Abstract
Plant mitochondrial NAD-malic enzyme (NAD-ME), which is composed of α- and β-subunits in many species, participates in many plant biosynthetic pathways and in plant respiratory metabolism. However, little is known about the properties of woody plant NAD-MEs. In this study, we analyzed four NAD-ME genes (PtNAD-ME1 through PtNAD-ME4) in the genome of Populus trichocarpa. PtNAD-ME1 and -2 encode putative α-subunits, while PtNAD-ME3 and -4 encode putative β-subunits. The Populus NAD-MEs were expressed in Escherichia coli cells as GST-tagged fusion proteins. Each recombinant GST-PtNAD-ME protein was purified to near homogeneity by glutathione-Sepharose 4B affinity chromatography. Milligram quantities of each native protein were obtained from 1 L bacterial cultures after cleavage of the GST tag. Analysis of the enzymatic properties of these proteins in vitro indicated that α-NAD-MEs are more active than β-NAD-MEs and that α- and β-NAD-MEs presented different kinetic properties (Vmax, kcat and kcat/Km). The effect of different amounts of metabolites on the activities of Populus α- and β-NAD-MEs was assessed in vitro. While none of the metabolites evaluated in our assays activated Populus NAD-ME, oxalacetate and citrate inhibited all α- and β-NAD-MEs and glucose-6-P and fructose inhibited only the α-NAD-MEs.
1. Introduction
Malic enzyme (ME) catalyzes the oxidative decarboxylation of malate to pyruvate and CO2 in the presence of a divalent metal ion using NAD or NADP as a cofactor. The products of the reaction participate in many biosynthetic pathways and in respiratory metabolism [1]. MEs are classified into three different groups. The first includes the NAD-malic enzymes (NAD-MEs, EC 1.1.1.38) found in Ascaris suum and bacteria, which use NAD and can decarboxylate oxaloacetate (OAA) in addition to malate. The second group consists of the plant NAD-MEs (EC 1.1.1.39), which are not able to decarboxylate OAA. A third group includes the NADP-malic enzymes (NADP-MEs, EC 1.1.1.40). Plant NAD-ME isoforms are found in the mitochondria [2]. NAD-MEs in some C4 and CAM plants provide CO2 for the Calvin cycle during photosynthetic metabolism [3–5]. Furthermore, malate is decarboxylated in the mitochondria through the action of NAD-MEs to produce pyruvate, which is oxidized in the tricarboxylic acid (TCA) cycle [1,5]. Recent metabolic profiling of Arabidopsis plants completely lacking NAD-ME activity revealed that NAD-MEs have a major influence over nocturnal metabolism [6].
Plant NAD-MEs have been found to be composed of two dissimilar subunits (α- and β-) in some species such as potato (Solanum tuberosum; C3 plant) [7], Urochloa panicoides (C4-PEPCK plant) [8], Crassula argentea (CAM plant) [9], and Amaranthus hypochondriacus (C4-NAD-ME plant) [10]. The α- and β-subunits have similar primary amino acid sequences [2] and in vitro studies have provided evidence that both α- and β-subunits are required for NAD-ME activity [9,11]. Another study suggested that the β-subunit plays a regulatory role in NAD-ME activity [10]. However, recent studies showed that the separated recombinant proteins of Arabidopsis thaliana (AtNAD-ME1 and -2) showed NAD-ME activity and display distinct kinetic mechanisms [6,12].
The genes encoding the α- and β-subunits of plant NAD-MEs were identified from various plants, including potato, amaranth and Arabidopsis [6,10,13]. Plant α- and β-NAD-ME are encoded by single genes in the species analyzed so far. NAD-MEs assemble as heterodimers, heterotetramers or heterooctamers in some species, while it forms a homomer in others [6,12,14]. Some plant NAD-MEs are activated by fumarate and coenzyme A (CoA) [15,16] and are also potentially regulated via changes in aggregation state [7,11]. Little is known about the properties of NAD-MEs in woody plants. In this study, we characterized two putative α-NAD-MEs (PtNAD-ME1 and -2) and two putative β-NAD-MEs (PtNAD-ME3 and -4) of the woody model plant. The proteins were successfully expressed in E. coli cells as fusion proteins. The purified recombinant proteins were used to investigate their enzymatic properties.
