Design, Synthesis, Anti-Varicella-Zoster and Antimicrobial Activity of (Isoxazolidin-3-yl)Phosphonate Conjugates of N1-Functionalised Quinazoline-2,4-Diones
Abstract
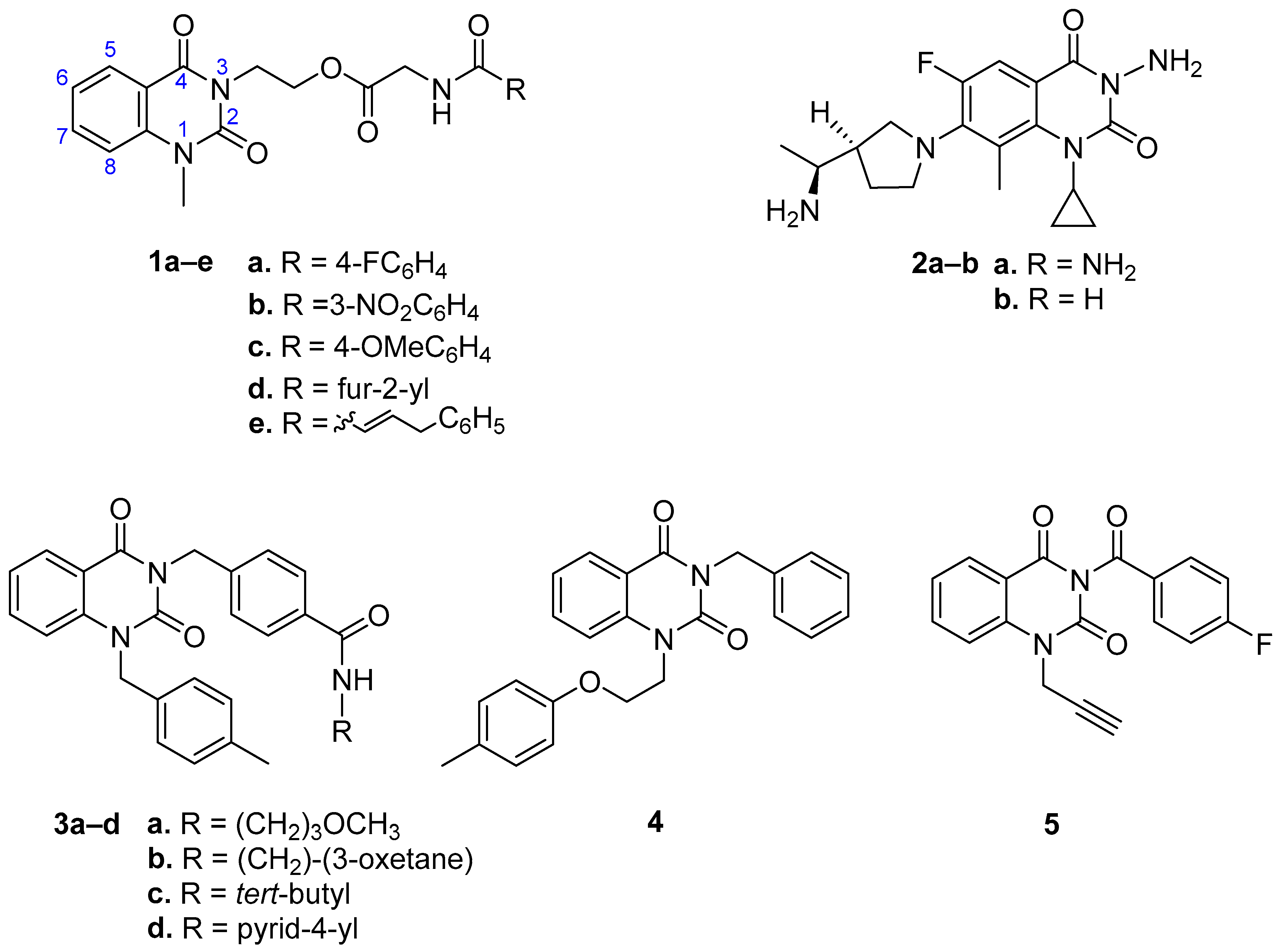
1. Introduction
2. Results and Discussion
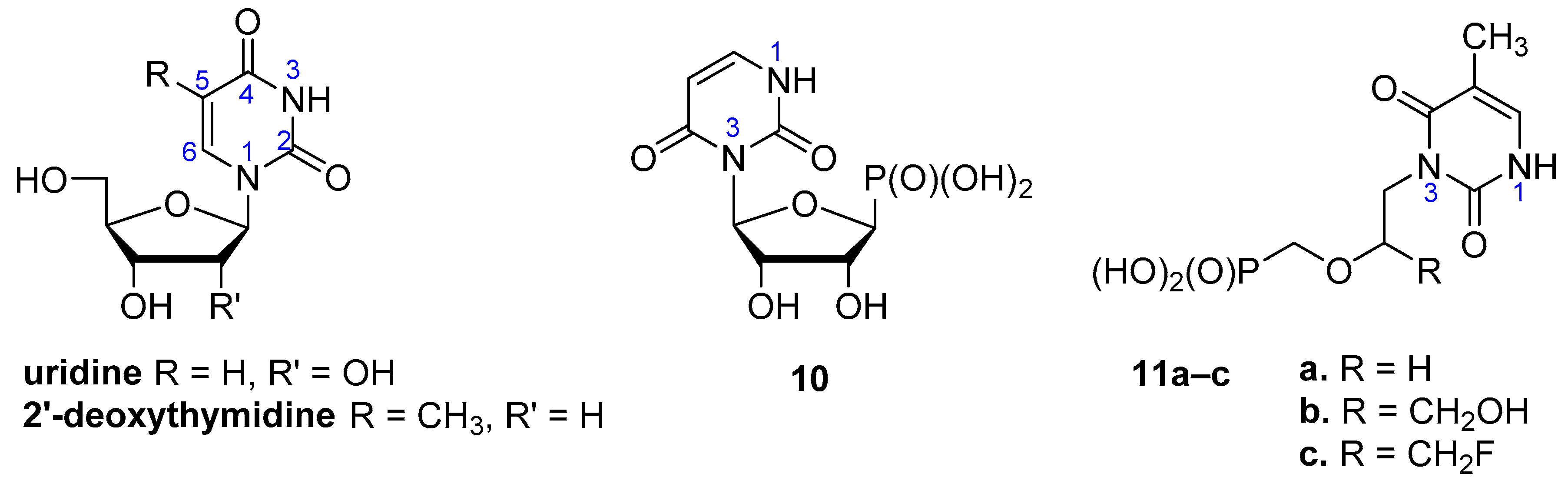
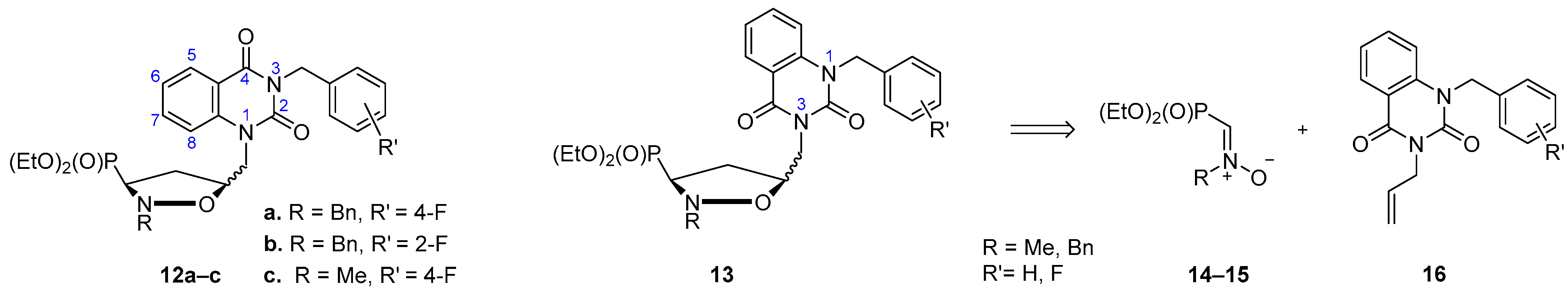
2.1. Chemistry
2.2. Antiviral and Antimicrobial Evaluation
2.2.1. Antiviral Activity
2.2.2. Antimicrobial Activity
3. Materials and Methods
3.1. General Information
3.2. General Procedure for Benzylation of 2H-Benzo[d][1,3]Oxazine-2,4-Diones 25
3.3. General Procedure for the Synthesis of 1-Benzylquinazoline-2,4-Diones 26a–d
3.4. General Procedure for Allylation of 1-Benzylquinazolin-2,4-Dione 27a–d
3.5. General Procedure for the Synthesis of Isoxazolidines cis-20 and trans-20 As Well As cis-21 and trans-21
3.6. Antiviral Activity Assays
3.7. Antibacterial Activity Assays
3.8. Microbial Mutagenicity Assay—The Ames Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Głowacka, I.E.; Balzarini, J.; Andrei, G.; Snoeck, R.; Schols, D.; Piotrowska, D.G. Design, synthesis, antiviral and cytostatic activity of omega-(1H-1,2,3-triazol-1-yl)(polyhydroxy)alkylphosphonates as acyclic nucleotide analogues. Bioorg. Med. Chem. 2014, 22, 3629–3641. [Google Scholar] [CrossRef] [PubMed]
- Kakuta, H.; Koiso, Y.; Nagasawa, K.; Hashimoto, Y. Fluorescent bioprobes for visualization of puromycin-sensitive aminopeptidase in living cells. Bioorg. Med. Chem. Lett. 2003, 13, 83–86. [Google Scholar] [CrossRef]
- Nencini, A.; Pratelli, C.; Quinn, J.M.; Salerno, M.; Tunici, P.; De Robertis, A.; Valensin, S.; Mennilo, F.; Rossi, M.; Bakker, A.; et al. Structure-activity relationship and properties optimization of a series of quinazoline-2,4-diones as inhibitors of the canonical Wnt pathway. Eur. J. Med. Chem. 2015, 95, 526–545. [Google Scholar] [CrossRef] [PubMed]
