(Substituted-quinoline-1-yl) Methylbenzylammonium Chloride: Quaternization Reaction Process, Corrosion Inhibition Behavior, and Calculation Process
Abstract
1. Introduction
2. Results and Discussion
2.1. Surface Morphology Analysis
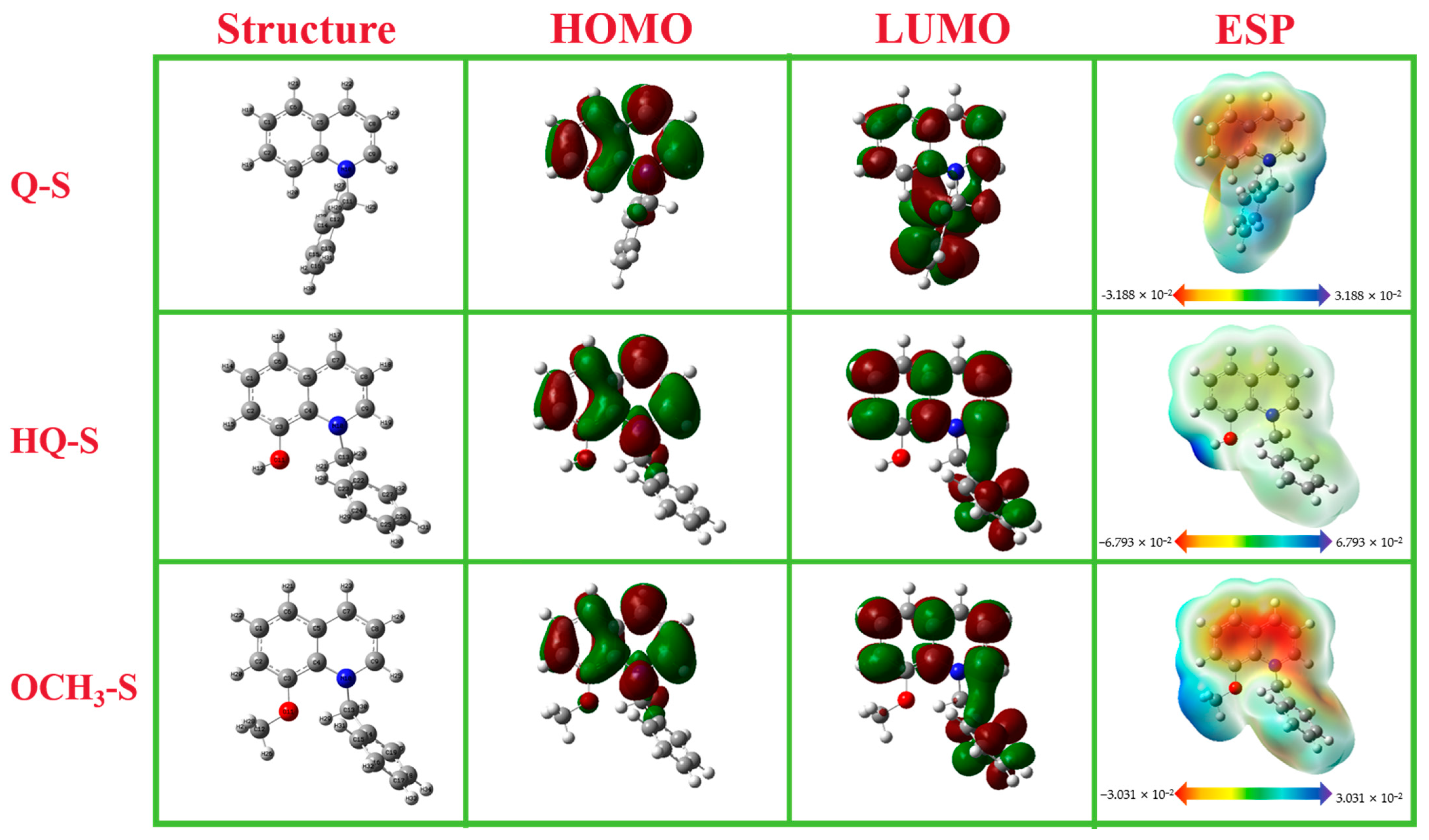
2.2. Quantum Chemical Calculation
2.3. Molecular Dynamics Simulation Process
2.4. Adsorption Isotherm Models
3. Experimental Methods
3.1. Materials
3.2. Physicochemical Characterization
3.3. Electrochemical Measurements
3.4. Calculation Details
3.5. Synthesis of Quinoline-Based Quaternary Ammonium Salts
4. Anti-Corrosion Mechanism
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tasic, Z.Z.; Mihajlovic, M.B.P.; Antonijevic, M.M. The influence of chloride ions on the anti-corrosion ability of binary inhibitor system of 5-methyl-1H-benzotriazole and potassium sorbate in sulfuric acid solution. J. Mol. Liq. 2016, 222, 1–7. [Google Scholar] [CrossRef]
- Sudheer; Quraishi, M.A. Electrochemical and theoretical investigation of triazole derivatives on corrosion inhibition behavior of copper in hydrochloric acid medium. Corros. Sci. 2013, 70, 161–169. [Google Scholar] [CrossRef]
- Alsaiari, R.A.; Kamel, M.M.; Mohamed, M.M. Inhibition of Expired Cefazolin Drug on Copper Metal in Dilute Hydrochloric Acid Solution: Practical and Theoretical Approaches. Molecules 2024, 29, 1157. [Google Scholar] [CrossRef]
- Cong, Y.F.; Liu, Y.; Zhang, C.; Jiang, G.M.; Huang, W. Modification of lmidazoline Quaternary Ammonium Salt. Liaoning Univ. Petro. Chem. Technol. 2014, 34, 5–8. [Google Scholar] [CrossRef]
- Ren, Z.B.; Lu, S.; Liu, L. Corrosion Inhibition Performance of Two Kinds of Schiff Base Inhibitors of o-Phenylenediamine. J. Petrochem. Univ. 2019, 32, 8–14. [Google Scholar] [CrossRef]
- Song, W.W.; Zhang, J.; Du, M. Research progress of bisquaternary ammonium compound inhibitors. Chem. Ind. Eng. Prog. 2011, 30, 842–847. [Google Scholar] [CrossRef]
- Ser, C.T.; Uvela, P.; Wong, M.W. Prediction of corrosion inhibition efficiency of pyridines and quinolines on an iron surface using machine learning-powered quantitative structure-property relationships. Appl. Surf. Sci. 2020, 512, 145612. [Google Scholar] [CrossRef]
- Verma, C.; Quraishi, M.A.; Ebenso, E.E. Quinoline and its derivatives as corrosion inhibitors: A review. Surf. Interfaces 2020, 21, 100634. [Google Scholar] [CrossRef]
- Zhou, K.; Jiang, L.; Xiang, B. Toxicity prediction of three quinoline derivatives and their corrosion inhibition effect on carbon steel. Surf. Technol. 2018, 47, 219–224. [Google Scholar] [CrossRef]