2. Results and Discussion
2.1. Characterization and Expression Pattern of the PtNAD-ME Gene Family from Populus
Using the sequences of Arabidopsis NAD-MEs (At2g13560 and At4g00570) [6], four NAD-ME genes were found in the genome of Populus ( http://genome.jgi-psf.org/Poptr1_1/Poptr1_1.home.html). These genes were named PtNAD-ME1, PtNAD-ME2, PtNAD-ME3, and PtNAD-ME4 (Table S1). RT-PCR and DNA sequencing analysis showed that these four PtNAD-MEs are all expressed genes in Populus. We further validated their full-length CDS in the Populus genome database (data not shown). We examined transcript levels in the different Populus tissues by quantitative real-time PCR. The genes PtNAD-ME1, PtNAD-ME2, PtNAD-ME3 and PtNAD-ME4 were expressed in various tissues (Figure 1A). Compared with their relative transcript levels, PtNAD-ME1 and PtNAD-ME2 have transcript levels similar to one another in unlignified stem, immature leaf, and root tissues. The corresponding transcript levels of PtNAD-ME3 and PtNAD-ME4 are also similar (Figure 1B). In root tissue, the transcript levels of PtNAD-ME1 and PtNAD-ME2 were higher than those of PtNAD-ME3 and PtNAD-ME4 (Figure 1B). To gain further insight into the structural diversity of PtNAD-ME genes, we compared the exon/intron organization and showed that PtNAD-ME1 and -2 and PtNAD-ME3 and -4 share similar exon/intron structure, respectively (Figure S1). The amino acid sequence analysis revealed high identity among the PtNAD-ME families: 96% identity for PtNAD-ME1 versus PtNAD-ME2 and 92% identity for PtNAD-ME3 versus PtNAD-ME4 (Table S2 and Figure S2). The phylogenetic tree, constructed with the neighbor joining (NJ) method using the program MEGA 4.0 [17], indicated that PtNAD-MEs are divided into two clades (Figure 1C). Some plant NAD-MEs are composed of α- and β-subunits, with molecular masses of approximately 62 and 58 kDa, respectively [9]. PtNAD-ME1 and PtNAD-ME2 belong to the α-NAD-MEs, and PtNAD-ME3 and PtNAD-ME4 clustered with β-NAD-MEs. All Populus NAD-MEs are predicted to contain a mitochondrial targeting peptide using TargetP ( http://www.cbs.dtu.dk/services/TargetP) and SignalP ( http://www.cbs.dtu.dk/services/SignalP). To the best of our knowledge, only one gene encoding α-NAD-ME or β-NAD-ME exists in Arabidopsis, potato, Urochloa panicoides, and Crassula argentea, while the enzymes isolated from Eleusine coracana, Panicum dichotomiflorum, or Amaranthus tricolor were postulated to be composed of identical subunits [6–8,10,18,19].
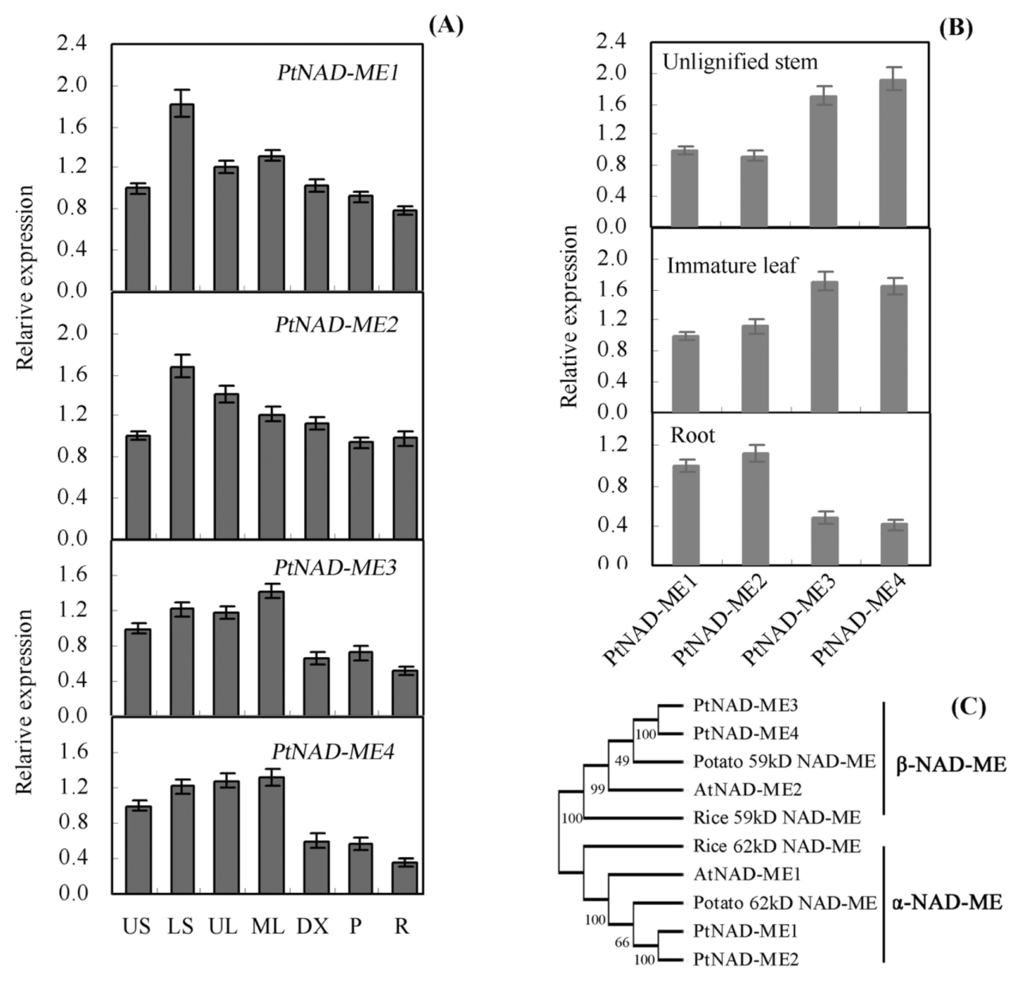
Figure 1.
