The Tryptophan Metabolite Indole-3-Carboxaldehyde Alleviates Mice with DSS-Induced Ulcerative Colitis by Balancing Amino Acid Metabolism, Inhibiting Intestinal Inflammation, and Improving Intestinal Barrier Function
Abstract
1. Introduction
2. Results
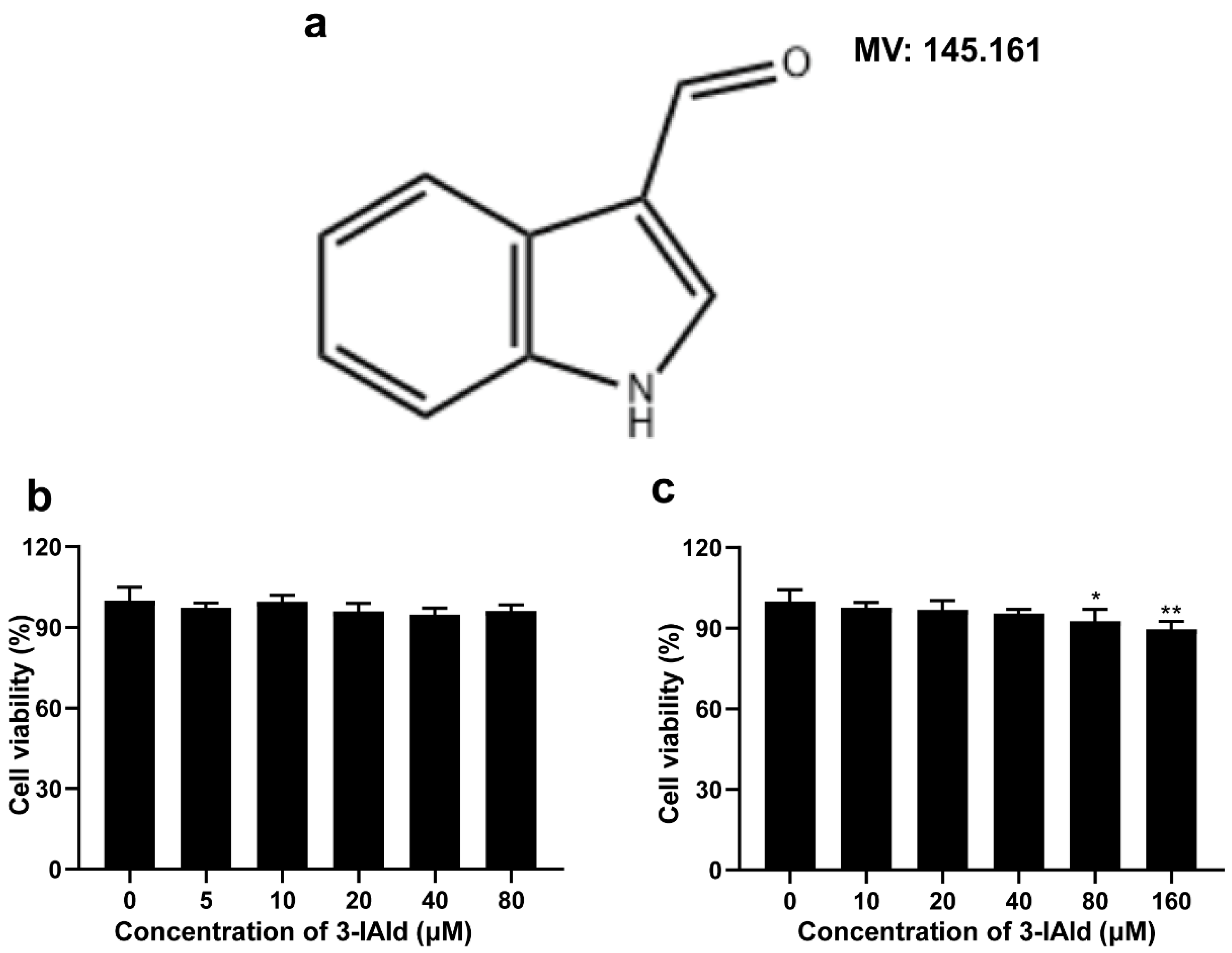
2.1. Structure of 3-Iald and Its Effect on Cell Viability of RAW264.7 and Caco2 Cells
2.2. 3-Iald Inhibited the Expression Levels of NO, TNF-α, IL-6, and IL-1β Induced by LPS in RAW264.7 Cells
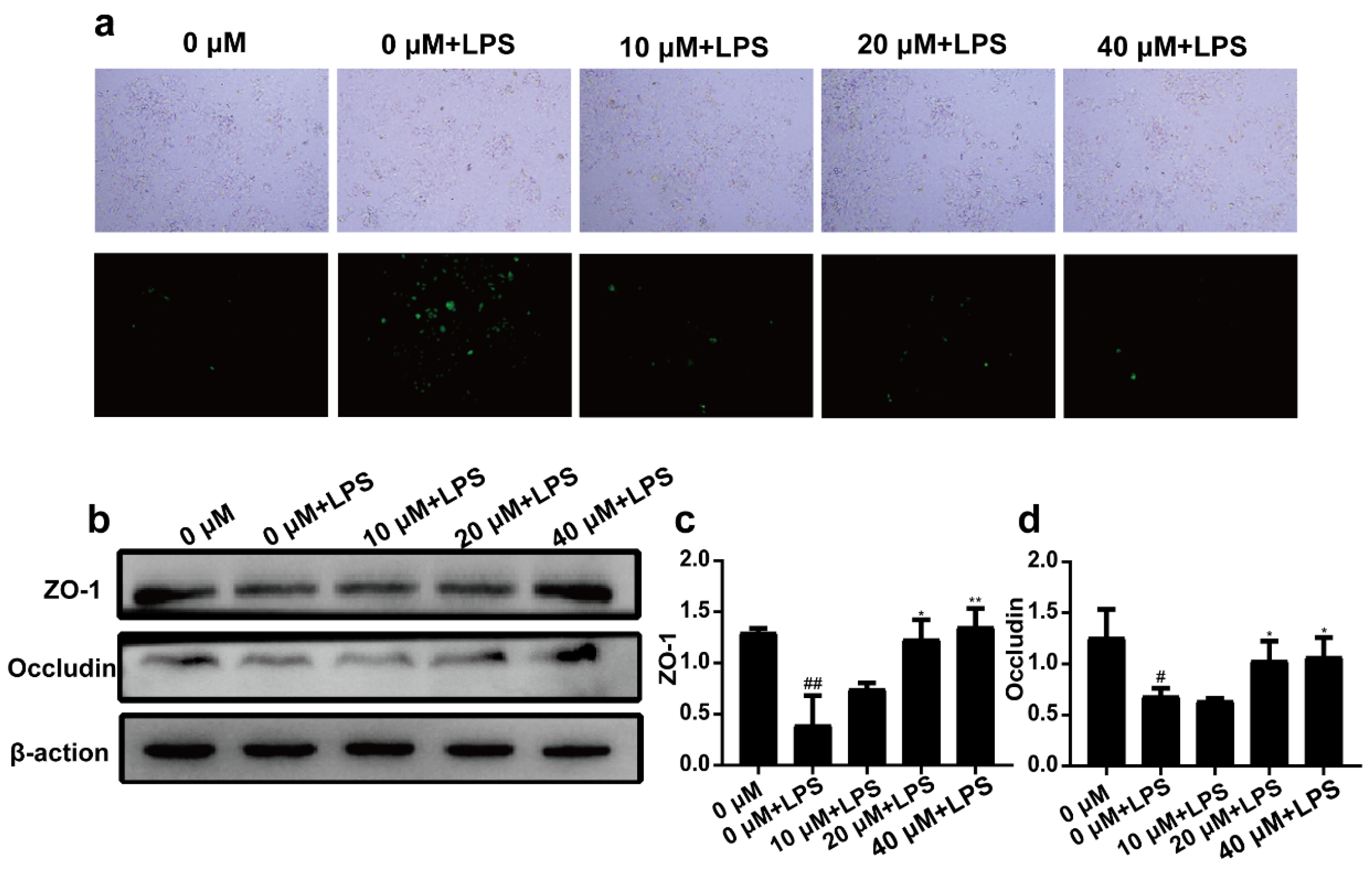
2.3. 3-Iald Reduced LPS Induced ROS Production and Up-Regulated the Expression of ZO-1 and Occludin
2.4. 3-Iald Improved the Shortening of Colon Length and Pathological Damage Caused by DSS in Mice
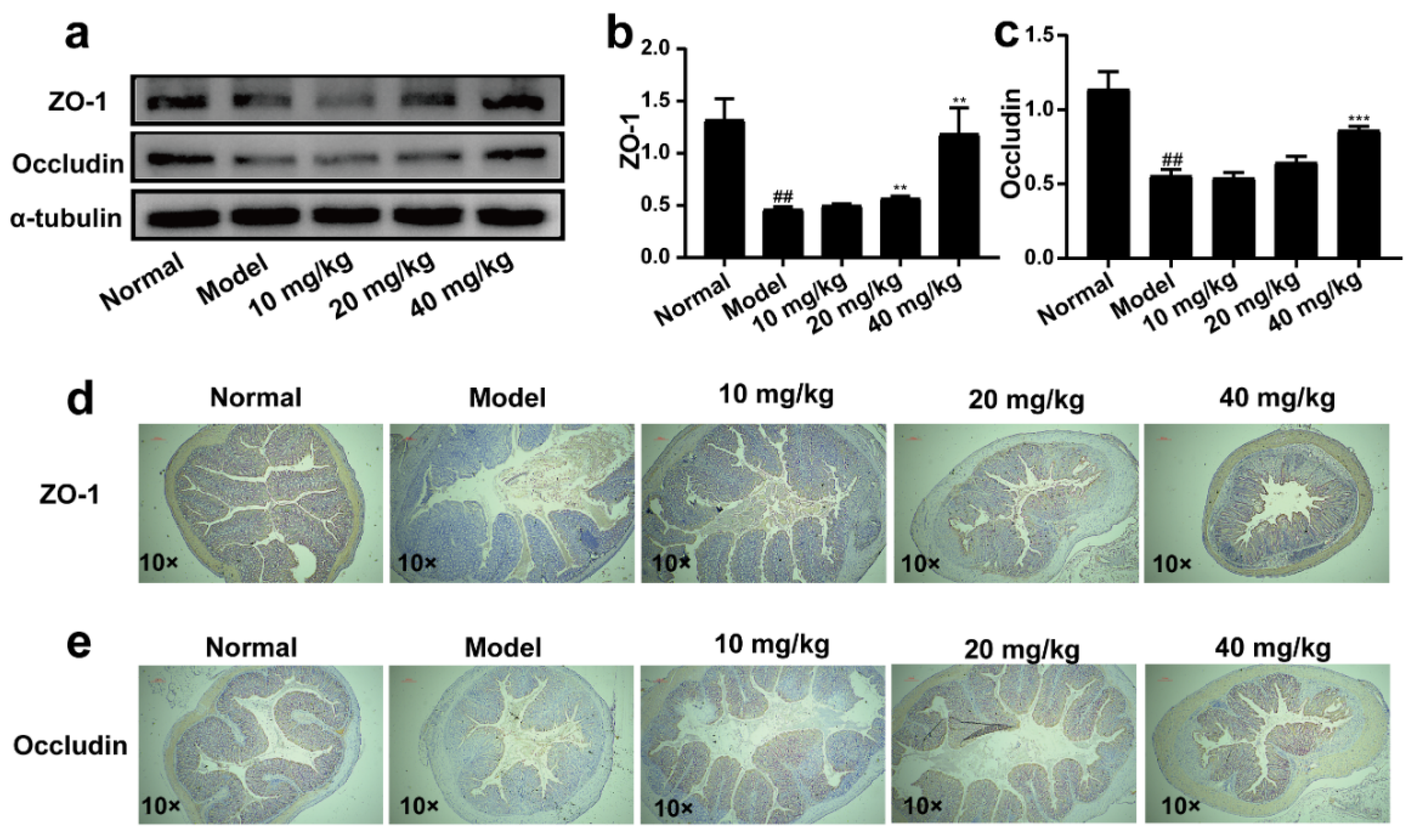
2.5. 3-IAld Alleviated the Disruption of the Colonic Barrier Caused by DSS in Mice
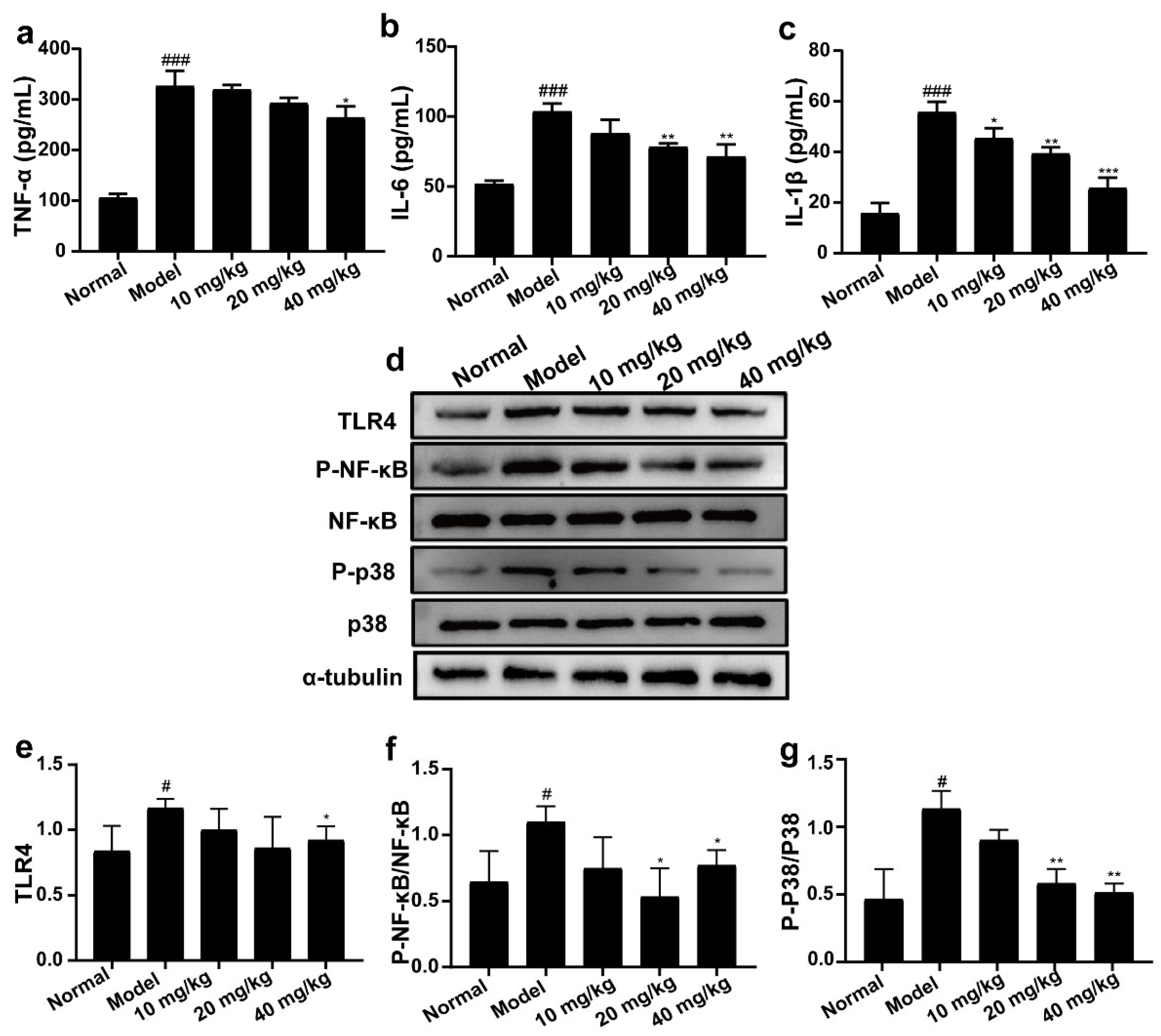
2.6. 3-IAld Inhibited the Expression Levels of TNF-α, IL-6, and IL-1β, and Down-Regulated the Expression Levels of TLR4, P-NF-κB, and P-P38 Proteins in DSS-Induced UC Mice
2.7. Effect of 3-IAld on Serum Metabolism in DSS-Induced C57BL/6 Mice
3. Discussion
4. Materials and Methods
4.1. Drugs and Reagents
4.2. Cell Lines and Cell Culture
4.3. Cell Viability Assessment
4.4. Enzyme-Linked Immunosorbent Assay (ELISA)
