The Chemokine CXCL16 Is a New Biomarker for Lymph Node Analysis of Colon Cancer Outcome
Abstract
:1. Introduction
2. Results
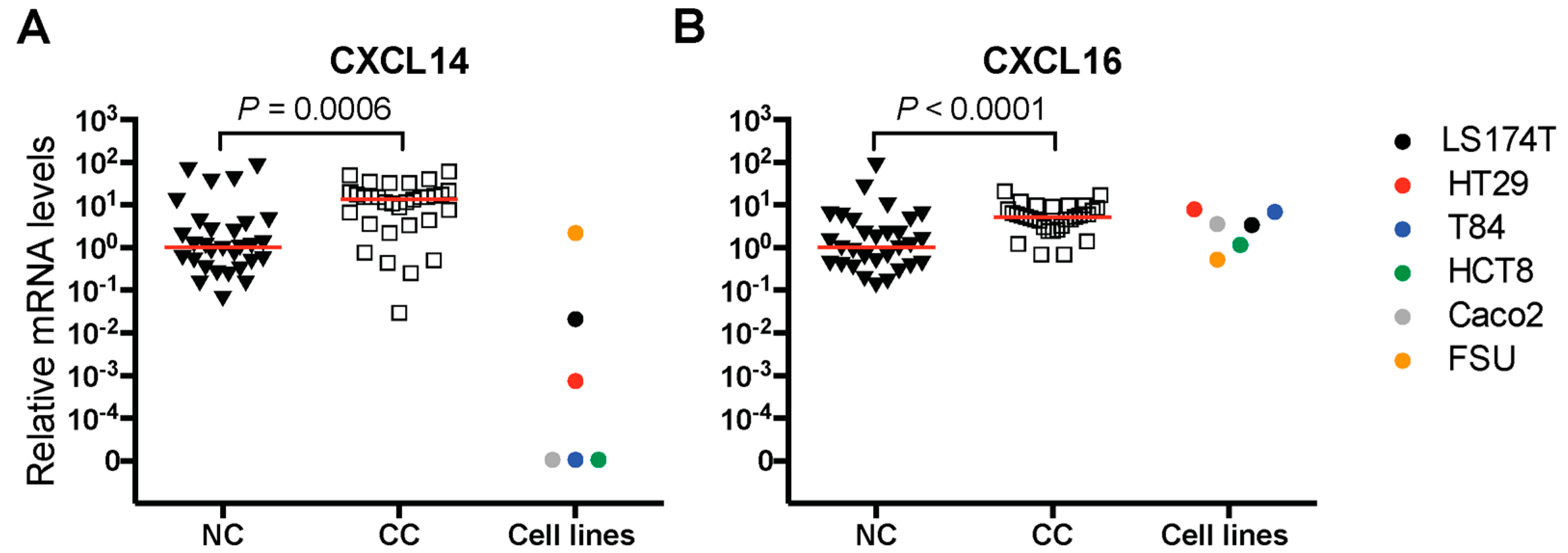
2.1. mRNA Levels of Chemokines CXCL14 and CXCL16 in Primary Colon Tumors and Colon Carcinoma Cell Lines
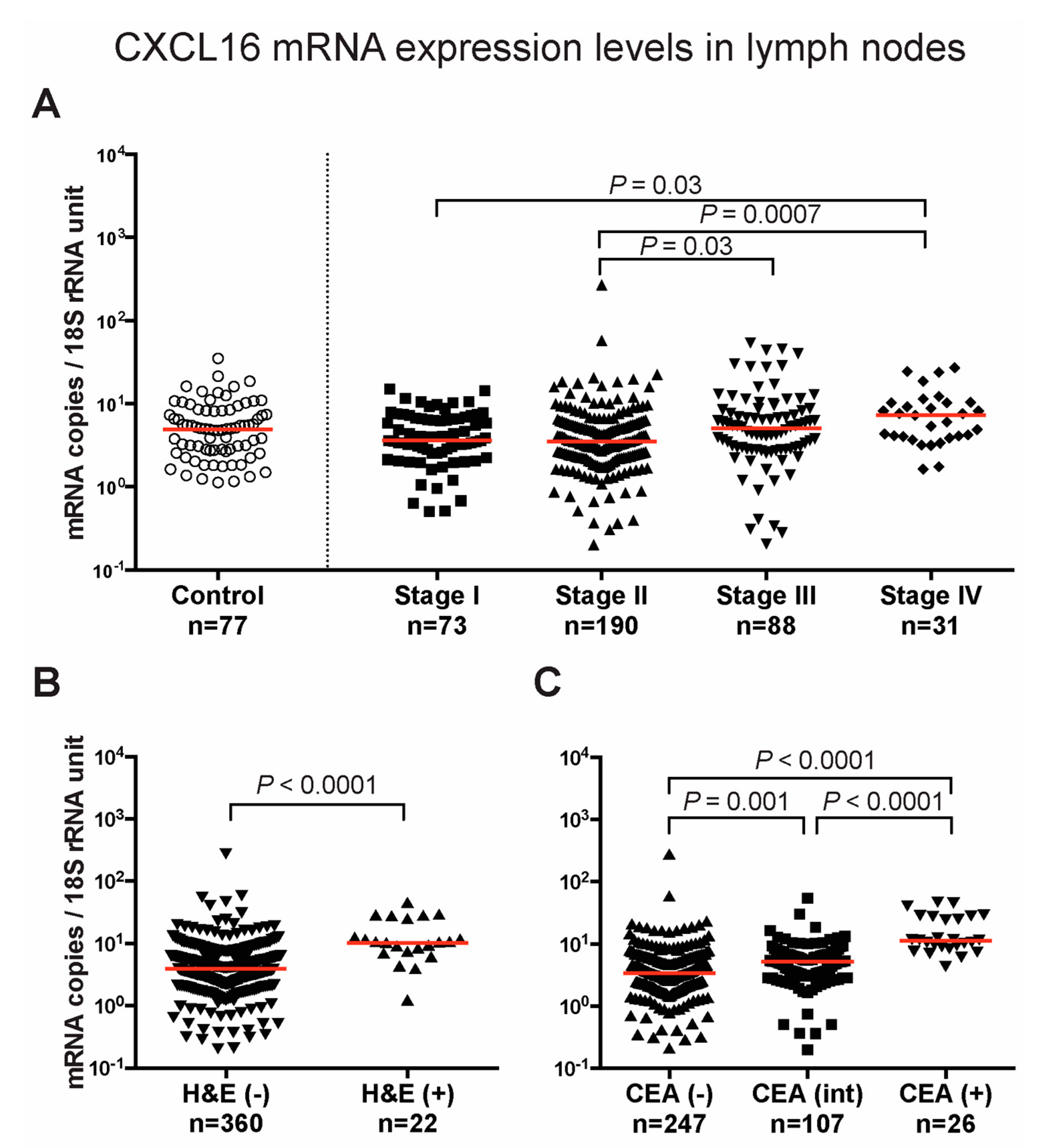
2.2. mRNA Levels of Chemokines CXCL14 and CXCL16 in Regional Lymph Nodes of CC Patients
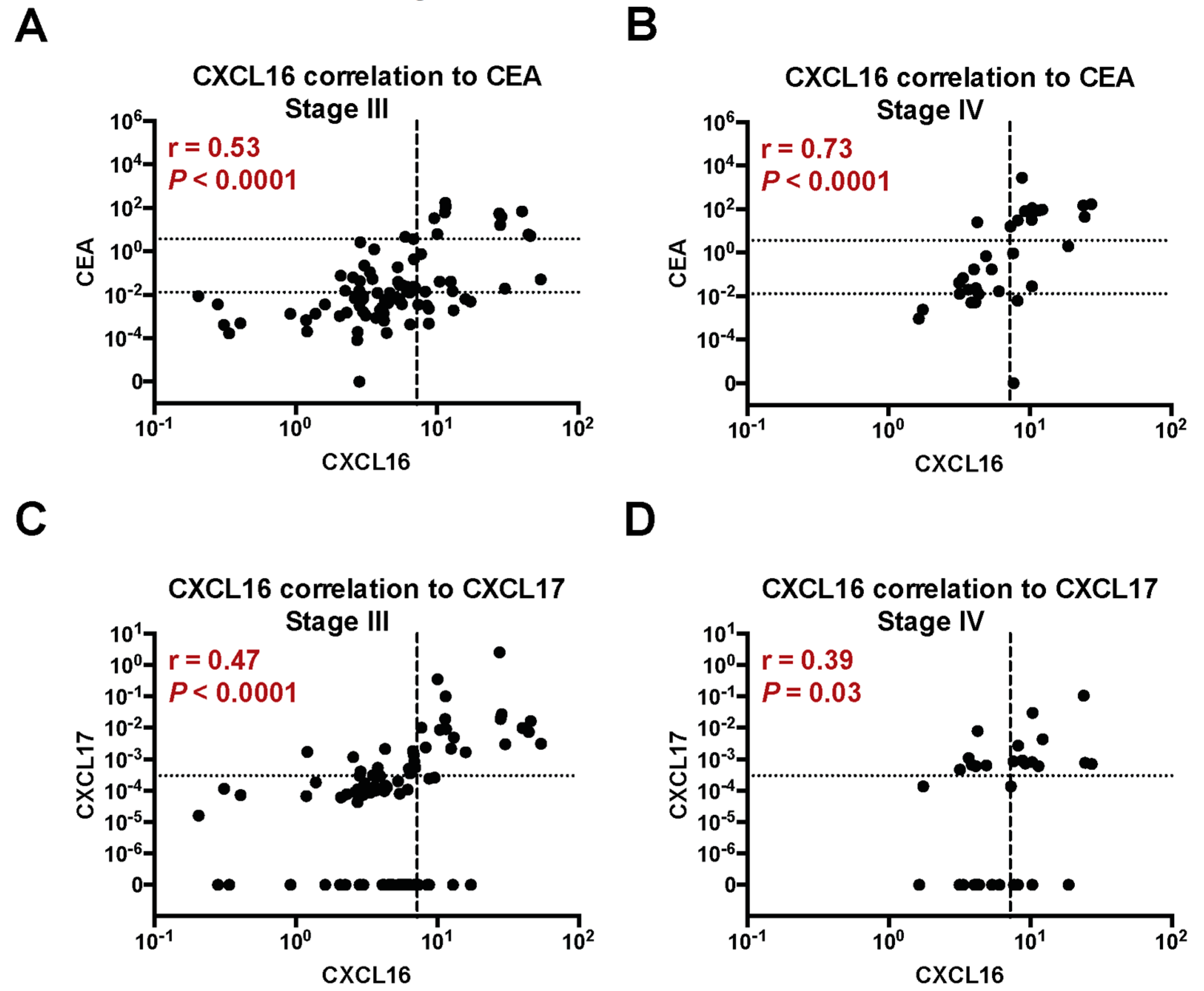
2.3. Correlation between Expression Levels of CXCL16 mRNA, CEA mRNA, and CXCL17 mRNA in Lymph Nodes of CC Patients
2.4. CXCL16 mRNA Expression in Regional Lymph Nodes can Predict Disease Recurrence
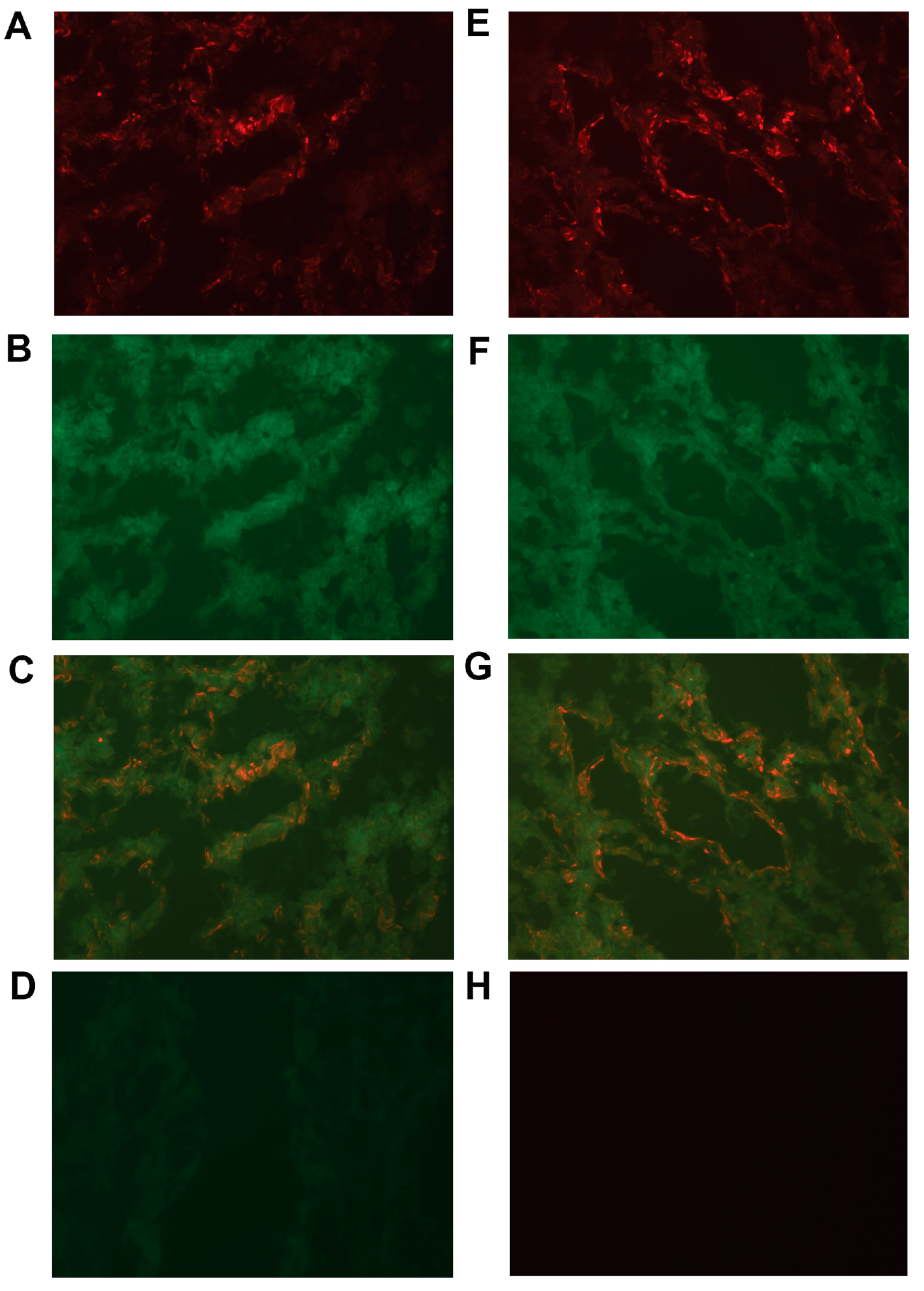
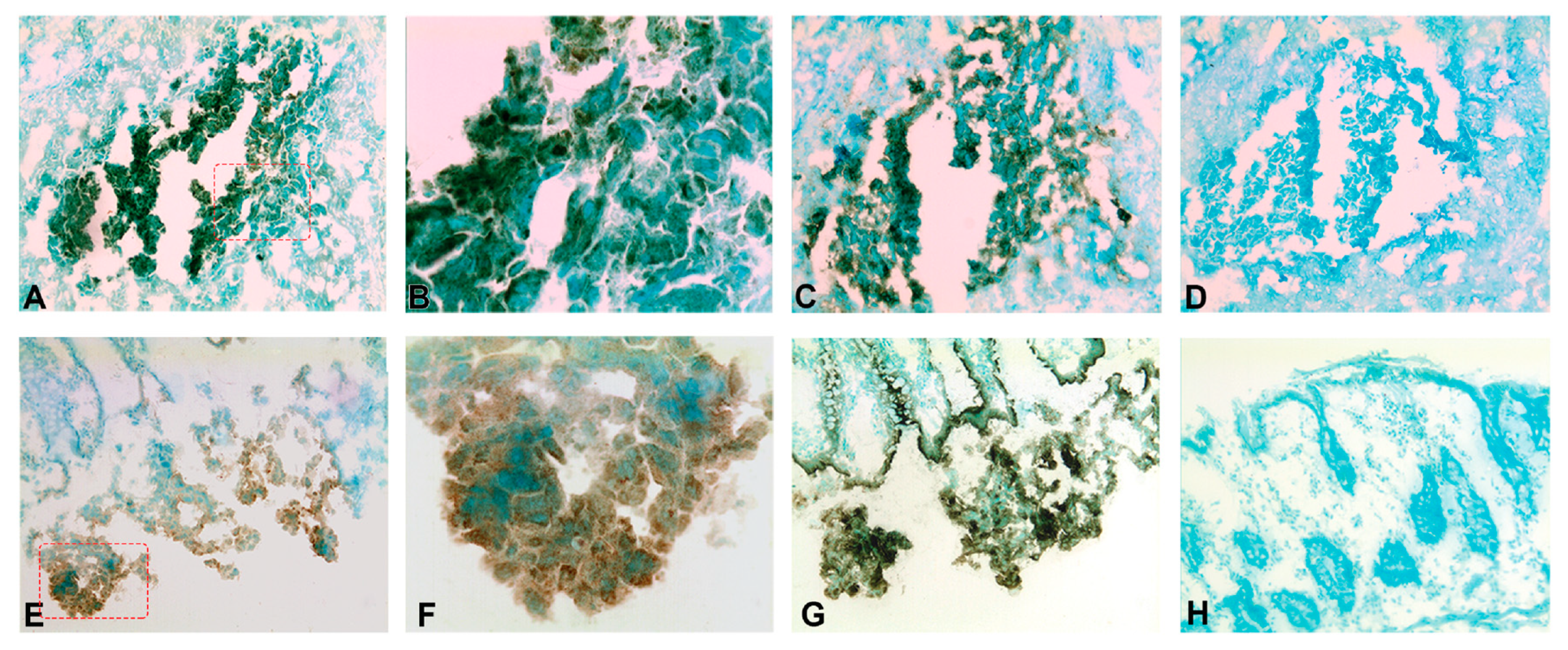
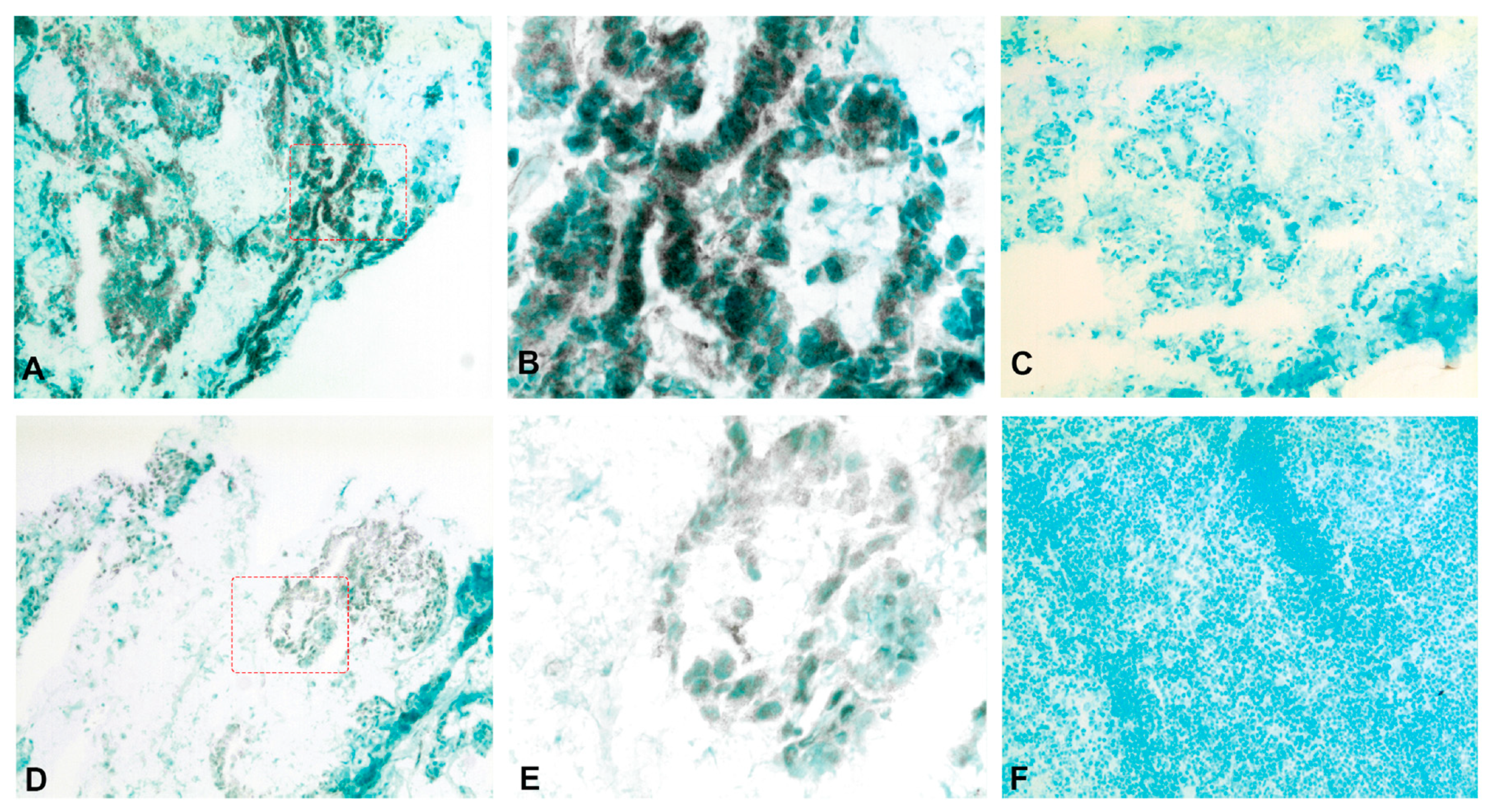
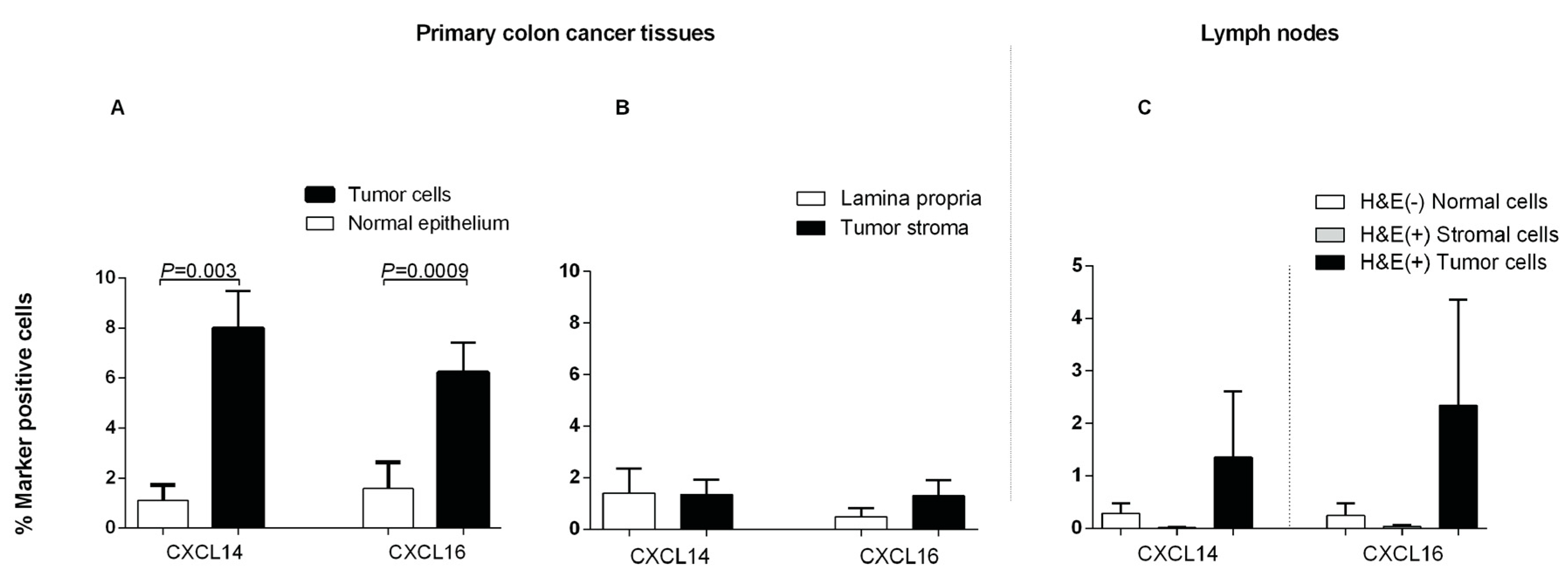
2.5. Expression of CXCL14 and CXCL16 Proteins in CC Tumors and Normal Colon Tissue and Lymph Nodes as Determined by IHC and IF
3. Discussion
4. Materials and Methods
4.1. Patients and Tissue Specimens for mRNA Analysis
4.2. Patients and Tissue Specimens for Immunohistochemistry