- Mourya, P.; Banerjee, S.; Singh, M.M. Corrosion inhibition of mild steel in acidic solution by Tagetes erecta (Marigold flower) extract as a green inhibitor. Corros. Sci. 2014, 85, 352–363. [Google Scholar] [CrossRef]
- Zong, P. Synthetise and Corrosion Inhibition Mechanism Investigation of Quaternary Ammonium Corrosion Inhibitors for Acidification. Master’s Thesis, China University of Petroleum (East China), Qingdao, China, April 2008. [Google Scholar]
- Peme, T.; Olasunkanmi, L.O.; Bahadur, I.; Adekunle, A.S.; Kabanda, M.M.; Ebenso, E.E. Adsorption and Corrosion Inhibition Studies of Some Selected Dyes as Corrosion Inhibitors for Mild Steel in Acidic Medium: Gravimetric, Electrochemical, Quantum Chemical Studies and Synergistic Effect with Iodide Ions. Molecules 2014, 20, 16004–16029. [Google Scholar] [CrossRef]
- Tian, H.W.; Cheng, Y.F.; Li, W.H.; Hou, B.R. Triazolyl-acylhydrazone derivatives as novel inhibitors for copper corrosion in chloride solutions. Corros. Sci. 2015, 100, 341–352. [Google Scholar] [CrossRef]
- Li, M.L.; Cao, S.Y.; Meng, L.B.; Gui, J.Z.; Zhang, L.; Liu, D. Corrosion Inhibition of Q235 Steel by Acid Ionic Liquidin in HCl Solution. J. Liaoning Univ. Pet. Chem. Technol. 2015, 35, 17–31. [Google Scholar] [CrossRef]
- Deyab, M.A. Decyl glucoside as a corrosion inhibitor for magnesium–air battery. J. Power Sources 2016, 325, 98–103. [Google Scholar] [CrossRef]
- Yıldız, R.; Doğan, T.; Dehri, İ. Evaluation of corrosion inhibition of mild steel in 0.1 M HCl by 4-amino-3-hydroxynaphthalene-1-sulphonic acid. Corros. Sci. 2014, 85, 215–221. [Google Scholar] [CrossRef]
- Qiang, Y.J.; Zhang, S.T.; Yan, S.; Zou, X.; Chen, S. Three indazole derivatives as corrosion inhibitors of copper in a neutral chloride solution. Corros. Sci. 2017, 126, 295–304. [Google Scholar] [CrossRef]
- Sigircik, G.; Yildirim, D.; Tiiken, T. Synthesis and inhibitory effect of N, N-bis (1-phenylethanol) ethylenediamine against steel corrosion in HCl Media. Corros. Sci. 2017, 120, 184–193. [Google Scholar] [CrossRef]
- Sherif, E.S.; Erasmus, R.M.; Comins, J.D. Inhibition of copper corrosion in acidic chloride pickling solutions by 5-(3aminophenyl)-tetrazole as a corrosion inhibitor. Corros. Sci. 2008, 50, 3439–3445. [Google Scholar] [CrossRef]
- Alibakhshi, E.; Ramezanzadeh, M.; Bahlakeh, G.; Ramezanzadeh, B.; Mahdavian, M.; Motamedi, M. Glycyrrhiza glabra leaves extract as a green corrosion inhibitor for mild steel in 1 M hydrochloric acid solution: Experimental, molecular dynamics, Monte Carlo and quantum mechanics study. J. Mol. Liq. 2018, 255, 185–198. [Google Scholar] [CrossRef]
