Assessment of ADC Higher Order Structure Through 2D NMR Analysis
Abstract
1. Introduction
2. Results
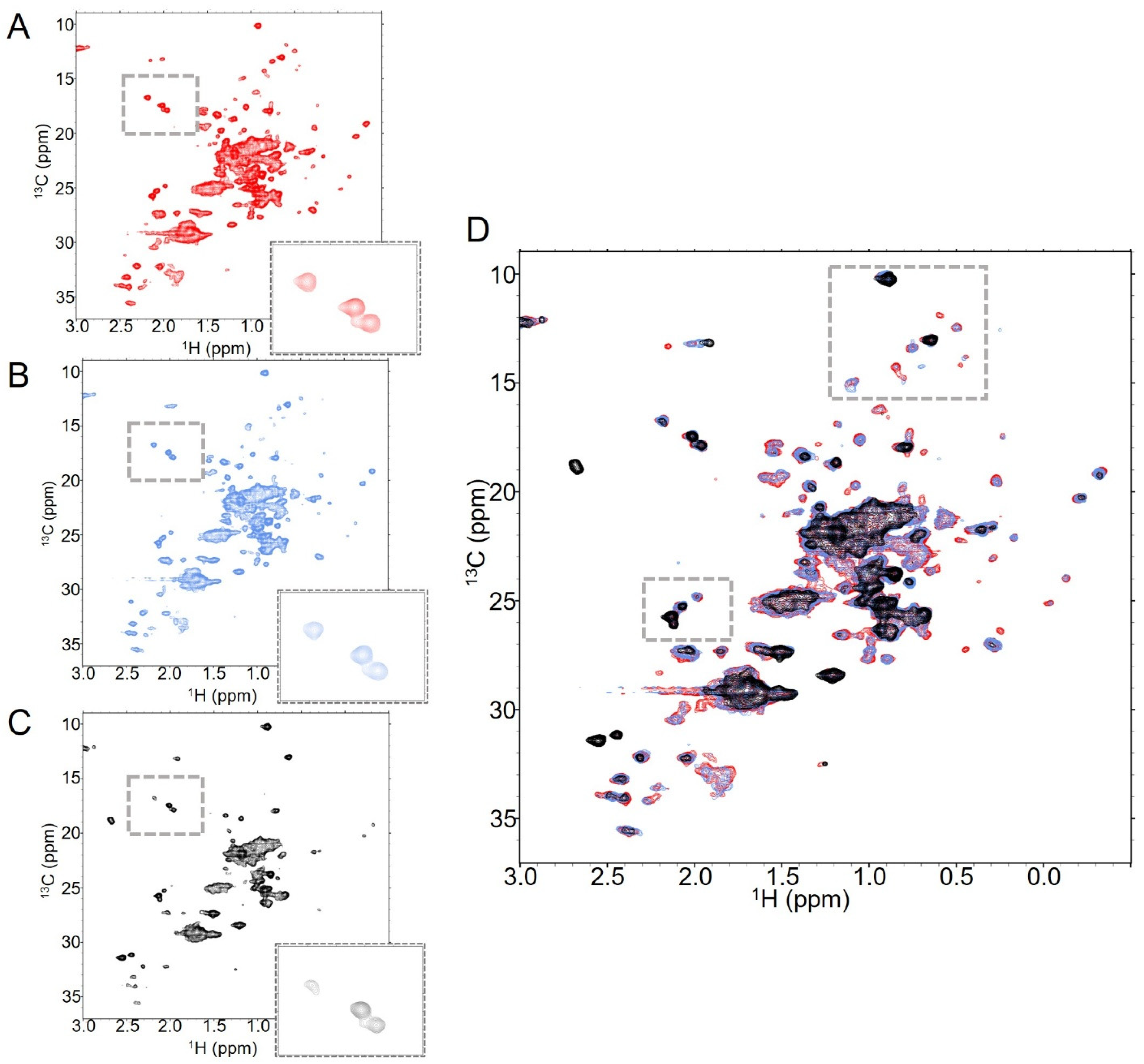
2.1. Trastuzumab Generates Consistent NMR Spectra Under Various Conditions
2.2. T-MMAE NMR Spectra Indicate That the Antibody Structure Is Largely Unchanged by Attachment of the Payload
2.3. T-MMAE NMR Spectra Show Hallmarks of Aggregation and Instability with Increased DAR
2.4. T-DXd and T-MMAE NMR Spectra Are Remarkably Similar at Low DAR
2.5. T-DXd NMR Spectra Show Fewer Characteristics of Aggregation at Higher DAR
3. Discussion
4. Materials and Methods
4.1. Conjugation of T-MMAE and T-DXd
4.2. Characterization of DAR and Monomer
4.3. Trastuzumab NMR Sample Preparation
4.4. ADC NMR Sample Preparation
4.5. NMR Data Collection and Analysis
4.6. Cell Line Expression and Growth
4.7. IC50 Determination
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADC | Antibody–drug conjugate |
| DAR | Drug-antibody ratio |
| NMR | Nuclear magnetic resonance |
| MMAE | monomethyl auristatin E |
| DXd | deruxtecan |
| T-MMAE | Trastuzumab-mc-vc-PAB-MMAE |
| T-DXd | Trastuzumab-deruxtecan |
| IC50 | Half maximal inhibitory concentration |
References
- Hafeez, U.; Parakh, S.; Gan, H.K.; Scott, A.M. Antibody–Drug Conjugates for Cancer Therapy. Molecules 2020, 25, 4764. [Google Scholar] [CrossRef]
- Baah, S.; Laws, M.; Rahman, K.M. Antibody–Drug Conjugates—A Tutorial Review. Molecules 2021, 26, 2943. [Google Scholar] [CrossRef]
- Jain, N.; Smith, S.W.; Ghone, S.; Tomczuk, B. Current ADC Linker Chemistry. Pharm. Res. 2015, 32, 3526–3540. [Google Scholar] [CrossRef] [PubMed]
- Tsuchikama, K.; Anami, Y.; Ha, S.Y.Y.; Yamazaki, C.M. Exploring the next Generation of Antibody–Drug Conjugates. Nat. Rev. Clin. Oncol. 2024, 21, 203–223. [Google Scholar] [CrossRef] [PubMed]
- Long, R.; Zuo, H.; Tang, G.; Zhang, C.; Yue, X.; Yang, J.; Luo, X.; Deng, Y.; Qiu, J.; Li, J.; et al. Antibody-Drug Conjugates in Cancer Therapy: Applications and Future Advances. Front. Immunol. 2025, 16, 1516419. [Google Scholar] [CrossRef]
- Conilh, L.; Sadilkova, L.; Viricel, W.; Dumontet, C. Payload Diversification: A Key Step in the Development of Antibody–Drug Conjugates. J. Hematol. Oncol. 2023, 16, 3. [Google Scholar] [CrossRef]
- Tong, J.T.W.; Harris, P.W.R.; Brimble, M.A.; Kavianinia, I. An Insight into FDA Approved Antibody-Drug Conjugates for Cancer Therapy. Molecules 2021, 26, 5847. [Google Scholar] [CrossRef]
- Donaghy, H. Effects of Antibody, Drug and Linker on the Preclinical and Clinical Toxicities of Antibody-Drug Conjugates. mAbs 2016, 8, 659–671. [Google Scholar] [CrossRef]