Characterization of the PtNAD-ME gene family in Populus. (A) Analysis of PtNAD-ME gene expression in Populus by real-time PCR. US, unlignified stem (internodes 1–3 from top); LS, lignified stem (from internodes 8–10); UL, immature leaf (from internodes 2–3); ML, mature leaf (from internodes 8–10); DX, developing xylem (from the basal internodes); Phloem (P, from the basal internodes); R, Root; (B) Comparison of four PtNAD-ME gene expression levels in unlignified stem, immature leaf, and root tissues of Populus, respectively; (C) Phylogenetic analysis of the Populus NAD-MEs with other NAD-MEs by neighbor joining method using MEGA 4.0 software. Accession numbers and sources of the NAD-MEs are as follows: AtNAD-ME1 (At2g13560) and AtNAD-ME2 (At4g00570) from Arabidopsis, rice 59 and 62 kDa NAD-MEs (LOC_Os07g31380 and LOC_Os10g35960 ) and potato 59 and 62 kDa NAD-MEs.
2.2. Expression and Purification of PtNAD-ME Proteins in E. coli
To confirm the proposed function of the putative PtNAD-ME genes, the coding regions of each mature PtNAD-ME were amplified by PCR without signal peptide sequences, cloned in pGEX-6p-3 vector and heterologously expressed in E. coli BL21 cells under the control of the T7 promoter. After 2 h induction with 1 mM IPTG, the presence of each PtNAD-ME fusion protein with the GST tag increased significantly, as determined by SDS-PAGE (Figure S3). The molecular masses of GST-PtNAD-ME1 and GST-PtNAD-ME2 fusion proteins with the 26 kDa GST tag were approximately 93 and 92 kDa, respectively, and approximately 87 kDa for GST-PtNAD-ME3 and GST-PtNAD-ME4 (Figure 2).
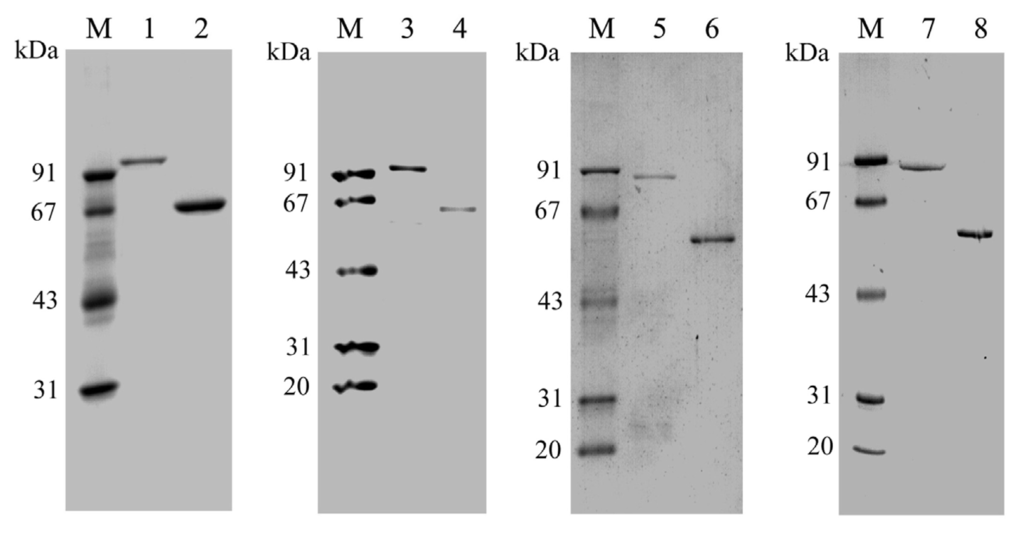
Figure 2.
SDS-PAGE analysis of the recombinant Populus NAD-MEs purified from E. coli BL21 cells. M, marker; 1–2, GST-PtNAD-ME1 (1 μg) and PtNAD-ME1 (5 μg); 3–4, GST-PtNAD-ME2 (2 μg) and PtNAD-ME2 (1 μg); 5–6, GST-PtNAD-ME3 (1 μg) and PtNAD-ME3 (2 μg); 7–8, GST-PtNAD-ME4 (1 μg) and PtNAD-ME4 (2 μg).
Under optimal growth conditions, the cells were collected from 1 L bacterial cultures and lysed with lysozyme. The recombinant active GST-tagged PtNAD-ME proteins (GST-PtNAD-ME1 to GST-PtNAD-ME4) were purified in a single step to near homogeneity by glutathione-Sepharose 4B affinity chromatography (Figure 2). After cleavage of the GST tags using PreScission protease, the molecular masses of native proteins of PtNAD-ME1 to PtNAD-ME4 were approximately 67, 66, 61, and 61 kDa on SDS-PAGE gels, respectively (Figure 2). The results of PtNAD-ME purification are summarized in Table 1.

Table 1.
Summary of purification of four PtNAD-ME recombinant proteins from E. coli BL21 cells in 1 L cultures.
