Abietane Diterpenes of the Genus Plectranthus sensu lato
Abstract
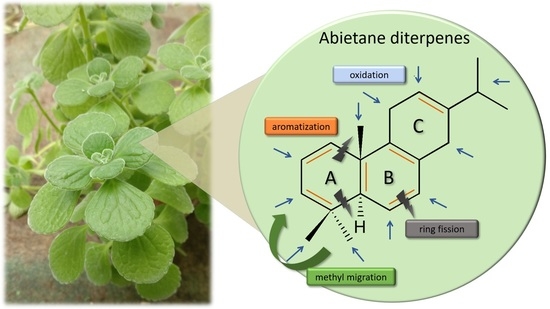
1. Introduction
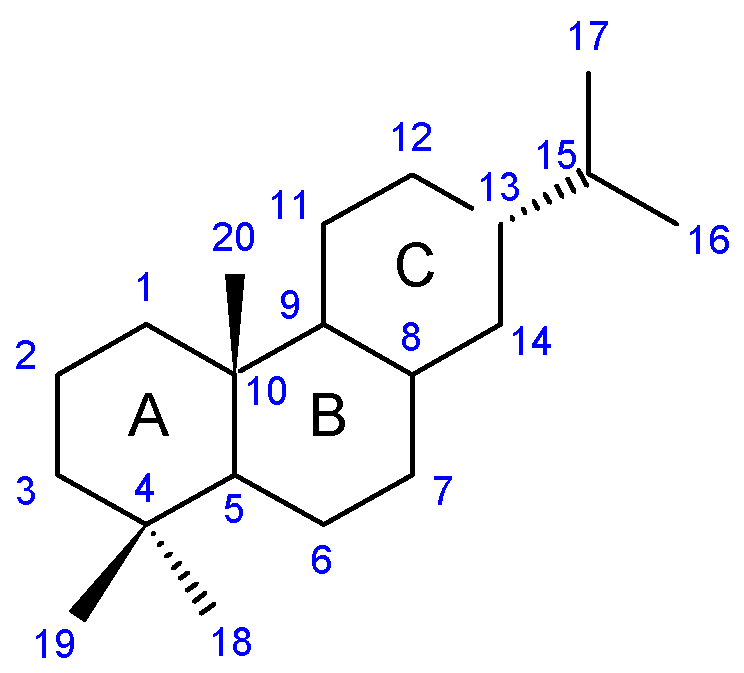
2. Abietane Diterpenes of the Genus Plectranthus s.l.
2.1. Royleanones
| (a) Royleanones with an Isopropyl Side Chain | |||||||
|---|---|---|---|---|---|---|---|
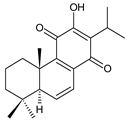 6,7-dehydroroyleanone (1) Coleus (Ethiopia) [33,34], C. amboinicus [35], C. bishopianus [36,37], C. forsteri ‘Marginatus’ [38], C. grandidentatus [39,40], C. lanuginosus [23], C. maculosus [41], C. madagascariensis [42,43] | |||||||
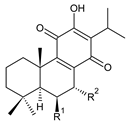 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | Source [References] | ||||
| royleanone (2) | –H | –H | C. (Ethiopia) [33,34], C. amboinicus [35], C. grandidentatus [39,40], C. maculosus [41] | ||||
| 6β-hydroxyroyleanone (3) | –OH | –H | C. amboinicus [35], C. grandidentatus [39,40], C. maculosus [41], C. sanguineus [28] | ||||
| horminone (syn. 7α-hydroxyroyleanone) (4) | –H | –OH | C. (Ethiopia) [33,34], C. amboinicus [35], C. grandidentatus [39,40], C. hereroensis [40,44,45], C. madagascariensis [46], C. sanguineus [28] | ||||
| 7-O-formylhorminone (5) | –H | –OCHO | C. sanguineus [28] | ||||
| 7α-acetoxyroyleanone (6) | –H | –OCOCH3 | C. (Ethiopia) [34], C. amboinicus [35] | ||||
| 6β,7α-dihydroxyroyleanone (7) | –OH | –OH | C. (Ethiopia) [33,34], C. (Rwanda) [29], C. amboinicus [35], C. argentatus [47], C. bishopianus [36,37], C. grandidentatus [40,48,49], C. hereroensis [50], C. maculosus subsp. edulis [26], C. madagascariensis [46], C. sanguineus [28] | ||||
| 7α-acetoxy-6β-hydroxyroyleanone (8) | –OH | –OCOCH3 | C. (Ethiopia) [33,34], C. amboinicus [35], C. argentatus [47], C. grandidentatus [39,40,48,49,51], C. hadiensis [16,52], C. madagascariensis [42,43,46], C. sanguineus [28] | ||||
| 7α-acyloxy-6β-hydroxyroyleanone (mixture of esters) (9) | –OH | –palmityloxy –stearyloxy –oleyloxy –n-heptadecanoyloxy –n-pentadecanoyloxy –myristyloxy | C. grandidentatus [39,48,49] | ||||
| 7α-formyloxy-6β-hydroxyroyleanone (10) | –OH | –OCHO | C. argentatus [47], C. hadiensis [16], C. hereroensis [50], C. madagascariensis [53], C. sanguineus [28] | ||||
| 6β-formyloxy-7α-hydroxyroyleanone (11) | –OCHO | –OH | C. argentatus [47] | ||||
| 6β-hydroxy-7α-methoxyroyleanone (12) | –OH | –OCH3 | C. bishopianus [36,37] | ||||
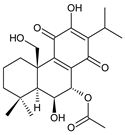 7α-acetoxy-6β,20-dihydroxyroyleanone (13) C. amboinicus [35] | |||||||
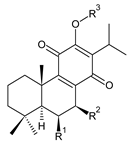 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | Source [References] | |||
| taxoquinone (syn. 7β-hydroxyroyleanone) (14) | –H | –OH | –H | C. (Ethiopia) [33,34], C. maculosus [41] | |||
| 7β-acetoxyroyleanone (15) | –H | –OCOCH3 | –H | C. maculosus [41] | |||
| 6β,7β-dihydroxyroyleanone (16) | –OH | –OH | –H | C. forsteri ‘Marginatus’ [38], C. hadiensis [52], C. maculosus [41], C. madagascariensis [42,43,54] | |||
| 7β-acetoxy-6β-hydroxyroyleanone (17) | –OH | –OCOCH3 | –H | C. forsteri ‘Marginatus’ [38], C. hadiensis [52], C. maculosus [41], C. madagascariensis [54] | |||
| 6β,7β-dihydroxy-12-O-methylroyleanone (18) | –OH | –OH | –CH3 | C. maculosus [41] | |||
| 7β-acetoxy-6β-hydroxy-12-O-methylroyleanone (19) | –OH | –OCOCH3 | –CH3 | C. maculosus [41] | |||
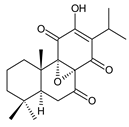 8α,9α-epoxy-7-oxoroyleanon (20) C. (Ethiopia) [34] | |||||||
 | |||||||
| Compound Name (Numerical ID) | R | Source [References] | |||||
| 8α,9α-epoxy-6-deoxycoleon U-quinone (21) | –H | C. maculosus [41] | |||||
| 8α,9α-epoxycoleon U-quinone (syn. 8α,9α-epoxy-8,9-dihydrocoleon U-quinone) (22) | –OH | C. argentatus [47], C. maculosus [41], C. sanguineus [28], C. xanthanthus [55] | |||||
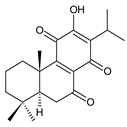 7-oxoroyleanone (syn. 7-ketoroyleanone) (23) C. (Ethiopia) [33,34] | 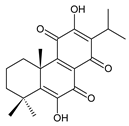 coleon U-quinone (24) C. argentatus [47], C. forsteri ‘Marginatus’ [56], C. maculosus [41], C. madagascariensis [46,54], C. sanguineus [28], C. xanthanthus [55] | ||||||
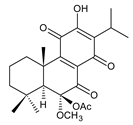 6β-acetoxy-6α-methoxy-7-oxoroyleanone (25) C. maculosus [41] | 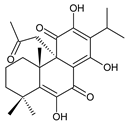 6,12,14-trihydroxy-9α-(2-oxopropyl)abieta-5,8(14),12-triene-7,11-dione (26) C. grandidentatus [51] | ||||||
| (b) Royleanones with a 1-Hydroxyprop-2-yl Side Chain | |||||||
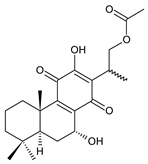 16-acetoxy-7α,12-dihydroxyabieta-8,12-diene-11,14-dione (27) C. hereroensis [40,45,57] | 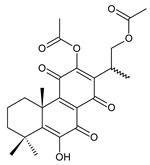 xanthanthusin H (28) C. xanthanthus [55] | 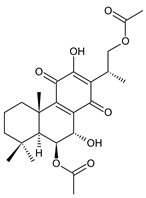 scutellarioidone B (29) C. scutellarioides [12] | |||||
| (c) Royleanones with a 2-Hydroxypropyl Side Chain | |||||||
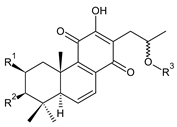 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | Source [References] | |||
| plectranthone G (30) | –H | –OH | –COCH3 | C. (Rwanda) [29] | |||
| plectranthroyleanone A (31) | –OH | –H | –CH3 | C. engleri [58] | |||
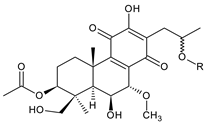 | |||||||
| Compound Name (Numerical ID) | R | Source [References] | |||||
| plectranthroyleanone B (32) | –CH3 | C. engleri [58] | |||||
| plectranthroyleanone C (33) | –H | C. engleri [58] | |||||
 17(15→16)-abeo-3α,18-bis(acetoxy)-6β,7α,16ξ-trihydroxyroyleanone (34) C. barbatus var. barbatus [25] | 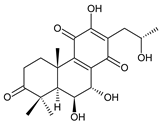 scutellarioidone A (35) C. scutellarioides [12,59] | 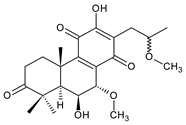 sincoetsin C (36) C. scutellarioides [59] | |||||
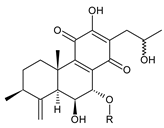 | |||||||
| Compound Name (Numerical ID) | R | Source [References] | |||||
| 17(15→16),19(4→3)-bis(abeo)-6β,7α,16ξ-trihydroxyroyleanone (37) | –H | C. barbatus var. barbatus [25], C. lanuginosus [60] | |||||
| 17(15→16),19(4→3)-bis(abeo)-6β, 16ξ-dihydroxy- 7α-methoxyroyleanone (38) | –CH3 | C. barbatus var. barbatus [25] | |||||
 17(15→16),18(4→3)-bis(abeo)-6β,7α,16ξ-trihydroxyroyleanone (39) C. lanuginosus [60], C. maculosus subsp. edulis [26] | |||||||
| (d) Royleanones with an Allyl Side Chain | |||||||
 | |||||||
| Compound Name (Numerical ID) | R | Source [References] | |||||
| lanugon A (40) | –H | C. lanuginosus [23], C. maculosus subsp. edulis [26] | |||||
| lanugon B (41) | –OH | C. lanuginosus [23] | |||||
| lanugon C (42) | –OCHO | C. lanuginosus [23] | |||||
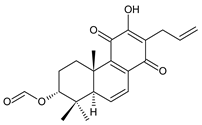 3α-formyloxylanugon A (43) C. maculosus subsp. edulis [26] | 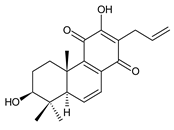 plectranthone F (44) C. (Rwanda) [29] | ||||||
 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | R5 | R6 | Source [References] |
| 7α,12-dihydroxy-17(15→16)-abeo-abieta-8,12,16- triene-11,14-dione (45) | –H | –H | –H | –H | –H | –OH | C. hereroensis [40,44] |
| 6β,7α-dihydroxy(allyl)royleanone (46) | –H | –H | –H | –H | –OH | –OH | C. sanguineus [28] |
| plectranthone H (47) | –H | –OH | –H | –H | –OCOCH3 | –H | C. (Rwanda) [29] |
| plectranthone I (48) | –H | –OH | –H | –H | –OCOCH3 | –OH | C. (Rwanda) [29] |
| lanugon D (49) | –H | –H | –OCHO | –H | –OH | –OH | C. lanuginosus [23] |
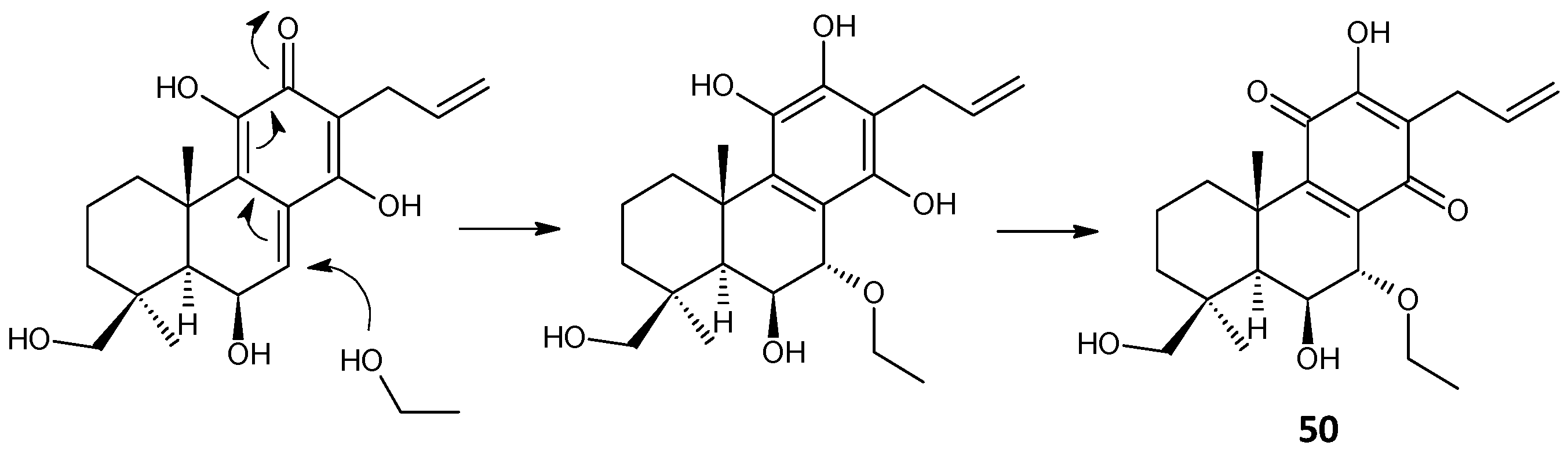
| lanugon E (50) | –H | –H | –OH | –H | –OH | –OCH2CH3 | C. lanuginosus [23] |
| 3α-formyloxy-6β,7α-dihydroxy(allyl)royleanone (51) | –OCHO | –H | –H | –H | –OH | –OH | C. maculosus subsp. edulis [26] |
| 18-acetoxy-3α-formyloxy-6β,7α-dihydroxy(allyl)royleanone (52) | –OCHO | –H | –H | –OCOCH3 | –OH | –OH | C. lanuginosus [60] |
