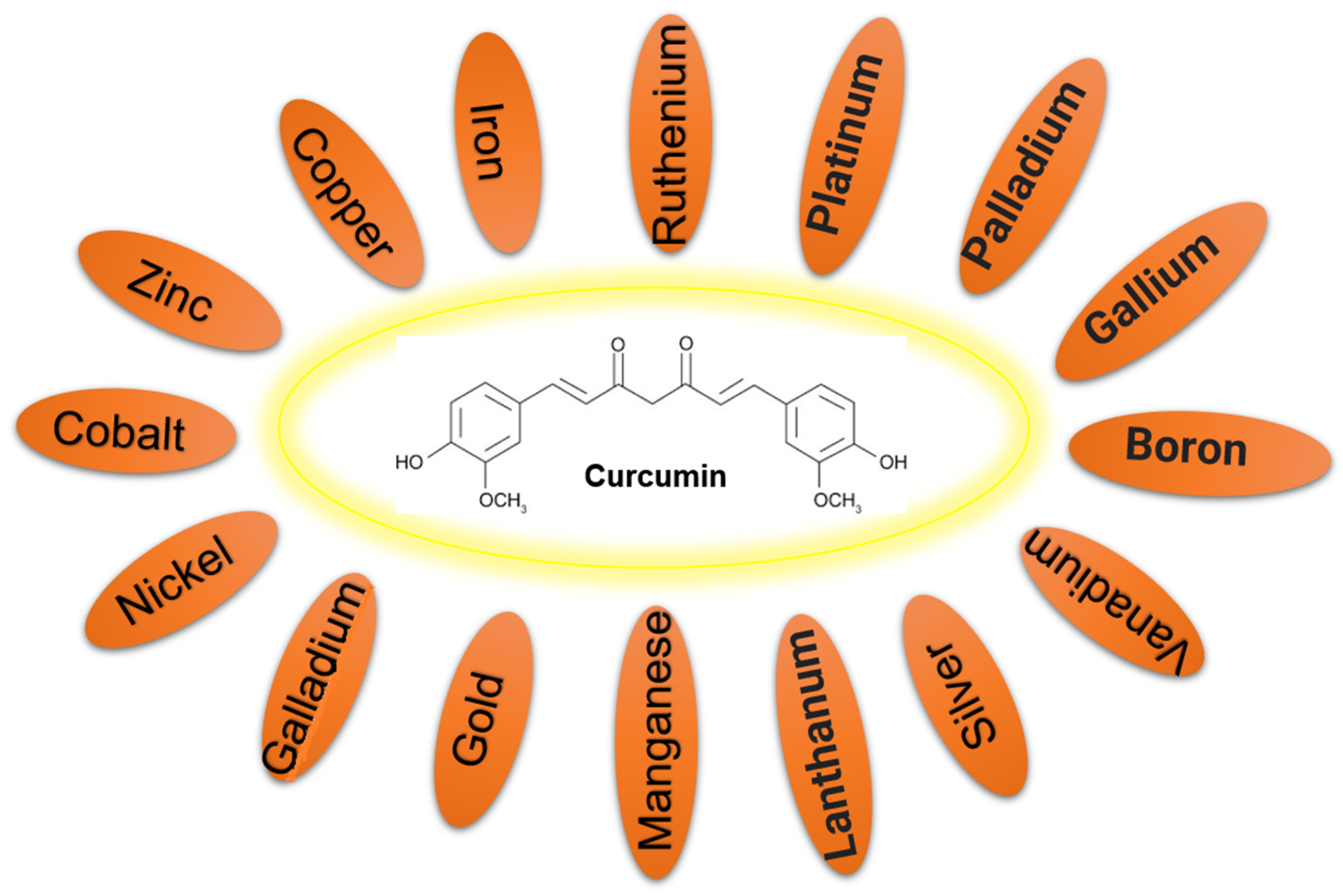
2. Metal–Curcumin Interactions
3. Synthesis of the Curcumin–Metal Complexes
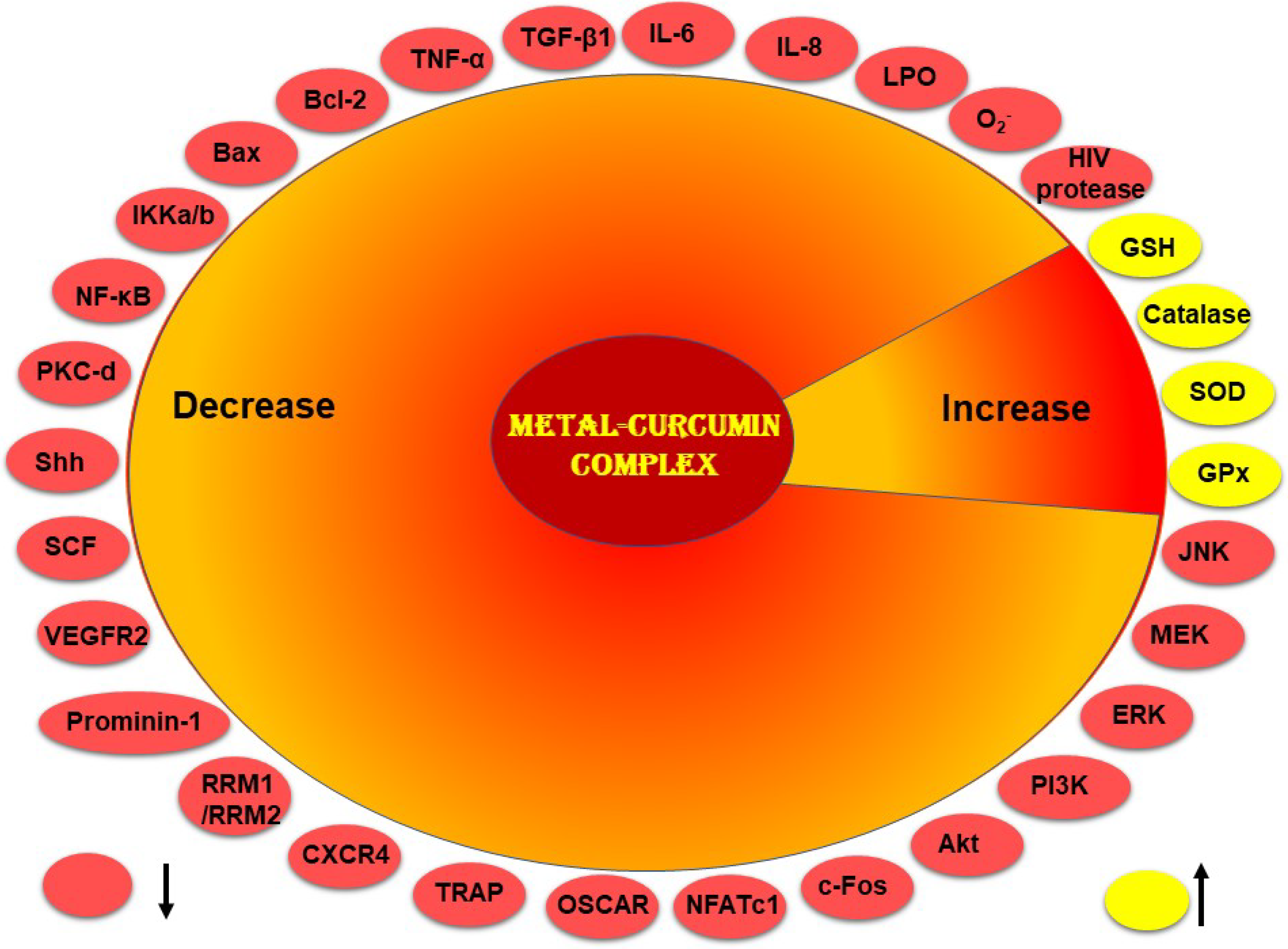
4. Pharmacological Effects of Metal–Curcumin Complex
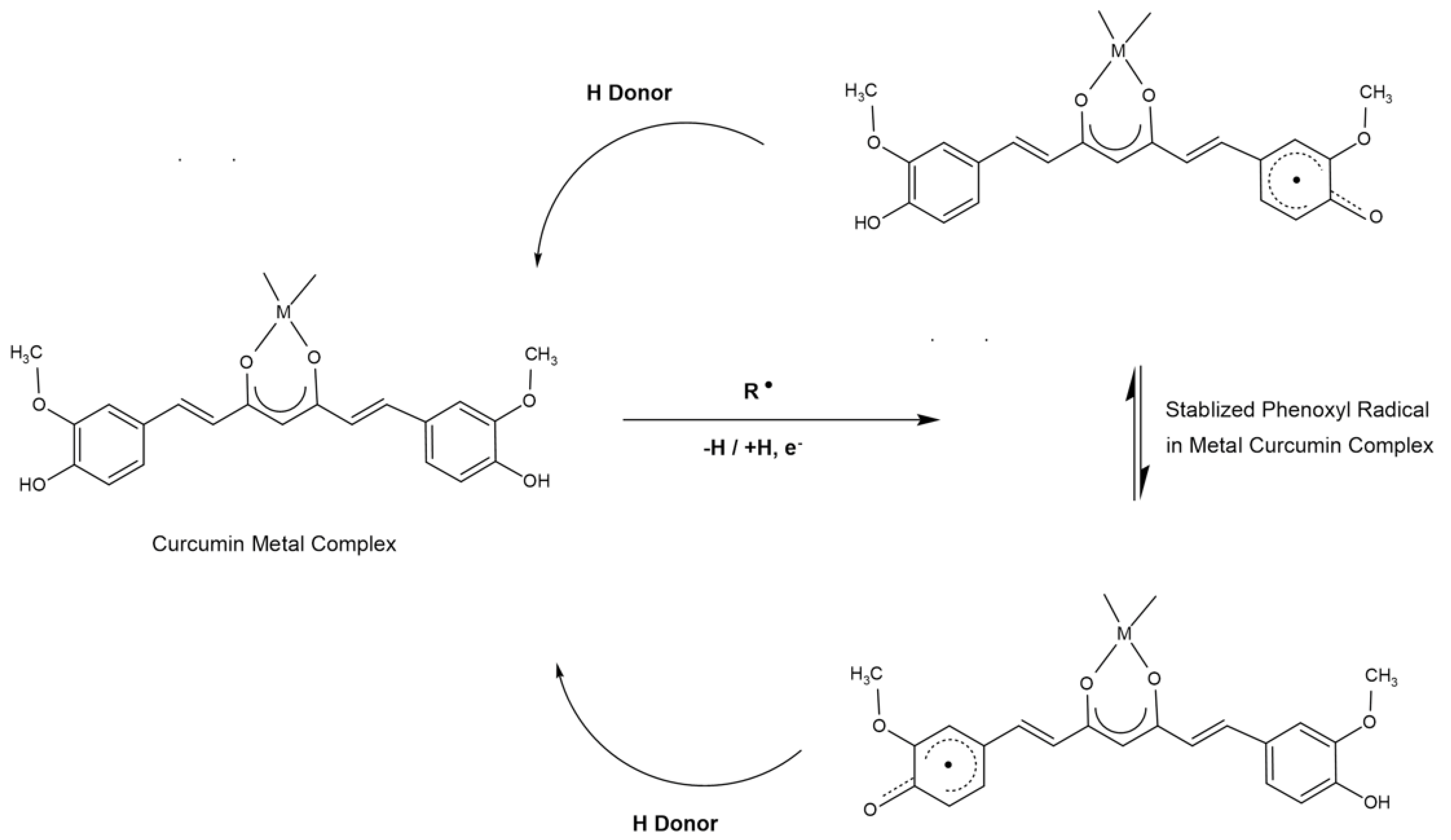
4.1. Antioxidant
4.2. Anti-Inflammatory
4.3. Antimicrobial
4.4. Antiviral
5. Prevention and Treatment of Chronic Inflammatory Diseases by Metal–Curcumin Complex
5.1. Cancer
5.2. Arthritis
5.3. Osteoporosis
5.4. Neurological Disorders
5.5. Other
6. Application of Metal–Curcumin Complex on Biological Imaging and Radioimaging
7. Solubility, Stability, and Cellular Uptake of Metal–Curcumin Complexes
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gupta, S.C.; Sung, B.; Kim, J.H.; Prasad, S.; Li, S.; Aggarwal, B.B. Multitargeting by turmeric, the golden spice: From kitchen to clinic. Mol. Nutr. Food Res. 2013, 57, 1510–1528. [Google Scholar] [CrossRef] [PubMed]
- Kunnumakkara, A.B.; Bordoloi, D.; Padmavathi, G.; Monisha, J.; Roy, N.K.; Prasad, S.; Aggarwal, B.B. Curcumin, the golden nutraceutical: Multitargeting for multiple chronic diseases. Br. J. Pharmacol. 2017, 174, 1325–1348. [Google Scholar] [CrossRef] [Green Version]
- Prasad, S.; Gupta, S.C.; Tyagi, A.K.; Aggarwal, B.B. Curcumin, a component of golden spice: From bedside to bench and back. Biotechnol. Adv. 2014, 32, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B.; Deb, L.; Prasad, S. Curcumin differs from tetrahydrocurcumin for molecular targets, signaling pathways and cellular responses. Molecules 2015, 20, 185–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Priyadarsini, K.I. The chemistry of curcumin: From extraction to therapeutic agent. Molecules 2014, 19, 20091–20112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salem, M.; Rohani, S.; Gillies, E.R. Curcumin, a promising anti-cancer therapeutic: A review of its chemical properties, bioactivity and approaches to cancer cell delivery. RSC Adv. 2014, 4, 10815–10829. [Google Scholar] [CrossRef]
- Mary, C.P.V.; Vijayakumar, S.; Shankar, R. Metal chelating ability and antioxidant properties of curcumin–metal complexes—A DFT approach. J. Mol. Gr. Modell. 2018, 79, 1–14. [Google Scholar] [CrossRef]
- Gupta, H.; Gupta, M.; Bhargava, S. Potential use of turmeric in COVID-19. Clin. Exp. Dermatol. 2020, 45, 902–903. [Google Scholar] [CrossRef] [PubMed]
- Vellampatti, S.; Chandrasekaran, G.; Mitta, S.B.; Lakshmanan, V.K.; Park, S.H. Metallo-curcumin-conjugated DNA complexes induces preferential prostate cancer cells cytotoxicity and pause growth of bacterial cells. Sci. Rep. 2018, 8, 14929–14939. [Google Scholar] [CrossRef] [PubMed]
- Khalil, M.I.; Al-Zahem, A.M.; Al-Qunaibit, M.H. Synthesis, characterization, mössbauer parameters, and antitumor activity of Fe(III) curcumin complex. Bioinorg. Chem. Appl. 2013, 2013, 982423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barik, A.; Mishra, B.; Kunwar, A.; Kadam, R.M.; Shen, L.; Dutta, S.; Padhye, S.; Satpati, A.K.; Zhang, H.Y.; Indira Priyadarsini, K. Comparative study of Copper(II)-curcumin complexes as superoxide dismutase mimics and free radical scavengers. Eur. J. Med. Chem. 2007, 42, 431–439. [Google Scholar] [CrossRef]
- Leung, M.H.M.; Harada, T.; Kee, T.W. Delivery of curcumin and medicinal effects of the Copper(II)-curcumin complexes. Curr. Pharm. Des. 2013, 19. [Google Scholar] [CrossRef]
- Vajragupta, O.; Boonchoong, P.; Watanabe, H.; Tohda, M.; Kummasud, N.; Sumanont, Y. Manganese complexes of curcumin and its derivatives: Evaluation for the radical scavenging ability and neuroprotective activity. Free Rad. Biol. Med. 2003, 35, 1632–1644. [Google Scholar] [CrossRef] [PubMed]
- Hieu, T.Q.; Thao, D.T.T. Enhancing the solubility of curcumin metal complexes and investigating some of their biological activities. J. Chem. 2019, 2019, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Subhan, M.A.; Alam, K.; Rahaman, M.S.; Rahman, M.A.; Awal, R. Synthesis and characterization of metal complexes containing curcumin (C21H20O6) and study of their anti-microbial activities and DNA-binding properties. J. Sci. Res. 2013, 6, 97–109. [Google Scholar] [CrossRef] [Green Version]
- Shakeri, A.; Panahi, Y.; Johnston, T.P.; Sahebkar, A. Biological properties of metal complexes of curcumin. BioFactors 2019, 45, 304–317. [Google Scholar] [CrossRef]