- El-Adl, K.; El-Helby, A.G.A.; Sakr, H.; El-Hddad, S.S.A. Design, synthesis, molecular docking, and anticancer evaluations of 1-benzylquinazoline-2,4(1H,3H)-dione bearing different moieties as VEGFR-2 inhibitors. Arch. Pharm. 2020, 353, e2000068. [Google Scholar] [CrossRef] [PubMed]
- Aboelmagd, A.; Salem, E.M.S.; Ali, I.A.I.; Gomaa, M.S. Synthesis of quinazolindionyl amino acid and hydrazone derivatives as possible antitumour agents. Arkivoc 2018, 20–35. [Google Scholar] [CrossRef]
- Havera, H.J.; Vidrio, H. Derivatives of 1,3-disubstituted 2,4(1H,3H)-quinazolinediones as possible peripheral vasodilators or antihypertensive agents. J. Med. Chem. 1979, 22, 1548–1550. [Google Scholar] [CrossRef]
- Goto, S.; Tsuboi, H.; Kanoda, M.; Mukai, K.; Kagara, K. The process development of a novel aldose reductase inhibitor, FK366. Part 1. Improvement of discovery process and new syntheses of 1-substituted quinazolinediones. Org. Process Res. Dev. 2003, 7, 700–706. [Google Scholar] [CrossRef]
- Prashanth, M.K.; Madaiah, M.; Revanasiddappa, H.D.; Veeresh, B. Synthesis, anticonvulsant, antioxidant and binding interaction of novel N-substituted methylquinazoline-2,4(1H,3H)-dione derivatives to bovine serum albumin: A structure-activity relationship study. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 110, 324–332. [Google Scholar] [CrossRef]
- Ji, Q.G.; Yang, D.; Deng, Q.; Ge, Z.Q.; Yuan, L.J. Design, synthesis, and evaluation of novel 1-methyl-3-substituted quinazoline-2,4-dione derivatives as antimicrobial agents. Med. Chem. Res. 2014, 23, 2169–2177. [Google Scholar] [CrossRef]
