Abstract
Under high pressure conditions, the Diels-Alder reaction involving N,N-diethyl-1-tosyl-3-indoleglyoxylamide and 1-(N-acetyl-N-propylamino)-1,3-butadiene produces a highly functionalized intermediate for the synthesis of Indole Alkaloids, in shorter times and higher yields than under thermal conditions.
Introduction
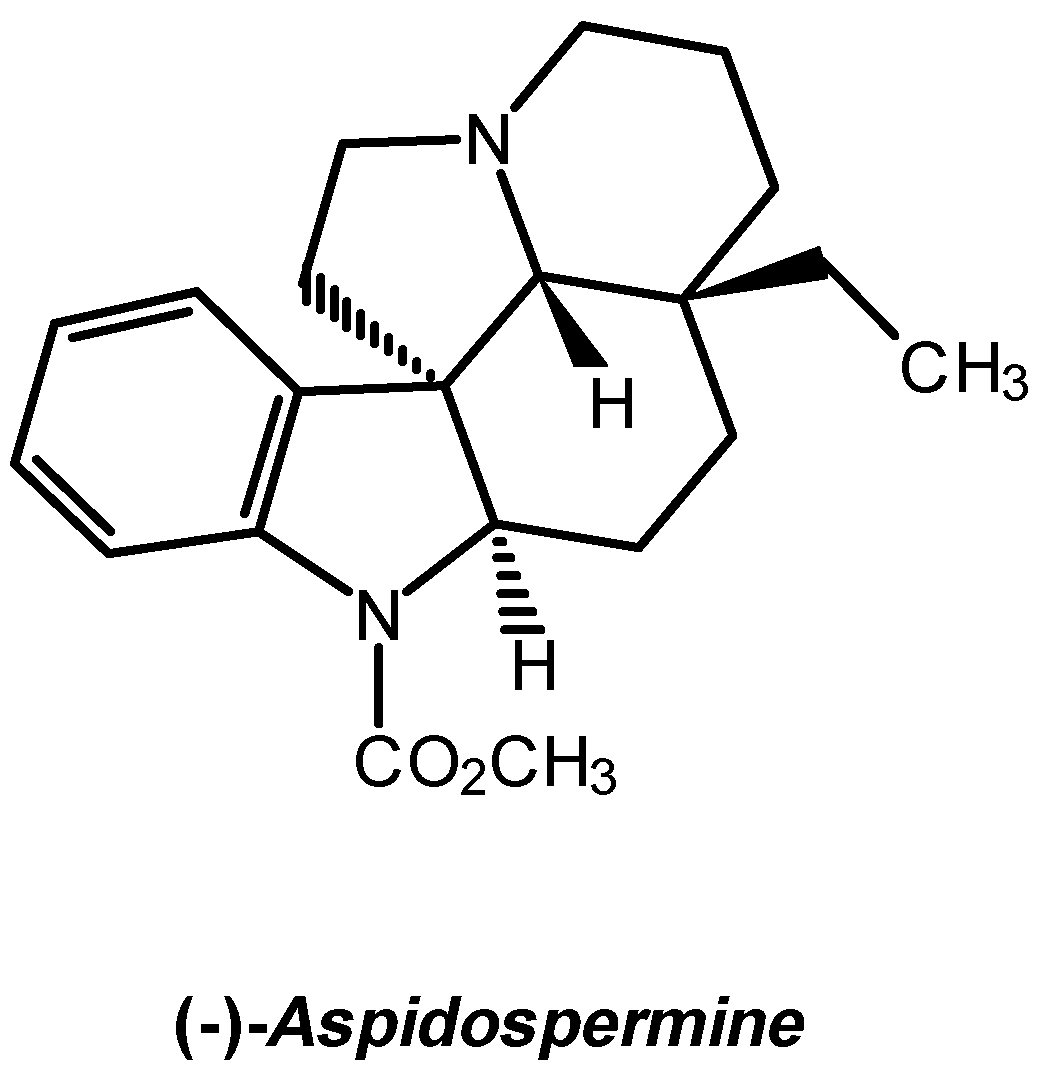
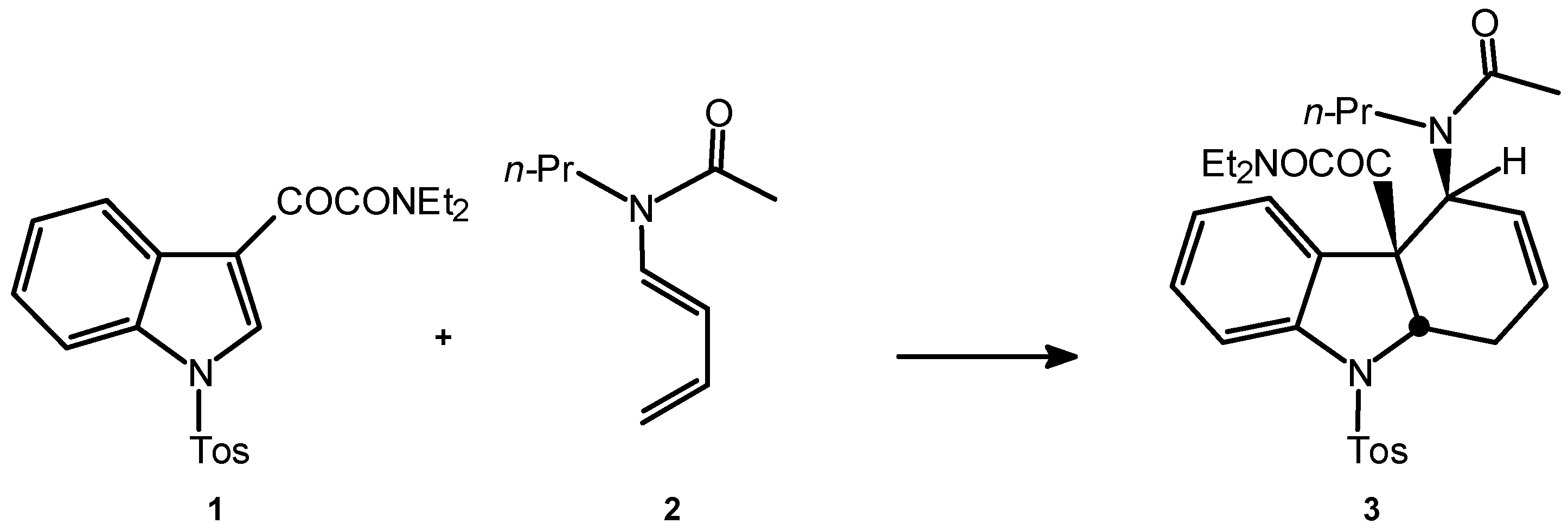
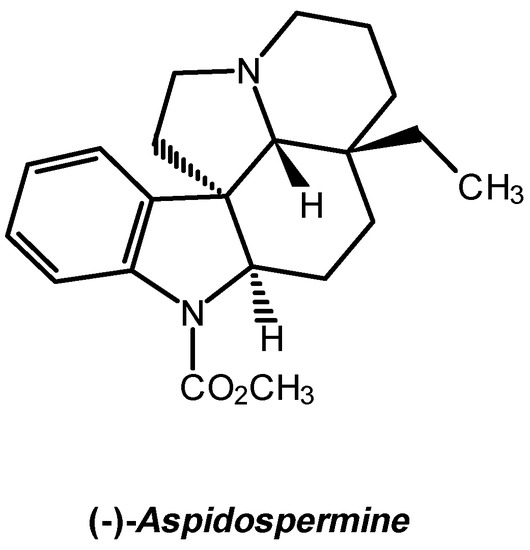
From the limited number of heteroaromatic compounds which can act as dienophiles in normal Diels-Alder (D-A) reactions [1], 1-tosyl-3-nitroindole proves to be the most reactive, leading to high yields in dihydrocarbazoles with nitrous acid extrusion in the reactions involving isoprene (155°C, 26 hours), 1-(N-acetyl-N-propylamino)-1,3-butadiene (90°C, 96 hours) and 1-(N-benzoyl-N-benzylamino)-1,3-butadiene (130°C, 96 hours) [1,2]. The reactions with Danishefsky diene (65°C, 24 hours) produces adducts that keep the original functionality [3]. Under hyperbar conditions (12 kbar, room temperature), the named reactions offer products keeping the nitro-substitution, except with the dien-amides, where the dihydrocarbazole is still the main product [3]. The N,N-diethyl-1-tosyl-3-indoleglyoxylamide 1 is, between the acyl-substituted indoles, the one that produces the highest yields reacting with isopren [1], constituting therefore a potentially suitable substrate for the comparative study of the D-A reactions with 1-(N-acetyl-N-propylamino)-1,3-butadiene 2 under hyperbar and thermal conditions. Complementary, it would allow to synthesise properly substituted intermediates for the advanced synthesis of Indole Alkaloids, such as (-)-Aspidospermine (Scheme 1).
