Abstract
An efficient synthetic route to highly substituted dihydroquinolines and dihydronaphthyridines has been developed using a domino reaction of Morita-Baylis-Hillman (MBH) acetates with primary aliphatic and aromatic amines in DMF at 50–90 °C. The MBH substrates incorporate a side chain acetate positioned adjacent to an acrylate or acrylonitrile aza-Michael acceptor as well as an aromatic ring activated toward SNAr ring closure. A control experiment established that the initial reaction was an SN2′-type displacement of the side chain acetate by the amine to generate the alkene product with the added nitrogen nucleophile positioned trans to the SNAr aromatic ring acceptor. Thus, equilibration of the initial alkene geometry is required prior to cyclization. A further double bond migration was observed for several reactions targeting dihydronaphthyridines from substrates with a side chain acrylonitrile moiety. MBH acetates incorporating a 2,5-difluorophenyl moiety were found to have dual reactivity in these annulations. In the absence of O2, the expected dihydroquinolines were formed, while in the presence of O2, quinolones were produced. All of the products were isolated in good to excellent yields (72–93%). Numerous cases (42) are reported, and mechanisms are discussed.
1. Introduction
We recently disclosed a synthesis of 1,3,6-trisubstituted naphthalenes and 6,8-disubstituted quinolines from Morita-Baylis-Hillman (MBH) acetates and active methylene compounds (AMCs) in DMF with K2CO3 at 23–90 °C [1]. This process formally involved SN2′ displacement of the acetoxy group by the stabilized AMC anion, a second deprotonation, SNAr cyclization and elimination to give the aromatic product. However, contrary to previous reports, our experiments indicated that the initial adduct was formed with the added nucleophile trans to the SNAr acceptor ring, necessitating double bond equilibration before cyclization. The current project sought to develop an efficient route to dihydroquinolines and dihydronaphthyridines from MBH acetates. This work differs from our earlier contribution [1] in providing a route to the title heterocycles wherein a dihydropyridine is fused to a preexisting ring and cannot aromatize. The sequence was initiated by addition of a nitrogen rather than a carbon nucleophile and required mild heating, but no added base. Despite the more active nucleophile, the reaction followed a mechanism similar to our previous report. An interesting double bond migration was observed in several naphthyridines generated from acrylonitrile-derived precursors. Finally, in addition to yielding dihydroquinolines under anaerobic conditions, a difluoro-substituted MBH substrate showed disparate reactivity in generating a quinolone when reacted in the presence of O2. These findings complement earlier cyclizations by our group to form 1-alkyl-2,3-dihydro-4(1H)-quinolinones [2] and 1-alkyl-2,3-dihydro-1,8-naphthyridine-4(1H)-ones [3] by a Michael-SNAr process.
MBH adducts hold great potential in drug synthesis [4,5] due to their high functional density, which makes them prime candidates for domino reactions to prepare a wide range of cyclic targets. 1,2-Dihydroquinolines exhibit valuable biological activity against a wide range of human and animal health disorders [6,7,8,9]. The corresponding 1,2-dihydro-1,8-naphthyridine derivatives appear to have fewer current uses [10,11], but this may stem from a lack of procedures to generate a diverse selection of these targets. Fluoroquinolones were first introduced as antibiotics in the early 1960s and many derivatives have been reported [12,13]. Although bacteria have developed a resistance to some derivatives [12,13], there are many analogs that remain in use to treat various illnesses [14,15,16,17]. Addition of primary amines to MBH acetates permits rapid assembly of each of these privileged structures with a minimum of purifications.
Previous work has described a number of syntheses of nitrogen heterocycles from MBH precursors. For example, a synthesis of 3-quinolinecarboxylic esters has been reported from ethyl 2-((2-chlorophenyl)((tosylsulfonamido)methyl)acrylate and catalytic tosylsulfonamide [18] (Figure 1A). A second study described an approach to N-substituted-1,2-dihydrobenzo[b][1,8]-naphthyridines by reaction of MBH acetates derived from 2-chloroquinoline-3-carboxaldehyde with primary amines [19] (Figure 1B). A later account detailed a synthesis of dihydroacridines from this same MBH precursor with AMCs [20] (Figure 1C). Basavaiah and co-workers reacted nitroalkane anions, from nitroalkanes and K2CO3, with MBH acetates to give adducts that were subjected to a reduction-cyclization sequence to afford γ-lactams [21] (Figure 1D). In a slightly different application, this same group converted MBH alcohols to 3,4-dihydrobenzo[b][1,8]naphthyridin-2(1H)-ones by a Johnson–Claisen rearrangement with triethyl orthoesters to give ethyl (Z)-4-cyano-5-(2-nitrophenyl)-4-pentenoate derivatives which were converted to the heterocycles using a domino cyclization initiated by reduction of the nitro function [22] (Figure 1E). Finally, an approach to fused polycyclic quinolines starting from MBH acetates and methyl prolinate or imidazole-2-carboxaldehyde has also appeared [23] (Figure 1F). Several of the cited papers assumed the initial SN2′ reaction resulted in the correct alkene geometry for cyclization [18,21,22]. Only a few noted that the initial addition–elimination process afforded a Z alkene (or a mixture of Z and E) which lacked the geometry needed for direct cyclization [19,20]. In the current study, MBH acetates were reacted with primary amines to produce 3,6-disubstituted 1-alkyl/phenyl-1,2-dihydroquinolines and 3-substituted 1-alkyl/phenyl-1,2-dihydro-1,8-naphthyridines in good to excellent yields. In our work, only the Z alkene was observed, and thus would require a Z→E alkene equilibration prior to cyclization.
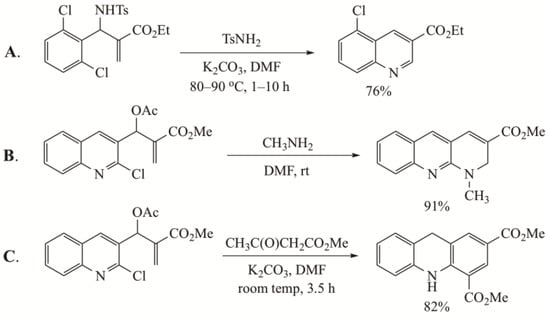
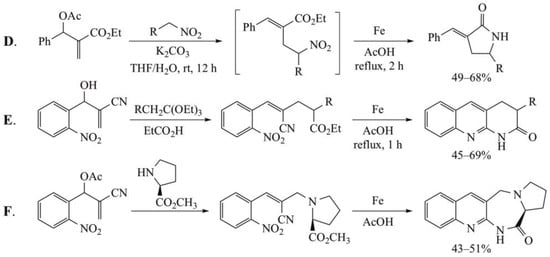
Figure 1.
Some previous nitrogen heterocycle syntheses from MBH precursors. (A) Formation of quinolines; (B) formation of N-substituted-1,2-dihydrobenzo[b][1,8]-naphthyridines; (C) formation of dihydroacridines; (D) formation of γ-lactams; (E) formation of 3,4-dihydrobenzo[b][1,8]naphthyridin-2(1H)-ones; (F) formation of fused polycyclic quinolines.
2. Results and Discussion
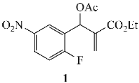
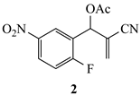
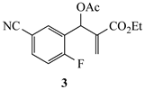
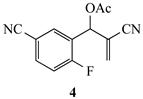
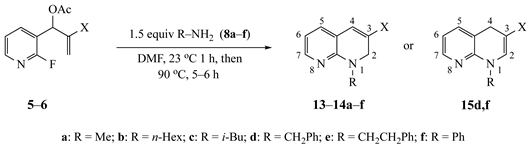
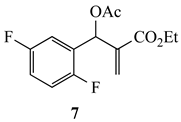
Our study focused on MBH acetates 1–7, four with 2-fluoroaromatic rings activated toward SNAr ring closure by C5 nitro or cyano groups, and two with a 2-fluoropyridine group and one with a 2,5-difluoroaromatic ring (Figure 2). The Michael acceptors embedded in the side chains were either acrylate esters or acrylonitriles. These precursors were prepared by a Morita-Baylis-Hillman reaction using 1 equiv of the aromatic aldehyde, 2 equiv of the activated alkene and 1.2 equiv of 1,4-diazabicyclo[2.2.2]octane (DABCO) [1] in acetonitrile (2 days, 23 °C) to afford 86–98% yields. Acylation of the alcohol adducts with no allylic inversion was achieved in ≥95% yield after 30 min at 0 °C by treatment with acetic anhydride and catalytic trimethylsilyl trifluoromethanesulfonate, as has been previously described [1,24]. These substrates required no purification and were used directly in the annulation procedure.
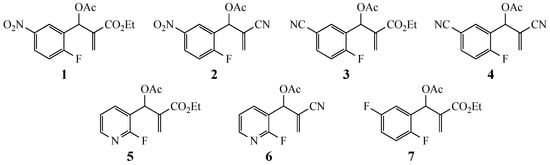
Figure 2.
Substrates for the preparation of 3,6-disubstituted 1-alkyl/phenyl-1,2-dihydroquinolines (1–4 and 7), 3-substituted 1-alkyl/phenyl-1,2-dihydro-1,8-naphthyridines (5,6) and quinolones (7).
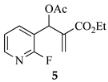
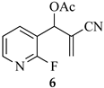
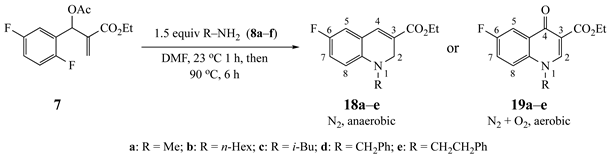
The results from reactions of 1–4 (1 equiv) with primary aliphatic or aromatic amines 8 (1.5 equiv) in DMF (1 h, 23 °C, and 1–5 h, 50 °C) gave the 3,6-disubstituted 1-alkyl/phenyl-1,2-dihydroquinolines 9–12 shown in Table 1. A similar protocol was used to generate dihydronaphthyridines 13 and 14 from precursors 5 and 6, respectively (Table 2). Finally, the difluoro-substituted substrate 7 showed dual reactivity under anaerobic versus aerobic conditions, giving dihydroquinolines 18 in the absence of O2 and quinolones 19 when O2 was present (Table 3). As in our previous work [1], it was also noted that the 2-fluoropyridine (and 2,5-difluorophenyl) precursors were less reactive, requiring higher temperatures and longer reaction times (1 h, 23 °C and 5–6 h, 90 °C) for complete conversion. This appears to reflect the decreased activation of these acceptor rings in the final SNAr ring-closing step. Furthermore, while the 2-fluoropyridine-bound acrylate 5 successfully delivered dihydronaphthyridines 13, the corresponding acrylonitrile 6 often yielded products 14 contaminated by uncyclized material. Although acrylates and acrylonitriles are among the most reactive acceptors in aza-Michael reactions, alkyl substitution α to the electron-withdrawing group (EWG) on these substrates slows the addition, and this may impact their relative reactivities toward nucleophiles [25]. Additionally, reactions of acetates 6d and 6f also yielded double bond-migrated products 15d and 15f, respectively (Figure 3). Finally, discussion of the unique reactivity of difluorophenyl substrate 7 is provided at the end of the article.

Table 1.
Synthesis of 3,6-disubstituted 1-alkyl/phenyl-1,2-dihydroquinolines.

Table 2.
Synthesis of 3-substituted 1-alkyl/phenyl-1,2 and 1,4-dihydronaphthyridines.

Table 3.
Synthesis of ethyl 1-alkyl-1,2 and 1,4-dihydronaphthyridine-3-carboxylates.
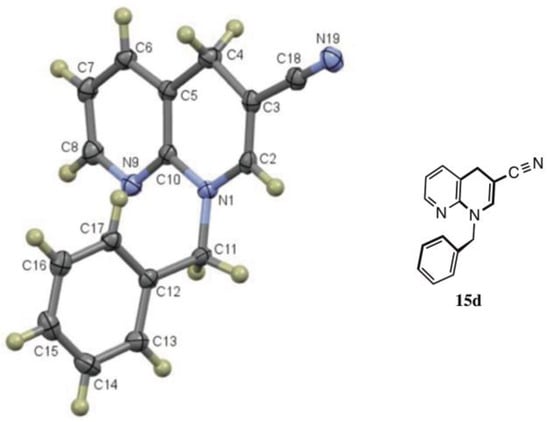
Figure 3.
X-ray structure of 1-benzyl-1,4-dihydro-1,8-naphthyridine-3-carbonitrile (15d, CCDC 2035027).
