Human Platelet Lysate Supports Efficient Expansion and Stability of Wharton’s Jelly Mesenchymal Stromal Cells via Active Uptake and Release of Soluble Regenerative Factors
Abstract
1. Introduction
2. Results
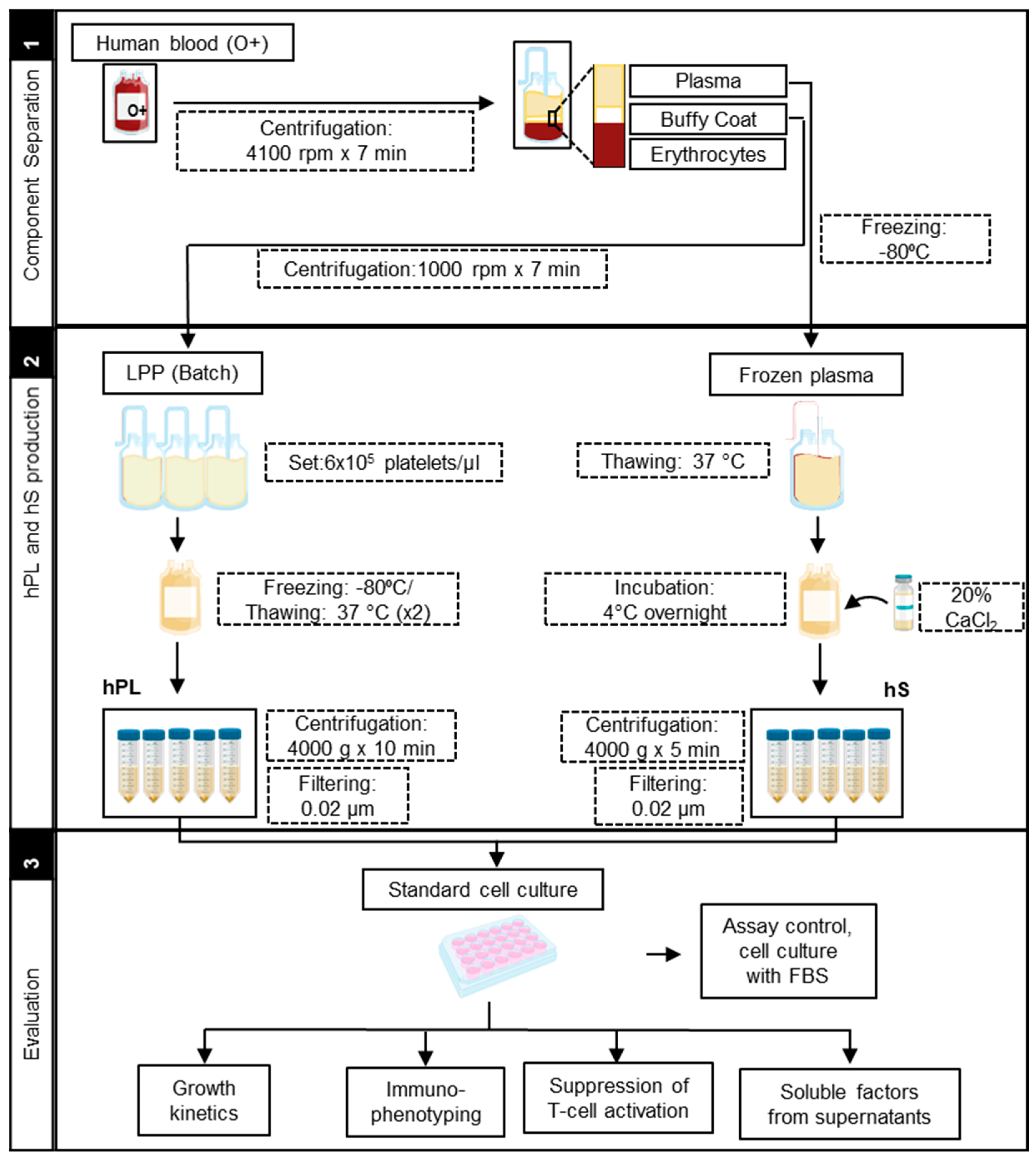
2.1. Production of hPL and hS for Culture of WJ-MSC
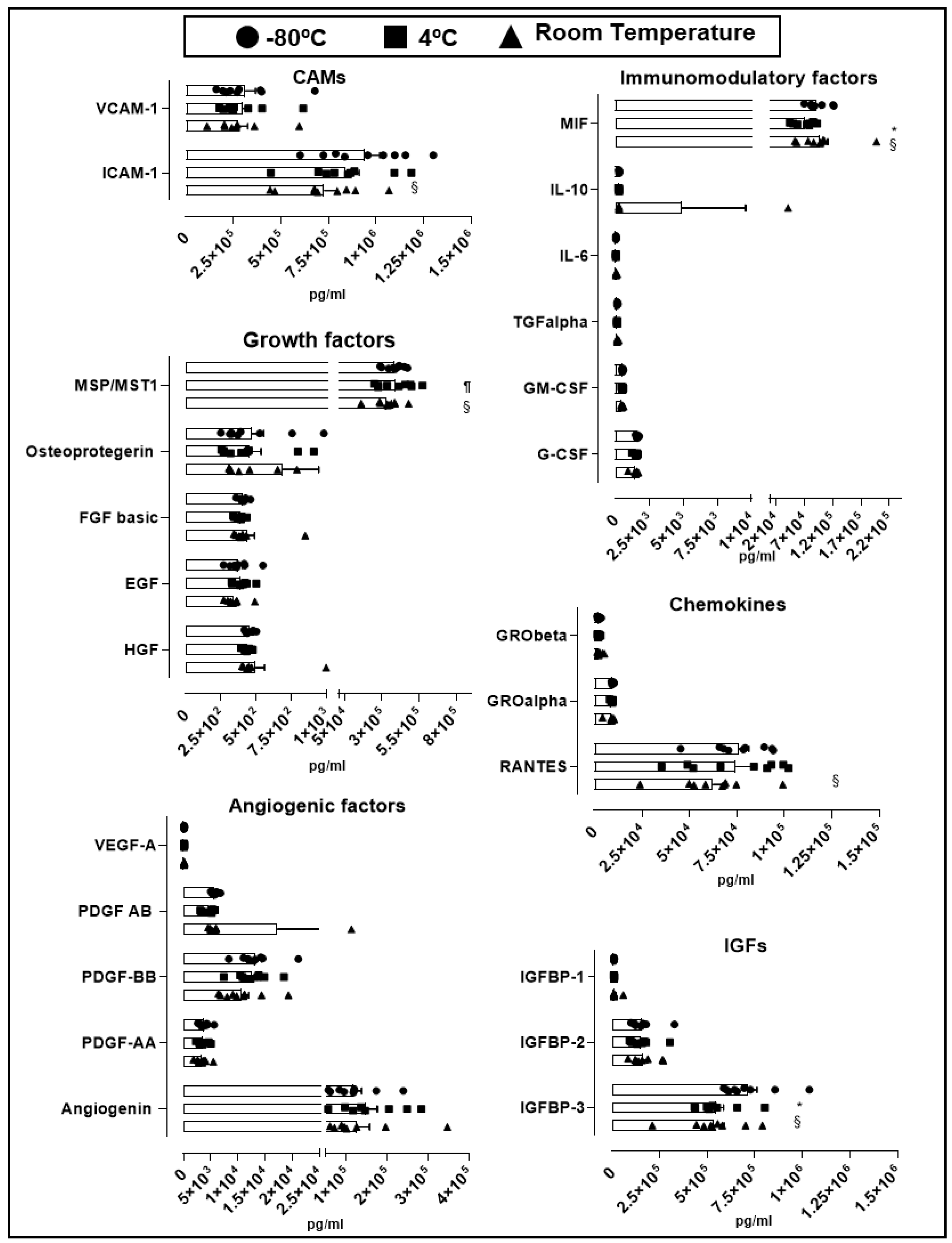
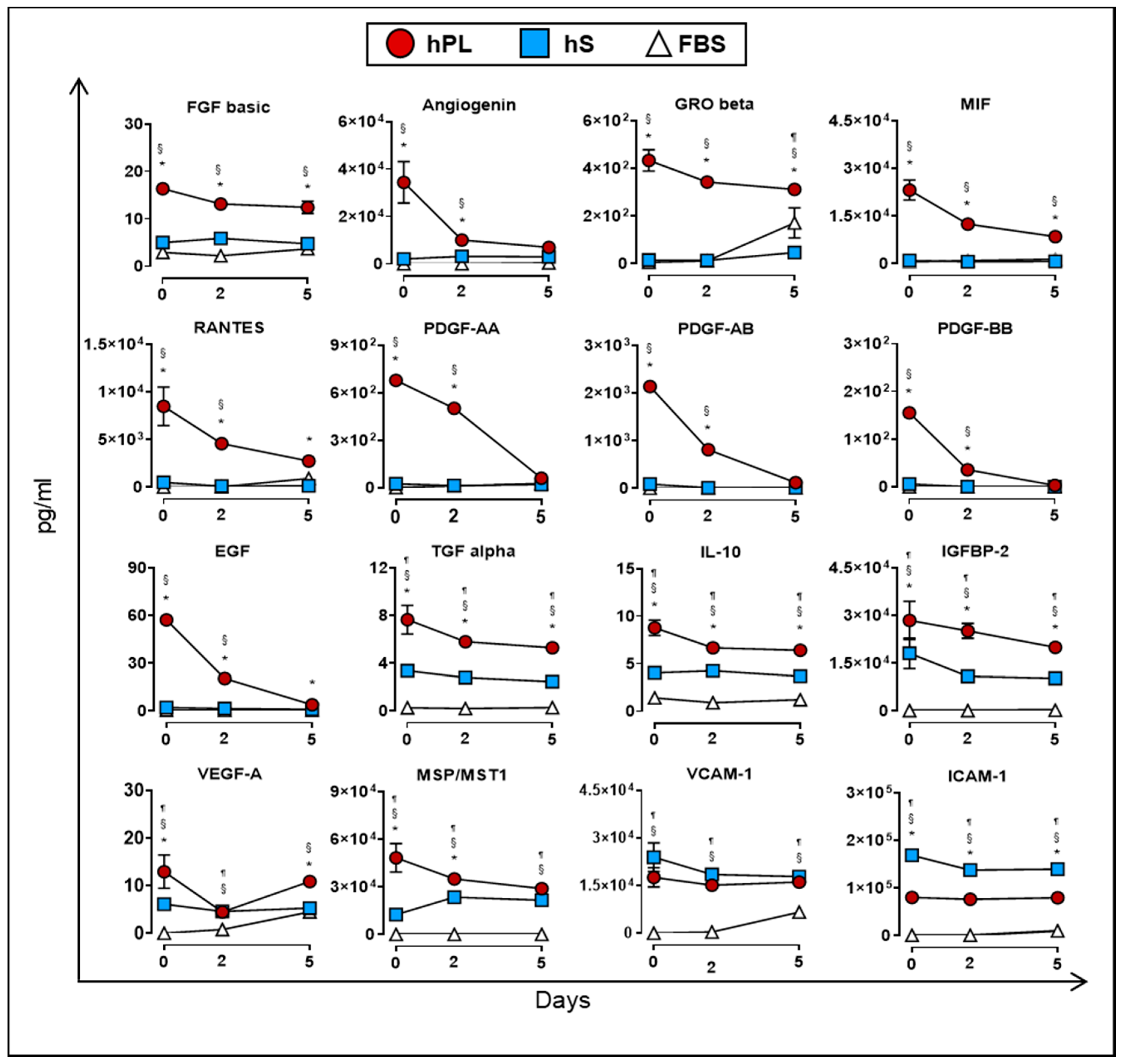
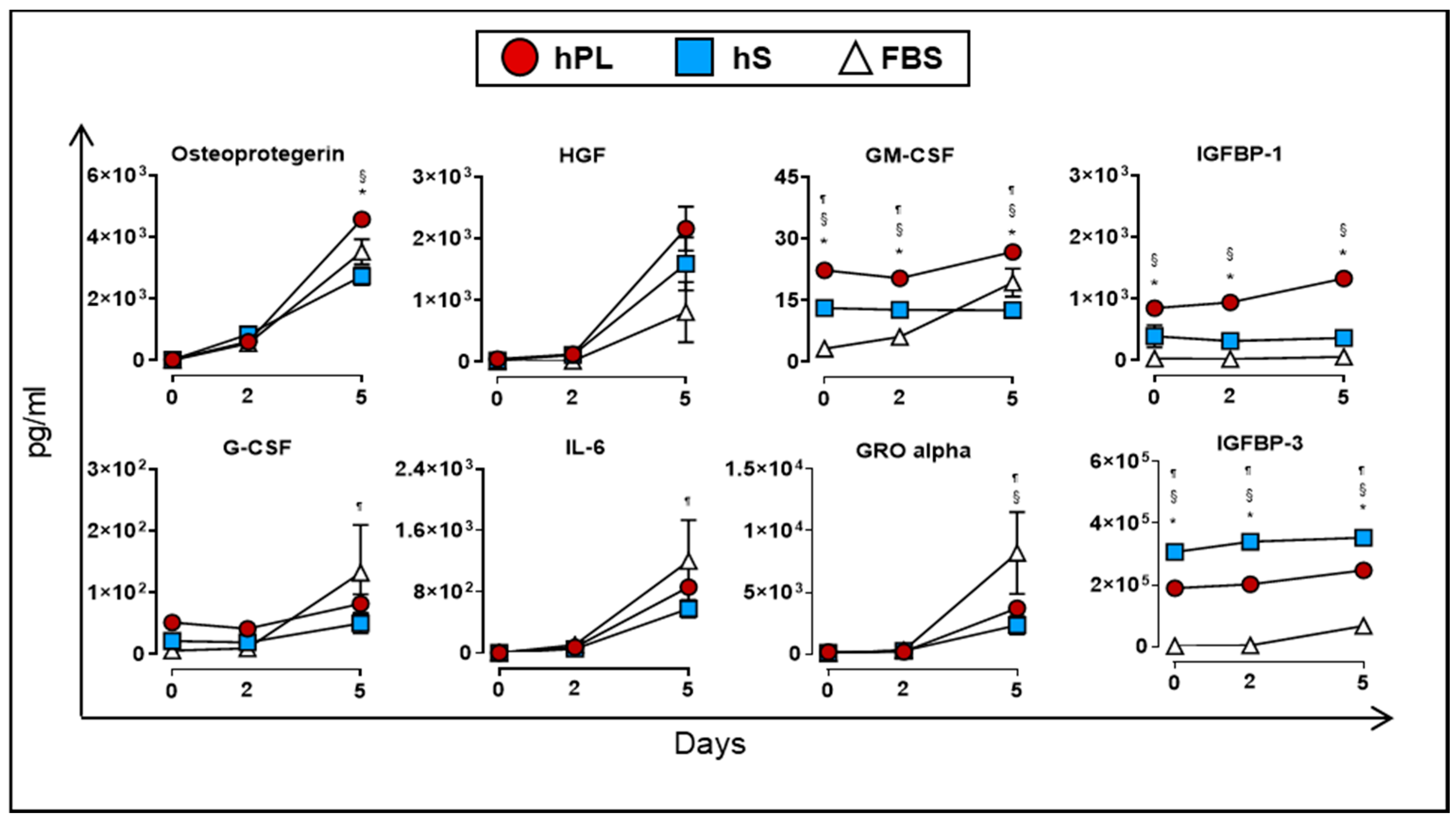
2.2. Cytokine and Growth Factor Stability Present in hPL
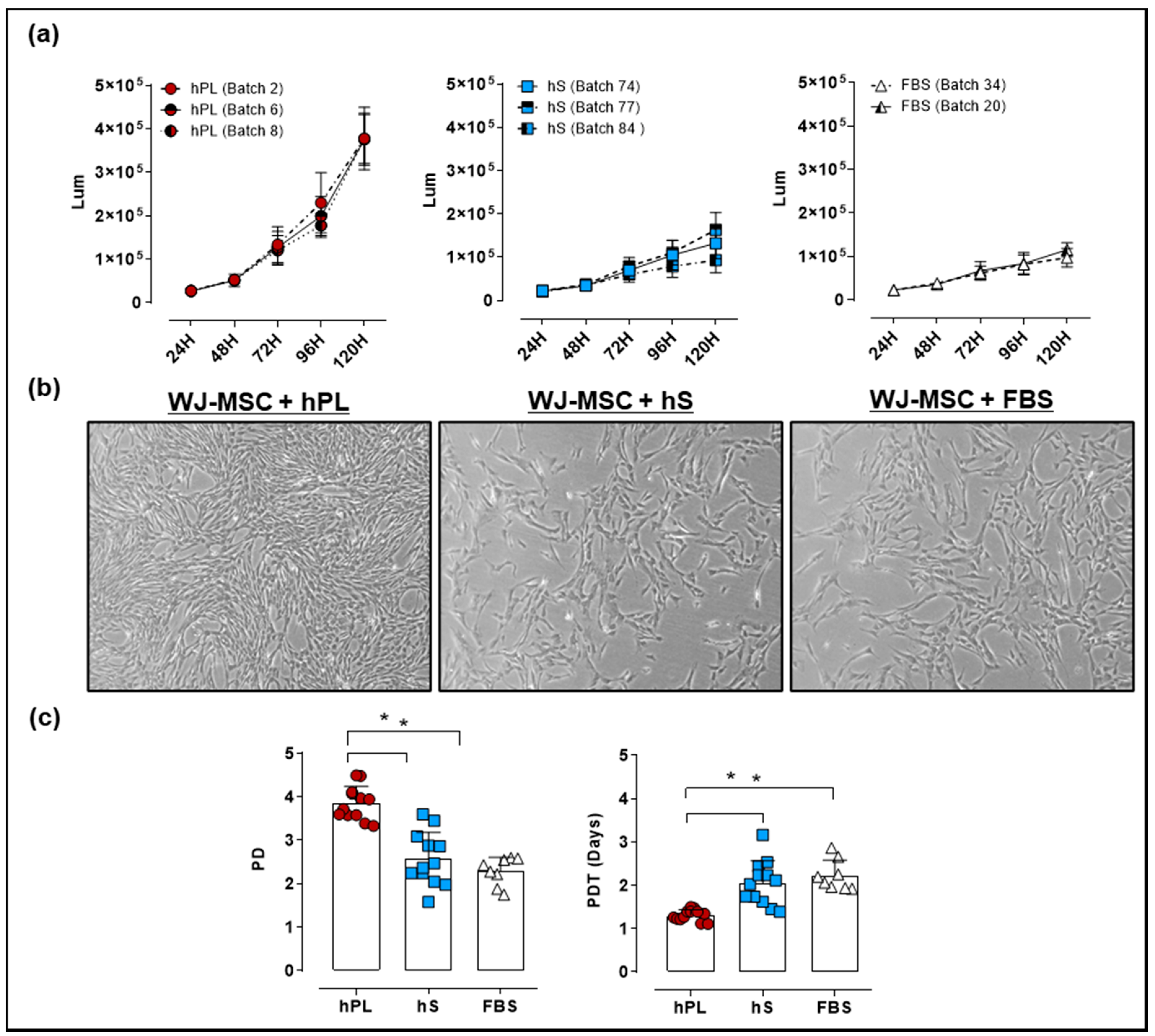
2.3. Comparative Assessment of hPL, hS and FBS in the Expansion of WJ-MSC
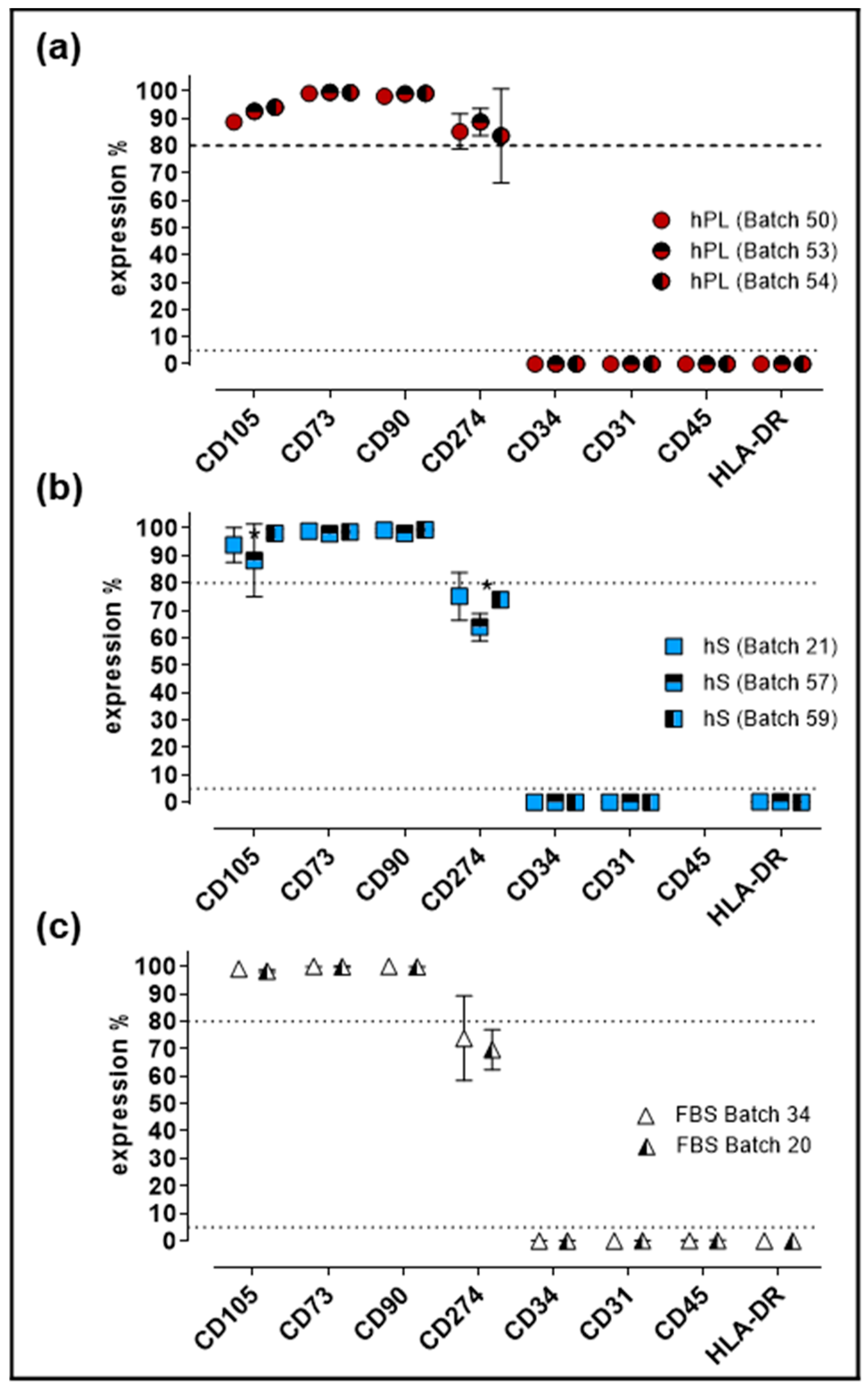
2.4. Impact of hPL, hS and FBS in Cell Identity of WJ-MSC
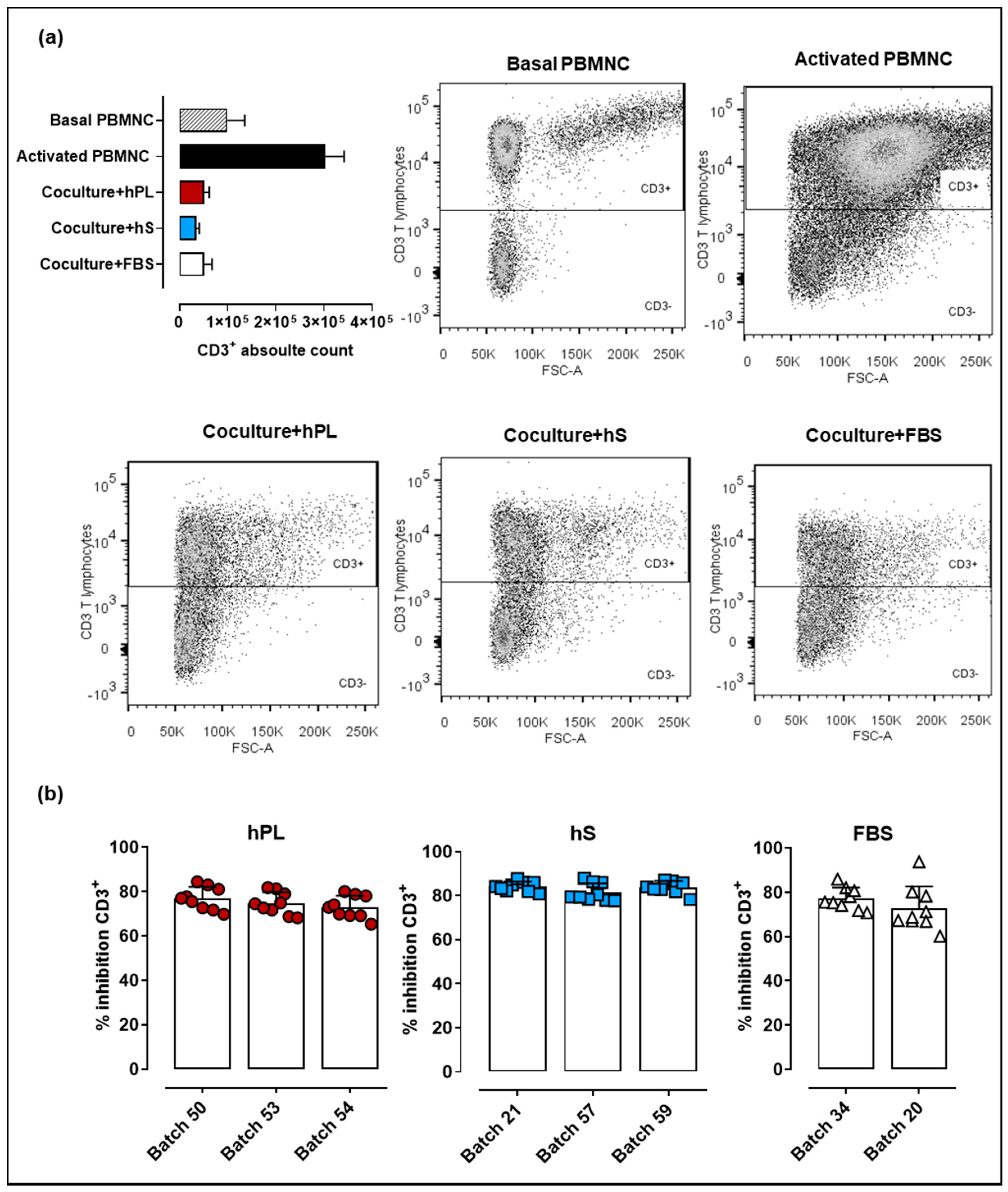
2.5. WJ-MSC Expanded with hPL, hS and FBS Maintain their Immune Suppressive Potency
2.6. Analysis of Cytokines and Growth Factors Present in Culture Supernatants of WJ-MSC
3. Discussion
4. Materials and Methods
4.1. Human Platelet Lysate (hPL) Production
4.2. Human SERUM (hS) Production
4.3. Isolation and Culture of WJ-MSC
4.4. Evaluation of Cell Growth Kinetics in hPL, hS and FBS-Supplemented Media
4.5. WJ-MSC Immunophenotype Analyses
4.6. Assessment T Lymphocyte Suppression by WJ-MSC
4.7. Determination of Cytokine and Growth Factor Levels by Multiplex Bead Analysis
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| WJ-MSC | Wharton’s Jelly Mesenchymal Stromal Cells |
| hPL | Human Platelet Lysate |
| hS | Human Serum |
| FBS | Fetal bovine serum |
| ATMP | Advanced Therapy Medicinal Products |
References
- Juhl, M.; Tratwal, J.; Follin, B.; Søndergaard, R.H.; Kirchhoff, M.; Ekblond, A.; Kastrup, J.; Haack-Sørensen, M. Comparison of clinical grade human platelet lysates for cultivation of mesenchymal stromal cells from bone marrow and adipose tissue. Scand. J. Clin. Lab. Investig. 2016, 76, 93–104. [Google Scholar] [CrossRef]