4.3. Cell Lines
4.4. Real-Time qRT-PCR
4.5. Antibodies and Substrate
4.6. Immunohistochemistry
4.7. Two-Color Immunofluorescence
4.8. Immunomorphometry
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
Ethical Considerations
References
- Vassilatis, D.K.; Hohmann, J.G.; Zeng, H.; Li, F.; Ranchalis, J.E.; Mortrud, M.T.; Brown, A.; Rodriguez, S.S.; Weller, J.R.; Wright, A.C.; et al. The G protein-coupled receptor repertoires of human and mouse. Proc. Natl. Acad. Sci. USA 2003, 100, 4903–4908. [Google Scholar] [CrossRef] [PubMed]
- Chow, M.T.; Luster, A.D. Chemokines in cancer. Cancer Immunol. Res. 2014, 2, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, L.; Hammarström, M.-L.; Lindmark, G.; Hammarström, S.; Sitohy, B. Ectopic expression of the chemokine CXCL17 in colon cancer cells. Br. J. Cancer 2016, 114, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Rashad, Y.; Olsson, L.; Israelsson, A.; Öberg, Å.; Lindmark, G.; Hammarström, M.-L.; Hammarström, S.; Sitohy, B. Lymph node CXCL17 messenger RNA: A new prognostic biomarker for colon cancer. Tumor Biol. 2018, 40, 1010428318799251. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Zou, R.; Lin, F.; Zheng, S.; Shen, X.; Xue, X. Expression and effect of CXCL14 in colorectal carcinoma. Mol. Med. Rep. 2014, 10, 1561–1568. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Yang, X.; Cheng, L.; Liu, R.; Lei, Y.; Dong, D.; Li, F.; Lau, Q.C.; Deng, L.; Nice, E.C.; et al. Chemokine CXCL14 is associated with prognosis in patients with colorectal carcinoma after curative resection. J. Transl. Med. 2013, 11, 6. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Yang, Y.; Pan, Y.; Jia, Y.; Brock, M.V.; Herman, J.G.; Guo, M. Epigenetic silencing of CXCL14 induced colorectal cancer migration and invasion. Discov. Med. 2013, 16, 137–147. [Google Scholar] [PubMed]
- Matloubian, M.; David, A.; Engel, S.; Ryan, J.E.; Cyster, J.G. A transmembrane CXC chemokine is a ligand for HIV-coreceptor Bonzo. Nat. Immunol. 2000, 1, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Gough, P.J.; Garton, K.J.; Wille, P.T.; Rychlewski, M.; Dempsey, P.J.; Raines, E.W. A disintegrin and metalloproteinase 10-mediated cleavage and shedding regulates the cell surface expression of CXC chemokine ligand 16. J. Immunol. 2004, 172, 3678–3685. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Kunkel, E.J.; Boisvert, J.; Johnston, B.; Campbell, J.J.; Genovese, M.C.; Greenberg, H.B.; Butcher, E.C. Bonzo/CXCR6 expression defines type 1–polarized T-cell subsets with extralymphoid tissue homing potential. J. Clin. Investig. 2001, 107, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Johnston, B.; Butcher, E.C.; Lipp, M. Trafficking machinery of NKT cells: Shared and differential chemokine receptor expression among Vα24(+)Vβ 11(+) NKT cell subsets with distinct cytokine-producing capacity. Blood 2002, 100, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Hojo, S.; Koizumi, K.; Tsuneyama, K.; Arita, Y.; Cui, Z.; Shinohara, K.; Minami, T.; Hashimoto, I.; Nakayama, T.; Sakurai, H.; et al. High-Level Expression of Chemokine CXCL16 by Tumor Cells Correlates with a Good Prognosis and Increased Tumor-Infiltrating Lymphocytes in Colorectal Cancer. Cancer Res. 2007, 67, 4725–4731. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, K.; Toiyama, Y.; Tanaka, K.; Saigusa, S.; Hiro, J.; Uchida, K.; Inoue, Y.; Kusunoki, M. Soluble CXCL16 in Preoperative Serum is a Novel Prognostic Marker and Predicts Recurrence of Liver Metastases in Colorectal Cancer Patients. Ann. Surg. Oncol. 2012, 19, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Fahlgren, A.; Baranov, V.; Frangsmyr, L.; Zoubir, F.; Hammarström, M.-L.; Hammarström, S. Interferon-gamma Tempers the Expression of Carcinoembryonic Antigen Family Molecules in Human Colon Cells: A Possible Role in Innate Mucosal Defence. Scand. J. Immunol. 2003, 58, 628–641. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, D.; Zhang, C.; Zhang, Z.; Chen, X.; Lian, J.; Liu, J.; Wang, G.; Yuan, W.; Sun, Z.; et al. Identification of liver metastasis-associated genes in human colon carcinoma by mRNA profiling. Chin. J. Cancer Res. 2018, 30, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Bas, A.; Forsberg, G.; Hammarström, S.; Hammarström, M.-L. Utility of the housekeeping genes 18S rRNA, β-actin and glyceraldehyde-3-phosphate-dehydrogenase for normalization in real-time quantitative reverse transcriptase-polymerase chain reaction analysis of gene expression in human T lymphocytes. Scand. J. Immunol. 2004, 59, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Sitohy, B.; Hammarström, S.; Danielsson, Å.; Hammarström, M.-L. Basal lymphoid aggregates in ulcerative colitis colon: A site for regulatory T cell action. Clin. Exp. Immunol. 2008, 151, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Weibel, E. Point counting methods. In Stereological Methods: Practical Methods for Biological Morphometery; Academic Press: London, UK, 1979. [Google Scholar]
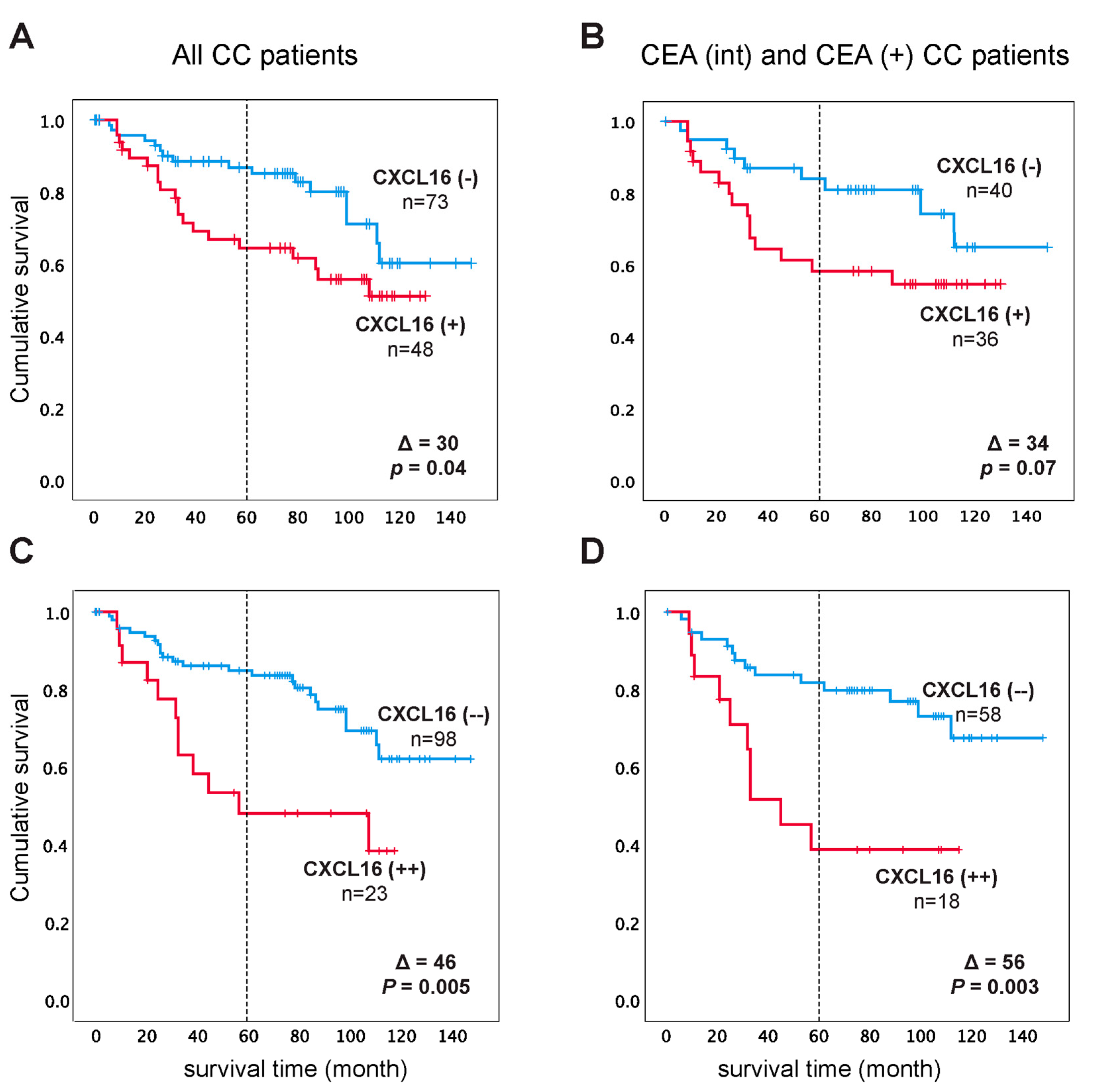
| Patient Group | Category | 5 Year Follow-Up after Surgery | 12 Year Follow-Up after Surgery | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Disease-Free Survival | Risk for Recurrence | Disease-Free Survival | Risk for Recurrence | ||||||||