The process is very efficient, requiring only one step from the MBH acetates and one column chromatography (SiO2, 10% EtOAc in hexanes) for each final product. The heterocycles were formed in good to excellent yields (72–93% for all substrates except 6, see below) and were fully characterized by analytical and spectroscopic methods. The IR spectra indicated the presence of the expected functional groups (conjugated CO2Et or CN, polarized conjugated double bonds and NO2). The 1H NMR of the dihydroquinolines showed a doublet (J1,3 < 3.0 Hz), a doublet of doublets (J1,2 and 1,3 = large and small) and a doublet (J1,2 > 9.0 Hz), respectively, for protons on C5, C7 and C8 with chemical shifts in accord with those expected for protons adjacent to electron-withdrawing or donating functionality. The dihydropyridine subunit of these products showed a broad singlet at δ 7.0–7.5 for the C4 alkene proton and a small doublet (Jallylic < 2 Hz) at δ 4.5–5.0 for the C2 ring methylene. The 1H NMR spectra of the dihydronaphthyridines showed the expected chemical shifts and coupling patterns for the protons at C5, C6 and C7 of the system, while the protons at C4 and C2 of these derivatives were similar to those of the dihydroquinolines. For all products, the ethyl ester, if present, was also clearly discernible as were proton signals for the added amine fragment. The 13C NMR spectra, for all derivatives, were appropriate with respect to the number of carbonyl, nitrile, aromatic and aliphatic carbons. The mass spectra showed an ion corresponding to the molecular weight of the compound and the elemental analyses confirmed the formulas and purity.
An interesting observation was noted in cyclizations of substrate 6, which consisted of a 2-fluoropyridine having an acrylonitrile moiety in the side chain. These reactions required heating for 6 h at 90 °C to assure complete conversion. While n-hexylamine and phenethylamine gave normal 1,2-dihydronaphthyridine products 14b (82%) and 14e (80%), respectively, benzylamine and aniline gave 1,4-dihydronaphthyridines 15d (82%) and 15f (72%) as the major products, resulting from migration of the double bond from C3-C4 to C2-C3. X-ray analysis confirmed the structure of double bond-migrated product 15d from benzylamine (8d) with 6 (see Figure 3 and the SI). The 13C NMR of these systems was distinguished by the presence of the β-enamine carbon bearing the CN group (C3) which appeared at δ 75–80 in the spectrum. A reason for this isomerization is not clear since all of the reactions were performed under the same conditions. However, there is stabilization available to the enaminonitrile, as the dihydropyridine π system is still conjugated with the fused pyridine ring, although the required resonance contributor places a positive charge on an electronegative nitrogen.
Two basic mechanisms are possible for this transformation: (1) initial SNAr attack of the amino group at the aromatic ring, followed by conjugate addition to the side chain double bond with elimination of acetoxy (OAc) or (2) initial SN2′-type displacement of OAc from the side chain, followed by an SNAr cyclization. Option 1 would unequivocally predict the observed product of the reaction and would not be dependent on alkene geometry in the intermediate for ring closure. This was ruled out for the dihydronaphthyridines by a control experiment wherein benzylamine (8d) was stirred with 2-fluoropyridine for 12 h at 50 °C to afford none of the SNAr product. In the dihydroquinoline series, substitution by 8d was noted after 5 h at 50 °C for acceptor rings activated by a C4 NO2 (complete conversion) and CN (modest conversion), but evidently this was slower than addition to the side chain aza-Michael acceptors (see below). Option 2 would require a diastereoselective addition to give the E alkene that positions the amine nitrogen cis to the SNAr acceptor ring. To examine this aspect of the reaction, the adduct from addition of 8d to the acrylonitrile moiety of 2 was intercepted after 30 min at 23 °C. Although our earlier paper utilized a similar control to probe the reaction chronology for a carbon nucleophile, this same experiment was necessary in the current study since nitrogen, which is generally more reactive in the SNAr reaction, could potentially attack the aryl ring first and result in a different sequence of events. From this control reaction, intermediate 16 was isolated in 76% yield and was accompanied by 11% of the ring closed dihydroquinoline product 10d. X-ray analysis of intermediate 16 revealed it to be (Z)-2-((benzyl-amino)methyl)-3-(2-fluoro-5-nitrophenyl)acrylonitrile having the amine nitrogen trans to the SNAr acceptor ring (see Figure 4 and the SI). Thus, Option 2 above likely predominates, followed by double bond equilibration to permit the SNAr ring closure. No other intermediates were observed as the reaction proceeded. Presumably, any E alkene produced in the reaction rapidly cyclized and eluded detection by standard methods used to monitor reactions. Further evidence for double bond equilibration was established by heating of 16 in DMF for 1 h at 50 °C, which afforded 10d in 60% yield (unoptimized).
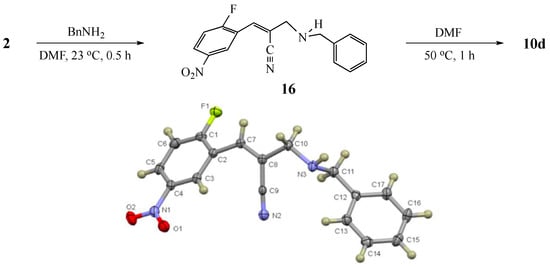
Figure 4.
Control reaction for selective conjugate addition and formation of (Z)-2-((benzylamino)methyl)-3-(2-fluoro-5-nitrophenyl)acrylonitrile (16, CCDC 2035028) and its further conversion to 10d.
In reactions of the side chain allylic acetate, there are two mechanistic scenarios to explain the addition of amines to MBH acetates. The first involves the SN2′ process shown in Scheme 1 for the addition of benzylamine (8d) to MBH acetate 2 to give 10d. The initial conformation of 2 would allow minimal steric interaction between the aromatic ring, the OAc substituent and the EWG [26]. While the OAc impedes approach to the top face of the Michael acceptor, the planar aryl group should not significantly hinder approach to the bottom face of this moiety. Addition of 8d to the bottom face of the molecule would result in a build-up of negative charge on the top face of the allylic system as shown in A, leading to elimination of acetic acid and formation of the Z alkene 16 [27]. Elimination of acetic acid would require a 120° rotation to bring the OAc anti to the site of negative charge. This rotation should also move the large 2-fluoro-5-nitrophenyl SNAr acceptor away from the newly added nucleophile. Such a rotation would be relatively easy when EWG is a sterically small group such as CN, but could be more difficult when EWG is a larger CO2Et. Equilibration of 16 to 17 [28,29,30,31,32] and attack of the side chain nitrogen at the fluorine-bearing aromatic carbon would give Meisenheimer intermediate C. Rearomatization via loss of fluoride and deprotonation of the ammonium nitrogen would lead to dihydroquinoline 10d. A similar process with the fluoropyridine precursors could be invoked for the formation of dihydronaphthyridines.
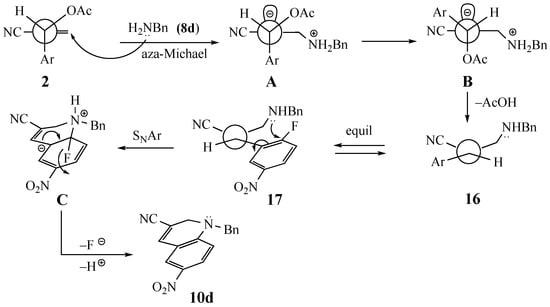
Scheme 1.
Plausible mechanism for the formation of dihydroquinoline 10d from MBH acetate 2 and amine 8d.
An alternative mechanism proposed for SN2′ reactions must also be considered [33]. According to this hypothesis, the substrate could lose the OAc group to give the aryl allyl stabilized carbocations D and E (Scheme 2), which should then add the amine nucleophiles to the least hindered terminal carbon of the allylic system. While steric congestion around the alkene terminus is lower in carbocation E, it is difficult to envision any selectivity in producing only the Z alkene 16 with the added amine trans to the SNAr acceptor. In our studies, here and elsewhere [1], a product having the added nucleophile cis to the acceptor ring was not observed. This could potentially rule out this mechanistic option, unless the cyclization of 17 is sufficiently fast that it is not possible to observe or isolate this intermediate.
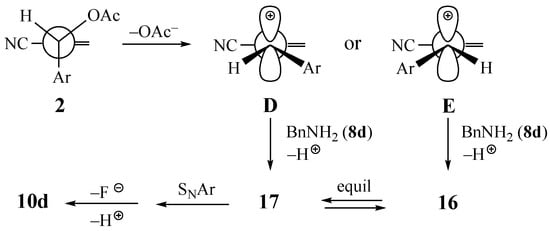
Scheme 2.
Possible carbocation mechanism for the formation of dihydroquinoline 10d.
Two possible pathways for the equilibration of 16 to 17 required prior to SNAr cyclization are depicted in Scheme 3. The first, and most likely, is a well-precedented aza-Michael–reverse aza-Michael sequence [28,29,30,31,32] involving the excess amine in the reaction. A second option is analogous to an intramolecular addition–elimination process previously described [1]. This alternative mechanism would require addition of the amine nitrogen in 16 to the aryl-conjugated double bond to give the stabilized aziridinyl benzylic anion F. This scenario is viable only if the aromatic ring is sufficiently electron-deficient to stabilize the anionic center. Bond rotation to give G and ring opening would then deliver the E alkene needed for cyclization. Once equilibration occurs, ring closure and deprotonation to furnish 10d should be facile. Even if the equilibrium does not strongly favor the necessary alkene geometry, product formation should gradually funnel the initial adduct over to the fused heterocyclic target.
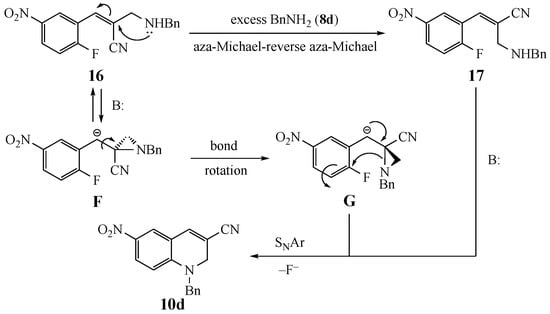
Scheme 3.
Plausible mechanisms for double bond equilibration.
In the current reaction, dihydropyridine annulation would require SNAr ring closure from the E alkene (aryl and added amine cis). An alternative progression where the SNAr occurred first would preclude the necessity for stereoselective formation of an E alkene for cyclization but evidence for this pathway was lacking. While several previous studies assumed the formation of the E alkene intermediate with the added amine proximal to the SNAr acceptor ring, our experiments permitted isolation of only the Z alkene, and this appears to be the major pathway. If any of the E alkene intermediate was formed, it likely underwent rapid ring closure. Thus, an equilibration of the alkene geometry was essential to provide high yields of cyclized products. This equilibration is evidently facile as further treatment of isolated intermediate 16 in DMF for 1 h at 50 °C afforded dihydroquinoline 10d.
We were also interested in extending this reaction to other substrates with activating functionality. Treatment of ethyl 2-(acetoxy(2,5-difluorophenyl)methyl)acrylate (7) with amines 8 in the same molar ratio afforded ethyl 1-alkyl-6-fluoro-1,2-dihydroquinoline-3-carboxylates 18 (Table 3). These reactions also required heating at 90 °C to proceed to completion. An interesting discovery was made, however, when the amine reaction was run using an older bottle of anhydrous DMF. In this experiment, ethyl 1-alkyl-6-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylates (e.g., fluoroquinolones 19) were isolated rather than the expected dihydroquinolines (Table 3). It was rationalized that this could be due to the presence of dissolved O2 in the older bottle of solvent. It is known that O2 readily dissolves in DMF up to a concentration of ca 4.5 mM at 25 °C [34]. Indeed, when O2 was bubbled through DMF from a fresh bottle of anhydrous DMF for 15 s using a gas dispersion tube, the reaction produced the quinolone as the only product. On the small scale studied, the dissolved O2 was sufficient for complete conversion, and an O2 atmosphere was not needed. This competing reaction is likely facilitated by the higher temperatures necessary to drive the ring closure. Spectroscopically, the 1H NMR spectra of the fluorodihydroquinolines displayed the typical signals with additional line broadening or coupling by the fluorine. The fluoroquinolones exhibited a singlet at δ 7.4 for the quinolone β-enone proton as well as a singlet at δ 8.0–8.5, a doublet of doublets at δ 8.3 (J = ca 9.0, 3.0 Hz), and a doublet at δ 7.4 (J = ca 5–6 Hz when R = CH3 or i-Bu; this signal was obscured when R = CH2Ph or CH2CH2Ph) for protons at C5, C7 and C8, respectively. Additionally, the C2 ring methylene, observed for the dihydroquinolines, was absent. The 13C NMR spectra, showed the correct number and types of carbons with added coupling in the fluoroaromatic rings. IR and mass spectra were consistent with the structures indicated. Proof of the quinolone structures was obtained by X-ray analysis of 19e, derived from reaction of 7 with phenethylamine (8e, See Figure 5s and SI).
