Overcoming Shellfish Allergy: How Far Have We Come? †
Abstract
1. Background
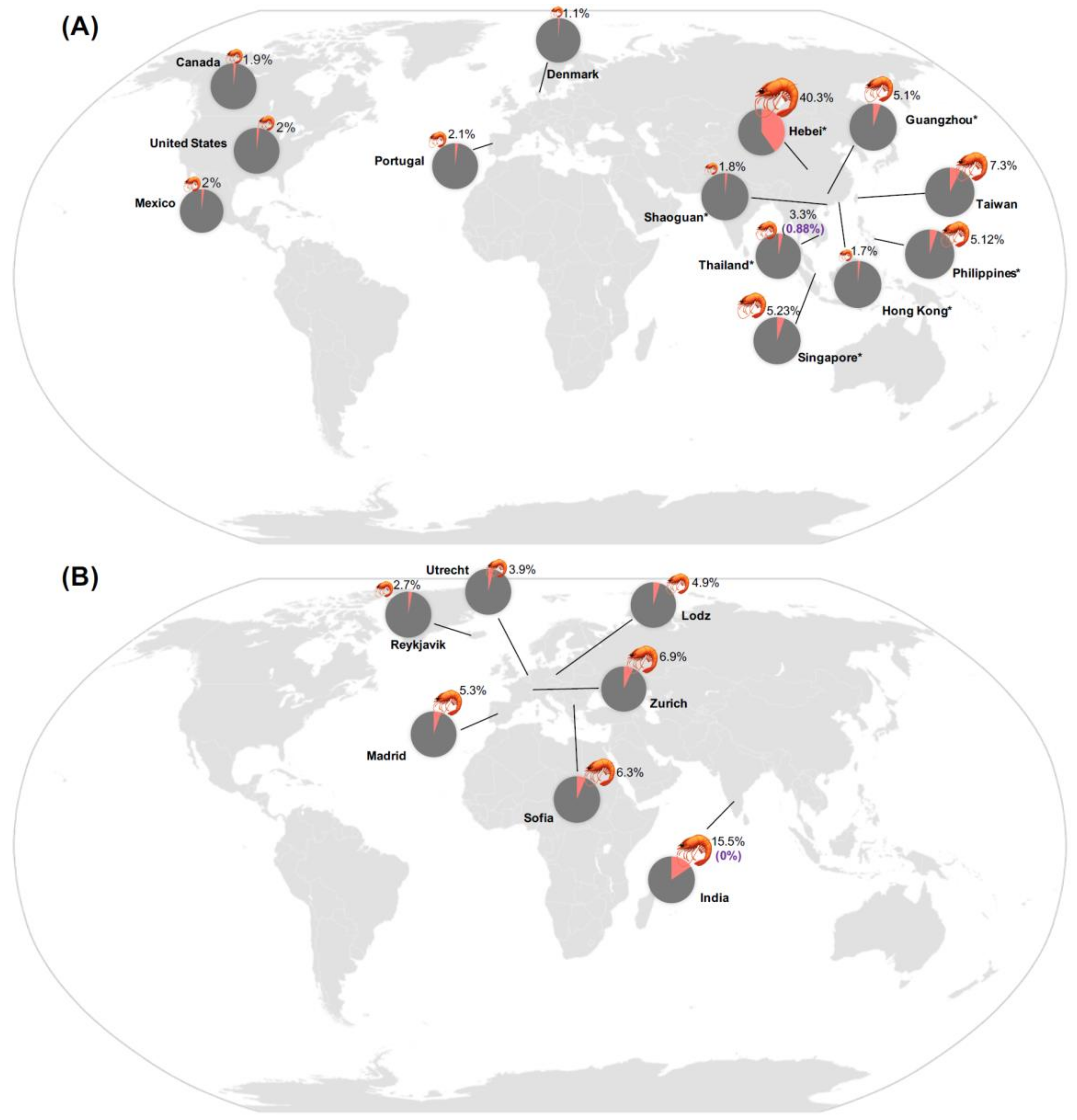
1.1. Epidemiology
1.2. Cross-Reactivity among Shellfish Allergens
1.3. Diagnosis of Shellfish Allergy
1.4. Shellfish-Specific Immunotherapy
1.5. Shrimp Extract and Allergens
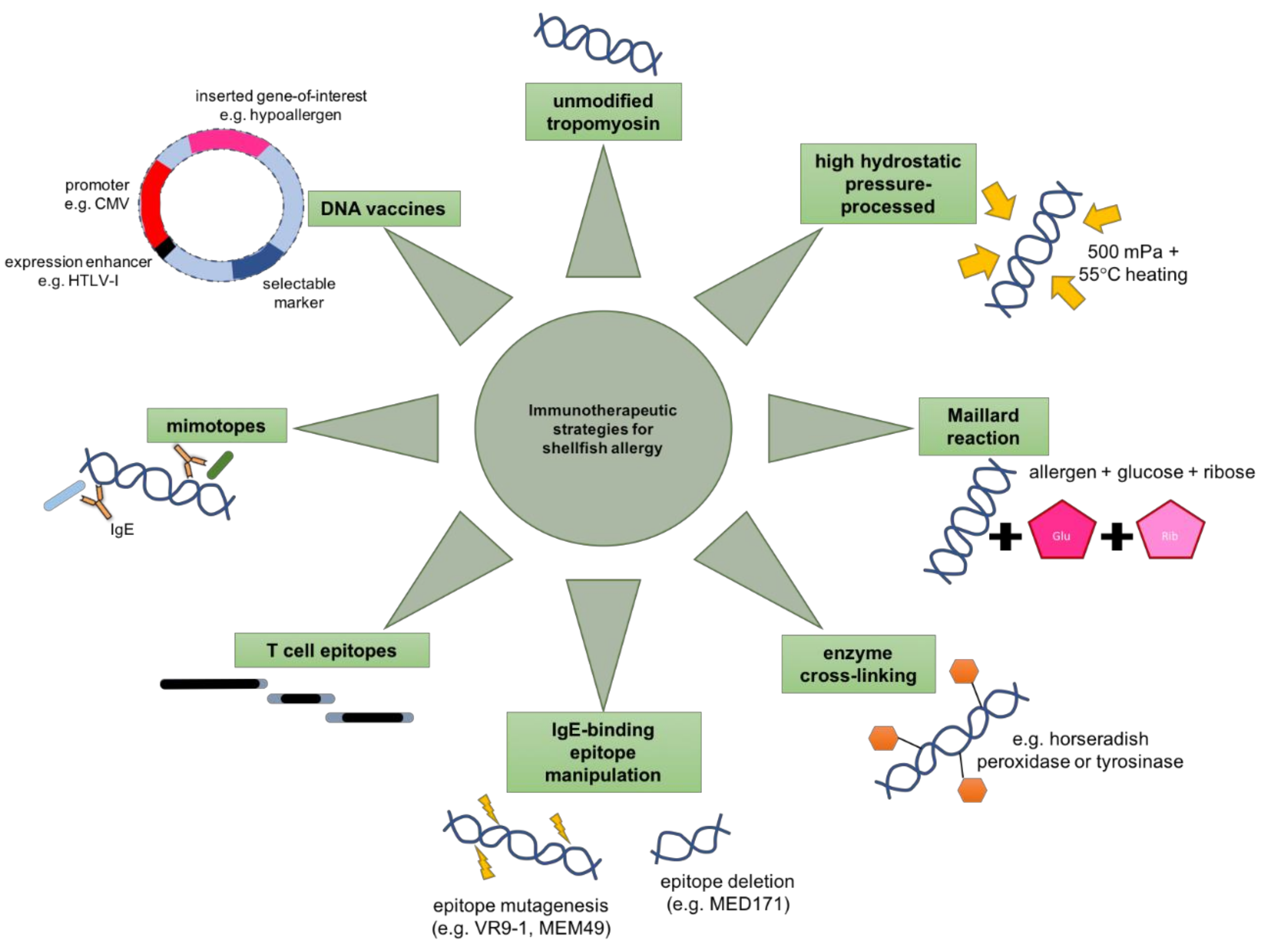
1.6. Hypoallergens
1.7. High Hydrostatic Pressure Processing
1.8. Chemical Modification
1.9. Polypeptide Fragmentation
1.10. Epitope Manipulation
1.11. Immunoregulatory Peptides
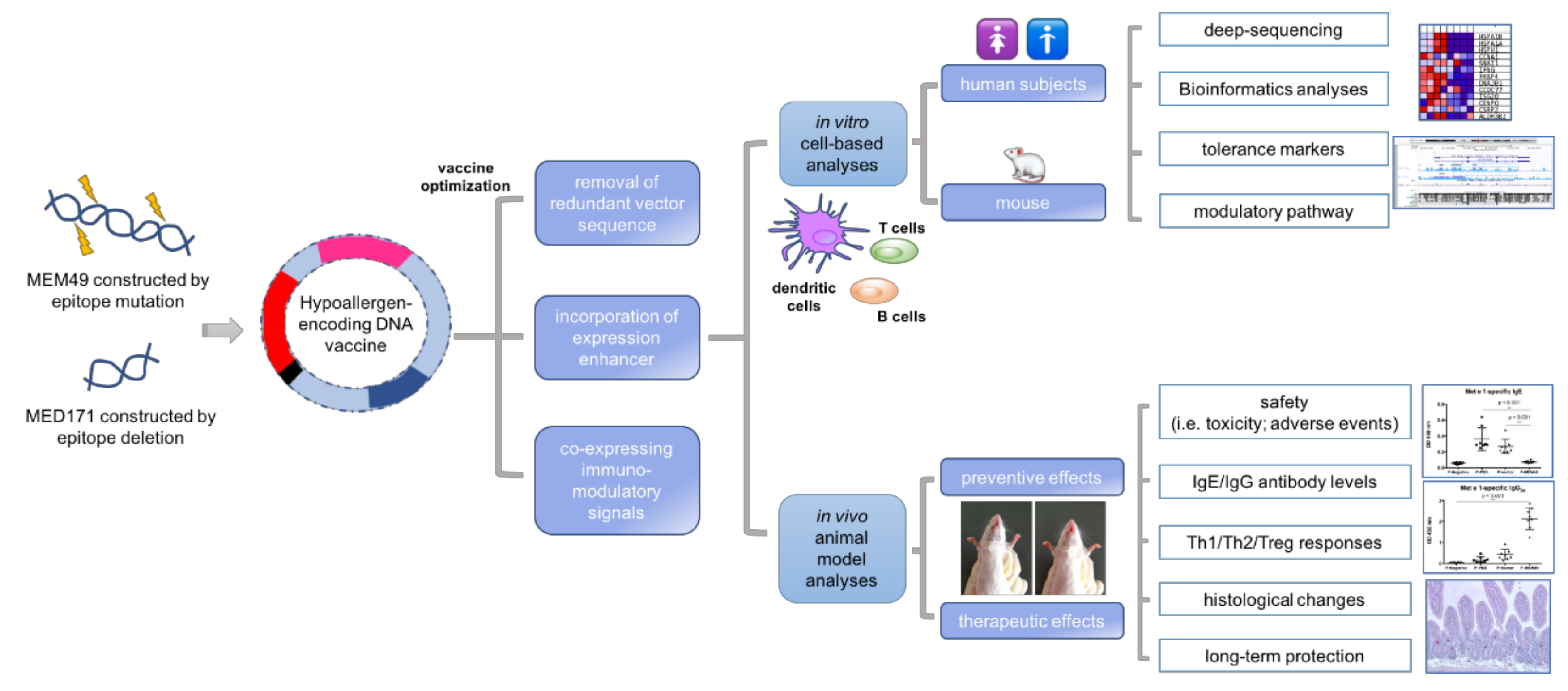
1.12. DNA Vaccine
2. Conclusions
Author Contributions
Funding
Conflicts of Interest
Patents
References
- Venter, C.; Patil, V.; Grundy, J.; Glasbey, G.; Twiselton, R.; Arshad, S.H.; Dean, T. Prevalence and cumulative incidence of food hyper-sensitivity in the first 10 years of life. Pediatr. Allergy Immunol. 2016, 27, 452–458. [Google Scholar] [CrossRef]
- Thalayasingam, M.; Gerez, I.F.; Yap, G.C.; Llanora, G.V.; Chia, I.P.; Chua, L.; Lee, C.J.; Ta, L.D.; Cheng, Y.K.; Thong, B.Y.; et al. Clinical and immunochemical profiles of food challenge proven or anaphylactic shrimp allergy in tropical Singapore. Clin. Exp. Allergy 2015, 45, 687–697. [Google Scholar] [CrossRef]
- Gonzalez-Estrada, A.; Silvers, S.K.; Klein, A.; Zell, K.; Wang, X.F.; Lang, D.M. Epidemiology of anaphylaxis at a tertiary care center: A report of 730 cases. Ann. Allergy Asthma Immunol. 2017, 118, 80–85. [Google Scholar] [CrossRef]
- Lertnawapan, R.; Maek-a-nantawat, W. Anaphylaxis and biphasic phase in Thailand: 4-year observation. Allergol. Int. 2011, 60, 283–289. [Google Scholar] [CrossRef]
- Goh, S.H.; Soh, J.Y.; Loh, W.; Lee, K.P.; Tan, S.C.; Heng, W.J.K.; Ibrahim, I.; Lee, B.W.; Chiang, W.C. Cause and clinical presentation of anaphylaxis in Singapore: From infancy to old age. Int. Arch. Allergy Immunol. 2018, 175, 91–98. [Google Scholar] [CrossRef]
- Smit, D.V.; Cameron, P.A.; Rainer, T.H. Anaphylaxis presentations to an emergency department in Hong Kong: Incidence and predictors of biphasic reactions. J. Emerg. Med. 2005, 28, 381–388. [Google Scholar] [CrossRef]
- Cummings, A.J.; Knibb, R.C.; King, R.M.; Lucas, J.S. The psychosocial impact of food allergy and food hypersensitivity in children, adolescents and their families: A review. Allergy 2010, 65, 933–945. [Google Scholar] [CrossRef]
- Leung, T.F.; Yung, E.; Wong, Y.S.; Li, C.Y.; Wong, G.W. Quality-of-life assessment in Chinese families with food-allergic children. Clin. Exp. Allergy 2009, 39, 890–896. [Google Scholar] [CrossRef]
- Moonesinghe, H.; Mackenzie, H.; Venter, C.; Kilburn, S.; Turner, P.; Weir, K.; Dean, T. Prevalence of fish and shellfish allergy: A systematic review. Ann. Allergy Asthma Immunol. 2016, 117, 264–272.e264. [Google Scholar] [CrossRef]
- Kattan, J. The prevalence and natural history of food allergy. Curr. Allergy Asthma Rep. 2016, 16, 47. [Google Scholar] [CrossRef]
- Sicherer, S.H.; Munoz-Furlong, A.; Sampson, H.A. Prevalence of seafood allergy in the United States determined by a random telephone survey. J. Allergy Clin. Immunol. 2004, 114, 159–165. [Google Scholar] [CrossRef]
- Acker, W.W.; Plasek, J.M.; Blumenthal, K.G.; Lai, K.H.; Topaz, M.; Seger, D.L.; Goss, F.R.; Slight, S.P.; Bates, D.W.; Zhou, L. Prevalence of food allergies and intolerances documented in electronic health records. J. Allergy Clin. Immunol. 2017, 140, 1587–1591. [Google Scholar] [CrossRef]