- Bieback, K. Platelet lysate as replacement for fetal bovine serum in mesenchymal stromal cell cultures. Transfus. Med. Hemother. 2013, 40, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Kocaoemer, A.; Kern, S.; Klüter, H.; Bieback, K. Human AB Serum and Thrombin-Activated Platelet-Rich Plasma Are Suitable Alternatives to Fetal Calf Serum for the Expansion of Mesenchymal Stem Cells from Adipose Tissue. Stem Cells 2007, 25, 1270–1278. [Google Scholar] [CrossRef] [PubMed]
- Bieback, K.; Fernandez-Muñoz, B.; Pati, S.; Schäfer, R. Gaps in the knowledge of human platelet lysate as a cell culture supplement for cell therapy: A joint publication from the AABB and the International Society for Cell & Gene Therapy. Cytotherapy 2019, 21, 911–924. [Google Scholar] [PubMed]
- Kandoi, S.; Patra, B.; Vidyasekar, P.; Sivanesan, D.; Vijayalakshmi, S.; Rajagopal, K.; Verma, R.S. Evaluation of platelet lysate as a substitute for FBS in explant and enzymatic isolation methods of human umbilical cord MSCs. Sci. Rep. 2018, 8, 12439. [Google Scholar] [CrossRef] [PubMed]
- Bieback, K.; Hecker, A.; Kocaömer, A.; Lannert, H.; Schallmoser, K.; Strunk, D.; Klüter, H. Human Alternatives to Fetal Bovine Serum for the Expansion of Mesenchymal Stromal Cells from Bone Marrow. Stem Cells 2009, 27, 2331–2341. [Google Scholar] [CrossRef]
- Antoninus, A.A.; Widowati, W.; Wijaya, L.; Agustina, D.; Puradisastra, S.; Sumitro, S.B.; Widodo, M.; Bachtiar, I. Human platelet lysate enhances the proliferation of Wharton’s jelly-derived mesenchymal stem cells. Biomark. Genom. Med. 2015, 7, 87–97. [Google Scholar] [CrossRef][Green Version]
- Sara, C.; Mareschi, K.; Labanca, L.; Lucania, G.; Leone, M.; Sanavio, F.; Castello, L.; Rustichelli, D.; Signorino, E.; Gunetti, M.; et al. Inactivated human platelet lysate with psoralen: A new perspective for mesenchymal stromal cell production in Good Manufacturing Practice conditions. Cytotherapy 2014, 16, 750–763. [Google Scholar] [CrossRef]