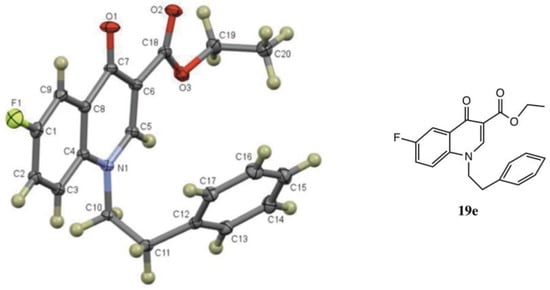
Figure 5.
ORTEP drawing of quinolone 19e (CCDC 2057847).
The oxidation process observed for the difluoro substrate 7 has no precedent under the current conditions and we have proposed a possible mechanism in Scheme 4 for its reaction with benzylamine (8d) to give 19d. The MBH acetate has a very labile allylic-benzylic C–H bond between the side chain Michael acceptor and the aromatic ring. Insertion of O2 into this bond would generate an intermediate hydroperoxide H as the initial intermediate. Addition of the amine to the side chain acrylate with loss of the OAc would then give the unsaturated MBH hydroperoxide I. SNAr ring closure (via direct addition or equilibration-addition) to Meisenheimer intermediate J and fluoride elimination would afford hydroperoxide K. Final elimination of water from this intermediate, promoted by excess amine, would then provide quinolone 19d.
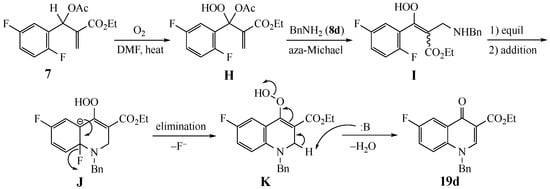
Scheme 4.
Possible mechanism for the conversion of difluoro substrate 7 to fluoroquinolone 19d.
This novel oxidation extends the possible uses of MBH acetates to access heterocycles with potential medicinal value. The difluorinated acetate 7 with less SNAr activation required higher temperatures for reaction and this led to a new reaction pathway when O2 was present. More reactive substrates 1–6 were converted to dihydroquinolines at lower temperature and did not undergo competitive oxidation to the quinolones. Attempts to induce this oxidation in 1–6 using O2-saturated DMF at 90 °C failed, and only dihydroquinolines were isolated, albeit in lower yields. The current reaction represents a new approach to fluoroquinolones, which may be further developed to produce previously inaccessible analogs.
3. Materials and Methods
3.1. General Methods
Unless otherwise indicated, all reactions were carried out under dry N2 in oven-dried glassware. All reagents and solvents were used as received. Reactions were monitored by thin layer chromatography on silica gel GF plates (Analtech No 21521, Newark, DE, USA). Preparative separations were performed by flash chromatography on silica gel (Davisil®, grade 62, 60–200 mesh, Sorbent Technologies, Norcross, GA, USA) containing UV-active phosphor (Sorbent Technologies No. UV-05) slurry packed into quartz columns. Band elution for all chromatographic separations was monitored using a hand-held UV lamp (Fisher Scientific, Pittsburgh, PA, USA). Melting points were obtained using a MEL-TEMP apparatus (Cambridge, MA, USA) and are uncorrected. FT-IR spectra were run using a Varian Scimitar FTS 800 spectrophotometer (Randolph, MA, USA) as thin films on NaCl disks. 1H- and 13C-NMR spectra were measured using a Bruker Avance 400 system (Billerica, MA, USA) in the indicated solvents at 400 MHz and 101 MHz, respectively, with (CH3)4Si as the internal standard; coupling constants (J) are given in Hz. Low-resolution mass spectra were obtained using a Hewlett-Packard Model 1800A GCD GC–MS system (Palo Alto, CA, USA). Details of the X-ray structure determinations are given in the SI. Elemental analyses (±0.4%) were determined by Atlantic Microlabs (Norcross, GA, USA).
3.2. Syntheses of the MBH Acetates
The syntheses of MBH acetates 1–5 have been previously described [1].
3.2.1. 2-Cyano-1-(2-fluoropyridin-3-yl)allyl Acetate (6)
Compound 6 was prepared from the MBH alcohol according to the procedure given for the preparation of compound 5 [1] and did not require any further purification. Yield: 209 mg (0.95 mmol, 95%) as a colorless oil; IR: 2231, 1756, 1609 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.25 (apparent s, 1H), 7.99 (apparent t, J = 8.5 Hz, 1H), 7.30 (m, 1H), 6.53 (s, 1H), 6.18 (s, 1H), 6.16 (s, 1H), 2.21 (s, 3H); 13C NMR (101 MHz, CDCl3): δ 168.8, 160.2 (d, J = 240.0 Hz), 148.4 (d, J = 15.0 Hz), 138.8 (d, J = 3.7 Hz), 133.8, 122.4 (d, J = 4.4 Hz), 120.9, 118.5 (d, J = 27.9 Hz), 115.4, 68.6 (d, J = 2.9 Hz), 20.8; MS (EI): m/z 220; Anal. Calcd for C11H9FN2O2: C, 60.00; H, 4.12; N, 12.72. Found: C, 59.88; H, 4.08; N, 12.64. After optimization, this procedure was scaled up by a factor of five with no significant decrease in yield.
3.2.2. Ethyl-2-(acetoxy(2,5-difluorophenyl)methyl)acrylate (7)
Compound 7 was prepared according to the procedure given for the preparation of compound 1 [1] and did not require any further purification. Yield: 278 mg (0.98 mmol, 98%) as a colorless oil; IR: 1752, 1723, 1638 cm−1; 1H NMR: δ 7.05–6.95 (complex, 3H), 6.88 (s, 1H), 6.47 (s, 1H), 5.82 (s, 1H), 4.18 (q, J = 7.1 Hz, 2H), 2.12 (s, 3H), 1.24 (t, J = 7.1 Hz, 3H); 13C NMR: δ 169.0, 166.4, 164.5, 158.5 (dd, J = 242.7, 2.3 Hz), 156.2 (dd, J = 245.6, 2.5 Hz), 138.2, 126.9 (m, 2C), 116.7 (dd, J = 24.7, 8.5 Hz), 115.4 (dd, J = 24.9, 3.7 Hz), 67.0, 61.1, 20.8, 13.9; MS (EI): m/z 284; Anal. Calcd for C14H14F2O4: C, 59.16; H, 4.96. Found: C, 59.19; H, 4.99. After optimization, this procedure was scaled up by a factor of five with no significant decrease in yield.
3.3. Representative Procedure for the Synthesis of Dihydroheteroaromatics Using MBH Acetates and 1° Alkyl or Aromatic Amines
In a 25 mL, round-bottomed flask, a solution of MBH acetate (1 mmol) in DMF (2 mL) was treated with the corresponding amine (1.5 mmol), and the mixture was stirred for 1 h at room temperature (23 °C). For the dihydroquinolines, the temperature was gradually increased to 50 °C, and the reaction was stirred for an additional 4 h. For the dihydronaphthyridines, the temperature was increased gradually to 90 °C and stirring was continued for 4 h. At this time, TLC (20% EtOAc/hexane) indicated complete consumption of the starting material, the solution was poured into de-ionized water (15 mL), and extracted with EtOAc (3 × 10 mL). The organic layer was washed with 10% aq. NH4Cl (2 × 10 mL), followed by saturated aq. NaHCO3 (2 × 10 mL). The combined organic layers were washed with saturated aq. NaCl and dried (Na2SO4). Removal of the solvent under vacuum gave a crude product, which was further purified by column chromatography (10% EtOAc in hexane) to afford the dihydroheteroaromatic compounds.
3.3.1. Ethyl-1-methyl-6-nitro-1,2-dihydroquinoline-3-carboxylate (9a) from 1 and Methylamine (8a)
Yield: 244 mg (93%) as a yellow solid, m.p. 117–118 °C; IR: (nujol) 1692, 1593, 1317 cm−1; 1H NMR (400 MHz, DMSO-d6): δ 8.01 (d, J = 2.7 Hz, 1H), 7.95 (dd, J = 9.2, 2.7 Hz, 1H), 7.42 (s, 1H), 6.60 (d, J = 9.2 Hz, 1H), 4.45 (br d, J = 1.1 Hz, 2H), 4.20 (q, J = 7.1 Hz, 2H), 2.92 (s, 3H), 1.26 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, DMSO-d6): δ 164.5, 151.9, 136.7, 133.9, 128.9, 125.8, 123.1, 118.6, 109.8, 66.0, 51.3, 37.9, 14.6; MS (EI): m/z 262 [M]+·; Anal. Calcd for C13H14N2O4: C, 59.54; H, 5.38; N, 10.68. Found: C, 59.51; H, 5.37; N, 10.75.
3.3.2. Ethyl-1-hexyl-6-nitro-1,2-dihydroquinoline-3-carboxylate (9b) from 1 and n-hexylamine (8b)
Yield: 289 mg (0.87 mmol, 87%) as a yellow solid, m.p. 98–99 °C; IR: 1703, 1656, 1600, 1516, 1321 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.00 (dd, J = 9.2, 2.7 Hz, 1H), 7.84 (d, J = 2.7 Hz, 1H), 7.28 (s, 1H), 7.25, 6.38 (d, J = 9.2 Hz, 1H), 4.51 (d, J = 1.5 Hz, 2H), 4.27 (q, J = 7.1 Hz, 2H), 3.26 (t, J = 7.8 Hz, 2H), 1.63 (quintet, J = 7.8 Hz, 2H), 1.42–1.31 (complex, 9H), 0.91 (t, J = 7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.6, 150.3, 137.8, 137.3, 133.8, 128.9, 128.7, 128.5, 127.0, 125.9, 122.3, 118.5, 108.7, 61.1, 52.5, 50.1, 31.5, 14.3; MS (EI): m/z 332 [M]+·; Anal. Calcd for C18H24N2O4: C, 65.04; H, 7.28; N, 8.43. Found: C, 64.97; H, 7.24; N, 8.34.
3.3.3. Ethyl-1-isobutyl-6-nitro-1,2-dihydroquinoline-3-carboxylate (9c) from 1 and Isobutylamine (8c)
Yield: 160 mg (93%) as a yellow solid, m.p. 104–105 °C; IR: (nujol) 1704, 1654, 1601, 1575, 1322 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.97 (dd, J = 9.3, 2.7 Hz, 1H), 7.84 (d, J = 2.7 Hz, 1H), 7.28 (s, 1H), 6.41 (d, J = 9.3 Hz, 1H), 4.52 (d, J = 1.5 Hz, 2H), 4.27 (q, J = 7.1 Hz, 2H), 3.10 (d, J = 7.5 Hz, 2H), 2.15 (nonet, J = 6.9 Hz, 1H), 1.35 (t, J = 7.1 Hz, 3H), 1.01 (d, J = 6.7 Hz, 6H); 13C NMR (101 MHz, CDCl3): δ 164.7, 151.1, 137.0, 134.0, 128.4, 126.0, 122.1, 118.4, 109.2, 61.1, 58.3, 50.9, 26.1, 20.2, 14.3; MS (EI): m/z 304 [M]+·; Anal. Calcd for C16H20N2O4: C, 63.14; H, 6.62; N, 9.20. Found: C, 63.19; H, 6.65; N, 9.13.
3.3.4. Ethyl-1-benzyl-6-nitro-1,2-dihydroquinoline-3-carboxylate (9d) from 1 and Benzylamine (8d)
Yield: 313 mg (92%) as a yellow solid, m.p. 197–199 °C; IR: 1703, 1654, 1601, 1571, 1320 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.94 (dd, J = 9.2, 2.7 Hz, 1H), 7.89 (d, J = 2.7 Hz, 1H), 7.40–7.29 (complex, 3H), 7.38 (s, 1H), 7.25 (d, J = 7.5 Hz, 2H), 6.43 (d, J = 9.3 Hz, 1H), 4.57 (d, J = 1.1 Hz, 2H), 4.54 (s, 2H), 4.26 (q, J = 7.1 Hz, 2H), 1.33 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.5, 150.8, 137.7, 134.5, 133.7, 129.1, 128.5, 127.9, 126.7, 125.8, 122.8, 118.5, 109.4, 61.1, 54.2, 50.5, 14.3; MS (EI): m/z 338 [M]+·; Anal. Calcd for C19H18N2O4: C, 67.45; H, 5.36; N, 8.28. Found: C, 67.39; H, 5.35; N, 8.19.
