Disubstituted Meldrum’s Acid: Another Scaffold with SuFEx-like Reactivity
Abstract
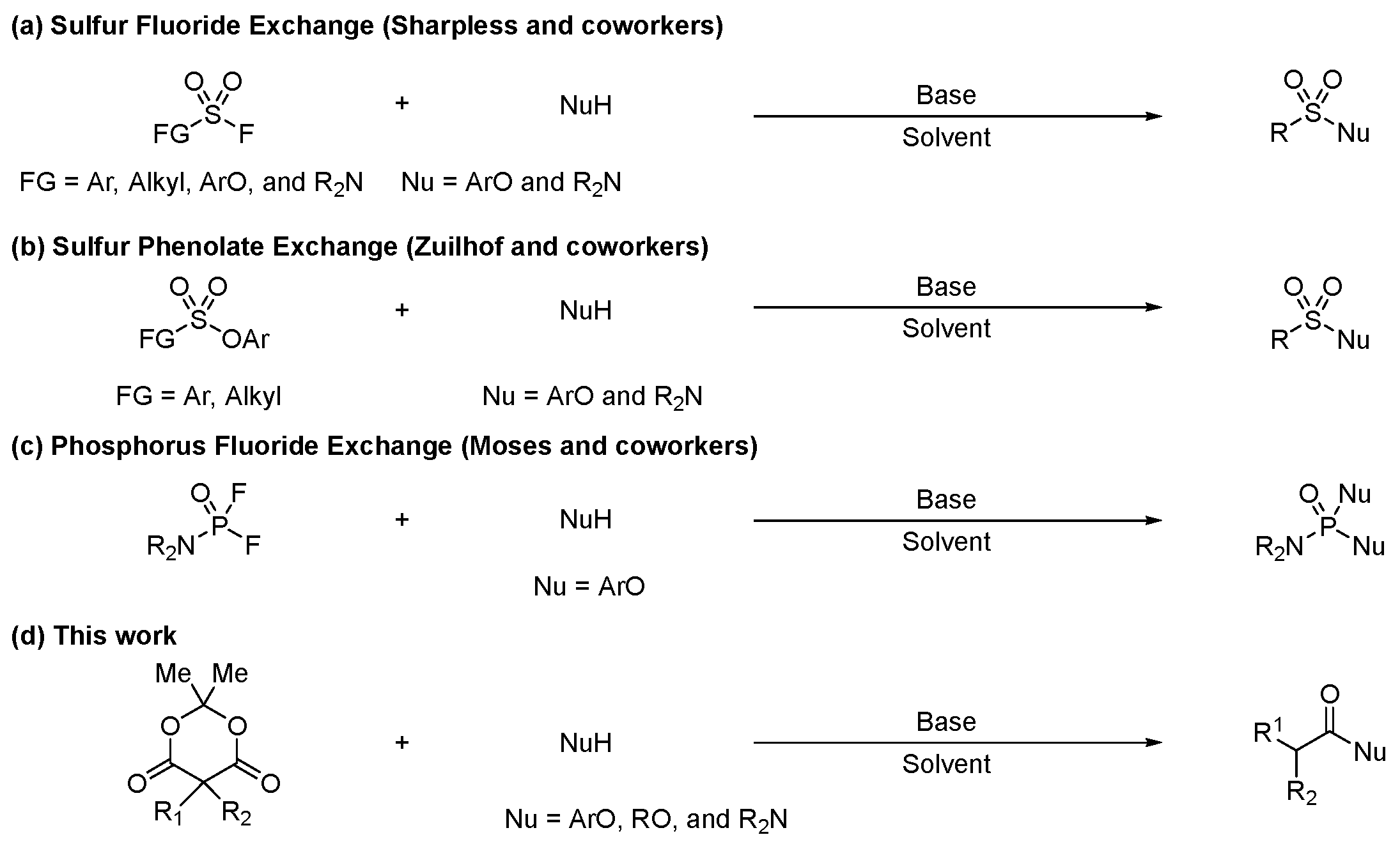
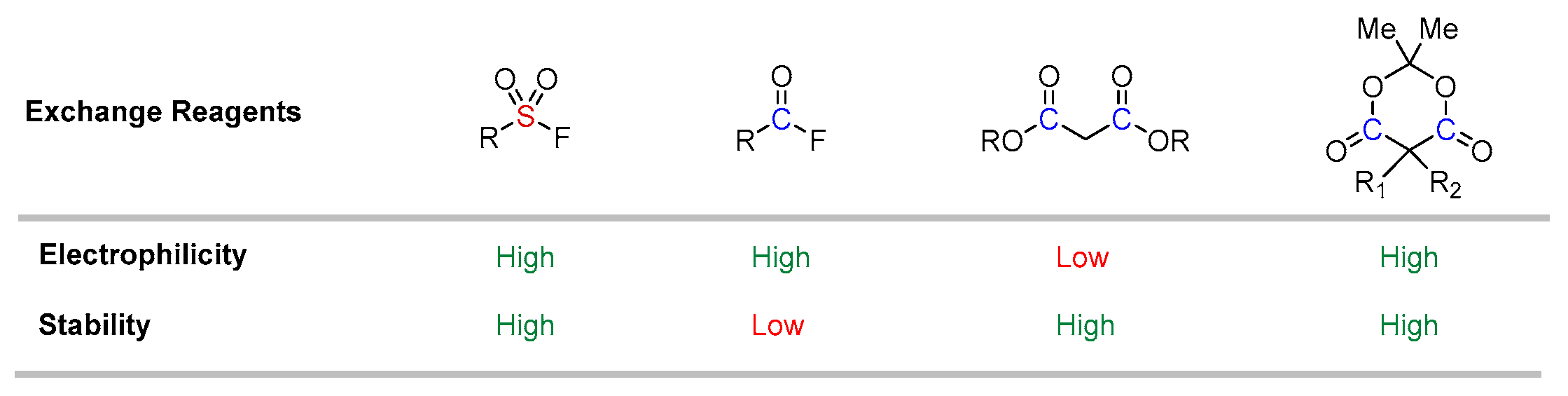
1. Introduction
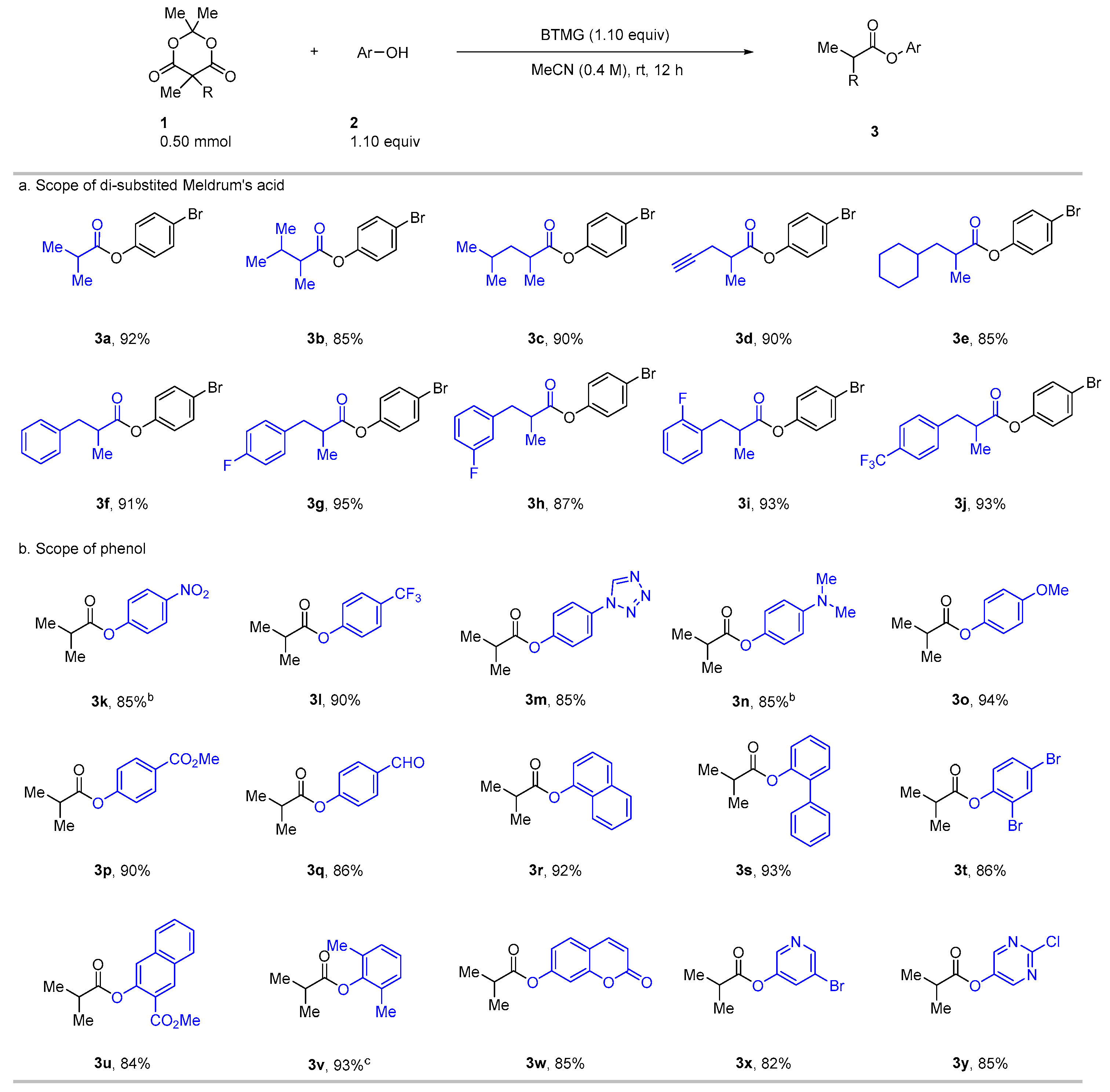
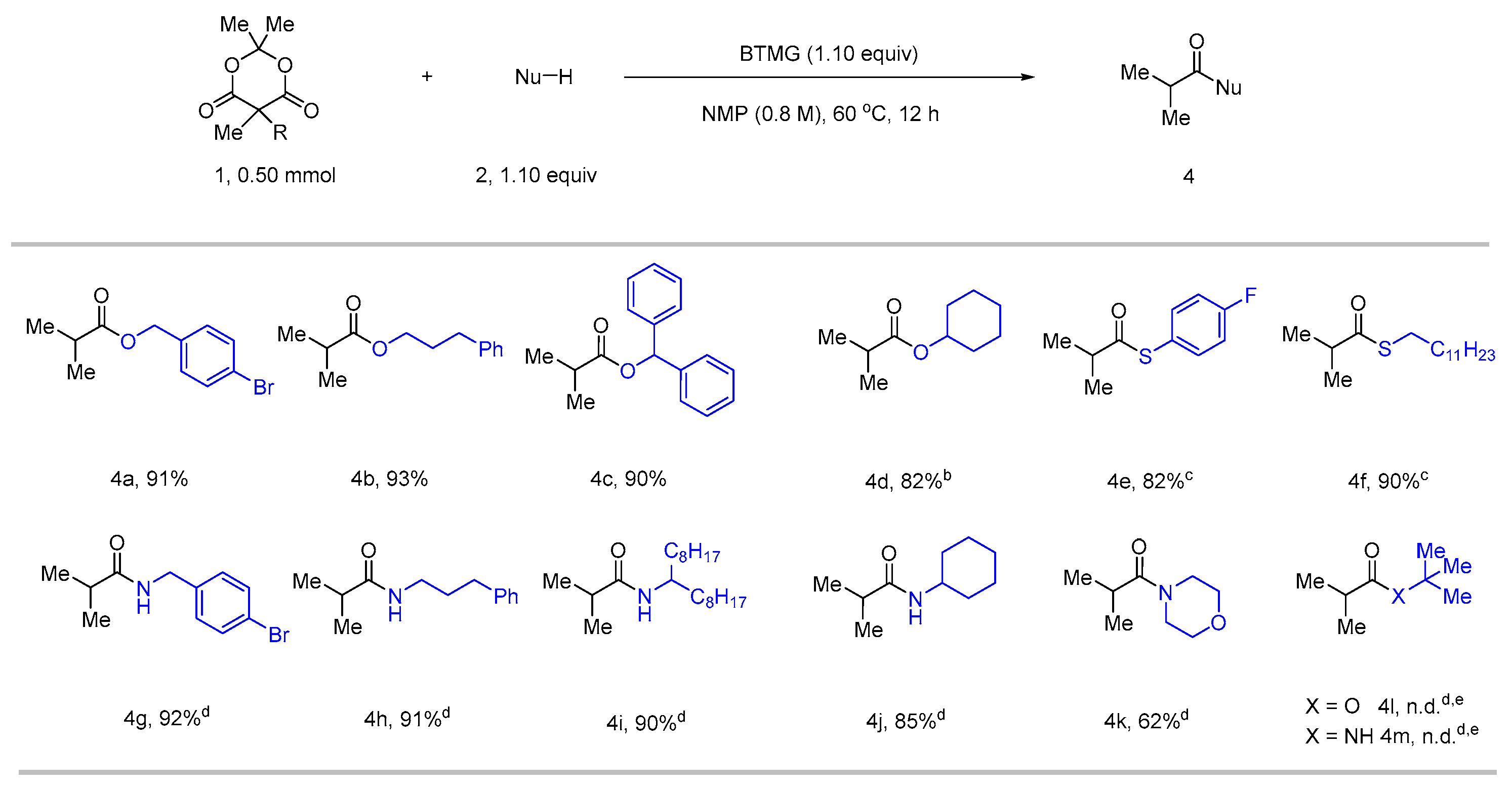
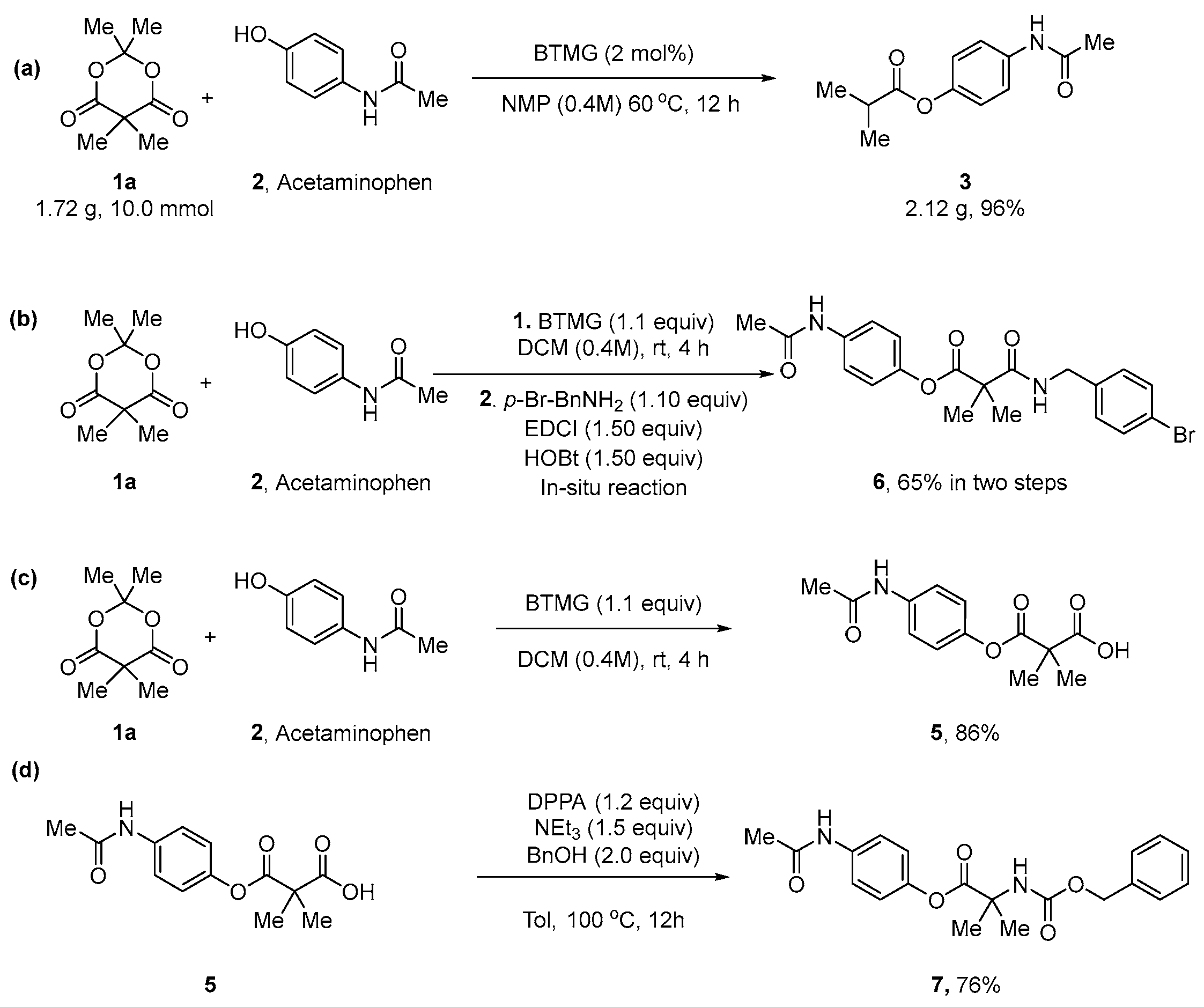
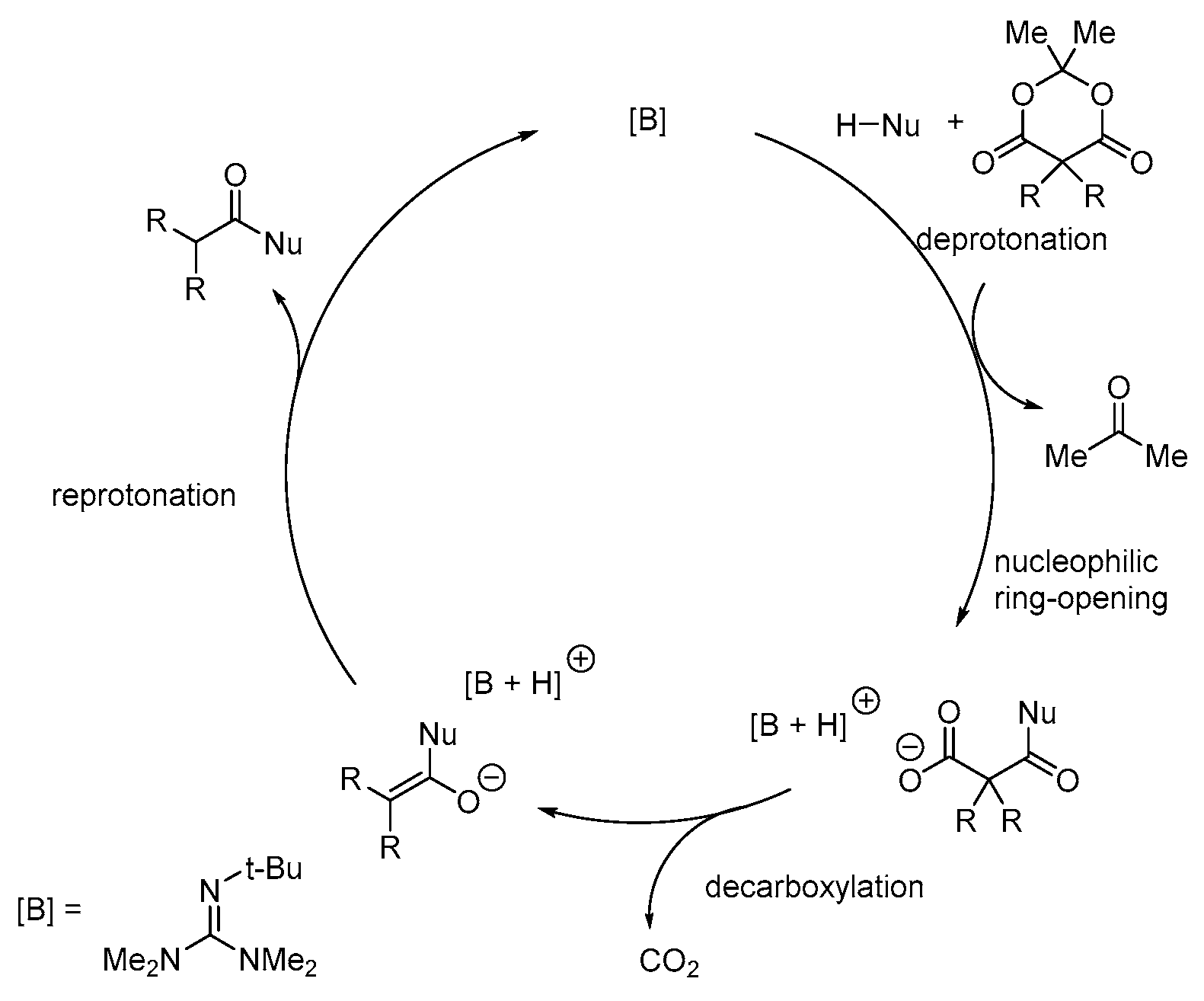
2. Results
3. Materials and Methods
3.1. Reagents and General Methods
3.2. Synthetic Procedures
3.2.1. A General Procedure for the 5-Substituted-5-methyl-1,3-dioxane-4,6-dione
3.2.2. A General Procedure for the Preparation of the Esters 3
3.2.3. A General Procedure for Alcohols, Thiol, Thiophenol, and Amines
3.2.4. Gram-Scale Preparation of 4-Acetamidophenyl Isobutyrate (3z)
3.2.5. Synthesis of 4-Acetamidophenyl 2-(((benzyloxy)carbonyl)amino)-2-Methylpropanoate (6)
3.2.6. One-Pot Preparation of 4-Acetamidophenyl 3-((4-bromobenzyl)amino)-2,2-dimethyl-3-oxopropanoate (7)
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Staudinger, H.; Meyer, J. Über neue organische Phosphorverbindungen III. Phosphinmethylenderivate und Phosphinimine. Helv. Chim. Acta 1919, 2, 635–646. [Google Scholar] [CrossRef]
- Dong, J.J.; Krasnova, L.; Finn, M.G.; Sharpless, K.B. Sulfur(VI) Fluoride Exchange (SuFEx): Another Good Reaction for Click Chemistry. Angew. Chem. Int. Ed. 2014, 53, 9430–9448. [Google Scholar] [CrossRef]
- Barrow, A.S.; Smedley, C.J.; Zheng, Q.; Li, S.; Dong, J.; Moses, J.E. The growing applications of SuFEx click chemistry. Chem. Soc. Rev. 2019, 48, 4731–4758. [Google Scholar] [CrossRef]
- Lou, T.S.-B.; Willis, M.C. Sulfonyl fluorides as targets and substrates in the development of new synthetic methods. Nat. Rev. Chem. 2022, 6, 146–162. [Google Scholar] [CrossRef]
- Homer, J.A.; Xu, L.; Kayambu, N.; Zheng, Q.H.; Choi, E.J.; Kim, B.M.; Sharpless, K.B.; Zuilhof, H.; Dong, J.J.; Moses, J.E. Sulfur fluoride exchange. Nat. Rev. Methods Primers 2023, 3, 58. [Google Scholar] [CrossRef]
- Rojas, J.J.; Bull, J.A. Sulfonyl fluorides as novel click reagents: Unconventional reactivity of sulfonyl fluorides. Trends Chem. 2025, 7, 124–133. [Google Scholar] [CrossRef]
- Zheng, Q.H.; Dong, J.J.; Sharpless, K.B. Ethenesulfonyl Fluoride (ESF): An On-Water Procedure for the Kilogram-Scale Preparation. J. Org. Chem. 2016, 81, 11360–11362. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Zhou, T.; Zhang, E. A Method for Sulfur(VI) Fluoride Exchange (SuFEx) Reaction. CN 107266392 A, 20 October 2017. [Google Scholar]
- Zheng, Q.H.; Woehl, J.L.; Kitamura, S.; Santos-Martins, D.; Smedley, C.J.; Li, G.C.; Forli, S.; Moses, J.E.; Wolan, D.W.; Sharpless, K.B. SuFEx-enabled chemoproteomics platform. Proc. Natl. Acad. Sci. USA 2019, 116, 18808–18814. [Google Scholar] [CrossRef]
- Meng, G.Y.; Guo, T.J.; Ma, T.C.; Zhang, J.; Shen, Y.C.; Sharpless, K.B.; Dong, J.J. A Modular click chemistry libraries for functional screens using a diazotizing reagent. Nature 2019, 574, 86–89. [Google Scholar] [CrossRef]
- Zheng, M.-M.; Cai, L.; Ma, T.C.; Tan, H.-D.; Lai, X.Y.; Dong, J.J.; Xue, X.-S. Computational analysis of modular diazotransfer reactions for the development of predictive reactivity models and diazotransfer reagents. Nat. Synth. 2024, 3, 1507–1517. [Google Scholar] [CrossRef]
- Cao, L.; Yu, B.C.; Klauser, P.C.; Zhang, P.; Li, S.S.; Wang, L. SuFEx Click Chemistry in Peptide Synthesis. Angew. Chem. Int. Ed. 2024, 63, e202412843. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.; Woroch, C.P.; Cleary, L.; Rusznak, M.; Franzese, R.W.; Reese, M.R.; Tucker, J.W.; Humphrey, J.M.; Etuk, S.M.; Kwan, S.C.; et al. Sulfonamide Synthesis via Calcium Triflimide Activation of Sulfonyl Fluorides. Org. Lett. 2018, 20, 3943–3947. [Google Scholar] [CrossRef]
