Do Ganoderma Species Represent Novel Sources of Phenolic Based Antimicrobial Agents?
Abstract
1. Introduction
2. Results and Discussion
2.1. Mycochemical Profile by LC-MS/MS Detection
2.2. Antimicrobial Activity
2.2.1. Antibacterial Activity
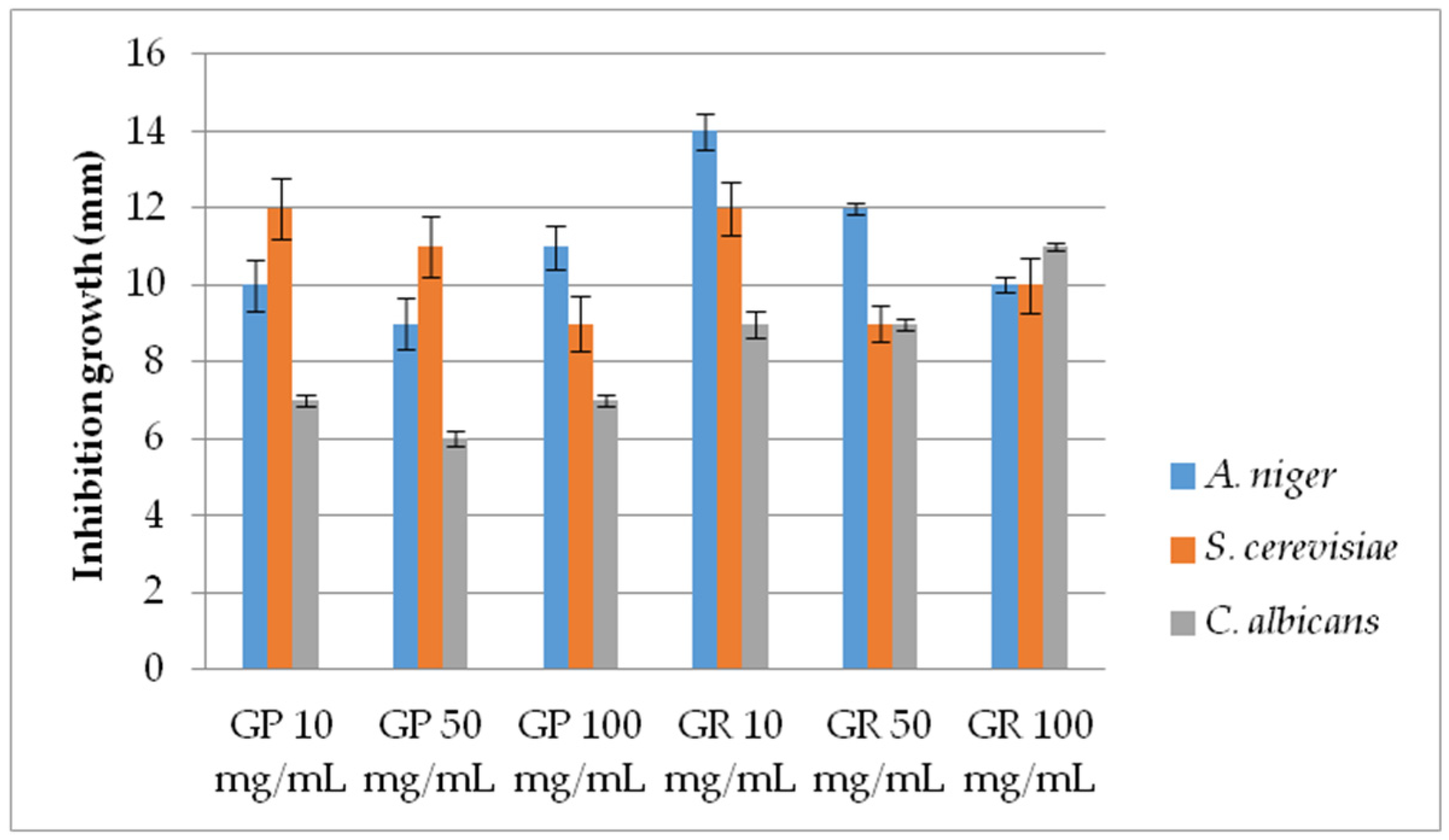
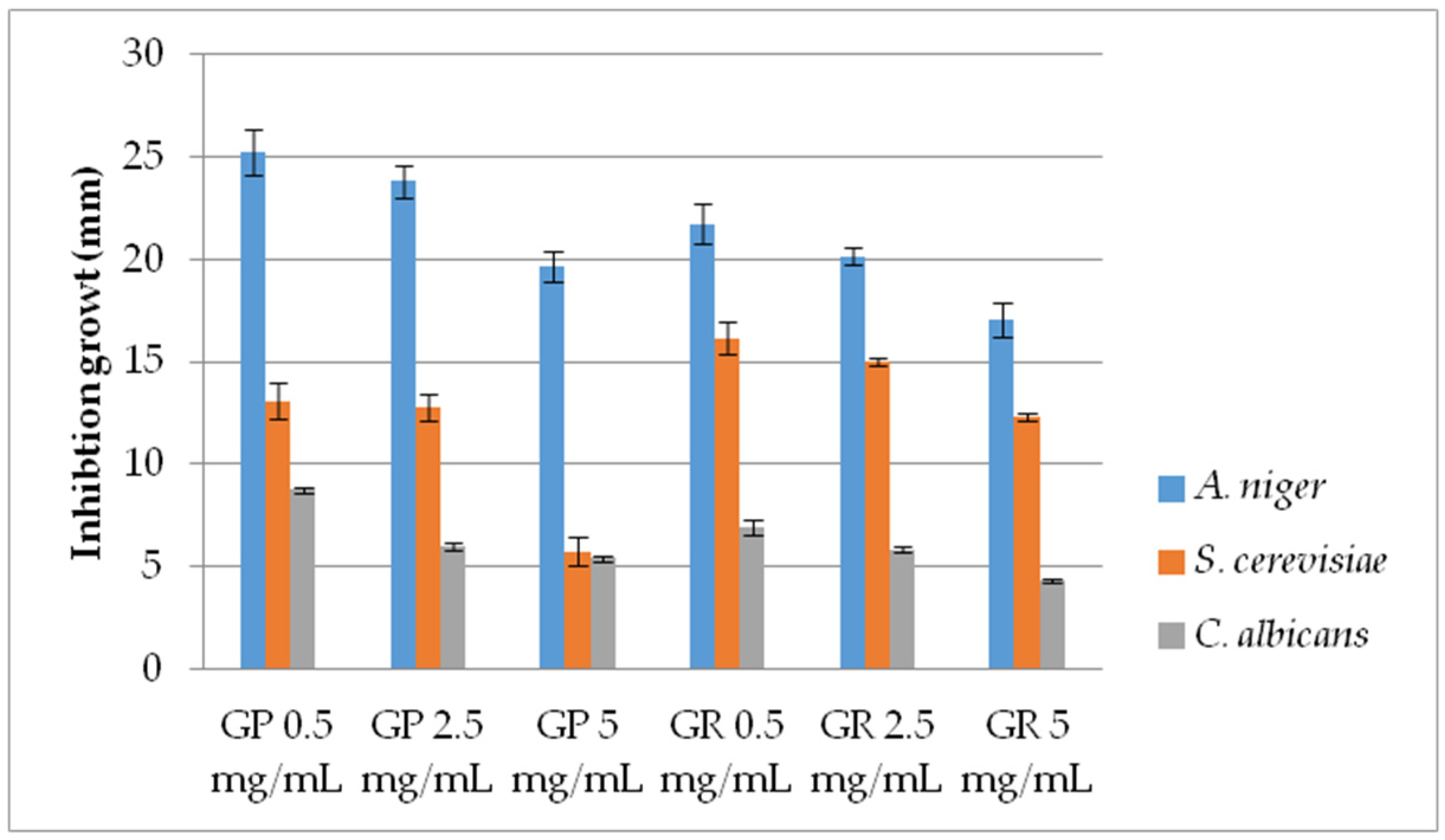
2.2.2. Antifungal Activity
2.2.3. Antiviral Activity