3.3.5. Ethyl-6-nitro-1-phenethyl-1,2-dihydroquinoline-3-carboxylate (9e) from 1 and Phenethylamine (8e)
Yield: 269 mg (83%) as a yellow solid, m.p. 139–140 °C; IR: 1704, 1652, 1601, 1572, 1324 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.99 (dd, J = 9.2, 2.7 Hz, 1H), 7.86 (d, J = 2.7 Hz, 1H), 7.34 (t, J = 7.4 Hz, 2H), 7.28 (s, 1H), 7.27–7.22 (complex, 3H), 6.41 (d, J = 9.2 Hz, 1H), 4.47 (d, J = 1.5 Hz, 2H), 4.27 (q, J = 7.1 Hz, 2H), 3.51 (t, J = 7.7 Hz, 2H), 2.92 (t, J = 7.7. Hz, 2H), 1.34 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.6, 150.3, 137.8, 137.3, 133.8, 128.9, 128.7, 128.5, 127.0, 125.9, 122.3, 118.5, 108.7, 61.1, 52.5, 50.1, 31.5, 14.3; MS (EI): m/z 352 [M]+·; Anal. Calcd for C20H20N2O4: C, 68.17; H, 4.97; N, 6.84. Found: C, 68.12; H, 4.91; N, 6.77.
3.3.6. Ethyl-6-nitro-1-phenyl-1,2-dihydroquinoline-3-carboxylate (9f) from 1 and Aniline (8f)
Yield: 313 mg (89%) as a yellow solid, m.p. 149–150 °C; IR: 1692,1601,1518,1318 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.99 (dd, J = 9.2, 2.7 Hz, 1H), 7.86 (d, J = 2.7 Hz, 1H), 7.34 (t, J = 7.4 Hz, 2H), 7.28 (s, 1H), 7.25 (obscured, 2H), 6.41 (d, J = 9.2 Hz, 1H), 4.47 (d, J = 1.5 Hz, 2H), 4.27 (q, J = 7.1 Hz, 2H), 3.51 (t, J = 8.0 Hz, 2H), 2.92 (t, J = 8.0 Hz, 2H), 1.35 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.5, 150.7, 143.7, 138.7, 133.4, 130.4, 127.4, 125.7, 125.6, 123.2, 119.8, 112.6, 61.2, 51.1, 14.3 (1 aromatic carbon unresolved); MS (EI): m/z 324 [M]+; Anal. Calcd for C18H16N2O4: C, 66.66; H, 4.97; N, 8.64. Found: C, 66.59; H, 4.94; N, 8.69.
3.3.7. 1-Methyl-6-nitro-1,2-dihydroquinoline-3-carbonitrile (10a) from 2 and 8a
Yield: 189 mg (88%) as an orange solid, m.p. 186–187 °C; IR: 2210, 1640, 1515, 1321 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.05 (dd, J = 9.2, 2.7 Hz, 1H), 7.82 (d, J = 2.7 Hz, 1H), 7.00 (br s, 1H), 6.47 (d, J = 9.2 Hz, 1H), 4.41 (d, J = 1.6 Hz, 2H), 2.94 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 150.5, 139.0, 137.7, 128.7, 125.0, 117.5, 116.5, 109.6, 103.7, 51.5, 37.6; MS (EI): m/z 215 [M]+·. Anal. Calcd for C11H9N3O2: C, 61.39; H, 4.22; N, 19.53. Found: C, 61.41; H, 4.25; N, 19.44.
3.3.8. 1-Hexyl-6-nitro-1,2-dihydroquinoline-3-carbonitrile (10b) from 2 and 8b
Yield: 222 mg (78%) as a yellow solid, m.p. 110–111 °C; IR: 2228, 1647, 1532, 1344 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.02 (dd, J = 9.3, 2.7 Hz, 1H), 7.80 (d, J = 2.7 Hz, 1H), 6.97 (br s, 1H), 6.44 (d, J = 9.3 Hz, 1H), 4.43 (s, 2H), 3.25 (t, J = 7.2 Hz, 2H), 1.62 (quintet, J = 7.5 Hz, 2H), 1.36 (m, 6H), 0.92 (t, J = 6.8 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 149.8, 139.2, 137.6, 128.9, 125.5, 117.4, 116.7, 109.6, 103.3, 50.9, 50.3, 31.5, 26.6, 25.0, 22.6, 14.0; MS (EI): m/z 285 [M]+·; Anal. Calcd for C16H19N3O2: C, 67.35; H, 6.71; N, 14.73. Found: C, 67.46; H, 6.76; N, 14.63.
3.3.9. 1-Isobutyl-6-nitro-1,2-dihydroquinoline-3-carbonitrile (10c) from 2 and 8c
Yield: 203 mg (79%) as a yellow solid, m.p. 158–159 oC; IR: 2208, 1642, 1519, 1324 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.00 (d, J = 9.3 Hz, 1H), 7.80 (s, 1H), 6.96 (s, 1H), 6.47 (d, J = 9.3 Hz, 1H), 4.44 (s, 2H), 3.08 (d, J = 7.5 Hz, 2H), 2.11 (septet, J = 6.7 Hz, 1H), 1.02 (d, J = 6.7 Hz, 6H); 13C NMR (101 MHz, CDCl3): δ 150.3, 139.3, 137.6, 128.7, 125.6, 117.4, 116.7, 110.2, 103.1, 58.4, 51.3, 26.2, 20.2, 14.3; MS (EI): m/z 257 [M]+·; Anal. Calcd for C14H15N3O2: C, 65.36; H, 5.88; N, 16.33. Found: C, 65.41; H, 5.91; N, 16.25.
3.3.10. 1-Benzyl-6-nitro-1,2-dihydroquinoline-3-carbonitrile (10d) from 2 and 8d
Yield: 233 mg (80%) as a yellow solid, m.p. 154–155 °C; IR: 2231, 1648, 1530, 1345 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.99 (dd, J = 9.2, 2.6 Hz, 1H), 7.86 (d, J = 2.6 Hz, 1H), 7.43–7.32 (complex, 3H), 7.23 (d, J = 8.5 Hz, 2H), 7.04 (s, 1H), 6.54 (d, J = 9.2 Hz, 1H), 4.52 (s, 2H), 4.44 (d, J = 1.6 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 150.1, 139.0, 138.3, 133.8, 129.3, 128.9, 128.3, 126.8, 125.5, 117.6, 116.5, 110.4, 103.9, 54.1, 50.4; MS (EI): m/z 291 [M]+·; Anal. Calcd for C17H13N3O2: C, 70.09; H, 4.50; N, 14.42. Found: C, 70.04; H, 4.53; N, 14.38.
3.3.11. 6-Nitro-1-phenethyl-1,2-dihydroquinoline-3-carbonitrile (10e) from 2 and 8e
Yield: 250 mg (82%) as a yellow solid, m.p. 121–122 °C; IR: 2239, 1623, 1568, 1344 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.02 (dd, J = 9.2, 2.7 Hz, 1H), 7.81 (d, J = 2.7 Hz, 1H), 7.38–7.18 (complex, 5H), 6.95 (s, 1H), 6.47 (d, J = 9.3 Hz, 1H), 4.28 (s, 2H), 3.52 (t, J = 7.4 Hz, 2H), 2.92 (t, J = 7.4 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 149.5, 139.0, 137.9, 137.5, 129.1, 128.9, 128.7, 127.2, 125.6, 117.5, 116.5, 109.6, 103.4, 52.6, 50.7, 31.8; MS (EI): m/z 305 [M]+·; Anal. Calcd for C18H15N3O2: C, 70.81; H, 4.95; N, 13.76. Found: C, 70.76; H, 4.89; N, 13.68.
3.3.12. 6-Nitro-1-phenyl-1,2-dihydroquinoline-3-carbonitrile (10f) from 2 and 8f
Yield: 224 mg (81%) as a yellow solid, m.p. 161–163 °C; IR: 2210, 1633, 1570, 1326 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.94 (d, J = 2.7 Hz, 1H), 7.88 (dd, J = 9.3, 2.7 Hz, 1H), 7.52 (t, J = 7.5 Hz, 2H), 7.39 (t, J = 7.5 Hz, 1H), 7.28 (d, J = 7.5 Hz, 2H), 7.14 (s, 1H), 6.41 (d, J = 9.3 Hz, 1H), 4.69 (d, J = 1.5 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 149.9, 143.0, 139.1, 138.7, 130.6, 127.9, 127.8, 125.5, 125.2, 118.8, 116.5, 113.5, 103.9, 51.4; MS (EI): m/z 277 [M]+·; Anal. Calcd for C16H11N3O2: C, 69.31; H, 4.00; N, 15.15. Found: C, 69.37; H, 3.98; N, 15.03.
3.3.13. Ethyl-6-cyano-1-methyl-1,2-dihydroquinoline-3-carboxylate (11a) from 3 and 8a
Yield: 209 mg (91%) as a yellow solid, m.p. 140–141 °C; IR: 2201, 1708, 1652 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.35 (dd, J = 8.6, 2.1 Hz, 1H), 7.23 (s, 1H), 7.17 (d, J = 2.1 Hz, 1H), 6.42 (d, J = 8.6 Hz, 1H), 4.41 (d, J = 1.5 Hz, 2H), 4.27 (q, J = 7.1 Hz, 2H), 2.87 (s, 3H), 1.34 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.8, 149.6, 135.9, 133.7, 133.0, 122.7, 119.9, 119.8, 109.9, 98.6, 61.0, 51.0, 37.4, 14.3; MS (EI): m/z 242 [M]+·; Anal. Calcd for C14H14N2O2: C, 69.41; H, 5.82; N, 11.56. Found: C, 69.36; H, 5.78; N, 11.43.
3.3.14. Ethyl-6-cyano-1-hexyl-1,2-dihydroquinoline-3-carboxylate (11b) from 3 and 8b
Yield: 256 mg (82%) as a yellow semisolid; IR: 2216, 1705, 1651 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.31 (dd, J = 8.8, 2.1 Hz, 1H), 7.20 (s, 1H), 7.15 (d, J = 2.1 Hz, 1H), 6.40 (d, J = 8.8 Hz, 1H), 4.44 (d, J = 1.5 Hz, 2H), 4.26 (q, J = 7.1 Hz, 2H), 3.19 (t, J = 7.8 Hz, 2H), 1.56 (m, 2H), 1.34 (superimposed m, 6H and t, J = 7.1 Hz, 3H), 0.91 (t, J = 6.7 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.9, 148.8, 135.8, 133.8, 133.4, 122.0, 120.0, 119.5, 109.8, 97.7, 61.0, 50.4, 49.5, 31.6, 26.6, 24.8, 22.6, 14.3, 14.0; MS (EI): m/z 312 [M]+·; Anal. Calcd for C19H24N2O2: C, 73.05; H, 7.74; N, 8.97. Found: C, 73.14; H, 7,79; N, 8.89.
3.3.15. Ethyl-6-cyano-1-isobutyl-1,2-dihydroquinoline-3-carboxylate (11c) from 3 and 8c
Yield: 241 mg (85%) as a yellow solid, m.p. 117–118 °C; IR: 2214, 1690, 1639, 1603, 1515 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.29 (dd, J = 8.8 Hz, 1H), 7.20 (s, 1H), 7.16 (d, J = 2.0 Hz, 1H), 6.42 (d, J = 8.8 Hz, 1H), 4.46 (d, J = 1.5 Hz, 2H), 4.26 (q, J = 7.1 Hz, 2H), 3.03 (d, J = 7.5 Hz, 2H), 2.10 (nonet, J = 6.7 Hz, 1H); 1.34 (t, J = 7.1 Hz, 3H), 0.98 (d, J = 6.7 Hz, 6H); 13C NMR (101 MHz, CDCl3): δ 164.9, 149.3, 135.6, 133.9, 133.5, 121.8, 119.9, 119.5, 110.3, 97.7, 61.0, 58.0, 50.5, 26.0, 20.2, 14.3; MS (EI): m/z 284 [M]+·; Anal. Calcd for C17H20N2O2: C, 71.81; H, 7.09; N, 9.85. Found: C, 71.85; H, 7.12; N, 9.79.
3.3.16. Ethyl-1-benzyl-6-cyano-1,2-dihydroquinoline-3-carboxylate (11d) from 3 and 8d
Yield: 256 mg (79%) as a yellow solid, m.p. 97–98 °C; IR: 2219, 1703, 1642, 1602, 1517 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.36 (t, J = 7.5 Hz, 2H), 7.33–7.20 (complex, 6H), 6.44 (d, J = 8.6 Hz, 1H), 4.51 (d, J = 1.5 Hz, 2H), 4.48 (s, 2H), 4.25 (q, J = 7.1 Hz, 2H), 1.32 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.7, 149.0, 137.9, 134.9, 133.6, 133.3, 129.1, 127.7, 126.7, 122.4, 119.7, 119.6, 110.5, 98.7, 61.0, 53.8, 50.1, 14.3; MS (EI): m/z 318 [M]+·; Anal. Calcd for C20H18N2O2: C, 75.45; H, 5.70; N, 8.80. Found: C, 75.38; H, 5.68; N, 8.67.
