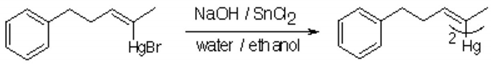
The general part of the experimental section [1] has been presented elsewhere. To a stirred solution of sodium hydroxide (20 eq.) in distilled water (4 ml) was addded stannous chloride (1.5 eq.) in distilled water (10 ml), A suspension of (Z)-5-Phenyl-2-penten-2-ylmercury bromide (2 mmol) in water (10 ml) and ethanol (10 ml) was then added over 1 hour, and the mixture was stirred at room temperature overnight, diluted with water (50 ml) and extracted with ether (3x40 ml). The combined ether extracts were dried (Na2SO4), filtered and evaporated under reduced pressure. Kugelrohr distillation gave the title compound in 85% yield.
B.p. 200°/0.1 mmHg.
UV (ethanol) 272(sh) (1234), 264sh (1728), 237sh (5266) nm.
IR (film) 3026, 2928, 2902(s), 2841, 1602(s), 1496, 1453(s), 842, 747(s), 698(s) cm-1.
1H-NMR (90 MHz, CDCl3) 1.83 (3H, bs, J199Hg,H 87 Hz, CH3), 2.10-2.88 (4H, m, 2xCH2), 6.51 (1H, bt, J 6.6 Hz, =CH), 6.82-7.42 (5H, m, ArH).
EI-MS 146(28%), 145(87), 144(51), 129(27), 117(22), 104(10), 92(18), 91(100), 77(10), 65(18).
Acknowledgment: The authors gratefully acknowledge financial support from the Australian Research Council and The University of Sydney.
References and Notes
- Moloney, M.G.; Pinhey, J.T.; Stoermer, M.J. "Vinyl Cation Formation by Decomposition of Vinyl-lead Triacetates. The reactions of Vinylmercury and Vinyltin Compounds with Lead Tetraacetate". J. Chem. Soc. Perkin Trans. 1 1990, 10, 2645. [Google Scholar] [CrossRef]
Sample Availability: No sample available. |
© 1998 MDPI. All rights reserved. Molecules website http://www.mdpi.org/molecules/