- Mei, X.; Xu, D.; Xu, S.; Zheng, Y.; Xu, S. Gastroprotective and antidepressant effects of a New Zinc(II)-curcumin complex in rodent models of gastric ulcer and depression induced by stresses. Pharmacol. Biochem. Behav. 2011, 99, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.A.; Hasan, M.N.; Bagchi, D.; Altass, H.M.; Morad, M.; Jassas, R.S.; Hameed, A.M.; Patwari, J.; Alessa, H.; Alharbi, A.; et al. Combating essential metal toxicity: Key information from optical spectroscopy. ACS Omega 2020, 5, 15666–15672. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.K.; Jagannathan, R.; Khandelwal, P.; Abraham, P.M.; Poddar, P. In Situ synthesis and surface functionalization of gold nanoparticles with curcumin and their antioxidant properties: An experimental and density functional theory investigation. Nanoscale 2013, 5, 1882–1893. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Y.; Yu, M.; Liu, Q.; Zhang, Q.; Liu, Z.; Tian, Y.; Li, D.; Changdao, M. Synthesis of silver nanoparticles using oxidized amylose and combination with curcumin for enhanced antibacterial activity. Carbohydr. Polym. 2020, 230, 115573. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.S.; Sun, J.L.; Xie, W.H.; Shen, L.; Ji, H.F. Neuroprotective effects and mechanisms of curcumin–Cu(II) and–Zn(II) complexes systems and their pharmacological implications. Nutrients 2018, 10, 28. [Google Scholar] [CrossRef] [Green Version]
- Singh, A.; Dutta, P.K. Green synthesis, characterization and biological evaluation of chitin glucan based zinc oxide nanoparticles and its curcumin conjugation. Int. J. Biol. Macromol. 2020, 156, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Antonyan, A.; De, A.; Vitali, L.A.; Pettinari, R.; Marchetti, F.; Gigliobianco, M.R.; Pettinari, C.; Camaioni, E.; Lupidi, G. Evaluation of (Arene)Ru(II) complexes of curcumin as inhibitors of dipeptidyl peptidase IV. Biochimie 2014, 99, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Jahangoshaei, P.; Hassani, L.; Mohammadi, F.; Hamidi, A.; Mohammadi, K. Investigating the effect of gallium curcumin and gallium diacetylcurcumin complexes on the structure, function and oxidative stability of the peroxidase enzyme and their anticancer and antibacterial activities. J. Biol. Inorg. Chem. 2015, 20, 1135–1146. [Google Scholar] [CrossRef] [PubMed]
- Mei, X.; Xu, D.; Xu, S.; Zheng, Y.; Xu, S. Novel role of Zn(II)-curcumin in enhancing cell proliferation and adjusting proinflammatory cytokine-mediated oxidative damage of ethanol-induced acute gastric ulcers. Chem. Biol. Interact. 2012, 197, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Mei, X.; Luo, X.; Xu, S.; Xu, D.; Zheng, Y.; Xu, S.; Lv, J. Gastroprotective effects of a new Zinc(II)-curcumin complex against pylorus-ligature-induced gastric ulcer in rats. Chem. Biol. Interact. 2009, 181, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Kunwar, A.; Narang, H.; Indira Priyadarsini, K.; Krishna, M.; Pandey, R.; Sainis, K.B. Delayed activation of PKCδ and NFκB and higher radioprotection in splenic lymphocytes by Copper (II)-Curcumin (1:1) complex as compared to curcumin. J. Cell. Biochem. 2007, 102, 1214–1224. [Google Scholar] [CrossRef]
- Seeta Rama Raju, G.; Pavitra, E.; Purnachandra Nagaraju, G.; Ramesh, K.; El-Rayes, B.F.; Yu, J.S. Imaging and curcumin delivery in pancreatic cancer cell lines using PEGylated α-Gd2(MoO4)3 mesoporous particles. Dalton Trans. 2014, 43, 3330–3338. [Google Scholar] [CrossRef] [PubMed]
- Kareem, A.; Arshad, M.; Nami, S.A.A.; Nishat, N. Herbo-mineral based schiff base ligand and its metal complexes: Synthesis, characterization, catalytic potential and biological applications. J. Photochem. Photobiol. B Biol. 2016, 160, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou, A.; Ketikidis, I.; Stathopoulou, M.E.K.; Banti, C.N.; Papachristodoulou, C.; Zoumpoulakis, L.; Agathopoulos, S.; Vagenas, G.V.; Hadjikakou, S.K. Innovative material containing the natural product curcumin, with enhanced antimicrobial properties for active packaging. Mater. Sci. Eng. C 2018, 84, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, P.; Shukla, M.; Kaul, G.; Chopra, S.; Patra, A.K. Rationally designed curcumin based Ruthenium(II) antimicrobials effective against drug-resistant: Staphylococcus aureus. Dalton Trans. 2019, 48, 11822–11828. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Mukhopadhyay, K.; Saha, T.; Kumar, P.; Sepay, N.; Ganguly, D.; Tiwari, K. Multitargeting antibacterial activity of a synthesized Mn2+ complex of curcumin on gram-positive and gram-negative bacterial strains. ACS Omega 2020, 5, 16342–16357. [Google Scholar] [CrossRef]
- Chauhan, G.; Rath, G.; Goyal, A.K. In-vitro anti-viral screening and cytotoxicity evaluation of copper-curcumin complex. Artif. Cells Nanomed. Biotechnol. 2013, 41, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Gholami, M.; Zeighami, H.; Bikas, R.; Heidari, A.; Rafiee, F.; Haghi, F. Inhibitory activity of metal-curcumin complexes on quorum sensing related virulence factors of Pseudomonas Aeruginosa PAO1. AMB Express 2020, 10, 111–120. [Google Scholar] [CrossRef]
- Katsipis, G.; Tsalouxidou, V.; Halevas, E.; Geromichalou, E.; Geromichalos, G.; Pantazaki, A.A. In vitro and in silico evaluation of the inhibitory effect of a curcumin-based Oxovanadium (IV) complex on alkaline phosphatase activity and bacterial biofilm formation. Appl. Microbiol. Biotechnol. 2021, 105, 147–168. [Google Scholar] [CrossRef] [PubMed]