- Mahapatra, S.; Woroch, C.P.; Butler, T.W.; Carneiro, S.N.; Kwan, S.C.; Khasnavis, S.R.; Gu, J.; Dutra, J.K.; Vetelino, B.C.; Bellenger, J.; et al. Unified Strategy to Access Sulfamides, Sulfamates and Sulfonamides from Sulfur(VI) Fluorides. Org. Lett. 2020, 22, 4389–4394. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Zhang, L.; Zheng, Q.H.; Zhou, F.; Klivansky, L.M.; Lu, J.M.; Liu, Y.; Dong, J.J.; Wu, P.; Sharpless, K.B. Bifluoride-catalysed sulfur(VI) fluoride exchange reaction for the synthesis of polysulfates and polysulfonates. Nat. Chem. 2017, 9, 1083–1088. [Google Scholar] [CrossRef]
- Wei, M.J.; Liang, D.C.; Cao, X.H.; Luo, W.J.; Ma, G.J.; Liu, Z.Y.; Li, L. A Broad-Spectrum Catalytic Amidation of Sulfonyl Fluorides and Fluorosulfates. Angew. Chem. Int. Ed. 2021, 60, 7397–7404. [Google Scholar] [CrossRef]
- Smedley, C.J.; Homer, J.A.; Gialelis, T.L.; Barrow, A.S.; Koelln, R.A.; Moses, J.E. Accelerated SuFEx Click Chemistry For Modular Synthesis. Angew. Chem. Int. Ed. 2022, 61, e202112375. [Google Scholar] [CrossRef]
- Lin, M.Z.; Luo, J.Y.; Xie, Y.; Du, G.F.; Cai, Z.H.; Dai, B.; He, L. SuFEx Reactions of Sulfonyl Fluorides, Fluorosulfates, and Sulfamoyl Fluorides Catalyzed by N-Heterocyclic Carbenes. ACS Catal. 2023, 13, 14503–14512. [Google Scholar] [CrossRef]
- Yassa, T.D.; Fang, Y.X.; Ravelo, L.K.; Anand, S.; Arora, S.; Ball, N.D. Lewis Acid-Catalyzed Sulfur Fluoride Exchange. Org. Lett. 2024, 26, 9897–9902. [Google Scholar] [CrossRef]
- Homer, J.A.; Koelln, R.A.; Barrow, A.S.; Gialelis, T.L.; Boiarska, Z.; Steinohrt, N.S.; Lee, E.F.; Yang, W.H.; Johnson, R.M.; Chung, T.M.; et al. Modular synthesis of functional libraries by accelerated SuFEx click chemistry. Chem. Sci. 2024, 15, 3879–3892. [Google Scholar] [CrossRef]
- Narayanan, A.; Jones, L.H. Sulfonyl fluorides as privileged warheads in chemical biology. Chem. Sci. 2015, 6, 2650–2659. [Google Scholar] [CrossRef] [PubMed]
- Baranczak, A.; Liu, Y.; Connelly, S.; Du, W.G.H.; Greiner, E.R.; Genereux, J.C.; Wiseman, R.L.; Eisele, Y.S.; Bradbury, N.C.; Dong, J.J.; et al. A Fluorogenic Aryl Fluorosulfate for Intraorganellar Transthyretin Imaging in Living Cells and in Caenorhabditis elegans. J. Am. Chem. Soc. 2015, 137, 7404–7414. [Google Scholar] [CrossRef]
- Chen, W.T.; Dong, J.J.; Plate, L.; Mortenson, D.E.; Brighty, G.J.; Li, S.H.; Liu, Y.; Galmozzi, A.; Lee, P.S.; Hulce, J.J.; et al. Arylfluorosulfates Inactivate Intracellular Lipid Binding Protein(s) through Chemoselective SuFEx Reaction with a Binding Site Tyr Residue. J. Am. Chem. Soc. 2016, 138, 7353–7364. [Google Scholar] [CrossRef]
- Chen, W.; Dong, J.; Li, S.; Liu, Y.; Wang, Y.; Yoon, L.; Wu, P.; Sharpless, K.B.; Kelly, J.W. Synthesis of Sulfotyrosine-Containing Peptides by Incorporating Fluorosulfated Tyrosine Using an Fmoc-Based Solid-Phase Strategy. Angew. Chem. Int. Ed. 2016, 55, 1835–1838. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.X.; Yang, B.; Fu, C.Y.; Zhu, H.; Zheng, F.; Kobayashi, T.; Liu, J.; Li, S.S.; Ma, C.; Wang, P.G.; et al. Genetically Encoding Fluorosulfate-L-tyrosine To React with Lysine, Histidine, and Tyrosine via SuFEx in Proteins in Vivo. J. Am. Chem. Soc. 2018, 140, 4995–4999. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wang, H.; Li, S.; Bare, G.A.L.; Chen, X.; Wang, C.; Moses, J.E.; Wu, P.; Sharpless, K.B. Biocompatible SuFEx Click Chemistry: Thionyl Tetrafluoride (SOF4)-Derived Connective Hubs for Bioconjugation to DNA and Proteins. Angew. Chem. Int. Ed. 2019, 58, 8029–8033. [Google Scholar] [CrossRef]
- Yang, L.Y.; Yuan, Z.N.; Li, Y.K.; Yang, S.Y.; Yu, B.C. Sulfur (VI) fluoride exchange (SuFEx): A versatile tool to profile protein-biomolecule interactions for therapeutic development. Med. Chem. Res. 2024, 33, 1315–1329. [Google Scholar] [CrossRef]