- Soller, L.; Ben-Shoshan, M.; Harrington, D.W.; Knoll, M.; Fragapane, J.; Joseph, L.; Pierre, Y.S.; La Vieille, S.; Wilson, K.; Elliott, S.J.; et al. Prevalence and predictors of food allergy in Canada: A focus on vulnerable populations. J. Allergy Clin. Immunol. Pract. 2015, 3, 42–49. [Google Scholar] [CrossRef]
- Ontiveros, N.; Valdez-Meza, E.E.; Vergara-Jimenez, M.J.; Canizalez-Roman, A.; Borzutzky, A.; Cabrera-Chavez, F. Parent-reported prevalence of food allergy in Mexican schoolchildren: A population-based study. Allergol. Immunopathol. (Madr.) 2016, 44, 563–570. [Google Scholar] [CrossRef]
- Burney, P.G.; Potts, J.; Kummeling, I.; Mills, E.N.; Clausen, M.; Dubakiene, R.; Barreales, L.; Fernandez-Perez, C.; Fernandez-Rivas, M.; Le, T.M.; et al. The prevalence and distribution of food sensitization in European adults. Allergy 2014, 69, 365–371. [Google Scholar] [CrossRef]
- Lozoya-Ibanez, C.; Morgado-Nunes, S.; Rodrigues, A.; Lobo, C.; Taborda-Barata, L. Prevalence and clinical features of adverse food reactions in Portuguese adults. Allergy Asthma Clin. Immunol. 2016, 12, 36. [Google Scholar] [CrossRef]
- Venkataraman, D.; Erlewyn-Lajeunesse, M.; Kurukulaaratchy, R.J.; Potter, S.; Roberts, G.; Matthews, S.; Arshad, S.H. Prevalence and longitudinal trends of food allergy during childhood and adolescence: Results of the Isle of Wight Birth Cohort study. Clin. Exp. Allergy 2018, 48, 394–402. [Google Scholar] [CrossRef]
- Osterballe, M.; Hansen, T.K.; Mortz, C.G.; Host, A.; Bindslev-Jensen, C. The prevalence of food hypersensitivity in an unselected population of children and adults. Pediatr. Allergy Immunol. 2005, 16, 567–573. [Google Scholar] [CrossRef]
- Hao, G.; Lai, X.; Song, Z.; Wang, Z.; Kong, X.A.; Zhong, H.; Hui, S.F.; Zheng, Y. Self-reported questionnaire survey on the prevalence and symptoms of adverse food reactions in patients with chronic inhalant diseases in Tangshan city, China. Allergy Asthma Clin. Immunol. 2018, 14, 3. [Google Scholar] [CrossRef]
- Wu, T.C.; Tsai, T.C.; Huang, C.F.; Chang, F.Y.; Lin, C.C.; Huang, I.F.; Chu, C.H.; Lau, B.H.; Wu, L.; Peng, H.J.; et al. Prevalence of food allergy in Taiwan: A questionnaire-based survey. Intern. Med. J. 2012, 42, 1310–1315. [Google Scholar] [CrossRef]
- Leung, T.F.; Yung, E.; Wong, Y.S.; Lam, C.W.; Wong, G.W. Parent-reported adverse food reactions in Hong Kong Chinese pre-schoolers: Epidemiology, clinical spectrum and risk factors. Pediatr. Allergy Immunol. 2009, 20, 339–346. [Google Scholar] [CrossRef]
- Ho, M.H.; Lee, S.L.; Wong, W.H.; Ip, P.; Lau, Y.L. Prevalence of self-reported food allergy in Hong Kong children and teens—A population survey. Asian Pac. J. Allergy Immunol. 2012, 30, 275–284. [Google Scholar]
- Yang, Z.; Zhao, J.; Wei, N.; Feng, M.; Xian, M.; Shi, X.; Zheng, Z.; Su, Q.; Wong, G.W.K.; Li, J. Cockroach is a major cross-reactive allergen source in shrimp-sensitized rural children in southern China. Allergy 2018, 73, 585–592. [Google Scholar] [CrossRef]
- Li, J.; Ogorodova, L.M.; Mahesh, P.A.; Wang, M.H.; Fedorova, O.S.; Leung, T.F.; Fernandez-Rivas, M.; Mills, E.N.C.; Potts, J.; Kummeling, I.; et al. Comparative study of food allergies in children from China, India, and Russia: The EuroPrevall-INCO surveys. J. Allergy Clin. Immunol. Pract. 2019. [Google Scholar] [CrossRef]
- Shek, L.P.; Cabrera-Morales, E.A.; Soh, S.E.; Gerez, I.; Ng, P.Z.; Yi, F.C.; Ma, S.; Lee, B.W. A population-based questionnaire survey on the prevalence of peanut, tree nut, and shellfish allergy in 2 Asian populations. J. Allergy Clin. Immunol. 2010, 126, 324–331. [Google Scholar] [CrossRef]