- Sui, Z.; Salto, R.; Li, J.; Craik, C.; Ortiz de Montellano, P.R. Inhibition of the HIV-1 and HIV-2 proteases by curcumin and curcumin boron complexes. Bioorg. Med. Chem. 1993, 1, 415–422. [Google Scholar] [CrossRef]
- Refat, M.S. Synthesis and characterization of ligational behavior of curcumin drug towards some transition metal ions: Chelation effect on their thermal stability and biological activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 105, 326–337. [Google Scholar] [CrossRef] [PubMed]
- Barclay, L.R.C.; Vinqvist, M.R.; Mukai, K.; Goto, H.; Hashimoto, Y.; Tokunaga, A.; Uno, H. On the antioxidant mechanism of curcumin: Classical methods are needed to determine antioxidant mechanism and activity. Org. Lett. 2000, 2, 2841–2843. [Google Scholar] [CrossRef]
- Gupta, N.; Verma, K.; Nalla, S.; Kulshreshtha, A.; Lall, R.; Prasad, S. Free radicals as a double-edged sword: The cancer preventive and therapeutic roles of curcumin. Molecules 2020, 25, 5390. [Google Scholar] [CrossRef] [PubMed]
- Jakubczyk, K.; Drużga, A.; Katarzyna, J.; Skonieczna-żydecka, K. Antioxidant potential of curcumin—A meta-analysis of randomized clinical trials. Antioxidants 2020, 9, 1092. [Google Scholar] [CrossRef] [PubMed]
- Priya, R.S.; Balachandran, S.; Daisy, J.; Mohanan, P.V. Reactive centers of curcumin and the possible role of metal complexes of curcumin as antioxidants. Univ. J. Phys. Appl. 2015, 9, 6–16. [Google Scholar] [CrossRef]
- Thakam, A.; Saewan, N. Antioxidant activities of curcumin–metal complexes. Thail. J. Agric. Sci. 2011, 44, 188–193. [Google Scholar]
- Eybl, V.; Kotyzová, D.; Lešetický, L.; Bludovská, M.; Koutenský, J. The influence of curcumin and manganese complex of curcumin on cadmium-induced oxidative damage and trace elements status in tissues of mice. J. Appl. Toxicol. 2006, 26, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Meza-Morales, W.; Mirian Estévez-Carmona, M.; Alvarez-Ricardo, Y.; Obregón-Mendoza, M.A.; Cassani, J.; Ramírez-Apan, M.T.; Escobedo-Martínez, C.; Soriano-García, M.; Reynolds, W.F.; Enríquez, R.G. Full structural characterization of homoleptic complexes of diacetylcurcumin with Mg, Zn, Cu, and Mn: Cisplatin-level cytotoxicity in vitro with minimal acute toxicity in vivo. Molecules 2019, 24, 1598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, S.; Tyagi, A.K. Curcumin and its analogues: A potential natural compound against HIV infection and AIDS. Food Funct. 2015, 6, 3412–3419. [Google Scholar] [CrossRef] [PubMed]
- Sandhya, P.; Renu, K. Stabilisation of curcumin with ferrous ion and evaluation of its pharmacological property. Int. J. Pharmacogn. Phytochem. Res. 2015, 7, 943–947. [Google Scholar]
- Kali, A.; Bhuvaneshwar, D.; Charles, P.M.V.; Seetha, K. Antibacterial synergy of curcumin with antibiotics against biofilm producing clinical bacterial isolates. J. Basic Clin. Pharm. 2016, 7, 93–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agel, M.R.; Baghdan, E.; Pinnapireddy, S.R.; Lehmann, J.; Schäfer, J.; Bakowsky, U. Curcumin loaded nanoparticles as efficient photoactive formulations against gram-positive and gram-negative bacteria. Coll. Surf. B Biointerfaces 2019, 178, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Jeyaraman, P.; Samuel, M.; Johnson, A.; Raman, N. Synthesis, characterization, ADMET, in vitro and in vivo studies of mixed ligand metal complexes from a curcumin schiff base and lawsone. Nucleosides Nucleotides Nucl. Acids 2020, 40, 242–263. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.M.; Xu, J.P.; Ding, L.; Hou, Q.; Liu, J.W.; Zhu, Z.L. Syntheses, characterization and biological activities of rare earth metal complexes with curcumin and 1,10-phenanthroline-5,6-dione. J. Inorg. Biochem. 2009, 103, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Li, J.; Yu, J.; Wang, Y.; Liu, H.; Lin, G.; He, Z.; Wang, Y. Unique flower-like cur-metal complexes loaded liposomes for primary and metastatic breast cancer therapy. Mater. Sci. Eng. C 2021, 121, 111835. [Google Scholar] [CrossRef] [PubMed]
- Goswami, T.K.; Gadadhar, S.; Gole, B.; Karande, A.A.; Chakravarty, A.R. Photocytotoxicity of Copper(II) complexes of curcumin and N-Ferrocenylmethyl-l-amino acids. Eur. J. Med. Chem. 2013, 63, 800–810. [Google Scholar] [CrossRef] [PubMed]
- John, V.D.; Kuttan, G.; Krishnankutty, K. Anti-tumour studies of metal chelates of synthetic curcuminoids. J. Exp. Clin. Cancer Res. 2002, 21, 219–224. [Google Scholar] [PubMed]
- Greish, K.; Pittalà, V.; Taurin, S.; Taha, S.; Bahman, F.; Mathur, A.; Jasim, A.; Mohammed, F.; El-Deeb, I.M.; Fredericks, S.; et al. Curcumin–copper complex nanoparticles for the management of triple-negative breast cancer. Nanomaterials 2018, 8, 884. [Google Scholar] [CrossRef] [Green Version]