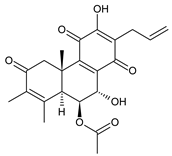 plectranthone J (53) C. (Rwanda) [29], C. barbatus [61] | |||||||
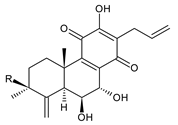 | |||||||
| Compound Name (Numerical ID) | R | Source [References] | |||||
| 17(15→16),18(4→3)-bis(abeo)-6β,7α,12-trihydroxyabieta-4(19),8,12,16-tetraene-11,14-dione (54) | –H | C. maculosus subsp. edulis [26] | |||||
| 3β-acetoxy-6β,7α,12-trihydroxy-17(15→16),18(4→3)-bis(abeo)-abieta-4(19),8,12,16-tetraene-11,14-dione (55) | –OCOCH3 | C. hereroensis [39,45] | |||||
| (e) Royleanones with a 3-Hydroxypropyl Side Chain | |||||||
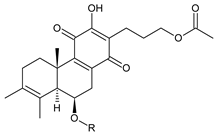 | |||||||
| Compound Name (Numerical ID) | R | Source [References] | |||||
| fredericon B (56) | –H | C. fredericii [62], C. scutellarioides [12] | |||||
| 6-acetylfredericone B (57) | –COCH3 | C. scutellarioides [12] | |||||
 scutellarioidone D (58) C. scutellarioides [12] | |||||||
| (f) Royleanones with a Dihydropyrane Side Chain | |||||||
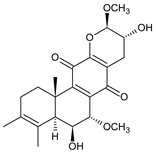 fredericone C (59) C. fredericii [62] | |||||||
| (g) Seco-Royleanones | |||||||
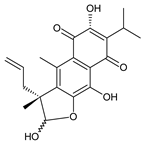 coleon A (60) C. barbatus var. grandis [63], C. aff. Gracilis [56], C. igniarius [64,65] | |||||||
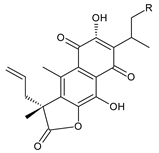 | |||||||
| Compound Name (Numerical ID) | R | Source [References] | |||||
| coleon A-lactone (61) | –H | C. gracilis [56], C. maculosus subsp. Edulis [26] | |||||
| 16-acetoxycoleon A-lactone (62) | –OCOCH3 | C. maculosus subsp. Edulis [26] | |||||
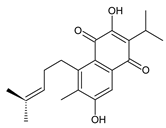 6,12-dihydroxysapriparaquinone (63) C. maculosus [41] | |||||||
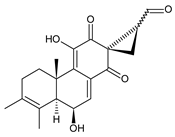
 plectrabarbene (64) C. barbatus [66] |  xanthanthusin E (65) C. xanthanthus [55] |  sanguinon A (66) C. sanguineus [28] | |||||
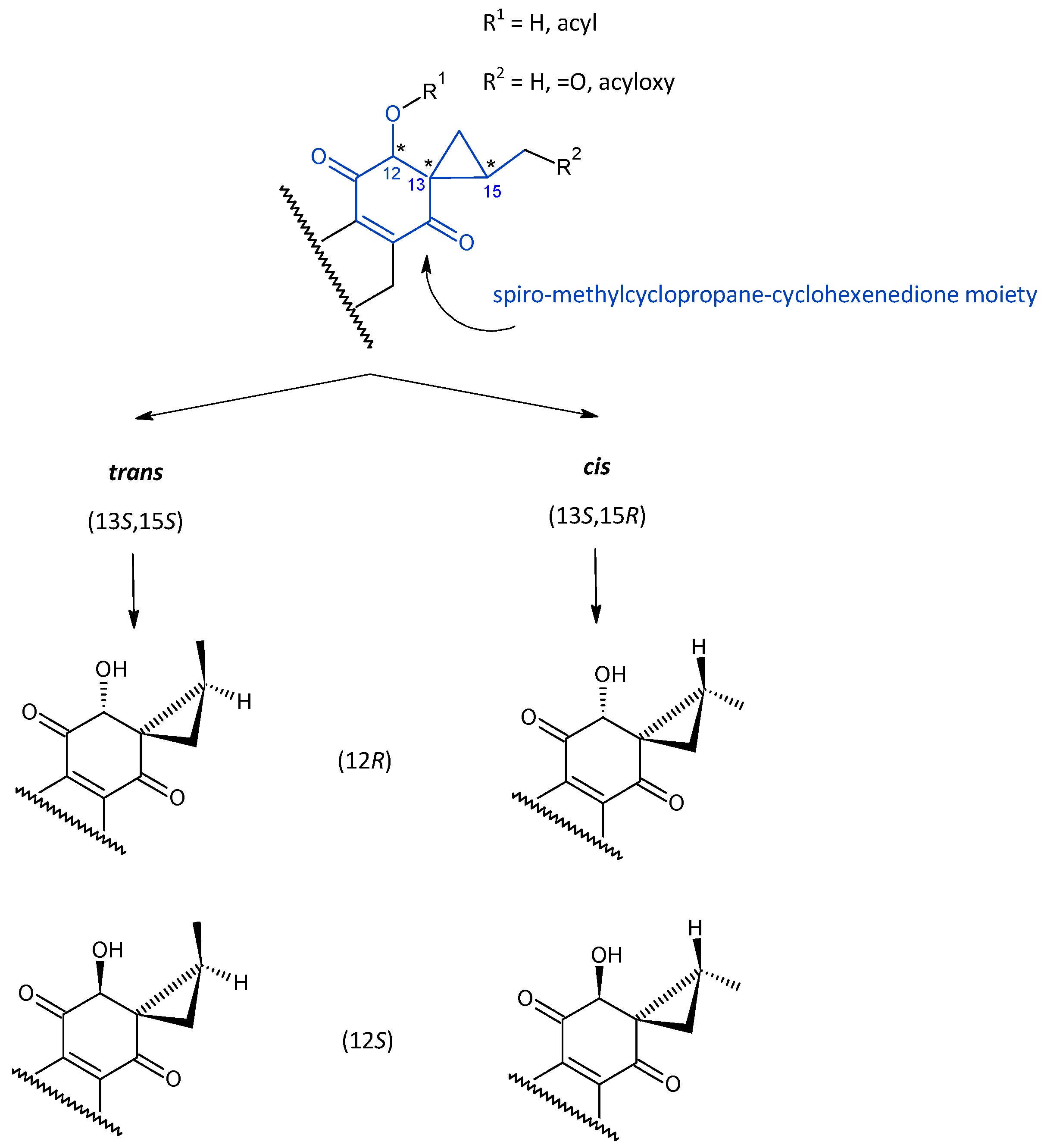
2.2. Spirocoleons
| (a) Compounds with the Unspecified Orientation in the Cyclopropane Ring | |||||||
|---|---|---|---|---|---|---|---|
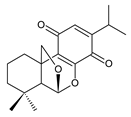
 fredericon A (67) C. fredericii [62] | |||||||
| (b) Type (13S,15S) | |||||||
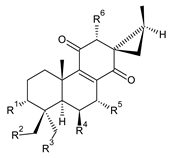 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | R5 | R6 | Source [References] |
| lanugon K (68) | –H | –H | –H | –OH | –OCOCH3 | –OH | C. lanuginosus [23], C. scutellarioides [12] |
| lanugon K’ (69) | –H | –H | –H | –OH | –OCHO | –OH | C. lanuginosus [23] |
| coleon Q (70) | –H | –H | –H | –OH | –OH | –OCOCH3 | C. caninus [24] |
| 12-O-desacetylcoleon Q (syn. 15-epilanugon F) (71) | –H | –H | –H | –OH | –OH | –OH | C. harmandii [71], C. lanuginosus [60] |
| 7-O-acetyl-12-O-desacetyl-19-hydroxycoleon Q (72) | –H | –OH | –H | –OH | –OCOCH3 | –OH | C. lanuginosus [60] |
| 18-acetoxy-12-O-desacetylcoleon Q (73) | –H | –H | –OCOCH3 | –OH | –OH | –OH | C. harmandii [71], C. lanuginosus [60] |
| 6-O-acetyl-19-acetoxycoleon Q (74) | –H | –OCOCH3 | –H | –OCOCH3 | –OH | –OCOCH3 | C. autranii [72], C. garckeanus [72] |
| 12-O-desacetyl-6-O-acetyl-19-acetoxycoleon Q (75) | –H | –OCOCH3 | –H | –OCOCH3 | –OH | –OH | C. harmandii [71] |
| 12-O-desacetyl-7-O-acetyl-19-acetoxycoleon Q (76) | –H | –OCOCH3 | –H | –OH | –OCOCH3 | –OH | C. autranii [72], C. garckeanus [72] |
| coleon R (77) | –OCOCH3 | –H | –H | –OCOCH3 | –OH | –OCOCH3 | C. caninus [24], C. comosus [73] |
| 12-O-desacetylcoleon R (78) | –OCOCH3 | –H | –H | –OCOCH3 | –OH | –OH | C. barbatus var. barbatus [25] |
| 6,12-bis(O-desacetyl)coleon R (79) | –OCOCH3 | –H | –H | –OH | –OH | –OH | C. barbatus var. barbatus [25] |
| coleon Y (80) | –OCOCH3 | –H | –OCOCH3 | –OH | –OH | –OH | C. barbatus var. barbatus [25], C. lanuginosus [60] |
| 3-O-desacetyl-3-O-formylcoleon Y (81) | –OCHO | –H | –OCOCH3 | –OH | –OH | –OH | C. barbatus var. barbatus [25], C. lanuginosus [60] |
| 3,18-bis(O-desacetyl)3,18-bis(O-formyl)coleon Y (82) | –OCHO | –H | –OCHO | –OH | –OH | –OH | C. lanuginosus [60] |
| (13S,15S)-6β,7α,12α,19-tetrahydroxy-13β,16-cycloabiet-8-ene-11,14-dione (83) | –H | –OH | –H | –OH | –OH | –OH | C. porcatus [74] |
| (13S,15S)-6β,12α-bis(acetoxy)-3α-formyloxy-7α-hydroxy-13β,16-cycloabiet-8-ene-11,14-dione (84) | –OCHO | –H | –H | –OCOCH3 | –OH | –OCOCH3 | C. maculosus subsp. edulis [26] |
| (13S,15S)-3α,7α-diformyloxy-6β,12α-dihydroxy-13,16-cycloabiet-8-ene-11,14-dione (85) | –OCHO | –H | –H | –OH | –OCHO | –OH | C. maculosus subsp. edulis [26] |
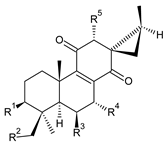 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | R5 | Source [References] | |
| 12-O-desacetyl-7-O-acetyl-3β,19-bis(acetoxy)coleon Q (86) | –OCOCH3 | –OCOCH3 | –OH | –OCOCH3 | –OH | C. autranii [72], C. garckeanus [72] | |
| 3β-hydroxy-3-deoxybarbatusine (87) | –OH | –H | –OCOCH3 | –OH | –OCOCH3 | C. barbatus [75], C. barbatus var. grandis [76], C. comosus [77] | |
| ornatin G (88) | –OH | –H | –OCOCH3 | –OH | –OH | C. comosus [77] | |
 barbatusine (89) C. barbatus [68,75,78], C. barbatus var. grandis [76], C. caninus [24] | 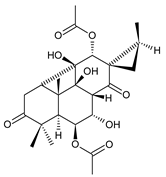 cyclobutatusine (90) C. barbatus [68,75,78] | ||||||
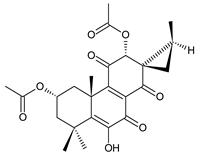 xanthanthusin I (91) C. xanthanthus [55] | 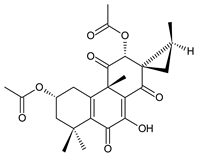 xanthanthusin J (92) C. xanthanthus [55] | 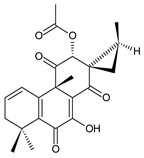 xanthanthusin K (93) C. xanthanthus [55] | |||||
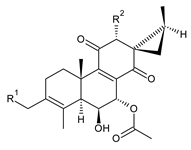 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | Source [References] | ||||
| coleon M (94) | –H | –OCOCH3 | C. caninus [24] | ||||
| coleon O (95) | –H | –OH | C. autranii [72], C. barbatus [79], C. barbatus var. barbatus [25], C. caninus [24], C. garckeanus [72], C. lanuginosus [24,60], C. scutellarioides [12,80] | ||||
| 19-acetoxycoleon O (syn. syl-C) (96) | –OCOCH3 | –OH | C. autranii [72,81] | ||||
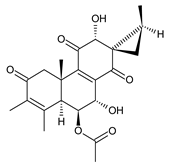 plectrin (97) C. barbatus [61,79] | |||||||
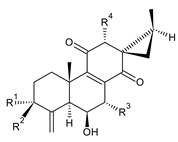 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | Source [References] | ||
| coleon G (98) | –CH3 | –H | –OCOCH3 | –OH | C. lanuginosus [60,69], C. scutellarioides [12] | ||
| coleon J (99) | –CH3 | –H | –OH | –OH | C. lanuginosus [60,69] | ||
| coleon N (100) | –H | –CH3 | –OCOCH3 | –OCOCH3 | C. caninus [24] | ||
| 7,12-bis(O-desacetyl)coleon N (101) | –H | –CH3 | –OH | –OH | C. barbatus var. barbatus [25] | ||
| 12-O-desacetylcoleon N (102) | –H | –CH3 | –OCOCH3 | –OH | C. barbatus var. barbatus [25] | ||
| 7-desoxy-12-O-desacetyl-3-acetoxycoleon N (103) | –OCOCH3 | –CH3 | –H | –OH | C. autranii [72] | ||
 syl-B (104) C. autranii [81] |  coleon Z (syn. syl-D) (105) C. autranii [72,81], C. garckeanus [72], C. maculosus subsp. edulis [26] | 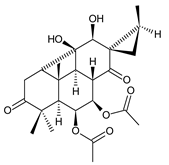 7β-acetyl-12-desacetoxycyclobutatusine (106) C. barbatus [78] | |||||
 | |||||||
| Compound Name (Numerical ID) | R | Source [References] | |||||
| spirocoleon-7-O-β-D-glucoside (107) | –H | C. scutellarioides [59,82] | |||||
| 3-hydroxyspirocoleon-7-O-β-D-glucoside (108) | –OH | C. scutellarioides [59] | |||||
| (c) Type (13S,15R) | |||||||
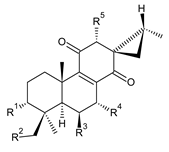 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | R5 | Source [References] | |
| lanugon F (109) | –H | –H | –OH | –OH | –OH | C. lanuginosus [23], C. maculosus subsp. edulis [26] | |
| lanugon G (110) | –H | –H | –OH | –OCHO | –OH | C. lanuginosus [23], C. maculosus subsp. edulis [26] | |
| lanugon H (111) | –H | –OH | –OH | –OCHO | –OH | C. lanuginosus [23] | |
| lanugon I (112) | –H | –OCHO | –OH | –OH | –OH | C. lanuginosus [23] | |
