Immunomodulatory Responses of Two Synthetic Peptides against Salmonella Typhimurium Infection
Abstract
:1. Introduction
2. Results
2.1. Pin2[G] and FA1 Are Innocuous to Viability of Macrophages
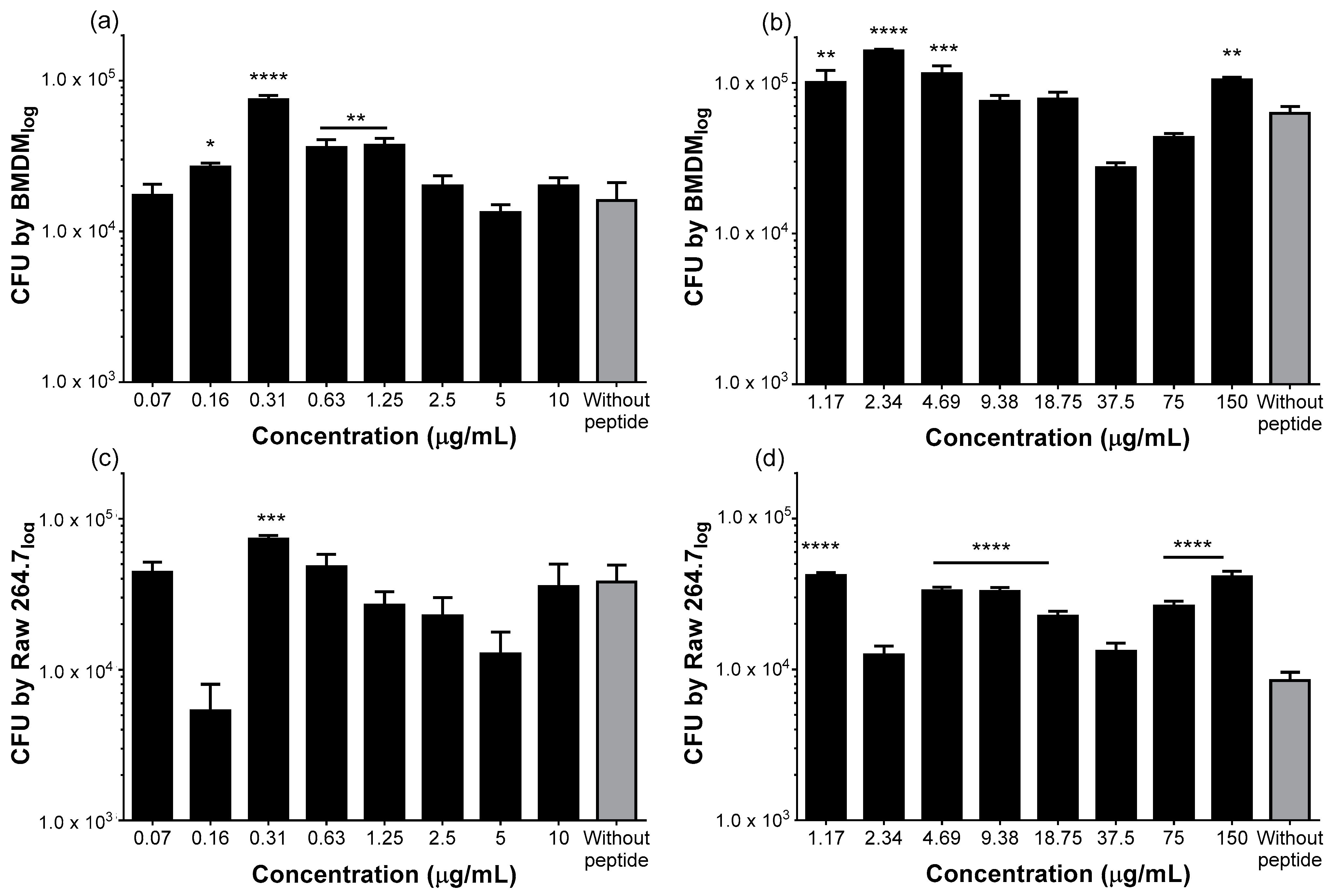
2.2. Both Peptides Stimulate Phagocytosis of S. Typhimurium by Macrophages
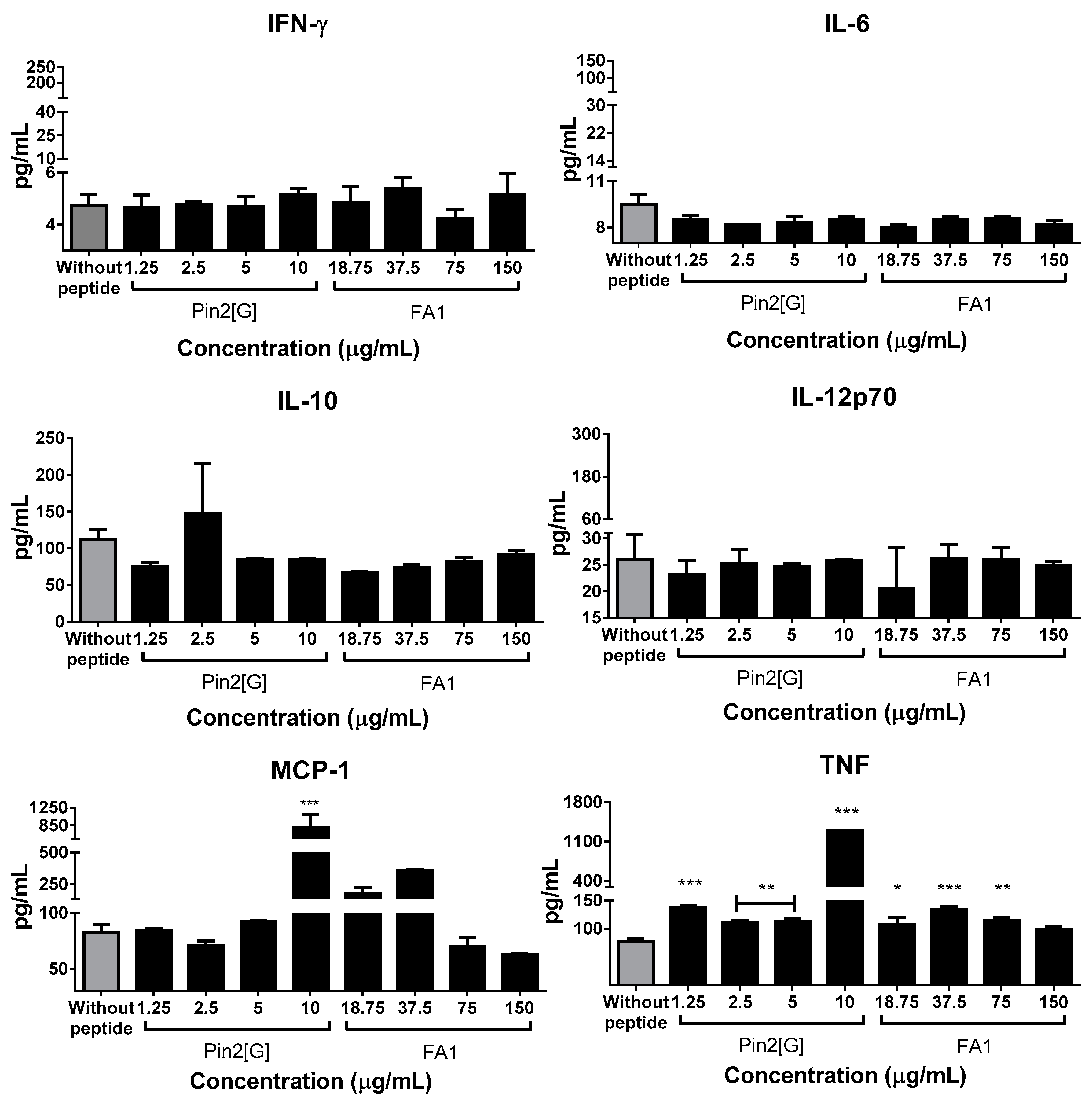
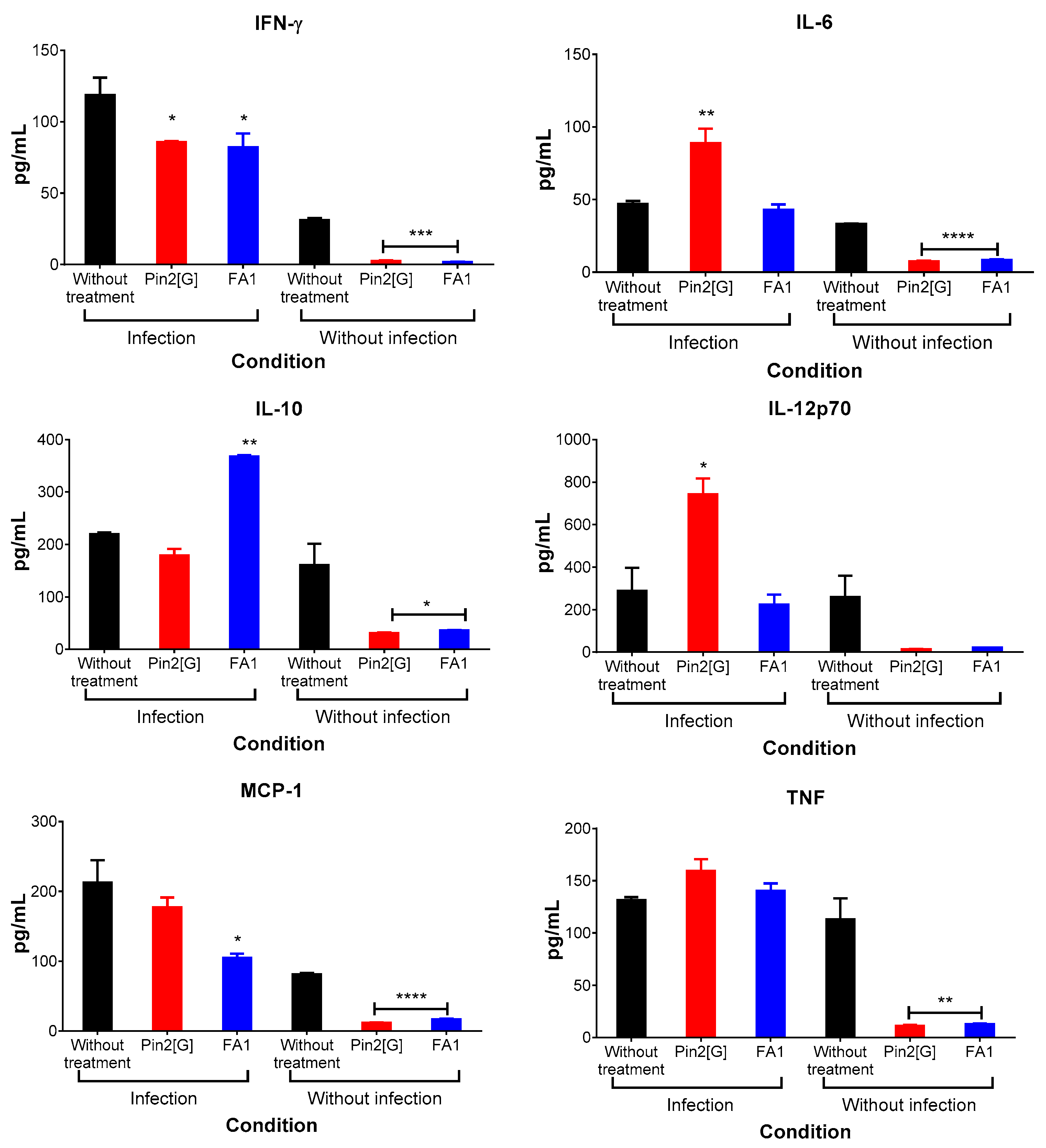
2.3. Pin2[G] and FA1 Influence the Cytokine Profiles of Macrophages Infected with S. Typhimurium
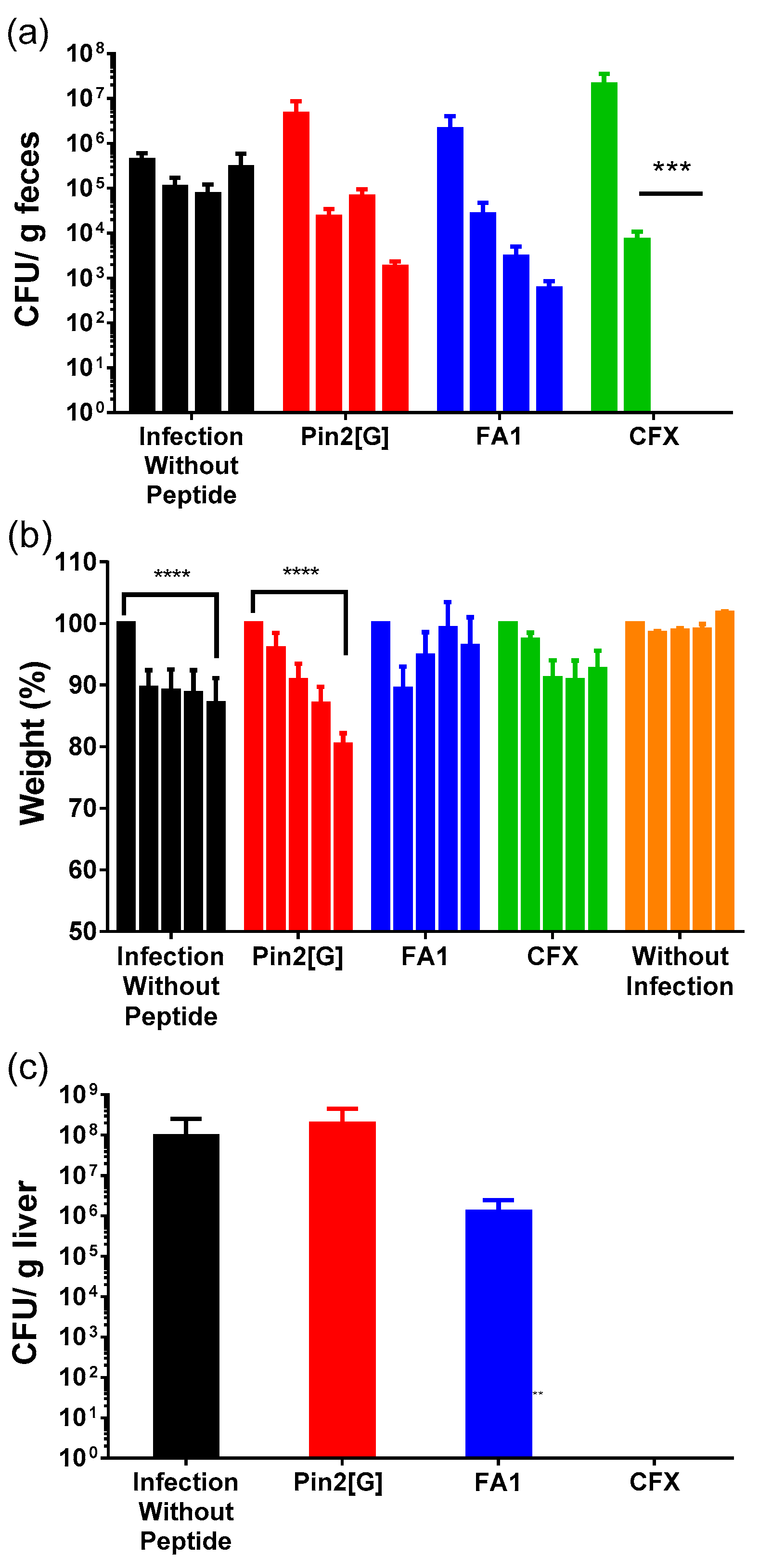
2.4. Both HDP Decreases Salmonella Typhimurium Gastrointestinal Infection
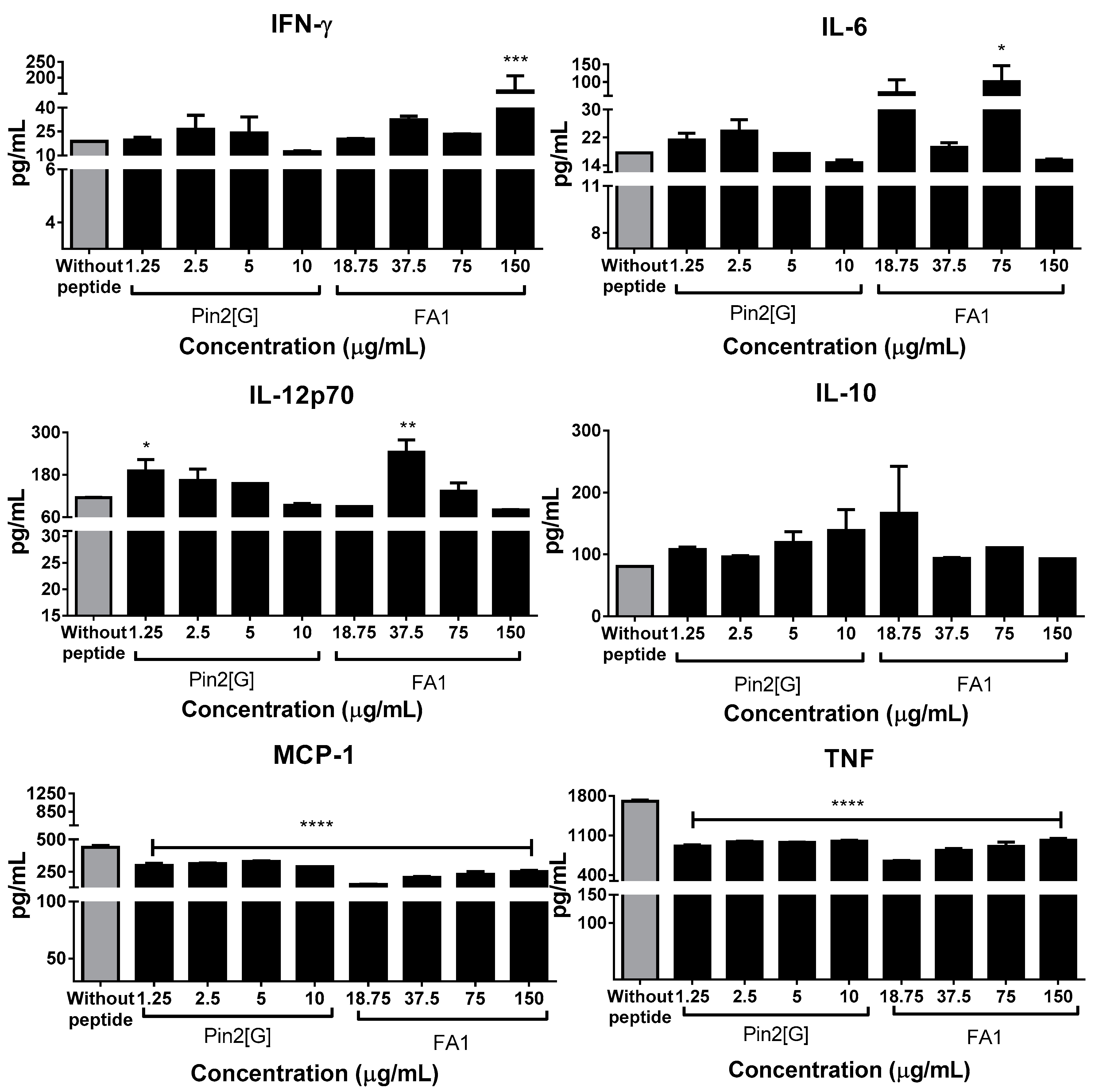
2.5. Pin2[G] Promotes Inflammatory Cytokines While FA1 Stimulates an Anti-Inflammatory Profile
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Cell Lines
4.2. Animals
4.3. Viability Assay
4.4. Phagocytosis Stimulation
4.5. Gastric Model Infection
4.6. Bacterial Quantification
4.7. Cytokine Profiles
4.8. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- World Health Organization. Antimicrobial Resistance: Global Report on Surveillance; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- GBD 2017 Typhoid and Paratyphoid Collaborators. The global burden of typhoid and paratyphoid fevers: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Infect. Dis. 2019, 19, 369–381. [Google Scholar] [CrossRef] [Green Version]