- Alio, J.L.; Rodriguez, A.E.; WróbelDudzińska, D. Eye platelet-rich plasma in the treatment of ocular surface disorders. Curr. Opin. Ophthalmol. 2015, 26, 325–332. [Google Scholar] [CrossRef]
- Alio, J.L.; Arnalich-Montiel, F.; Rodriguez, A.E. The Role of “Eye Platelet Rich Plasma” (E-Prp) for Wound Healing in Ophthalmology. Curr. Pharm. Biotechnol. 2012, 13, 1257–1265. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.-H.; Im, C.-J.; Huh, J.-Y.; Suh, J.-J.; Lee, S.-H. Effect of platelet-rich plasma on bone regeneration in autogenous bone graft. Int. J. Oral Maxillofac. Surg. 2004, 33, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Barro, L.; Su, Y.; Nebie, O.; Wu, Y.-W.; Huang, Y.; Koh, M.B.; Knutson, F.; Burnouf, T. A double-virally-inactivated (Intercept-solvent/detergent) human platelet lysate for in vitro expansion of human mesenchymal stromal cells. Transfusion 2019, 59, 2061–2073. [Google Scholar] [CrossRef] [PubMed]
- Barro, L.; Nebie, O.; Chen, M.-S.; Wu, Y.-W.; Koh, M.B.; Knutson, F.; Watanabe, N.; Takahara, M.; Burnouf, T. Nanofiltration of growth media supplemented with human platelet lysates for pathogen-safe xeno-free expansion of mesenchymal stromal cells. Cytotherapy 2020, 22, 458–472. [Google Scholar] [CrossRef] [PubMed]
- Marino, L.; Castaldi, M.A.; Rosamilio, R.; Ragni, E.; Vitolo, R.; Fulgione, C.; Castaldi, S.G.; Serio, B.; Bianco, R.; Guida, M.; et al. Mesenchymal Stem Cells from the Wharton’s Jelly of the Human Umbilical Cord: Biological Properties and Therapeutic Potential. Int. J. Stem Cells 2019, 12, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Liau, L.; Ruszymah, B.; Ng, M.; Law, J.X. Characteristics and clinical applications of Wharton’s jelly-derived mesenchymal stromal cells. Curr. Res. Transl. Med. 2020, 68, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Shih, D.T.