Abstract
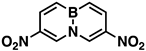
Mono- and dinitro-BN-naphthalenes, i.e., 1-nitro-, 3-nitro-, 1,6-dinitro-, 3,6-dinitro-, and 1,8-dinitro-BNN, were generated in the nitration of 9,10-BN-naphthalene (BNN), a boron–nitrogen (BN) bond-embedded naphthalene, with AcONO2 and NO2BF4 in acetonitrile. The nitrated products were isolated and characterized by NMR, GC-MS, IR, and X-ray single crystallography. The effects of the nitration on the electron density and aromaticity of BNN were evaluated by B-11 NMR analysis and HOMA calculations.
1. Introduction
As part of a continuing program in the synthesis of nitro-substituted, boron–nitrogen (BN) bond-embedded aromatics as next-generation energetic materials, we undertook the nitration of 9,10-BN-naphthalene (BNN) with AcONO2 and NO2BF4 in acetonitrile. The nitration led to the formation of mono- and dinitro-BN-naphthalenes. To our knowledge, dinitro-BN aromatics have not been reported previously.
BN aromatics have been widely investigated in recent years since the materials possess attractive physical and chemical properties and hold potential applications in the light-emitting polymer [1,2,3,4,5,6,7,8,9,10,11,12], semiconductor [13,14,15,16,17], and other research fields [18,19,20,21,22,23,24,25,26,27,28,29]; however, there have been relatively few reports on the synthesis of nitrated BN aromatics. Thus far, only five mononitro-BN aromatics have been reported [30,31]. Dewar and their co-workers [30], in 1959, reported the first nitration of a BN aromatic, BN-phenanthrene (BNP), yielding a mixture of mononitro-substituted BNPs (1). In their work, the nitrating reagent, acetyl nitrate (AcONO2), was generated by treating Ac2O with HNO3 in acetic acid. This is a typical procedure for generating AcONO2 in organic synthesis, but in our hands, AcONO2 generated by this method failed to nitrate BN-naphthalene, giving only decomposition. Fang and co-workers [31], in 2017, successfully nitrated azaborazines to nitro-azaborazines (2) and BNN to 3-nitro-BNN (3) using AcONO2 generated from hydrated metal nitrates and acetyl chloride (AcCl) in CH2Cl2. The nitration provided highly positional selectivity at the para-position relative to the boron atom, but the nitration did not produce higher nitrated BN derivatives even at the nitration temperature of 95 °C [31].
2. Results and Discussions
2.1. Nitration of BN-Naphthalene
As mentioned above, a successful nitration of boron–nitrogen bond-embedded aromatics critically depends on the nitration conditions. In our preliminary trial, it was found that BN-naphthalene is sensitive to certain nitration reagents, such as HNO3/H2SO4, N2O4/AcOH, HNO3/Ac2O/AcOH/, and HNO3/Ac2O. These acidic nitration conditions seem to decompose BNN. Therefore, neutral nitration conditions such as AcONO2 and NO2BF4 in acetonitrile were investigated in the hope that BNN would survive under the reaction conditions and produce higher nitrated BNN products.
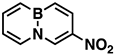
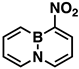
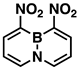
Indeed, when AcONO2 was generated from AgNO3 and AcCl and the nitration was carried out in anhydrous acetonitrile, mono- and dinitro-BN-naphthalenes were formed. Using one equivalent of the nitration reagent, only mononitro-BNNs were observed, with about 50% conversion, as judged by GC-MS and NMR. By increasing the nitration reagent to 2.7 equivalents, the starting material BNN was consumed completely with the formation of a mixture of mono- and dinitro-BNNs. The procedure involved treating BNN [32] (1.7 mmol) with 2.7 equivalents of AcONO2 generated from 2.7 equivalents of AgNO3 and 3.1 equivalents of AcCl in anhydrous acetonitrile at 10 °C and stirring the mixture at 20 °C for 2 h. The products were isolated by silica gel column chromatography, eluting with CH2Cl2/hexane. The first product eluted was identified as the known 3-nitro-BNN (3) [31]. Further elution gave a second, previously unreported product, 1-nitro-BN-naphthalene (4). The molecular structure was confirmed by GC-MS and 1H and 13C NMR and further confirmed by X-ray crystallographic analysis (all spectra can be found in Supplementary Materials). The third product eluted from the column was assigned as 3,6-dinitro-BN-naphthalene (5) based on 1H and 13C NMR, and high-resolution mass spectroscopy (m/z = 219.0459, corresponding to dinitro-BNN: C8H6BN3O4, calculated 219.0451). Following compound 5, two other dinitro-BNNs, 1,6- (6) and 1,8-dinitro-BNN (7), were isolated. Both structures were assigned by NMR, GC-MS, and X-ray single-crystal analysis. The ratio of 3, 4, 5, 6, and 7 isolated from the nitration is 45:23:6:16:11, with a total yield of 37%.