- Ghaffari, S.B.; Sarrafzadeh, M.H.; Fakhroueian, Z.; Shahriari, S.; Khorramizadeh, M.R. Functionalization of ZnO nanoparticles by 3-mercaptopropionic acid for aqueous curcumin delivery: Synthesis, characterization, and anticancer assessment. Mater. Sci. Eng. C 2017, 79, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Chakravarty, A.R. Metal complexes of curcumin for cellular imaging, targeting, and photoinduced anticancer activity. Acc. Chem. Res. 2015, 48, 2075–2083. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.P.; Wei, Z.Z.; Wang, Z.F.; Huang, X.L.; Tan, M.X.; Zou, H.H.; Liang, H. Imaging and therapeutic applications of Zn(Ii)-cryptolepine-curcumin molecular probes in cell apoptosis detection and photodynamic therapy. Chem. Commun. 2020, 56, 3999–4002. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Mei, X.; Ye, Y.; Xue, T.; Wang, J.; Sun, W.; Lin, C.; Xue, R.; Zhang, J.; Xu, D. Zn(II)-curcumin solid dispersion impairs hepatocellular carcinoma growth and enhances chemotherapy by modulating gut microbiota-mediated zinc homeostasis. Pharmacol. Res. 2019, 104454–104463. [Google Scholar] [CrossRef]
- Li, S.; Xu, G.; Zhu, Y.; Zhao, J.; Gou, S. Bifunctional ruthenium(II) polypyridyl complexes of curcumin as potential anticancer agents. Dalton Trans. 2020, 49, 9454–9463. [Google Scholar] [CrossRef] [PubMed]
- Castonguay, A.; Doucet, C.; Juhas, M.; Maysinger, D. New Ruthenium(II)-letrozole complexes as anticancer therapeutics. J. Med. Chem. 2012, 55, 8799–8806. [Google Scholar] [CrossRef] [Green Version]
- Bonfili, L.; Pettinari, R.; Cuccioloni, M.; Cecarini, V.; Mozzicafreddo, M.; Angeletti, M.; Lupidi, G.; Marchetti, F.; Pettinari, C.; Eleuteri, A.M. Arene-RuII complexes of curcumin exert antitumor activity via proteasome inhibition and apoptosis induction. ChemMedChem 2012, 7, 2010–2020. [Google Scholar] [CrossRef]
- Garai, A.; Pant, I.; Banerjee, S.; Banik, B.; Kondaiah, P.; Chakravarty, A.R. Photorelease and cellular delivery of mitocurcumin from its cytotoxic Cobalt(III) complex in visible light. Inorg. Chem. 2016, 55, 6027–6035. [Google Scholar] [CrossRef] [PubMed]
- Tunc, D.; Dere, E.; Karakas, D.; Cevatemre, B.; Yilmaz, V.T.; Ulukaya, E. Cytotoxic and apoptotic effects of the combination of palladium (II) 5,5-diethylbarbiturate complex with bis(2-pyridylmethyl)amine and curcumin on non small lung cancer cell lines. Bioorg. Med. Chem. 2017, 25, 1717–1723. [Google Scholar] [CrossRef] [PubMed]
- Valentini, A.; Conforti, F.; Crispini, A.; de Martino, A.; Condello, R.; Stellitano, C.; Rotilio, G.; Ghedini, M.; Federici, G.; Bernardini, S.; et al. Synthesis, oxidant properties, and antitumoral effects of a heteroleptic palladium(II) complex of curcumin on human prostate cancer cells. J. Med. Chem. 2009, 52, 484–491. [Google Scholar] [CrossRef]
- Fischer-Fodor, E.; Mikláš, R.; Rišianová, L.; Cenariu, M.; Grosu, I.G.; Virag, P.; Schrepler, M.P.; Tomuleasa, C.; Neagoe, I.B.; Devínsky, F.; et al. Novel palladium(II) complexes that influence prominin-1/CD133 expression and stem cell factor release in tumor cells. Molecules 2017, 22, 561. [Google Scholar] [CrossRef] [PubMed]
- Banaspati, A.; Raza, M.K.; Goswami, T.K. Ni(II) curcumin complexes for cellular imaging and photo-triggered in vitro anticancer activity. Eur. J. Med. Chem. 2020, 204, 112632. [Google Scholar] [CrossRef] [PubMed]
- Raza, M.K.; Mitra, K.; Shettar, A.; Basu, U.; Kondaiah, P.; Chakravarty, A.R. Photoactive platinum(II) β-diketonates as dual action anticancer agents. Dalton Trans. 2016, 45, 13234–13243. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.; Gautam, S.; Ramu, V.; Kondaiah, P.; Chakravarty, A.R. Photocytotoxic cancer cell-targeting platinum(II) complexes of glucose-appended curcumin and biotinylated 1,10-phenanthroline. Dalton Trans. 2019, 48, 17556–17565. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, Y.; Xu, X.; Chen, C.; Xue, X.; Xu, B.; Li, T.; Chen, Z. Synthesis characterization of platinum (IV) complex curcumin backboned polyprodrugs: In vitro drug release anticancer activity. Polymers 2021, 13, 67. [Google Scholar] [CrossRef]
- Mitra, K.; Gautam, S.; Kondaiah, P.; Chakravarty, A.R. The cis-diammineplatinum(II) complex of curcumin: A dual action DNA crosslinking and photochemotherapeutic agent. Angew. Chem. Int. Ed. 2015, 54, 13989–13993. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, C.; Zhang, X.; He, C.; Zhao, P.; Li, M.; Fan, T.; Yan, R.; Lu, Y.; Lee, R.J.; et al. Platinum complexes of curcumin delivered by dual-responsive polymeric nanoparticles improve chemotherapeutic efficacy based on the enhanced anti-metastasis activity and reduce side effects. Acta Pharm. Sin. B 2020, 10, 1106–1121. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, U.; Kumar, B.; Garai, A.; Bhattacharyya, A.; Kumar, A.; Banerjee, S.; Kondaiah, P.; Chakravarty, A.R. Curcumin “drug” stabilized in oxidovanadium(IV)-BODIPY conjugates for mitochondria-targeted photocytotoxicity. Inorg. Chem. 2017, 56, 12457–12468. [Google Scholar] [CrossRef] [PubMed]