- Huband, M.D.; Cohen, M.A.; Zurack, M.; Hanna, D.L.; Skerlos, L.A.; Sulavik, M.C.; Gibson, G.W.; Gage, J.W.; Ellsworth, E.; Stier, M.A.; et al. In vitro and in vivo activities of PD 0305970 and PD 0326448, new bacterial gyrase/topoisomerase inhibitors with potent antibacterial activities versus multidrug-resistant gram-positive and fastidious organism groups. Antimicrob. Agents Chemother. 2007, 51, 1191–1201. [Google Scholar] [CrossRef]
- Matharu, D.S.; Flaherty, D.P.; Simpson, D.S.; Schroeder, C.E.; Chung, D.; Yan, D.; Noah, J.W.; Jonsson, C.B.; White, E.L.; Aubé, J.; et al. Optimization of potent and selective quinazolinediones: Inhibitors of respiratory syncytial virus that block RNA-dependent RNA-polymerase complex activity. J. Med. Chem. 2014, 57, 10314–10328. [Google Scholar] [CrossRef] [PubMed]
- Novikov, M.S.; Valuev-Elliston, V.T.; Babkov, D.A.; Paramonova, M.P.; Ivanov, A.V.; Gavryushov, S.A.; Khandazhinskaya, A.L.; Kochetkov, S.N.; Pannecouque, C.; Andrei, G.; et al. N1,N3-disubstituted uracils as nonnucleoside inhibitors of HIV-1 reverse transcriptase. Bioorg. Med. Chem. 2013, 21, 1150–1158. [Google Scholar] [CrossRef] [PubMed]
- Głowacka, I.E.; Andrei, G.; Schols, D.; Snoeck, R.; Gawron, K. Design, Synthesis, and the biological evaluation of a new series of acyclic 1,2,3-triazole nucleosides. Arch. Pharm. 2017, 350, e1700166. [Google Scholar] [CrossRef] [PubMed]
- Głowacka, I.E.; Balzarini, J.; Wróblewski, A.E. The synthesis, antiviral, cytostatic and cytotoxic evaluation of a new series of acyclonucleotide analogues with a 1,2,3-triazole linker. Eur. J. Med. Chem. 2013, 70, 703–722. [Google Scholar] [CrossRef] [PubMed]
- Andreeva, O.V.; Garifullin, B.F.; Zarubaev, V.V.; Slita, A.V.; Yesaulkova, I.L.; Saifina, L.F.; Shulaeva, M.M.; Belenok, M.G.; Semenov, V.E.; Kataev, V.E. Synthesis of 1,2,3-triazolyl nucleoside analogues and their antiviral activity. Mol. Divers. 2021, 25, 473–490. [Google Scholar] [CrossRef]