-B.; Burnouf, T. Preparation, quality criteria, and properties of human blood platelet lysate supplements for ex vivo stem cell expansion. New Biotechnol. 2015, 32, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Silva-Cote, I.; Cruz-Barrera, M.; Cañas-Arboleda, M.; Correa-Araujo, L.; Méndez, L.; Jagielska, J.; Camacho, B.; Salguero, G. Strategy for the Generation of Engineered Bone Constructs Based on Umbilical Cord Mesenchymal Stromal Cells Expanded with Human Platelet Lysate. Stem Cells Int. 2019, 2019, 7198215. [Google Scholar] [CrossRef]
- Prat, C.; Rodríguez-Gómez, L.; Aylagas, M.; Nieto-Nicolau, N.; Gastelurrutia, P.; Agustí, E.; Gálvez-Montón, C.; Jorba, I.; Teis, A.; Monguió-Tortajada, M.; et al. First-in-human PeriCord cardiac bioimplant: Scalability and GMP manufacturing of an allogeneic engineered tissue graft. EBioMedicine 2020, 54, 102729. [Google Scholar] [CrossRef]
- Abbaszadeh, H.; Ghorbani, F.; Derakhshani, M.; Movassaghpour, A.A.; Yousefi, M.; Talebi, M.; Shamsasenjan, K. Regenerative potential of Wharton’s jelly-derived mesenchymal stem cells: A new horizon of stem cell therapy. J. Cell. Physiol. 2020. [Google Scholar] [CrossRef]
- Davies, J.E.; Walker, J.T.; Keating, A. Concise Review: Wharton’s Jelly: The Rich, but Enigmatic, Source of Mesenchymal Stromal Cells. Stem Cells Transl. Med. 2017, 6, 1620–1630. [Google Scholar] [CrossRef]
- Mohamed, H.E.; Asker, M.E.; Kotb, N.S.; El Habab, A.M. Human platelet lysate efficiency, stability, and optimal heparin concentration required in culture of mammalian cells. Blood Res. 2020, 55, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Laner-Plamberger, S.; Oeller, M.; Mrazek, C.; Hartl, A.; Sonderegger, A.; Rohde, E.; Strunk, D.; Schallmoser, K. Upregulation of mitotic bookmarking factors during enhanced proliferation of human stromal cells in human platelet lysate. J. Transl. Med. 2019, 17, 432. [Google Scholar] [CrossRef] [PubMed]