Scheme 1.
Experimental
The thermal reaction between the indoleglyoxamide 1 and the dienamide 2 should be carried out at high temperatures in order to induce the dienophilic character of 1. The maximum limit of temperature is set in 130°C, due to the thermal instability of the dienamide 2. Under thermal conditions (120°C, 96 hours) the reaction leads to two diastereomeric adducts (total regioselectivity) in very low yields (ca. 9%). Under hyperbar conditions (11.5 kbar, 40°C, 48 hours), the reaction leads to 50% of a single iso-mer 3 (Scheme 3), which arises from the exo addition (Scheme 2). This experimental condition allows to recover ca. 48% of the unreacted dienophile.
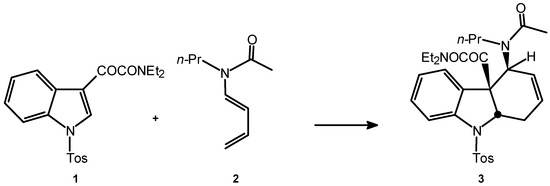
Scheme 3.
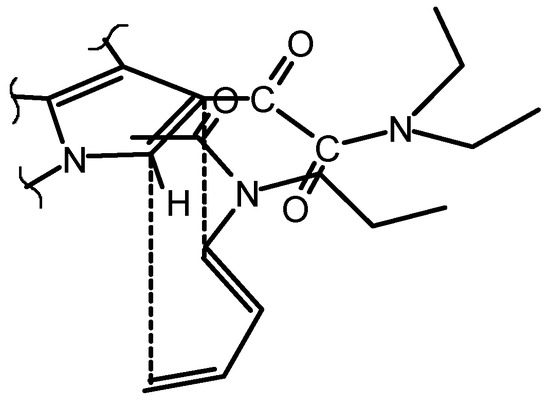
Scheme 2.
Results and Discussions
The D-A reaction between N,N-diethyl-1-tosyl-3-indoleglyoxamide and 1-(N-acetyl-N-propylamino)-1,3-butadiene, carried out under hyperbar conditions, is clean, fast and leads to higher yields compared to thermal conditions, since it allows to produce a single adduct which holds the basic skeleton and appropriate functionality of (-)-Aspidospermine and related Plumerane alkaloids.
Acknowledgements
We are indebted to the Science and Technology Secretariat - Universidad Na-cional del Litoral, República Argentina, for the financial support, CAI+D Program (Projects 94-0858-007-056 y 96-00-024-161). The authors want to thank Prof. Serge Piettre from IRCOF - Université de Rouen, France, for his valuable advice, and Dr. Gonzalez Sierra from the IQUIOS – UNR (Argentina) for the NMR experiments.
References and Notes
- Wenkert, E.; Moeller, P. D. R.; Piettre, S. J. Am. Chem. Soc. 1988, 110, 7188.
- Biolatto, B.; Kneeteman, M.; Mancini, P. Tetrahedron Lett. 1999, 40, 3343.
- Biolatto, B.; Kneeteman, M.; Gonzalez Sierra, M.; Mancini, P. M. Unpublished results.