| Average a | Difference | P-Value | Hazard Ratio | P-Value | Average a | Difference | P-Value | Hazard Ratio | P-Value | ||
| (Months) | (Months) | (95% CI) b | (Months) | (Months) | (95% CI) b | ||||||
| All CC patients | CXCL16(−) c | 55 | 118 | ||||||||
| CXCL16(+) | 47 | 8 | 0.01 | 2.4 | 0.013 | 88 | 30 | 0.041 | 2.0 | 0.045 | |
| (1.2–4.8) | (1.0–3.8) | ||||||||||
| CXCL16(−−) d | 53 | 117 | |||||||||
| CXCL16(++) | 45 | 8 | 0.01 | 2.5 | 0.013 | 71 | 46 | 0.005 | 2.6 | 0.007 | |
| (1.2–5.2) | (1.3–5.2) | ||||||||||
| CEA(int) plus CEA(+) e CC patients | CXCL16(−) | 54 | 119 | ||||||||
| CXCL16(+) | 46 | 8 | 0.06 | 2.2 | 0.067 | 85 | 34 | 0.075 | 2.1 | 0.082 | |
| (0.9–5.0) | (0.9–4.8) | ||||||||||
| CXCL16(−−) | 53 | 117 | |||||||||
| CXCL16(++) | 42 | 11 | 0.008 | 2.9 | 0.011 | 61 | 56 | 0.003 | 3.3 | 0.005 | |
| (1.3–6.5) | (1.4–7.4) | ||||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