- Singh, A.; Ansari, K.R.; Chauhan, D.S.; Quraishi, M.A.; Lgaz, H.; Chung, I.M. Comprehensive investigation of steel corrosion inhibition at macro/micro level by ecofriendly green corrosion inhibitor in 15% HCl medium. J. Colloid Interface Sci. 2020, 560, 225–236. [Google Scholar] [CrossRef]
- Zhang, G.H.; Liu, J.; Dong, Q.C.; Ni, M.L.; Luo, J.; Yang, D.D. Synthesis and corrosion inhibition of two kinds of 2-(dimethylamino) ethyl methacrylate quaternary ammonium salts. Fine Chem. 2020, 37, 1056–1064. [Google Scholar] [CrossRef]
- Pearson, R.G. Absolute electronegativity and hardness: Application to inorganic chemistry. J. Org. Chem. 1988, 27, 734. [Google Scholar] [CrossRef]
- Mourya, P.; Singh, P.; Tewari, A.K.; Rastogi, R.B.; Singh, M.M. Relationship between structure and inhibition behaviour of quinolinium salts for mild steel corrosion: Experimental and theoretical approach. Corros. Sci. 2015, 95, 71–87. [Google Scholar] [CrossRef]
- Zarrouk, A.; Zarrok, H.; Salghi, R.; Hammouti, B.; Touzani, R.; Bouachrine, M.; Warad, I.; Hadda, T.; Arabia, S. A Theoretical Investigation on the Corrosion Inhibition of Copper by Quinoxaline Derivatives in Nitric Acid Solution. Int. J. Electrochem. Sci. 2012, 7, 6353–6364. [Google Scholar] [CrossRef]
- Pareek, S.; Jain, D.; Hussain, S.; Biswas, A.; Shrivastava, R.; Parida, S.K.; Kisan, H.K.; Lgaz, H.; Chung, I.M.; Behera, D. A New Insight into Corrosion Inhibition Mechanism of Copper in Aerated 3.5% (by weight) NaCl Solution by Eco-friendly Imidazopyrimidine Dye: Experimental and Theoretical Approach. Chem. Eng. J. 2019, 358, 725–742. [Google Scholar] [CrossRef]
- Obot, I.B.; Obi-Egbedi, N.O.; Ebens, E.E.; Afolabi, A.S.; Oguzie, E.E. Experimental, quantum chemical calculations, and molecular dynamic simulations insight into the corrosion inhibition properties of 2-(6-methylpyridin-2-yl)oxazolo[5,4-f][1,10]phenanthroline on mild steel. Res. Chem. Intermed. 2013, 39, 1927–1948. [Google Scholar] [CrossRef]
- El-Lateef, H.M.A.; Sayed, A.R.; Gomha, S.M.; Bakir, E.M.; Shalabi, K. Synthesis and study of poly[(hydrazinylazo)]thiazoles as potent corrosion inhibitors for cast iron-carbon alloy in molar HCl: A collective computational and experiential methods. J. Mol. Liq. 2021, 337, 116555. [Google Scholar] [CrossRef]
- Simovi, A.R.; Novakovi, J.; Milovanovi, B.; Etinski, M.; Janakovi, P.; Bajat, J.B. Nature-Derived corrosion inhibition of steel in HCl by Pani spruce essential Oil: Insights from experimental and Computational Approaches. Appl. Surf. Sci. 2025, 698, 163048. [Google Scholar] [CrossRef]