4.5. Intracellular ROS Assay
4.6. Western Blotting Assay
4.7. DSS Mouse Models
4.8. Hematoxylin and Eosin (H&E) Staining
4.9. Immunohistochemistry (IHC)
4.10. Analysis of The Effect of 3-IAld on Serum Metabolism in UC Mice Using the Non-Targeted Metabolomics Technique
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bantel, H.; Berg, C.; Vieth, M.; Stolte, M.; Kruis, W.; Schulze-Osthoff, K. Mesalazine inhibits activation of transcription factor NF-kappaB in inflamed mucosa of patients with ulcerative colitis. Am. J. Gastroenterol. 2000, 95, 3452–3457. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, G.G. The global burden of IBD: From 2015 to 2025. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Bai, G.; Tang, L.; Liu, L.; Li, Y.; Jiang, W. Changes in MMP-2, MMP-9, inflammation, blood coagulation and intestinal mucosal permeability in patients with active ulcerative colitis. Exp. Ther. Med. 2020, 20, 269–274. [Google Scholar] [CrossRef]
- Diesing, A.K.; Nossol, C.; Danicke, S.; Walk, N.; Post, A.; Kahlert, S.; Rothkotter, H.J.; Kluess, J. Vulnerability of polarised intestinal porcine epithelial cells to mycotoxin deoxynivalenol depends on the route of application. PLoS ONE 2011, 6, e17472. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, M.C.; Madsen, K.; Doyle, J.; Meddings, J. Reducing small intestinal permeability attenuates colitis in the IL10 gene-deficient mouse. Gut 2009, 58, 41–48. [Google Scholar] [CrossRef]