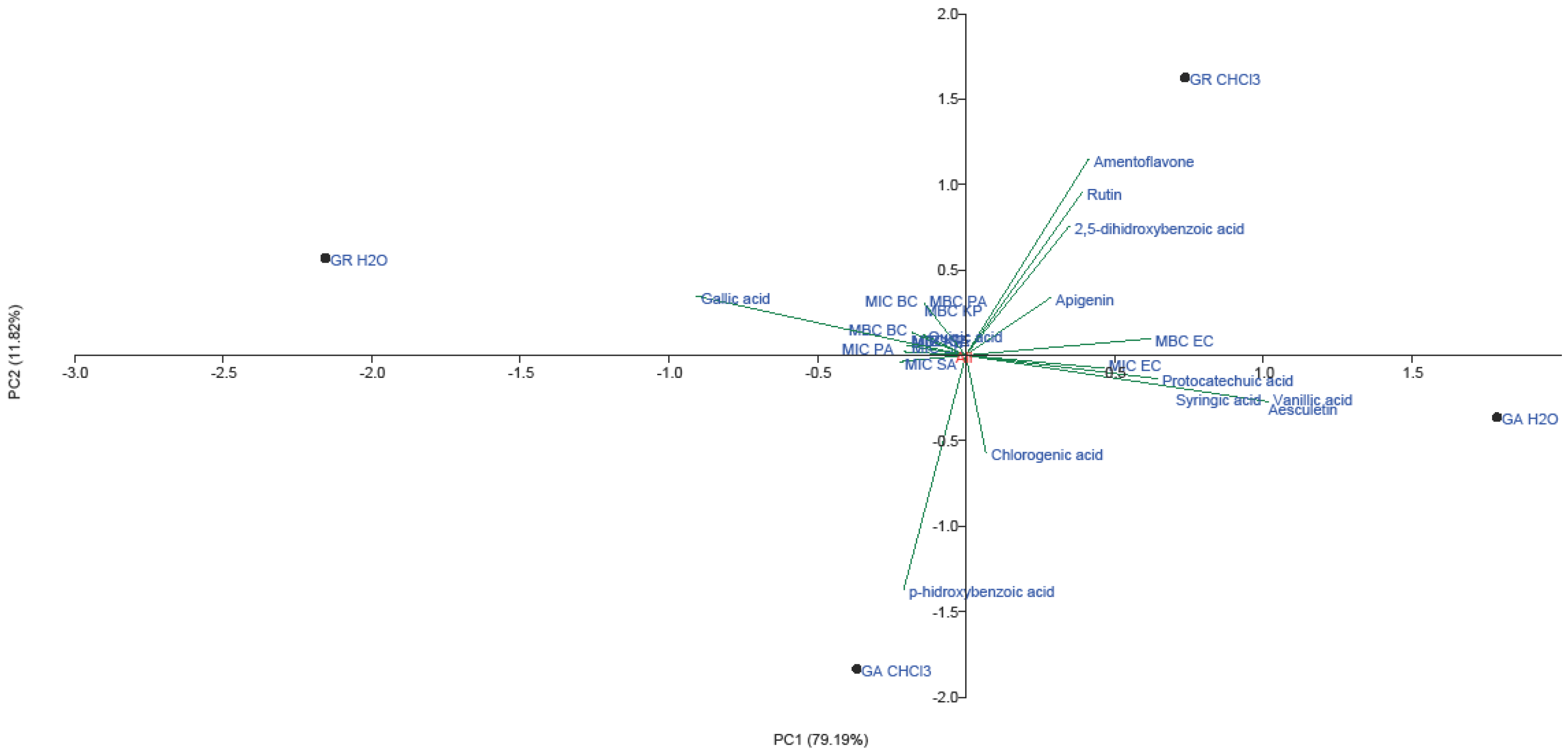
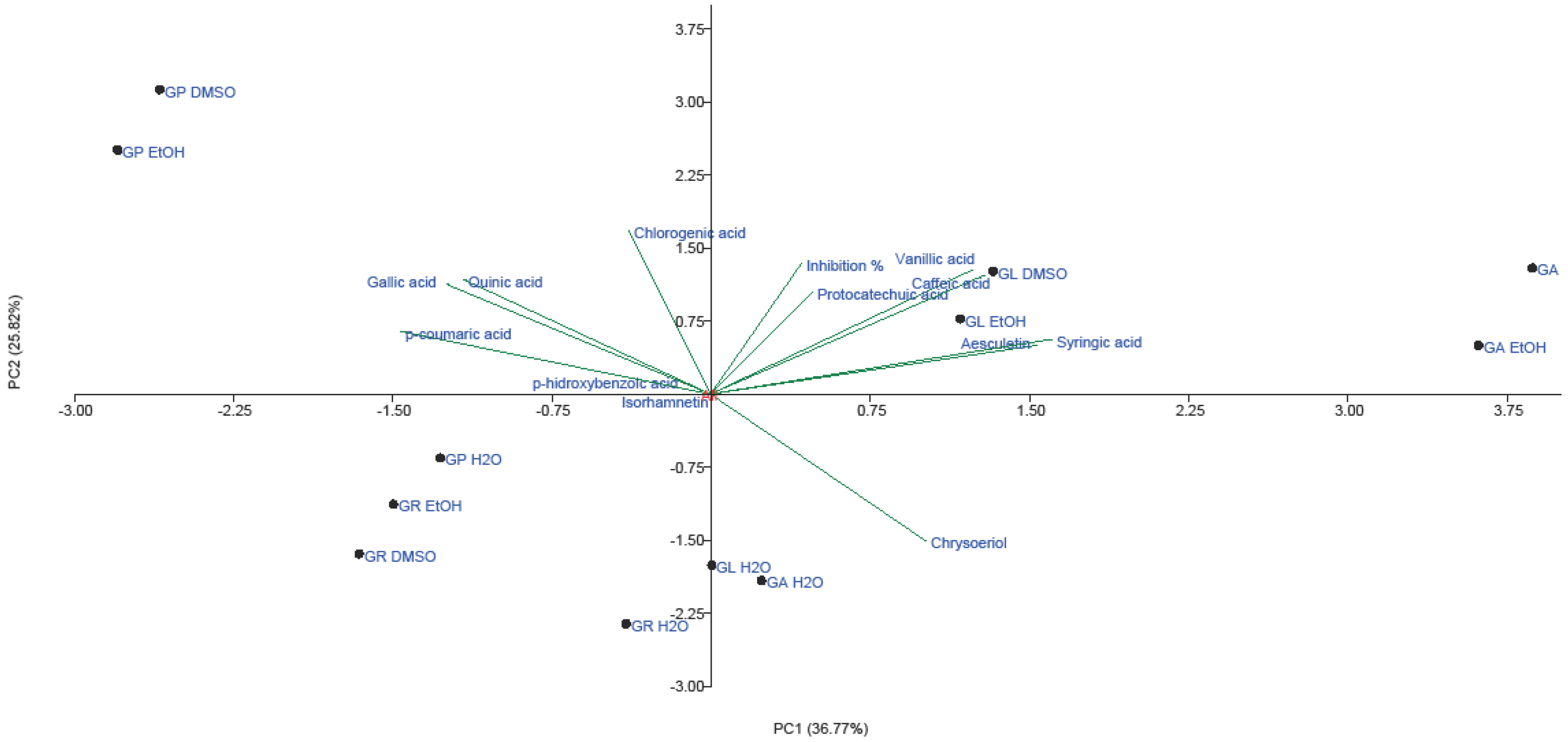
2.3. PCA Analysis
3. Materials and Methods
3.1. Fungal Material
3.2. Preparation of Extracts
3.3. LC-MS/MS Analysis of Selected Phenolic Compounds
3.4. Antimicrobial Activity
3.4.1. Nutrient Media
3.4.2. Antibacterial and Antifungal Activity
Bacterial and Fungal Strains
Antibiogram
Diffusion Assays
Pour Plate Method
Microdilution Assay
3.4.3. Antiviral Activity
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thapa, R.; Maharjan, R.; Tamang, P.; Gautam, P.; Adhikari, R.; Maharjan, S. Antimicrobial assessment and phytochemical screening of medicinal plants and Ganoderma lucidum. IJASBT 2022, 10, 228–236. [Google Scholar] [CrossRef]
- WHO. Antibiotic Resistance; World Health Organization: Geneva, Switzerland, 2016.