- Balaji, B.; Balakrishnan, B.; Perumalla, S.; Karande, A.A.; Chakravarty, A.R. Photoactivated cytotoxicity of ferrocenyl-terpyridine oxovanadium(IV) complexes of curcuminoids. Eur. J. Med. Chem. 2014, 85, 458–467. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Setua, S.; Kumari, S.; Dan, N.; Massey, A.; bin Hafeez, B.; Yallapu, M.M.; Stiles, Z.E.; Alabkaa, A.; Yue, J.; et al. superparamagnetic iron oxide nanoparticles of curcumin enhance gemcitabine therapeutic response in pancreatic cancer. Biomaterials 2019, 208, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, F.; Rashid-Doubell, F.; Taha, S.; Cassidy, S.; Fredericks, S. Effects of curcumin complexes on MDA-MB-231 breast cancer cell proliferation. Int. J. Oncol. 2020, 57, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Xu, N.; Xu, L.; Wang, L.; Li, Z.; Ma, W.; Zhu, Y.; Xu, C.; Kotov, N.A. Multifunctional magnetoplasmonic nanoparticle assemblies for cancer therapy and diagnostics (theranostics). Macromol. Rapid Commun. 2010, 31, 228–236. [Google Scholar] [CrossRef] [Green Version]
- Thompson, K.H.; Böhmerle, K.; Polishchuk, E.; Martins, C.; Toleikis, P.; Tse, J.; Yuen, V.; McNeill, J.H.; Orvig, C. Complementary inhibition of synoviocyte, smooth muscle cell or mouse lymphoma cell proliferation by a vanadyl curcumin complex compared to curcumin alone. J. Inorg. Biochem. 2004, 98, 2063–2070. [Google Scholar] [CrossRef]
- Sharma, K.K.; Chandra, S.; Basu, D.K. Synthesis and antiarthritic study of a new orally active diferuloyl methane (curcumin) gold complex. Inorg. Chim. Acta 1987, 135, 47–48. [Google Scholar] [CrossRef]
- Mawani, Y.; Orvig, C. Improved separation of the curcuminoids, syntheses of their rare earth complexes, and studies of potential antiosteoporotic activity. J. Inorg. Biochem. 2014, 132, 52–58. [Google Scholar] [CrossRef]
- Heo, D.N.; Ko, W.K.; Moon, H.J.; Kim, H.J.; Lee, S.J.; Lee, J.B.; Bae, M.S.; Yi, J.K.; Hwang, Y.S.; Bang, J.B.; et al. Inhibition of osteoclast differentiation by gold nanoparticles functionalized with cyclodextrin curcumin complexes. ACS Nano 2014, 8, 12049–12062. [Google Scholar] [CrossRef]
- Kochi, A.; Lee, H.; Vithanarachchi, S.; Padmini, V.; Allen, M.; Lim, M. Inhibitory activity of curcumin derivatives towards metal-free and metal-induced amyloid-β aggregation. Curr. Alzheimer Res. 2016, 12, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Hu, X.; Zhou, L.; Tu, Y.; Shi, S.; Yao, T. Orientation-inspired perspective on molecular inhibitor of tau aggregation by curcumin conjugated with ruthenium(II) complex scaffold. J. Phys. Chem. B 2020, 124, 2343–2353. [Google Scholar] [CrossRef] [PubMed]
- Bicer, N.; Yildiz, E.; Yegani, A.A.; Aksu, F. Synthesis of curcumin complexes with iron(III) and manganese(II), and effects of curcumin-iron(III) on Alzheimer’s disease. New J. Chem. 2018, 42, 8098–8104. [Google Scholar] [CrossRef]
- Al-Ali, K.; Fatah, H.S.A.; El-Badry, Y.A.M. Dual effect of curcumin-zinc complex in controlling diabetes mellitus in experimentally induced diabetic rats. Biol. Pharm. Bull. 2016, 39, 1774–1780. [Google Scholar] [CrossRef] [Green Version]
- Lu, W.P.; Mei, X.T.; Wang, Y.; Zheng, Y.P.; Xue, Y.F.; Xu, D.H. Zn(II)-curcumin protects against oxidative stress, deleterious changes in sperm parameters and histological alterations in a male mouse model of cyclophosphamide-induced reproductive damage. Environ. Toxicol. Pharmacol. 2015, 39, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Garufi, A.; Trisciuoglio, D.; Porru, M.; Leonetti, C.; Stoppacciaro, A.; D’Orazi, V.; Avantaggiati, M.L.; Crispini, A.; Pucci, D.; D’Orazi, G. A fluorescent curcumin-based Zn(II)-complex reactivates mutant (R175H and R273H) P53 in cancer cells. J. Exp. Clin. Cancer Res. 2013, 32, 72–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banerjee, S.; Pant, I.; Khan, I.; Prasad, P.; Hussain, A.; Kondaiah, P.; Chakravarty, A.R. remarkable enhancement in photocytotoxicity and hydrolytic stability of curcumin on binding to an oxovanadium(IV) moiety. Dalton Trans. 2015, 44, 4108–4122. [Google Scholar] [CrossRef]
- Caruso, F.; Pettinari, R.; Rossi, M.; Monti, E.; Gariboldi, M.B.; Marchetti, F.; Pettinari, C.; Caruso, A.; Ramani, M.V.; Subbaraju, G.V. The in vitro antitumor activity of arene-ruthenium(II) curcuminoid complexes improves when decreasing curcumin polarity. J. Inorg. Biochem. 2016, 162, 44–51. [Google Scholar] [CrossRef]
- Bharti, A.C.; Takada, Y.; Aggarwal, B.B. Curcumin (diferuloylmethane) inhibits receptor activator of NF-ΚB ligand-induced NF-ΚB activation in osteoclast precursors and suppresses osteoclastogenesis. J. Immunol. 2004, 172, 5940–5947. [Google Scholar] [CrossRef] [Green Version]