- Yang, R.; Hui, Q.; Jiang, Q.; Liu, S.; Zhang, H.; Wu, J.; Lin, F.; O, K.; Yang, C. Effect of Manitoba-Grown Red-Osier Dogwood Extracts on Recovering Caco-2 Cells from H(2)O(2)-Induced Oxidative Damage. Antioxidants 2019, 8, 250. [Google Scholar] [CrossRef]
- Cui, L.; Feng, L.; Zhang, Z.H.; Jia, X.B. The anti-inflammation effect of baicalin on experimental colitis through inhibiting TLR4/NF-kappaB pathway activation. Int. Immunopharmacol. 2014, 23, 294–303. [Google Scholar] [CrossRef]
- Fukata, M.; Abreu, M.T. TLR4 signalling in the intestine in health and disease. Biochem. Soc. Trans. 2007, 35, 1473–1478. [Google Scholar] [CrossRef]
- Seo, J.H.; Lim, J.W.; Kim, H. Differential Role of ERK and p38 on NF- kappa B Activation in Helicobacter pylori-Infected Gastric Epithelial Cells. J. Cancer Prev. 2013, 18, 346–350. [Google Scholar] [CrossRef]
- Wang, C.; Sun, H.; Song, Y.; Ma, Z.; Zhang, G.; Gu, X.; Zhao, L. Pterostilbene attenuates inflammation in rat heart subjected to ischemia-reperfusion: Role of TLR4/NF-kappaB signaling pathway. Int. J. Clin. Exp. Med. 2015, 8, 1737–1746. [Google Scholar] [PubMed]