- Mahesh, P.A.; Wong, G.W.K.; Ogorodova, L.; Potts, J.; Leung, T.F.; Fedorova, O.; Holla, A.D.; Fernandez-Rivas, M.; Mills, E.N.C.; Kummeling, I.; et al. Prevalence of food sensitization and probable food allergy among adults in India: The EuroPrevall INCO study. Allergy 2016, 71, 1010–1019. [Google Scholar] [CrossRef]
- Lao-araya, M.; Trakultivakorn, M. Prevalence of food allergy among preschool children in northern Thailand. Pediatr. Int. 2012, 54, 238–243. [Google Scholar] [CrossRef]
- Woo, C.K.; Bahna, S.L. Not all shellfish “allergy” is allergy! Clin. Transl. Allergy 2011, 1, 3. [Google Scholar] [CrossRef]
- Shanti, K.N.; Martin, B.M.; Nagpal, S.; Metcalfe, D.D.; Rao, P.V.S. Identification of tropomyosin as the major shrimp allergen and characterization of its IgE-binding epitopes. J. Immunol. 1993, 151, 5354–5363. [Google Scholar]
- Reese, G.; Jeoung, B.J.; Daul, C.B.; Lehrer, S.B. Characterization of recombinant shrimp allergen Pen a 1 (tropomyosin). Int. Arch. Allergy Immunol. 1997, 113, 240–242. [Google Scholar] [CrossRef]
- Leung, P.S.C.; Chu, K.H.; Chow, W.K.; Ansari, A.; Bandea, C.I.; Kwan, H.S.; Nagy, S.M.; Gershwin, M.E. Cloning, expression, and primary structure of Metapenaeus ensis tropomyosin, the major heat-stable shrimp allergen. J. Allergy Clin. Immunol. 1994, 94, 882–890. [Google Scholar] [CrossRef]
- DeWitt, A.M.; Mattsson, L.; Lauer, I.; Reese, G.; Lidholm, J. Recombinant tropomyosin from Penaeus aztecus (rPen a 1) for measurement of specific immunoglobulin E antibodies relevant in food allergy to crustaceans and other invertebrates. Mol. Nutr. Food Res. 2004, 48, 370–379. [Google Scholar] [CrossRef]
- Giuffrida, M.G.; Villalta, D.; Mistrello, G.; Amato, S.; Asero, R. Shrimp allergy beyond tropomyosin in Italy: Clinical relevance of arginine kinase, sarcoplasmic calcium binding protein and hemocyanin. Eur. Ann. Allergy Clin. Immunol. 2014, 46, 172–177. [Google Scholar]
- Ayuso, R.; Grishina, G.; Bardina, L.; Carrillo, T.; Blanco, C.; Ibanez, M.D.; Sampson, H.A.; Beyer, K. Myosin light chain is a novel shrimp allergen, Lit v 3. J. Allergy Clin. Immunol. 2008, 122, 795–802. [Google Scholar] [CrossRef]
- Ayuso, R.; Grishina, G.; Ibanez, M.D.; Blanco, C.; Carrillo, T.; Bencharitiwong, R.; Sanchez, S.; Nowak-Wegrzyn, A.; Sampson, H.A. Sarcoplasmic calcium-binding protein is an EF-hand-type protein identified as a new shrimp allergen. J. Allergy Clin. Immunol. 2009, 124, 114–120. [Google Scholar] [CrossRef]
- Bauermeister, K.; Wangorsch, A.; Garoffo, L.P.; Reuter, A.; Conti, A.; Taylor, S.L.; Lidholm, J.; Dewitt, A.M.; Enrique, E.; Vieths, S.; et al. Generation of a comprehensive panel of crustacean allergens from the North Sea Shrimp Crangon crangon. Mol. Immunol. 2011, 48, 1983–1992. [Google Scholar] [CrossRef]
- Ayuso, R.; Grishina, G.; Pascal, M.; Sanchez-Garcia, S.; Towle, D.; Smith, C.; Ibanez, M.; Sampson, H.A. Hemocyanin, troponin C and Fatty Acid-binding Protein (FABP) may be cross-reactive allergens between crustaceans, cockroach and dust mites. J. Allergy Clin. Immunol. 2011, 127, AB235. [Google Scholar] [CrossRef]
- Faber, M.A.; Pascal, M.; El Kharbouchi, O.; Sabato, V.; Hagendorens, M.M.; Decuyper, I.I.; Bridts, C.H.; Ebo, D.G. Shellfish allergens: Tropomyosin and beyond. Allergy 2017, 72, 842–848. [Google Scholar] [CrossRef]
- Piboonpocanun, S.; Jirapongsananuruk, O.; Tipayanon, T.; Boonchoo, S.; Goodman, R.E. Identification of hemocyanin as a novel non-cross-reactive allergen from the giant freshwater shrimp Macrobrachium rosenbergii. Mol. Nutr. Food Res. 2011, 55, 1492–1498. [Google Scholar] [CrossRef]
