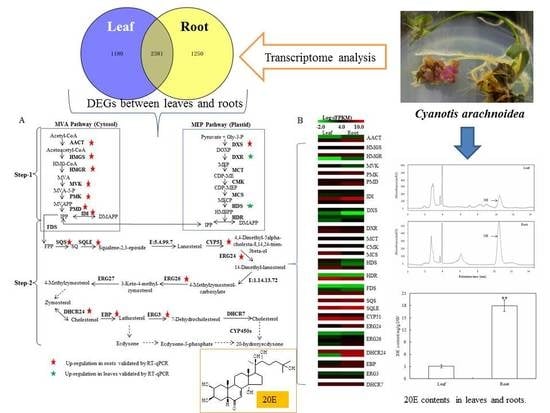
Comparative Transcriptome Analysis Identifies Genes Putatively Involved in 20-Hydroxyecdysone Biosynthesis in Cyanotis arachnoidea
Abstract
1. Introduction
2. Results and Discussion
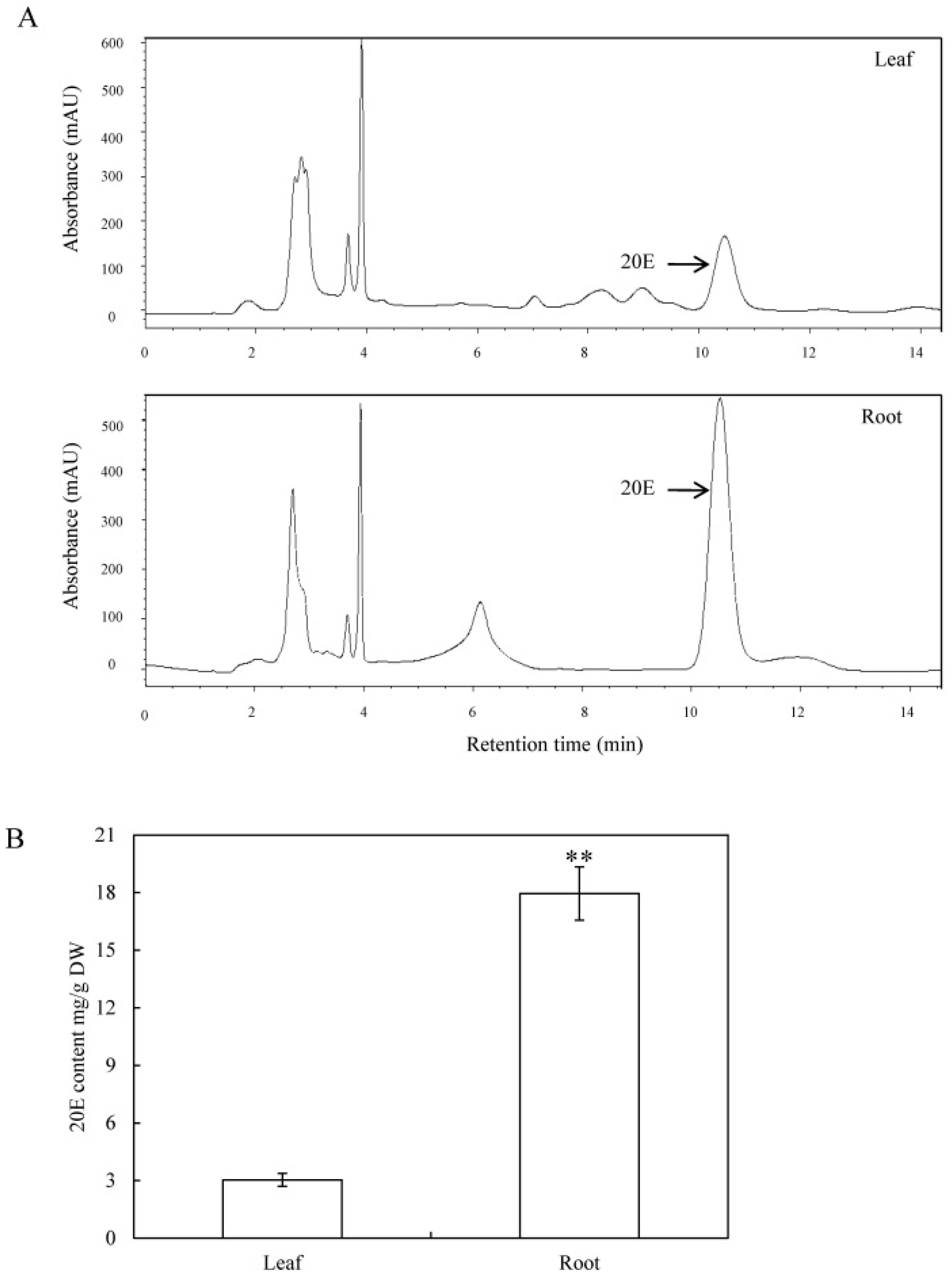
2.1. Quantification of 20E from C. arachnoidea
2.2. Library Sequencing and De Novo Assembly and Annotation
2.3. Different Expression Genes (DEGs) Identified Between Leaves and Roots
2.4. Identification of Unigenes Involved in Secondary Metabolism
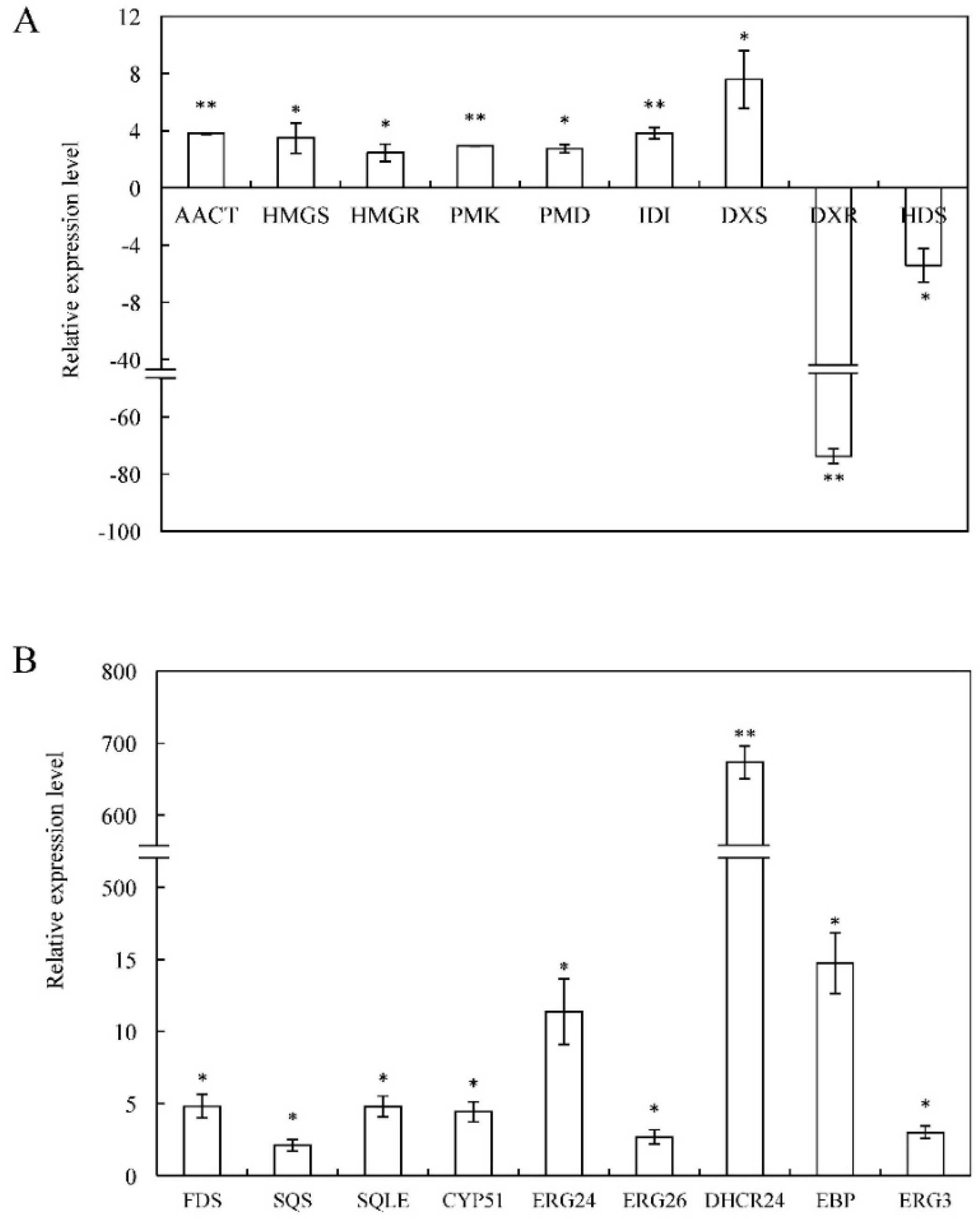
2.5. Identification of Unigenes Related to 20E Biosynthesis
2.6. Validation of Differentially Expressed CYP450s
2.7. Transcription Factors Predicted and Statistics of Simple Sequence Repeats
3. Materials and Methods
3.1. Plant Materials
3.2. 20E Extraction and Analysis
3.3. cDNA Library Construction, Sequencing and Quality Control
3.4. De Novo Assembly and Sequence Annotation
3.5. Identification of DEGs
3.6. Real-Time Quantitative PCR Analysis
3.7. Identification of Simple Sequence Repeats
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tan, C.Y.; Wang, J.H.; Xiao, W.; Li, X. A new phytosterone from Cyanotis arachnoidea. J. Asian Nat. Prod. Res. 2002, 4, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.Y.; Wang, J.H.; Li, X. Phytoecdysteroid constituents from Cyanotis arachnoidea. J. Asian Nat. Prod. Res. 2003, 5, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Trivedy, K.; Nair, K.S.; Ramesh, M.; Gopal, N.; Kumar, S.N. Effect of phytoecdysteroid on maturation of silkworm, Bombyx mori L. Indian J. Seric. 2003, 42, 75–77. [Google Scholar]
- Mellon, D.F.; Greer, E. Induction of precocious molting and claw transformation in Alpheid shrimps by exogenous 20-hydroxyecdysone. Biol. Bull. 1987, 172, 350–356. [Google Scholar] [CrossRef]
- Dinan, L.; Lafont, R. Effects and applications of arthropod steroid hormones (ecdysteroids) in mammals. J. Endocrinol. 2006, 191, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Thiem, B.; Kikowska, M.; Maliński, M.P.; Kruszka, D.; Napierała, M.; Florek, E. Ecdysteroids: Production in plant in vitro cultures. Phytochem. Rev. 2017, 16, 603–622. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Yang, S.C.; Guan, D.J.; Yang, T.; Wen, G.S.; Zhang, W.M. A study on accumulation of β-ecdysone and optimal harvest time for Cyanotis arachnoidea C. B. Clarke. J. Yunnan Agric. Univ. 2011, 26, 194–198. (In Chinese) [Google Scholar]
