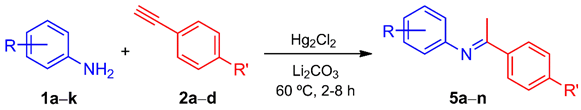
Abstract
An efficient synthesis of ketimines was achieved through a regioselective Hg(I)-catalyzed hydroamination of terminal acetylenes in the presence of anilines. The Pd(II)-catalyzed cyclization of these imines into the 2-substituted indoles was satisfactorily carried out by a C-H activation. In a single-step approach, a variety of 2-substituted indoles were also generated via a Hg(I)/Pd(II)-catalyzed, one-pot, two-step process, starting from anilines and terminal acetylenes. The arylacetylenes proved to be more effective than the alkyl derivatives.
1. Introduction
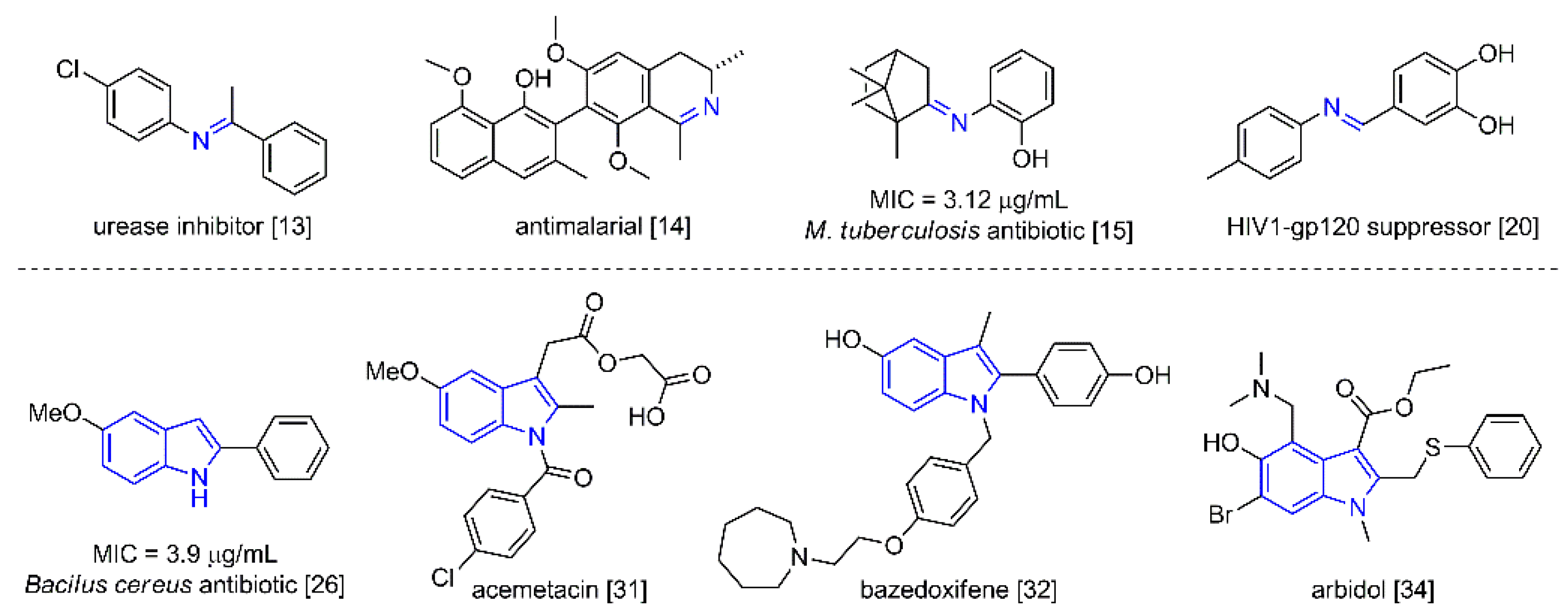
Imines are one of the most versatile functional groups in synthesis. Numerous studies have focused on their preparation [1,2,3,4,5], chemical structure and tautomeric or transimination equilibrium [6,7,8], and use as a precursor for the construction of more complex structures [9,10,11,12]. Due to their versatility, they play an important role in medicinal chemistry, serving as urease and protease inhibitors [13] as well as antimalarial, antibacterial, and antifungal agents [14]. They also exhibit activity against Mycobacterium tuberculosis [15] and tumor cells [16,17,18,19], and act as suppressors of HIV1-gp120, related to the human immunodeficiency virus (HIV) [20] (Figure 1). Interestingly, imines are present in phytoplankton and shellfish as toxins [21].