3.3.17. Ethyl-6-cyano-1-phenethyl-1,2-dihydroquinoline-3-carboxylate (11e) from 3 and 8e
Yield: 270 mg (85%) as a yellow solid, m.p. 131–132 °C; IR: 2217, 1701, 1650 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.36–7.29 (complex, 3H), 7.25–7.18 (complex, 4H), 7.18 (d, J = 2.1 Hz, 1H), 6.44 (d, J = 8.7 Hz, 1H), 4.43 (d, J = 1.6 Hz, 2H), 4.26 (q, J = 7.1 Hz, 2H), 3.45 (t, J = 7.9 Hz, 2H), 2.88 (t, J = 7.9 Hz, 2H), 1.33 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.8, 148.5, 138.1, 135.8, 132.7, 133.4, 128.9, 128.7, 126.8, 122.1, 119.9, 119.7, 109.8, 98.2, 61.0, 52.1, 49.6, 31.3, 14.3; MS (EI): m/z 332 [M]+·; Anal. Calcd for C21H20N2O2: C, 75.88; H, 6.06; N, 8.43. Found: C, 75.81; H, 6.05; N, 8.35.
3.3.18. Ethyl-6-cyano-1-phenyl-1,2-dihydroquinoline-3-carboxylate (11f) from 3 and 8f
Yield: 252 mg (83%) as a yellow solid, m.p. 129–130 °C; IR: 2254, 1698, 1644 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.47 (t, J = 7.8 Hz, 2H), 7.36 (s, 1H), 7.33–7.24 (complex, 4H), 7.19 (dd, J = 8.7, 2.0 Hz, 1H), 6.44 (d, J = 8.7 Hz, 1H), 4.71 (d, J = 1.5 Hz, 2H), 4.27 (q, J = 7.1 Hz, 2H), 1.33 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.6, 148.8, 143.9, 134.8, 133.22, 133.16, 130.2, 126.8, 125.4, 123.1, 121.1, 119.6, 113.8, 100.3, 61.1, 50.4, 14.3; MS (EI): m/z 304 [M]+·; Anal. Calcd for C19H16N2O2: C, 74.98; H, 5.30; N, 9.20. Found: C, 75.07; H, 5.31; N, 9.08.
3.3.19. 1-Methyl-1,2-dihydroquinoline-3,6-dicarbonitrile (12a) from 4 and 8a
Yield: 160 mg (82%) as a yellow solid, m.p. 205–206 °C; IR: 2253, 1625 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.39 (d, J = 8.6 Hz, 1H), 7.15 (s, 1H), 6.94 (s, 1H), 6.48 (d, J = 8.6 Hz, 1H), 4.33 (s, 2H), 2.87 (s, 3H); 13C NMR (101 MHz, CDCl3): δ 148.9, 139.0, 136.4, 132.7, 119.2, 119.0, 116.8, 110.8, 103.7, 99.8, 51.4, 37.4; MS (EI): m/z 195 [M]+·; Anal. Calcd for C12H9N3: C, 73.83; H, 4.65; N, 21.52. Found: C, 73.79; H, 4.63; N, 21.37.
3.3.20. 1-Hexyl-1,2-dihydroquinoline-3,6-dicarbonitrile (12b) from 4 and 8b
Yield: 204 mg (77%) as a yellow solid, m.p. 178–179 °C; IR: 2254, 1651 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.76 (s, 1H), 8.09 (s, 1H), 7.96 (d, J = 8.9 Hz, 1H), 7.60 (d, J = 8.9 Hz, 1H), 4.22 (t, J = 7.4 Hz, 2H), 1.91 (quintet, J = 7.4 Hz, 2H), 1.50–1.28 (complex, 8H), 0.91 (t, J = 6.7 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 172.8, 149.5, 141.2, 135.5, 132.9, 127.0, 117.5, 117.3, 114.5, 109.7, 97.9, 54.8, 31.2, 28.8, 26.3, 22.4, 13.9; MS (EI): m/z 265 [M]+·; Anal. Calcd for C17H19N3: C, 76.95; H, 7.22; N, 15.84. Found: C, 77.03; H, 7.18; N, 15.69.
3.3.21. 1-Isobutyl-1,2-dihydroquinoline-3,6-dicarbonitrile (12c) from 4 and 8c
Yield 192 mg (81%) as a yellow solid, m.p. 131–132 °C; IR: 2229, 1626 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.33 (dd, J = 8.8, 2.1 Hz, 1H), 7.14 (d, J = 2.1 Hz, 1H), 6.92 (s, 1H), 6.48 (d, J = 8.8 Hz, 1H), 4.38 (d, J = 1.5 Hz, 2H), 3.02 (d, J = 7.5 Hz, 2H), 2.06 (septet, J = 6.6 Hz, 1H), 1.00 (d, J = 6.6 Hz, 6H); 13C NMR (101 MHz, CDCl3): δ 148.6, 139.1, 136.1, 133.2, 119.3, 118.7, 116.9, 111.2, 102.7, 98.9, 58.1, 50.9, 26.2, 20.2; MS (EI): m/z 237 [M]+·; Anal. Calcd for C15H15N3: C, 75.92; H, 6.37; N, 17.71. Found: C, 75.97; H, 6.41; N, 17.60.
3.3.22. 1-Benzyl-1,2-dihydroquinoline-3,6-dicarbonitrile (12d) from 4 and 8d
Yield: 231 mg (86%) as a yellow solid, m.p. 140–141 °C; IR: 2215, 1639, 1603, 1514 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.42–7.30 (complex, 4H), 7.23 (d, J = 7.0 Hz, 2H), 7.20(d, J = 2.0 Hz, 1H), 6.97 (s, 1H), 6.54 (d, J = 8.8 Hz, 1H), 4.46 (s, 2H), 4.38 (d, J = 1.5 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 148.4, 138.8, 136.4, 134.2, 133.0, 129.2, 128.1, 126.8, 119.1, 118.7, 116.8, 111.4, 103.6, 99.9, 53.8, 50.1; MS (EI): m/z 271 [M]+·; Anal. Calcd for C18H13N3: C, 79.68; H, 4.83; N, 15.49. Found: C, 79.73; H, 4.84; N, 15.41.
3.3.23. 1-Phenethyl-1,2-dihydroquinoline-3,6-dicarbonitrile (12e) from 4 and 8e
Yield: 251 mg (88%) as a yellow solid, m.p. 129–130 °C; IR: 2215, 1639, 1603, 1514 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.37–7.27 (complex, 4H), 7.21 (d, J = 8.3 Hz, 2H), 7.15 (d, J = 2.0 Hz, 1H), 6.89 (t, J = 1.6 Hz, 1H), 6.50 (d, J = 9.0 Hz, 1H), 4.25 (d, J = 1.6 Hz, 2H), 3.46 (t, J = 7.4 Hz, 2H), 2.89 (t, J = 7.4 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 147.8, 138.8, 137.7, 136.3, 133.2, 129.0, 128.7, 127.1, 119.2, 118.8, 116.8, 110.7, 103.0, 99.3, 52.2, 50.3, 31.6; MS (EI): m/z 285 [M]+·; Anal. Calcd for C19H15N3: C, 79.98; H, 5.30; N, 14.73. Found: C, 79.88; H, 5.26; N, 14.61.
3.3.24. Ethyl-1-methyl-1,2-dihydro-1,8-naphthyridine-3-carboxylate (13a) from 5 and 8a
Yield: 191 mg (88%) as a yellow solid, m.p. 65–66 °C; IR: 1701, 1646, 1556, 1395, 1222 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.95 (dd, J = 5.1, 1.8 Hz, 1H), 7.23 (t, J = 1.2 Hz, 1H) 7.10 (dd, J = 7.2, 1.8 Hz, 1H) 6.41 (dd, J = 7.2, 5.1 Hz, 1H) 4.48 (d, J = 1.2 Hz, 2H), 4.26 (q, J = 7.1 Hz, 2H), 3.00 (s, 3H), 1.33 (t, J = 7.2 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 165.1, 156.7, 150.1, 135.9, 134.5, 122.1, 114.6, 112.5, 60.8, 51.1, 35.2, 14.3; MS (EI): m/z 218 [M]+·; Anal. Calcd for C12H14N2O2: C, 66.04; H, 6.47; N, 12.84. Found: C, 65.95; H, 6.43; N, 12.87.
3.3.25. Ethyl-1-hexyl-1,2-dihydro-1,8-naphthyridine-3-carboxylate (13b) from 5 and 8b
Yield: 230 mg (80%) as a yellow oil; IR: 1703, 1647 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.92 (dd, J = 5.1, 1.9 Hz, 1H), 7.19 (t, J = 1.4 Hz, 1H), 7.06 (dd, J = 7.2, 1.9 Hz, 1H), 6.35 (dd, J = 7.2, 5.1 Hz, 1H), 4.51 (d, J = 1.4 Hz, 2H), 4.25 (q, J = 7.1 Hz, 2H), 3.46 (t, J = 7.6 Hz, 2H), 1.60 (quintet, J = 7.6 Hz, 2H), 1.33 (overlapping t, J = 7.1 Hz, 3H and m, 6 H), 0.89 (t, J = 6.8 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 165.2, 156.3, 150.2, 136.0, 134.6, 121.6, 114.2, 112.0, 60.7, 48.9, 47.5, 31.8, 26.7, 25.6, 22.7, 14.3, 14.1; MS (EI): m/z 288 [M]+·; Anal. Calcd for C17H24N2O2: C, 70.80; H, 8.39; N, 9.71. Found: C, 70.92; H, 8.34; N, 9.66.
3.3.26. Ethyl-1-isobutyl-1,2-dihydro-1,8-naphthyridine-3-carboxylate (13c) from 5 and 8c
Yield: 208 mg (80%) as a yellow oil; IR: 1702, 1648 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.91 (dd, J = 5.0, 1.9 Hz, 1H), 7.20 (t, J = 1.5 Hz, 1H), 7.07 (dd, J = 7.2, 1.9 Hz, 1H), 6.36 (dd, J = 7.2, 5.0 Hz, 1H), 4.50 (d, J = 1.5 Hz, 2H), 4.25 (q, J = 7.2 Hz, 2H), 3.34 (d, J = 7.6 Hz, 2H), 2.13 (nonet, J = 6.9 Hz, 1H), 1.33 (t, J = 7.2 Hz, 3H), 0.95 (d, J = 6.7 Hz, 6H); 13C NMR (101 MHz, CDCl3): δ 165.2, 156.7, 150.1, 136.1, 134.7, 121.5, 114.1, 112.0, 60.7, 54.5, 49.5, 25.7, 20.1, 14.3; MS (EI): m/z 260 [M]+·; Anal. Calcd for C15H20N2O2: C, 69.20; H, 7.74; N, 10.76. Found: C, 69.29; H, 7.70; N, 10.63.
3.3.27. Ethyl-1-benzyl-1,2-dihydro-1,8-naphthyridine-3-carboxylate (13d) from 5 and 8d
Yield: 238 mg (81%) as a yellow oil; IR: 1702, 1651 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.96 (dd, J = 5.0, 1.9 Hz, 1H), 7.35–7.26 (complex, 4H), 7.25 (m, 1H), 7.22 (s, 1H), 7.13 (dd, J = 7.2, 1.9 Hz, 1H), 6.44 (dd, J = 7.2, 5.0 Hz, 1H), 4.78 (s, 2H), 4.42 (d, J = 1.5 Hz, 2H), 4.21 (q, J = 7.1 Hz, 2H), 1.29 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 165.1, 156.2, 150.2,. 137.1, 136.3, 134.3, 128.6, 128.0, 127.2, 122.0, 114.0, 112.7, 60.8, 50.4, 48.6, 14.3; MS (EI): m/z 294 [M]+·; Anal. Calcd for C18H18N2O2: C, 73.45: H, 6.16; N, 9.52. Found: C, 73.51; H, 6.11; N, 9.45.