| lanugon J (113) | –H | –OCHO | –OH | –OCHO | –OH | C. lanuginosus [23] | |
| coleon P (114) | –H | –H | –OH | –OH | –OCOCH3 | C. caninus [24], revision [70] | |
| 12-O-desacetyl- 7-O-acetyl-19-acetoxycoleon P (115) | –H | –OCOCH3 | –OH | –OCOCH3 | –OH | C. autranii [72] | |
| plectranthone K (116) | –H | –H | –OH | –OCOCH3 | –OH | C. (Rwanda) [29] | |
| (13S,15R)-6β-acetoxy-3α-formyloxy-7α,12α-dihydroxy-13β,16-cycloabiet-8-ene-11,14-dione (117) | –OCHO | –H | –OCOCH3 | –OH | –OH | C. maculosus subsp. edulis [26] | |
| (13S,15R)-3α-formyloxy-6β,7α,12α-trihydroxy-13β,16-cycloabiet-8-ene-11,14-dione (118) | –OCHO | –H | –OH | –OH | –OH | C. maculosus subsp. edulis [26] | |
| (13S,15R)-3α,7α-diformyloxy-6β,12α-dihydroxy-13β,16-cycloabiet-8-ene-11,14-dione (119) | –OCHO | –H | –OH | –OCHO | –OH | C. maculosus subsp. edulis [26] | |
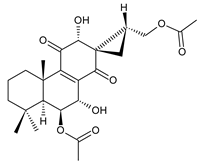 12-O-desacetyl-6-O-acetyl-17-acetoxycoleon P (syn. syl-A) (120) C. autranii [72,81], C. garckeanus [72] | 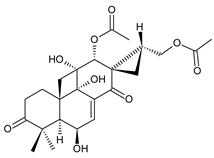 spiroscutelone A (121) C. scutellarioides [83] |  spiroscutelone B (122) C. scutellarioides [83] | |||||
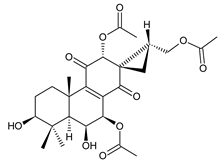 spiroscutelone C (123) C. scutellarioides [83] | 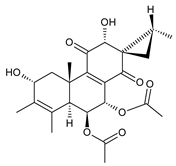 plectranthone L (124) C. (Rwanda) [29] | ||||||
 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | Source [References] | ||
| 15-epi-coleon J (125) | –CH3 | –H | –OH | –OH | C. maculosus subsp. edulis [26] | ||
| 7,12-bis(O-acetyl)-15-epi-coleon J (126) | –CH3 | –H | –OCOCH3 | –OCOCH3 | C. maculosus subsp. edulis [26] | ||
| 7-O-formyl-12-O-acetyl-15-epi-coleon J (127) | –CH3 | –H | –OCHO | –OCOCH3 | C. maculosus subsp. edulis [26] | ||
| 7-O-acetyl-15-epi-coleon J (128) | –CH3 | –H | –OCOCH3 | –OH | C. maculosus subsp. edulis [26] | ||
| 12-O-acetyl-15-epi-coleon J (129) | –CH3 | –H | –OH | –OCOCH3 | C. maculosus subsp. edulis [26] | ||
| 7-O-formyl-15-epi-coleon J (130) | –CH3 | –H | –OCHO | –OH | C. maculosus subsp. edulis [26] | ||
| 19(4→3)-abeo-3α,7α-bis(acetoxy)-6β,12α-dihydroxy-13β,16-cycloabieta-4(18),8-diene-11,14-dione (131) | –OCOCH3 | –CH3 | –OCOCH3 | –OH | C. maculosus subsp. edulis [26] | ||
 | |||||||
| Compound Name (Numerical ID) | R1 | R2 | Source [References] | ||||
| 12-O-acetyl-15-epi-coleon Z (syn.12-epi-mon-A) (132) | –OCOCH3 | –H | C. monostachyus [81,84], revision [70] | ||||
| 12-O-acetyl-7-O-formyl-7-O-desacetyl-15-epi-coleon Z (syn. 12-epi-mon-B) (133) | –OCHO | –H | C. monostachyus [81,84], revision [70] | ||||
| 12-O-acetyl-17-acetoxy-15-epi-coleon Z (syn. 12-epi-mon-C) (134) | –OCOCH3 | –OCOCH3 | C. monostachyus [81,84], revision [70] | ||||
| 12-O-acetyl-17-formyloxy-15-epi-coleon Z (135) | –OCOCH3 | –OCHO | C. monostachyus [81,84], revision [70] | ||||
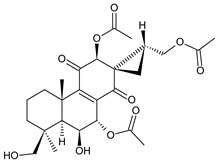 ornatin F (136) C. comosus [77] | |||||||
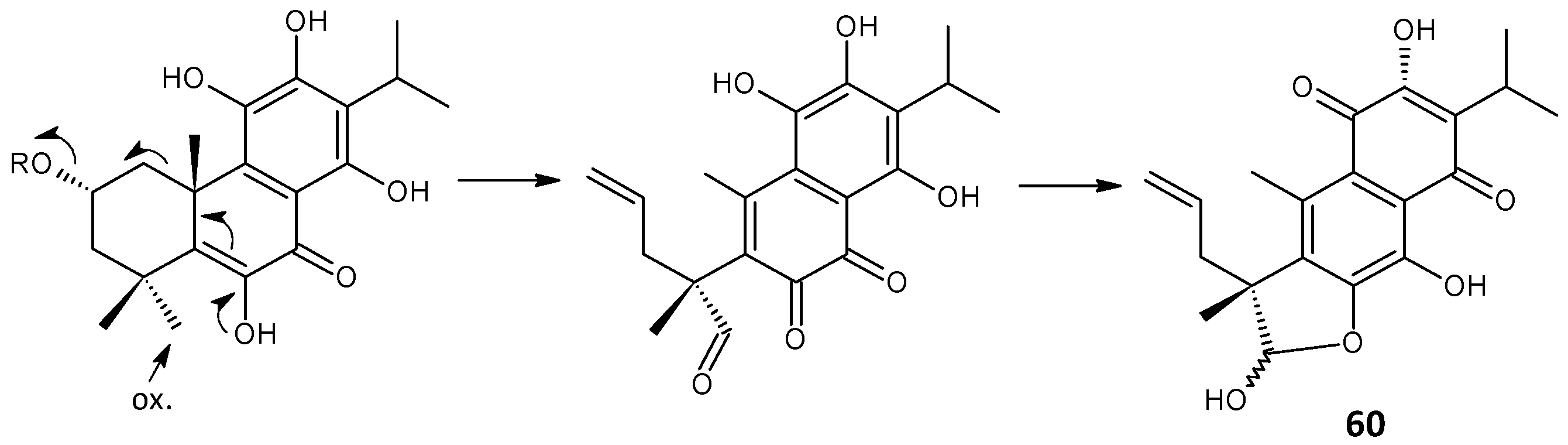
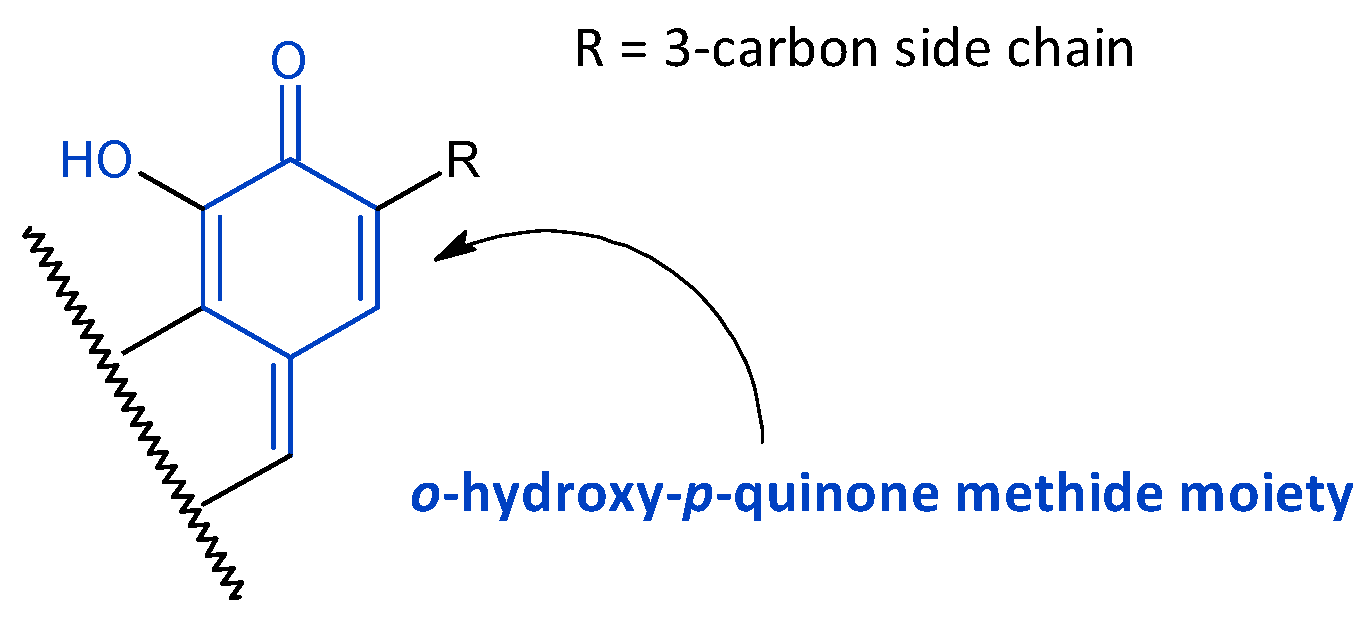
2.3. Quinone Methides
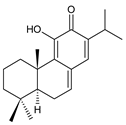 11-hydroxy-12-oxo-7,9(11),13-abietatriene (137) P. elegans [86] | ||
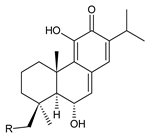 | ||
| Compound Name (Numerical ID) | R | Source [References] |
| 19-senecioyloxytaxodone (138) | (CH3)2C=CH–COO– | P. purpuratus [14] |
| 19-isovaleroyloxytaxodone (139) | (CH3)2CH–CH2–COO– | P. purpuratus [14] |
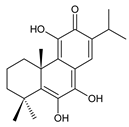 5,6-didehydro-7-hydroxytaxodone (140) C. barbatus [87] | ||
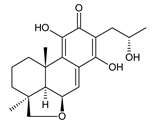 lanugon L (141) C. lanuginosus [23] | 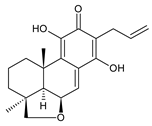 lanugon M (142) C. lanuginosus [23] | 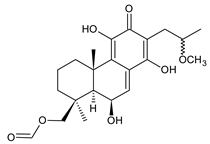 lanugon N (143) C. lanuginosus [23] |
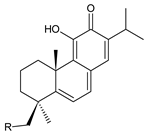 | ||
| Compound Name (Numerical ID) | R | Source [References] |
| parviflorone A (144) | (CH3)2C=CH–COO– | C. australis [88], P. lucidus [16], P. purpuratus [14], P. purpuratus subsp. purpuratus [16], P. strigosus [89] |
| 11-hydroxy-19-(3-methylbutanoyloxy)-5,7,9 (11),13-abietatetraen-12-one (145) | (CH3)2CH–CH2–COO– | P. purpuratus [14] |
| parviflorone B (146) | 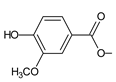 | C. australis [88], P. strigosus [89] |
| parviflorone C (147) |  | C. australis [88], P. pururatus subsp. tongaensis [16], P. strigosus [89] |
| parviflorone E (148) |  | C. australis [88], P. purpuratus subsp. tongaensis [16], P. strigosus [89], P. verticillatus [90] |
 | ||
| Compound Name (Numerical ID) | R | Source [References] |
| parviflorone D (149) | 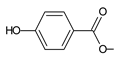 | C. australis [88], P. ecklonii [16,42,43,91,92], P. lucidus [16], P. strigosus [89,93] |
| parviflorone F (150) | 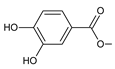 | C. australis [88], P. ecklonii [16,91], P. strigosus [89], P. verticillatus [90] |
| parviflorone G (151) | 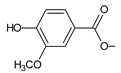 | P. strigosus [89] |
 parviflorone H (152) P. strigosus [89] |  fredericone D (153) C. fredericii [62] | |
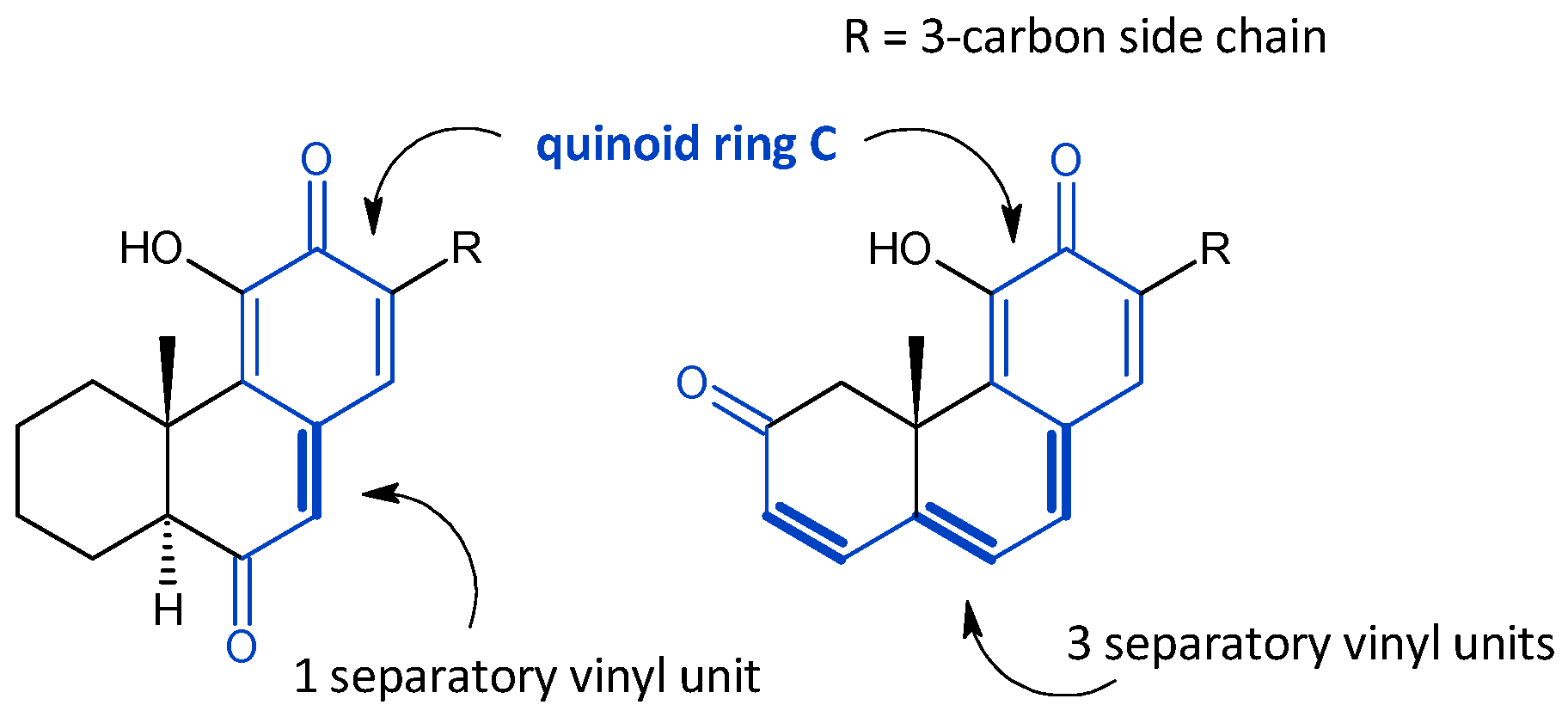
2.4. Vinylogous Quinones
| (a) Vinylogous Quinones with One Separatory Unit | |||
|---|---|---|---|
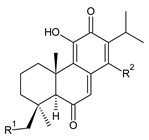 | |||
| Compound Name (Numerical ID) | R1 | R2 | Source [References] |
| taxodione (154) | –H | –H | C. barbatus [87] |
| 19-senecioyloxytaxodione (155) | (CH3)2C=CH–COO– | –H | P. purpuratus [14] |
| 19-isovaleroyloxytaxodione (156) | (CH3)2CH–CH2–COO– | –H | P. purpuratus [14] |
| 14-hydroxytaxodione (157) | –H | –OH | C. grandidentatus [95] |
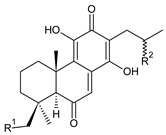 | |||
| Compound Name (Numerical ID) | R1 | R2 | Source [References] |
| 17(15→16)-abeo-16ξ-acetoxy-11,14-dihydroxy-7,9(11),13-abietatriene-6,12-dione (158) | –H | –OCOCH3 | C. maculosus subsp. edulis [26] |
| 17(15→16)-abeo-11,14,16ξ -trihydroxy-7,9(11),13-abietatriene-6,12-dione (159) | –H | –OH | C. maculosus subsp. edulis [26] |
| lanugon P (160) | –OCHO | –OH | C. lanuginosus [23] |
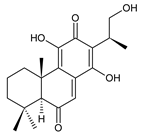 lanugon O (161) C. lanuginosus [23], C. maculosus subsp. edulis [26] | 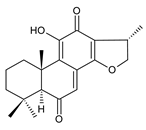 lanugon Q (162) C. lanuginosus [23] | ||
| (b) Vinylogous Quinones with Three Separatory Units | |||
 | |||
| Compound Name (Numerical ID) | R | Source [References] | |
| coleon E (163) | –CH2CH(OH)CH3 | C. (Rwanda) [29], C. barbatus [61,67,79,96] | |
| coleon F (164) | –CH2CH=CH2 | C. (Rwanda) [29], C. barbatus [61,97] | |
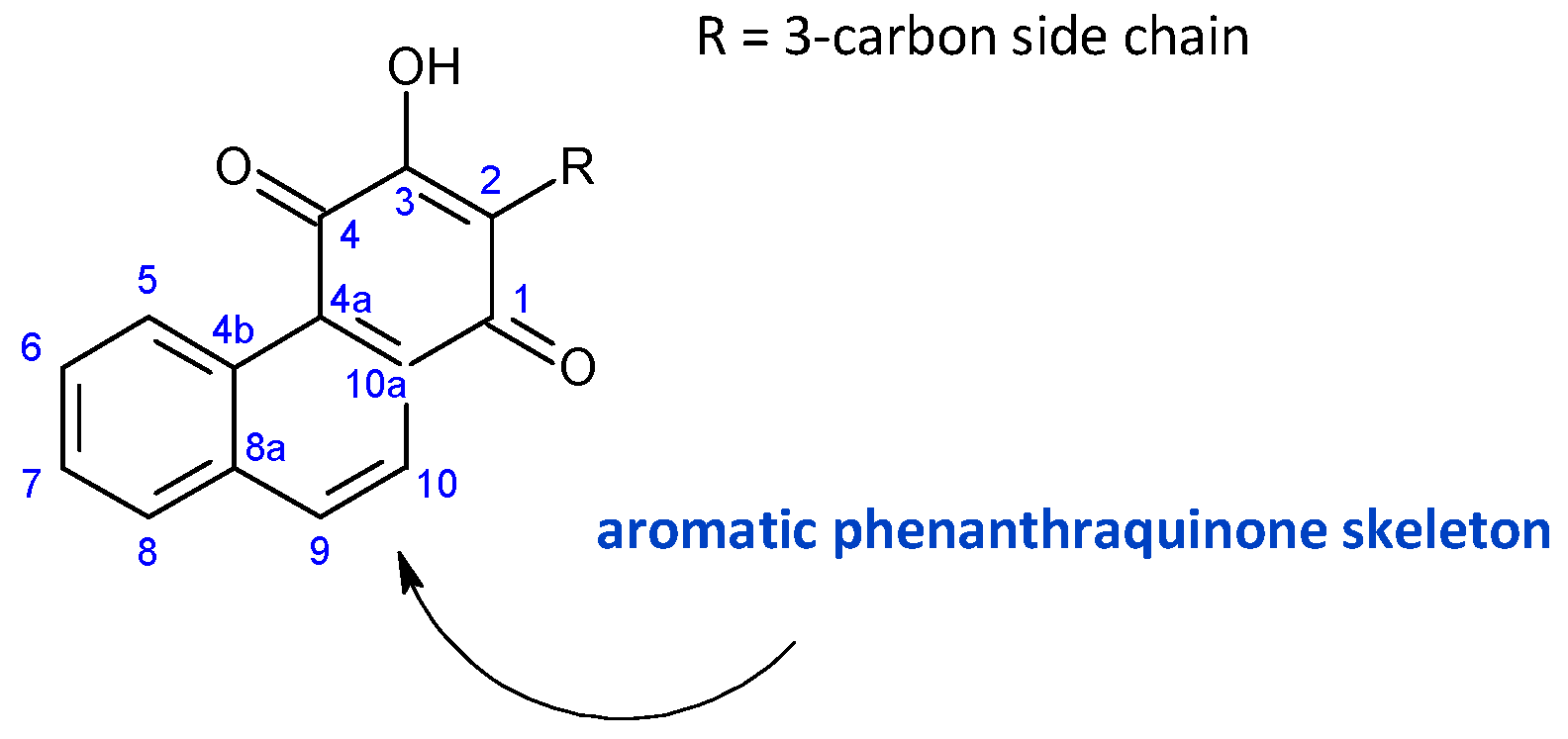
2.5. 1,4-Phenathraquinones
 | ||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | Source [References] |
| plectranthone A (165) | –CH3 | –H | –H | C. (Rwanda) [29,98] |
| plectranthone C (166) | –H | –H | –H | C. (Rwanda) [29,98] |
| plectranthone D (167) | –H | –H | –CH3 | C. (Rwanda) [29,98] |
| plectranthone E (168) | –H | –OH | –H | C. (Rwanda) [29] |
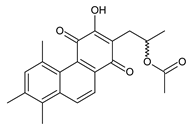 plectranthone B (169) C. (Rwanda) [29,98] | ||||
2.6. Dimeric Diterpenes
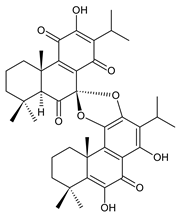 grandidone A (170) C. grandidentatus [48,49,95], C. hereroensis [95], C. sanguineus [28] |  7-epi-grandidone A (171) C. grandidentatus [95], C. sanguineus [28] | |
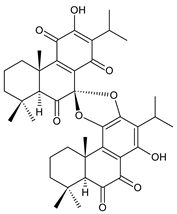 grandidone B (172) C. amboinicus [95], C. grandidentatus [95], C. sanguineus [28] | 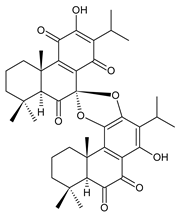 7-epi-grandidone B (173) C. amboinicus [95], C. grandidentatus [95], C. sanguineus [28] | |