- Ye, H.Y.; Jin, J.; Jin, L.W.; Chen, Y.; Zhou, Z.H.; Li, Z.Y. Chlorogenic Acid Attenuates Lipopolysaccharide-Induced Acute Kidney Injury by Inhibiting TLR4/NF-kappaB Signal Pathway. Inflammation 2017, 40, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Ngo, V.L.; Abo, H.; Maxim, E.; Harusato, A.; Geem, D.; Medina-Contreras, O.; Merlin, D.; Gewirtz, A.T.; Nusrat, A.; Denning, T.L. A cytokine network involving IL-36gamma, IL-23, and IL-22 promotes antimicrobial defense and recovery from intestinal barrier damage. Proc. Natl. Acad. Sci. USA 2018, 115, E5076–E5085. [Google Scholar] [CrossRef]
- Zhuang, H.; Li, B.; Xie, T.; Xu, C.; Ren, X.; Jiang, F.; Lei, T.; Zhou, P. Indole-3-aldehyde alleviates chondrocytes inflammation through the AhR-NF-kappaB signalling pathway. Int. Immunopharmacol. 2022, 113, 109314. [Google Scholar] [CrossRef] [PubMed]
- D’Onofrio, F.; Renga, G.; Puccetti, M.; Pariano, M.; Bellet, M.M.; Santarelli, I.; Stincardini, C.; Mosci, P.; Ricci, M.; Giovagnoli, S.; et al. Indole-3-Carboxaldehyde Restores Gut Mucosal Integrity and Protects from Liver Fibrosis in Murine Sclerosing Cholangitis. Cells 2021, 10, 1622. [Google Scholar] [CrossRef]