3.3.28. Ethyl-1-phenethyl-1,2-dihydro-1,8-naphthyridine-3-carboxylate (13e) from 5 and 8e
Yield: 256 mg (83%) as a yellow oil; IR: 1702, 1647 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.97 (dd, J = 5.1, 1.9 Hz, 1H), 7.30 (apparent s, 2H), 7.29 (apparent s, 2H), 7.19 (superimposed m, 1H and s, 1H), 7.08 (dd, J = 7.2, 1.9 Hz, 1H), 6.40 (dd, J = 7.2, 5.1 Hz, 1H), 4.48 (d, J = 1.5 Hz, 2H), 4.24 (q, J = 7.1 Hz, 2H), 3.70 (t, J = 7.7 Hz, 2H), 2.92 (t, J = 7.7 Hz, 2H), 1.32 (t, J = 7.1 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 165.1, 156.0, 150.3, 139.6, 136.0, 134.5, 128.9, 128.4, 126.2, 121.7, 114.3, 112.3, 60.8, 49.5, 49.3, 32.1, 14.3; MS (EI): m/z 308 [M]+·; Anal. Calcd for C19H20N2O2: C, 74.00: H, 6.54; N, 9.08. Found: C, 73.88; H, 6.51; N, 8.96.
3.3.29. Ethyl-1-phenyl-1,2-dihydro-1,8-naphthyridine-3-carboxylate (13f) from 5 and 8f
Yield: 215 mg (77%) as a yellow oil; IR: 1703, 1647 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.95 (dd, J = 5.0, 1.8 Hz, 1H), 7.42 (t, J = 7.8 Hz, 2H), 7.38–7.32 (complex, 3H), 7.28 (dd, J = 7.3, 1.8 Hz, 1H), 7.21 (t, J = 7.2 Hz, 1H), 6.59 (dd, J = 7.3, 5.0 Hz, 1H), 4.84 (d, J = 1.4 Hz, 2H), 4.26 (q, J = 7.2 Hz, 2H), 1.32 (t, J = 7.2 Hz, 3H); 13C NMR (101 MHz, CDCl3): δ 164.8, 155.8, 150.0, 144.1, 136.5, 133.8, 129.1, 125.5, 125.0, 123.0, 116.1, 114.7, 60.9, 50.5, 14.3; MS (EI): m/z 280 [M]+·; Anal. Calcd for C17H16N2O2: C, 72.84: H, 5.75: N, 9.99. Found: C, 72.71; H, 5.68; N, 9.92.
3.3.30. 1-Hexyl-1,2-dihydro-1,8-naphthyridine-3-carbonitrile (14b) from 6 and 8b
Yield: 198 mg (0.82 mmol, 82%) as a yellow semisolid; IR: 2254, 1639 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.97 (dd, J = 5.0, 1.8 Hz, 1H), 7.04 (dd, J = 7.3, 1.8 Hz, 1H), 6.87 (t, J = 1.4 Hz, 1H), 6.42 (dd, J = 7.3, 5.0 Hz, 1H), 4.44 (d, J = 1.4 Hz, 2H), 3.45 (t, J = 7.4 Hz, 2H), 1.58 (quintet, J = 7.2 Hz, 2H), 1.38–1.28 (complex, 6H), 0.90 (t, J = 6.9 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 155.5, 151.0, 139.8, 135.9, 117.5, 113.4, 112.7, 102.4, 49.6, 47.4, 31.7, 26.6, 25.7, 22.6, 14.1; MS (EI): m/z 241 [M]+·; Anal. Calcd for C15H19N3: C, 74.65; H, 7.94; N, 17.41. Found: C, 74.77; H, 7.98; N, 17.29.
3.3.31. 1-Benzyl-1,2-dihydro-1,8-naphthyridine-3-carbonitrile (14d) from 6 and 8d
Yield: 14 mg (0.057 mmol, 5.7%) as a yellow oil; IR: 2209, 1638 cm−1: 1H NMR (400 MHz, CDCl3): δ 8.02 (dd, J = 5.0, 1.9 Hz, 1H), 7.37–7.28 (complex, 5H), 7.11 (dd, J = 7.3, 1.9 Hz, 1H), 6.90 (t, J = 1.5 Hz, 1H), 6.51 (dd, J = 7.3, 5.0 Hz, 1H), 4.76 (s, 2H), 4.31 (d, J = 1.5 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 155.4, 150.9, 139.4, 136.3, 136.1, 128.7, 128.2, 127.6, 117.3, 113.4, 113.2, 102.9, 50.2, 49.8; MS (EI): m/z 247 [M]+·; Anal. Calcd for C16H13N3: C, 77.71; H, 5.30; N, 16.99. Found: C, 77.67; H, 5.24; N, 16.87.
3.3.32. 1-Benzyl-1,4-dihydro-1,8-naphthyridine-3-carbonitrile (15d) from 6 and 8d
Double bond-migrated product; Yield: 203 mg (0.82 mmol, 82%) as a white powder (slowly darkens on exposure to air and light), m.p. 117–118 °C; IR: 2000, 1651 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.09 (d, J = 4.9 Hz, 1H), 7.37–7.25 (complex, 6H), 6.87 (dd, J = 7.4, 4.9 Hz, 1H), 6.84 (s, 1H), 4.99 (s, 2H), 3.81 (s, 2H); 13C NMR (101 MHz, CDCl3): δ 149.6, 146.4, 144.0, 137.3, 137.1, 128.8, 127.7, 127.6, 120.3, 119.2, 115.3, 78.8, 51.0, 27.9; MS (EI): m/z 247 [M]+·; Anal. Calcd for C16H13N3: C, 77.71; H, 5.30; N, 16.99. Found: C,77.69; H, 5.29; N, 16.94. The X-ray structure for 15d (CCDC 2035027) and the thermal ellipsoid plot appears in Figure 3 and the SI.
3.3.33. 1-Phenethyl-1,2-dihydro-1,8-naphthyridine-3-carbonitrile (14e) from 6 and 8e
Yield: 209 mg (0.80 mmol, 80%) as a yellow semisolid; IR: 2254, 1639 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.02 (dd, J = 5.0, 1.9 Hz, 1H), 7.35–7.21 (complex, 5H), 7.06 (dd, J = 7.3, 1.9 Hz, 1H), 6.86 (t, J = 1.4 Hz, 1H), 6.46 (dd, J = 7.3, 5.0 Hz, 1H), 4.32 (d, J = 1.4 Hz, 2H), 3.68 (t, J = 7.4 Hz, 2H), 2.92 (t, J = 7.1 Hz, 2H); 13C NMR (100 MHz, CDCl3): δ 155.2, 151.1, 139.6, 135.8, 128.9, 128.6, 126.5, 117.3, 113.5, 113.0, 102.5, 50.3, 49.6, 32.4; MS (EI): m/z 261 [M]+·; Anal. Calcd for C17H15N3: C, 78.13; H, 5.79; N, 16.08. Found: C, 78.06; H, 5.77; N, 16.01.
3.3.34. 1-Phenyl-1,4-dihydro-1,8-naphthyridine-3-carbonitrile (15f) from 6 and 8f
Double bond-migrated product; Yield: 168 mg (0.72 mmol, 72%) as a white powder (slowly darkens on exposure to air and light), m.p. 162–163 °C; IR: 2193, 1645 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.15 (dd, J = 5.0, 1.6 Hz, 1H), 7.82 (ddd, J = 9.5, 7.3, 1.9 Hz, 1H), 7.39–7.30 (complex, 3H), 7.23 (ddd, J = 7.3, 5.0, 1.6 Hz, 1H), 7.06 (t, J = 7.4 Hz, 1H), 6.93 (d, J = 7.4 Hz, 2H), 3.53 (s, 2H); 13C NMR (101 MHz, CDCl3): δ 146.5, 146.3, 141.1, 140.9, 139.9, 129.9, 123.4, 122.3, 121.8, 119.4, 115.6, 80.5, 25.7; MS (EI): m/z 233 [M]+·; Anal. Calcd for C15H11N3: C, 77.23; H, 4.75; N, 18.01. Found: C, 77.12; H, 4.69; N, 17.93.
3.4. Control Experiment Attempted Substitution of Benzylamine (8d) on 2-fluoropyridine and other SNAr Activated Rings
A 50 mL round-bottomed flask was charged with 2-fluoropyridine (97 mg, 1.0 mmol) and benzylamine (8d, 164 mg, 1.5 mmol) in DMF (2 mL) under N2. The mixture was stirred for 1 h at 23 °C and then for 12 h at 50 °C. Analysis of the mixture by TLC indicated that no substitution had occurred. Substitution was noted under these conditions, however, for fluoroaromatic rings activated by a C4 NO2 (complete conversion) and CN (modest conversion).
3.5. Control Experiment Capture of Intermediate (Z)-2-((benzylamino)methyl)-3-(2-fluoro-5-nitrophenyl)acrylonitrile (16)
A 50 mL, round-bottomed flask equipped with a condenser and a stir bar was charged with 2-cyano-1-(2-fluoro-5-nitrophenyl)allyl acetate (2, 264 mg, 1.0 mmol) in DMF (2 mL) under N2. Benzylamine (8d, 164 mg, 1.5 mmol) was added at 23 °C with continued stirring. After 30 min, the solution was poured into de-ionized water (15 mL), and the mixture was extracted with EtOAc (3 × 15 mL). The combined organic layers were washed with saturated aq. NaCl and dried (Na2SO4). Removal of the solvent under vacuum gave the crude product, which was purified by silica gel column chromatography (10% EtOAc in hexane) to afford 10d (band 1, 46 mg, 11%) as a yellow solid and (Z)-2-((benzylamino)methyl)-3-(2-fluoro-5-nitrophenyl)acrylonitrile (16, band 2, 236 mg, 76%) as a light yellow solid, m.p. 51–52 °C; IR: 3344, 2216, 1623, 1530, 1351 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.92 (dd, J = 6.3, 2.8 Hz, 1H), 8.30 (ddd, J = 9.1, 4.5, 2.8 Hz, 1H), 7.37–7.34 (complex, 4H), 7.33 (br s, 1H), 7.29 (t, J = 9.1 Hz, 1H), 7.29 (obscured, 1H), 3.87 (s, 2H), 3.65 (d, J = 1.6 Hz, 2H), 1.70 (br s, 1H); 13C NMR (101 MHz, CDCl3): δ 163.3 (d, J = 262.1 Hz), 144.4, 139.1, 133.1 (d, J = 5.1 Hz), 128.6, 128.2, 127.5, 127.0 (d, J = 10.5 Hz), 124.7 (d, J = 4.1 Hz), 122.9 (d, J = 14.7 Hz), 117.2, 117.1 (d, J = 21.2 Hz), 116.8 (d, J = 10.0 Hz), 52.5, 52.3; MS: m/z 311 [M]+·. Anal. Calcd for C17H14FN3O2: C, 65.59; H, 4.53; N, 13.50. Found: C, 65.51; H, 4.54; N, 13.38. The X-ray structure of 16 (CCDC 2035028) was obtained and the thermal ellipsoid plot is shown in Figure 4 and the SI. Further heating of 16 (200 mg, 0.64 mmol) at 50 °C in DMF (2 mL) for 1 h afforded 10d (112 mg, 60% unoptimized).
3.6. Representative Procedure for Preparation of Fluorodihydroquinolines Using MBH Acetates and Primary Amines
A 50 mL, round-bottomed flask equipped with a condenser, stir bar and vacuum adapter, was charged with MBH acetate 7 (1 mmol) in DMF (2 mL) under N2. The amine (1.5 mmol) was added in DMF (2 mL). The reaction was stirred at 23 °C for 1 h and gradually increased to 90 °C for 6 h. After TLC analysis (30% EtOAc/hexane) indicated completion of reaction, the solution was poured into de-ionized water (15 mL), and the mixture was extracted with EtOAc (3 × 10 mL). The combined organic layers were washed with saturated NaCl and dried (Na2SO4). Removal of the solvent under vacuum gave the crude product, which was purified by silica gel column chromatography (10% EtOAc in hexane) to afford the pure fluorodihydroquinoline analogs.
3.6.1. Ethyl-6-fluoro-1-methyl-1,2-dihydroquinoline-3-carboxylate (18a) from 7 and 8a
Yield: 214 mg (0.91 mmol, 91%) as a light yellow solid, m.p. 61–62 °C; IR: 1690, 1639, 1236 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.21 (s, 1H), 6.84–6.75 (complex, 2H), 6.66 (dd, J = 8.8, 4.7 Hz, 1H), 4.19 (q, J = 7.1 Hz, 2H), 3.75 (s, 2H), 3.20 (s, 3H), 1.29 (t, J = 7.1 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 167.7, 158.8 (d, J = 242.0 Hz), 142.9, 135.3 (d, J = 2.3 Hz), 125.3 (d, J = 7.4 Hz), 116.2 (d, J = 22.5 Hz), 113.5 (d, J = 8.1 Hz), 113.3 (d, J = 22.4 Hz), 96.1, 59.6, 39.1, 26.6, 14.6; MS (EI): m/z 235 [M]+·; Anal. Calcd for C13H14FNO2: C, 66.37; H, 6.00; N, 5.95. Found: C, 66.40; H, 5.96; N, 5.87.