- Salehi, B.; Calina, D.; Docea, A.; Koirala, N.; Aryal, S.; Lombardo, D.; Pasqua, L.; Taheri, Y.; Marina Salgado Castillo, C.; Martorell, M.; et al. Curcumin’s nanomedicine formulations for therapeutic application in neurological diseases. J. Clin. Med. 2020, 9, 430. [Google Scholar] [CrossRef] [Green Version]
- Caito, S.; Aschner, M. Neurotoxicity of metals. Handb. Clin. Neurol. 2015, 131, 169–189. [Google Scholar] [CrossRef]
- Jiang, T.; Zhou, G.R.; Zhang, Y.H.; Sun, P.C.; Du, Q.M.; Zhou, P. Influence of curcumin on the Al(III)-induced conformation transition of silk fibroin and resulting potential therapy for neurodegenerative diseases. RSC Adv. 2012, 2, 9106–9113. [Google Scholar] [CrossRef]
- Lu, Q.; Wu, C.J.; Liu, Z.; Niu, G.; Yu, X. Fluorescent AIE-active materials for two-photon bioimaging applications. Front. Chem. 2020, 8, 617463. [Google Scholar] [CrossRef]
- Pi, Z.; Wang, J.; Jiang, B.; Cheng, G.; Zhou, S. A Curcumin-based TPA four-branched copper(II) complex probe for in vivo early tumor detection. Mater. Sci. Eng. C 2015, 46, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Bhat, I.A.; Jain, R.; Siddiqui, M.M.; Saini, D.K.; Mukherjee, P.S. Water-soluble Pd8L4 self-assembled molecular barrel as an aqueous carrier for hydrophobic curcumin. Inorg. Chem. 2017, 56, 5352–5360. [Google Scholar] [CrossRef] [PubMed]
- Sahub, C.; Tumcharern, G.; Chirawatkul, P.; Ruangpornvisuti, V.; Ekgasit, S.; Wanichweacharungruang, S.; Tuntulani, T.; Palaga, T.; Tomapatanaget, B. Self-assembly of Gd3+/SDS/HEPES complex and curcumin entrapment for enhanced stability, fluorescence image in cellular system. Coll. Surf. B Biointerfaces 2017, 156, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, F.; Rashid-Doubell, F.; Cassidy, S.; Henari, F. A comparative study of the spectral, fluorometric properties and photostability of natural curcumin, iron- and boron- complexed curcumin. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 183, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Alberti, D.; Protti, N.; Franck, M.; Stefania, R.; Bortolussi, S.; Altieri, S.; Deagostino, A.; Aime, S.; Geninatti Crich, S. Theranostic nanoparticles loaded with imaging probes and rubrocurcumin for combined cancer therapy by folate receptor targeting. ChemMedChem 2017, 12, 502–509. [Google Scholar] [CrossRef]
- Hussain, A.; Somyajit, K.; Banik, B.; Banerjee, S.; Nagaraju, G.; Chakravarty, A.R. Enhancing the photocytotoxic potential of curcumin on terpyridyl lanthanide(III) complex formation. Dalton Trans. 2013, 42, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Tyagi, A.K.; Aggarwal, B.B. Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: The golden pigment from golden spice. Cancer Res. Treat. 2014, 46, 2–18. [Google Scholar] [CrossRef] [Green Version]
- Khorasani, M.Y.; Langari, H.; Sany, S.B.T.; Rezayi, M.; Sahebkar, A. The role of curcumin and its derivatives in sensory applications. Mater. Sci. Eng. C 2019, 103, 109792. [Google Scholar] [CrossRef] [PubMed]
- Wanninger, S.; Lorenz, V.; Subhan, A.; Edelmann, F.T. Metal complexes of curcumin-synthetic strategies, structures and medicinal applications. Chem. Soc. Rev. 2015, 44, 4986–5002. [Google Scholar] [CrossRef] [Green Version]
- Zebib, B.; Mouloungui, Z.; Noirot, V. Stabilization of curcumin by complexation with divalent cations in glycerol/water system. Bioinorg. Chem. Appl. 2010, 2010, 292760–292767. [Google Scholar] [CrossRef] [PubMed]
- Sareen, R.; Jain, N.; Dhar, K.L. Curcumin–Zn(II) complex for enhanced solubility and stability: An approach for improved delivery and pharmacodynamic effects. Pharm. Develop. Technol. 2016, 21, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Grabner, S.; Modec, B. Zn(II) curcuminate complexes with 2,20-bipyridine and carboxylates. Molecules 2019, 24, 2540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaurav, C.; Goutam, R.; Rohan, K.N.; Sweta, K.T.; Abhay, C.S.; Amit, G.K. (Copper-curcumin) β-cyclodextrin vaginal gel: Delivering a novel metal-herbal approach for the development of topical contraception prophylaxis. Eur. J. Pharm. Sci. 2014, 65, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Moussa, Z.; Hmadeh, M.; Abiad, M.G.; Dib, O.H.; Patra, D. Encapsulation of curcumin in cyclodextrin-metal organic frameworks: Dissociation of loaded CD-MOFs enhances stability of curcumin. Food Chem. 2016, 212, 485–494. [Google Scholar] [CrossRef]
- Kamalasanan, K.; Deepa, M.K.; Sharma, C.P. Supramolecular curcumin-barium prodrugs for formulating with ceramic particles. Coll. Surf. B Biointerfaces 2014, 122, 301–308. [Google Scholar] [CrossRef]
- Bettini, S.; Pagano, R.; Valli, L.; Giancane, G. Drastic nickel ion removal from aqueous solution by curcumin-capped ag nanoparticles. Nanoscale 2014, 6, 10113–10117. [Google Scholar] [CrossRef]
- Orteca, G.; Pisaneschi, F.; Rubagotti, S.; Liu, T.W.; Biagiotti, G.; Piwnica-Worms, D.; Iori, M.; Capponi, P.C.; Ferrari, E.; Asti, M. Development of a potential gallium-68-labelled radiotracer based on DOTA-curcumin for colon-rectal carcinoma: From synthesis to in vivo studies. Molecules 2019, 24, 644. [Google Scholar] [CrossRef] [Green Version]