- Polákova, I.; Buděšínský, M.; Točík, Z.; Rosenberg, I. Tetrofuranose nucleoside phosphonic acids: Synthesis and properties. Collect. Czech. Chem. Commun. 2011, 76, 503–536. [Google Scholar] [CrossRef]
- Pomeisl, K.; Holý, A.; Votruba, I.; Pohl, R. Syntheses of N-3-substituted thymine acyclic nucleoside phosphonates and a comparison of their inhibitory effect towards thymidine phosphorylase. Bioorg. Med. Chem. Lett. 2008, 18, 1364–1367. [Google Scholar] [CrossRef]
- Piotrowska, D.G.; Andrei, G.; Schols, D.; Snoeck, R.; Lysakowska, M. Synthesis, anti-varicella-zoster virus and anti-cytomegalovirus activity of quinazoline-2,4-diones containing isoxazolidine and phosphonate substructures. Eur. J. Med. Chem. 2017, 126, 84–100. [Google Scholar] [CrossRef]
- Piotrowska, D.G. N-Substituted C-diethoxyphosphorylated nitrones as useful synthons for the synthesis of alpha-aminophosphonates. Tetrahedron Lett. 2006, 47, 5363–5366. [Google Scholar] [CrossRef]
- Hardtmann, G.E.; Koletar, G.; Pfister, O.R. Chemistry of 2H-3,1-benzoxazine-2,4(1H)dione (isatoic anhydrides). 1. Synthesis of N-substituted 2H-3,1-benzoxazine-2,4(1H)diones. J. Heterocycl. Chem. 1975, 12, 565–572. [Google Scholar] [CrossRef]
- Łysakowska, M.; Balzarini, J.; Piotrowska, D.G. Design, synthesis, antiviral, and cytostatic evaluation of novel isoxazolidine analogs of homonucleotides. Arch. Pharm. Chem. Life Sci. 2014, 347, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, D.G.; Wróblewski, A.E.; Balzarini, J.; Andrei, G.; Schols, D.; Snoeck, R.; Felczak, A.; Wrońska, N.; Lisowska, K.; Głowacka, I. Synthesis and biological activity of novel phenyl-conjugates of isoxazolidines. Acta Pol. Pharm. 2017, 74, 1091–1100. [Google Scholar]