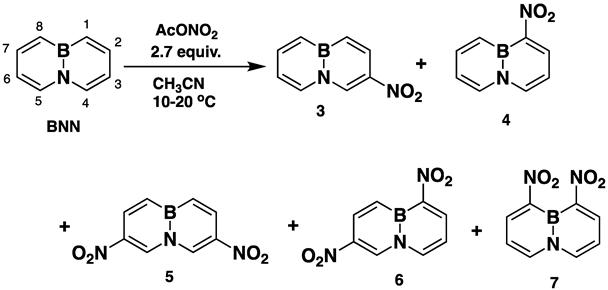
Further increasing the number of equivalents of the nitration reagent increases the amounts of dinitro-BNNs but decreases the overall yield. When the nitration reagent was increased to 6.0 equivalents, no detectable mono- and dinitro-BNNs nor trinitro- or higher nitrated BN derivatives were found.
Nitronium tetrafluoroborate (NO2BF4) is a relatively powerful nitration reagent which has been previously reported to give poly-nitro-substituted aromatics under mild conditions [33,34]. Applying the reagent to BNN and carrying out the nitration with three equivalents of NO2BF4 under similar reaction conditions to the AcONO2 nitration (anhydrous CH3CN, 10–20 °C), we isolated only 3,6-dinitro-(5) and 1,6-dinitro-BNN (6), with no detectable amounts of mononitro-BNNs nor 1,8-dinitro-BNN (7). The ratio of 5 and 6 is 53:47 based on 1H NMR integration, with a total isolated yield of 39%.
To look more closely at the relative reactivities of the nitration reagents to BN-naphthalene, we carefully treated BNN with one equivalent of AcONO2 and NO2BF4, under identical reaction conditions, with the assumption that only mono-nitrated products would be formed. Indeed, AcONO2 nitration, according to NMR analysis, gave only mononitro-BNN (3) and (4) and un-reacted BNN. The ratio of 3:4:BNN was estimated to be 34:17:49 by integration of the NMR signals (Figure 1a). Replacing AcONO2 with NO2BF4, we observed not only mono-nitrated products 3 and 4 but also dinitro-BNN (6), even though the starting material BNN was also not completely consumed. The product ratio of 3:4:6:BNN was 23:33:13:31 (Figure 1b). Different from AcONO2 nitration, NO2BF4 nitration gave 1-nitro-BNN (4) as the major product, while AcONO2 nitration gave predominately 3-nitro-BNN (3).
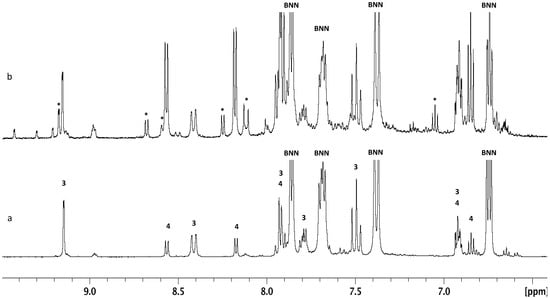
Figure 1.
1 HNMR analysis of the nitration mixture from the reaction of BNN with AcONO2 and NO2BF4 in CH3CN at 10–20 °C. The products were measured in CD2Cl2. (a) AcONO2 prepared from 1.0 equiv. of AgNO3 and 1.1 equiv. of AcCl. (b) NO2BF4, 1.0 equivalent (dinitro-BNN (6) labeled as *).
2.2. The Spectral and Structural Features of Nitro-BNNs
The introduction of nitro groups to BNN results in a downfield chemical shift for all protons. Similar to carbon–carbon bond-based aromatics, nitro groups, being strongly electron-withdrawing, lower the electron density on the BNN heterocyclic rings, resulting in significant deshielding of the ring protons. However, the nitro groups do not move B-11 chemical shifts in the same manner; B-11 resonances are shifted both upfield and downfield depending on the position where the nitro group is attached. The chemical shift of BNN itself appears at 27.8 ppm (28.4 ppm in n-hexane and 27.9 ppm in AcOH [35]). The introduction of a nitro group to position-1 (4) results in a 2.2 ppm upfield shift to 25.6 ppm. A higher upfield shift of 5.2 ppm is observed when the second nitro group is introduced to position-8 (7), giving a B-11 chemical shift at 22.6 ppm. However, when the nitro group is attached to position-3 (3), the B-11 shifts downfield by 1.7 ppm to 29.5 ppm. Further introducing a nitro group to position-6 (5) leads to a further downfield shift by 2.1 ppm to 29.9 ppm (Table 1).

Table 1.