The purified recombinant PtNAD-ME1 and -2 (α-NAD-MEs) have high specific activities of 34.0 U/mg and 40.5 U/mg, respectively. However, the activities of PtNAD-ME3 and -4 (β-NAD-MEs) are very low (Table 1). Previous reports showed that in some plant species both α- and β-subunits are required for NAD-ME activity [9–11]. Taken together, our data suggest that Populus NAD-ME might be composed of dissimilar subunits where the α-subunit would contribute to catalysis and the β-subunit may have a regulatory role. However, Arabidopsis α- and β-subunit proteins displayed high NAD-ME activity and distinct kinetic mechanisms [6,12].
2.3. Optimum pH of Recombinant PtNAD-ME Proteins
The effect of pH on the activity of each recombinant PtNAD-ME protein was investigated. Specific activity was determined from measurements of the oxidative decarboxylation of malate at different pH values (6.2–8.6). When the pH values ranged between 7.0 and 7.8, each recombinant PtNAD-ME enzyme retained more than 60% activity. The optimum pH values of the Populus α-and β-NAD-MEs were 7.2 and 7.4, respectively (Figure 3). For comparison, the optimal pH values for purified NAD-MEs from other plants are reported to be 7.2 in the CAM plant Mesembryanthemum crystallinum [4], 6.4 to 6.6 in the C3 plant Arabidopsis [6] and 7.4 in the C4 plant Panicum miliaceum [20]. The optimal pH values for Populus NAD-MEs are similar to those of Panicum miliaceum and Mesembryanthemum crystallinum.
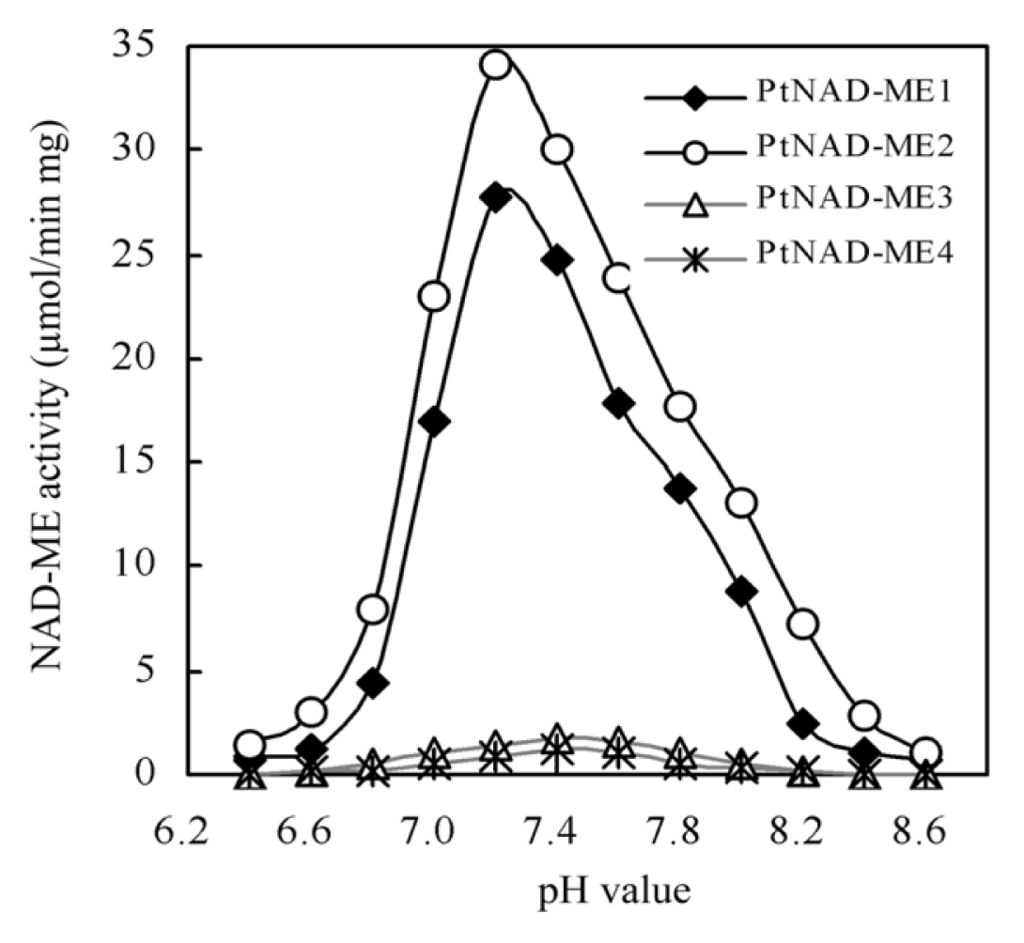
Figure 3.
Effect of pH on the activity four purified PtNAD-ME recombinant proteins. The activity of NAD-ME was determined from measurements of the oxidative decarboxylation of malate as described by Tronconi et al. [6]. Reactions were started by the addition of purified PtNAD-ME recombinant proteins. Values indicate the means of three independent measurements.