- Mahmood, I. Clinical Pharmacology of Antibody-Drug Conjugates. Antibodies 2021, 10, 20. [Google Scholar] [CrossRef] [PubMed]
- Buecheler, J.W.; Winzer, M.; Tonillo, J.; Weber, C.; Gieseler, H. Impact of Payload Hydrophobicity on the Stability of Antibody–Drug Conjugates. Mol. Pharm. 2018, 15, 2656–2664. [Google Scholar] [CrossRef]
- Jin, Y.; Schladetsch, M.A.; Huang, X.; Balunas, M.J.; Wiemer, A.J. Stepping Forward in Antibody-Drug Conjugate Development. Pharmacol. Ther. 2022, 229, 107917. [Google Scholar] [CrossRef]
- Colombo, R.; Rich, J.R. The Therapeutic Window of Antibody Drug Conjugates: A Dogma in Need of Revision. Cancer Cell 2022, 40, 1255–1263. [Google Scholar] [CrossRef] [PubMed]
- Hamblett, K.J.; Senter, P.D.; Chace, D.F.; Sun, M.M.C.; Lenox, J.; Cerveny, C.G.; Kissler, K.M.; Bernhardt, S.X.; Kopcha, A.K.; Zabinski, R.F.; et al. Effects of Drug Loading on the Antitumor Activity of a Monoclonal Antibody Drug Conjugate. Clin. Cancer Res. 2004, 10, 7063–7070. [Google Scholar] [CrossRef]
- Brinson, R.G.; Marino, J.P.; Delaglio, F.; Arbogast, L.W.; Evans, R.M.; Kearsley, A.; Gingras, G.; Ghasriani, H.; Aubin, Y.; Pierens, G.K.; et al. Enabling Adoption of 2D-NMR for the Higher Order Structure Assessment of Monoclonal Antibody Therapeutics. mAbs 2019, 11, 94–105. [Google Scholar] [CrossRef]
- Beaumont, V.A.; Liu, L.; Shi, H.; Rouse, J.C.; Kim, H.-Y. Application of NMR and Chemometric Analyses to Better Understand the Quality Attributes in pH and Thermally Degraded Monoclonal Antibodies. Pharm. Res. 2023, 40, 2457–2467. [Google Scholar] [CrossRef]
- Hodgson, D.J.; Ghasriani, H.; Aubin, Y. Assessment of the Higher Order Structure of Humira®, Remicade®, Avastin®, Rituxan®, Herceptin®, and Enbrel® by 2D-NMR Fingerprinting. J. Pharm. Biomed. Anal. 2019, 163, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Freedberg, D.I.; Keire, D.A. NMR Profiling of Biomolecules at Natural Abundance Using 2D 1H–15N and 1H–13C Multiplicity-Separated (MS) HSQC Spectra. J. Magn. Reson. 2015, 251, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Arbogast, L.W.; Brinson, R.G.; Marino, J.P. Application of Natural Isotopic Abundance 1H–13C- and 1H–15N-Correlated Two-Dimensional NMR for Evaluation of the Structure of Protein Therapeutics. In Methods in Enzymology; Kelman, Z., Ed.; Isotope Labeling of Biomolecules-Applications; Academic Press: New York, NY, USA, 2016; Volume 566, pp. 3–34. [Google Scholar]