- Pelgrift, R.Y.; Friedman, A.J. Nanotechnology as a therapeutic tool to combat microbial resistance. Adv. Drug Deliv. Rev. 2013, 65, 1803–1815. [Google Scholar] [CrossRef]
- Cetinkaya, Y.; Falk, P.; Mayhall, C.G. Vancomycin-resistant enterococci. Clin. Microbiol. Rev. 2000, 13, 686–707. [Google Scholar] [CrossRef]
- Betts, J.W.; Hornsey, M.; La Ragione, R.M. Novel antibacterials: Alternatives to traditional antibiotics. Adv. Microb. Physiol. 2018, 73, 123–169. [Google Scholar] [PubMed]
- Spencer, C.A.; Brubaker, K.R.; Garneau-Tsodikova, S. Systemic fungal infections: A pharmacist/researcher perspective. Fungal Biol. Rev. 2023, 44, 100293. [Google Scholar] [CrossRef]
- Hussain, M.K.; Ahmed, S.; Khan, A.; Siddiqui, A.J.; Khatoon, S.; Jahan, S. Mucormycosis: A hidden mystery of fungal infection, possible diagnosis, treatment and development of new therapeutic agents. Eur. J. Med. Chem. 2023, 246, 115010. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, J.Q.; Zhang, J.; Li, Z.; Liu, H.; Wang, Y. Traditional uses, chemical components and pharmacological activities of the genus Ganoderma P. Karst.: A review. RSC Adv. 2020, 10, 42084–42097. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Wu, S.H.; Dai, Y.C. Species clarification of the prize medicinal Ganoderma mushroom “Lingzhi”. Fungal Divers 2012, 56, 49–62. [Google Scholar] [CrossRef]
- Karaman, M.; Čapelja, E.; Rašeta, M.; Rakić, M. Diversity, Chemistry, and Environmental Contamination of Wild Growing Medicinal Mushroom Species as Sources of Biologically Active Substances (Antioxidants, Anti-Diabetics, and AChE Inhibitors). In Biology, Cultivation and Applications of Mushrooms; Arya, A., Rusevska, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2022; Volume 8, pp. 203–257. [Google Scholar]
- Al-Fatimi, M.; Wurster, M.; Kreisel, H.; Lindequist, U. Antimicrobial, cytotoxic and antioxidant activity of selected basidiomycetes from Yemen. Die Pharmazie-Int. J. Pharm. 2005, 60, 776–780. [Google Scholar]
- Karaman, M.; Mimica-Dukić, N.; Knežević, P.; Svirčev, Z.; Matavuly, M.N. Antibacterial properties of selected lignicolous mushrooms and fungi from Northern Serbia. Int. J. Med. Mushrooms 2009, 11, 269–279. [Google Scholar] [CrossRef]
- Karaman, M.; Matavulj, M.; Janjić, L. Antibacterial agents from lignicolous macrofungi. In Antimicrobial Agents; Chapter 18; Varaprasad, B., Ed.; InTech: Rijeka, Croatia, 2012; pp. 361–386. [Google Scholar]
- Karaman, M.; Stahl, M.; Vesic, M.; Novakovic, M.; Janjic, L.; Matavuly, M. Bioactive properties of wild-growing mushroom species Ganoderma applanatum (Pers.) Pat. from Fruška gora forest (Serbia). In Recent Progress in Medicinal Plants, RPMP Ethnomedicine and Therapeutic Validation; Govil, J.N., Ed.; Studium Press: New Delhi, India, 2012; Volume 32, pp. 361–377. [Google Scholar]
- Dora, J.; Hena, V.; Ranjan. Antimicrobial activity of Ganoderma lucidum fruiting body extract from Himachal Pradesh. IJSDR 2020, 5, 263–266. [Google Scholar]
- Ede, S.O.; Aguiyi, J.C.; Omale, S.; Ede, R.F. Anti-trypanosomal, antioxidant and antimicrobial activities of the fruiting bodies of Ganoderma lucidum (W. Curt.: Fr)(Ganodermataceae) aqueous extract. JPB 2021, 18, 172–181. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Gholami, A.; Omidifar, N.; Chiang, W.H.; Neralla, V.R.; Shokripour, M. Ganoderma lucidum methanolic extraction as a potent phytoconstituent: Characterization, in-vitro antimicrobial and cytotoxic activity. Res. Sq. 2022. preprint. [Google Scholar] [CrossRef]
- Suansia, A.; John, P. Antimicrobial and Antioxidant properties of medicinal mushroom Ganoderma P. Karst. GSCBPS 2021, 17, 106–112. [Google Scholar]
- Al-Fatimi, M.; Wurster, M.; Lindequist, U. Chemical composition, antimicrobial and antioxidant activities of the volatile oil of Ganoderma pfeifferi Bres. Medicines 2016, 3, 10. [Google Scholar] [CrossRef]
- Sharma, C.; Bhardwaj, N.; Sharma, A.; Tuli, H.S.; Batra, P.; Beniwal, V.; Gupta, G.K.; Sharma, A. Bioactive metabolites of Ganoderma lucidum: Factors, mechanism and broad-spectrum therapeutic potential. J. Herb. Med. 2019, 17–18, 100268. [Google Scholar] [CrossRef]
- Stamets, P.E.; Naeger, N.L.; Evans, J.D.; Han, J.O.; Hopkins, B.K.; Lopez, D.L.; Moershel, H.M.; Nally, R.; Sumerlin, D.; Taylor, A.; et al. Extracts of polypore mushroom mycelia reduce viruses in honeybees. Sci. Rep. 2018, 8, 13936. [Google Scholar] [CrossRef]
- Stojković, D.S.; Barros, L.; Calhelha, R.C.; Glamočlija, J.; Ćirić, A.; van Griensven, L.J.; Soković, M.D.; Ferreira, I.C. A detailed comparative study between chemical and bioactive properties of Ganoderma lucidum from different origins. Int. J. Food Sci. Nutr. 2014, 65, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Rašeta, M.; Karaman, M.; Jakšić, M.; Šibul, F.; Kebert, M.; Novaković, A.; Popović, M. Mineral composition, antioxidant and cytotoxic biopotentials of wild-growing Ganoderma species (Serbia): G. lucidum (Curtis) P. Karst vs. G. applanatum (Pers.). Pat. Int. J. Food Sci. Technol. 2016, 51, 2583–2590. [Google Scholar] [CrossRef]
- Zengin, G.; Sarikurkcu, C.; Gunes, E.; Uysal, A.; Ceylan, R.; Uysal, S.; Gungor, H.; Aktumseka, A. Two Ganoderma species: Profiling of phenolic compounds by HPLC-DAD, antioxidant, antimicrobial and inhibitory activities on key enzymes linked to diabetes mellitus, Alzheimer’s disease and skin disorders. Food Funct. 2015, 6, 2794–2802. [Google Scholar] [CrossRef] [PubMed]
- Rašeta, M.; Popović, M.; Čapo, I.; Stilinović, N.; Vukmirović, S.; Karaman, M. Antidiabetic effect of two different Ganoderma species tested in alloxan diabetic rats. RSC Adv. 2020, 10, 10382–10393. [Google Scholar] [CrossRef]