2.4. Kinetic Parameters for Recombinant PtNAD-ME Proteins
Kinetic reaction of the recombinant Populus α- and β-NAD-MEs were determined by varying the concentrations of one substrate while keeping the concentrations of other substrates and the cofactor at saturation levels. When malate was the limiting substrate (0.008–16 mM), the activities of α-NAD-MEs (PtNAD-ME1 and -2) and β-NAD-MEs (PtNAD-ME3 and -4) were shown in Figure 4A. The result showed that high concentrations of malate (from 5 to 16 mM) inhibited neither α- nor β-NAD-MEs. Similarly, the activities of Arabidopsis recombinant NAD-ME1 and NAD-ME2 (α- and β-NAD-MEs) are unaffected at a saturating concentration of malate [12,14]. When NAD was the limiting substrate (0.008–16 mM) and the saturated concentration of malate was 4 mM, the activities of Populus α- and β-NAD-MEs were shown in Figure 4B.
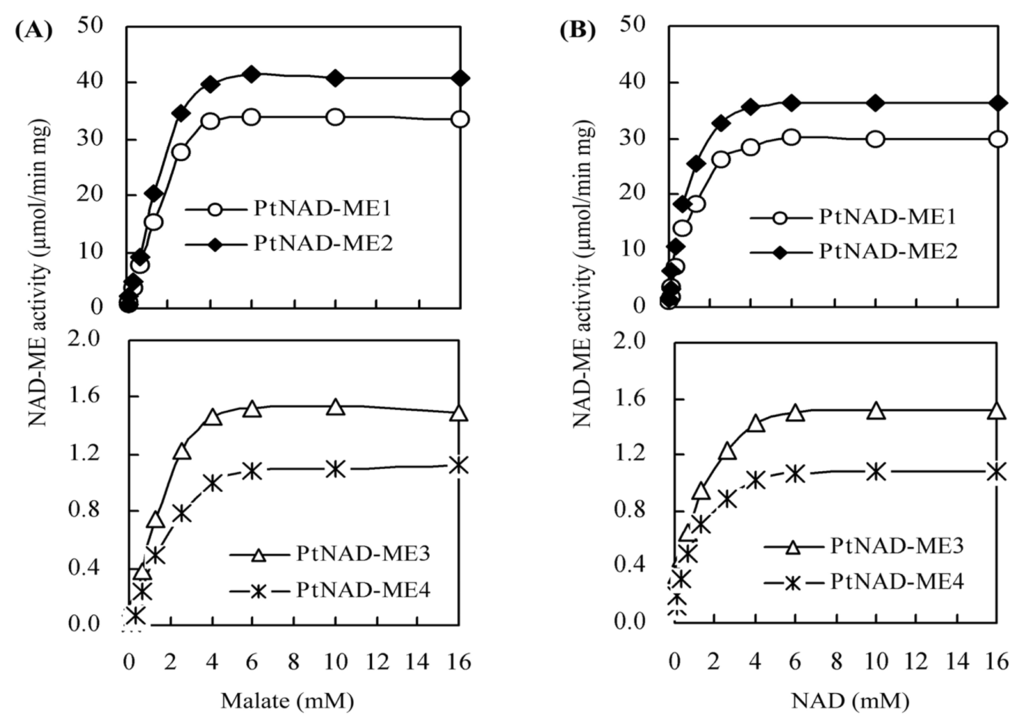
Figure 4.
Activity curves of recombinant PtNAD-ME. Assays were performed at the optimum pH of each PtNAD-ME. The results are presented as the mean of triplicate determinations.
Kinetic parameters were determined for the recombinant Populus NAD-MEs (Table 2). The kinetic properties of both α-NAD-MEs were comparable. In addition, the kinetic properties of both β-NAD-MEs were similar. The α- and β-NAD-MEs showed different kinetic properties. The velocities of the reactions (Vmax and kcat) catalyzed by the α-NAD-MEs were approximately 20 times higher than those catalyzed by the β-NAD-MEs. In addition, the Km values for malate and NAD were in the same order for all PtNAD-MEs. The efficiencies of the reactions catalyzed by the α-NAD-MEs were thus much higher than those of the β-NAD-MEs (Table 2).

Table 2.
Kinetic parameters of four recombinant PtNAD-ME proteins purified from E. coli. The values are presented as the mean of triplicate determinations.
2.5. Effect of Catabolites on the Activities of Recombinant PtNAD-MEs
The activities of recombinant Populus α- and β-NAD-ME proteins in the malate decarboxylation reaction were evaluated in the presence of different metabolites from mitochondrial respiration, the tricarboxylic acid cycle and glycolytic metabolism (Figure 5). Among the metabolites assayed, OAA and citrate inhibited the activities of all á- and â-NAD-MEs. In addition, glucose-6-P and fructose also inhibited the â-NAD-MEs. The plant mitochondrial NAD-ME enzyme has a key role in mitochondrial carbon metabolism, providing a means whereby organic acids can be partitioned between replenishment of mitochondrial pools and complete oxidation [13]. None of the metabolites evaluated in this assay activated Populus NAD-MEs. However, Arabidopsis NAD-ME1 was strongly activated by OAA and fumarate while PEP, FBP, acetyl-CoA and CoA enhanced the activity of NAD-ME2 [14].