11 B NMR chemical shifts of nitrated BNN and corresponding boron π-electron density.
The chemical shift of B-11 in homogeneous solution has a close linear relationship with the boron π-electron density [36]. Applying Michl’s equation [37], the boron electron density for each nitro-BNN based on the chemical shifts was calculated (Table 1). This shows that the nitro groups attached to the carbons at position-1 and -8 increase the boron π-electron density, but at position-3 and -6, they decrease the density. Baranac-Stojanovic and Stojanovic [38] pointed out that nitro-1,2-azaborazine with a nitro group at the α-position to the boron atom is more stable than its isomer where the nitro group is in the para-position. It was reasoned that the electron-withdrawing nitro group on the carbon at the α-position to the boron atom results in the electron density being drawn to the electron-deficient boron from the neighboring nitrogen to provide a better electrostatic stabilization energy to the molecule [38].
The other important features of a BN aromatic compound are bond lengths and bond angles. These parameters are related to molecular aromaticity and stability. Therefore, we tried to prepare single crystals for all nitro-BNNs to determine the parameters, but only 4, 6, and 7 with a nitro group at position-1 provided crystals of sufficient quality for X-ray analysis. Crystals 4 and 6 are in the orthorhombic space group, with densities of 1.406 and 1.583 g/cm3, respectively. Compound 7 is monoclinic, with a density of 1.553 g/cm3 (BNN [39], ρ = 1.215 g/cm3). The crystallographic analysis confirms that the introduction of nitro groups to BNN rings does not significantly distort the structure of the molecule. The rings in 4 and 6 are completely planar. In 7, the rings have a slight torsional angle of ~7.3° centered on the boron atom. The nitro groups on 4 and 6 are co-planar with BNN rings, but the nitro groups in 7 are rotated about 39° out of the plane of the heterocyclic rings due to the steric crowding of the nitro groups.
The bond lengths of 4, 6, and 7 are summarized in Table 2, along with the reported bond lengths of the parent BNN [39,40]. All other crystal structure data can be found in the supporting information.

Table 2.
Bond lengths of 4, 6, and 7 from X-ray crystallographic analysis a.
The B–N bond length is usually used as a measure of the relative stability of BN aromatics. Table 2 shows that the introduction of nitro groups to BNN does not significantly change the B–N bond length. When a nitro group is attached to BNN at position-1 (to 4), the B–N bond length is lengthened slightly by 0.011 Å, from 1.461 to 1.472 Å. With a further introduction of a second nitro group to position-8 (to 7), the length of the B–N bond is not lengthened but slightly shortened, changing from 1.472 to 1.460 Å. However, when the second nitro group is introduced to 4 at position-6 (to 6), the B–N bond length falls between the B–N bond lengths of 4 and 7 at 1.469 Å (Table 1). To look closely at how the bond lengths of nitro-BNNs impact the molecular aromaticity, we introduced the HOMA index [41,42] by using bond lengths collected from the X-ray crystallographic analysis. The results are depictured in Figure 2.
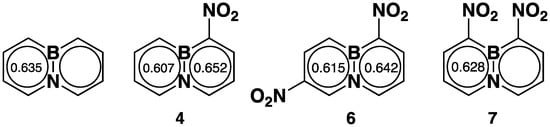
Figure 2.
Aromaticity of nitro-BNNs calculated using HOMA model.
According to HOMA calculations, BN-naphthalene (BNN) possesses 81% of the aromaticity of naphthalene [41]. The introduction of one nitro group to position-1 slightly changes the aromaticity of BNN (±2%). The same results were observed when the second nitro group was introduced to position-6 or -8, suggesting that dinitro-BNNs retained a significant amount of aromaticity.
3. Conclusions
We demonstrated the first reported formation of dinitro-substituted-BN-naphthalenes using AcONO2 and NO2BF4 as the nitrating reagents in acetonitrile. These new boron–nitrogen bond-embedded nitrocompounds were fully characterized by NMR, GC-MS, IR, and X-ray crystallographic analysis. In addition, the positional effect of nitro substitution on the electron density of the boron atom of BNN, along with a comparison of the positional effect of nitration on the aromaticity of nitrated BNN with respect to the parent BNN, was discussed.
4. Experimental Section
All reagents and anhydrous solvents were purchased from commercial suppliers and used without further purification except nitronium tetrafluoroborate (NO2BF4). The purity of acetyl chloride was ≥ 99%, AgNO3 ≥ 99%, and anhydrous CH3CN, ≥ 99.9%. NO2BF4 was purified by washing the commercial product with anhydrous nitromethane and CH2Cl2 under argon before use. NO2BF4 (commercial product, ≥ 95%, 5 g) was placed into a Schlenk tube equipped with a fritted filter. Anhydrous nitromethane (10 mL) was added, and the mixture was stirred with a glass bar under argon for 10–20 s. The liquid was then drawn under argon. The solid was washed with anhydrous nitromethane (10 mL) for one more time, followed by anhydrous CH2Cl2 (3 × 10 mL), and dried under slow-flowing argon for 2 h. 9,10-BN-Naphthalene was prepared from di-(3-butene-1-yl)amine using a slight modification of Dewar’s method [32] and purified by column chromatography (silica gel, CH2Cl2:Hexane, 1:5, Rf = 0.5), followed by recrystallization from cold pentane.