- Boshta, N.M.; El-Essawy, F.A.; Alshammari, M.B.; Noreldein, S.G.; Darwesh, O.M. Discovery of Quinazoline-2,4(1H,3H)-Dione Derivatives as Potent Antibacterial Agent: Design, Synthesis, and Their Antibacterial Activity. Molecules 2022, 27, 3853. [Google Scholar] [CrossRef]
- Noureldin, N.A.; Kothayer, H.; Lashine, E.M.; Baraka, M.M.; Huang, Y.; Li, B.; Ji, Q. Design, synthesis and biological evaluation of novel quinazoline-2,4-diones conjugated with different amino acids as potent chitin synthase inhibitors. Eur. J. Med. Chem. 2018, 152, 560–569. [Google Scholar] [CrossRef] [PubMed]
- Ji, Q.; Yang, D.; Wang, X.; Chen, C.; Deng, Q.; Ge, Z.; Yuan, L.; Yang, X.; Liao, F. Design, synthesis and evaluation of novel quinazoline-2,4-dione derivatives as chitin synthase ihbibitors and antifungal agents. Bioorg. Med. Chem. 2014, 22, 3405–3413. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.T.; Tallent, S.M. Screening food for Bacillus cereus toxins using whole genome sequencing. Food Microbial. 2019, 78, 164–170. [Google Scholar] [CrossRef]
- Holzel, C.S.; Tetens, J.L.; Schwaiger, K. Unreveling the Role of Vegetables in Spreading Antimicrobial-Resistant Bacteria: A Need for Quantitative Risk Assessment. Foodborne Pathog. Dis. 2018, 15, 671–688. [Google Scholar] [CrossRef]
- Tejs, S. The Ames test: A methodological short review. Environ. Biotechnol. 2008, 4, 7–14. [Google Scholar]
- OECD. Test No. 471: Bacterial Reverse Mutation Test, OECD Guidelines for the Testing of Chemicals; OECD: Paris, France, 1997. [Google Scholar]
- ISO 10993-3:2014(E); Biological Evaluation of Medical Devices—Part 4: Tests for Genotoxicity, Carcinogenicity and Reproductive Toxicity. ISO: Geneva, Switzerland, 2014.
- ISO 10993-33:2015; Biological Evaluation of Medical Devices—Part 33: Guidance on Tests to Evaluate Genotoxicity—Supplement to ISO 10993-3. ISO: Geneva, Switzerland, 2015.