- Guo, H.; Xu, Y.; Huang, W.; Zhou, H.; Zheng, Z.; Zhao, Y.; He, B.; Zhu, T.; Tang, S.; Zhu, Q. Kuwanon G Preserves LPS-Induced Disruption of Gut Epithelial Barrier In Vitro. Molecules 2016, 21, 1597. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Liu, Z.; Cui, X.; Zhao, Q.; Liu, T. Intestinal vitamin D receptor knockout protects from oxazolone-induced colitis. Cell Death Dis. 2020, 11, 461. [Google Scholar] [CrossRef]
- Wang, H.; Jiang, Y.; Li, H.; Wang, J.; Li, C.; Zhang, D. Carbachol protects the intestinal barrier in severe acute pancreatitis by regulating Cdc42/F-actin cytoskeleton. Exp. Ther. Med. 2020, 20, 2828–2837. [Google Scholar] [CrossRef]
- Bender, M.J.; McPherson, A.C.; Phelps, C.M.; Pandey, S.P.; Laughlin, C.R.; Shapira, J.H.; Sanchez, L.M.; Rana, M.; Richie, T.G.; Mims, T.S.; et al. Dietary tryptophan metabolite released by intratumoral Lactobacillus reuteri facilitates immune checkpoint inhibitor treatment. Cell 2023, in press. [Google Scholar] [CrossRef]
- Li, Y.Y.; Wang, X.J.; Su, Y.L.; Wang, Q.; Huang, S.W.; Pan, Z.F.; Chen, Y.P.; Liang, J.J.; Zhang, M.L.; Xie, X.Q.; et al. Baicalein ameliorates ulcerative colitis by improving intestinal epithelial barrier via AhR/IL-22 pathway in ILC3s. Acta Pharmacol. Sin. 2022, 43, 1495–1507. [Google Scholar] [CrossRef]