1H and 13C NMR spectra were acquired on either a Bruker 500 MHz spectrometer (500 and 150 MHz, respectively) or an Anasazi Instruments Eft-90 MHz spectrometer with a Varian magnet (90 and 22.5 MHz, respectively). 11B NMR was performed on an Anasazi Instruments Eft-90 MHz spectrometer (28.1 MHz) using BF3.Et2O as the reference (0 ppm). 1H and 13C NMR chemical shifts were reported relative to the residual solvent as internal standard, such as CD2Cl2 (5.32 ppm for proton and 53.5 ppm for C-13). Infrared spectra (thin films) were collected using a Bruker Alpha ZnSe ATR FTIR. GC-MS was performed on an Agilent 7890A gas chromatograph equipped with a 5975 mass spectrometer and an NCI detector.
X-ray crystallography: Colorless crystals were mounted on α MiteGen MicroMesh by using a small amount of Cargille Immersion oil. Data were collected on a Bruker three-circle platform diffractometer equipped with a SMART APEX II CCD detector. The crystals were irradiated by using graphite-monochromated MoKα radiation (λ = 0.71073). An Oxford Cobra low-temperature device was used to maintain the crystals at a constant 150(2) K during data collection. Data collection was performed, and the unit cell was initially refined by using APEX2 (v2010.3–0). Data reduction was performed using SAINT (v7.68 A) and XPREP (v2008/2). Corrections were applied for Lorentz, polarization, and absorption effects by using SADABS (v2008/1). The structure was solved and refined with the aid of the programs in the SHELXTL-plus (v2008/4) system of programs. The full-matrix least squares refinement on F2 included atomic coordinates and anisotropic thermal parameters for all non-hydrogen atoms.
4.1. Nitration of BNN with AcONO2
A flask equipped with a thermometer, gas inlet, and stir bar was charged with BN-naphthalene [5] (0.22g, 1.7 mmol), anhydrous CH3CN (7 mL), and AgNO3 (0.79 g, 4.7 mmol). With vigorous stirring, acetyl chloride (0.42 g, 5.4 mmol) in 1 mL of anhydrous CH3CN was added dropwise at 10 °C. The reaction temperature did not change during the addition. The nitration mixture was warmed to 20 °C and stirred for 2.0 h. The precipitated AgCl was removed by filtration through a layer of Celite and the solvent was removed in vacuo. The oily residue was diluted with CH2Cl2 (20 mL), quickly washed with water (2 × 3 ml), and dried over MgSO4. After filtration and concentration, the residue was loaded onto a silica gel column chromatograph and eluted with a 2:1 CH2Cl2 and hexane solvent mixture. The first fraction was identified as compound 3-Nitro-BN-naphthalene (3) [31] (Rf = 0.57), the second was 1-Nitro-BN-naphthalene (4) (Rf = 0.37), the third was 3,6-Dinitro-BN-naphthalene (5) (Rf = 0.26), and the fourth was 1,6-Dinitro-BN-naphthalene (6)(Rf = 0.17). Compound 1,8-Dinitro-BN-naphthalene (7) (Rf = 0.09) was eluted with CH2Cl2. The total yield was 37%.
3-Nitro-BN-naphthalene (3), white solid, 50.0 mg (17%), mp. 78–79 °C. 1H NMR (CD2Cl2): δ 9.14 (s, 1H), 8.41 (d, J = 12.5 Hz, 1H), 7.92 (d, J = 6.9 Hz, 1H), 7.79 (q, J = 11.0, 6.5 Hz, 1H), 7.49 (q, J = 12.3, 11.0 Hz, 2H), 6.92 (t, J = 6.7 Hz, 1H). 13C NMR (CD2Cl2): δ 140.75, 138.96 (C-NO2), 136.05, 134.13, 132.23, 131 (b, C-B), 117.08. 11B NMR (CD2Cl2) δ 29.52. GC-MS m/z (EI): 174.2 (m, 100%), 173.2 (22%). IR (film) 1627, 1537, 1515, 1473, 1399, 1336, 748 cm−1.