- Mortenson, D.E.; Brighty, G.J.; Plate, L.; Bare, G.; Chen, W.; Li, S.; Wang, H.; Cravatt, B.F.; Forli, S.; Powers, E.T.; et al. “Inverse Drug Discovery” Strategy To Identify Proteins That Are Targeted by Latent Electrophiles As Exemplified by Aryl Fluorosulfates. J. Am. Chem. Soc. 2017, 140, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.L.; Li, J.; Li, S.H.; Li, G.C.; Sharpless, K.B.; Wu, P. SuFEx Click Chemistry Enabled Late-Stage Drug Functionalization. J. Am. Chem. Soc. 2018, 140, 2919–2925. [Google Scholar] [CrossRef]
- Martín-Gago, P.; Olsen, C.A. Arylfluorosulfate-Based Electrophiles for Covalent Protein Labeling: A New Addition to the Arsenal. Angew. Chem. Int. Ed. 2019, 58, 957–966. [Google Scholar] [CrossRef]
- Xu, H.T.; Ma, F.; Wang, N.; Hou, W.; Xiong, H.; Lu, F.P.; Li, J.; Wang, S.Y.; Ma, P.X.; Yang, G.; et al. DNA-Encoded Libraries: Aryl Fluorosulfonates as Versatile Electrophiles Enabling Facile On-DNA Suzuki, Sonogashira, and Buchwald Reactions. Adv. Sci. 2019, 6, 1901551. [Google Scholar] [CrossRef]
- Smedley, C.J.; Li, G.C.; Barrow, A.S.; Gialelis, T.L.; Giel, M.C.; Ottonello, A.; Cheng, Y.F.; Kitamura, S.; Wolan, D.W.; Sharpless, K.B.; et al. Diversity Oriented Clicking (DOC): Divergent Synthesis of SuFExable Pharmacophores from 2-Substituted-Alkynyl-1-Sulfonyl Fluoride (SASF) Hubs. Angew. Chem. Int. Ed. 2020, 59, 12460–12469. [Google Scholar] [CrossRef]
- Kitamura, S.; Zheng, Q.H.; Woehl, J.L.; Solania, A.; Chen, E.; Dillon, N.; Hull, M.V.; Kotaniguchi, M.; Cappiello, J.R.; Kitamura, S.; et al. Sulfur(VI) Fluoride Exchange (SuFEx)-Enabled High-Throughput Medicinal Chemistry. J. Am. Chem. Soc. 2020, 142, 10899–10904. [Google Scholar] [CrossRef]
- Kline, G.M.; Nugroho, K.; Kelly, J.W. Inverse Drug Discovery identifies weak electrophiles affording protein conjugates. Curr. Opin. Chem. Biol. 2022, 67, 102113. [Google Scholar] [CrossRef]
- Liu, Z.W.; Li, J.; Li, S.H.; Li, G.C.; Sharpless, K.B.; Wu, P. Sulfur-fluoride exchange (SuFEx)-enabled lead discovery of AChE inhibitors by fragment linking strategies. Eur. J. Med. Chem. 2023, 257, 115502. [Google Scholar] [CrossRef]
- Huang, H.; Jones, L.H. Covalent drug discovery using sulfur(VI) fluoride exchange warheads. Expert Opin. Drug Discov. 2023, 18, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.S.; Qin, R.Z.; Rakesh, K.P.; Sharath, K.K.S.; Qin, H.L. Chemical and biology of sulfur fluoride exchange (SuFEx) click chemistry for drug discovery. Bioorg. Chem. 2023, 130, 106227. [Google Scholar] [CrossRef]
- Du, S.Q.; Hu, X.P.; Lindsley, C.W.; Zhan, P. New Applications of Sulfonyl Fluorides: A Microcosm of the Deep Integration of Chemistry and Biology in Drug Design. J. Med. Chem. 2024, 67, 16925–16927. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.J.; Sharpless, K.B.; Kwisnek, L.; Oakdale, J.S.; Fokin, V.V. SuFEx-Based Synthesis of Polysulfates. Angew. Chem. Int. Ed. 2014, 53, 9466–9470. [Google Scholar] [CrossRef]
- Yatvin, J.; Brooks, K.; Locklin, J. SuFEx on the Surface: A Flexible Platform for Postpolymerization Modification of Polymer Brushes. Angew. Chem. Int. Ed. 2015, 54, 13370–13373. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, F.; Ren, G.R.; Zheng, Q.H.; Chen, H.L.; Gao, B.; Klivansky, L.; Liu, Y.; Wu, B.; Xu, Q.F.; et al. SuFEx-Based Polysulfonate Formation from Ethenesulfonyl Fluoride–Amine Adducts. Angew. Chem. Int. Ed. 2017, 56, 11203–11208. [Google Scholar] [CrossRef]
- Hmissa, T.; Zhang, X.F.; Dhumal, N.R.; McManus, G.J.; Zhou, X.; Nulwala, H.B.; Mirjafari, A. Autocatalytic Synthesis of Bifluoride Ionic Liquids by SuFEx Click Chemistry. Angew. Chem. Int. Ed. 2018, 57, 16005–16009. [Google Scholar] [CrossRef] [PubMed]