- Brinson, R.G.; Arbogast, L.W.; Marino, J.P.; Delaglio, F. Best Practices in Utilization of 2D-NMR Spectral Data as the Input for Chemometric Analysis in Biopharmaceutical Applications. J. Chem. Inf. Model. 2020, 60, 2339–2355. [Google Scholar] [CrossRef]
- Ma, J.; Pathirana, C.; Liu, D.Q.; Miller, S.A. NMR Spectroscopy as a Characterization Tool Enabling Biologics Formulation Development. J. Pharm. Biomed. Anal. 2023, 223, 115110. [Google Scholar] [CrossRef]
- Arbogast, L.W.; Delaglio, F.; Brinson, R.G.; Marino, J.P. Assessment of the Higher-Order Structure of Formulated Monoclonal Antibody Therapeutics by 2D Methyl Correlated NMR and Principal Component Analysis. Curr. Protoc. Protein Sci. 2020, 100, e105. [Google Scholar] [CrossRef]
- Yandrofski, K.; Mouchahoir, T.; De Leoz, M.L.; Duewer, D.; Hudgens, J.W.; Anderson, K.W.; Arbogast, L.; Delaglio, F.; Brinson, R.G.; Marino, J.P.; et al. Interlaboratory Studies Using the NISTmAb to Advance Biopharmaceutical Structural Analytics. Front. Mol. Biosci. 2022, 9, 876780. [Google Scholar] [CrossRef] [PubMed]
- Tokunaga, Y.; Takeuchi, K. Role of NMR in High Ordered Structure Characterization of Monoclonal Antibodies. Int. J. Mol. Sci. 2020, 22, 46. [Google Scholar] [CrossRef] [PubMed]
- Baldisseri, D.; Luo, S.; Ancajas, C.A.F.; Ortega-Rodriguez, U.; Fischer, C.; Zou, G.; Gu, J.; Keire, D.; Piotto, M.; Zhang, B. NMR-Based Structural Integrity Analysis of Therapeutic Monoclonal Antibodies: A Comparative Study of Humira and Its Biosimilars. mAbs 2025, 17, 2551208. [Google Scholar] [CrossRef]
- Yagi, H.; Zhang, Y.; Yagi-Utsumi, M.; Yamaguchi, T.; Iida, S.; Yamaguchi, Y.; Kato, K. Backbone 1H, 13C, and 15N Resonance Assignments of the Fc Fragment of Human Immunoglobulin G Glycoprotein. Biomol. NMR Assign. 2015, 9, 257–260. [Google Scholar] [CrossRef]
- Gagné, D.; Aramini, J.M.; Aubin, Y. Backbone and Methyl Side-Chain Resonance Assignments of the Single Chain Fab Fragment of Trastuzumab. Biomol. NMR Assign. 2024, 18, 119–128. [Google Scholar] [CrossRef]
- Schaefer, C.; Cornet, E.; Piotto, M. NMR Coupled with Multivariate Data Analysis for Monitoring the Degradation of a Formulated Therapeutic Monoclonal Antibody. Int. J. Pharm. 2024, 667, 124894. [Google Scholar] [CrossRef]
- Joshi, S.; Khatri, L.R.; Kumar, A.; Rathore, A.S. NMR Based Quality Evaluation of mAb Therapeutics: A Proof of Concept Higher Order Structure Biosimilarity Assessment of Trastuzumab Biosimilars. J. Pharm. Biomed. Anal. 2022, 214, 114710. [Google Scholar] [CrossRef]