- Marchello, C.S.; Hong, C.Y.; Crump, J.A. Global typhoid fever incidence: A systematic review and meta-analysis. Clin Infect Dis. 2019, 68 (Suppl. S2), S105–S116. [Google Scholar] [CrossRef] [Green Version]
- Majowicz, S.E.; Musto, J.; Scallan, E.; Angulo, F.J.; Kirk, M.; Brien, S.J.O.; Jones, T.F.; Fazil, A.; Hoekstra, R.M. The Global Burden of Nontyphoidal Salmonella Gastroenteritis. Clin. Infect. Dis. 2010, 50, 882–889. [Google Scholar] [CrossRef] [Green Version]
- Broz, P.; Ohlson, M.B.; Monack, D.M. Innate immune response to Salmonella Typhimurium, a model enteric pathogen. Gut Microbes 2012, 3, 62–70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hurley, D.; McCusker, M.P.; Fanning, S.; Martins, M. Salmonella–Host Interactions–Modulation of the Host Innate Immune System. Front. Immunol. 2014, 5, 481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boto, A.; De La Lastra, J.M.P.; González, C.C. The road from host-defense peptides to a new generation of antimicrobial drugs. Molecules 2018, 23, 311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mwangi, J.; Hao, X.; Lai, R.; Zhang, Z.Y. Antimicrobial peptides: New hope in the war against multidrug resistance. Zool. Res. 2019, 40, 488–505. [Google Scholar] [CrossRef] [PubMed]
- Fjell, C.D.; Hiss, J.A.; Hancock, R.E.W.; Schneider, G. Designing antimicrobial peptides: Form follows function. Nat. Rev. Drug Discov. 2012, 11, 37–51. [Google Scholar] [CrossRef]