3.6.2. Ethyl-6-fluoro-1-hexyl-1,2-dihydroquinoline-3-carboxylate (18b) from 7 and 8b
Yield: 244 mg (0.80 mmol, 80%) as a yellow oil; IR: 1688, 1637, 1237 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.22 (s, 1H), 6.68 (apparent d, J = 8.1 Hz, 2H), 6.67 (dd, J = 8.9, 4.8 Hz, 1H), 4.20 (q, J = 7.1 Hz, 2H), 3.75 (s, 2H), 3.51 (t, J = 7.4 Hz, 2H), 1.66 (quintet, J = 7.4 Hz, 2H), 1.39–1.26 (complex, 6H), 1.30 (t, J = 7.1 Hz, 3H), 0.89 (t, J = 6.6 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 167.8, 158.6 (d, J = 242.0 Hz), 142.3, 134.1 (d, J = 2.5 Hz), 125.6 (d, J = 7.2 Hz), 116.5 (d, J = 22.3 Hz), 113.8 (d, J = 8.1 Hz), 113.3 (d, J = 22.4 Hz), 95.5, 59.6, 51.6, 31.5, 27.8, 26.7, 26.4, 22.6, 14.6, 14.0; MS (EI): m/z 305 [M]+·; Anal. Calcd for C18H24FNO2: C, 70.79; H, 7.92; N, 4.59. Found: C, 70.72; H, 7.95; N, 4.49.
3.6.3. Ethyl-6-fluoro-1-isobutyl-1,2-dihydroquinoline-3-carboxylate (18c) from 7 and 8c
Yield: 244 mg (0.88 mmol, 88%) as a yellow oil; IR: 1688, 1639, 1234 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.19 (s, 1H), 6.82–6.74 (complex, 2H), 6.65 (dd, J = 9.0, 4.7 Hz, 1H), 4.19 (q, J = 7.1 Hz, 2H), 3.75 (s, 2H), 3.30 (d, J = 7.3 Hz, 2H), 2.07 (nonet, J = 6.7 Hz, 1H), 1.29 (t, J = 7.1 Hz, 3H), 0.96 (d, J = 7.0 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 167.8, 158.6 (d, J = 242.1 Hz), 142.9, 134.3 (d, J = 2.5 Hz), 125.5 (d, J = 7.2 Hz), 116.4 (d, J = 22.1 Hz), 114.1 (d, J = 8.0 Hz), 113.3 (d, J = 22.4 Hz), 95.4, 59.6, 59.3, 26.7, 26.6, 20.0, 14.6; MS (EI): m/z 277 [M]+·; Anal. Calcd for C16H20FNO2: C, 69.29; H, 7.27; N, 5.05. Found: C, 69.18; H, 7.29; N, 4.98.
3.6.4. Ethyl-1-benzyl-6-fluoro-1,2-dihydroquinoline-3-carboxylate (18d) from 7 and 8d
Yield: 261 mg (0.84 mmol, 84%) as a light yellow solid, m.p. 57–58 °C; IR: 1686, 1643, 1242 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.37 (s, 1H), 7.37–7.32 (complex, 2H), 7.30–7.22 (complex, 3H), 6.79 (dd, J = 8.8, 2.9 Hz, 1H), 6.64 (td, J = 8.4, 2.9 Hz, 1H), 6.52 (dd, J = 8.9, 4.7 Hz, 1H), 4.76 (s, 2H), 4.21 (q, J = 7.1 Hz, 2H), 3.83 (s, 2H), 1.30 (t, J = 7.1 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 167.7, 158.7 (d, J = 242.3 Hz), 142.8, 136.4, 134.2 (d, J = 2.4 Hz), 129.0, 127.7, 126.1, 125.4 (d, J = 7.4 Hz), 116.4 (d, J = 22.4 Hz), 114.9 (d, J = 8.2 Hz), 113.4 (d, J = 22.4 Hz), 96.9, 59.7, 55.3, 26.7, 14.6; MS (EI): m/z 311 [M]+·; Anal. Calcd for C19H18FNO2: C, 73.30; H, 5.83; N, 4.50. Found: C, 73.22; H, 5.78; N, 4.48.
3.6.5. Ethyl-6-fluoro-1-phenethyl-1,2-dihydroquinoline-3-carboxylate (18e) from 7 and 8e
Yield: 289 mg (0.89 mmol, 89%) as a yellow oil; IR: 1687, 1643, 1241 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.32 (t, J = 7.5 Hz, 2H), 7.27–7.22 (complex, 1H), 7.19 (d, J = 7.5 Hz, 2H), 7.08 (s, 1H), 6.86–6.74 (complex, 3H), 4.16 (d, J = 7.1 Hz, 2H), 3.74 (t, J = 7.4 Hz, 2H), 3.73 (s, 2H), 2.94 (t, J = 7.0 Hz, 2H), 1.27 (t, J = 7.1 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 167.7, 158.7 (d, J = 242.2 Hz), 142.0, 137.9, 133.9 (d, J = 2.5 Hz), 128.81, 128.75, 126.9, 125.6 (d, J = 7.3 Hz), 116.7 (d, J = 21.8 Hz), 113.7 (d, J = 8.1 Hz), 113.5 (d, J = 22.4 Hz), 96.4, 59.6, 52.9, 34.3, 26.6, 14.6; MS (EI): m/z 325 [M]+·; Anal. Calcd for C20H20FNO2: C, 73.83; H, 6.20; N, 4.30. Found: C, 73.80; H, 6.22; N, 4.33.
3.7. Representative Procedure for Synthesis of Fluoroquinolones
Prior to the reaction, the anhydrous DMF solvent was treated with dry O2, introduced via a gas dispersion tube, for 15 s. A 50 mL, round-bottomed flask equipped with a condenser, stir bar and vacuum adapter, was charged with MBH acetate 7 (1 mmol) in DMF/O2 (2 mL) under N2. The (1.5 mmol) amine was added in DMF/O2 (2 mL). The reaction was stirred at 23 °C for 1 h and then at 90 °C for 6 h. After TLC analysis (30% EtOAc/hexane) indicated completion of the reaction, the solution was poured into de-ionized water (15 mL), and the mixture was extracted with EtOAc (3 × 10 mL). The combined organic layers were washed with saturated NaCl and dried (Na2SO4). Removal of the solvent under vacuum gave the crude product, which was purified by trituration with 5% pentane in ether to afford the fluoroquinolone derivatives.
3.7.1. Ethyl-6-fluoro-1-methyl-4-oxo-1,4-dihydroquinoline-3-carboxylate (19a) from 7 and 8a
Yield: 212 mg (0.85 mmol, 85%) as a light tan solid, m.p. 203–204 °C; IR: 1728, 1694 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.46 (s, 1H), 8.18 (d, J = 8.9 Hz, 1H), 7.44 (d, J = 5.7 Hz, 2H), 4.40 (q, J = 7.1 Hz, 2H), 3.90 (s, 3H), 1.42 (t, J = 7.1 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 173.5 (d, J = 2.0 Hz), 165.7, 160.1 (d, J = 232.0 Hz), 149.6, 136.3 (d, J = 6.6 Hz), 130.8 (d, J = 6.8 Hz), 121.0 (d, J = 24.7 Hz), 117.8 (d, J = 7.7 Hz), 113.0 (d, J = 23.2 Hz), 110.4, 61.6, 41.6, 14.4; MS (EI): m/z 249; Anal. Calcd for C13H12FNO3: C, 62.65; H, 4.85; N, 5.62. Found: C, 62.74; H, 4.79; N, 5.56.
3.7.2. Ethyl-6-fluoro-1-isobutyl-4-oxo-1,4-dihydroquinoline-3-carboxylate (19c) from 7 and 8c
Yield: 250 mg (0.86 mmol, 86%) as a light tan solid, m.p. 150–151 °C; IR: 1727, 1695 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.43 (s, 1H), 8.21 (m, 1H), 7.42 (m, 2H), 4.41 (q, J = 7.1 Hz, 2H), 3.97 (d, J = 7.5 Hz, 2H), 2.26 (nonet, J = 7.0 Hz, 1H), 1.42 (t, J = 7.1 Hz, 3H), 1.02 (d, J = 6.6 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 173.4 (d, J = 2.4 Hz), 165.9, 159.9 (d, J = 247.8 Hz), 149.4, 135.4 (d, J = 1.9 Hz), 131.2 (d, J = 6.8 Hz), 120.8 (d, J = 28.1 Hz), 118.1 (d, J = 7.8 Hz), 113.1 (d, J = 23.0 Hz), 109.9, 61.6, 61.0, 27.7, 19.9, 14.4; MS (EI): m/z 291; Anal. Calcd for C16H18FNO3: C, 65.97; H, 6.23; N, 4.81. Found: C, 66.01; H, 6.20; N, 4.76.
3.7.3. Ethyl-1-benzyl-6-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylate (19d) from 7 and 8d
Yield: 269 mg (0.83 mmol, 83%) as a light tan solid, m.p. 169–170 °C; IR: 1727, 1691, 1619 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.60 (s, 1H), 8.18 (dd, J = 8.9, 2.9 Hz, 1H), 7.41–7.24 (complex, 5H), 7.16 (d, J = 6.3 Hz, 2H), 5.40 (s, 2H), 4.41 (q, J = 7.1 Hz, 2H), 1.42 (t, J = 7.1 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 173.5 (d, J = 2.3 Hz), 165.8, 160.0 (d, J = 248.0 Hz), 149.6, 135.6 (d, J = 1.8 Hz), 133.9, 131.2 (d, J = 7.0 Hz), 129.5, 128.8, 126.0, 121.0 (d, J = 25.3 Hz), 118.9 (d, J = 7.8 Hz), 113.0 (d, J = 23.0 Hz), 110.6, 61.1, 57.8, 14.4; MS (EI): m/z 325; Anal. Calcd for C19H16FNO3: C, 70.14; H, 4.96; N, 4.31. Found: C, 70.04; H, 4.92; N, 4.23.
3.7.4. Ethyl-6-fluoro-4-oxo-1-phenethyl-1,4-dihydroquinoline-3-carboxylate (19e) from 7 and 8e
Yield: 284 mg (0.84 mmol, 84%) as a light tan solid, m.p. 151–152 °C; IR: 1736, 1691, 1618 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.21 (dd, J = 8.9, 3.0 Hz, 1H), 8.14 (s, 1H), 7.50 (dd, J = 9.2, 4.1 Hz, 1H), 7.44 (m, 1H), 7.33–7.23 (complex, 3H), 7.06 (d, J = 7.5 Hz, 2H), 4.40 (t, J = 7.1 Hz, 2H), 4.34 (q, J = 7.1 Hz, 2H), 3.16 (t, J = 7.1 Hz, 2H), 1.36 (t, J = 7.1 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 173.3 (d, J = 2.3 Hz), 165.4, 159.9 (d, J = 247.9 Hz), 149.0, 136.1, 135.1 (d, J = 1.8 Hz), 131.2 (d, J = 6.8 Hz), 129.1, 128.7, 127.6, 121.0 (d, J = 25.2 Hz), 117.7 (d, J = 7.7 Hz), 113.3 (d, J = 23.0 Hz), 110.1, 60.9, 55.4, 35.2, 14.4; MS (EI): m/z 339; Anal. Calcd for C20H18FNO3: C, 70.78; H, 5.35; N, 4.13. Found: C, 70.69; H, 5.33; N, 4.08. The X-ray structure for 19e (CCDC 2057847) and the thermal ellipsoid plot appears in Figure 5 and the SI.
4. Conclusions
We have investigated the synthesis of dihydroquinolines and dihydronaphthyridines from Morita-Baylis-Hillman acetates and primary amines in DMF. The formation of dihydroquinolines occurs at 50 °C while dihydronaphthyridines required heating to 90 °C. Substrates for the dihydroquinolines were MBH acetates bearing 2-fluorophenyl rings activated toward SNAr ring closure by a C5 nitro or cyano group. Dihydronaphthyridine precursors were activated only by the electron-withdrawing nitrogen in a 2-fluoropyridine moiety, and were thus less reactive in the final cyclization. The transformation most likely involves a domino SN2′-SNAr process. An initial SN2′ mechanism yields the alkene having the nitrogen nucleophile predominantly trans to the SNAr acceptor ring. Under the reaction conditions, a reversible Michael addition by the excess amine or an internal addition–elimination process can equilibrate the double bond geometry to yield the alkene isomer needed for ring closure. The isolation of intermediates from the reaction disputes several earlier reports that assumed the reaction directly generated the alkene needed for cyclization. Once equilibrated, subsequent SNAr ring closure by the nitrogen nucleophile and deprotonation affords the product. Much of this selectivity appears to be guided by steric factors. Two dihydronaphthyridines derived from precursors incorporating a side chain acrylonitrile inexplicably underwent migration of the C3–C4 double bond to the C2–C3 position to afford an enaminonitrile. We have extended our study to a difluorophenyl MBH acetate and found divergent reactivity, depending upon the presence or absence of O2 in the DMF solvent. Under anaerobic conditions at 90 °C, these substrates afforded the expected dihydroquinolines while aerobic conditions led to the formation of fluoroquinolones. The difluoro MBH derivatives represent a new approach to fluoroquinolones affording yet another valuable ring system that can be generated from these versatile precursors. Good to excellent yields (72–93%) were isolated for all examples.