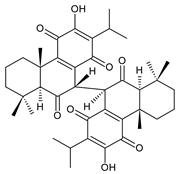 grandidone C (174) C. grandidentatus [39,95] | 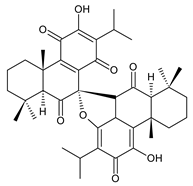 grandidone D (175) C. grandidentatus [39,95] |  7-epi-grandidone D (176) C. grandidentatus [39,95] |
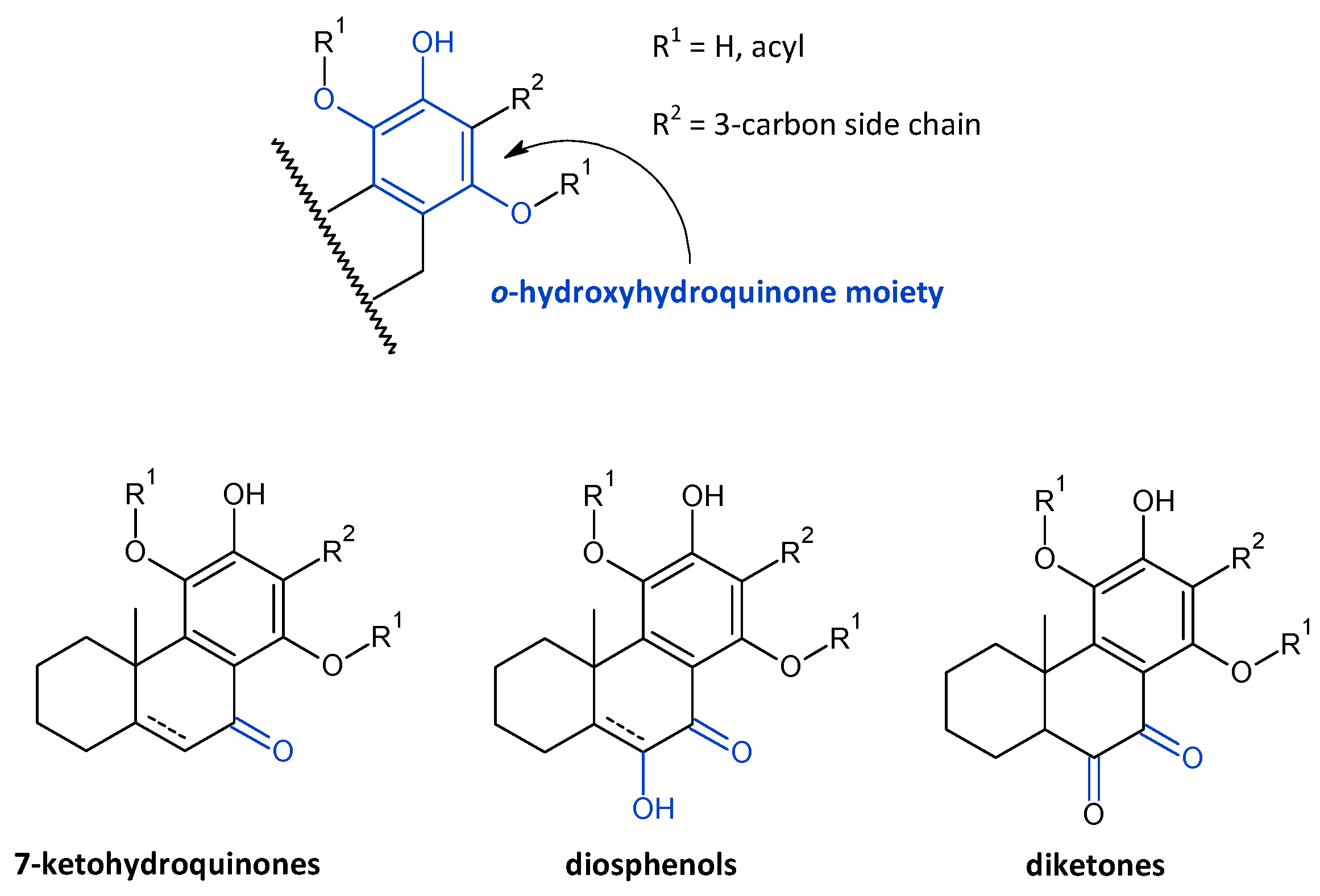
2.7. Hydroquinones
| (a) 7-Ketohydroquinones | ||||||
|---|---|---|---|---|---|---|
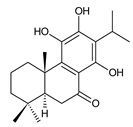 demethylinuroyleanol (177) C. maculosus [41] | ||||||
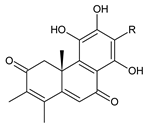 | ||||||
| Compound Name (numerical ID) | R | Source [References] | ||||
| plectrinone A (178) | –CH2CH(OH)CH3 | C. (Rwanda) [29], C. barbatus [61,99] | ||||
| plectrinone B (179) | –CH2CH=CH2 | C. (Rwanda) [29], C. barbatus [61] | ||||
| (b) Diosphenols | ||||||
 5,6-dihydrocoleon U (180) C. argentatus [47], C. sanguineus [28] |  coleon B (181) C. igniarius [64,100] | |||||
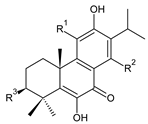 | ||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | Source [References] | ||
| coleon S (182) | –OH | –OH | –OH | C. caninus [101] | ||
| coleon U (183) | –OH | –OH | –H | C. amboinicus [35], C. argentatus [47], C. forsteri ‘Marginatus’ [56], C. grandidentatus [40,48,49,51,95], C. hereroensis [50], C. maculosus subsp. edulis [26], C. sanguineus [28], C. xanthanthus [55] | ||
| 14-O-acetylcoleon U (184) | –OH | –OCOCH3 | –H | C. grandidentatus [102] | ||
| coleon U 11-acetate (185) | –OCOCH3 | –OH | –H | C. xanthanthus [55] | ||
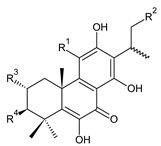 | ||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | Source [References] | |
| 11,16-bis(O-acetoxy)coleon U (186) | –OCOCH3 | –OCOCH3 | –H | –H | C. xanthanthus [55] | |
| coleon C (187) | –OH | –OH | –H | –H | C. (North Madagascar) [103], C. barbatus var. barbatus [25], C. lanuginosus [23], C. maculosus subsp. edulis [26,104], C. monostachyus [84] | |
| (15R)-2α-formyloxy-coleon C (188) | –OH | –OH | –OCHO | –H | C. monostachyus [84] | |
| (15S)-2α-acetoxycoleon C (189) | –OH | –OH | –OCOCH3 | –H | C. (Rwanda) [29] | |
| 16-O-acetylcoleon C (190) | –OH | –OCOCH3 | –H | –H | C. barbatus var. barbatus [25], C. maculosus subsp. edulis [26], C. xanthanthus [55] | |
| coleon H (191) | –OH | –OH | –H | –OCOCH3 | C. (Rwanda) [29], C. comosus [77], C. lanuginosus [60,105], C. monostachyus [84], revision [106] | |
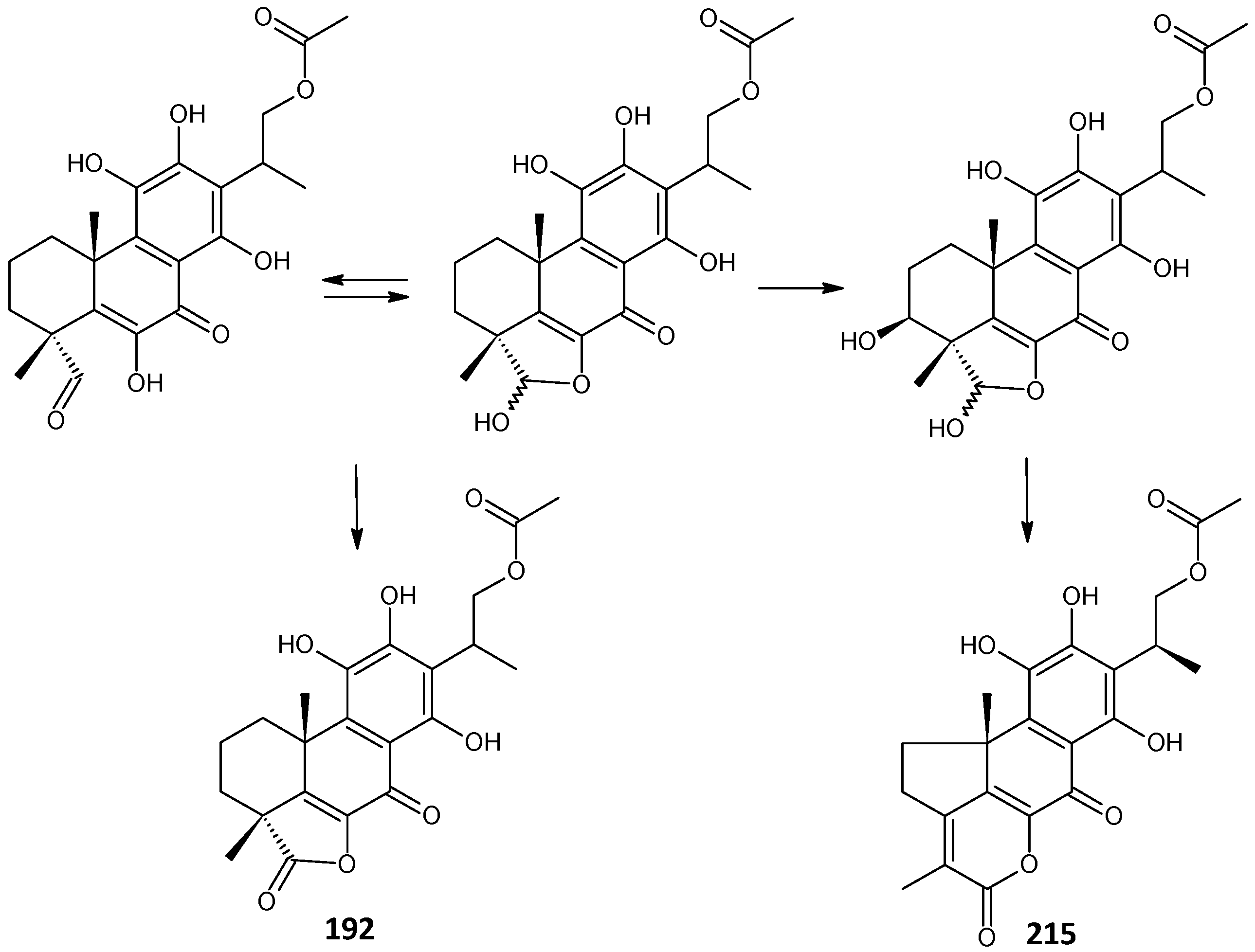
 lactone derivative of 16-O-acetylcoleon C (192) C. maculosus subsp. edulis [26] | ||||||
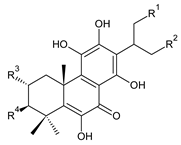 | ||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | Source [References] | |
| coleon L (193) | –OCOCH3 | –OH | –H | –OCOCH3 | C. comosus [77], C. lanuginosus [60,106] | |
| coleon W (194) | –OCOCH3 | –OH | –H | –H | C. autranii [72], C. barbatus var. barbatus [25], C. garckeanus [72], C. hereroensis [50] | |
| 16(or 17)-O-acetylcoleon W (195) | –OCOCH3 | –OCOCH3 | –H | –H | C. autranii [72], C. garckeanus [72] | |
| 2α-acetoxycoleon W (196) | –OCOCH3 | –OH | –OCOCH3 | –H | C. scutellarioides [12,83,107] | |
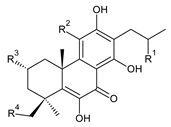 | ||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | Source [References] | |
| lanugon S (197) | –OH | –OH | –H | –OCHO | C. lanuginosus [23] | |
| 13-desisopropyl-13-(2-hydroxypropyl)coleon U (198) | –OH | –OH | –H | –H | C. maculosus subsp. edulis [26] | |
| xanthanthusin F (199) | –OH | –OCOCH3 | –OCOCH3 | –H | C. xanthanthus [55] | |
| xanthanthusin G (200) | –OCH(CH3)2 | –OCOCH3 | –OCOCH3 | –H | C. xanthanthus [55] | |
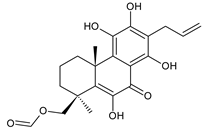 lanugon R (201) C. lanuginosus [23] | 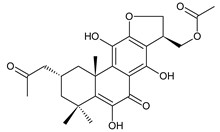 scutellarioidone C (202) C. scutellarioides [12] | |||||
| (c) Diketones | ||||||
 | ||||||
| Compound Name (Numerical ID) | R1 | R2 | Source [References] | |||
| coleon T (203) | –OH | –OH | C. caninus [101] | |||
| coleon V (204) | –OH | –H | C. amboinicus [35], C. argentatus [47], C. barbatus var. barbatus [25], C. grandidentatus [95], C. hereroensis [50], C. maculosus [41], C. sanguineus [28] | |||
| 14-O-formylcoleon V (205) | –OCHO | –H | C. hereroensis [50] | |||
 | ||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | Source [References] | ||
| coleon D (206) | –OH | –H | –H | C. (North Madagascar) [103], C. barbatus var. barbatus [25], C. lanuginosus [23,60], C. maculosus subsp. edulis [26,108] | ||
| (15S)-2α -acetoxycoleon D (207) | –OH | –OCOCH3 | –H | C. (Rwanda) [29] | ||
| 16-O-acetylcoleon D (208) | –OCOCH3 | –H | –H | C. barbatus var. barbatus [25], C. maculosus subsp. edulis [26] | ||
| coleon I (209) | –OH | –H | –OCOCH3 | C. (North Madagascar) [103], C. (Rwanda) [29], C. lanuginosus [60,105], revision [106] | ||
| coleon I’ (210) | –OH | –H | –OCHO | C. (North Madagascar) [103], revision [106] | ||
 | ||||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | Source [References] | ||
| coleon K (211) | –OCOCH3 | –OH | –OCOCH3 | C. lanuginosus [60,105], revision [106] | ||
| coleon X (212) | –OCOCH3 | –OH | –H | C. autranii [72], C. garckeanus [72] | ||
| 16(or 17)-O-acetylcoleon X (213) | –OCOCH3 | –OCOCH3 | –H | C. garckeanus [72] | ||
 13-desisopropyl-13-(2-hydroxypropyl)coleon V (214) C. maculosus subsp. edulis [26] | ||||||
| (d) 5(4→3)-Abeo-Acylhydroquinones | ||||||
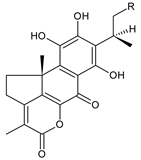 | ||||||
| Compound Name (Numerical ID) | R | Source [References] | ||||
| edulon A (215) | –OCOCH3 | C. maculosus subsp. edulis [26,109,110] | ||||
| desacetyledulon A (216) | –OH | C. maculosus subsp. edulis [26] | ||||
| (e) Seco-Hydroquinones | ||||||
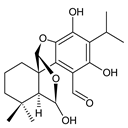 cariocal (217) C. barbatus [111] | ||||||
2.8. Other Phenolic Abietanes
| (a) 12-Hydroxylated Aromatic Abietanes | |||||
|---|---|---|---|---|---|
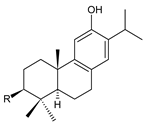 | |||||
| Compound Name (Numerical ID) | R | Source [References] | |||
| ferruginol (218) | –H | C. barbatus [13,112,113] | |||
| hinokiol (219) | –OH | P. strigosus [93] | |||
 sugiol (220) C. barbatus [66,112,114] |  5,6-dehydrosugiol (221) C. barbatus [112] | ||||
| (b) 11,12-Dihydroxylated Aromatic Abietanes | |||||
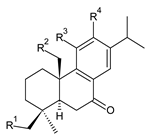 | |||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | R4 | Source [References] |
| 11-hydroxysugiol (222) | –H | –H | –OH | –OH | C. hadiensis [115] |
| 11,20-dihydroxysugiol (223) | –H | –OH | –OH | –OH | C. hadiensis [115] |
| 19-O-senecioylester of 11,19-dihydroxysugiol (224) | (CH3)2C=CH–COO– | –H | –OH | –OH | P. purpuratus [14] |
| 19-O-isovaleroylester of 11,19-dihydroxysugiol (225) | (CH3)2CH–CH2–COO– | –H | –OH | –OH | P. purpuratus [14] |
| plectranthol A (226) | 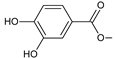 | –H | –OH | –OH | P. verticillatus [90] |
| plectranthol B (227) |  | –H | –OH | (CH3)2C=CH–COO– | P. verticillatus [90] |
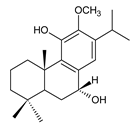 7α,11-dihydroxy-12-methoxy-8,11,13-abietatriene (228) P. elegans [86] | 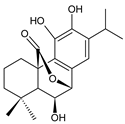 6β- hydroxycarnosol (229) C. barbatus [113] | ||||
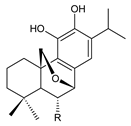 | |||||
| Compound Name (Numerical ID) | R | Source [References] | |||
| 20-deoxocarnosol (230) | –H | C. barbatus [87,113] | |||
| 6α,11,12-trihydroxy-7β,20-epoxy-8,11,13-abietatriene (231) | –OH | C. barbatus [87] | |||
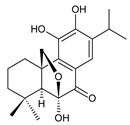 carnosolon (232) C. amboinicus [35], C. hadiensis [115], C. madagascariensis [46] | 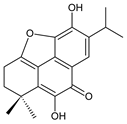 1,11-epoxy-6,12-dihydroxy-20-norabieta-1(10),5,8,11,13-pentaen-7-one (233) C. hadiensis [115] | ||||
| (c) 11,14-Dihydroxylated Aromatic Abietane | |||||