- Harrison, P.L.; Abdel-Rahman, M.A.; Miller, K.; Strong, P.N. Antimicrobial peptides from scorpion venoms. Toxicon 2014, 88, 115–137. [Google Scholar] [CrossRef]
- Corzo, G.; Escoubas, P.; Villegas, E.; Barnham, K.J.; He, W.; Norton, R.S.; Nakajima, T. Characterization of unique amphipathic antimicrobial peptides from venom of the scorpion Pandinus imperator. Biochem. J. 2001, 45, 35–45. [Google Scholar] [CrossRef]
- Rodríguez, A.; Villegas, E.; Montoya-rosales, A.; Rivas-santiago, B.; Corzo, G. Characterization of Antibacterial and Hemolytic Activity of Synthetic Pandinin 2 Variants and Their Inhibition against Mycobacterium tuberculosis. PLoS ONE 2014, 9, e101742. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Vásquez, L.; Silva-Sánchez, J.; Jiménez-Vargas, J.M.; Rodríguez-Romero, A.; Muñoz-Garay, C.; Rodríguez, M.C.; Possani, L.D. Enhanced antimicrobial activity of novel synthetic peptides derived from vejovine and hadrurin. BBA Gen. Subj. 2013, 1830, 3427–3436. [Google Scholar] [CrossRef]
- Arenas, I.; Ibarra, M.A.; Santana, F.L.; Villegas, E.; Hancock, R.E.W.; Corzo, G. In Vitro and In Vivo Antibiotic Capacity of Two Host Defense Peptides. Antimicrob. Agents Chemother. 2020, m64, e00145-20. [Google Scholar] [CrossRef]