- Pascal, M.; Grishina, G.; Yang, A.C.; Sanchez-Garcia, S.; Lin, J.; Towle, D.; Ibanez, M.D.; Sastre, J.; Sampson, H.A.; Ayuso, R. Molecular diagnosis of shrimp allergy: Efficiency of several allergens to predict clinical reactivity. J. Allergy Clin. Immunol. Pract. 2015, 3, 521–529. [Google Scholar] [CrossRef]
- QinWai, C.Y.Y.; Leung, N.Y.H.; Leung, A.S.Y.; Lam, M.C.Y.; Xu, K.; Shum, Y.; Wong, G.W.K.; Leung, T.F. Troponin C is the major shrimp allergen among Chinese patients with shellfish allergy. J. Allergy Clin. Immunol. 2019, 143, AB270. [Google Scholar]
- Leung, N.Y.H.; Wai, C.Y.Y.; Shu, S.; Wang, J.; Kenny, T.P.; Chu, K.H.; Leung, P.S.C. Current immunological and molecular biological perspectives on seafood allergy: A comprehensive review. Clin. Rev. Allergy Immunol. 2014, 46, 180–197. [Google Scholar] [CrossRef] [PubMed]
- Vidal, C.; Bartolome, B.; Rodriguez, V.; Armisen, M.; Linneberg, A.; Gonzalez-Quintela, A. Sensitization pattern of crustacean-allergic individuals can indicate allergy to molluscs. Allergy 2015, 70, 1493–1496. [Google Scholar] [CrossRef] [PubMed]
- Klaewsongkram, J. High prevalence of shellfish and house dust mite allergies in Asia-Pacific: Probably not just a coincidence. Asian Pac. J. Allergy Immunol. 2012, 30, 247–248. [Google Scholar]
- Wong, L.; Huang, C.H.; Lee, B.W. Shellfish and house dust mite allergies: Is the link tropomyosin? Allergy Asthma Immunol. Res. 2016, 8, 101–106. [Google Scholar] [CrossRef]
- Yu, C.J.; Lin, Y.F.; Chiang, B.L.; Chow, L.P. Proteomics and immunological analysis of a novel shrimp allergen, Pen m 2. J. Immunol. 2003, 170, 445–453. [Google Scholar] [CrossRef]
- Broekman, H.; Verhoeckx, K.C.; Jager, C.F.D.; Kruizinga, A.G.; Pronk-Kleinjan, M.; Remington, B.C.; Bruijnzeel-Koomen, C.A.; Houben, G.F.; Knulst, A.C. Majority of shrimp-allergic patients are allergic to mealworm. J. Allergy Clin. Immunol. 2016, 137, 1261–1263. [Google Scholar] [CrossRef]
- Jirapongsananuruk, O.; Sripramong, C.; Pacharn, P.; Udompunturak, S.; Chinratanapisit, S.; Piboonpocanun, S.; Visitsunthorn, N.; Vichyanond, P. Specific allergy to Penaeus monodon (seawater shrimp) or Macrobrachium rosenbergii (freshwater shrimp) in shrimp-allergic children. Clin. Exp. Allergy 2008, 38, 1038–1047. [Google Scholar] [CrossRef]
- Lieberman, J.A.; Sicherer, S.H. Diagnosis of food allergy: Epicutaneous skin tests, in vitro tests, and oral food challenge. Curr. Allergy Asthma Rep. 2011, 11, 58–64. [Google Scholar] [CrossRef]
- Sicherer, S.H.; Sampson, H.A. Food allergy: A review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J. Allergy Clin. Immunol. 2018, 141, 41–58. [Google Scholar] [CrossRef]
- Gamez, C.; Sanchez-Garcia, S.; Ibanez, M.D.; Lopez, R.; Aguado, E.; Lopez, E.; Sastre, B.; Sastre, J.; del Pozo, V. Tropomyosin IgE-positive results are a good predictor of shrimp allergy. Allergy 2011, 66, 1375–1383. [Google Scholar] [CrossRef]
- Asero, R.; Scala, E.; Villalta, D.; Pravettoni, V.; Arena, A.; Billeri, L.; Colombo, G.; Cortellini, G.; Cucinelli, F.; De Cristofaro, M.L.; et al. Shrimp allergy: Analysis of commercially available extracts for in vivo diagnosis. J. Investig. Allergol. Clin. Immunol. 2017, 27, 175–182. [Google Scholar] [CrossRef]
- Fernandez-Rivas, M.; Barreales, L.; Mackie, A.R.; Fritsche, P.; Vazquez-Cortes, S.; Jedrzejczak-Czechowicz, M.; Kowalski, M.L.; Clausen, M.; Gislason, D.; Sinaniotis, A.; et al. The EuroPrevall outpatient clinic study on food allergy: Background and methodology. Allergy 2015, 70, 576–584. [Google Scholar] [CrossRef]