- Gahtory, D.; Sen, R.; Pujari, S.; Li, S.H.; Zheng, Q.H.; Moses, J.E.; Sharpless, K.B.; Zuilhof, H. Quantitative and Orthogonal Formation and Reactivity of SuFEx Platforms. Chem. Eur. J. 2018, 24, 10550–10556. [Google Scholar] [CrossRef]
- Yang, C.J.; Flynn, J.P.; Niu, J. Facile Synthesis of Sequence-Regulated Synthetic Polymers Using Orthogonal SuFEx and CuAAC Click Reactions. Angew. Chem. Int. Ed. 2018, 57, 16194–16199. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Song, H.; Ko, N.; Kim, C.; Kim, K.; Lee, E. SuFEx in Metal−Organic Frameworks: Versatile Postsynthetic Modification Tool. ACS Appl. Mater. Interfaces 2018, 10, 33785–33789. [Google Scholar] [CrossRef]
- Demay-Drouhard, P.; Du, K.; Samanta, K.; Wan, X.T.; Yang, W.W.; Srinivasan, R.; Sue, A.C.H.; Zuilhof, H. Functionalization at Will of Rim-Differentiated Pillar[5]arenes. Org. Lett. 2019, 21, 3976–3980. [Google Scholar] [CrossRef]
- Kulow, W.; Wu, J.W.; Kim, C.; Michaudel, Q. Synthesis of unsymmetrical sulfamides and polysulfamides via SuFEx click chemistry. Chem. Sci. 2020, 11, 7807–7812. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Lu, K.Y.; Wang, L.; Hao, Q.; Liu, J.R.; Wang, Y.; Wu, Z.Q.; Chen, H. Introducing SuFEx click chemistry into aliphatic polycarbonates: A novel toolbox/platform for post-modification as biomaterials. Mater. Chem. B 2022, 10, 5203–5210. [Google Scholar] [CrossRef]
- Wang, M.L.; Hou, J.M.; Do, H.; Wang, C.; Zhang, X.H.; Du, Y.; Dong, Q.X.; Wang, L.J.; Ni, K.; Ren, F.Z.; et al. Intramolecular chalcogen bonding activated SuFEx click chemistry for efficient organic-inorganic linking. Nat. Commun. 2024, 15, 6849. [Google Scholar] [CrossRef]
- Li, S.H.; Li, G.C.; Gao, B.; Pujari, S.P.; Chen, X.Y.; Kim, H.; Zhou, F.; Klivansky, L.M.; Liu, Y.; Driss, H.; et al. SuFExable polymers with helical structures derived from thionyl tetrafluoride. Nat. Chem. 2021, 13, 858–867. [Google Scholar] [CrossRef]
- Kim, H.; Zhao, J.Y.; Bae, J.; Klivansky, L.M.; Dailing, E.A.; Liu, Y.; Cappiello, J.R.; Sharpless, K.B.; Wu, P. Chain-Growth Sulfur(VI) Fluoride Exchange Polycondensation: Molecular Weight Control and Synthesis of Degradable Polysulfates. ACS Cent. Sci. 2021, 7, 1919–1928. [Google Scholar] [CrossRef]
- Kim, M.P.; Sahoo, M.K.; Chun, J.-H.; Hong, S.Y. The First Decade of SuFEx Chemistry: Advancements in SuFEx Polymerization, Non-Canonical SuFEx Reactions, and SuFEx Radiochemistry. Synthesis 2025, 57, 1551–1568. [Google Scholar] [CrossRef]
- Qin, H.L.; Zheng, Q.H.; Bare, G.A.L.; Wu, P.; Sharpless, K.B. A Heck–Matsuda Process for the Synthesis of β-Arylethenesulfonyl Fluorides: Selectively Addressable Bis-electrophiles for SuFEx Click Chemistry. Angew. Chem. Int. Ed. 2016, 55, 14155–14158. [Google Scholar] [CrossRef] [PubMed]
- Li, S.H.; Wu, P.; Moses, J.E.; Sharpless, K.B. Multidimensional SuFEx Click Chemistry: Sequential Sulfur(VI) Fluoride Exchange Connections of Diverse Modules Launched From An SOF4 Hub. Angew. Chem. Int. Ed. 2017, 56, 2903–2908. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Li, S.H.; Wu, P.; Moses, J.E.; Sharpless, K.B. SuFEx Chemistry of Thionyl Tetrafluoride (SOF4) with Organolithium Nucleophiles: Synthesis of Sulfonimidoyl Fluorides, Sulfoximines, Sulfonimidamides, and Sulfonimidates. Angew. Chem. Int. Ed. 2018, 57, 1939–1943. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.J.; Meng, G.Y.; Zhan, X.J.; Yang, Q.; Ma, T.C.; Xu, L.; Sharpless, K.B.; Dong, J.J. A New Portal to SuFEx Click Chemistry: A Stable Fluorosulfuryl Imidazolium Salt Emerging as an “F@SO2+” Donor of Unprecedented Reactivity, Selectivity, and Scope. Angew. Chem. Int. Ed. 2018, 57, 2605–2610. [Google Scholar] [CrossRef]