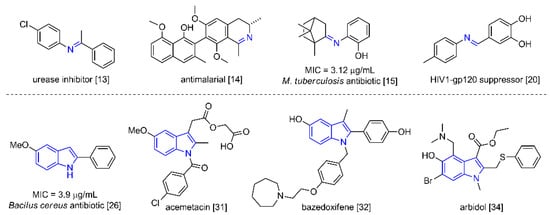
Figure 1.
Imine- and indole-containing compounds with pharmacological activity.
Likewise, indoles are widely distributed in natural products [22,23,24]. They occupy a prominent place as pharmacologically active compounds [25,26,27,28], including commercial therapeutic chemicals and active pharmaceutical ingredients [29,30,31]. The U.S. Food and Drug Administration (FDA) has approved at least 43 indole-containing drugs [32,33]. For example, arbidol has shown efficient in vitro activity against the SARS-CoV-2 infections [34] and has been administered to infected patients [35,36,37] (Figure 1). Hence, the molecular structures of imines and indoles are of great value for medicinal and synthetic purposes.
Despite the variety of methods for the preparation of imines [1,2,3,4,5], their synthesis is mainly carried out with conventional procedures, such as the acid-catalyzed condensation of a carbonyl compound with a primary amine [1]. Among the relevant methodologies are the reduction of nitriles, oximes, and aliphatic nitro derivatives [38,39,40,41], the addition of an organometallic reagent to nitriles and amides [42,43], and the oxidation of alcohols and amines [44,45]. These methods are only practical, however, if there is an existing carbon–nitrogen formal bond in the substrate.
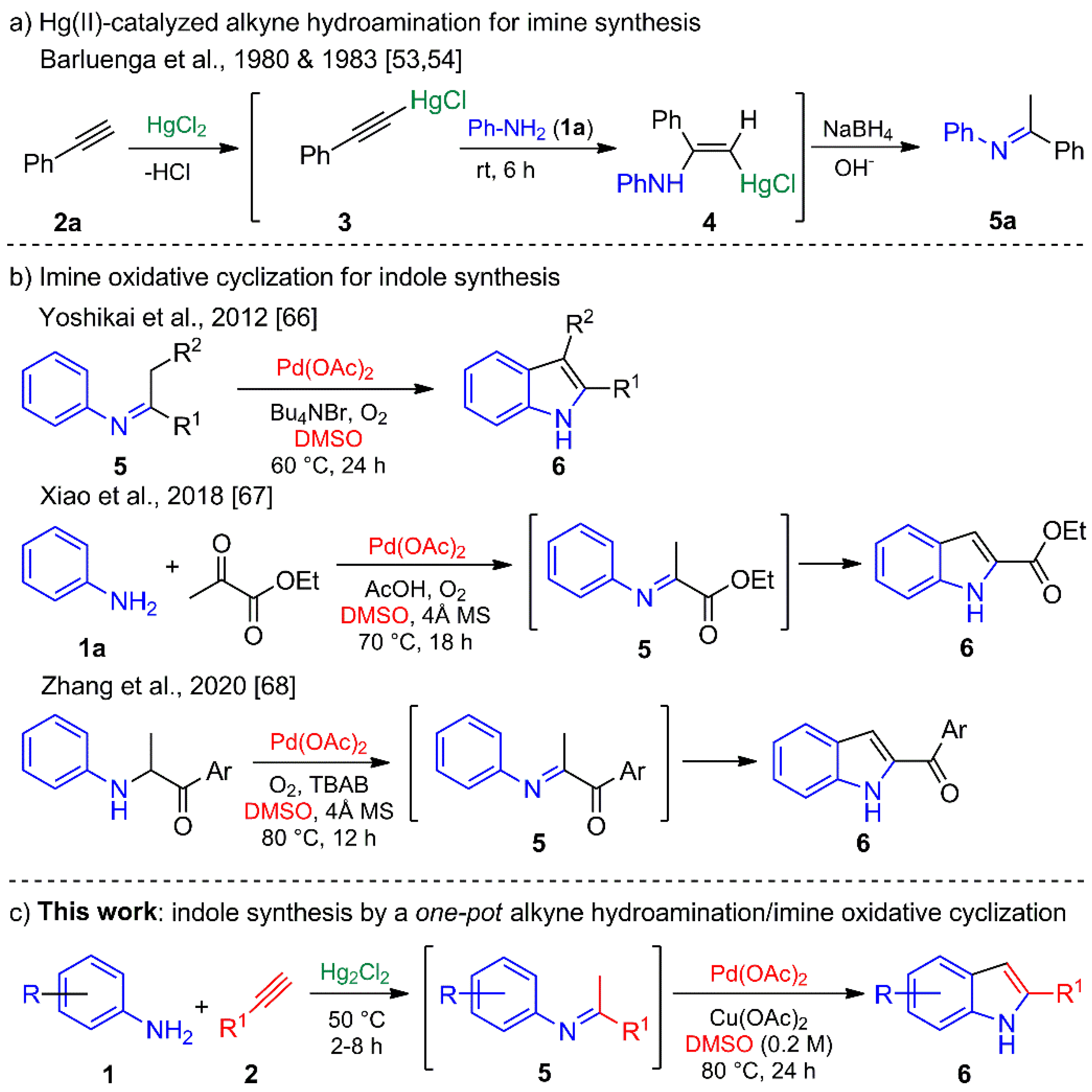
Intermolecular imine is usually synthesized by condensation reactions with an azeotropic agent [1], a desiccant [46], or a water scavenger [47] to remove water. Alkyne hydroamination arose as an alternative, being an attractive and elegant pathway with complete atom economy [48,49,50,51]. Barluenga et al. described several catalytic and noncatalytic hydroamination methods with terminal acetylenes (e.g., 2a) promoted by Tl(III) or Hg(II) salts [52]. They found that HgCl2 acts through a nucleophilic attack of the aniline (1a) on the internal acetylene carbon, firstly forming terminal acetylene-HgCl complex 3 followed by its possible conversion into the aminoalkenylmercurial(II) chloride intermediate 4. The latter regenerates the Hg(II) catalyst by protonolysis to give the enamine, which is converted into the desired imine 5a (Scheme 1a) [53,54].