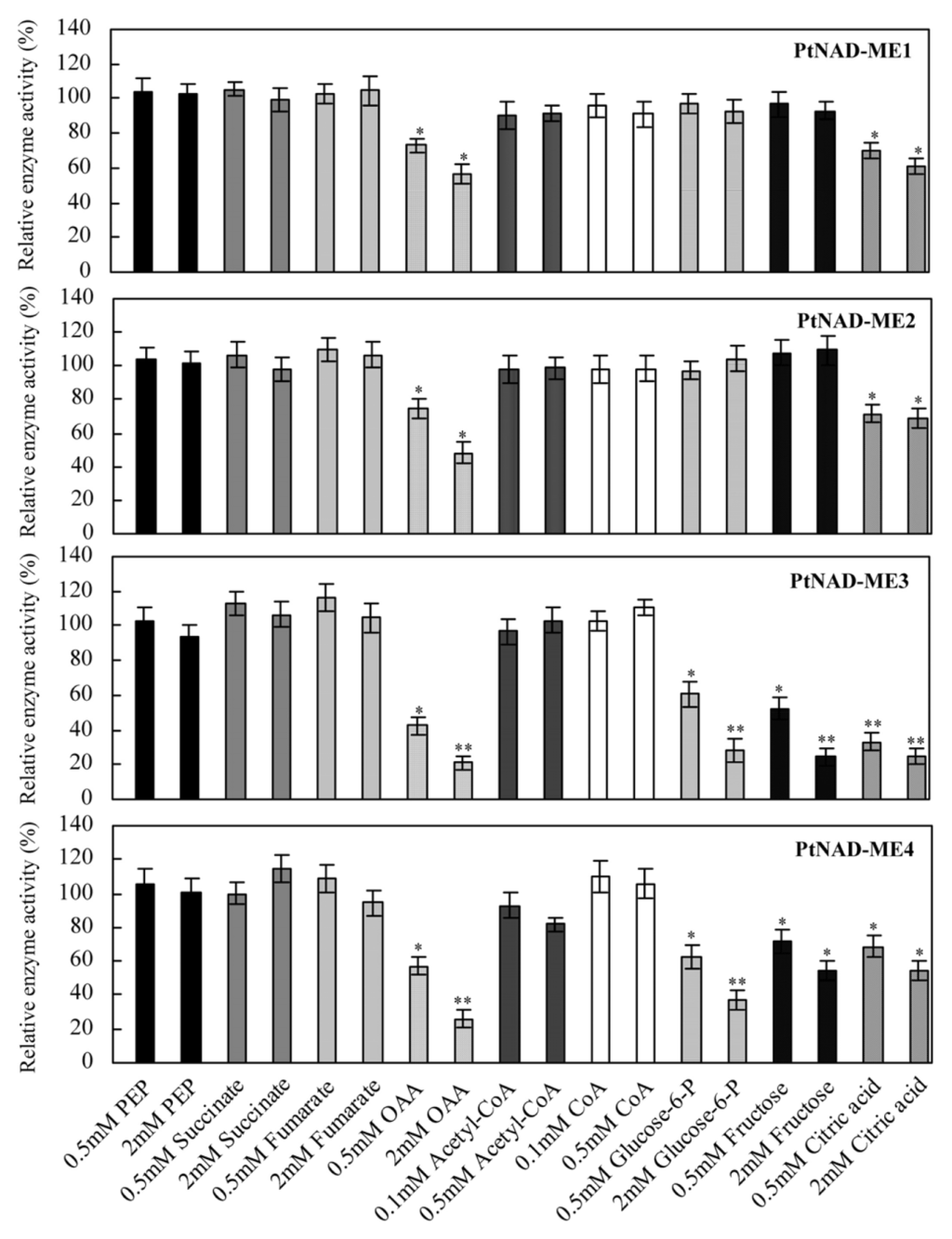
Figure 5.
Effect of different metabolites on the activity of each purified PtNAD-ME recombinant proteins. Activities were measured at the optimum pH of each PtNAD-ME in the absence or presence of each effector. The results represent the percent of activity in the presence of each effector relative to the activity measured in the absence of the metabolites (100%, control). Values are presented as the mean of triplicate determinations. * and ** differ significantly from the control with p < 0.05 and p < 0.01, respectively, by Student’s t-test.
3. Experimental Section
3.1. Plant Growth
Populus trichocarpa was grown in a greenhouse under long day conditions (16 h light/8 h dark) at temperatures of 25–28 °C. Unlignified stem (internodes 1–3 from top), lignified stem (from internodes 8–10), immature leaf (from internodes 2–3), mature leaf (from internodes 8–10), terminal bud, developing xylem (from the basal internodes), phloem (from the basal internodes) and root tissues were separately harvested from one-year-old trees. All samples were immediately frozen in liquid nitrogen and stored at −80 °C before the isolation of total RNA.
3.2. Isolation of PtNAD-ME cDNAs and Real-Time PCR Analysis
Total RNA was isolated from 0.2 g of different tissues of Populus using the pBIOZOL plant total RNA Extraction Reagent according to the manufacturer’s instructions (BioFlux, Hangzhou, China). The quantity and purity of isolated total RNA were determined spectrophotometrically, and the integrity of the RNA was verified by 1.5% denaturing formaldehyde agarose gel electrophoresis. Total cDNAs were synthesized with RNA LA PCR™ Kit (AMV) Ver.1.1 (Takara Company, Dalian, China). Full-length sequences of Populus NAD-ME genes were obtained by RT-PCR (the primers used are described in Table S3) using total cDNAs as templates, subcloned into the entry vector pENTR/D-TOPO (Invitrogen, Carlsbad, NM, USA) and completely sequenced. Real-time PCR analysis was performed to determine the expression levels of the four PtNAD-ME genes in different Populus tissues. The pairs of primers used are described in Table S4.