- Meng, Y.P.; Wang, S.M.; Fang, W.Y.; Xie, Z.Z.; Leng, J.; Alsulami, H.; Qin, H.L. Ethenesulfonyl Fluoride (ESF) and Its Derivatives in SuFEx Click Chemistry and More. Synthesis 2020, 52, 673–687. [Google Scholar] [CrossRef]
- Liang, D.D.; Streefkerk, D.E.; Jordaan, D.; Wagemakers, J.; Baggerman, J.; Zuilhof, H. Silicon-Free SuFEx Reactions of Sulfonimidoyl Fluorides: Scope, Enantioselectivity, and Mechanism. Angew. Chem. Int. Ed. 2020, 59, 7494–7500. [Google Scholar] [CrossRef]
- Nie, X.L.; Xu, T.X.; Hong, Y.H.; Zhang, H.H.; Mao, C.X.; Liao, S.H. Introducing A New Class of Sulfonyl Fluoride Hubs via Radical Chloro-Fluorosulfonylation of Alkynes. Angew. Chem. Int. Ed. 2021, 60, 22035–22042. [Google Scholar] [CrossRef]
- Zeng, D.M.; Ma, Y.H.; Deng, W.P.; Wang, M.; Jiang, X.F. The Linkage of Sulfonimidoyl Fluorides and Unactivated Alkenes via Hydrosulfonimidoylation. Angew. Chem. Int. Ed. 2022, 61, e202207100. [Google Scholar] [CrossRef]
- Zeng, D.M.; Deng, W.P.; Jiang, X.F. Advances in the construction of diverse SuFEx linkers. Natl. Sci. Rev. 2023, 10, nwad123. [Google Scholar] [CrossRef]
- Erchinger, J.E.; Hoogesteger, R.; Laskar, R.; Dutta, S.; Huempel, C.; Rana, D.; Daniliuc, C.G.; Glorius, F. EnT-Mediated N−S Bond Homolysis of a Bifunctional Reagent Leading to Aliphatic Sulfonyl Fluorides. J. Am. Chem. Soc. 2023, 145, 2364–2374. [Google Scholar] [CrossRef]
- Aleksandrenko, S.; Dobrydnev, A.V.; Zhersh, S.; Grygorenko, O.O. Saturated Heterocyclic Sulfamoyl Fluorides: Building Blocks for the SuFEx Chemistry. Eur. J. Org. Chem. 2024, 27, e202400611. [Google Scholar] [CrossRef]
- Li, H.Y.; Wang, Y. Recent Advances in SuFEx Chemistry. Synthesis 2025, 57, 1690–1706. [Google Scholar]
- Serbetci, D.; Marraffa, L.; Natho, P.; Andresini, M.; Luisi, R. A Practical Guide to SuFEx Chemistry: An Overview of S(VI)-SuFEx Linkers and Their Reactivity. Synthesis 2025, 57, 1569–1582. [Google Scholar] [CrossRef]
- Chao, Y.; Krishna, A.; Subramaniam, M.; Liang, D.D.; Pujari, S.P.; Sue, A.C.H.G.; Li, G.N.; Miloserdov, F.M.; Zuilhof, H. Sulfur–Phenolate Exchange: SuFEx-Derived Dynamic Covalent Reactions and Degradation of SuFEx Polymers. Angew. Chem. Int. Ed. 2022, 61, e202207456. [Google Scholar] [CrossRef] [PubMed]
- Boom, A.F.J.; Subramaniam, M.; Zuilhof, H. Sulfur-Phenolate Exchange As a Fluorine-Free Approach to S(VI) Exchange Chemistry on Sulfonyl Moieties. Org. Lett. 2022, 24, 8621–8626. [Google Scholar] [CrossRef] [PubMed]
- Boom, A.F.J.; Zuilhof, H. Sulfur-Phenolate Exchange as a Mild, Fast, and High-Yielding Method toward the Synthesis of Sulfonamides. Org. Lett. 2023, 25, 788–793. [Google Scholar] [CrossRef] [PubMed]
- Chao, Y.; Subramaniam, M.; Namitharan, K.; Zhu, Y.M.; Koolma, V.; Hao, Z.T.; Li, S.K.; Wang, Y.X.; Hudoynazarov, I.; Miloserdov, F.M.; et al. Synthesis of Large Macrocycles with Chiral Sulfur Centers via Enantiospecific SuFEx and SuPhenEx Click Reactions. J. Org. Chem. 2023, 88, 15658–15665. [Google Scholar] [CrossRef]
- Sun, S.J.; Homer, J.A.; Smedley, C.J.; Cheng, Q.-Q.; Sharpless, K.B.; Moses, J.E. Phosphorus fluoride exchange: Multidimensional catalytic click chemistry from phosphorus connective hubs. Chem 2023, 9, 2128–2143. [Google Scholar] [CrossRef]
- Cao, L.; Yu, B.C.; Li, S.S.; Zhang, P.; Li, Q.K.; Wang, L. Genetically enabling phosphorus fluoride exchange click chemistry in proteins. Chem 2024, 10, 1868–1884. [Google Scholar] [CrossRef]
- Homer, J.A.; Sun, S.J.; Koelln, R.A.; Moses, J.E. Protocol for producing phosphoramidate using phosphorus fluoride exchange click chemistry. STAR Protoc. 2024, 5, 102824. [Google Scholar] [CrossRef]