- Burnouf, T.; Strunk, D.; Koh, M.B.; Schallmoser, K. Human platelet lysate: Replacing fetal bovine serum as a gold standard for human cell propagation? Biomaterials 2016, 76, 371–387. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, D.; Harloff, S.; Liu, L.; Schlenke, P.; Wedel, T.; Geerling, G. Epitheliotrophic capacity of a growth factor preparation produced from platelet concentrates on corneal epithelial cells: A potential agent for the treatment of ocular surface defects? Transfusion 2004, 44, 1724–1731. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, S.; Wang, S.; Soares, F.; Fischer, M.; Meng, F.-L.; Du, Z.; Lin, C.; Meyer, C.; DeCaprio, J.A.; et al. Transcriptional landscape of the human cell cycle. Proc. Natl. Acad. Sci. USA 2017, 114, 3473–3478. [Google Scholar] [CrossRef]
- Chen, X.; Whitney, E.M.; Gao, S.Y.; Yang, V.W. Transcriptional Profiling of Krüppel-like Factor 4 Reveals a Function in Cell Cycle Regulation and Epithelial Differentiation. J. Mol. Biol. 2003, 326, 665–677. [Google Scholar] [CrossRef]
- Yang, J.; Sung, E.; Donlin-Asp, P.G.; Corces, V.G. A subset of Drosophila Myc sites remain associated with mitotic chromosomes colocalized with insulator proteins. Nat. Commun. 2013, 4, 1464. [Google Scholar] [CrossRef]
- Gharibi, B.; Hughes, F.J. Effects of Medium Supplements on Proliferation, Differentiation Potential, and In Vitro Expansion of Mesenchymal Stem Cells. Stem Cells Transl. Med. 2012, 1, 771–782. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, X.; Cao, W.; Shi, Y. Plasticity of mesenchymal stem cells in immunomodulation: Pathological and therapeutic implications. Nat. Immunol. 2014, 15, 1009–1016. [Google Scholar] [CrossRef]