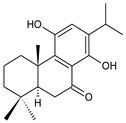 11,14-dihydroxy-8,11,13-abietatrien-7-one (234) C. barbatus [66,114] | |||||
| (d) Seco-Abietanes with Phenolic Ring C | |||||
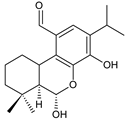 seco-abietane 1 (235) C. barbatus [30] | 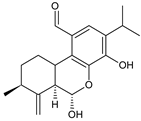 seco-abietane 2 (236) C. barbatus [30] | ||||
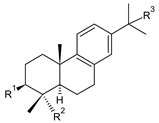
2.9. Non-Phenolic Aromatic Abietanes
 | ||||
| Compound Name (Numerical ID) | R1 | R2 | R3 | Source [References] |
| dehydroabietane (abietatriene) (237) | –H | –CH3 | –H | C. barbatus [87] |
| 4-epi-triptobenzene L (238) | –OH | –CH2OH | –H | C. harmandii [71] |
| dehydroabietic acid (239) | –H | –COOH | –H | C. harmandii [71] |
| 15- hydroxydehydroabietic acid (240) | –H | –COOH | –OH | C. harmandii [71] |
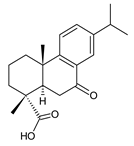 7-oxodehydroabietic acid (241) C. harmandii [71] | 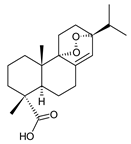 9α-13α-epidioxyabiet-8(14)-en-18-oic acid (242) C. harmandii [71] | 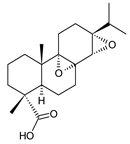 8α,9α,13α,14α-diepoxyabietan-18-oic acid (243) C. harmandii [71] | ||
3. Occurrence and Isolation of Abietane Diterpenes
4. Biological Activity of Abietane Diterpenes
4.1. Antitumoral and Cytotoxic Activity
4.2. Antimicrobial Activity
4.3. Antiprotozoal Activity
4.4. Gastroprotective, Antisecretory and Antiulcer Activity
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Süntar, I. Importance of ethnopharmacological studies in drug discovery: Role of medicinal plants. Phytochem. Rev. 2020, 19, 1199–1209. [Google Scholar] [CrossRef]
- Paton, A.; Mwanyambo, M.; Culham, A. Phylogenetic study of Plectranthus, Coleus and allies (Lamiaceae): Taxonomy, distribution and medicinal use. Bot. J. Linn. Soc. 2018, 188, 355–376. [Google Scholar] [CrossRef]
- Paton, A.J.; Mwanyambo, M.; Govaerts, R.H.A.; Smitha, K.; Suddee, S.; Phillipson, P.B.; Wilson, T.C.; Forster, P.I.; Culham, A. Nomenclatural changes in Coleus and Plectranthus (Lamiaceae): A tale of more than two genera. PhytoKeys 2019, 129, 1–158. [Google Scholar] [CrossRef]
- Van Jaarsveld, E.; Thomas, V. The Southern African Plectranthus: And the Art of Turning Shade to Glade; Fernwood Press: Pretoria, South Africa, 2006. [Google Scholar]
- Lukhoba, C.W.; Simmonds, M.S.J.; Paton, A.J. Plectranthus: A review of ethnobotanical uses. J. Ethnopharmacol. 2006, 103, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Rice, L.J.; Brits, G.J.; Potgieter, C.J.; van Staden, J. Plectranthus: A plant for the future? S. Afr. J. Bot. 2011, 77, 947–959. [Google Scholar] [CrossRef]
- Grayer, R.J.; Eckert, M.R.; Lever, A.; Veitch, N.C.; Kite, G.C.; Paton, A.J. Distribution of exudate flavonoids in the genus Plectranthus. Biochem. Syst. Ecol. 2010, 38, 335–341. [Google Scholar] [CrossRef]
- Abdel-Mogib, M.; Albar, H.A.; Batterjee, S.M. Chemistry of the genus Plectranthus. Molecules 2002, 7, 271–301. [Google Scholar] [CrossRef]
- Waldia, S.; Joshi, B.C.; Pathak, U.; Joshi, M.C. The Genus Plectranthus in India and Its Chemistry. Chem. Biodivers. 2011, 8, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Rijo, P.; Faustino, C.; Simões, M.F. Antimicrobial natural products from Plectranthus plants. In Microbial Pathogens and Strategies for Combating Them: Science, Technology and Education; Méndez-Vilas, A., Ed.; Formatex: Badajoz, Spain, 2013; pp. 922–931. [Google Scholar]
- Grayer, R.J.; Paton, A.J.; Simmonds, M.S.; Howes, M.J.R. Differences in diterpenoid diversity reveal new evidence for separating the genus Coleus from Plectranthus. Nat. Prod. Rep. 2021, 38, 1720–1728. [Google Scholar] [CrossRef]
- Cretton, S.; Saraux, N.; Monteillier, A.; Righi, D.; Marcourt, L.; Genta-Jouve, G.; Wolfender, J.L.; Cuendet, M.; Christen, P. Anti-inflammatory and antiproliferative diterpenoids from Plectranthus scutellarioides. Phytochemistry 2018, 154, 39–46. [Google Scholar] [CrossRef]
- Kelecom, A. Isolation, structure determination, and absolute configuration of barbatusol, a new bioactive diterpene with a rearranged abietane skeleton from the Labiate Coleus barbatus. Tetrahedron 1983, 39, 3603–3608. [Google Scholar] [CrossRef]
- Katti, S.B.; Rüedi, P.; Eugster, C.H. Diterpenoide Chinomethane, vinyloge Chinone und ein Phyllocladan-Derivat aus Plectranthus purpuratus HARV. (Labiatae). Helv. Chim. Acta 1982, 65, 2189–2197. [Google Scholar] [CrossRef]
- Ladeiras, D.; Monteiro, C.M.; Pereira, F.; Reis, C.P.; Afonso, C.A.M.; Rijo, P. Reactivity of Diterpenoid Quinones: Royleanones. Curr. Pharm. Des. 2016, 22, 1682–1714. [Google Scholar] [CrossRef]
- van Zyl, R.L.; Khan, F.; Edwards, T.J.; Drewes, S.E. Antiplasmodial activities of some abietane diterpenes from the leaves of five Plectranthus species. S. Afr. J. Sc. 2008, 104, 62–64. [Google Scholar]
- Bao, H.; Zhang, Q.; Ye, Y.; Lin, L. Naturally occurring furanoditerpenoids: Distribution, chemistry and their pharmacological activities. Phytochem. Rev. 2017, 16, 235–270. [Google Scholar] [CrossRef]
- García, P.A.; de Oliveira, A.B.; Batista, R. Occurrence, biological activities and synthesis of kaurane diterpenes and their glycosides. Molecules 2007, 12, 455–483. [Google Scholar] [CrossRef] [PubMed]
- González, M.A. Aromatic abietane diterpenoids: Their biological activity and synthesis. Nat. Prod. Rep. 2015, 32, 684–704. [Google Scholar] [CrossRef] [PubMed]
- Otto, A.; Wilde, V. Sesqui-, Di-, and Triterpenoids as Chemosystematic Markers in Extant Conifers—A Review. Bot. Rev. 2001, 67, 141–238. [Google Scholar] [CrossRef]
- Talapatra, S.K.; Talapatra, B. Diterpenoids (C20). In Chemistry of Plant Natural Products; Springer: Berlin, Germany, 2015; pp. 469–510. [Google Scholar]
- Lin, L.G.; Ung, C.O.L.; Feng, Z.L.; Huang, L.; Hu, H. Naturally Occurring Diterpenoid Dimers: Source, Biosynthesis, Chemistry and Bioactivities. Planta Med. 2016, 82, 1309–1328. [Google Scholar] [CrossRef]
- Schmid, J.M.; Rüedi, P.; Eugster, C.H. Diterpenoide Drüsenfarbstoffe aus Labiaten: 22 neue Coleone und Royleanone aus Plectranthus lanuginosus. Helv. Chim. Acta 1982, 65, 2136–2163. [Google Scholar] [CrossRef]
- Arihara, S.; Rüedi, P.; Eugster, C.H. Neue spiro-cyclopropyl-cyclohexendion-diterpene: Coleone M, N, P, Q, R sowie barbatusin aus Plectranthus caninus Roth und coleon O aus Coleus somaliensis S. Moore. Helv. Chim. Acta 1975, 58, 343–356. [Google Scholar] [CrossRef]
- Grob, K.; Rüedi, P.; Eugster, C.H. Drüsenfarbstoffe aus Labiaten: Strukturen von 16 Diterpenen (Coleone und Royleanone) aus Coleus coerulescens GÜRKE. Helv. Chim. Acta 1978, 61, 871–884. [Google Scholar] [CrossRef]
- Künzle, J.M.; Rüedi, P.; Eugster, C.H. Isolierung und Strukturaufklärung von 36 Diterpenoiden aus Trichomen von Plectranthus edulis (VATKE) T.T. AYE. Helv. Chim. Acta 1987, 70, 1911–1929. [Google Scholar] [CrossRef]
- Eugster, C.H. Terpenoide, insbesondere diterpenoide Farbstoffe. Ber. Deutsch. Bot. Ges. 1975, 88, 141–161. [Google Scholar]
- Matloubi-Moghadam, F.; Rüedi, P.; Eugster, C.H. Drüsefarbstoffe aus Labiaten: Identifizierung von 17 abietanoiden aus Plectranthus sanguineus BRITTEN. Helv. Chim. Acta 1987, 70, 975–983. [Google Scholar] [CrossRef]
- Alder, A.C.; Rüedi, P.; Prewo, R.; Bieri, J.H.; Eugster, C.H. Neue Diterpenoide aus Blattdrüsen einer Plectranthus sp. aus Rwanda. Helv. Chim. Acta 1986, 69, 1395–1417. [Google Scholar] [CrossRef]
- Kelecom, A.; Dos Santos, T.C.; Medeiros, W.L.B. Secoabietane diterpenes from Coleus barbatus. Phytochemistry 1987, 26, 2337–2340. [Google Scholar] [CrossRef]