1-Nitro-BN-naphthalene (4), white solid, 25.0 mg (8%), mp. 124–126 °C. 1H NMR (CD2Cl2): δ 8.56 (d, J = 7.6 Hz, 1H), 8.18 (d, J = 6.8 Hz, 1H), 7.93 (d, J = 11.3 Hz, 1H), 7.91 (d, J = 7.1 Hz, 1H), 7.86 (q, J = 11.4, 6.3 Hz, 1H), 6.91 (t, J = 6.8, 1.3 Hz, 1H), 6.84 (t, J = 7.3 Hz, 1H). 13C NMR (CD2Cl2): δ 140.97, 140.92, 137.40, 133.59, 131.0 (b, C-B), 116.14, 110.71. 11B NMR (CD2Cl2) δ 25.63. GC-MS m/z (EI): 174.1 (m, 100%), 173.1 (27%). IR (film) 1617, 1502, 1333, 1313, 1232, 751cm−1.
3,6-Dinitro-BN-naphthalene (5), white solid, 8 mg (2%), mp. 191–192 °C. 1H NMR (CD2Cl2): δ 9.21 (s, J = 1.5 Hz, 2H), 8.51 (d, J = 12.5 Hz, 2H), 7.65 (d, J = 12.5 Hz, 2H). 13C NMR (CD2Cl2): δ 140.84 (C-NO2), 135.81, 134.50, 133 (b, C-B). 11B NMR (CD2Cl2) δ 29.91. GC-MS m/z (EI): 219.0 (m, 100%), 218.0 (23%). HRMS m/z = 219.0459, corresponding to dinitro-BNN: C8H6BN3O4, calculated 219.0451. IR (film) 1625, 1529, 1337, 1240, 1110, 835 cm−1.
1,6-Dinitro-BN-naphthalene (6), white solid, 22 mg (6%), mp. 179–180 °C. 1H NMR (CD2Cl2): δ 9.17 (s, 1H), 8.68 (d, J = 7.5 Hz, 1H), 8.58 (d, J = 12.5 Hz, 1H), 8.25 (d, J = 7.0 Hz, 1H), 8.12 (d, J = 12.5 Hz, 1H), 7.05 (dd, J = 7.0, 7.5 Hz, 1H). 13C NMR (CD2Cl2): δ 140.83, 140.11, 139.52, 135.09, 134.76, 132.0 (b, C-B), 113.63. 11B NMR (CD2Cl2) δ 26.64. GC-MS m/z (EI): 219.0 (m, 100%), 218.0 (29%). IR (film) 1625, 1529, 1337, 1240, 1110, 835, 800 cm−1.
1,8-Dinitro-BN-naphthalene (7), white solid, 15 mg (4%), mp. 250 °C (decomp). 1H NMR (CD2Cl2): δ 8.33 (d, J = 7.5 Hz, 2H), 8.13 (d, J = 7.0 Hz, 2H), 6.97 (dd, J = 7.0, 7.5 Hz, 2H). 13C NMR (CD2Cl2): δ 138.66, 137.64, 112.64. 11B NMR (CD2Cl2) δ 22.58. GC-MS m/z (EI): 219.0 (m, 100%), 218.0 (29%). IR (film) 1618, 1500, 1363, 809, 744, 693 cm−1.
4.2. Nitration of BNN with NO2BF4
A flask equipped with a gas inlet and a stir bar was charged with BN-naphthalene (9.0 mg, 0.07 mmol) and anhydrous CH3CN (0.5 mL). The mixture was stirred in a water bath (10 °C), and NO2BF4 (28 mg, 0.21 mmol) was added in one portion. The reaction mixture was warmed to 20 °C and stirred at the temperature for 2 h. The reaction mixture was concentrated in vacuo at 20 °C, and the residue was diluted with 1.0 mL of ice water. The products were extracted with methylene chloride, washed with water, and dried over MgSO4. The drying reagent was filtered, and the filtrated product was passed through a layer of silica gel. The solvent was removed in vacuo to give a solid, 6.0 mg. The NMR spectrum showed that the mixture only contained 3,6-dinitro- (5) and 1,6-dinitro-BNN (6). The total yield was about 39%.
4.3. Nitration of BNN with One Equivalent of AcONO2 in CH3CN
Under an argon atmosphere, AcCl (6.6 mg, 0.084 mmol) in 0.29 mL of anhydrous CH3CN was added dropwise via a syringe to a solution of BNN (10 mg, 0.078 mmol) and AgNO3 (13.1 mg, 0.078 mmol) in 0.2 mL of anhydrous CH3CN at 10 °C. After the addition was completed, the water bath was warmed to 20 °C, and the reaction mixture was stirred for 2.0 h. The solvent was removed in vacuo at 20 °C, and the residue was diluted with 2 mL of CH2Cl2 and washed with water (2 × 0.5 mL). The organic phase was dried over MgSO4. The drying reagent was filtered, and the filtrate was treated by passing through a layer of silica gel, eluted with CH2Cl2. Removal of the solvent gave an oily mixture, 13 mg. The mixture was dissolved in CD2Cl2 for NMR analysis (Figure 1a).