- Tarkowská, D.; Strnad, M. Plant ecdysteroids: Plant sterols with intriguing distributions, biological effects and relations to plant hormones. Planta 2016, 244, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Rohmer, M. The discovery of a mevalonate-independent pathway for isoprenoid biosynthesis in bacteria, algae and higher plants. Nat. Prod. Rep. 1999, 16, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Devarenne, T.P.; Sen-Michael, B.; Adler, J.H. Biosynthesis of ecdysteroids in Zea mays. Phytochemistry 1995, 40, 1125–1131. [Google Scholar] [CrossRef]
- Nakagawa, T.; Hara, N.; Fujimoto, Y. Biosynthesis of 20-hydroxyecdysone in Ajuga hairy roots: Stereochemistry of C-25 hydroxylation. Tetrahedron Lett. 1997, 38, 2701–2704. [Google Scholar] [CrossRef]
- Adler, J.H.; Grebenok, R.J. Biosynthesis and distribution of insect-molting hormones in plants—A review. Lipids 1995, 30, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Tsukagoshi, Y.; Ohyama, K.; Seki, H.; Akashi, T.; Muranaka, T.; Suzuki, H.; Fujimoto, Y. Functional characterization of CYP71D443, a cytochrome P450 catalyzing C-22 hydroxylation in the 20-hydroxyecdysone biosynthesis of Ajuga hairy roots. Phytochemistry 2016, 127, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Q.; Fang, X.; Wu, X.M.; Mao, Y.B.; Wang, L.J.; Chen, X.Y. Transcriptional regulation of plant secondary metabolism. J. Integr. Plant Biol. 2012, 54, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Tomás, J.; Camps, F.; Claveria, E.; Coll, J.; Melé, E.; Messeguer, J. Composition and location of phytoecdysteroids in Ajuga reptans in vivo and in vitro cultures. Phytochemistry 1992, 31, 1585–1591. [Google Scholar] [CrossRef]
- Tomás, J.; Camps, F.; Coll, J.; Melé, E.; Messeguer, J. Phytoecdysteroid production by Ajuga reptans tissue cultures. Phytochemistry 1993, 32, 317–324. [Google Scholar] [CrossRef]
- Yang, L.; Ding, G.H.; Lin, H.Y.; Cheng, H.N.; Kong, Y.; Wei, Y.K.; Fang, X.; Liu, R.Y.; Wang, L.; Chen, X.Y.; et al. Transcriptome analysis of medicinal plant Salvia miltiorrhiza and identification of genes related to tanshinone biosynthesis. PLoS ONE 2013, 8, e80464. [Google Scholar] [CrossRef]