- Zhukareva, V.; Obrocka, M.; Houlé, J.D.; Fischer, I.; Neuhuber, B. Secretion profile of human bone marrow stromal cells: Donor variability and response to inflammatory stimuli. Cytokine 2010, 50, 317–321. [Google Scholar] [CrossRef]
- Czapla, J.; Matuszczak, S.; Kulik, K.; Wiśniewska, E.; Pilny, E.; Jarosz, M.; Smolarczyk, R.; Sirek, T.; Zembala, M.O.; Zembala, M.; et al. The effect of culture media on large-scale expansion and characteristic of adipose tissue-derived mesenchymal stromal cells. Stem Cell Res. Ther. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Waterman, R.S.; Tomchuck, S.L.; Henkle, S.L.; Betancourt, A.M. A New Mesenchymal Stem Cell (MSC) Paradigm: Polarization into a Pro-Inflammatory MSC1 or an Immunosuppressive MSC2 Phenotype. PLoS ONE 2010, 5, e10088. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Rebollo, E.; Mentrup, B.; Ebert, R.; Franzen, J.; Abagnale, G.; Sieben, T.; Ostrowska, A.; Hoffmann, P.; Roux, P.-F.; Rath, B.; et al. Human Platelet Lysate versus Fetal Calf Serum: These Supplements Do Not Select for Different Mesenchymal Stromal Cells. Sci. Rep. 2017, 7, 5132. [Google Scholar] [CrossRef] [PubMed]
- Tancharoen, W.; Aungsuchawan, S.; Pothacharoen, P.; Bumroongkit, K.; Puaninta, C.; Pangjaidee, N.; Narakornsak, S.; Markmee, R.; Laowanitwattana, T.; Thaojamnong, C. Human platelet lysate as an alternative to fetal bovine serum for culture and endothelial differentiation of human amniotic fluid mesenchymal stem cells. Mol. Med. Rep. 2019, 19, 5123–5132. [Google Scholar] [CrossRef] [PubMed]
- Rauch, C. Alternatives to the use of fetal bovine serum: Human platelet lysates as a serum substitute in cell culture media. ALTEX Altern. Anim. Exp. 2011, 28, 305–316. [Google Scholar] [CrossRef]