- Zabka, M.; Pavela, R. Antifungal efficacy of some natural phenolic compounds against significant pathogenic and toxinogenic filamentous fungi. Chemosphere 2013, 93, 1051–1056. [Google Scholar] [CrossRef]
- Mothana, R.A.; Jansen, R.; Jülich, W.D.; Lindequist, U. Ganomycins A and B, new antimicrobial farnesyl hydroquinones from the basidiomycete Ganoderma pfeifferi. J. Nat. Prod. 2000, 63, 416–418. [Google Scholar] [CrossRef]
- Zhang, W.; Tao, J.; Yang, X.; Yang, Z.; Zhang, L.; Liu, H.; Wu, K.; Wu, J. Antiviral effects of two Ganoderma lucidum triterpenoids against Enterovirus 71 infection. Biochem. Biophys. Res. Commun. 2014, 449, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef]
- Carvalho, R.S.; Carollo, C.A.; de Magalhães, J.C.; Palumbo, J.M.C.; Boaretto, A.G.; Sa, I.C.N.; Ferraz, A.C.; Lima, W.G.; de Siqueira, J.M.; Ferreira, J.M.S. Antibacterial and antifungal activities of phenolic compound-enriched ethyl acetate fraction from Cochlospermum regium (mart. Et. Schr.) Pilger roots: Mechanisms of action and synergism with tannin and gallic acid. S. Afr. J. Bot. 2018, 114, 181–187. [Google Scholar] [CrossRef]
- Yang, S.K.; Yusoff, K.; Ajat, M.; Thomas, W.; Abushelaibi, A.; Akseer, R.; Lim, S.E.; Lai, K.S. Disruption of KPC-producing Klebsiella pneumoniae membrane via induction of oxidative stress by cinnamon bark (Cinnamomum verum J. Presl) essential oil. PLoS ONE 2019, 14, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Griffin, S.G.; Wyllie, S.G.; Markham, J.L.; Leach, D.N. The role of structure and molecular properties of terpenoids in determining their antimicrobial activity. Flavour Fragr. J. 1999, 14, 322–332. [Google Scholar] [CrossRef]
- Rašeta, M.; Popović, M.; Beara, I.; Šibul, F.; Zengin, G.; Krstić, S.; Karaman, M. Anti-inflammatory, antioxidant and enzyme inhibition activities in correlation with mycochemical profile of selected indigenous Ganoderma spp. from Balkan region (Serbia). Chem. Biodivers 2020, 17, e2000828. [Google Scholar] [CrossRef] [PubMed]
- Nagaraj, K.; Mallikarjun, N.; Naika, R.; Venugopal, T.M. Phytochemical analysis and in vitro antimicrobial potential of Ganoderma applanatum (Pers.) Pat. of Shivamogga district-Karnataka, India. Int. J. Pharm. Sci. Rev. Res 2013, 23, 36–41. [Google Scholar]
- Niedermeyer, T.H.J.; Lindequist, U.; Mentel, R.; Gördes, D.; Schmidt, E.; Thurow, K.; Lalk, M. Antiviral terpenoid constituents of Ganoderma pfeifferi. J. Nat. Prod. 2005, 68, 1728–1731. [Google Scholar] [CrossRef]
- Celal, B. Antioxidant and antimicrobial capacities of Ganoderma lucidum. MedCrave 2019, 7, 5–7. [Google Scholar]
- Cör, D.; Knez, Ž.; Knez Hrnčič, M. Antitumour, antimicrobial, antioxidant and antiacetylcholinesterase effect of Ganoderma lucidum terpenoids and polysaccharides: A review. Molecules 2018, 23, 649. [Google Scholar] [CrossRef] [PubMed]
- Karaman, M.; Jovin, E.; Malbaša, R.; Matavulj, M.; Popović, M. Medicinal and edible lignicolous fungi as natural sources of antioxidative and antibacterial agents. Phytother. Res. 2010, 24, 1473–1481. [Google Scholar] [CrossRef]
- Ofodile, L.N.; Uma, N.U.; Kokubun, T.; Grayer, R.J.; Ogundipe, O.T.; Simmonds, M.S.J. Antimicrobial activity of some Ganoderma species from Nigeria. Phytother. Res.: An Int. J. Pharm. Toxicol. Eval. Nat. Prod. Deriv. 2005, 19, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Smania, A.J.; Monache, F.D.; Smania, E.F.; Cuneo, R.S. Antibacterial activity of steroidal compounds isolated from Ganoderma applanatum (Pers.) Pat. (Aphyllophoromycetidae) fruit body. Int. J. Med. Mushrooms 1999, 1, 325–330. [Google Scholar] [CrossRef]
- Ameri, A.; Vaidya, J.G.; Deokule, S.S. In vitro evaluation of anti-staphylococcal activity of Ganoderma lucidum, Ganoderma praelongum and Ganoderma resinaceum from Pune, India. Afr. J. Microbiol. Res. 2011, 5, 328–333. [Google Scholar]
- Keypour, S.; Riahi, H.; Moradali, M.F.; Rafati, H. Investigation of the antibacterial activity of a chlorophorm extract of Ling Zhi or Reishi medicinal mushroom Ganoderma lucidum (W. Curt.: Fr) P. Karst. (Aphyllophoromzcetideae) from Iran. Int. J. Med. Mushrooms 2008, 10, 345–349. [Google Scholar] [CrossRef]
- Araruna, M.K.; Brito, S.A.; Morais-Braga, M.F.; Santos, K.K.; Souza, T.M.; Leite, T.R.; Costa, J.G.; Coutinho, H.D. Evaluation of antibiotic and antibiotic modifying activity of pilocarpine and rutin. Indian J. Med. Res. 2012, 135, 252–254. [Google Scholar]
- Dubey, S.; Ganeshpurkar, A.; Bansal, D.; Dubey, N. Experimental studies on bioactive potential of rutin. Chron. Young Sci. 2013, 4, 153–157. [Google Scholar]
- Arima, H.; Ashida, H.; Danno, G. Rutin-enhanced antibacterial activities of flavonoids against Bacillus cereus and Salmonella enteritidis. Biosci. Biotechnol. Biochem. 2002, 66, 1009–1014. [Google Scholar] [CrossRef]
- Ganeshpurkar, A.; Saluja, A.K. The pharmacological potential of rutin. Saudi Pharm. J. 2017, 25, 149–164. [Google Scholar] [CrossRef]
- Jorcin, G.; Barneche, S.; Vázquez, A.; Cerdeiras, M.P.; Alborés, S. Effects of culture conditions on antimicrobial activity of Ganoderma resinaceum (Agaricomycetes) extracts. Int. J. Med. Mushrooms 2017, 19, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Alves, M.J.; Ferreira, I.C.; Froufe, H.J.; Abreu, R.M.; Martins, A.; Pintado, M. Antimicrobial activity of phenolic compounds identified in wild mushrooms, SAR analysis and docking studies. J. Appl. Microbiol. 2013, 113, 466–475. [Google Scholar] [CrossRef]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual, 3rd ed; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2001. [Google Scholar]