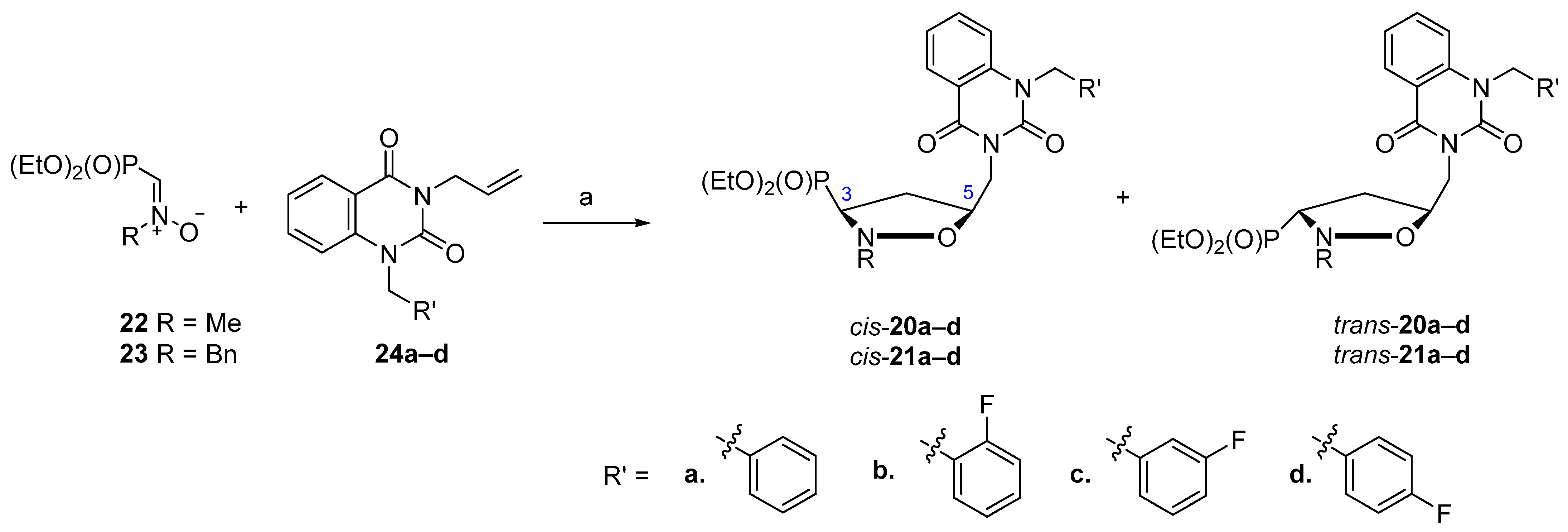
| Nitrone 22/23 (R) | Alkene 24 (R′) 13 | cis:trans Ratio | Yield (%) |
|---|---|---|---|
| 22 (Me) | Ph | 10:90 | cis-20a + trans-20a (87) a |
| 22 (Me) | 2-F-C6H4 | 8:92 | cis-20b + trans-20b (80) a |
| 22 (Me) | 3-F-C6H4 | 6:94 | cis-20c + trans-20c (96) a |
| 22 (Me) | 4-F-C6H4 | 10:90 | cis-20d + trans-20d (92) a |
| 23 (Bn) | Ph | 10:90 | cis-21a + trans-21a (95) a |
| 23 (Bn) | 2-F-C6H4 | 15:85 | cis-21b + trans-21b (71) a |
| 23 (Bn) | 3-F-C6H4 | 10:90 | cis-21c + trans-21c (91) a |
| 23 (Bn) | 3-F-C6H4 | 15:85 | cis-21d + trans-21d (84) a |
| Compound | R | R′ | Antiviral Activity EC50 (μM) a | Cytotoxicity (μM) | |
|---|---|---|---|---|---|
| TK+ VZV Strain (OKA) | TK– VZV Strain (07-1) | Cell Morphology MCC b | |||
| cis-20a/trans-20a (10:90) | Me | Ph | 31.68 | 100 | >100 |
| cis-20b/trans-20b (8:92) | Me | 2-F-C6H4 | 31.68 | 58.48 | >100 |
| cis-20c/trans-20c (6:94) | Me | 3-F-C6H4 | 100 | 58.48 | >100 |
| cis-20d/trans-20d (10:90) | Me | 4-F-C6H4 | 58.48 | 38.07 | >100 |
| cis-21a/trans-21a (10:90) | Bn | Ph | 14.5 | 34.2 | 100 |
| cis-21b/trans-21b (15:85) | Bn | 2-F-C6H4 | 12.63 | 27.59 | 100 |
| cis-21c/trans-21c (10:90) | Bn | 3-F-C6H4 | 20 | 14.49 | 20 |
| cis-21d/trans-21d (15:85) | Bn | 4-F-C6H4 | 20 | 20 | 100 |
| 24a | - | Ph | 100 | 100 | >100 |
| 24b | - | 2-F-C6H4 | >100 | 64.47 | >100 |
| 24c | - | 3-F-C6H4 | >100 | >100 | >100 |
| 24d | - | 4-F-C6H4 | 54.69 | >20 | 100 |
| Acyclovir | 0.49 | 23.22 | >440 | ||
| Brivudin | 0.026 | 12.01 | >300 | ||
| Compound | E. faecalis ATCC 29212 | S. aureus ATCC 2593 | B. cereus PCM 1948 | E. coli ATCC 25922 | P. aeruginosa ATCC 27853 | C. albicans ATCC 10241 | A. brasiliensis ATCC 16404 |
|---|---|---|---|---|---|---|---|
| MIC a/MBC b (mg/mL) | MIC a = MBC b (mg/mL) | ||||||
| cis-20a/trans-20a (10:90) | >5/>5 | 2.5 | 1.25 | 1.25 | 2.5 | 2.5 | 5 |
| cis-20b/trans-20b (8:92) | >5/>5 | 5 | 1.25 | 2.5 | 2.5 | 2.5 | 5 |
| cis-20c/trans-20c (6:94) | >5/>5 | 5 | 0.625 | 2.5 | 2.5 | 2.5 | 5 |
| cis-20d/trans-20d (10:90) | >5/>5 | 5 | 1.25 | 2.5 | 2.5 | 2.5 | 5 |