- Shane, A.L.; Mody, R.K.; Crump, J.A.; Tarr, P.I.; Steiner, T.S.; Kotloff, K.; Langley, J.M.; Wanke, C.; Warren, C.A.; Cheng, A.C.; et al. 2017 Infectious Diseases Society of America Clinical Practice Guidelines for the Diagnosis and Management of Infectious Diarrhea. Clin. Infect. Dis. 2017, 65, e45–e80. [Google Scholar] [CrossRef] [Green Version]
- Veeraraghavan, B.; Pragasam, A.K.; Bakthavatchalam, Y.D.; Ralph, R. Typhoid fever: Issues in laboratory detection, treatment options & concerns in management in developing countries. Future Sci. OA 2018, 4, FSO312. [Google Scholar] [CrossRef] [Green Version]
- Haney, E.F.; Straus, S.K.; Hancock, R.E.W. Reassessing the Host Defense Peptide Landscape. Front. Chem. 2019, 7, 43. [Google Scholar] [CrossRef] [Green Version]
- Fusco, A.; Savio, V.; Cammarota, M.; Alfano, A.; Schiraldi, C.; Donnarumma, G. Beta-Defensin-2 and Beta-Defensin-3 Reduce Intestinal Damage Caused by Salmonella Typhimurium Modulating the Expression of Cytokines and Enhancing the Probiotic Activity of Enterococcus faecium. J. Immunol. Res. 2017, 2017, 6976935. [Google Scholar] [CrossRef] [Green Version]
- Mookherjee, N.; Anderson, M.A.; Haagsman, H.P.; Davidson, D.J. Antimicrobial host defense peptides: Functions and clinical potential. Nat. Rev. Drug Discov. 2020, 19, 311–332. [Google Scholar] [CrossRef] [PubMed]
- Yacoub, T.; Rima, M.; Karam, M.; Fajloun, J.-M.S.A.Z. Antimicrobials from Venomous Animals: An Overview. Molecules 2020, 25, 2402. [Google Scholar] [CrossRef] [PubMed]