3.3. Construction of Expression Plasmids
The coding regions of each mature PtNAD-ME (without signal peptide sequences) were amplified using the primers shown in Table S5. The PCR products were ligated into the vector pGEX-6p-3 after it had been treated with the appropriate restriction enzymes. The resulting plasmids were confirmed by DNA sequencing and were named pGEX-6p-3-PtNAD-MEs. E. coli BL21 cells with individual pGEX-6p-3-PtNAD-ME plasmids were used to express the recombinant PtNAD-ME proteins fused with GST tags.
3.4. Expression and Purification of Populus PtNAD-ME Proteins in E. coli
BL21 cells were cultured overnight in LB medium containing 100 μg/mL ampicillin at 37 °C, diluted 1:100 with fresh pre-warmed LB medium containing ampicillin and incubated at 28 °C with shaking at 150 rpm. When OD600 had reached a value of 1.0, expression of GST-PtNAD-ME was induced by addition of IPTG to a final concentration of 0.1 mM, and the cells were incubated for an additional 6 h. The cells were pelleted by centrifugation at 6000× g for 5 min. Cell pellets were resuspended in a pre-cooled lysis buffer (150 mM NaC1, 50 mM Tris-HCl, pH 8.0, 1 mM EDTA, 1 mM DTT, and 1 mM PMSF), and lysozyme was added to the suspension to a final concentration of 1 mg/mL. The suspension was incubated on ice for 1 h and centrifuged at 40,000× g for 30 min. The supernatant, containing the fusion proteins, was loaded onto a glutathione-Sepharose 4B column (GE, Piscataway, NJ, USA) pre-equilibrated with buffer A (100 mM Tris-HCl, pH 8.0, 10 mM MgCl2, 1 mM DTT, and 1 mM PMSF). Non-specifically bound proteins were removed by washing with buffer A, and the bound fusion proteins with GST tags were eluted with buffer containing 50 mM Tris-HCl, pH 8.0, and 10 mM reduced glutathione.
3.5. Cleavage of the GST Tag
Recombinant GST-PtNAD-ME proteins bound to the column were digested by the addition of PreScission Protease (GE, Piscataway, NJ, USA) in elution buffer (50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1 mM EDTA, 1 mM DTT, and 0.01% Triton X-100) and incubated for 16 h at 4 °C. The desired recombinant proteins (each PtNAD-ME isoform) were eluted from the column and either used immediately for activity tests or stored at −80 °C for later use.
3.6. SDS-PAGE
Protein samples were analyzed by SDS-polyacrylamide gel electrophoresis (SDS-PAGE) as described by Laemmli [21] and stained with Coomassie brilliant blue R-250. The BCA protein assay (Pierce, Rockford, IL, USA) was used to determine the protein concentration.
3.7. Assay of NAD-ME Activity
NAD-ME activity was determined spectrophotometrically by monitoring NADH production at 340 nm. The standard reaction mixture contained 150 mM Mes/NaOH, 10 mM MnCl2, 4 mM NAD, and 10 mM malate in a final volume of 1 mL. One unit of enzyme activity is defined as the amount of enzyme resulting in the production of 1 μmol of NADH per minute. The reaction was started by the addition of NAD-ME. The optimum pH for NAD-ME was determined by measuring the oxidative decarboxylation of malate, which was described by Tronconi et al. [6]. When testing different compounds as possible inhibitors or activators of enzymatic activity, NAD-ME activity was measured in the presence of nonsaturating concentrations of malate while maintaining NAD concentrations at saturating levels, as described by Tronconi et al. [14]. The parameters were calculated by triplicate determinations, and the values are the average of three replicates differing by <5%.
3.8. Kinetic Studies on the Recombinant PtNAD-ME Proteins
The kinetic parameters were determined as described by Tronconi et al. [14]. Initial velocity studies were performed by varying the concentration of one substrate around its Km while keeping other substrate concentrations at saturating levels. All kinetic parameters were calculated by triplicate determinations and adjusted to nonlinear regression. The concentrations of malate and NAD each varied between 0.008 and 16 mM. The values are the average of three replicates differing by <5%.
4. Conclusions
In this study, we have found that two α- and two β-NAD-ME genes exist in the genome of Populus trichocarpa and are expressed in various tissues. The characterization of recombinant Populus NAD-MEs in vitro indicates different enzymatic properties between α- and β-NAD-MEs. Also, the effects of metabolites on the activities of recombinant α-NAD-MEs are distinct from those of β-NAD-MEs. It suggests that Populus NAD-ME might be composed of dissimilar subunits where the α-subunit would contribute to catalysis and the β-subunit may have a regulatory role. Further, analysis of the deletion of α- or β-NAD-ME in future will reveal the function of PtNAD-ME in Populus biosynthetic pathway and respiratory metabolism.
Supplementary Information
ijms-14-12994-s001.pdfAcknowledgments
This work was supported by the National High Technology Research and Development Program of China (863 Program) (2011AA100202), the Natural Scientific Foundation of China (30872006) and the Fundamental Research Funds for the Central Universities (DL12DA02).