- Orhan, D.D.; Özçelik, B.; Özgen, S.; Ergun, F. Antibacterial, antifungal, and antiviral activities of some flavonoids. Microbiol. Res. 2010, 165, 496–504. [Google Scholar] [CrossRef]
- Ai, H.; Wang, F.; Xia, Y.; Chen, X.; Lei, C. Antioxidant, antifungal and antiviral activities of chitosan from the larvae of housefly, Musca domestica L. Food Chem. 2012, 132, 493–498. [Google Scholar] [CrossRef]
- Guaní-Guerra, E.; Santos-Mendoza, T.; Lugo-Reyes, S.; Teran, L.M. Antimicrobial peptides: General overview and clinical implications in human health and diseases. Clin. Immunol. 2010, 135, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ng, T.B. Ganodermin, an antifungal protein from fruiting bodies of the medicinal mushroom Ganoderma lucidum. Peptides 2006, 27, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Yan, H.; Chen, J.; Zhang, X. Bioactive proteins from mushrooms. Biotechnol. Adv. 2011, 29, 667–674. [Google Scholar] [CrossRef]
- Chen, J.; Seviour, R. Medicinal importance of fungal—(1→3), (1→4)—glucans. Mycol. Res. 2007, 3, 635–652. [Google Scholar] [CrossRef]
- Smania, E.F.A.; Delle Monache, F.; Smania, J.A.; Yunes, R.A.; Cuneo, R.S. Antifungal activity of sterols and triterpenes isolated from Ganoderma annulare. Fitoterapia 2003, 74, 375–377. [Google Scholar] [CrossRef]
- Mothana, R.A.A.; Awadh Ali, N.A.; Jansen, R.; Wegner, U.; Mentel, R.D.; Lindequist, U. Antiviral lanostanoid triterpenes from the fungus Ganoderma pfeifferi. Fitoterapia 2003, 74, 177–180. [Google Scholar] [CrossRef]
- Roberts, L.M. Australian Ganoderma: Identification, growth and antibacterial properties. Ph.D. Thesis, Swinburne University of Technology, Melbourne, Australia, 2004. [Google Scholar]
- Bharadwaj, S.; Lee, K.E.; Dwivedi, V.D.; Yadava, U.; Panwar, A.; Lucas, S.J.; Pandey, A.; Kang, S.G. Discovery of Ganoderma lucidum triterpenoids as potential inhibitors against Dengue virus NS2B-NS3 protease. Sci. Rep. 2019, 9, 19059. [Google Scholar] [CrossRef]
- Ahmad, A.; Kaleem, M.; Ahmed, Z.; Shafiq, H. Therapeutic potential of flavonoids and their mechanism of action against microbial and viral infections- a review. Food Res. Int. 2015, 77, 221–235. [Google Scholar] [CrossRef]
- Kowalczyk, M.; Golonko, A.; Świsłocka, R.; Kalinowska, M.; Parcheta, M.; Swiergiel, A.; Lewandowski, W. Drug design strategies for the treatment of viral disease. Plant phenolic compounds and their derivatives. Front. Pharmacol. 2021, 12, 709104. [Google Scholar] [CrossRef]
- Mani, J.S.; Johnson, J.B.; Steel, J.C.; Broszczak, D.A.; Neilsen, P.M.; Walsh, K.B.; Naiker, M. Natural product-derived phytochemicals as potential agents against coronaviruses: A review. Virus. Res. 2020, 284, 197989. [Google Scholar] [CrossRef] [PubMed]
- Lindequist, U.; Niedermeyer, T.H.J.; Jülich, W.D. The pharmacological potential of mushrooms. Evid. Based Complement. Altern. Med. 2005, 2, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Razumov, I.A.; Kosogova, T.A.; Kazachinskaia, E.I.; Puchkova, L.I.; Shcherbakova, N.S.; Gorbunova, I.; Mikhaĭlovskaia, I.N.; Loktev, V.B.; Tepliakova, T.V. Antiviral activity of aqueous extracts and polysaccharide fractions from mycelium and fruit bodies of higher fungi. Antibiot. Chemoter. 2010, 55, 14–18. [Google Scholar]
- Eo, S.K.; Kim, Y.S.; Lee, C.K.; Han, S.S. Possible mode of antiviral activity of acidic protein bound polysaccharide isolated from Ganoderma lucidum on herpes simplex viruses. J. Ethnopharmacol. 2000, 72, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Nie, S.; Zhang, H.; Li, W.; Xie, M. Current development of polysaccharides from Ganoderma: Isolation, structure and bioactivities. Bioact. Carbohydr. Diet. Fibre 2013, 1, 10–20. [Google Scholar] [CrossRef]
- Cör Andrejč, D.; Knez, Ž.; Knez Marevci, M. Antioxidant, antibacterial, antitumor, antifungal, antiviral, anti-inflammatory, and nevro-protective activity of Ganoderma lucidum: An overview. Front Pharmacol. 2022, 13, 934982. [Google Scholar] [CrossRef]
- Cheng, P.G.; Teoh, T.C.; Rizman-Idid, M. Chemical compounds and computational prediction of their inhibitory effects on the HIV-1 gp120 receptor by lingzhi or reishi medicinal mushroom, Ganoderma lucidum (agaricomycetes), with antler-like morphology of fruiting bodies. Int. J. Med. Mushrooms 2021, 23, 63–77. [Google Scholar] [CrossRef]
- Al-jumaili, M.M.; Al-Dulaimi, F.; Ajeel, M.A. The role of Ganoderma lucidum uptake on some hematological and immunological response in patients with coronavirus (COVID-19). Syst. Rev. Pharm. 2020, 11, 537–541. [Google Scholar]
- Cheng, P.W.; Ng, L.T.; Chiang, L.C.; Lin, C.C. Antiviral effects of saikosaponins on human coronavirus 229E in vitro. Clin. Exp. Pharmacol. Physiol. 2006, 33, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, A.; Kostanjsek, R.; Rákhely, G.; Knezevic, P. The first Siphoviridae family bacteriophages infecting Bordetella bronchiseptica isolated from environment. Microb. Ecol. 2017, 73, 368–377. [Google Scholar] [CrossRef]
- Ryvarden, L. The Polyporaceae of North Europe. Albatrellus Incrustoporia 1976, 1, 214. [Google Scholar]
- Bernicchia, A. Fungi Europaei, Polyporaceae. Lomazzo 2005, 808. [Google Scholar]
- Orčić, D.; Francišković, M.; Bekvalac, K.; Svirčev, E.; Beara, I.; Lesjak, M.; Mimica-Dukić, N. Quantitative determination of plant phenolics in Urtica dioica extracts by high-performance liquid chromatography coupled with tandem massspectrometric detection. Food Chem. 2014, 143, 48–53. [Google Scholar] [CrossRef]
- M27-A2; Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts. Approved. Standard. 2nd ed. NCCLS: Wayne, PA, USA, 2002; ISBN 1-56238-469-4.