- Edwards, O.E.; Feniak, G.; Los, M. Diterpenoid quinones of Inula royleana DC. Can. J. Chem. 1962, 40, 1540–1546. [Google Scholar] [CrossRef]
- Brieskorn, C.H.; Buchberger, L. Diterpenchinone aus Labiatenwurzeln1. Planta Med. 1973, 24, 190–195. [Google Scholar] [CrossRef]
- Hensch, M.; Rüedi, P.; Eugster, C.H. Horminon, Taxochinon und weitere Royleanone aus 2 abessinischen Plectranthus-Spezies (Labiatae). Helv. Chim. Acta 1975, 58, 1921–1934. [Google Scholar] [CrossRef]
- Rüedi, P. 8α, 9α-Epoxy-7-oxoroyleanon, ein Diterpen-Epoxychinon aus einer abessinischen Plectranthus-Art (Labiatae). Helv. Chim. Acta 1984, 67, 1116–1120. [Google Scholar] [CrossRef]
- Yoshizaki, F.; Rüedi, P.; Eugster, C.H. Diterpenoide Drüsenfarbstoffe aus Labiaten: 11 Coleone und Royleanone aus Coleus carnosus HASSK. Helv. Chim. Acta 1979, 62, 2754–2762. [Google Scholar] [CrossRef]
- Syamasundar, K.V.; Vinodh, G.; Srinivas, K.V.N.S.; Srinivasulu, B. A New Abietane Diterpenoid from Plectranthus bishopianus Benth. Helv. Chim. Acta 2012, 95, 643–646. [Google Scholar] [CrossRef]
- Vinodh, G.; Naveen, P.; Venkatesan, C.S.; Rajitha, G.; Shree, A.J. Pharmacological Evaluation of Abietane Diterpenoids from Plectranthus bishopianus as Potent Antibacterial, Antioxidant and Their Cytotoxic Agents. Nat. Prod. J. 2019, 9, 229–237. [Google Scholar] [CrossRef]
- Kubínová, R.; Švajdlenka, E.; Schneiderová, K.; Hanáková, Z.; Dall’Acqua, S.; Farsa, O. Polyphenols and diterpenoids from Plectranthus forsteri ‘Marginatus’. Biochem. Syst. Ecol. 2013, 49, 39–42. [Google Scholar] [CrossRef]
- Teixeira, A.P.; Batista, O.; Simões, M.F.; Nascimento, J.; Duarte, A.; de la Torrre, M.C.; Rodríguez, B. Abietane diterpenoids from Plectranthus grandidentatus. Phytochemistry 1997, 44, 325–327. [Google Scholar] [CrossRef]
- Gaspar-Marques, C.; Rijo, P.; Simões, M.F.; Duarte, M.A.; Rodriguez, B. Abietanes from Plectranthus grandidentatus and P. hereroensis against methicillin-and vancomycin-resistant bacteria. Phytomedicine 2006, 13, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Abdissa, N.; Frese, M.; Sewald, N. Antimicrobial Abietane-Type Diterpenoids from Plectranthus punctatus. Molecules 2017, 22, 1919. [Google Scholar] [CrossRef]
- Sitarek, P.; Toma, M.; Ntungwe, E.; Kowalczyk, T.; Skała, E.; Wieczfinska, J.; Śliwiński, T.; Rijo, P. Insight the Biological Activities of Selected Abietane Diterpenes Isolated from Plectranthus spp. Biomolecules 2020, 10, 194. [Google Scholar] [CrossRef]
- Śliwiński, T.; Sitarek, P.; Skała, E.; Isca, V.M.S.; Synowiec, E.; Kowalczyk, T.; Bijak, M.; Rijo, P. Diterpenoids from Plectranthus spp. as Potential Chemotherapeutic Agents via Apoptosis. Pharmaceuticals 2020, 13, 123. [Google Scholar] [CrossRef]
- Batista, O.; Duarte, A.; Nascimento, J.; Simões, M.F.; de la Torre, M.C.; Rodríguez, B. Structure and antimicrobial activity of diterpenes from the roots of Plectranthus hereroensis. J. Nat. Prod. 1994, 57, 858–861. [Google Scholar] [CrossRef] [PubMed]
- Batista, O.; Simões, M.F.; Nascimento, J.; Riberio, S.; Duarte, A.; Rodríguez, B.; de la Torre, M.C. A rearranged abietane diterpenoid from Plectranthus hereroensis. Phytochemistry 1996, 41, 571–573. [Google Scholar] [CrossRef]
- Ndjoubi, K.O.; Sharma, R.; Badmus, J.A.; Jacobs, A.; Jordaan, A.; Marnewick, J.; Warner, D.F.; Hussein, A.A. Antimycobacterial, Cytotoxic, and Antioxidant Activities of Abietane Diterpenoids Isolated from Plectranthus madagascariensis. Plants 2021, 10, 175. [Google Scholar] [CrossRef] [PubMed]
- Alder, A.C.; Rüedi, P.; Eugster, C.H. Drüsenfarbstoffe aus Labiaten: Die polaren Diterpenoide aus Plectranthus argentatus ST BLAKE. Helv. Chim. Acta 1984, 67, 1523–1530. [Google Scholar] [CrossRef]
- Gaspar-Marques, C.; Pedro, M.; Simões, M.F.A.; Nascimento, M.S.J.; Pinto, M.M.M.; Rodríguez, B. Effect of Abietane Diterpenes from Plectranthus grandidentatus on the Growth of Human Cancer Cell Lines. Planta Med. 2002, 68, 839–840. [Google Scholar] [CrossRef]
- Cerqueira, F.; Cordeiro-Da-Silva, A.; Gaspar-Marques, C.; Simoes, F.; Pinto, M.M.M.; Nascimento, M.S.J. Effect of abietane diterpenes from Plectranthus grandidentatus on T-and B-lymphocyte proliferation. Bioorg. Med. Chem. 2004, 12, 217–223. [Google Scholar] [CrossRef]
- Miyase, T.; Rüedi, P.; Eugster, C.H. Diterpenoide Drüsenfarbstoffe aus Labiaten: Coleone U, V, W und 14-O-Formyl-coleon-V sowie 2 Royleanone aus Plectranthus myrianthus BRIQ.; cis-und trans-A/B-6, 7-Dioxoroyleanon. Helv. Chim. Acta 1977, 60, 2770–2779. [Google Scholar] [CrossRef]
- Gaspar-Marques, C.; Simões, M.F.; Rodríguez, B. A trihomoabietane Diterpenoid from Plectranthus grandidentatus and an Unusual Addition of Acetone to the ortho-Quinone System of Cryptotanshinone. J. Nat. Prod. 2005, 68, 1408–1411. [Google Scholar] [CrossRef]
- Mehrotra, R.; Vishwakarma, R.A.; Thakur, R.S. Abietane diterpenoids from Coleus zeylanicus. Phytochemistry 1989, 28, 3135–3137. [Google Scholar] [CrossRef]
- Matias, D.; Nicolai, M.; Saraiva, L.; Pinheiro, R.; Faustino, C.; Diaz Lanza, A.; Pinto Reis, C.; Stankovic, T.; Dinic, J.; Pesic, M.; et al. Cytotoxic Activity of Royleanone Diterpenes from Plectranthus madagascariensis Benth. ACS Omega 2019, 4, 8094–8103. [Google Scholar] [CrossRef]
- Kubínova, R.; Pořízková, R.; Navrátilová, A.; Farsa, O.; Hanáková, Z.; Bačinská, A.; Čížek, A.; Valentová, M. Antimicrobial and enzyme inhibitory activities of the constituents of Plectranthus madagascariensis (Pers.) Benth. J. Enzym. Inhib. Med. Chem. 2014, 29, 749–752. [Google Scholar] [CrossRef] [PubMed]
- Mei, S.X.; Jiang, B.; Niu, X.M.; Li, M.L.; Yang, H.; Na, Z.; Lin, Z.W.; Li, C.M.; Sun, H.D. Abietane Diterpenoids from Coleus xanthanthus. J. Nat. Prod. 2002, 65, 633–637. [Google Scholar] [CrossRef] [PubMed]
- Wellsow, J.; Grayer, R.J.; Veitch, N.C.; Kokubun, T.; Lelli, R.; Kite, G.C.; Simmonds, M.S.J. Insect-antifeedant and antibacterial activity of diterpenoids from species of Plectranthus. Phytochemistry 2006, 67, 1818–1825. [Google Scholar] [CrossRef] [PubMed]
- Batista, O.; Simões, M.F.; Duarte, A.; Valdeira, M.L.; de la Torre, M.C.; Rodríguez, B. An antimicrobial abietane from the root of Plectranthus hereroensis. Phytochemistry 1995, 38, 167–169. [Google Scholar] [CrossRef]
- Nzogong, R.T.; Nganou, B.K.; Tedonkeu, A.T.; Awouafack, M.D.; Tene, M.; Ito, T.; Tane, P.; Morita, H. Three New Abietane-Type Diterpenoids from Plectranthus africanus and Their Antibacterial Activities. Planta Med. 2018, 84, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Jurkaninová, S.; Kubínová, R.; Nejezchlebová, M.; Gazdová, M.; Hanáková, Z.; Dall’Acqua, S. Anti-MRSA activity of abietane diterpenes from Coleus blumei Benth. Nat. Prod. Res. 2021, 35, 3033–3039. [Google Scholar] [CrossRef]
- Matloubi-Moghadam, F.; Rüedi, P.; Eugster, C.H. Neue Coleone und Royleanone aus Coleus somaliensis S. MOORE. Helv. Chim. Acta 1984, 67, 201–208. [Google Scholar] [CrossRef]
- Rüedi, P. Neue Diterpene aus Blattdrüsen von Plectranthus barbatus (Labiatae). Die absolute Konfiguration der 2-Hydroxypropyl-Seitenkette in Coleon, E. Helv. Chim. Acta 1986, 69, 972–984. [Google Scholar] [CrossRef]
- Zhu, Z.Y.; Nayeshiro, H.; Prewo, R.; Rüedi, P.; Eugster, C.H. Fredericone, neue hochfunktionalisierte Abietanoide aus Blattdrüsen von Coleus fredericii G. TAYL. Helv. Chim. Acta 1988, 71, 577–587. [Google Scholar] [CrossRef]
- Baxter, R.L.; Blake, A.J.; Gould, R.O. The absolute configuration of coleon A: A seco-diterpenoid pigment from Coleus spp. Phytochemistry 1995, 38, 195–197. [Google Scholar] [CrossRef]