- Nakamura, R.; Uchida, Y.; Higuchi, M.; Nakamura, R.; Tsuge, I.; Urisu, A.; Teshima, R. A convenient and sensitive allergy test: IgE crosslinking-induced luciferase expression in cultured mast cells. Allergy 2010, 65, 1266–1273. [Google Scholar] [CrossRef]
- Jarupalee, T.; Chatchatee, P.; Komolpis, K.; Suratannon, N.; Roytraku, S.; Yingchutraku, Y.; Yimchuen, W.; Bun, P.; Jacquet, A.; Palaga, T. Detecting allergens from black tiger shrimp penaeus monodon that can bind and cross-link IgE by ELISA, western blot, and a humanized rat basophilic leukemia reporter cell line RS-ATL8. Allergy Asthma Immunol. Res. 2018, 10, 62–76. [Google Scholar] [CrossRef]
- Yu, W.; Freeland, D.M.; Nadeau, K.C. Food allergy: Immune mechanisms, diagnosis and immunotherapy. Nat. Rev. Immunol. 2016, 16, 751–765. [Google Scholar] [CrossRef]
- Refaat, M.M.; Attia, M.Y.; Saber, H.M. Desensitization efficacy by sublingual immunotherapy of shrimps extract in asthmatic, rhinitis and urticaria allergic patients. Food Nutr. Sci. 2014, 5, 1704–1710. [Google Scholar] [CrossRef]
- Leung, N.Y.H.; Wai, C.Y.Y.; Shu, S.A.; Chang, C.C.; Chu, K.H.; Leung, P.S.C. Low-dose allergen-specific immunotherapy induces tolerance in a murine model of shrimp allergy. Int. Arch. Allergy Immunol. 2017, 174, 86–96. [Google Scholar] [CrossRef]
- Mahajan, A.; Youssef, L.A.; Cleyrat, C.; Grattan, R.; Lucero, S.R.; Mattison, C.P.; Erasmus, M.F.; Jacobson, B.; Tapia, L.; Hlavacek, W.S.; et al. Allergen valency, dose, and FcepsilonRI occupancy set thresholds for secretory responses to Pen a 1 and motivate design of hypoallergens. J. Immunol. 2017, 198, 1034–1046. [Google Scholar] [CrossRef]
- Myrset, H.R.; Faeste, C.K.; Kristiansen, P.E.; Dooper, M.M. Mapping of the immunodominant regions of shrimp tropomyosin Pan b 1 by human IgE-binding and IgE receptor crosslinking studies. Int. Arch. Allergy Immunol. 2013, 162, 25–38. [Google Scholar] [CrossRef]
- Wang, C.Y.; Huang, H.W.; Hsu, C.P.; Yang, B.B. Recent advances in food processing using high hydrostatic pressure technology. Crit. Rev. Food Sci. Nutr. 2016, 56, 527–540. [Google Scholar] [CrossRef] [PubMed]
- Long, F.Y.; Yang, X.; Wang, R.R.; Hu, X.S.; Chen, F. Effects of combined high pressure and thermal treatments on the allergenic potential of shrimp (Litopenaeus vannamei) tropomyosin in a mouse model of allergy. Innov. Food Sci. Emerg. Technol. 2015, 29, 119–124. [Google Scholar] [CrossRef]
- Plundrich, N.J.; Kulis, M.; White, B.L.; Grace, M.H.; Guo, R.; Burks, A.W.; Davis, J.P.; Lila, M.A. Novel strategy to create hypoallergenic peanut protein-polyphenol edible matrices for oral immunotherapy. J. Agric. Food Chem. 2014, 62, 7010–7021. [Google Scholar] [CrossRef] [PubMed]
- Schulke, S.; Burggraf, M.; Waibler, Z.; Wangorsch, A.; Wolfheimer, S.; Kalinke, U.; Vieths, S.; Toda, M.; Scheurer, S. A fusion protein of flagellin and ovalbumin suppresses the TH2 response and prevents murine intestinal allergy. J. Allergy Clin. Immunol. 2011, 128, 1340–1348. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Kawasaki, H.; Hsu, S.C.; Lee, R.T.; Yao, X.; Plunkett, B.; Fu, J.; Yang, K.; Lee, Y.C.; Huang, S.K. Oral tolerance to food-induced systemic anaphylaxis mediated by the C-type lectin SIGNR1. Nat. Med. 2010, 16, 1128–1133. [Google Scholar] [CrossRef] [PubMed]