- Rubagotti, S.; Croci, S.; Ferrari, E.; Orteca, G.; Iori, M.; Capponi, P.C.; Versari, A.; Asti, M. Uptake of Ga-curcumin derivatives in different cancer cell lines: Toward the development of new potential 68Ga-labelled curcuminoids-based radiotracers for tumour imaging. J. Inorg. Biochem. 2017, 173, 113–119. [Google Scholar] [CrossRef]
- Glenister, A.; Chen, C.K.J.; Tondl, E.M.; Paterson, D.; Hambley, T.W.; Renfrew, A.K. Targeting curcumin to specific tumour cell environments: The influence of ancillary ligands. Metallomics 2017, 9, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Mierzecki, A.; Strecker, D.; Radomska, K. A pilot study on zinc levels in patients with rheumatoid arthritis. Biol. Trace Elem. Res. 2011, 143, 854–862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, T.C.; Chang, W.T.; Hu, Y.C.; Hsieh, B.S.; Cheng, H.L.; Yen, J.H.; Chiu, P.R.; Chang, K.L. Zinc protects articular chondrocytes through changes in Nrf2-mediated antioxidants, cytokines and matrix metalloproteinases. Nutrients 2018, 10, 471. [Google Scholar] [CrossRef] [Green Version]
- Read, S.A.; Obeid, S.; Ahlenstiel, C.; Ahlenstiel, G. The role of zinc in antiviral immunity. Adv. Nutr. 2019, 10, 696–710. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Metal–Curcumin Complex | Effects | References |
|---|---|---|
| Antioxidant | ||
| Zinc–curcumin | Increases antioxidant enzymes and decreases MDA level caused by cold-restraint stress in mice | [17] |
| Iron–curcumin | Improves antioxidant activity | [18] |
| Gold NP–curcumin | Shows good antioxidant activity | [19] |
| Silver–curcumin | Presents good antioxidant activity and cell compatibility | [20] |
| Copper–curcumin Zinc–curcumin | Enhances the superoxide dismutase, catalase, and glutathione peroxidase activities and attenuates MDA levels in PC12 cells | [21] |
| Copper–curcumin | Exhibits antioxidant potential by scavenging DPPH radicals | [11] |
| Zinc NP–curcumin | Exerts antioxidant activity as it scavenges ABTS+ and DPPH free radicals | [22] |
| Ruthenium–curcumin | Exhibits potent antioxidant activity | [23] |
| Gallium–diacetylcurcumin | Presents an increase in the antioxidant efficiency | [24] |
| Anti-inflammatory | ||
| Zinc–curcumin | Suppresses NF-κB, TGF-β1, IL-8, IL-6, TNF-α in rat model | [25,26] |
| Copper–curcumin | Suppresses PKCδ and NF-κB | [27] |
| Zinc–curcumin Copper–curcumin | Inhibits H2O2-induced NF-κB in PC12 cells | [21] |
| Gadolinium–curcumin | Inhibits pIKKα, pIKKα/β, and NF-κB in pancreatic cancer cells | [28] |
| Anti-microbial | ||
| Cobalt–, Nickel–, Copper–curcumin | Inhibits growth of E. coli, S. aureus, K. pneumoniae, S. pyogenes, and P. aeruginosa | [29] |
| Zinc–curcumin | Inhibits growth of P. aeruginosa | [30] |
| Zinc–curcumin | Inhibits growth of B. subtilis, E. coli | [22] |
| Ruthenium–curcumin | Inhibits growth of drug resistant S. aureus, reduces mean bacterial counts in murine model of Staphylococcus infection | [31,32] |
| Manganese–curcumin | Inhibits growth of S. aureus, E. coli | [32] |
| Silver–curcumin | Combats multiple bacteria-induced infections on wound surfaces | [20] |
| Copper–curcumin | Exhibits antimicrobicidal activity | [33] |
| Copper–curcumin | Inhibits growth of P. aeruginosa | [34] |
| Orthovanadium–curcumin | Inhibits bacterial biofilm formation, growth of S. aureus and E. coli | [35] |
| Copper–curcumin | Inhibits growth of P. aeruginosa and biofilm formation | [34] |
| Ruthenium–curcumin | Exhibits strong antibiofilm activity | [31] |
| Zinc–curcumin | Exerts potency against biofilm formation | [30] |
| Antiviral | ||
| Boron–curcumin | Inactivates HIV proteases | [36] |
| Copper–curcumin | Inhibits multiplication of multiple types of viruses | [33] |
| Metal–Curcumin Complex | Models | Mechanisms | References |
|---|---|---|---|
| Cancer | |||
| Copper–curcumin | Breast cancer cells | Promotes apoptosis, inhibits proliferation and angiogenesis | [51] |
| Copper–curcumin | Cervical and breast cancer cells | Induces photocytotoxicity | [52] |
| Copper–curcumin | Mice | Increases life span of ascites tumor bearing animals and reduces tumor growth | [53] |
| Copper–curcumin | Murine | Inhibits growth of human breast tumors in animals | [54] |
| Zinc–curcumin | Breast cancer cells | Induces more cytotoxicity compared to free curcumin | [55] |
| Zinc–curcumin | Prostate cancer, neuroblastoma cell lines | Induces cytotoxicity | [56] |
| Zinc–curcumin | In vitro and in vivo | Promotes mitochondria-mediated apoptosis | [57] |
| Zinc–curcumin | In vitro and in vivo | Enhances cell death response to therapeutic drugs like doxorubicin | [58] |
| Ruthenium–curcumin | Various cancer cells | Induces cytotoxic effect | [56] |