- Eugster, C.H.; Küng, H.P.; Kühnis, H.; Karrer, P. Isolierung und Charakterisierung zweier neuer Blattfarbstoffe: Coleon A und Coleon, B. Chinontrennungen an Polyamidpulver. Helv. Chim. Acta 1963, 46, 530–543. [Google Scholar] [CrossRef]
- Karanatsios, D.; Eugster, C.H. Struktur von Coleon A. Helv. Chim. Acta 1965, 48, 471–508. [Google Scholar] [CrossRef]
- Musayeib, N.M.A.; Amina, M.; Al-Hamoud, G.A.; Mohamed, G.A.; Ibrahim, S.R.; Shabana, S. Plectrabarbene, a New Abietane Diterpene from Plectranthus barbatus Aerial Parts. Molecules 2020, 25, 2365. [Google Scholar] [CrossRef]
- Rüedi, P.; Eugster, C.H. Struktur von Coleon E, einem neuen diterpenoiden Methylenchinon aus der Coleus barbatus-Gruppe (Labiatae). Helv. Chim. Acta 1972, 55, 1994–2014. [Google Scholar] [CrossRef]
- Wang, A.H.; Paul, I.C.; Zelnik, R.; Lavie, D.; Levy, E.C. Structure and Stereochemistry of Cyclobutatusin, a Diterpenoid Containing a Four-Membered Ring. J. Am. Chem. Soc. 1974, 96, 580–581. [Google Scholar] [CrossRef] [PubMed]
- Moir, M.; Rüedi, P.; Eugster, C.H. Diterpenoids from Coleus somaliensis (S. Moore): Coleons, G., and J. Helv. Chim. Acta 1973, 56, 2539–2548. [Google Scholar] [CrossRef]
- Rüedi, P.; Schmid, J.M.; Prewo, R.; Bieri, J.H.; Eugster, C.H. Spirocoleone: Synthese und Charakterisierung von vier diastereomeren Spiro(methylcyclopropan)-Substrukturen; Revision der Konfiguration an C (12) und C (15) von Coleon P und Derivaten sowie von Coleon-Z-Derivaten; Röntgenstrukturanalysen von Lanugon J und weiteren Spirocoleonen. Helv. Chim. Acta 1983, 66, 429–449. [Google Scholar]
- Lekphrom, R.; Kanokmedhakul, S.; Kanokmedhakul, K. Bioactive Diterpenes from the Aerial Parts of Anisochilus harmandii. Planta Med. 2010, 76, 726–728. [Google Scholar] [CrossRef]
- Miyase, T.; Yoshizaki, F.; Kabengele, N.T.; Rüedi, P.; Eugster, C.H. Strukturen von 13 Diterpenen (Coleonen) aus Blattdrüsen von Solenostemon sylvaticus und Coleus garckeanus (Labiatae). Helv. Chim. Acta 1979, 62, 2374–2383. [Google Scholar] [CrossRef]
- Ávila, F.N.; Pinto, F.C.; Sousa, T.S.; Torres, M.C.M.; Costa-Lotufo, L.V.; Rocha, D.D.; de Vasconcelos, M.A.; Cardoso-Sá, N.; Teixeira, E.H.; Albuquerque, M.R.J.R.; et al. Miscellaneous Diterpenes from the Aerial Parts of Plectranthus ornatus Codd. J. Braz. Chem. Soc. 2017, 28, 1014–1022. [Google Scholar] [CrossRef]
- Simões, M.F.; Rijo, P.; Duarte, A.; Barbosa, D.; Matias, D.; Delgado, J.; Cirilo, N.; Rodríguez, B. Two new diterpenoids from Plectranthus species. Phytochem. Lett. 2010, 3, 221–225. [Google Scholar] [CrossRef]
- Zelnik, R.; Lavie, D.; Levy, E.C.; Wang, A.H.J.; Paul, I.C. Barbatusin and cyclobutatusin, two novel diterpenoids from Coleus barbatus Bentham. Tetrahedron 1977, 33, 1457–1467. [Google Scholar] [CrossRef]
- de Araújo Rodrigues, P.; de Morais, S.M.; de Souza, C.M.; Araújo Silva, A.R.; de Andrade, G.M.; Vasconcelos Silva, M.G.; Albuquerque, R.L.; Rao, V.S.; Santos, F.A. Gastroprotective effect of barbatusin and 3-beta-hydroxy-3-deoxibarbatusin, quinonoid diterpenes isolated from Plectranthus grandis, in ethanol-induced gastric lesions in mice. J. Ethnopharmacol. 2010, 127, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, L.S.F.; Matos, T.S.; do Nascimento Ávila, F.; da Silva Batista, A.; Moura, A.F.; de Moraes, M.O.; da Silva, M.C.M.; Ferreira, T.L.A.; Nascimento, N.R.F.; Monteiro, N.K.V.; et al. Diterpenoids from Leaves of Cultivated Plectranthus ornatus. Planta Med. 2021, 87, 124–135. [Google Scholar] [CrossRef]
- Albuquerque, R.L.D.; Kentopff, M.R.; Machado, M.I.L.; Silva, M.G.V.; Matos, F.J.D.A.; Morais, S.M.; Braz-Filho, R. Diterpenos tipo abietano isolados de Plectranthus barbatus Andrews. Quím. Nova 2007, 30, 1882–1886. [Google Scholar] [CrossRef]
- Kubo, I.; Matsumoto, T.; Tori, M.; Asakawa, Y. Structure of plectrin, an aphid antifeedant diterpene from Plectranthus barbatus. Chem. Lett. 1984, 13, 1513–1516. [Google Scholar] [CrossRef]
- Devriese, E.G.; Buffel, K.; Geuns, J.M. Stimulation of adventitious root formation on mung bean cuttings by coleon O. Phytochemistry 1988, 27, 293–294. [Google Scholar] [CrossRef]
- Miyase, T.; Rüedi, P.; Eugster, C.H. Unusual Rearranged Abietanoic Diterpenoids from Solenostemon Species. J. Chem. Soc. Chem. Commun. 1977, 859–860. [Google Scholar] [CrossRef]
- Kubínová, R.; Gazdová, M.; Hanáková, Z.; Jurkaninová, S.; Dall’Acqua, S.; Cvačka, J.; Humpa, O. New diterpenoid glucoside and flavonoids from Plectranthus scutellarioides (L.) R. Br. S. Afr. J. Bot. 2019, 120, 286–290. [Google Scholar] [CrossRef]
- Ito, T.; Rakainsa, S.K.; Nisa, K.; Morita, H. Three new abietane-type diterpenoids from the leaves of Indonesian Plectranthus scutellarioides. Fitoterapia 2018, 127, 146–150. [Google Scholar] [CrossRef]
- Miyase, T.; Rüedi, P.; Eugster, C.H. Struktur von sechs Coleonen (Diterpenen) aus Solenostemon monostachys (P. BEAUV.) BRIQ. (Labiatae). Helv. Chim. Acta 1980, 63, 95–101. [Google Scholar] [CrossRef]
- Zhou, Q. Natural diterpene and triterpene quinone methides: Structures, synthesis and biological potentials. In Quinone Methides; Rokita, S.E., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2009; pp. 269–295. [Google Scholar]
- Dellar, J.E.; Cole, M.D.; Waterman, P.G. Antimicrobial abietane diterpenoids from Plectranthus elegans. Phytochemistry 1996, 41, 735–738. [Google Scholar] [CrossRef]
- Mothana, R.A.; Al-Said, M.S.; Al-Musayeib, N.M.; Gamal, A.A.E.; Al-Massarani, S.M.; Al-Rehaily, A.J.; Abdulkader, M.; Maes, L. In Vitro Antiprotozoal Activity of Abietane Diterpenoids Isolated from Plectranthus barbatus Andr. Int. J. Mol. Sci. 2014, 15, 8360–8371. [Google Scholar] [CrossRef] [PubMed]
- Rüedi, P.; Eugster, C.H. Diterpenoide Drüsenfarbstoffe aus Labiaten: 6 neue p-Chinomethane aus Plectranthus parviflorus WILLD. Helv. Chim. Acta 1978, 61, 709–715. [Google Scholar] [CrossRef]
- Alder, A.C.; Rüedi, P.; Eugster, C.H. Drüsenfarbstoffe aus tropischen Labiaten: Parviflorone aus Plectranthus strigosus BENTH. Helv. Chim. Acta 1984, 67, 1531–1534. [Google Scholar] [CrossRef]
- Narukawa, Y.; Shimizu, N.; Shimotohno, K.; Takeda, T. Two new diterpenoids from Plectranthus nummularius Briq. Chem. Pharm. Bull. 2001, 49, 1182–1184. [Google Scholar] [CrossRef]
- Nyila, M.A.; Leonard, C.M.; Hussein, A.A.; Lall, N. Bioactivities of Plectranthus ecklonii Constituents. Nat. Prod. Commun. 2009, 4, 1934578X0900400903. [Google Scholar] [CrossRef]
- Burmistrova, O.; Perdomo, J.; Simões, M.F.; Rijo, P.; Quintana, J.; Estevez, F. The abietane diterpenoid parvifloron D from Plectranthus ecklonii is a potent apoptotic inducer in human leukemia cells. Phytomedicine 2015, 22, 1009–1016. [Google Scholar] [CrossRef]
- Gaspar-Marques, C.; Fátima Simões, M.; Luísa Valdeira, M.; Rodriguez, B. Terpenoids and phenolics from Plectranthus strigosus, bioactivity screening. Nat. Product Res. 2008, 22, 167–177. [Google Scholar] [CrossRef]
- Fuson, R.C. The principle of Vinylogy. Chem. Rev. 1935, 16, 1–27. [Google Scholar] [CrossRef]
- Uchida, M.; Miyase, T.; Yoshizaki, F.; Bieri, J.H.; Rüedi, P.; Eugster, C.H. 14-Hydroxytaxodion als Hauptditerpen in Plectranthus grandidentatus GÜRKE.; Isolierung von sieben neuen dimeren Diterpenen aus P. grandidentatus, P. myrianthus BRIQ. und Coleus carnosus HASSK.: Strukturen der Grandidone A, 7-Epi-A, B, 7-Epi-B, C, D und 7-Epi-D. Helv. Chim. Acta 1981, 64, 2227–2250. [Google Scholar]