- Fukata, M.; Shang, L.; Santaolalla, R.; Sotolongo, J.; Pastorini, C.; Espana, C.; Ungaro, R.; Harpaz, N.; Cooper, H.S.; Elson, G.; et al. Constitutive activation of epithelial TLR4 augments inflammatory responses to mucosal injury and drives colitis-associated tumorigenesis. Inflamm. Bowel Dis. 2011, 17, 1464–1473. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.A.; Lee, I.A.; Gu, W.; Hyam, S.R.; Kim, D.H. beta-Sitosterol attenuates high-fat diet-induced intestinal inflammation in mice by inhibiting the binding of lipopolysaccharide to toll-like receptor 4 in the NF-kappaB pathway. Mol. Nutr. Food Res. 2014, 58, 963–972. [Google Scholar] [CrossRef] [PubMed]
- Gan, H.T.; Chen, Y.Q.; Ouyang, Q. Sulfasalazine inhibits activation of nuclear factor-kappaB in patients with ulcerative colitis. J. Gastroenterol. Hepatol. 2005, 20, 1016–1024. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Yue, B.; Yu, Z.; Ren, Y.; Zhang, J.; Ren, J.; Wang, Z.; Dou, W. Obacunone Protects Against Ulcerative Colitis in Mice by Modulating Gut Microbiota, Attenuating TLR4/NF-kappaB Signaling Cascades, and Improving Disrupted Epithelial Barriers. Front. Microbiol. 2020, 11, 497. [Google Scholar] [CrossRef]
- Hashimoto, T.; Perlot, T.; Rehman, A.; Trichereau, J.; Ishiguro, H.; Paolino, M.; Sigl, V.; Hanada, T.; Hanada, R.; Lipinski, S.; et al. ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature 2012, 487, 477–481. [Google Scholar] [CrossRef]


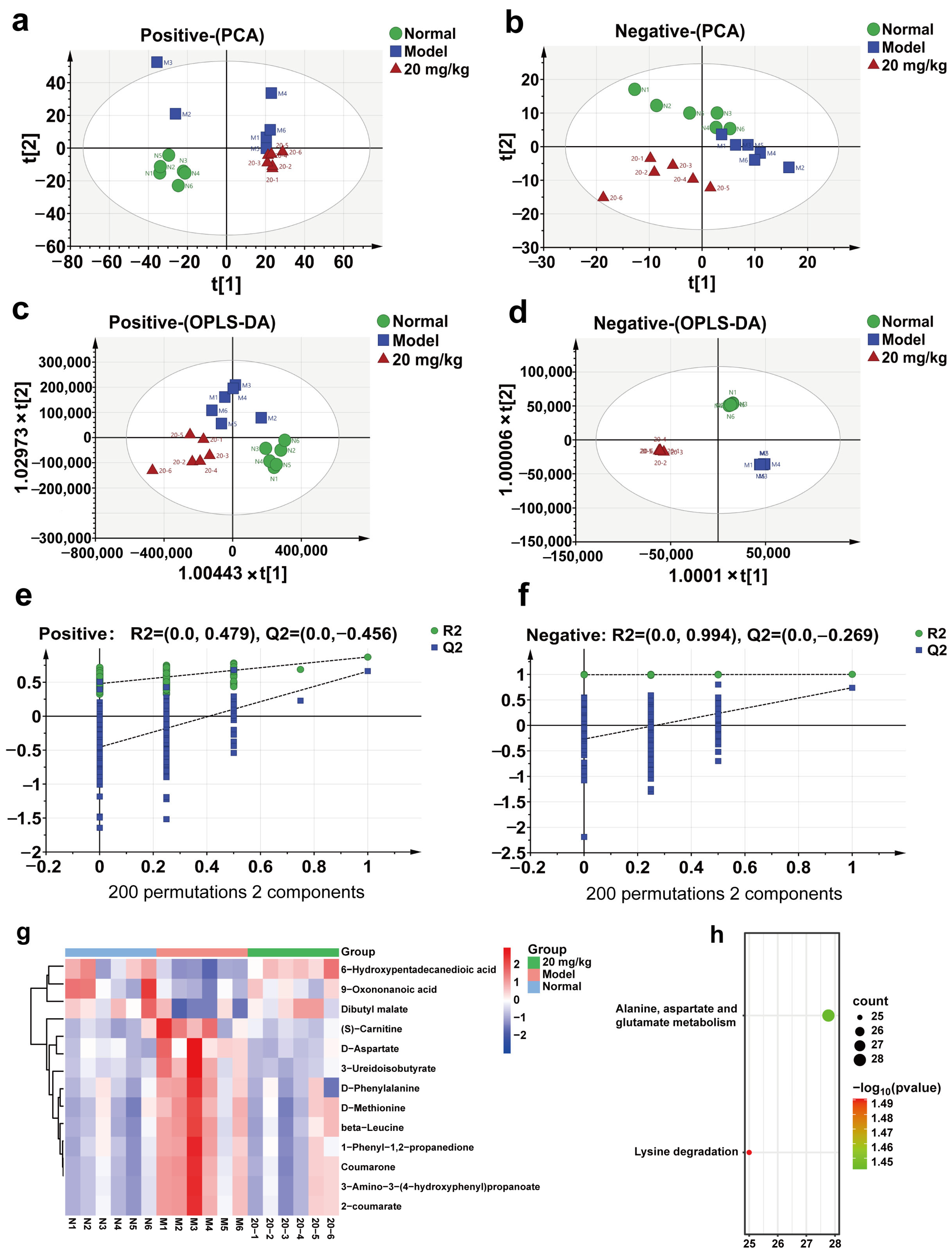