- Arenas, I.; Villegas, E.; Walls, O.; Barrios, H.; Rodríguez, R.; Corzo, G. Antimicrobial Activity and Stability of Short and Long Based Arachnid Synthetic Peptides in the Presence of Commercial Antibiotics. Molecules 2016, 21, 225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasilchenko, A.S.; Rogozhin, E.A. Sub-inhibitory Effects of Antimicrobial Peptides. Front. Microbiol. 2019, 10, 1160. [Google Scholar] [CrossRef] [Green Version]
- Lei, J.; Sun, L.; Huang, S.; Zhu, C.; Li, P.; He, J.; Mackey, V.; Coy, D.H.; He, Q. The antimicrobial peptides and their potential clinical applications. Am J Transl Res. 2019, 11, 3919–3931. [Google Scholar]
- Ji, S.Y.; Lee, H.; Hwangbo, H.; Hong, S.-H.; Cha, H.-J.; Park, C.; Kim, D.-H.; Kim, G.-Y.; Kim, S.; Kim, H.-S.; et al. A Novel Peptide Oligomer of Bacitracin Induces M1 Macrophage Polarization by Facilitating Ca(2+) Influx. Nutrients 2020, 12, 1603. [Google Scholar] [CrossRef]
- Payoungkiattikun, W.; Joompang, A.; Thongchot, S.; Nowichai, B.; Jangpromma, N.; Klaynongsruang, S. Evidence of multi-functional peptide activity: Potential role of KT2 and RT2 for anti-inflammatory, anti-oxidative stress, and anti-apoptosis properties. Appl. Biol. Chem. 2020, 63, 5. [Google Scholar] [CrossRef]