- Gupta, P.; Goel, R.; Pathak, S.; Srivastava, A.; Singh, S.P.; Sangwan, R.S.; Asif, M.H.; Trivedi, P.K. De novo assembly, functional annotation and comparative analysis of Withania somnifera leaf and root transcriptomes to identify putative genes involved in the withanolides biosynthesis. PLoS ONE 2013, 8, e62714. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.J.; Zheng, L.P.; Sima, Y.H.; Yuan, H.Y.; Wang, J.W. Methyl jasmonate stimulates 20-hydroxyecdysone production in cell suspension cultures of Achyranthes bidentata. Plant Omics 2013, 6, 116–120. [Google Scholar]
- Wang, Q.J.; Zheng, L.P.; Zhao, P.F.; Zhao, Y.L.; Wang, J.W. Cloning and characterization of an elicitor-responsive gene encoding 3-hydroxy-3-methylglutaryl coenzyme A reductase involved in 20-hydroxyecdysone production in cell cultures of Cyanotis arachnoidea. Plant Physiol. Biochem. 2014, 84, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.D.; Zhang, Y.X.; Ye, J.Y.; Wu, C.L.; Yu, L.; Yang, X. Effect of different size of ground Cyanotis arachnoidea particles on the release of β-ecdysone. Agric. Sci. Technol. Hunan 2011, 12, 1318–1326. [Google Scholar]
- Niwa, R.; Niwa, R.S. Enzymes for ecdysteroid biosynthesis: Their biological functions in insects and beyond. Biosci. Biotechnol. Biochem. 2014, 78, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Rewitz, K.F.; O’Connor, M.B.; Gilbert, L.I. Molecular evolution of the insect Halloween family of cytochrome P450s: Phylogeny, gene organization and functional conservation. Insect Biochem. Mol. Biol. 2007, 37, 741–753. [Google Scholar] [CrossRef] [PubMed]
- Thagun, C.; Imanishi, S.; Kudo, T.; Nakabayashi, R.; Ohyama, K.; Mori, T.; Kawamoto, K.; Nakamura, Y.; Katayama, M.; Nonaka, S.; et al. Jasmonate-responsive ERF transcription factors regulate steroidal glycoalkaloid biosynthesis in tomato. Plant Cell Physiol. 2016, 57, 961–975. [Google Scholar] [CrossRef] [PubMed]
- Kalia, R.K.; Rai, M.K.; Kalia, S.; Singh, R.; Dhawan, A.K. Microsatellite markers: An overview of the recent progress in plants. Euphytica 2011, 177, 309–334. [Google Scholar] [CrossRef]
- Sharopova, N. Plant simple sequence repeats: Distribution, variation, and effects on gene expression. Genome 2008, 51, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Bao, S.; Corke, H.; Sun, M. Microsatellites in starch-synthesizing genes in relation to starch physicochemical properties in waxy rice (Oryza sativa L.). Theor. Appl. Genet. 2002, 105, 898–905. [Google Scholar] [PubMed]