- M38; Reference Method for Broth Dilution Antifungal Susceptibility Test of Filamentous Fungi. 3rd ed. Clinical and Laboratory Standard Institute: Wayne, PA, USA, 2017; ISBN 1-56238-830-4.
- M45; Methods for Antimicrobial Dilution and Disk Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria. 3rd ed. Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2016.
- Hudzicki, J. Kirby-Bauer Disk Diffusion Susceptibility Test Protocol; American Society for Microbiology: Washington, DC, USA, 2009. [Google Scholar]
- Ngai, P.H.; Ng, T.B. Lectin, a novel and potent antifungal protein from shitake mushroom with inhibitory effects on activity of human immunodeficiency virus-1 reverse transcriptase and proliferation of leukemia cells. Life Sci. 2003, 73, 63–74. [Google Scholar] [CrossRef]
- M27-A3; Supplement Reference method for broth dilution antifungal susceptibility testing of yeasts. approved standard. 3rd ed. Clinical Laboratory Standards Institute: Wayne, PA, USA, 2008.
- M100; Performance Standards for Antimicrobial Susceptibility Testing. 27th ed. CLSI supplement. Clinical and Laboratory Standards Institute CLSI: Wayne, PA, USA, 2017.
- Carlson, K. Working with bacteriophages: Common techniques and methodological approaches. In Bacteriophages: Biology and Applications; Kutter, E., Sulakvelidze, A., Eds.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]

| Class | Compound | G. applanatum | G. resinaceum |
|---|---|---|---|
| Biflavonoid | Amentoflavone | 4.13 | 18.95 |
| Flavone | Apigenin | 35.12 | 47.87 |
| Flavonol | Rutin | 26.20 | 32.18 |
| Hydroxybenzoic acids | p-Hydroxybenzoic acid | 57.85 | <48.85 |
| 2,5-dihydroxybenzoic acid | 60.12 | 79.50 | |
| Chlorogenic acid | 5-O-Caffeoylquinic acid | 8.22 | 7.50 |
| G. resinaceum | G. pfeifferi | G. lucidum | G. applanatum | |||||
|---|---|---|---|---|---|---|---|---|
| Agar-well diffusion assay—inhibition zone (mm) | ||||||||
| B. cereus ATCC 11778 | 13 | 19 | 11 | 13 | ||||
| S. aureus ATCC 255923 | 11 | 13 | - | 16 | ||||
| S. aureus ATCC 6538 | 7 | 13 | - | - | ||||
| E. faecalis ATCC 19433 | - | - | - | - | ||||
| E. coli ATCC 11775 | - | 21 | 16 | - | ||||
| E. coli ATCC 11229 | - | 16 | 7 | - | ||||
| P. aeruginosa ATCC 3554 | - | - | - | - | ||||
| K. aerogenes ATCC 13048 | - | - | 9 | - | ||||
| P. mirabilis ATCC 12453 | - | - | - | - | ||||
| Disk-diffusion assay—inhibition zone (mm) | ||||||||
| B. cereus ATCC 11778 | 10 | 13 | 11 | - | ||||
| S. aureus ATCC 255923 | 11 | - | - | 21 | ||||
| S. aureus ATCC 6538 | - | - | 11 | 9 | ||||
| E. faecalis ATCC 19433 | 10 | - | - | - | ||||
| E. coli ATCC 11775 | 7 | 11 | - | - | ||||
| E. coli ATCC 11229 | 8 | 9 | - | - | ||||
| P. aeruginosa ATCC 3554 | 10 | - | - | - | ||||
| K. aerogenes ATCC 13048 | 10 | 10 | - | - | ||||
| P. mirabilis ATCC 12453 | - | - | - | - | ||||
| Microdilution assay—MIC * and MBC ** | ||||||||
| MIC (mg/mL) | MBC (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | |
| B. cereus ATCC 11778 | 100 | 100 | 200 | ↑ 200 | 200 | ↑ 200 | 100 | ↑ 100 |
| S. aureus ATCC 255923 | 100 | ↑ 100 | 200 | ↑ 200 | - | - | 100 | ↑ 100 |
| S. aureus ATCC 6538 | 100 | ↑↑ 100 | 200 | ↑ 200 | - | - | 100 | ↑ 100 |
| E. faecalis ATCC 19433 | - | - | - | - | - | - | - | - |
| E. coli ATCC 11775 | - | - | - | - | - | - | - | - |
| E. coli ATCC 11229 | 100 | ↑ 100 | 100 | - | - | - | - | - |
| P. aeruginosa ATCC 3554 | - | - | - | - | - | - | - | - |
| K. aerogenes ATCC 13048 | - | - | - | - | - | - | - | - |
| P. mirabilis ATCC 12453 | 100 | ↑ 100 | 200 | ↑ 200 | 200 | ↑ 200 | - | - |
| Bacterial Strain | G. applanatum | G. resinaceum | Referent Antibiotic- Streptomicin | |||
|---|---|---|---|---|---|---|