- Wang, Z.F.; Vishwakarma, D.S.; Sun, S.J.; Huang, Q.Y.; Pati, S.; Johnson, R.M.; Rufrano, R.M.; Gembicky, M.J.; Homer, J.A.; Moses, J.E. Phosphorus Fluoride Exchange (PFEx) Click Chemistry: 2-Substituted-Alkynyl-1-Cyclotriphosphazene (SACP) Hubs for Diversity Oriented Clicking. Adv. Synth. Catal. 2025, 367, e202401573. [Google Scholar] [CrossRef]
- Arnett, E.M.; Harrelson, J.A., Jr. A Spectacular Example of the Importance of Rotational Barriers: The Ionization of Meldrum’s Acid. J. Am. Chem. Soc. 1987, 109, 809–812. [Google Scholar] [CrossRef]
- Zhang, X.M.; Bordwell, F.G. Acidities and Homolytic Bond Dissociation Enthalpies (BDEs) of the Acidic H-A Bonds in Acyclic and Cyclic Alkoxycarbonyl Compounds (Esters and Carbamates). J. Org. Chem. 1994, 59, 6456–6458. [Google Scholar] [CrossRef]
- Byun, K.; Mo, Y.; Gao, J. New Insight on the Origin of the Unusual Acidity of Meldrum’s Acid from ab Initio and Combined QM/MM Simulation Study. J. Am. Chem. Soc. 2001, 123, 3974–3979. [Google Scholar] [CrossRef]
- Nakamura, S.; Hirao, H.; Ohwada, T. Rationale for the Acidity of Meldrum’s Acid. Consistent Relation of C-H Acidities to the Properties of Localized Reactive Orbital. J. Org. Chem. 2004, 69, 4309–4316. [Google Scholar] [CrossRef]
- Dumas, A.M.; Fillion, E. Meldrum’s Acids and 5-Alkylidene Meldrum’s Acids in Catalytic Carbon-Carbon Bond-Forming Processes. Acc. Chem. Res. 2010, 43, 440–454. [Google Scholar] [CrossRef]
- Chen, B.-C. Meldrum’s Acid in Orgnaic Synthesis. Heterocycles 1991, 32, 529–597. [Google Scholar] [CrossRef]
- Brosge, F.; Singh, P.; Almqvist, F.; Bolm, C. Selected applications of Meldrum’s acid – a tutorial. Org. Biomol. Chem. 2021, 19, 5014–5027. [Google Scholar] [CrossRef]
- Hiratake, J.; Shibata, K.; Baba, N.; Oda, J. Enantiotopic-Group Differentiation. Asymmetric Monoesterification of Malonic Acids Using Cinchona Alkaloid Derivatives. Synthesis 1988, 4, 278–280. [Google Scholar] [CrossRef]
- Korotkov, V.S.; Larionov, O.V.; Hofmeister, A.; Magull, J.; Meijere, A.D. GaCl3-Catalyzed Insertion of Diazene Derivatives into the Cyclopropane Ring. J. Org. Chem. 2007, 72, 7504–7510. [Google Scholar] [CrossRef]
- Lau, K.-N.; Chow, H.-F.; Chan, M.-C.; Wong, K.-W. Dendronized Polymer Organogels from Click Chemistry: A Remarkable Gelation Property Owing to Synergistic Functional-Group Binding and Dendritic Size Effects. Angew. Chem. Int. Ed. 2008, 47, 6912–6916. [Google Scholar] [CrossRef]
- Chang, T.T.; More, S.V.; Lu, I.-H.; Hsu, J.-C.; Chen, T.-J.; Jen, Y.C.; Lu, C.-K.; Li, W.-S. Isomalyngamide A, A-1 and their analogs suppress cancer cell migration in vitro. Eur. J. Med. Chem. 2011, 46, 3810–3819. [Google Scholar] [CrossRef]
- Choi, L.-S.; Chow, H.-F. Vetteerrsatile Synthesis of Amphiphilic Oligo(Aliphatic-Glycerol) Layer-Block Dendrons with Different Hydrophilic-Lipophilic Balance Values. Synlett 2013, 24, 201–206. [Google Scholar] [CrossRef]
- Ariyarathna, Y.; Tunge, J.A. Decarboxylative allylations of ester enolate equivalents. Org. Biomol. Chem. 2014, 12, 8386–8389. [Google Scholar] [CrossRef] [PubMed]
- Ambrogio, I.; Cacchi, S.; Fabrizi, G.; Goggiamani, A.; Iazzetti, A. Palladium-Catalyzed Nucleophilic Substitution of Propargylic Carbonates and Meldrum’s Acid Derivatives. Eur. J. Org. Chem. 2015, 14, 3147–3151. [Google Scholar] [CrossRef]
- Sui, X.L.; Gutekunst, W.R. Cascade Alternating Metathesis Cyclopolymerization of Diynes and Dihydrofuran. ACS Macro Lett. 2022, 11, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Ronzon, Q.; Zhang, W.; Charote, T.; Casaretto, N.; Frison, G.; Nay, B. Total Synthesis of (+)-Cinereain and (-)-Janoxepin through a Fragment Coupling/Retro-Claisen Rearrangement Cascade. Angew. Chem. Int. Ed. 2022, 61, e202212855. [Google Scholar] [CrossRef]