- Małaczewska, J.; Kaczorek-Łukowska, E.; Wójcik, R.; Rękawek, W.; Siwicki, A.K. In vitro immunomodulatory effect of nisin on porcine leucocytes. J. Anim. Physiol. Anim. Nutr. 2019, 103, 882–893. [Google Scholar] [CrossRef] [PubMed]
- Scott, M.G.; Dullaghan, E.; Mookherjee, N.; Glavas, N.; Waldbrook, M.; Thompson, A.; Wang, A.; Lee, K.; Doria, S.; Hamill, P.; et al. An anti-infective peptide that selectively modulates the innate immune response. Nat. Biotechnol. 2007, 25, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Dupont, A.; Kaconis, Y.; Yang, I.; Albers, T.; Woltemate, S.; Heinbockel, L.; Anderson, M.; Suerbaum, S.; Brandenburg, K.; Hornef, M.W. Intestinal mucus affinity and biological activity of an orally administered antibacterial and anti-inflammatory peptide. Gut 2015, 64, 222–232. [Google Scholar] [CrossRef]
- Marin, M.; Holani, R.; Blyth, G.A.D.; Drouin, D.; Odeón, A.; Cobo, E.R. Human cathelicidin improves colonic epithelial defenses against Salmonella Typhimurium by modulating bacterial invasion, TLR4 and pro-inflammatory cytokines. Cell Tissue Res. 2019, 376, 433–442. [Google Scholar] [CrossRef]
- Wuerth, K.C.; Falsafi, R.; Hancock, R.E.W. Synthetic host defense peptide IDR-1002 reduces inflammation in Pseudomonas aeruginosa lung infection. PLoS ONE 2017, 12, e0187565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marques-Neto, L.M.; Trentini, M.M.; das Neves, R.C.; Resende, D.P.; Procopio, V.O.; da