| The Sequencing Data | Leaf | Root |
|---|---|---|
| Clean bases | 5,671,005,500 | 5,136,914,500 |
| Clean reads | 45,368,044 | 41,095,316 |
| Q30 (%) | 90.63 | 89.90 |
| GC content (%) | 43.50 | 44.50 |
| Unigenes (≥ 300 bp) | 69,782 | 70,556 |
| All unigenes (≥ 300 bp) | 79,835 | |
| N50 (bp) | 1268 | |
| Average length (bp) | 894 | |
| Secondary Metabolites Biosynthesis Pathway | Total | Up-Regulated Unigenes | Down-Regulated Unigenes |
|---|---|---|---|
| Phenylpropanoid biosynthesis (ko00940) | 289 | 62 | 17 |
| Stilbenoid, diarylheptanoid, and gingerol biosynthesis (ko00945) | 85 | 9 | 5 |
| Terpenoid backbone biosynthesis (ko00900) | 79 | 1 | 1 |
| Flavonoid biosynthesis (ko00941) | 79 | 2 | 12 |
| Limonene and pinene degradation (ko00903) | 68 | 4 | 8 |
| Ubiquinone and other terpenoid-quinone biosynthesis (ko00130) | 67 | 4 | 12 |
| Isoquinoline alkaloid biosynthesis (ko00950) | 63 | 8 | 2 |
| Steroid biosynthesis (ko00100) | 59 | 4 | 2 |
| Carotenoid biosynthesis (ko00906) | 43 | 1 | 6 |
| Tropane, piperidine and pyridine alkaloid biosynthesis (ko00960) | 39 | 1 | 2 |
| Brassinosteroid biosynthesis (ko00905) | 37 | 7 | 0 |
| Diterpenoid biosynthesis (ko00904) | 35 | 3 | 2 |
| Zeatin biosynthesis (ko00908) | 34 | 3 | 1 |
| Flavone and flavonol biosynthesis (ko00944) | 24 | 0 | 1 |
| Isoflavonoid biosynthesis (ko00943) | 16 | 0 | 1 |
| Monoterpenoid biosynthesis (ko00902) | 14 | 1 | 4 |
| Sesquiterpenoid and triterpenoid biosynthesis (ko00909) | 12 | 3 | 1 |
| Anthocyanin biosynthesis (ko00942) | 8 | 0 | 0 |
| Betalain biosynthesis (ko00965) | 6 | 0 | 0 |
| Geraniol degradation (ko00281) | 5 | 0 | 0 |
| Glucosinolate biosynthesis (ko00966) | 4 | 1 | 0 |
| Caffeine metabolism (ko00232) | 2 | 0 | 0 |
| Polyketide sugar unit biosynthesis (ko00523) | 1 | 0 | 0 |
| Indole alkaloid biosynthesis (ko00901) | 1 | 1 | 0 |
| Total | 1070 | 115 | 77 |
| Enzymes | Enzyme Commission Number | Unigenes ID | FPKM Value | |
|---|---|---|---|---|
| Leaf | Root | |||
| Step 1: MVA pathway | ||||
| acetyl CoA acetyltransferase (AACT) | 2.3.1.9 | comp60224_c0_seq7 | 1.35 | 4.97 |
| comp70319_c0_seq8 | 36.31 | 114.60 | ||