- Ariyarathna, Y.; Tunge, J.A. Multicomponent decarboxylative allylations. Chem. Commun. 2014, 50, 14049–14052. [Google Scholar] [CrossRef] [PubMed]
- Legros, F.; Martzel, T.; Brière, J.-F.; Oudeyer, S.; Levacher, V. Organocatalytic Enantioselective Decarboxylative Protonation Reaction of Meldrum’s Acid Derivatives under PTC Conditions. Eur. J. Org. Chem. 2018, 17, 1975–1983. [Google Scholar] [CrossRef]
- Martzel, T.; Annibaletto, J.; Levacher, V.; Brière, J.F.; Oudeyer, S. C5-Disubstituted Meldrum’s Acid Derivatives as Platform for the Organocatalytic Synthesis of C3-Alkylated Dihydrocoumarins. Adv. Synth. Catal. 2019, 361, 995–1000. [Google Scholar] [CrossRef]
- Oikawa, Y.; Hirasawa, H.; Yonemitsu, O. Meldrum’s acid in organic synthesis. 1. A convenient one-pot synthesis of ethyl indolepropionates. Tetrahedron Lett. 1978, 20, 1759–1762. [Google Scholar] [CrossRef]
- Cheawchan, S.; Koyama, Y.; Uchida, S.; Takata, T. Catalyst-free click cascade functionalization of unsaturated-bondcontaining polymers using masked-ketene-tethering nitrile N-oxide. Polymer 2013, 54, 4501–4510. [Google Scholar] [CrossRef]
- Nakatani, K.; Natsuhara, N.; Mori, Y.; Mukherjee, S.; Das, B.; Murata, A. Synthesis of Naphthyridine Dimers with Conformational Restriction and Binding to DNA and RNA. Chem. Asian J. 2017, 12, 3077–3087. [Google Scholar] [CrossRef] [PubMed]
- Adamson, N.J.; Wilbur, K.C.E.; Malcolmson, S.J. Enantioselective Intermolecular Pd-Catalyzed Hydroalkylation of Acyclic 1,3-Dienes with Activated Pronucleophiles. J. Am. Chem. Soc. 2018, 140, 2761–2764. [Google Scholar] [CrossRef]
- Liu, M.; Zhao, H.Y.; Li, C.K. Rh(I)-catalyzed regio- and enantioselective allylic alkylation of Meldrum’s acid. Chin. Chem. Lett. 2021, 32, 385–388. [Google Scholar] [CrossRef]
- Tabti, R.; Lamoureux, F.; Charrier, C.; Ory, B.; Heymann, D.; Bentouhami, E.; Desaubry, L. Development of prohibitin ligands against osteoporosis. Eur. J. Med. Chem. 2021, 210, 112961. [Google Scholar] [CrossRef]
- Das, B.; Nagano, K.; Kawai, G.; Murata, A.; Nakatani, K. 2-Amino-1,8-naphthyridine Dimer (ANP77), a High-Affinity Binder to the Internal Loops of C/CC and T/CC Sites in Double-Stranded DNA. J. Org. Chem. 2022, 87, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.-W.; Han, M.; Shen, M.-H.; Zhu, C.-F.; Xu, H.-D. Synthesis of functionalized γ-lactams by a lewis acid catalyzed ketene formation/cyclization/claisen rearrangement sequence of 5,5-disubstituted Meldrum’s acids. Tetrahedron Lett. 2022, 99, 153816. [Google Scholar] [CrossRef]
- Avula, S.K.; Ullah, S.; Ebrahimi, A.; Rostami, A.; Halim, S.A.; Khan, A.; Anwar, M.U.; Gibbons, S.; Csuk, R.; Al-Harrasi, A. Dihydrofolate reductase inhibitory potential of 1H-indole-based-meldrum linked 1H-1,2,3-triazoles as new anticancer derivatives: In-vitro and in-silico studies. Eur. J. Med. Chem. 2025, 283, 117174. [Google Scholar] [CrossRef]
- Tite, T.; Sabbah, M.; Levacher, V.; Brière, J.-F. Organocatalysed decarboxylative protonation process from Meldrum’s acid: Enantioselective synthesis of isoxazolidinones. Chem. Commun. 2013, 49, 11569–11571. [Google Scholar] [CrossRef]
- Engl, O.D.; Saadi, J.; Cosimi, E.; Wennemers, H. Synthesis of Monothiomalonates – Versatile Thioester Enolate Equivalents for C–C Bond Formations. Helv. Chim. Acta 2017, 100, e1700196. [Google Scholar] [CrossRef]
- Brown, R.F.C.; Eastwood, F.W.; Harrington, K.J. Methyleneketenes and Methylenecarbenes. I Formation of Arylmethyleneketenes and Alkylideneketenes by Pyrolysis of Substituted 2,2-Dimethyl-1,3-dioxan-4,6-diones. Aust. J. Chem. 1974, 27, 2373–2384. [Google Scholar] [CrossRef]