Conflict of Interest
The authors declare no conflict of interest.
References
- Maier, A.; Zell, M.B.; Maurino, V.G. Malate decarboxylases: Evolution and roles of NAD(P)-ME isoforms in species performing C(4) and C(3) photosynthesis. J. Exp. Bot 2011, 62, 3061–3069. [Google Scholar]
- Winning, B.M.; Bourguignon, J.; Leaver, C.J. Plant mitochondrial NAD-dependent malic enzyme, cDNA cloning, deduced primary structure of the 59- and 62-kDa subunit, import, gene complexity and expression analysis. J. Biol. Chem 1994, 269, 4780–4786. [Google Scholar]
- Hatch, M.D.; Kagawa, T. Activity, location and role of NAD malic enzyme in the leaves of C4 photosynthesis. Aust. J. Plant Physiol 1974, 1, 357–369. [Google Scholar]
- Artus, N.N.; Edwards, G.E. Properties of leaf NAD-malic enzyme from the inducible crassulacean acid metabolism species Mesembryanthemum crystallinum. Plant Cell Physiol 1985, 26, 341–350. [Google Scholar]
- Artus, N.N.; Edwards, G.E. NAD-malic enzyme from plants. FEBS Lett 1985, 182, 225–233. [Google Scholar]
- Tronconi, M.A.; Fahnenstich, H.; Gerrard Wheeler, M.C.; Andreo, C.S.; Flugge, U.I.; Drincovich, M.F.; Maurino, V.G. Arabidopsis NAD-malic enzyme functions as a homodimer and heterodimer and has a major impact during nocturnal metabolism. Plant Physiol 2008, 146, 1540–1552. [Google Scholar]
- Grover, S.D.; Wedding, R.T. Kinetic ramifications of the association-dissociation behavior of NAD-malic enzyme. Plant Physiol 1982, 70, 1169–1172. [Google Scholar]
- Burnell, J.N. Photosynthesis in phosphoenolpyruvate carboxykinase type C4 species: Properties of NAD-malic enzyme from Urochloa panicoides. Aust. J. Plant Physiol 1987, 14, 517–525. [Google Scholar]
- Willeford, K.O.; Wedding, R.T. Evidence for a multiple subunit composition of plant NAD malic enzyme. J. Biol. Chem 1987, 262, 8423–8429. [Google Scholar]
- Long, J.L.; Wang, J.L.; Berry, J.O. Cloning and analysis of the C4 photosynthetic NAD-dependent malic enzyme of Amaranth mitochondria. J. Biol. Chem 1994, 269, 2817–1833. [Google Scholar]
- Grover, S.D.; Wedding, R.T. Modulation of the activity of NAD malic enzyme from Solanum tuberosum by changes in oligomeric state. Arch. Biochem. Biophys 1984, 234, 418–425. [Google Scholar]
- Tronconi, M.A.; Gerrard Wheeler, M.C.; Maurino, V.G.; Drincovich, M.F.; Andreo, C.S. NAD-malic enzymes of Arabidopsis thaliana display distinct kinetic mechanisms that support differences in physiological control. Biochem J 2010, 430, 295–303. [Google Scholar]
- Jenner, H.L.; Winning, B.M.; Millar, A.H.; Tomlinson, K.L.; Leaver, C.J.; Hill, S.A. NAD malic enzyme and the control of carbohydrate metabolism in potato tubers. Plant Physiol 2001, 126, 1139–1149. [Google Scholar]
- Tronconi, M.A.; Maurino, V.G.; Andreo, C.S.; Drincovich, M.F. Three different and tissue-specific NAD-malic enzymes generated by alternative subunit association in Arabidopsis thaliana. J. Biol. Chem 2010, 285, 11870–11879. [Google Scholar]
- Day, D.A.; Neuberger, M.; Douce, R. Activation of NAD-linked malic enzyme in intact mitochondria by exogenous CoA. Arch Biochem. Biophys 1984, 231, 233–234. [Google Scholar]
- Grissom, C.B.; Canellas, P.F.; Wedding, R.T. Allosteric regulation of the NAD malic enzyme from cauliflower: Activation by fumarate and coenzyme A. Arch. Biochem. Biophys. 1983, 220, 133–144. [Google Scholar]
- Koichiro, T.; Joel, D.; Masatoshi, N.; Sudhir, K. MEGA4: Molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol. Biol. Evol 2007, 24, 1596–1599. [Google Scholar]
- Oshugi, R.; Murata, T. Leaf anatomy, post-illumination CO2 burst and NAD-malic enzyme activity in Panicum dichotomiflorum. Plant Cell Physiol 1980, 21, 1329–1333. [Google Scholar]
- Murata, T.; Oshugi, R.; Matsuoka, M.; Nakamoto, M. Purification and characterization of NAD ME from leaves of Eleusine coracana and Panicum dichotomiflorum. Plant Physiol 1989, 89, 316–324. [Google Scholar]
- Rathnam, C.K.M. Studies with isolated bundle sheath mitochondria: Evidence for NAD-malic enzyme-catalyzed decarboxylation of C4 acids in species representing the three C4 metabolic subtypes. FEBS Lett 1978, 96, 367–372. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).