- Gruber, P.; Vieths, S.; Wangorsch, A.; Nerkamp, J.; Hofmann, T. Maillard reaction and enzymatic browning affect the allergenicity of Pru av 1, the major allergen from cherry (Prunus avium). J. Agric. Food Chem. 2004, 52, 4002–4007. [Google Scholar] [CrossRef]
- Nakamura, A.; Sasaki, F.; Watanabe, K.; Ojima, T.; Ahn, D.H.; Saeki, H. Changes in allergenicity and digestibility of squid tropomyosin during the Maillard reaction with ribose. J. Agric. Food Chem. 2006, 54, 9529–9534. [Google Scholar] [CrossRef]
- Nakamura, A.; Watanabe, K.; Ojima, T.; Ahn, D.H.; Saeki, H. Effect of Maillard reaction on allergenicity of scallop tropomyosin. J. Agric. Food Chem. 2005, 53, 7559–7564. [Google Scholar] [CrossRef]
- Liu, G.Y.; Hu, M.J.; Sun, L.C.; Han, X.Y.; Liu, Q.M.; Alcocer, M.; Fei, D.X.; Cao, M.J.; Liu, G.M. Allergenicity and oral tolerance of enzymatic cross-linked tropomyosin evaluated using cell and mouse models. J. Agric. Food Chem. 2017, 65, 2205–2213. [Google Scholar] [CrossRef]
- Fei, D.X.; Liu, Q.M.; Chen, F.; Yang, Y.; Chen, Z.W.; Cao, M.J.; Liu, G.M. Assessment of the sensitizing capacity and allergenicity of enzymatic cross-linked arginine kinase, the crab allergen. Mol. Nutr. Food Res. 2016, 60, 1707–1718. [Google Scholar] [CrossRef]
- Reese, G.; Viebranz, J.; Leong-Kee, S.M.; Plante, M.; Lauer, I.; Randow, S.; Moncin, M.S.; Ayuso, R.; Lehrer, S.B.; Vieths, S. Reduced allergenic potency of VR9-1, a mutant of the major shrimp allergen Pen a 1 (tropomyosin). J. Immunol. 2005, 175, 8354–8364. [Google Scholar] [CrossRef] [PubMed]
- Ayuso, R.; Lehrer, S.B.; Reese, G. Identification of continuous, allergenic regions of the major shrimp allergen Pen a 1 (tropomyosin). Int. Arch. Allergy Immunol. 2002, 127, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.N.; Lin, H.; Pawar, R.; Li, Z.X.; Li, M.H. Mapping IgE binding epitopes of major shrimp (Penaeus monodon) allergen with immunoinformatics tools. Food Chem. Toxicol. 2011, 49, 2954–2960. [Google Scholar] [CrossRef] [PubMed]
- Wai, C.Y.Y.; Leung, N.Y.H.; Ho, M.H.; Gershwin, L.J.; Shu, S.A.; Leung, P.S.C.; Chu, K.H. Immunization with hypoallergens of shrimp allergen tropomyosin inhibits shrimp tropomyosin specific IgE reactivity. PLoS ONE 2014, 9, e111649. [Google Scholar] [CrossRef]
- Sugimoto, M.; Kamemura, N.; Nagao, M.; Irahara, M.; Kagami, S.; Fujisawa, T.; Kido, H. Differential response in allergen-specific IgE, IgGs, and IgA levels for predicting outcome of oral immunotherapy. Pediatr. Allergy Immunol. 2016, 27, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Cady, C.T.; Powell, M.S.; Harbeck, R.J.; Giclas, P.C.; Murphy, J.R.; Katial, R.K.; Weber, R.W.; Hogarth, P.M.; Johnson, S.; Bonvini, E.; et al. IgG antibodies produced during subcutaneous allergen immunotherapy mediate inhibition of basophil activation via a mechanism involving both FcgammaRIIA and FcgammaRIIB. Immunol. Lett. 2010, 130, 57–65. [Google Scholar] [CrossRef]
- Strait, R.T.; Morris, S.C.; Finkelman, F.D. IgG-blocking antibodies inhibit IgE-mediated anaphylaxis in vivo through both antigen interception and Fc gamma RIIb cross-linking. J. Clin. Investig. 2006, 116, 833–841. [Google Scholar] [CrossRef]
- Burton, O.T.; Tamayo, J.M.; Stranks, A.J.; Koleoglou, K.J.; Oettgen, H.C. Allergen-specific IgG antibody signaling through FcgammaRIIb promotes food tolerance. J. Allergy Clin. Immunol. 2017. [Google Scholar] [CrossRef]
- James, L.K.; Till, S.J. Potential mechanisms for IgG4 inhibition of immediate hypersensitivity reactions. Curr. Allergy Asthma Rep. 2016, 16, 23. [Google Scholar] [CrossRef]