- Baxter, G.J.; Brown, R.F.C.; Eastwood, F.W.; Harrington, K.J. Pyrolytic generation of carbonylcyclopropane (dimethylene ketene) and its dimerization to dispiro-[2,1,2,1]-octane-4,8-dione. Tetrahedron Lett. 1975, 16, 4283–4284. [Google Scholar] [CrossRef]
- Leibfarth, F.A.; Kang, M.; Ham, M.; Kim, J.; Campos, L.M.; Gupta, N.; Moon, B.; Hawker, C.J. A facile route to ketene-functionalized polymers for general materials applications. Nat. Chem. 2010, 2, 207–212. [Google Scholar] [CrossRef]
- Leibfarth, F.A.; Wolffs, M.; Campos, L.M.; Delany, K.; Treat, N.; Kade, M.J.; Moon, B.; Hawker, C.J. Low-temperature ketene formation in materials chemistry through molecular engineering. Chem. Sci. 2012, 3, 766–771. [Google Scholar] [CrossRef]
- Armarego, W.L.F.; Perrin, D.D. Purification of Laboratory Chemicals, 4th ed.; Butterworth-Heinemann: Oxford, UK, 1997. [Google Scholar]
- Sakakura, A.; Kawajiri, K.; Ohkubo, T.; Kosugi, Y.; Ishihara, K. Widely Useful DMAP-Catalyzed Esterification under Auxiliary Base- and Solvent-Free Conditions. J. Am. Chem. Soc. 2007, 129, 14775–14779. [Google Scholar] [CrossRef]
- Mensah, E.; Earl, L. Mild and Highly Efficient Copper(I) Inspired Acylation of Alcohols and Polyols. Catalysts 2017, 7, 33. [Google Scholar] [CrossRef]
- Yang, C.-H.; Fan, W.-W.; Liu, G.-Q.; Duan, L.; Li, L.; Li, Y.-M. On the Understanding of BF3·Et2O-Promoted Intra- and Intermolecular Amination and Oxygenation of Unfunctionalized Olefins. RSC Adv. 2015, 5, 61081–61093. [Google Scholar] [CrossRef]
- More, S.-V.; Chang, T.-T.; Chiao, Y.-P.; Jao, S.-C.; Lu, C.-K.; Li., W.-S. Glycosylation enhances the anti-migratory activities of isomalyngamide A analogs. Eur. J. Med. Chem. 2013, 64, 169–178. [Google Scholar] [CrossRef]
- Kawamoto, K.; Zhong, M.; Wang, R.; Olsen, B.D.; Johnson, J.A. Loops versus Branch Functionality in Model Click Hydrogels. Macromolecules 2015, 48, 8980–8988. [Google Scholar] [CrossRef]
- Li, J.-S.; Da, Y.-D.; Chen, G.-Q.; Yang, Q.; Li, Z.-W.; Yang, F.; Huang, P.-M. Solvent-, and Catalyst-Free Acylation of Anilines with Meldrum’s Acids: A Neat Access to Anilides. ChemistrySelect 2017, 2, 1770–1773. [Google Scholar] [CrossRef]
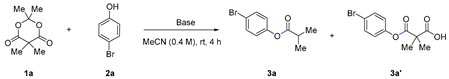 | ||||
|---|---|---|---|---|
| Entry | Base | Solvent | Time(h) | 3ab(%) |
| 1 | none | MeCN | 4 | 0 |
| 2 | Cs2CO3 | MeCN | 4 | 2 |
| 3 | K2CO3 | MeCN | 4 | 0 |
| 4 | K3PO4 | MeCN | 4 | 0 |
| 5 | Na2CO3 | MeCN | 4 | 0 |
| 6 | DMAP | MeCN | 4 | 0 |
| 7 | TEA | MeCN | 4 | 0 |
| 8 | DIPEA | MeCN | 4 | 2 |
| 9 | DBU | MeCN | 4 | 65 |
| 10 | BTMG | MeCN | 4 | 82 |
| 11 | BTMG | MeCN | 12 | 100 |
| 12 | BTMG | Dichloromethane | 12 | 87 |
| 13 | BTMG | Toluene | 12 | 100 |
| 14 | BTMG | Tetrahydrofuran | 12 | 100 |
| 15 | BTMG | N-methyl-pyrrolidinone | 12 | 100 |
| 16 | BTMG | Dimethyl sulfoxide | 12 | 99 |
| 17c | BTMG | Dimethyl sulfoxide | 1 | 87 |
| 18d | BTMG | Dimethyl sulfoxide | 1 | 80 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, B.; Wang, Z.; Peng, X.; Xie, J.; Sun, Z.; Li, L. Disubstituted Meldrum’s Acid: Another Scaffold with SuFEx-like Reactivity. Molecules 2025, 30, 3534. https://doi.org/10.3390/molecules30173534
Chen B, Wang Z, Peng X, Xie J, Sun Z, Li L. Disubstituted Meldrum’s Acid: Another Scaffold with SuFEx-like Reactivity. Molecules. 2025; 30(17):3534. https://doi.org/10.3390/molecules30173534
Chicago/Turabian StyleChen, Baoqi, Zhenguo Wang, Xiaole Peng, Jijun Xie, Zhixiu Sun, and Le Li. 2025. "Disubstituted Meldrum’s Acid: Another Scaffold with SuFEx-like Reactivity" Molecules 30, no. 17: 3534. https://doi.org/10.3390/molecules30173534
APA StyleChen, B., Wang, Z., Peng, X., Xie, J., Sun, Z., & Li, L. (2025). Disubstituted Meldrum’s Acid: Another Scaffold with SuFEx-like Reactivity. Molecules, 30(17), 3534. https://doi.org/10.3390/molecules30173534