- Lei, X.W.; Zhang, J.T.; Bai, Z.Q.; Feng, Y.R.; Zhang, J.X. Inhibition Effect of Quinoline Quaternary Ammonium Salt on Electrochemical Behavior of Super 13Cr Stainless Steel in Acid Medium. Corros. Sci. Prot. Technol. 2015, 27, 358–362. [Google Scholar] [CrossRef]
- Madkour, L.H.; Kaya, S.; Obot, I.B. Computational, Monte Carlo simulation and experimental studies of some arylazotriazoles (AATR) and their copper complexes in corrosion inhibition process. J. Mol. Liq. 2018, 260, 351–374. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Wang, X.J.; Tian, J.; Xing, J.J.; Liu, L. Epoxy composite coating with excellent anti-corrosion and self-healing properties based on mesoporous silica nano-containers. J. Mol. Struct. 2023, 1294, 136538. [Google Scholar] [CrossRef]
- Behera, D.; Kumar, S.; Yadav, M. Experimental and Quantum Chemical Studies on the Corrosion Inhibition Performance of Benzimidazole Derivatives for Mild Steel in HCl. Ind. Eng. Chem. Res. 2013, 52, 6318–6328. [Google Scholar] [CrossRef]
- Ibrahimi, B.E.; Jmiai, A.; Bazzi, L.; Lssami, S.E. Amino Acids and their Derivatives as Corrosion Inhibitors for Metals and Alloys. Ar. J. Chem. 2020, 13, 740–771. [Google Scholar] [CrossRef]









| Corrosion Inhibitor | C mM | Icorr A·cm−2 | Ecorr V | βa V·dec−1 | −βc V·dec−1 | ηp % |
|---|---|---|---|---|---|---|
| Blank | 0 | 2.1526× 10−5 | −0.34889 | 0.08388 | 0.06618 | —— |
| Q-S | 1 | 4.1545 × 10−6 | −0.30616 | 0.07125 | 0.06139 | 80.70 |
| 5 | 3.5549 × 10−6 | −0.30565 | 0.07150 | 0.06840 | 83.49 | |
| 10 | 2.0735 × 10−6 | −0.30513 | 0.07504 | 0.07399 | 90.37 | |
| 15 | 1.8562 × 10−6 | −0.29799 | 0.06011 | 0.06982 | 91.38 | |
| 20 | 1.6251 × 10−6 | −0.28749 | 0.05258 | 0.07104 | 92.45 | |
| HQ-S | 1 | 3.0785 × 10−6 | −0.34039 | 0.09234 | 0.06853 | 85.70 |
| 5 | 2.0665 × 10−6 | −0.31612 | 0.08294 | 0.07146 | 90.40 | |
| 10 | 1.8541 × 10−6 | −0.31092 | 0.11100 | 0.07277 | 91.39 | |
| 15 | 1.5353 × 10−6 | −0.29954 | 0.09374 | 0.07732 | 92.87 | |
| 20 | 1.3406 × 10−6 | −0.29171 | 0.08697 | 0.06592 | 93.77 | |
| OCH3-S | 1 | 1.7866 × 10−6 | −0.31597 | 0.11879 | 0.07518 | 91.70 |
| 5 | 1.4376 × 10−6 | −0.30185 | 0.10981 | 0.06692 | 93.32 | |
| 10 | 1.2667 × 10−6 | −0.29784 | 0.10945 | 0.06442 | 94.12 | |
| 15 | 1.0071 × 10−6 | −0.29585 | 0.09887 | 0.06726 | 95.32 | |
| 20 | 8.1103 × 10−7 | −0.28963 | 0.08090 | 0.07251 | 96.23 |
| Corrosion Inhibitor | C mM | Rf Ω cm2 | Rct Ω cm2 | Rp Ω cm2 | Cf μF cm−2 | n1 | Cdl μF cm−2 | n2 | η % |
|---|---|---|---|---|---|---|---|---|---|
| blank | — | — | 240 | 240 | — | — | 334.439 | 0.72 | — |
| Q-S | 1 | 19.0 | 1370 | 1389.0 | 121.670 | 0.85 | 118.439 | 0.70 | 82.48 |
| 5 | 40.6 | 1484 | 1524.6 | 31.810 | 0.79 | 87.793 | 0.66 | 83.83 | |