| 3-hydroxy-3-methyl-glutaryl coenzyme A synthase (HMGS) | 2.3.3.10 | comp69323_c0_seq6 | 15.40 | 26.53 |
| 3-hydroxy-3-methyl-glutaryl coenzyme A reductase (HMGR) | 1.1.1.34 | comp68996_c0_seq2 | 59.92 | 55.43 |
| comp52491_c0_seq3 | 0.29 | 2.98 | ||
| comp31434_c0_seq1 | 0 | 1.82 | ||
| mevalonate kinase (MVK) | 2.7.1.36 | comp69732_c0_seq13 | 10.58 | 5.18 |
| comp120320_c0_seq1 | 0 | 1.10 | ||
| phosphomevalonate kinase (PMK) | 2.7.4.2 | comp65552_c0_seq7 | 35.22 | 69.05 |
| diphosphomevalonate decarboxylase (PMD) | 4.1.1.33 | comp33484_c0_seq1 | 29.43 | 28.81 |
| comp59477_c0_seq1 | 30.56 | 72.50 | ||
| MEP pathway | ||||
| 1-deoxy-d-xylulose-5-phosphate synthase (DXS) | 2.2.1.7 | comp66526_c0_seq5 | 17.96 | 9.56 |
| comp24131_c0_seq1 | 2.04 | 0.25 | ||
| comp71989_c1_seq3 | 6.86 | 6.63 | ||
| comp59095_c0_seq1 | 3.92 | 12.75 | ||
| 1-deoxy-d-xylulose-5-phosphate reductoisomerase (DXR) | 1.1.1.267 | comp47417_c0_seq1 | 41.16 | 0 |
| comp71987_c0_seq5 | 31.46 | 22.10 | ||
| comp74054_c0_seq1 | 59.57 | 35.26 | ||
| 2-C-methyl-d-erythritol4-phosphate cytidylyi transferase (MCT) | 2.7.7.60 | comp71928_c2_seq77 | 21.02 | 15.51 |
| 4-diphosphocytidyl-2-C-methyl-d-erythritol kinase (CMK) | 2.7.1.148 | comp74100_c0_seq1 | 24.09 | 10.73 |
| 2-C-methyl-d-erythritol-2,4-cyclodiphosphate synthase (MCS) | 4.6.1.12 | comp30840_c0_seq1 | 88.79 | 22.58 |
| hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate synthase (HDS) | 1.17.7.1 | comp33211_c0_seq1 | 30.23 | 4.26 |
| comp53197_c0_seq1 | 7.95 | 2.19 | ||
| comp67980_c0_seq1 | 91.56 | 39.75 | ||
| hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate reductase (HDR) | 1.17.1.2 | comp90900_c0_seq1 | 0.82 | 0.69 |
| comp73401_c0_seq1 | 331.58 | 174.81 | ||
| isopentenyl diphosphate isomerase (IDI) | 5.3.3.2 | comp64868_c0_seq4 | 24.51 | 13.51 |
| comp61897_c0_seq2 | 8.51 | 10.42 | ||
| comp29765_c0_seq1 | 106.24 | 337.84 | ||
| comp74567_c0_seq1 | 19.86 | 25.58 | ||
| Step 2 | ||||
| farnesyl diphosphate synthase (FDS) | 2.5.1.10 | comp96291_c0_seq1 | 0.47 | 0.57 |
| comp60714_c0_seq9 | 6.56 | 3.03 | ||
| comp28732_c0_seq1 | 71.68 | 87.98 | ||
| squalene synthase (SQS) | 2.5.1.21 | comp68019_c0_seq1 | 105.92 | 144.18 |
| squalene monooxygenase (SQLE) | 1.14.13.132 | comp35298_c0_seq1 | 201.88 | 387.62 |