AbdelMageed, M.; Ali, H.; Olsson, L.; Lindmark, G.; Hammarström, M.-L.; Hammarström, S.; Sitohy, B. The Chemokine CXCL16 Is a New Biomarker for Lymph Node Analysis of Colon Cancer Outcome. Int. J. Mol. Sci. 2019, 20, 5793. https://doi.org/10.3390/ijms20225793
AbdelMageed M, Ali H, Olsson L, Lindmark G, Hammarström M-L, Hammarström S, Sitohy B. The Chemokine CXCL16 Is a New Biomarker for Lymph Node Analysis of Colon Cancer Outcome. International Journal of Molecular Sciences. 2019; 20(22):5793. https://doi.org/10.3390/ijms20225793
Chicago/Turabian StyleAbdelMageed, Manar, Haytham Ali, Lina Olsson, Gudrun Lindmark, Marie-Louise Hammarström, Sten Hammarström, and Basel Sitohy. 2019. "The Chemokine CXCL16 Is a New Biomarker for Lymph Node Analysis of Colon Cancer Outcome" International Journal of Molecular Sciences 20, no. 22: 5793. https://doi.org/10.3390/ijms20225793
APA StyleAbdelMageed, M., Ali, H., Olsson, L., Lindmark, G., Hammarström, M.-L., Hammarström, S., & Sitohy, B. (2019). The Chemokine CXCL16 Is a New Biomarker for Lymph Node Analysis of Colon Cancer Outcome. International Journal of Molecular Sciences, 20(22), 5793. https://doi.org/10.3390/ijms20225793