- Adem, Y.T.; Schwarz, K.A.; Duenas, E.; Patapoff, T.W.; Galush, W.J.; Esue, O. Auristatin Antibody Drug Conjugate Physical Instability and the Role of Drug Payload. Bioconjugate Chem. 2014, 25, 656–664. [Google Scholar] [CrossRef]
- Guo, J.; Kumar, S.; Chipley, M.; Marcq, O.; Gupta, D.; Jin, Z.; Tomar, D.S.; Swabowski, C.; Smith, J.; Starkey, J.A.; et al. Characterization and Higher-Order Structure Assessment of an Interchain Cysteine-Based ADC: Impact of Drug Loading and Distribution on the Mechanism of Aggregation. Bioconjugate Chem. 2016, 27, 604–615. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Ponte, J.F.; Yoder, N.C.; Laleau, R.; Coccia, J.; Lanieri, L.; Qiu, Q.; Wu, R.; Hong, E.; Bogalhas, M.; et al. Effects of Drug–Antibody Ratio on Pharmacokinetics, Biodistribution, Efficacy, and Tolerability of Antibody–Maytansinoid Conjugates. Bioconjugate Chem. 2017, 28, 1371–1381. [Google Scholar] [CrossRef] [PubMed]
- Ogitani, Y.; Aida, T.; Hagihara, K.; Yamaguchi, J.; Ishii, C.; Harada, N.; Soma, M.; Okamoto, H.; Oitate, M.; Arakawa, S.; et al. DS-8201a, A Novel HER2-Targeting ADC with a Novel DNA Topoisomerase I Inhibitor, Demonstrates a Promising Antitumor Efficacy with Differentiation from T-DM1. Clin. Cancer Res. 2016, 22, 5097–5108. [Google Scholar] [CrossRef]
- Nakada, T. Discovery Research and Translation Science of Trastuzumab Deruxtecan, from Non-Clinical Study to Clinical Trial. Transl. Regul. Sci. 2021, 3, 65–71. [Google Scholar] [CrossRef]
- Chiu, D.; Pan, L.; Fay, L.; Eakin, C.; Valliere-Douglass, J. Structural Characterization of a Monomethylauristatin-E Based ADC That Contains 8 Drugs Conjugated at Interchain Cysteine Residues. J. Pharm. Biomed. Anal. 2021, 205, 114309. [Google Scholar] [CrossRef]
- Bordeau, B.M.; Nguyen, T.D.; Polli, J.R.; Chen, P.; Balthasar, J.P. Payload-Binding Fab Fragments Increase the Therapeutic Index of MMAE Antibody-Drug Conjugates. Mol. Cancer Ther. 2023, 22, 459–470. [Google Scholar] [CrossRef]
- Singh, A.P.; Sharma, S.; Shah, D.K. Quantitative Characterization of In Vitro Bystander Effect of Antibody-Drug Conjugates. J. Pharmacokinet. Pharmacodyn. 2016, 43, 567–582. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhang, K.; Wang, W.; Liu, Y.; Huang, J.; Zheng, M.; Li, L.; Zhang, X.; Xu, M.; Chen, G.; et al. Combined Inhibition of HER2 and VEGFR Synergistically Improves Therapeutic Efficacy via PI3K-AKT Pathway in Advanced Ovarian Cancer. J. Exp. Clin. Cancer Res. CR 2024, 43, 56. [Google Scholar] [CrossRef] [PubMed]