| MIC (mg/mL) | MBC (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | MIC (μg/mL) | MBC (μg/mL) | |
| B. cereus HP | 12.5 | 25 | 25 | 25 | 8 | 32 |
| E. coli ATCC 11229 | 12.5 | ↑↑ 25 | 12.5 | 50 | 4 | 16 |
| K. pneumoniae HP | 25 | 25 | 25 | ↑↑ 50 | 2 | 8 |
| P. aeruginosa ATCC 3554 | 25 | 25 | 6.25 | 50 | 16 | 64 |
| S. aureus ATCC 6538 | 25 | 25 | 12.5 | 25 | 10.25 | 1 |
| S. enteritidis ATCC 13076 | 25 | 25 | 12.5 | 25 | 8 | 64 |
| Work Concentration of Extract (mg/mL) | Inhibition (%) | |||
|---|---|---|---|---|
| G. applanatum | G. lucidum | G. pfeifferi | G. resinaceum | |
| EtOH extracts | ||||
| 100 | 15.73 ± 1.74 a | 31.66 ± 1.76 a,1 | 10.95 ± 0.87 b | 33.69 ± 2.55 c,1 |
| 50 | 7.05 ± 0.87 a | 33.54 ± 7.31 a | 20.22 ± 2.14 b | 20.80 ± 3.14 c |
| 20 | 17.90 ± 0.43 a | 24.56 ±2.47 a | 25.72 ± 2.30 b | 21.96 ± 1.53 c |
| 10 | 4.87 ± 1.30 a | 23.70 ± 0.66 a | n.a. | 20.51 ± 3.04 c |
| 2 | 12.11 ± 0.90 a | 37.16 ±1.33 a | 28.76 ± 3.39 b | 20.22 ± 1.40 c |
| 0.2 | 16.17 ± 1.57 a | 40.93 ± 1.15 a | 20.95 ± 2.30 b | 20.22 ± 1.33 c |
| EtOH extract solution in 5% DMSO | ||||
| 200 | n.a. | n.a. | 70.03 ± 3.18 a | n.d. |
| 100 | 65.83 ± 1.75 a | n.a. | 51.12 ± 1.59 a | n.d. |
| 50 | 70.73 ± 1.70 a | 47.62 ± 0.64 a | 57.00 ± 4.72 a | n.d. |
| 20 | 48.46 ± 1.06 a | 58.82 ± 3.17 a | 17.37 ± 4.15 a | n.d. |
| 10 | 53.92 ± 3.99 a | 55.32 ± 3.26 a | 41.60 ± 1.68 a | n.d. |
| 2 | 57.00 ± 1.35 a | 57.00 ± 4.37 a | 42.02 ± 1.68 a | n.d. |
| 0.2 | n.a. | 73.39 ± 1.35 a | n.a. | n.a. |
| H2O extracts | ||||
| 100 | 11.02 ± 1.87 a | 3.27 ± 1.15 a,1 | 22.18 ± 1.31 b | 26.26 ± 1.31 c,1 |
| 50 | 20.00 ± 6.41 a | n.d. | 18.78 ± 5.93 b | 28.30 ± 3.32 c |
| 20 | 5.72 ± 1.08 a | 21.63 ± 0.82 a | 19.32 ± 1.03 b | 12.79 ± 3.43 c |
| 10 | n.d. | 29.25 ± 1.55 a | 8.16 ± 2.55 b | 29.39 ± 3.56 c |
| 2 | 21.90 ± 0.47 a | 27.21 ± 2.62 a | 1.77 ± 0.85 b | 25.85 ± 3.09 c |
| 0.2 | 4.35 ± 0.85 a | 12.38 ± 4.78 a | 10.34 ± 0.62 b | 22.99 ± 7.90 c |
| Species | Extract Type | Assays | Activity |
|---|---|---|---|
| G. resinaceum | H2O | Agar-well diffusion | AB |
| Disk-diffusion | |||
| Microdilution | |||
| CHCl3 | Microdilution | AB | |
| EtOH | Disk-diffusion | AF | |
| Pour plate | |||
| G. applanatum | H2O | Agar-well diffusion | AB |
| Disk-diffusion | |||
| Microdilution | |||
| CHCl3 | Microdilution | AB | |
| G. pfeifferi | H2O | Agar-well diffusion | AB |
| Disk-diffusion | |||
| Microdilution | |||
| EtOH | Disk-diffusion | AF | |
| Pour plate | |||
| G. lucidum | H2O | Agar-well diffusion | AB |
| Disk-diffusion | |||
| Microdilution |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rašeta, M.; Mišković, J.; Čapelja, E.; Zapora, E.; Petrović Fabijan, A.; Knežević, P.; Karaman, M. Do Ganoderma Species Represent Novel Sources of Phenolic Based Antimicrobial Agents? Molecules 2023, 28, 3264. https://doi.org/10.3390/molecules28073264
Rašeta M, Mišković J, Čapelja E, Zapora E, Petrović Fabijan A, Knežević P, Karaman M. Do Ganoderma Species Represent Novel Sources of Phenolic Based Antimicrobial Agents? Molecules. 2023; 28(7):3264. https://doi.org/10.3390/molecules28073264
Chicago/Turabian StyleRašeta, Milena, Jovana Mišković, Eleonora Čapelja, Ewa Zapora, Aleksandra Petrović Fabijan, Petar Knežević, and Maja Karaman. 2023. "Do Ganoderma Species Represent Novel Sources of Phenolic Based Antimicrobial Agents?" Molecules 28, no. 7: 3264. https://doi.org/10.3390/molecules28073264
APA StyleRašeta, M., Mišković, J., Čapelja, E., Zapora, E., Petrović Fabijan, A., Knežević, P., & Karaman, M. (2023). Do Ganoderma Species Represent Novel Sources of Phenolic Based Antimicrobial Agents? Molecules, 28(7), 3264. https://doi.org/10.3390/molecules28073264