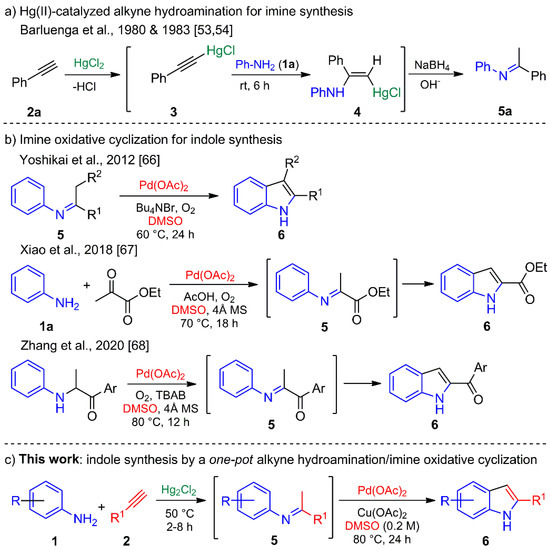
Scheme 1.
(a) Imine synthesis by a Hg(II)-promoted alkyne hydroamination. (b) Indole synthesis from imines derived by a condensation reaction and Pd(II)-catalyzed oxidative cyclization. (c) Pd(II)-catalyzed indole synthesis from imines derived from Hg(I)-catalyzed alkyne hydroamination.
In contrast, indoles have been elaborated by diverse protocols [55], such as the well-known Fischer [56,57], Julia [58], Bartoli [59], and Gassman [60] procedures, each of which include a sigmatropic rearrangement at a high temperature. Other efficient approaches have involved organometallic reagents, as with the syntheses of indoles described by Castro [61], Larock [62], Fagnou [63], and Söderberg [64,65]. Most of them demand the use of ortho-halogenated and ortho-vinylated N-protected anilines or nitrobenzenes to build the indole A ring. Recently, imine substrates have proven valuable for indole synthesis, as demonstrated by the groups of Yoshikai [66], Xiao [67], and Zhang [68] (Scheme 1b).
Our group has an ongoing interest in designing and carrying out the novel synthesis of heterocycles [69,70,71], and in particular of aza-heterocycles and indoles [72,73,74,75]. Accordingly, the aim of the present study was to assess the feasibility of preparing indoles in a one-pot process, involving the regioselective Hg(I)-catalyzed hydroamination of terminal alkynes to produce imines as intermediates, and their subsequent Pd(II)-catalyzed cross-coupling oxidative cyclization to afford the corresponding C-2-substituted indoles (Scheme 1c).
2. Results and Discussion
2.1. Synthesis of Imines 5a–n by Hg(I)-catalyzed Hydroamination of Alkynes 2a–d with Anilines 1a–k
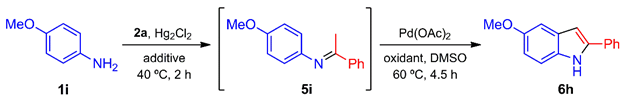
The properties of Hg(0) and Hg(II) salts have been examined [76], and these species have many chemical and industrial applications [77,78,79], including chemical transformations [80,81,82]. Curiously, the use of Hg(I) in organic synthesis is surprisingly rare and limited [83,84]. Our group has reported the inclusion of Hg2Cl2 as the catalyst for the solvent-free hydroamination of phenylacetylene (2a) with m-anisidine (1e) to furnish the corresponding imine 5e in quantitative yield [85]. Following up on that effort, the scope of the method was herein explored by varying the structure of anilines (1) and acetylenes (2) to develop an efficient synthesis of imines (5). Unfortunately, when employing the previously described reaction conditions (room temperature, 24 h), the yields of the resulting imines depended on the anilines (Table 1). For example, with p-anisidine (1i) and 2a, imine 5i was furnished in a modest yield (61%, entry 2). The yield was improved by increasing the reaction time (82%, entry 3) or by raising the temperature to 45 ºC with a reaction time of 4 h (87%, entry 4).