4.4. Nitration of BNN with One Equivalent of NO2BF4 in CH3CN
Under an argon atmosphere, NO2BF4-CH3CN solution, prepared from 100 mg of purified NO2BF4 in 2.73 g of anhydrous CH3CN (0.218g, 0.058 mmol of NO2BF4, assume the purity of NO2BF4 is 100%), was added dropwise from a syringe to a solution of BNN (7.0 mg, 0.054 mmol) in 0.2 mL of anhydrous CH3CN at 10 °C. After the addition was completed, the reaction mixture was warmed to 20 °C, and the reaction mixture was stirred for 2.0 h. The solvent was removed in vacuo at 20 °C. The residue was diluted with 2 mL of CH2Cl2 and washed with water (2 × 0.5 mL). The organic phase was dried over MgSO4, the drying reagent was filtered, and the filtrate was treated by passing through a layer of silica gel and eluted with CH2Cl2. Removal of the solvent gave an oily mixture, 8 mg. The mixture was dissolved in CD2Cl2 for NMR analysis (Figure 1b).
Supplementary Materials
1H, 13C, and 11B NMR and IR spectroscopy of compounds 3–7, and X-ray crystallographic analysis data (bond lengths and bond angles of 4, 6, and 7) are available in the supporting information.
Author Contributions
Chemistry, M.-X.Z.; N.B.Z. and P.F.P.; computing calculation, B.A.S., and I.-F.K.; crystallography, G.H.I. and D.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the US AFOSR (Award No. F1ATA02052G001), DoD/DOE Joint Munitions Program and the Office of Naval Research (Award No. N00014-13-AF-0-0002).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
This work was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory under Contract DE-AC52-07NA27344. We are grateful for financial support from the US AFOSR DoD/DOE Joint Munitions Program and the Office of Naval Research for financial support. Adele F. Panasci and Keith R. Coffee for HRMS analysis are also appreciated.
Conflicts of Interest
There are no conflict to declare.
Sample Availability
Samples of the compounds 3, 4, 5, 6, and 7 are not available from the authors.
References
- Yang, D.-T.; Zheng, J.; Peng, J.-B.; Wang, X.; Wang, S. Sequential and Diverse Synthesis of BN-Heterocycles and Investigation of Their Photoreactivity. J. Org. Chem. 2021, 86, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chen, W.; Qiao, Y.; Lu, X.; Zhou, G. BN-Embedded Polycyclic Aromatic Hydrocarbon Oligomers: Synthesis, Aromaticity, and Reactivity. Angew. Chem. Int. Ed. 2020, 59, 7122–7130. [Google Scholar] [CrossRef] [PubMed]
- Abengozar, A.; Sucunza, D.; Garcia-Garcia, P.; Perez-Redondo, A.; Vaquero, J.J.; Sampedro, D.A. New Member of the BN-Phenanthrene Family: Understanding the Role of the B-N Bond Position. J. Org. Chem. 2019, 84, 7113–7122. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, F.-D.; Sun, Z.-H.; Yao, Z.-F.; Chen, Q.-R.; Huang, Z.; Yang, J.-H.; Wang, J.-Y.; Pei, J. BN-Embedded Tetrabenzopentacene: A Pentacene Derivative with Improved Stability. Angew. Chem. Int. Ed. 2019, 58, 10708–10712. [Google Scholar] [CrossRef] [PubMed]
- Van de Wouw Heidi, L.; Klausen, R.S. BN Polystyrenes: Emerging Optical Materials and Versatile Intermediates. J. Org. Chem. 2019, 84, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Li, Z.; Xu, Y.; Liang, J.; Lin, C.; Wei, J.; Wang, Y. Achieving Efficient Blue Delayed Electrofluorescence by Shielding Acceptors with Carbazole Units. ACS Appl. Mater. Interfaces 2019, 11, 28096–28105. [Google Scholar] [CrossRef] [PubMed]
- Fukagawa, H.; Oono, T.; Iwasaki, Y.; Hatakeyama, T.; Shimizu, T. High-efficiency ultrapure green organic light-emitting diodes. Mater. Chem. Front. 2018, 2, 704–709. [Google Scholar] [CrossRef]
- Yoshida, K.; Osuka, A. Pyrrole-Modified Subporphyrins Bearing a Sulfur-Containing Heterocyclic Unit. Helv. Chim. Acta 2018, 101, e1800025. [Google Scholar] [CrossRef]