- Joo, M.W.; Chung, S.J.; Shin, S.H.; Chung, Y.G. The Effect of Autologous Platelet-Rich Plasma on Bone Regeneration by Autologous Mesenchymal Stem Cells Loaded onto Allogeneic Cancellous Bone Granules. Cells Tissues Organs 2017, 203, 327–338. [Google Scholar] [CrossRef]
- Oliver-Vila, I.; Ramírez-Moncayo, C.; Grau-Vorster, M.; Marín-Gallén, S.; Caminal, M.; Vives, J. Optimisation of a potency assay for the assessment of immunomodulative potential of clinical grade multipotent mesenchymal stromal cells. Cytotechnology 2018, 70, 31–44. [Google Scholar] [CrossRef]

| Batch | Platelets/µL | Leukocytes/µL | Erythrocytes/µL | pH | |||
|---|---|---|---|---|---|---|---|
| Initial | Adjusted | Initial | Adjusted | Initial | Adjusted | Final | |
| 2 | 9.7 × 105 | 6.1 × 105 | 1.0 × 102 | 6.0 × 101 | 5.0 × 104 | 3.0 × 104 | * |
| 5 | 8.3 × 105 | 5.8 × 105 | 5.9 × 102 | 4.0 × 102 | 4.0 × 104 | 3.0 × 104 | * |
| 6 | 1.1 × 106 | 5.8 × 105 | 2.9 × 102 | 1.8 × 102 | 8.0 × 104 | 4.0 × 104 | * |
| 8 | 6.1 × 105 | 5.9 × 105 | 1.4 × 102 | 1.3 × 102 | 3.0 × 104 | 3.0 × 104 | * |
| 23 | 1.1 × 106 | 5.8 × 105 | 1.2 × 102 | 7.0 × 101 | 5.0 × 104 | 2.0 × 104 | 7.25 |
| 25 | 7.8 × 105 | 6.0 × 105 | 1.2 × 102 | 7.0 × 101 | 2.0 × 104 | 1.0 × 104 | 7.66 |
| 26 | 1.1 × 106 | 5.8 × 105 | 2.3 × 102 | 1.3 × 102 | 6.0 × 104 | 3.0 × 104 | 7.21 |
| 27 | 6.9 × 105 | 6.1 × 105 | 2.6 × 102 | 2.2 × 102 | 3.0 × 104 | 2.0 × 104 | 7.53 |
| 28 | 1.1 × 106 | 5.8 × 105 | 3.5 × 102 | 1.8 × 102 | 3.0 × 104 | 2.0 × 104 | 7.50 |
| 29 | 1.1 × 106 | 5.9 × 105 | 1.5 × 102 | 8.0 × 101 | 5.0 × 104 | 3.0 × 104 | 7.25 |
| 30 | 8.0 × 105 | 6.0 × 105 | 1.1 × 102 | 8.0 × 101 | 3.0 × 104 | 2.0 × 104 | 7.63 |
| 31 | 8.5 × 105 | 5.9 × 105 | 2.5 × 102 | 1.8 × 102 | 3.0 × 104 | 2.0 × 104 | 7.54 |
| 33 | 1.3 × 106 | 5.9 × 105 | 2.5 × 102 | 1.5 × 102 | 1.9 × 105 | 9.0 × 104 | 6.04 |