- Falé, P.L.; Borges, C.; Madeira, P.J.A.; Ascensão, L.; Araújo, M.E.M.; Florêncio, M.H.; Serralheiro, M.L.M. Rosmarinic acid, scutellarein 4′-methyl ether 7-O-glucuronide and (16S)-coleon E are the main compounds responsible for the antiacetylcholinesterase and antioxidant activity in herbal tea of Plectranthus barbatus (“falso boldo”). Food Chem. 2009, 114, 798–805. [Google Scholar] [CrossRef]
- Rüedi, P.; Eugster, C.H. Struktur von Coleon, F. Helv. Chim. Acta 1973, 56, 1129–1132. [Google Scholar] [CrossRef]
- Alder, A.C.; Rüedi, P.; Eugster, C.H. Plectranthone, C und, D. Diterpenoide Phenanthren-1,4-dione aus Blattdrüsen einer Plectranthus sp. (Labiatae). Helv. Chim. Acta 1984, 67, 1003–1011. [Google Scholar] [CrossRef]
- Schultz, C.; Bossolani, M.P.; Torres, L.M.; Lima-Landman, M.T.R.; Lapa, A.J.; Souccar, C. Inhibition of the gastric H+, K+-ATPase by plectrinone A, a diterpenoid isolated from Plectranthus barbatus Andrews. J. Ethnopharmacol. 2007, 111, 1–7. [Google Scholar] [CrossRef]
- Ribi, M.; Sin-Ren, A.C.; Küng, H.P.; Eugster, C.H. Struktur und Reaktionen von Coleon B. 2. Mitteilung über Coleon B. Helv. Chim. Acta 1969, 52, 1685–1728. [Google Scholar] [CrossRef]
- Arihara, S.; Rüedi, P.; Eugster, C.H. Diterpenoide Drüsenfarbstoffe: Coleone S und T aus Plectranthus caninus Roth (Labiatae), ein neues Diosphenol/trans-A/B-6,7-Diketon-Paar aus der Abietanreihe. Helv. Chim. Acta 1977, 60, 1443–1447. [Google Scholar] [CrossRef]
- Rijo, P.; Gaspar-Marques, C.; Simões, M.F.; Jimeno, M.L.; Rodríguez, B. Further diterpenoids from Plectranthus ornatus and P. grandidentatus. Biochem. Syst. Ecol. 2007, 35, 215–221. [Google Scholar] [CrossRef]
- Rüedi, P.; Eugster, C.H. Coleone C, D, I, I′ aus einer madegassischen Plectranthus sp. nov. Interkonversion von cis-und trans-A/B-6, 7-Diketoditerpenen. Helv. Chim. Acta 1975, 58, 1899–1912. [Google Scholar] [CrossRef]
- Rüedi, P.; Eugster, C.H. Struktur von Coleon C, einem neuen Blattfarbstoff aus Coleus aquaticus Gürcke. Helv. Chim. Acta 1971, 54, 1606–1621. [Google Scholar] [CrossRef]
- Moir, M.; Rüedi, P.; Eugster, C.H. Diterpenoid Hydroquinones from Coleus somaliensis (S. Moore): Coleons H., I., and K. Helv. Chim. Acta 1973, 56, 2534–2539. [Google Scholar] [CrossRef]
- Rüedi, P.; Eugster, C.H. Diterpenoide Drüsenfarbstoffe: Coleon L, ein neues Diosphenol aus Coleus somaliensis S. MOORE; Revision der Strukturen von Coleon H., I., I′ und K. Helv. Chim. Acta 1977, 60, 1233–1238. [Google Scholar] [CrossRef]
- Ragasa, C.Y.; Templora, V.F.; Rideout, J.A. Diastereomeric Diterpenes from Coleus blumei. Chem. Pharm. Bull. 2001, 49, 927–929. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rüedi, P.; Eugster, C.H. Coleon D, ein Diterpen aus Coleus aquaticus Guercke mit trans-A/B-6, 7-Dioxostruktur. Helv. Chim. Acta 1972, 55, 1736–1745. [Google Scholar] [CrossRef]
- Buchbauer, G.; Rüedi, P.; Eugster, C.H. Edulon A, ein 2H-1-Oxapyrenderivat aus Plectranthus edulis (Labiatae). Helv. Chim. Acta 1978, 61, 1969–1974. [Google Scholar] [CrossRef]
- Künzle, J.M.; Rüedi, P.; Prewo, R.; Eugster, C.H. Struktur von Edulon A. Helv. Chim. Acta 1986, 69, 1513–1520. [Google Scholar] [CrossRef]
- Kelecom, A.; Dos Santos, T.C. Cariocal, a new seco-abietane diterpene from the Labiate Coleus barbatus. Tetrahedron Lett. 1985, 26, 3659–3662. [Google Scholar] [CrossRef]
- Mothana, R.A.; Khaled, J.M.; El-Gamal, A.A.; Noman, O.M.; Kumar, A.; Alajmi, M.F.; Al-Rehaily, A.J.; Al-Said, M.S. Comparative evaluation of cytotoxic, antimicrobial and antioxidant activities of the crude extracts of three Plectranthus species grown in Saudi Arabia. Saudi Pharm. J. 2019, 27, 162–170. [Google Scholar] [CrossRef]
- Kelecom, A. An abietane diterpene from the Labiate Coleus barbatus. Phytochemistry 1984, 23, 1677–1679. [Google Scholar] [CrossRef]
- Amina, M.; Al-Musayeib, N.M.; Alam, P.; Aleanizy, F.S.; Alqahtni, F.Y.; Al-Said, M.S.; Al-Rashidi, N.S.; Shakeel, F. Cytotoxic evaluation and concurrent analysis of two diterpenes in the chloroform extract of Plectranthus barbatus using a validated HPTLC-UV method. Bull. Chem. Soc. Ethiop. 2018, 32, 407–419. [Google Scholar] [CrossRef]
- Horvath, T.; Linden, A.; Yoshizaki, F.; Eugster, C.H.; Rüedi, P. Abietanes and a Novel 20-Norabietanoid from Plectranthus cyaneus (Lamiaceae). Helv. Chim. Acta 2004, 87, 2346–2353. [Google Scholar] [CrossRef]
- Garcia, C.; Teodósio, C.; Oliveira, C.; Oliveira, C.; Díaz-Lanza, A.; Reis, C.; Duarte, N.; Rijo, P. Naturally occurring Plectranthus-derived diterpenes with antitumoral activities. Curr. Pharm. Des. 2018, 24, 4207–4236. [Google Scholar] [CrossRef]
- Slameňová, D.; Mašterová, I.; Lábaj, J.; Horváthová, E.; Kubala, P.; Jakubíková, J.; Wsólová, L. Cytotoxic and DNA-damaging effects of diterpenoid quinones from the roots of Salvia officinalis L. on colonic and hepatic human cells cultured in vitro. Basic Clin. Pharmacol. Toxicol. 2004, 94, 282–290. [Google Scholar] [CrossRef]
- Fronza, M.; Murillo, R.; Ślusarczyk, S.; Adams, M.; Hamburger, M.; Heinzmann, B.; Laufer, S.; Merfort, I. In vitro cytotoxic activity of abietane diterpenes from Peltodon longipes as well as Salvia miltiorrhiza and Salvia sahendica. Bioorg. Med. Chem. 2011, 19, 4876–4881. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.; Silva, C.O.; Monteiro, C.M.; Nicolai, M.; Viana, A.; Andrade, J.M.; Barasoain, I.; Stankovic, T.; Quintana, J.; Hernandéz, I.; et al. Anticancer properties of the abietane diterpene 6, 7-dehydroroyleanone obtained by optimized extraction. Future Med. Chem. 2018, 10, 1177–1189. [Google Scholar] [CrossRef]
- Xing, X.; Wu, H.; Wang, X.; Huang, Y.; Li, Q.; Li, C.; Yang, Y.; Liu, Y.; Liu, J. Inhibition of tumor cell proliferation by coleon C. J. Chemother. 2008, 20, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Alegre-Gómez, S.; Sainz, P.; Simões, M.F.; Rijo, P.; Moiteiro, C.; González-Coloma, A.; Martínez-Díaz, R.A. Antiparasitic activity of diterpenoids against Trypanosoma cruzi. Planta Med. 2017, 83, 306–311. [Google Scholar] [CrossRef]
- Zadali, R.; Ebrahimi, S.N.; Tofighi, Z.; Es-Haghi, A.; Hamburger, M.; Kaiser, M.; D’ Ambola, M.; De Tommasi, N.; Hadjiakhoondi, A. Antiprotozoal activity of diterpenoids isolated from Zhumeria majdae- absolute configuration by circular dichroism. DARU J. Pharm. Sci. 2020, 28, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Areche, C.; Schmeda-Hirschmann, G.; Theoduloz, C.; Rodríguez, J.A. Gastroprotective effect and cytotoxicity of abietane diterpenes from the Chilean Lamiaceae Sphacele chamaedryoides (Balbis) Briq. J. Pharm. Pharmacol. 2009, 61, 1689–1697. [Google Scholar] [CrossRef]
- Murakami, S.; Muramatsu, M.; Tomisawa, K. Inhibition of gastric H+, K+-ATPase by flavonoids: A structure-activity study. J. Enzyme Inhib. 1999, 14, 151–166. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gáborová, M.; Šmejkal, K.; Kubínová, R. Abietane Diterpenes of the Genus Plectranthus sensu lato. Molecules 2022, 27, 166. https://doi.org/10.3390/molecules27010166
Gáborová M, Šmejkal K, Kubínová R. Abietane Diterpenes of the Genus Plectranthus sensu lato. Molecules. 2022; 27(1):166. https://doi.org/10.3390/molecules27010166
Chicago/Turabian StyleGáborová, Mária, Karel Šmejkal, and Renata Kubínová. 2022. "Abietane Diterpenes of the Genus Plectranthus sensu lato" Molecules 27, no. 1: 166. https://doi.org/10.3390/molecules27010166
APA StyleGáborová, M., Šmejkal, K., & Kubínová, R. (2022). Abietane Diterpenes of the Genus Plectranthus sensu lato. Molecules, 27(1), 166. https://doi.org/10.3390/molecules27010166