- Min, Y.; Dou, C.; Tian, H.; Geng, Y.; Liu, J.; Wang, L. n-Type Azaacenes Containing B→N Units. Angew. Chem. Int. Ed. 2018, 57, 2000–2004. [Google Scholar] [CrossRef]
- Wang, T.; Dou, C.; Liu, J.; Wang, L. Effects of the Substitutes of Boron Atoms on Conjugated Polymers Containing B←N Units. Chem. Eur. J. 2018, 24, 13043–13048. [Google Scholar] [CrossRef]
- Cassidy, S.J.; Brettell-Adams, I.; McNamara, L.E.; Smith, M.F.; Bautista, M.; Cao, H.; Vasiliu, M.; Gerlach, D.L.; Qu, F.; Hammer, N.I.; et al. Boranes with Ultra-High Stocks Shift Fluorescence. Organometallics 2018, 37, 3732–3741. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Wang, J.-Y.; Pei, J. BN Heterosuperbenzenes: Synthesis and Properties. Chem. Eur. J. 2015, 21, 3528–3539. [Google Scholar] [CrossRef]
- Min, Y.; Cao, X.; Tian, H.; Liu, J.; Wang, L. B←N Incorporated Dibenzoazaacene with Selective Near-Infrared Absorption and Visible Transparency. Chem. Eur. J. 2021, 27, 2065–2071. [Google Scholar] [CrossRef]
- Zhao, R.; Min, Y.; Dou, C.; Lin, B.; Ma, W.; Liu, J.; Wang, L. A Conjugated Polymer Containing a B←N Unit for Unipolar n-Type Organic Field-Effect Transistors. ACS Appl. Polym. Mater. 2020, 2, 19–25. [Google Scholar] [CrossRef] [Green Version]
- Muhammad, X.H.; Younis, U.; Qie, Y.; Aftab, W.; Sun, Q. A BN analog of two-dimensional triphenylene-graphdiyne: Stability and properties. Nanoscale 2019, 11, 9000–9007. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, J.S.A.; Darrigan, C.; Chrostowska, A.; Li, B.; Liu, S.-Y. A BN anthracene mimics the electronic structure of more highly conjugated systems. Dalton Trans. 2019, 48, 2807–2812. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, T.; Hashimoto, S.; Seki, S.; Nakamura, M. Synthesis of BN-fused Polycyclic Aromatics via Tandem Intramolecular Electrophilic Arene Borylation. J. Am. Chem. Soc. 2011, 133, 18614–18617. [Google Scholar] [CrossRef] [PubMed]
- Palomino-Ruiz, L.; Rodriguez-Gonzalez, S.; Fallaque, J.G.; Marquez, I.R.; Agrait, N.; Diaz, C.; Leary, E.; Cuerva, J.M.; Campana, A.G.; Martin, F.; et al. Single-Molecule Conductance of 1,4-Azaborine Derivatives as Models of BN-doped PAHs. Angew. Chem. Int. Ed. 2021, 60, 6609–6616. [Google Scholar] [CrossRef]
- Maar, R.R.; Katzman, B.D.; Boyle, P.D.; Staroverov, V.N.; Gilroy, J.B. Cationic boron formazanate dyes. Angew. Chem. Int. Ed. 2021, 60, 5152–5156. [Google Scholar] [CrossRef]
- Chen, M.; Unikela, K.S.; Ramalakshmi, R.; Li, B.; Darrigan, C.; Chrostowska, A.; Liu, S.-Y. A BN-doped cycloparaphenylene debuts. Angew. Chem. Int. Ed. 2021, 60, 1556–1560. [Google Scholar] [CrossRef]
- Zhang, Q.; Sun, Z.; Zhang, L.; Li, M.; Zi, L.; Liu, Z.; Zhen, B.; Sun, W.; Liu, X. Synthesis, Structures, and Properties of BN-Dinaphthothiophenes: Influence of B and N Placement on Photophysical Properties and Aromaticity. J. Org. Chem. 2020, 85, 7877–7883. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.D.; Wang, J.Y.; Pei, J. Recent Progress and Challenges in the Synthesis of Boron-nitrogen Fused Ploycyclic Aromatic Hydrocarbons. Sci. Sin. Chim. 2020, 50, 1205–1216. [Google Scholar]
- Tsuchiya, S.; Saito, H.; Nogi, K.; Yorimitsu, H. Aromatic Metamorphosis of Indoles into 1,2-Benzazaborins. Org. Lett. 2019, 21, 3855–3860. [Google Scholar] [CrossRef]
- Baranac-Stojanovic, M. A DFT Study of the Modulation of the Antiaromatic and Open-Shell Character of Dibenzo[a,f]pentalene by Employing Three Strategies: Additional Benzoannulation, BN/CC Isosterism, and Substitution. Chemistry 2019, 25, 9747–9757. [Google Scholar] [CrossRef]
- Anafcheh, M.; Ahmadi, E.; Zahedi, M. Addition of borazine to boron nitride nanotubes: [2+2] cycloaddition or bond cleavage. Mon. Chem. 2019, 150, 1019–1024. [Google Scholar] [CrossRef]