| 10 | 209.0 | 2397 | 2606.0 | 28.127 | 0.77 | 23.236 | 0.60 | 89.99 | |
| 15 | 210.2 | 2432 | 2642.2 | 20.176 | 0.74 | 19.405 | 0.49 | 90.13 | |
| 20 | 220.8 | 2944 | 3164.8 | 14.799 | 0.71 | 14.385 | 0.44 | 91.85 | |
| HQ-S | 1 | 72.2 | 1411 | 1483.2 | 43.412 | 0.86 | 188.526 | 0.49 | 83.99 |
| 5 | 68.4 | 2447 | 2515.4 | 17.015 | 0.74 | 15.447 | 0.46 | 90.19 | |
| 10 | 236.6 | 2725 | 2961.6 | 12.505 | 0.73 | 11.258 | 0.46 | 91.19 | |
| 15 | 106.9 | 3076 | 3182.9 | 11.635 | 0.71 | 9.674 | 0.43 | 91.20 | |
| 20 | 309.7 | 4567 | 4876.7 | 10.632 | 0.69 | 8.222 | 0.63 | 94.74 | |
| OCH3-S | 1 | 189.2 | 3163 | 3352.2 | 18.843 | 0.89 | 37.255 | 0.59 | 92.41 |
| 5 | 67.0 | 4848 | 4915.0 | 11.801 | 0.87 | 8.873 | 0.41 | 95.05 | |
| 10 | 98.6 | 5655 | 5753.6 | 9.994 | 0.81 | 7.255 | 0.50 | 95.76 | |
| 15 | 29.0 | 6978 | 7007.0 | 8.981 | 0.80 | 6.886 | 0.59 | 96.56 | |
| 20 | 409.7 | 7787 | 8196.7 | 8.425 | 0.75 | 6.297 | 0.83 | 96.92 |
| Corrosion Inhibitor | ELUMO (eV) | EHOMO (eV) | ΔE (eV) | χ (eV) | γ (eV) | ΔN | μ (Debye) |
|---|---|---|---|---|---|---|---|
| Q-S | −1.464 | −6.053 | 4.589 | 5.321 | 2.295 | 0.183 | 2.012 |
| HQ-S | −1.479 | −5.718 | 4.239 | 3.599 | 2.120 | −0.122 | 1.550 |
| OCH3-S | −1.416 | −5.633 | 4.217 | 3.525 | 2.109 | −0.136 | 2.109 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, J.; Zinatullin, R.; Qian, J.; Li, Y.; Kang, X.; Li, J.; Wang, Y.; Huang, H.; Xing, J. (Substituted-quinoline-1-yl) Methylbenzylammonium Chloride: Quaternization Reaction Process, Corrosion Inhibition Behavior, and Calculation Process. Molecules 2025, 30, 4782. https://doi.org/10.3390/molecules30244782
Tian J, Zinatullin R, Qian J, Li Y, Kang X, Li J, Wang Y, Huang H, Xing J. (Substituted-quinoline-1-yl) Methylbenzylammonium Chloride: Quaternization Reaction Process, Corrosion Inhibition Behavior, and Calculation Process. Molecules. 2025; 30(24):4782. https://doi.org/10.3390/molecules30244782
Chicago/Turabian StyleTian, Jianing, Roman Zinatullin, Jianhua Qian, Yanping Li, Xueming Kang, Junhua Li, Yanan Wang, He Huang, and Jinjuan Xing. 2025. "(Substituted-quinoline-1-yl) Methylbenzylammonium Chloride: Quaternization Reaction Process, Corrosion Inhibition Behavior, and Calculation Process" Molecules 30, no. 24: 4782. https://doi.org/10.3390/molecules30244782
APA StyleTian, J., Zinatullin, R., Qian, J., Li, Y., Kang, X., Li, J., Wang, Y., Huang, H., & Xing, J. (2025). (Substituted-quinoline-1-yl) Methylbenzylammonium Chloride: Quaternization Reaction Process, Corrosion Inhibition Behavior, and Calculation Process. Molecules, 30(24), 4782. https://doi.org/10.3390/molecules30244782