- Ravkov, E.V.; Pavlov, I.Y.; Martins, T.B.; Gleich, G.J.; Wagner, L.A.; Hill, H.R.; Delgado, J.C. Identification and validation of shrimp-tropomyosin specific CD4 T cell epitopes. Hum. Immunol. 2013, 74, 1542–1549. [Google Scholar] [CrossRef]
- Wai, C.Y.Y.; Leung, N.Y.H.; Leung, P.S.C.; Chu, K.H. T cell epitope immunotherapy ameliorates allergic responses in a murine model of shrimp allergy. Clin. Exp. Allergy 2016, 46, 491–503. [Google Scholar] [CrossRef] [PubMed]
- Knittelfelder, R.; Riemer, A.B.; Jensen-Jarolim, E. Mimotope vaccination—From allergy to cancer. Expert Opin. Biol. Ther. 2009, 9, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Wallmann, J.; Epstein, M.M.; Singh, P.; Brunner, R.; Szalai, K.; El-Housseiny, L.; Pali-Scholl, I.; Jensen-Jarolim, E. Mimotope vaccination for therapy of allergic asthma: Anti-inflammatory effects in a mouse model. Clin. Exp. Allergy 2010, 40, 650–658. [Google Scholar] [CrossRef]
- Wallmann, J.; Proell, M.; Stepanoska, T.; Hantusch, B.; Pali-Scholl, I.; Thalhamer, T.; Thalhamer, J.; Jensen-Jarolim, E.; Hartl, A. A mimotope gene encoding the major IgE epitope of allergen Phl p 5 for epitope-specific immunization. Immunol. Lett. 2009, 122, 68–75. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Leung, N.Y.H.; Wai, C.Y.Y.; Ho, M.H.; Liu, R.; Lam, K.S.; Wang, J.J.; Shu, S.A.; Chu, K.H.; Leung, P.S.C. Screening and identification of mimotopes of the major shrimp allergen tropomyosin using one-bead-one-compound peptide libraries. Cell. Mol. Immunol. 2017, 14, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Leung, N.Y.H.; Wai, C.Y.Y.; Chu, K.H.; Leung, P.S.C. Mimotope-based allergen-specific immunotherapy: Ready for prime time? Cell. Mol. Immunol. 2019, 16, 890–891. [Google Scholar] [CrossRef]
- Lee, M.F.; Song, P.P.; Lin, T.M.; Chiu, Y.T.; Chen, Y.H. For t 2 DNA vaccine prevents Forcipomyia taiwana (biting midge) allergy in a mouse model. Allergy 2016, 71, 522–531. [Google Scholar] [CrossRef]
- Su, Y.; Connolly, M.; Marketon, A.; Heiland, T. CryJ-LAMP DNA vaccines for Japanese Red Cedar allergy induce robust Th1-type immune responses in murine model. J. Immunol. Res. 2016, 2016, 4857869. [Google Scholar] [CrossRef]
- Li, L.; Petrovsky, N. Molecular mechanisms for enhanced DNA vaccine immunogenicity. Expert Rev. Vaccines 2016, 15, 313–329. [Google Scholar] [CrossRef]
- Rajcani, J.; Mosko, T.; Rezuchova, I. Current developments in viral DNA vaccines: Shall they solve the unsolved? Rev. Med. Virol. 2005, 15, 303–325. [Google Scholar] [CrossRef]
- Muthumani, K.; Block, P.; Flingai, S.; Muruganantham, N.; Chaaithanya, I.K.; Tingey, C.; Wise, M.; Reuschel, E.L.; Chung, C.; Muthumani, A.; et al. Rapid and long-term immunity elicited by DNA-encoded antibody prophylaxis and DNA vaccination against chikungunya virus. J. Infect. Dis. 2016, 214, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Ensell, M.; Zhou, Y.; Nair, U.; Glickstein, J.; Kermany, M.H.; Cai, Q.; Cai, C.; Liu, W.; Deng, Y.P.; et al. Prevention and treatment of DNA vaccine encoding cockroach allergen Bla g 1 in a mouse model of allergic airway inflammation. Allergy 2012, 67, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Hemmi, H.; Takeuchi, O.; Kawai, T.; Kaisho, T.; Sato, S.; Sanjo, H.; Matsumoto, M.; Hoshino, K.; Wagner, H.; Takeda, K.; et al. A Toll-like receptor recognizes bacterial DNA. Nature 2000, 408, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Chu, K.H.; Leung, P.S.C.; Wai, C.Y.Y.; Leung, N.Y.H. Met e 1 Tropomyosin Variants for Use in Allergen-Specific Immunotherapy. U.S. Patent No. 10,301,365, 28 May 2019. [Google Scholar]