- van de Wouw, H.L.; Awuyah, E.C.; Baris, J.I.; Klausen, R.S. An Organoborane Vinyl Monomer with Styrene-like Radical Reactivity: Reactivity Ratios and Role of Aromaticity. Macromolecules 2018, 51, 6359–6368. [Google Scholar] [CrossRef]
- Zhu, C.; Ji, X.; You, D.; Chen, T.L.; Mu, A.U.; Barker, K.P.; Klivansky, L.M.; Liu, Y.; Fang, L. Extraordinary Redox Activities in Ladder-Type Conjugated Molecules Enabled by B←N Coordination-Promoted Delocalization and Hyperconjugation. J. Am. Chem. Soc. 2018, 140, 18173–18182. [Google Scholar] [CrossRef] [PubMed]
- Abengózar, A.; Fernández-González, M.A.; Sucunza, D.; Frutos, L.M.; Salgado, A.; García-García, P.; Vaquero, J.J. C−H Functionalization of BN-Aromatics Promoted by Addition of Organolithium Compounds to the Boron Atom. Org. Lett. 2018, 20, 4902–4906. [Google Scholar] [CrossRef] [PubMed]
- Schraff, S.; Sun, Y.; Pammer, F. Tuning of Electronic Properties Vialabile N-B-coordiation in Conjugated Organoboranes. J. Mater. Chem. C 2017, 5, 1730–1741. [Google Scholar] [CrossRef]
- Dewar, M.J.S.; Kubba, V.P. New heteroaromatic compounds. IV. Nitration and chlorination of 10-methyl-10,9-borazarophenanthrene. Tetrahedron 1959, 7, 213–222. [Google Scholar] [CrossRef]
- Zhang, Y.; Dan, W.; Fang, X. Metal nitrate mediated regioselective nitration of BN-substituted arenes. Organometallics 2017, 36, 1677–1680. [Google Scholar] [CrossRef]
- Dewar, M.J.S.; Gleicher, G.J.; Robinson, B.P. Synthesis and Nuclear Magnetic Resonance Spectrum of 10,9-Borazanaphthalene. J. Am. Chem. Soc. 1964, 86, 5698–5699. [Google Scholar] [CrossRef]
- Olah, G.; Narang, S.; Olah, J.; Lammertsma, K. Recent aspects of nitration: New preparative methods and mechanistic studies (a review). Proc. Nat. Acad. Sci. USA 1982, 79, 4487–4494. [Google Scholar] [CrossRef] [Green Version]
- Olah, G.A.; Narang, S.C.; Olah, J.A. Nitration of naphthalene and remarks on the mechanism of electrophilic aromatic nitration. Proc. Nat. Acad. Sci. USA 1981, 78, 3298–3300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, F.A.; Dewar, M.J.S.; Jones, R. New heterocyclic compounds. XXVII. Boron-11 chemical shifts of some heterocyclic boron compounds. J. Am. Chem. Soc. 1968, 31, 706–708. [Google Scholar] [CrossRef]
- Hermanek, S. B-11 NMR spectra of boranes, main-group heteroboranes, and substituted derivatives. Factors influencing chemical shifts of skeletal atoms. Chem. Rev. 1992, 92, 324–362. [Google Scholar] [CrossRef]
- Michl, J. Borazaro analogues of aromatic hydrocarbons. II. Semiempirical calculations. Collect. Czechoslov. Chem. Commun. 1971, 36, 1248–1278. [Google Scholar] [CrossRef]
- Baranac-Stojanovic, M.; Stojanovic, M. Does aromaticity account for an enhanced thermodynamic stability? The case of monosubstituted azaborines and the stereoelectronic chameleonism of the NH2 group. Phys. Chem. Phys. 2019, 21, 9465–9476. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Yang, H.; Kampf, J.W.; Banaszak, H.; Mark, M.; Ashe, A.J., III. Synthesis of Ring-Fused B-N Heterocyclic Compounds. Organometallics 2006, 25, 513–518. [Google Scholar] [CrossRef]
- Pejlovas, A.M.; Daly, A.M.; Ashe, A.J., III; Kukolich, S.G. Microwave spectra, molecular structure, and aromatic character of 4a,8a-azaboranaphthalene. J. Chem. Phys. 2016, 144, 114303–114310. [Google Scholar] [CrossRef]
- Stojanovic, M.; Baranac-Stojanovic, M. Mono BN-substituted analogues of naphthalene: A theoretical analysis of the effect of BN position on stability, aromaticity, and frontier orbital energies. New J. Chem. 2018, 42, 12968–12976. [Google Scholar] [CrossRef] [Green Version]
- Krygowski, T.M.; Szatylowicz, H.; Stasyuk, O.A.; Dominikowska, J.; Palusiak, M. Aromaticity from the viewpoint of molecular geometry: Application to planar systems. Chem. Rev. 2014, 114, 6383–6422. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).