- Sheen, D.A.; Shen, V.K.; Brinson, R.G.; Arbogast, L.W.; Marino, J.P.; Delaglio, F. Chemometric Outlier Classification of 2D-NMR Spectra to Enable Higher Order Structure Characterization of Protein Therapeutics. Chemom. Intell. Lab. Syst. Int. J. Spons. Chemom. Soc. 2020, 199, 103973. [Google Scholar] [CrossRef]
- Rößler, P.; Mathieu, D.; Gossert, A.D. Enabling NMR Studies of High Molecular Weight Systems Without the Need for Deuteration: The XL-ALSOFAST Experiment with Delayed Decoupling. Angew. Chem. Int. Ed. Engl. 2020, 59, 19329–19337. [Google Scholar] [CrossRef] [PubMed]
- Mueller, L. Alternate HMQC Experiments for Recording HN and HC-Correlation Spectra in Proteins at High Throughput. J. Biomol. NMR 2008, 42, 129–137. [Google Scholar] [CrossRef]
- Arbogast, L.W.; Delaglio, F.; Tolman, J.R.; Marino, J.P. Selective Suppression of Excipient Signals in 2D 1H–13C Methyl Spectra of Biopharmaceutical Products. J. Biomol. NMR 2018, 72, 149–161. [Google Scholar] [CrossRef]
- Delaglio, F.; Grzesiek, S.; Vuister, G.W.; Zhu, G.; Pfeifer, J.; Bax, A. NMRPipe: A Multidimensional Spectral Processing System Based on UNIX Pipes. J. Biomol. NMR 1995, 6, 277–293. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Tonelli, M.; Markley, J.L. NMRFAM-SPARKY: Enhanced Software for Biomolecular NMR Spectroscopy. Bioinformatics 2015, 31, 1325–1327. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Brüschweiler, R. COLMARvista: An Open Source 2D and Pseudo-3D NMR Spectral Processing, Visualization, and Analysis Software in JavaScript. J. Biomol. NMR 2025, 79, 171–179. [Google Scholar] [CrossRef] [PubMed]




| εmAb 280 nm (1%) | 14.81 |
| εmAb 280 nm (M−1 cm−1) | 219,188 |
| εmAb 369 nm (M−1 cm−1) | 0 |
| εDXd 280 nm (M−1 cm−1) | 6275 |
| εDXd 369 nm (M−1 cm−1) | 21,176 |
| A280 C.F. (DXd) | 0.296 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grasso, E.M.; Marquard, A.N.; Sparta, Z.; Fry, D.; Jain, N. Assessment of ADC Higher Order Structure Through 2D NMR Analysis. Molecules 2025, 30, 4490. https://doi.org/10.3390/molecules30224490
Grasso EM, Marquard AN, Sparta Z, Fry D, Jain N. Assessment of ADC Higher Order Structure Through 2D NMR Analysis. Molecules. 2025; 30(22):4490. https://doi.org/10.3390/molecules30224490
Chicago/Turabian StyleGrasso, Emily M., Angela N. Marquard, Zachary Sparta, David Fry, and Nareshkumar Jain. 2025. "Assessment of ADC Higher Order Structure Through 2D NMR Analysis" Molecules 30, no. 22: 4490. https://doi.org/10.3390/molecules30224490
APA StyleGrasso, E. M., Marquard, A. N., Sparta, Z., Fry, D., & Jain, N. (2025). Assessment of ADC Higher Order Structure Through 2D NMR Analysis. Molecules, 30(22), 4490. https://doi.org/10.3390/molecules30224490