Supplementary Materials
The following are available online: copies of 1H NMR and 13C NMR spectra for 6, 7, 9a–f, 10a–f, 11a–f, 12a–e, 13a–f, 14b, 14d,e, 15d, 15f, 16, 18a–e and 19a–e and tables of crystal data for compounds 15d, 16 and 19e.
Author Contributions
Formal analysis, R.A.B.; investigation, J.K.A.-G., E.A. and K.M.; methodology, J.K.A.-G., E.A. and K.M.; project administration, R.A.B.; writing—original draft, R.A.B.; writing—review and editing, R.A.B., J.K.A.-G., E.A. and K.M. All authors have read and agreed to the published version of the manuscript.
Funding
Financial support for this work was obtained from the Oklahoma State University Foundation and the College of Arts and Sciences at Oklahoma State University. The University of Oklahoma Chemical Crystallography Laboratory was upgraded with support from the NSF (CHE-1726630). The Oklahoma State University State-wide NMR Facility was established with funds from the NSF (BIR-9512269), the OSU Regents for Higher Education, the W. M. Keck Foundation, and Conoco, Inc.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Copies of 1H and 13C NMR spectra for 6, 7, 9a–f, 10a–f, 11a–f, 12a–e, 13a–f, 14b, 14d,e, 15d, 15f, 16, 18a–e and 19a–e compounds are given in the SI. Data pertaining to the X-ray crystal structures for 15d, 16 and 19e are also included in the SI.
Acknowledgments
J.K.A.-G. and K.M. wish to thank K. Darrell Berlin and the OSU Foundation for Summer Scholarships. The authors also wish to thank Douglas Powell at the University of Oklahoma Chemical Crystallography Laboratory for obtaining the X-ray crystal structures. This facility was established with support from the NSF (CHE-1726630). Finally, the authors are indebted to the OSU College of Arts and Sciences for funds to purchase several departmental instruments including an FT-IR and a 400 MHz NMR unit for the State-wide NMR facility. The NMR facility was initially established with support from the NSF (BIR-9512269), the Oklahoma State Regents for Higher Education, the W. M. Keck Foundation, and Conoco, Inc.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Annor-Gyamfi, J.K.; Ametsetor, E.; Meraz, K.; Bunce, R.A. Naphthalenes and quinolines by domino reactions of Morita-Baylis-Hillman acetates. Molecules 2020, 25, 5168. [Google Scholar] [CrossRef] [PubMed]
- Bunce, R.A.; Nago, T. 1-Alkyl-2,3-dihydro-4(1H)-quinolinones by a tandem Michael-SNAr annulation reaction. J. Heterocyclic Chem. 2009, 46, 623–628. [Google Scholar] [CrossRef]
- Bunce, R.A.; Squires, S.T.; Nammalwar, B. 1-Alkyl- and (±)-1,2-dialkyl-2,3-dihydro-1,8-naphthyridine-4(1H)-ones by a tandem Michael-SNAr annulation reaction. J. Org. Chem. 2013, 78, 2144–2148. [Google Scholar] [CrossRef]
- Narendar Reddy, T.; Jayathirtha Rao, V. Importance of Baylis-Hillman adducts in modern drug discovery. Tetrahedron Lett. 2018, 59, 2859–2875. [Google Scholar] [CrossRef]
- Lima-Junior, C.G.; Vasconcellos, M.A.A. Morita-Baylis-Hillman adducts: Biological activities and potentialities to the discovery of new cheaper drugs. Bioog. Med. Chem. 2012, 20, 3954–3971. [Google Scholar] [CrossRef] [PubMed]
- Fotie, J.; Kaiser, M.; Delfin, D.A.; Manley, J.; Reid, C.S.; Paris, J.-M.; Wenzler, T.; Maes, L.; Mahasenan, K.V.; Li, C.; et al. Antitrypanosomal activity of 1,2-dihydroquinolin-6-ols and their ester derivatives. J. Med. Chem. 2010, 53, 966–982. [Google Scholar] [CrossRef]
- Bhanja, C.; Jena, S.; Nayak, S.; Mohapatra, S. Organocatalytic tandem Michael addition reactions: A powerful access to the enantioselective synthesis of functionalized chromenes, thiochromenes and 1,2-dihydroquinolines. Beilstein J. Org. Chem. 2012, 8, 1668–1694. [Google Scholar] [CrossRef]
- Pucheta, A.; Mendieta, A.; Madrigal, D.A.; Hernández-Benitez, R.I.; Romero, L.; Garduño-Siciliano, L.; Rugerio-Escalona, C.; Cruz-Lopez, M.C.; Jimenez, F.; Ramirez-Villalva, A.; et al. Synthesis and biological activity of fibrate-based acyl- and alkyl- phenoxyacetic methyl esters and 1,2-dihydroquinones. Med. Chem. Res. 2020, 29, 459–478. [Google Scholar] [CrossRef]
- Matsuda, M.; Mori, T.; Kawashima, K.; Nagatsuka, M.; Kobayashi, S.; Yamamoto, M.; Kato, M.; Takai, M.; Oda, T. Preparation of Novel 1,2-dihydroquinoline Derivatives Having Glucocorticoid Receptor Binding Activity. World Patent WO 2007032556 A1, 22 March 2007. [Google Scholar]
- Buschmann, N.; Fairhurst, R.A.; Furet, P.; Knoepfel, T.; LeBlanc, C.; Mah, R.; Mallet, F.; Martz, J.; Liao, L.; Xiong, J.; et al. Preparation of N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1-yl)methyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxamide Particles Useful in Pharmaceutical Compositions for the Treatment of Cancer. World Patent WO 2016151500 A1, 29 September 2016. [Google Scholar]
- Baruah, A.; Khanna, I.; Pillarisetti, S. Preparation and Pharmaceutical Compositions of Dihydro-Naphthyridine and Tetrahydropyrazolopyridine derivatives as Cholesteryl Ester-Transfer Protein Inhibitors. U.S. Patent 20060135551 A1, 22 June 2006. [Google Scholar]
- Zhanel, G.G.; Walkty, A.; Vercaigne, L.; Karlowsky, J.A.; Embil, J.; Gin, A.S.; Hoban, D.J. The new fluoroquinolones: A critical review. Can. J. Infect. Dis. 1999, 10, 207–238. [Google Scholar] [CrossRef]
- Pham, T.D.M.; Ziora, Z.M.; Blaskovich, M.A.T. Quinolone antibiotics. Med. Chem. Commun. 2019, 10, 1719–1739. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.; Pennefather, P.M.; Kaye, S.B.; Hart, C.A. Fluoroquinolones: Place in Ocular therapy. Drugs 2001, 61, 747–761. [Google Scholar] [CrossRef]
- Falagas, M.E.; Matthaiou, D.K.; Bliziotis, I.A. Systematic review: Fluoroquinolones for the treatment of intra-abdominal surgical infections. Aliment. Pharmacol. Ther. 2007, 25, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Hurley, M.; Smyth, A. Fluoroquinolones in the treatment of bronchopulmonary disease in cystic fibrosis. Ther. Adv. Respir. Dis. 2012, 6, 363–373. [Google Scholar] [CrossRef]
- Raz-Pasteur, A.; Shasha, D.; Paul, M. Fluoroquinolones or macrolides alone versus combined with β-lactams for adults with community-acquired pneumonia: Systematic review and meta-analysis. Int. J. Antimicrob. Agt. 2015, 46, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.N.; Lee, H.J.; Lee, K.Y.; Kim, H.S. Synthesis of 3-quinolinecarboxylic acid esters from the Baylis-Hillman adducts of 2-halobenzaldehyde N-tosylimines. Tetrahedron Lett. 2001, 42, 3737–3740. [Google Scholar] [CrossRef]
- Singh, B.; Chandra, A.; Singh, R.H. Base-free amination of BH acetates of 2-chloroquinolinyl-3-carboxaldehydes: A facile route to the synthesis of N-substituted-1,2-dihydrobenzo[b][1.8]naphthyridines. Tetrahedron 2011, 67, 2441–2446. [Google Scholar] [CrossRef]
- Gupta, T.; Bharadwaj, K.C.; Singh, R.M. Cascade SN2’-SNAr, Elimination and 1,5-hydride shift reactions by acetylacetone/acetoacetic esters: Synthesis of 9,10-dihydroacridines. Eur. J. Org. Chem. 2016, 4981–4984. [Google Scholar] [CrossRef]
- Basavaiah, D.; Rao, J.S. Applications of Baylis-Hillman acetates: One pot, facile and convenient synthesis of substituted γ-lactams. Tetrahedron Lett. 2004, 45, 1621–1625. [Google Scholar] [CrossRef]
- Basavaiah, D.; Reddy, K.R. Simple one-pot synthesis of tri and tetracyclic frameworks containing [1,8]-naphthyridin-2-one moiety from the Baylis-Hillman adducts. Tetrahedron 2010, 66, 1215–1219. [Google Scholar] [CrossRef]
- Singh, V.; Hutait, S.; Batra, S. Reductive cyclization-mediated synthesis of fused polycyclic quinolines from Baylis-Hillman adducts of acrylonitrile: Scope and limitations. Eur. J. Org. Chem. 2009, 3454–3466. [Google Scholar] [CrossRef]
- Procopiou, P.A.; Baugh, S.P.D.; Flack, S.S.; Inglis, G.G.A. An extremely powerful acylation reaction of alcohols with acid anhydrides catalyzed by trimethylsilyl trifluoromethanesulfonate. J. Org. Chem. 1998, 63, 2342–2347. [Google Scholar] [CrossRef]
- Genest, A.; Portinha, D.; Fleury, E.; Ganachaud, F. The aza-Michael reaction as an alternative strategy to generate advanced silicon-based (macro)molecules and materials. Prog. Polym. Sci. 2017, 72, 61–110. [Google Scholar] [CrossRef]
- Buchholz, R.; Hoffmann, H.M.R. α-Methylidene and α-Alkylidene-β-lactams from nonproteinogenic amino acids. Helv. Chim. Acta 1991, 74, 1213–1220. [Google Scholar] [CrossRef]
- Stohrer, W.-D. On the stereochemistry of the SN2’ reaction. Angew. Chem. Int. Ed. 1983, 22, 613–614. [Google Scholar] [CrossRef]
- Bergman, E.D.; Ginsburg, D.; Pappo, R. The Michael Reaction. Org. React. 1959, 10, 179–555. [Google Scholar] [CrossRef]
- Werner, R.M.; Williams, L.M.; Davis, J.T. The C-glycosyl analog of an N-linked glycoamino acid. Tetrahedron Lett. 1998, 39, 9135–9138. [Google Scholar] [CrossRef]
- Shao, H.; Ekthawatchai, S.; Chen, C.-S.; Wu, S.-H.; Zou, W. 1,2-Migration of 2’-oxoalkyl group and concomitant synthesis of 2-C-branched O-, S-glycosides and glycosyl azides via 1,2-cyclopropanated sugars. J. Org. Chem. 2005, 70, 4726–4734. [Google Scholar] [CrossRef] [PubMed]
- Henderson, A.P.; Bleasdale, C.; Delaney, K.; Lindstrom, A.B.; Rappaport, S.M.; Waidyanatha, S.; Watson, W.P.; Golding, B.T. Evidence for the formation of Michael adducts from reactions of [E,E]-muconaldehyde with glutathione and other thiols. Bioorg. Chem. 2005, 33, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Hintermann, L.; Dittmer, C. Asymmetric ion-pairing catalysis of the reversible cyclization of 2’- hydroxychalcone to flavanone: Asymmetric catalysis of an equilibrating reaction. Eur. J. Org. Chem. 2012, 5573–5584. [Google Scholar] [CrossRef]
- Bach, R.D.; Wolber, G.J. Stereochemistry of the concerted SN2’ reaction of 3-chloropropene: A theoretical study. J. Am. Chem. Soc. 1985, 107, 1352–1357. [Google Scholar] [CrossRef]
- Achord, J.M.; Hussey, C.L. Determination of dissolved oxygen in nonaqueous electrochemical solvents. Anal. Chem. 1980, 52, 601–602. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).