Costa, A.C.; Kipnis, A.; Mortari, M.R.; Schwartz, E.F.; Junqueira-Kipnis, A.P. Antimicrobial and Chemotactic Activity of Scorpion-Derived Peptide, ToAP2, against Mycobacterium massiliensis. Toxins 2018, 10, 219. [Google Scholar] [CrossRef] [Green Version]
- Bacalum, M.; Radu, M. Cationic Antimicrobial Peptides Cytotoxicity on Mammalian Cells: An Analysis Using Therapeutic Index Integrative Concept. Int. J. Pept. Res. Ther. 2015, 21, 47–55. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, F.; Huang, Z.; Liu, H.; Xie, C.; Zhang, J.; Thacker, P.A.; Qiao, S. Effects of the antimicrobial peptide cecropin AD on performance and intestinal health in weaned piglets challenged with Escherichia coli. Peptides 2012, 35, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Machado, R.J.A.; Estrela, A.B.; Nascimento, A.K.L.; Melo, M.M.A.; Torres-Rêgo, M.; Lima, E.O.; Rocha, H.A.O.; Carvalho, E.; Silva-Junior, A.A.; Fernandes-Pedrosa, M.F. Characterization of TistH, a multifunctional peptide from the scorpion Tityus stigmurus: Structure, cytotoxicity and antimicrobial activity. Toxicon 2016, 119, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Guide for the Care and Use of Laboratory Animals, 8th ed.; National Academies Press: Washington, DC, USA, 2011.
- de Aluja, A.S. Laboratory animals and official Mexican norms (NOM-062-ZOO-1999). Gac. Med. Mex. 2002, 138, 295–298. [Google Scholar] [PubMed]