| No. | Name | Formula | RT (min) | Ion mod | VIP | Model vs. Normal | 20 mg/kg vs. Model |
|---|---|---|---|---|---|---|---|
| 1 | 6-Hydroxypentadecanedioic acid | C15H28O5 | 5.39 | ESI− | 2.29035 | ↓ ## | ↑ **** |
| 2 | Dibutyl malate | C12H22O5 | 6.427 | ESI− | 1.2762 | ↓ # | ↑ * |
| 3 | 9-Oxononanoic acid | C9H16O3 | 6.323 | ESI− | 1.1328 | ↓ # | ↑ * |
| 4 | beta-Leucine | C6H13NO2 | 2.513 | ESI+ | 3.50429 | ↑ ## | ↓ * |
| 5 | (S)-Carnitine | C7H15NO3 | 1.544 | ESI+ | 1.19308 | ↑ # | ↓ * |
| 6 | 1-Phenyl-1,2-propanedione | C9H8O2 | 4.946 | ESI+ | 4.36873 | ↑ ## | ↓ * |
| 7 | 2-coumarate | C9H8O3 | 2.355 | ESI+ | 3.97959 | ↑ ## | ↓ * |
| 8 | 3-Amino-3-(4-hydroxyphenyl) propanoate | C9H11NO3 | 2.365 | ESI+ | 3.56817 | ↑ ## | ↓ * |
| 9 | 3-Ureidoisobutyrate | C5H10N2O3 | 1.421 | ESI+ | 2.07972 | ↑ # | ↓ ** |
| 10 | Coumarone | C8H6O | 2.342 | ESI+ | 1.73917 | ↑ ## | ↓ * |
| 11 | D-Aspartate | C4H7NO4 | 1.418 | ESI+ | 1.91917 | ↑ # | ↓ * |
| 12 | D-Methionine | C5H11NO2S | 1.976 | ESI+ | 2.60987 | ↑ ## | ↓ ** |
| 13 | D-Phenylalanine | C9H11NO2 | 4.92 | ESI+ | 4.66763 | ↑ ## | ↓ ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, M.; Wang, Y.; Xiang, H.; Guo, M.; Li, S.; Liu, M.; Yao, J. The Tryptophan Metabolite Indole-3-Carboxaldehyde Alleviates Mice with DSS-Induced Ulcerative Colitis by Balancing Amino Acid Metabolism, Inhibiting Intestinal Inflammation, and Improving Intestinal Barrier Function. Molecules 2023, 28, 3704. https://doi.org/10.3390/molecules28093704