- Wai, C.Y.Y.; Leung, N.Y.H.; Leung, P.S.C.; Chu, K.H. Modulating shrimp tropomyosin-mediated allergy: Hypoallergen DNA vaccines induce regulatory T cells to reduce hypersensitivity in mouse model. Int. J. Mol. Sci. 2019, 20, 4656. [Google Scholar] [CrossRef] [PubMed]
- Wai, C.Y.Y.; Qin, J.; Leung, N.Y.H.; Leung, P.S.C.; Chu, K.H. IgE signalling repression by a designer DNA vaccine to modulate shrimp hypersensitivity. EMJ Allergy Immunol. 2019, 4, 69–71. [Google Scholar]
- Wai, C.Y.Y.; Leung, N.Y.H.; Leung, P.S.C.; Chu, K.H. Immunotherapy of food allergy: A comprehensive review. Clin. Rev. Allergy Immunol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Zuidmeer-Jongejan, L.; Fernandez-Rivas, M.; Poulsen, L.K.; Neubauer, A.; Asturias, J.; Blom, L.; Boye, J.; Bindslev-Jensen, C.; Clausen, M.; Ferrara, R.; et al. FAST: Towards safe and effective subcutaneous immunotherapy of persistent life-threatening food allergies. Clin. Transl. Allergy 2012, 2, 5. [Google Scholar] [CrossRef]
| Allergen | Allergen Nomenclature | Molecular Weight (kDa) | Rate of IgE Sensitization | Route of Exposure | Cross-Reactivity |
|---|---|---|---|---|---|
| Tropomyosin | Lit v 1; Met e 1; Pen a 1; Pen m 1; Pen b 1 | 34–38 | 23–83% | Ingestion | Arthropods (crustaceans, HDM, cockroach), mollusks |
| Inhalation | |||||
| Arginine kinase | Lit v 2; Pen m 2 | 40–45 | 10–15% | Ingestion | Shrimp, lobster, crab and crawfish |
| Inhalation | |||||
| Sarcoplasmic calcium-binding protein | Lit v 4; Pen m 4 | 20–25 | 10–15% | Ingestion | None |
| Myosin light chain | Lit v 3; Pen m 3 | 17–20 | >50% | Ingestion | None |
| Troponin C | Cra c 6 | 21–21 | 20–30% | Ingestion | NR |
| Triose phosphate isomerase | Cra c 8; Pen m 8 | 28 | 20–30% | Ingestion | NR |
| Inhalation | |||||
| Fatty acid-binding protein | - | 15–20 | 10.3% | NR | NR |
| Hemocyanin | - | 60–80 | 29–47% | NR | Macrobrachium resenbergii, Penaeus monodon and HDM |
| Paramyosins | - | 100 | NR | NR | Mollusks |
| myosin heavy chain | - | 225 | NR | NR | NR |
| 𝛼-actin | - | 94–99 | NR | NR | Shrimp and HDM |
| β-actin | - | 41–46 | NR | NR | Shrimp and HDM |
| ubiquitin | - | 5–8.5 | NR | NR | Shrimp and HDM |
| Glyceraldehyde phosphate dehydrogenase | - | 37 | NR | NR | NR |
| Smooth endoplasmic reticulum Ca + + ATPase | - | 113 | NR | NR | NR |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wai, C.Y.Y.; Leung, N.Y.H.; Chu, K.H.; Leung, P.S.C.; Leung, A.S.Y.; Wong, G.W.K.; Leung, T.F. Overcoming Shellfish Allergy: How Far Have We Come? Int. J. Mol. Sci. 2020, 21, 2234. https://doi.org/10.3390/ijms21062234
Wai CYY, Leung NYH, Chu KH, Leung PSC, Leung ASY, Wong GWK, Leung TF. Overcoming Shellfish Allergy: How Far Have We Come? International Journal of Molecular Sciences. 2020; 21(6):2234. https://doi.org/10.3390/ijms21062234
Chicago/Turabian StyleWai, Christine Y.Y., Nicki Y.H. Leung, Ka Hou Chu, Patrick S.C. Leung, Agnes S.Y. Leung, Gary W.K. Wong, and Ting Fan Leung. 2020. "Overcoming Shellfish Allergy: How Far Have We Come?" International Journal of Molecular Sciences 21, no. 6: 2234. https://doi.org/10.3390/ijms21062234
APA StyleWai, C. Y. Y., Leung, N. Y. H., Chu, K. H., Leung, P. S. C., Leung, A. S. Y., Wong, G. W. K., & Leung, T. F. (2020). Overcoming Shellfish Allergy: How Far Have We Come? International Journal of Molecular Sciences, 21(6), 2234. https://doi.org/10.3390/ijms21062234