| Ruthenium–curcumin | Various cancer cell lines | Displays antiproliferative activity and causes apoptosis via DNA interaction and inhibiting MEK/ERK signaling pathway | [59,60] |
| Ruthenium–curcumin | Glioblastoma and breast cancer cells | Induces cell death likely through autophagy | [60] |
| Ruthenium–curcumin | Colon cancer cells | Induces apoptosis through the inhibition of proteasomes | [61] |
| Cobalt–curcumin | Breast cancer cells | Acts as a phototoxin, generates intracellular ROS and causes apoptosis | [62] |
| Palladium–curcumin | Lung cancer cells | Induces cytotoxicity and apoptosis | [63] |
| Palladium–curcumin | Prostate cancer cells | Inhibits cell growth and induces apoptosis via ROS, mitochondrial pathway, and JNK phosphorylation | [64] |
| Palladium–curcumin | Colorectal carcinoma | Exhibits antitumor activity through decrease of prominin-1 (CD133) and SCF release | [65] |
| Manganese–curcumin | Colon, breast, and lung cancer cells | Causes cytotoxic effects | [44] |
| Nickel–curcumin | Cervical carcinoma, lung cancer cells | Exerts anticancer activity by cell cycle arrest, production of ROS, and mitochondrial membrane depolarization | [66] |
| Gallium–curcumin | Bladder, breast, and prostate carcinoma cells | Exhibits cytotoxic effect | [24] |
| Platinum–curcumin | Cervical, liver, breast, and lung adenocarcinoma | Causes phototoxic and apoptosis through cellular ROS | [67,68] |
| Platinum based NP–curcumin | Drug resistant A549/DDP cells | Synergistically enhances the chemotherapeutic effect of drugs | [69] |
| Platinum–curcumin | Cancer cells | Induces apoptotic photocytotoxicity through platinum-bound DNA adducts | [70] |
| Platinum–curcumin | Non-small-cell lung cancer cells | Exhibits anticancer & antimetastatic activity by inhibiting PI3K/AKT, MMP2 and VEGFR2 | [71,72] |
| Oxidovandium–curcumin | Cervical and breast cancer cells | Shows photodynamic therapy effect | [72] |
| Oxovanadium–curcumin | Liver cancer cells | Induces apoptosis through ROS generation | [73] |
| Iron–curcumin | Pancreatic cancer cells | Targets tumor microenvironment via suppression of SHH pathway and CXCR4/CXCL12 signaling | [74] |
| Iron–curcumin | Breast cancer cells | Induces apoptosis and inhibits invasion | [75] |
| Iron–curcumin | Leukemic cells | Increases contrast magnetic resonance resulted in high apoptosis | [76] |
| Arthritis | |||
| Vanadium–curcumin | In vitro | Inhibits smooth muscle cell growth, synoviocyte proliferation, and mouse lymphoma cell growth | [77] |
| Gold–curcumin | Rats | Causes remission in adjuvant induced polyarthritis and reduces paw swelling | [78,79] |
| Osteoporosis | |||
| Lanthanide–curcumin | MG-63 cells | Induces toxicity toward MG-63 cells | [79] |
| Gold NP–curcumin | Bone marrow-derived macrophages and mice | Inhibits RANKL-induced osteoclastogenesis through inhibition of c-Fos, NFATc1, TRAP, and OSCAR; improves bone density and prevents bone loss in mice | [80] |
| Neurological disorders | |||
| Gadolinium–curcumin | Aβ aggregate protein | Inhibits amyloid-β plaques aggregation | [81] |
| Ruthenium–curcumin | Tau peptides | Inhibits aggregation of tau peptide | [82] |
| Copper–curcumin Zinc–curcumin | PC12 cells | Inhibits H2O2-induced neuronal cell death via downregulating NF-κB pathway and upregulating Bcl-2/Bax pathway | [21] |
| Iron–curcumin | Mice | Reduces accumulation of β-amyloid25–35 protein and strengthens memory | [83] |
| Other diseases | |||
| Zinc–curcumin | Rats | Prevents ethanol-induced formation of ulcer lesions | [25] |
| Zinc–curcumin | Rats | Blocks gastric lesions, reduces gastric volume, free acidity, total acidity, and pepsin | [26] |
| Zinc–curcumin | Rats | Exerts hypoglycemic effect | [84] |
| Zinc–curcumin | Mice | Protects reproductive system impairments | [85] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prasad, S.; DuBourdieu, D.; Srivastava, A.; Kumar, P.; Lall, R. Metal–Curcumin Complexes in Therapeutics: An Approach to Enhance Pharmacological Effects of Curcumin. Int. J. Mol. Sci. 2021, 22, 7094. https://doi.org/10.3390/ijms22137094
Prasad S, DuBourdieu D, Srivastava A, Kumar P, Lall R. Metal–Curcumin Complexes in Therapeutics: An Approach to Enhance Pharmacological Effects of Curcumin. International Journal of Molecular Sciences. 2021; 22(13):7094. https://doi.org/10.3390/ijms22137094
Chicago/Turabian StylePrasad, Sahdeo, Dan DuBourdieu, Ajay Srivastava, Prafulla Kumar, and Rajiv Lall. 2021. "Metal–Curcumin Complexes in Therapeutics: An Approach to Enhance Pharmacological Effects of Curcumin" International Journal of Molecular Sciences 22, no. 13: 7094. https://doi.org/10.3390/ijms22137094
APA StylePrasad, S., DuBourdieu, D., Srivastava, A., Kumar, P., & Lall, R. (2021). Metal–Curcumin Complexes in Therapeutics: An Approach to Enhance Pharmacological Effects of Curcumin. International Journal of Molecular Sciences, 22(13), 7094. https://doi.org/10.3390/ijms22137094