Liu M, Wang Y, Xiang H, Guo M, Li S, Liu M, Yao J. The Tryptophan Metabolite Indole-3-Carboxaldehyde Alleviates Mice with DSS-Induced Ulcerative Colitis by Balancing Amino Acid Metabolism, Inhibiting Intestinal Inflammation, and Improving Intestinal Barrier Function. Molecules. 2023; 28(9):3704. https://doi.org/10.3390/molecules28093704
Chicago/Turabian StyleLiu, Mingfei, Yuxuan Wang, Haixin Xiang, Meng Guo, Shirong Li, Ming Liu, and Jingchun Yao. 2023. "The Tryptophan Metabolite Indole-3-Carboxaldehyde Alleviates Mice with DSS-Induced Ulcerative Colitis by Balancing Amino Acid Metabolism, Inhibiting Intestinal Inflammation, and Improving Intestinal Barrier Function" Molecules 28, no. 9: 3704. https://doi.org/10.3390/molecules28093704
APA StyleLiu, M., Wang, Y., Xiang, H., Guo, M., Li, S., Liu, M., & Yao, J. (2023). The Tryptophan Metabolite Indole-3-Carboxaldehyde Alleviates Mice with DSS-Induced Ulcerative Colitis by Balancing Amino Acid Metabolism, Inhibiting Intestinal Inflammation, and Improving Intestinal Barrier Function. Molecules, 28(9), 3704. https://doi.org/10.3390/molecules28093704