- Noster, J.; Chao, T.C.; Sander, N.; Schulte, M.; Reuter, T.; Hansmeier, N.; Hensel, M. Proteomics of intracellular Salmonella enterica reveals roles of Salmonella pathogenicity island 2 in metabolism and antioxidant defense. PLoS Pathog. 2019, 15, e1007741. [Google Scholar] [CrossRef] [PubMed] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibarra-Valencia, M.A.; Espino-Solis, G.P.; Estrada, B.E.; Corzo, G. Immunomodulatory Responses of Two Synthetic Peptides against Salmonella Typhimurium Infection. Molecules 2021, 26, 5573. https://doi.org/10.3390/molecules26185573
Ibarra-Valencia MA, Espino-Solis GP, Estrada BE, Corzo G. Immunomodulatory Responses of Two Synthetic Peptides against Salmonella Typhimurium Infection. Molecules. 2021; 26(18):5573. https://doi.org/10.3390/molecules26185573
Chicago/Turabian StyleIbarra-Valencia, Marco Antonio, Gerardo Pável Espino-Solis, Blanca Elisa Estrada, and Gerardo Corzo. 2021. "Immunomodulatory Responses of Two Synthetic Peptides against Salmonella Typhimurium Infection" Molecules 26, no. 18: 5573. https://doi.org/10.3390/molecules26185573
APA StyleIbarra-Valencia, M. A., Espino-Solis, G. P., Estrada, B. E., & Corzo, G. (2021). Immunomodulatory Responses of Two Synthetic Peptides against Salmonella Typhimurium Infection. Molecules, 26(18), 5573. https://doi.org/10.3390/molecules26185573