| sterol 14-demethylase (CYP51) | 1.14.13.72 | comp67692_c0_seq1 | 33.34 | 24.69 |
| comp67692_c1_seq3 | 335.08 | 250.89 | ||
| δ14-sterol reductase (ERG24) | 1.3.1.70 | comp52043_c0_seq5 | 21.53 | 69.73 |
| sterol-4α-carboxylate 3-dehydrogenase (ERG26) | 1.1.1.170 | comp40656_c0_seq1 | 1.25 | 1.91 |
| comp63129_c0_seq5 | 8.84 | 18.50 | ||
| comp48599_c0_seq1 | 22.32 | 23.31 | ||
| comp69306_c0_seq26 | 7.47 | 29.19 | ||
| δ24-sterol reductase (DHCR24) | 1.3.1.72 | comp29143_c0_seq1 | 136.31 | 215.60 |
| comp48248_c0_seq1 | 1.45 | 272.54 | ||
| cholestenol δ-isomerase (EBP) | 5.3.3.5 | comp47084_c0_seq3 | 10.87 | 59.19 |
| comp60181_c0_seq1 | 40.63 | 54.68 | ||
| δ7-sterol 5-desaturase (ERG3) | 1.14.19.20 | comp50441_c0_seq1 | 5.89 | 5.25 |
| comp61289_c0_seq4 | 28.21 | 24.24 | ||
| 7-dehydrocholesterol reductase (DHCR7) | 1.3.1.21 | comp71639_c2_seq1 | 55.04 | 56.70 |
| Unigene ID | Description | Foldchange | RT-qPCR |
|---|---|---|---|
| comp36620_c0_seq1 | cytochrome P450 94C1-like | Inf | (1.67 ± 0.13) × 103 ** |
| comp56632_c0_seq1 | cytochrome P450 90B1-like | Inf | 38.29 ± 10.74 * |
| comp60467_c1_seq1 | cytochrome P450 710A1-like | Inf | 49.69 ± 0.24 ** |
| comp70580_c0_seq1 | cytochrome P450 704C1 | Inf | 32.05 ± 7.01 * |
| comp28108_c0_seq1 | cytochrome P450 CYP736A12 | 1013.73 | (1.26 ± 0.27) × 102 * |
| comp30002_c0_seq1 | cytochrome P450 90A1 isoform X2 | 388.30 | (1.83 ± 0.63) × 102 |
| comp67837_c0_seq4 | cytochrome P450 86B1-like isoform X2 | 147.93 | (5.98 ± 0.71) × 102 ** |
| comp57563_c0_seq2 | cytochrome P450 71A1-like | 124.00 | 19.39 ± 1.42 ** |
| comp61518_c0_seq1 | cytochrome P450 90A1-like | 115.05 | (4.67 ± 0.34) × 102 ** |
| comp71681_c0_seq1 | cytochrome P450 71A1-like | 101.70 | (4.15 ± 0.10) × 102 ** |
| comp69723_c0_seq2 | cytochrome P450 86B1-like | 82.30 | 43.66 ± 8.29 * |
| comp60312_c0_seq1 | cytochrome P450 734A1-like | 56.02 | (1.12 ± 0.07) × 102 ** |
| comp60108_c0_seq3 | cytochrome P450 90B1-like | 47.42 | 73.56 ± 3.60 ** |
| comp60517_c0_seq1 | cytochrome P450 734A6-like | 39.25 | 2.10 ± 0.19 ** |
| comp66356_c0_seq1 | cytochrome P450 71A1-like | 33.79 | 18.72 ± 2.78 ** |
| comp70580_c1_seq4 | cytochrome P450 704C1-like | 22.36 | 24.68 ± 3.93 ** |
| comp60316_c0_seq2 | cytochrome P450 714B3-like | 21.86 | 33.65 ± 4.75 ** |