| cis-21a/trans-21a (10:90) | >5/>5 | 5 | 1.25 | 2.5 | 2.5 | 5 | 5 |
| cis-21b/trans-21b (15:85) | >5/>5 | 5 | 1.25 | 2.5 | 2.5 | 2.5 | 5 |
| cis-21c/trans-21c (10:90) | >5/>5 | 5 | 2.5 | 1.25 | 2.5 | 2.5 | 5 |
| cis-21d/trans-21d (15:85) | >5/>5 | 5 | 1.25 | 1.25 | 2.5 | 2.5 | 5 |
| 24a | >5/>5 | 5 | 5 | 2.5 | 2.5 | 5 | 5 |
| 24b | >5/>5 | 5 | 2.5 | 2.5 | 2.5 | 5 | 5 |
| 24c | >5/>5 | 5 | 5 | 1.25 | 2.5 | 2.5 | 5 |
| 24d | >5/>5 | 5 | 5 | 2.5 | 2.5 | 2.5 | 5 |
| Amikacin | 0.000625 | 0.001 | 0.02 | 0.000625 | 0.02 | - | - |
| Fluconazole | - | - | - | - | - | 0.005/>0.005 | 0.005/>0.005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Łysakowska, M.; Głowacka, I.E.; Andrei, G.; Schols, D.; Snoeck, R.; Lisiecki, P.; Szemraj, M.; Piotrowska, D.G. Design, Synthesis, Anti-Varicella-Zoster and Antimicrobial Activity of (Isoxazolidin-3-yl)Phosphonate Conjugates of N1-Functionalised Quinazoline-2,4-Diones. Molecules 2022, 27, 6526. https://doi.org/10.3390/molecules27196526
Łysakowska M, Głowacka IE, Andrei G, Schols D, Snoeck R, Lisiecki P, Szemraj M, Piotrowska DG. Design, Synthesis, Anti-Varicella-Zoster and Antimicrobial Activity of (Isoxazolidin-3-yl)Phosphonate Conjugates of N1-Functionalised Quinazoline-2,4-Diones. Molecules. 2022; 27(19):6526. https://doi.org/10.3390/molecules27196526
Chicago/Turabian StyleŁysakowska, Magdalena, Iwona E. Głowacka, Graciela Andrei, Dominique Schols, Robert Snoeck, Paweł Lisiecki, Magdalena Szemraj, and Dorota G. Piotrowska. 2022. "Design, Synthesis, Anti-Varicella-Zoster and Antimicrobial Activity of (Isoxazolidin-3-yl)Phosphonate Conjugates of N1-Functionalised Quinazoline-2,4-Diones" Molecules 27, no. 19: 6526. https://doi.org/10.3390/molecules27196526
APA StyleŁysakowska, M., Głowacka, I. E., Andrei, G., Schols, D., Snoeck, R., Lisiecki, P., Szemraj, M., & Piotrowska, D. G. (2022). Design, Synthesis, Anti-Varicella-Zoster and Antimicrobial Activity of (Isoxazolidin-3-yl)Phosphonate Conjugates of N1-Functionalised Quinazoline-2,4-Diones. Molecules, 27(19), 6526. https://doi.org/10.3390/molecules27196526









