Abstract
Five disulfated steroids and a mixture of monosulfated steroids were isolated from the ethanolic extract of the antarctic ophiuroid Gorgonocephalus chilensis. The struc- tures were determined by 1H-NMR, 13C-NMR and FABMS.
Introduction
Sulfated polyhydroxysteroids have been described from a wide variety of marine organisms, in par- ticular sponges and echinoderms. These compounds have exhibited interesting biological activities, in particular, cytotoxic action, inhibition of protein tyrosine kinases and anti-HIV properties [1]. Re- cently, we have demonstrated the antiviral activity of sulfated steroids isolated from the patagonic ophiuroid Ophioplocus januarii against four different pathogenic viruses in humans [2]. We have also isolated three novel sulfated polyhydroxylated steroids from the antarctic ophiuroid Astrotoma agas- sizii [3]. These compounds showed antiviral activity against herpes simples virus, polio virus and Junin virus, which causes a severe disease in humans known as Argentine hemorrhagic fever [4].
Experimental
The animals were homogenized in ethanol and the aqueous extract obtained after evaporation of the solvent was partitioned between water and cyclohexane. The aqueous phase was extracted with n- buthanol and the buthanolic extract was purified by Sephadex LH20 (MeOH). Fractions containing the polar steroids were purified by vacuum-dry column chromatography on sílica gel C-18 (MeOH/H2O, MeOH) and HPLC. Structural determination of the purified compounds was performed by H-NMR, 13C-NMR, FABMS and by solvolysis reactions.
Results and Discussion
We were able to isolate and characterize five disulfated polyhydroxysteroids (1-5). The compounds possess a sulfate group at C-21 and with exception of 2, all have a sulfate group at C-3(α). Compounds 2 and 3 are isomers that differ only in the location of the sulfate group in ring A. Compound 2 presents a sulfate group at C-2(β). Recently, we have isolated steroid 2 from another antarctic ophiuroid As- trotoma agassizii (3) and demonstrated its antiviral activity against herpes simplex 2 virus (4).
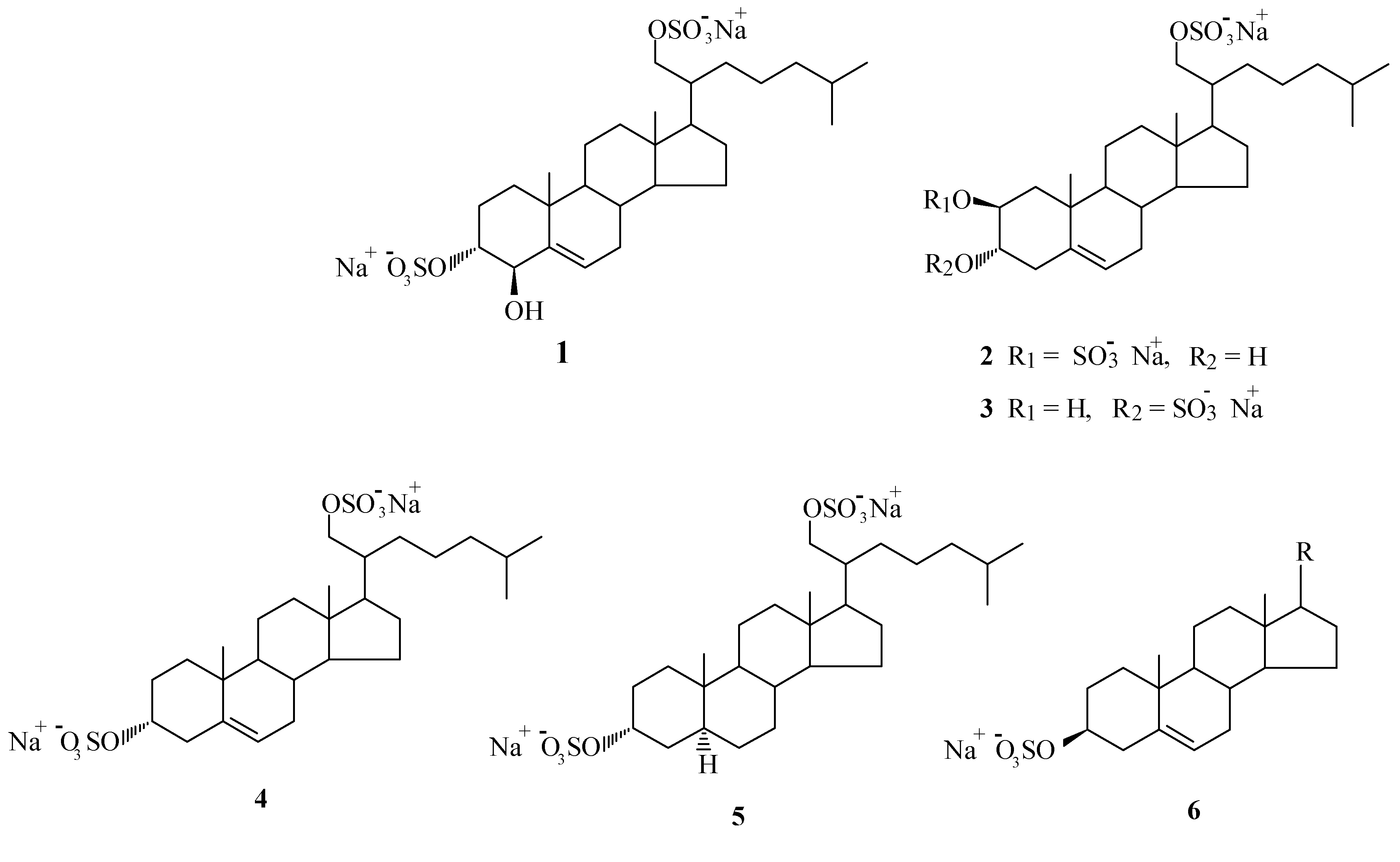
Compounds 4 and 5 differ in the insaturation in ring B and were separated by reversed phase HPLC. We have also isolated a mixture of monosulfated steroids at C-3(β). The composition of the mixture was determined by solvolysis of the sulfate group and analysis of the steroid mixture by glc.
Acknowledgements:
We are grateful to the University of Buenos Aires (Project TX-85) and the Inter- national Foundation for Science (Project F/1583-3) for partial financial support.
References and Notes
- McKee, T.C.; Cardellina, J.H.; Riccio, R.; D´Auria, M.V.; Iorizzi, M.; Minale, L.; Moran, R.A.; Gulakowski, R.J.; McMahon, J.B.; Buckheit, R.W.; Snader, K.M.; Boyd, M.R. J. Med. Chem. 1994, 37, 793. [PubMed]
- Roccatagliata, A.J.; Maier, M.S.; Seldes, A.M.; Pujol, C.A.; Damonte, E.B. J. Nat. Prod. 1996, 59, 887. [PubMed]
- Roccatagliata, A.J.; Maier, M.S.; Seldes, A.M. J. Nat. Prod. 1998, 61, 370. [PubMed]
- Comin, M.J.; Maier, M.S.; Roccatagliata, A.J.; Pujol, C.A.; Damonte, E.B. Steroids 1999, 64, 335. [PubMed]