| 50 | 1.5 × 106 | 6.4 × 105 | 1.7 × 102 | 7.0 × 101 | 7.0 × 104 | 3.0 × 104 | 7.20 |
| 53 | 1.0 × 106 | 6.0 × 105 | 5.0 × 101 | 3.0 × 101 | 4.0 × 104 | 2.0 × 104 | * |
| 54 | 1.4 × 106 | 5.7 × 105 | 7.0 × 101 | 3.0 × 101 | 2.2 × 105 | 9.0 × 104 | * |
| Mean | 10 × 105 | 5.9 × 105 | 2.0 × 102 | 1.3 × 102 | 6.4 × 104 | 3.3 × 104 | 7 |
| SD | 2.5 × 105 | 1.6 × 104 | 1.3 × 102 | 9.3 × 101 | 5.8 × 104 | 2.3 × 104 | 0.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cañas-Arboleda, M.; Beltrán, K.; Medina, C.; Camacho, B.; Salguero, G. Human Platelet Lysate Supports Efficient Expansion and Stability of Wharton’s Jelly Mesenchymal Stromal Cells via Active Uptake and Release of Soluble Regenerative Factors. Int. J. Mol. Sci. 2020, 21, 6284. https://doi.org/10.3390/ijms21176284
Cañas-Arboleda M, Beltrán K, Medina C, Camacho B, Salguero G. Human Platelet Lysate Supports Efficient Expansion and Stability of Wharton’s Jelly Mesenchymal Stromal Cells via Active Uptake and Release of Soluble Regenerative Factors. International Journal of Molecular Sciences. 2020; 21(17):6284. https://doi.org/10.3390/ijms21176284
Chicago/Turabian StyleCañas-Arboleda, Mariana, Karl Beltrán, Carlos Medina, Bernardo Camacho, and Gustavo Salguero. 2020. "Human Platelet Lysate Supports Efficient Expansion and Stability of Wharton’s Jelly Mesenchymal Stromal Cells via Active Uptake and Release of Soluble Regenerative Factors" International Journal of Molecular Sciences 21, no. 17: 6284. https://doi.org/10.3390/ijms21176284
APA StyleCañas-Arboleda, M., Beltrán, K., Medina, C., Camacho, B., & Salguero, G. (2020). Human Platelet Lysate Supports Efficient Expansion and Stability of Wharton’s Jelly Mesenchymal Stromal Cells via Active Uptake and Release of Soluble Regenerative Factors. International Journal of Molecular Sciences, 21(17), 6284. https://doi.org/10.3390/ijms21176284