| comp66583_c0_seq3 | cytochrome P450 714D1-like | 19.16 | 19.30 ± 7.73 |
| comp67012_c0_seq1 | cytochrome P450 734A6-like | 16.32 | 17.48 ± 4.38 ** |
| comp70760_c4_seq11 | Cytochrome P450 86A1 | 12.32 | (2.99 ± 0.07) × 102 ** |
| TF Family | Number of Unigenes Detected | Up-Regulated in Roots | Down-Regulated in Roots |
|---|---|---|---|
| bHLH | 146 | 13 | 10 |
| ERF | 80 | 3 | 1 |
| C2H2 | 79 | 6 | 3 |
| NAC | 78 | 4 | 4 |
| WRKY | 75 | 10 | 5 |
| MYB-related | 74 | 8 | 4 |
| bZIP | 63 | 7 | 0 |
| G2-like | 60 | 4 | 2 |
| MYB | 60 | 15 | 1 |
| HD-ZIP | 49 | 2 | 7 |
| Others | 548 | 18 | 21 |
| Total | 1312 | 90 | 58 |
| SSR Statistics | Number |
|---|---|
| Total number of sequences examined | 79,835 |
| Total size of examined sequences (bp) | 71,342,686 |
| Total number of identified SSRs | 19,158 |
| Number of SSR containing sequences | 15,313 |
| Sequences containing more than 1 SSR | 3067 |
| SSRs present in compound formation | 1194 |
| Mononucleotide repeats | 14,598 |
| Di-nucleotide repeat | 2100 |
| Tri-nucleotide repeat | 2270 |
| Tetra-nucleotide repeat | 155 |
| Penta-nucleotide repeat | 17 |
| Hexa-nucleotide repeat | 18 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lei, X.Y.; Xia, J.; Wang, J.W.; Zheng, L.P. Comparative Transcriptome Analysis Identifies Genes Putatively Involved in 20-Hydroxyecdysone Biosynthesis in Cyanotis arachnoidea. Int. J. Mol. Sci. 2018, 19, 1885. https://doi.org/10.3390/ijms19071885
Lei XY, Xia J, Wang JW, Zheng LP. Comparative Transcriptome Analysis Identifies Genes Putatively Involved in 20-Hydroxyecdysone Biosynthesis in Cyanotis arachnoidea. International Journal of Molecular Sciences. 2018; 19(7):1885. https://doi.org/10.3390/ijms19071885
Chicago/Turabian StyleLei, Xiu Yun, Jing Xia, Jian Wen Wang, and Li Ping Zheng. 2018. "Comparative Transcriptome Analysis Identifies Genes Putatively Involved in 20-Hydroxyecdysone Biosynthesis in Cyanotis arachnoidea" International Journal of Molecular Sciences 19, no. 7: 1885. https://doi.org/10.3390/ijms19071885
APA StyleLei, X. Y., Xia, J., Wang, J. W., & Zheng, L. P. (2018). Comparative Transcriptome Analysis Identifies Genes Putatively Involved in 20-Hydroxyecdysone Biosynthesis in Cyanotis arachnoidea. International Journal of Molecular Sciences, 19(7), 1885. https://doi.org/10.3390/ijms19071885