Table 1.
Optimization of the preparation of N-arylimines 5e and 5i a.
Other parameters were evaluated, such as solvent, catalyst, and additive. Although solvent-free reaction conditions are always recommended for designing an optimal green chemistry methodology [86], three chlorinated solvents (chloroform, 1,2-dichloroethane, and methylene chloride), THF, and DMSO were assessed by heating at 60 ºC for 2–12 h. Whereas imine 5i was obtained in fairly low yields (21–41%) with either the first or the third solvent, it was not detected with 1,2-dichloroethane, THF, and DMSO. When Ag2CO3 (Table 1, entry 5) and CuCl (not shown) were tested as the catalysts for 1e and 2a, the outcome was the recovery of the reactants but no trace of imine 5e.
Imines, usually water-sensitive, are converted into ketones by hydrolysis. For example, acetophenone (7a) can be formed from imines 5e or 5i. Therefore, anhydrous lithium carbonate was employed as the additive, which greatly improved the yield (Table 1, entries 6 and 7). Of course, under these optimal reaction conditions, the presence of Hg2Cl2 was essential for the transformation to take place. Due to a feasible light-induced Hg–Hg bond disproportionation [87], the generation of radicals was contemplated. Consequently, hydroquinone was added as a radical quencher, which did not modify the yield (entry 8) and thus ruled out this possibility.
In agreement with the findings of Barluenga et al. [52,53,54], the process was also catalyzed with HgCl2 under the same reaction conditions, but a lower yield (78%) was observed (Table 1, entry 9). This suggests that Hg(I) and Hg(II) are both probably the catalytic species involved in the process. Considering the feasible generation of Hg(0) as a secondary product during the decomposition of Hg2Cl2 under an analogous reaction [53,54], the process was carried out in the presence of Hg(0), but no imine was found (entry 10). Hence, the participation of Hg(0) as a catalyst is unlikely. Moreover, there was no visual evidence of a dark-silver mirror residue in the Hg(I) trials. Actually, Hg(0) is a well-known poisoning catalyst in heterogeneous/homogeneous reactions [88].
To gain insights into the mechanism, 2a was reacted with Hg2Cl2 to examine its possible conversion into acetophenone (7a) as an intermediate. After heating at 45 ºC for 1 h, however, only the recovery of 2a was achieved. Taking into account the potential formation of intermediate 7a in the middle of the reaction, a further trial was performed under the same reaction conditions (45 ºC for 1 h) and in the presence of 1i and Hg2Cl2. The result was again the recovery of the staring material. Finally, after reacting a mixture of 1i and Hg2Cl2 at 45 ºC for 1 h, no N-Hg complex was detected.
Once the reaction conditions were optimized (Table 1, entry 7), evaluation was performed for a series of anilines (1a–k) bearing substituents with diverse electron-demand at the three positions of the benzene ring, utilizing arylalkynes 2a–d as the terminal acetylenes (Table 2). In general, neither the electron-demand of the substituents nor their position in the benzene ring of the aniline showed any effect on the efficacy of the process. The only exception was the use of the halogenated anilines 1f and 1k, which gave rise to the corresponding imines in modest yields (Table 2, entries 6 and 11).

Table 2.
Preparation of the series of N-arylimines 5a–n through the Hg(I)-catalyzed hydroamination of 2a–d with anilines 1a–k a.
The high yields afforded by the unsubstituted phenylacetylene (2a) were also obtained with the terminal para-substituted phenylacetylenes 2b–d (Table 2, entries 9 and 12–14), supporting the idea that Hg(I) easily interacts with the triple bond regardless of the electron-demand of its substituent. Furthermore, the reaction was successful with terminal alkyl- and some aryl-acetylenes, including 4-cyanophenylacetylene (2c), propylacetylene (2e), cyclopropylacetylene (2f), and cyclohexylacetylene (2g), judging by the 1H NMR and MS/GC analyses of the crude mixtures. Nevertheless, the corresponding imines 5m (as a mixture of 5m/p-cyanoacetophenone (7b) (95:5)) and 5o–q were not stable enough to be isolated under the extraction conditions. Evidence of their formation was found unambiguously by the synthesis of the indoles (see Scheme 3).
Reactivity significantly decreased with the di-substitution of the triple bond. For instance, neither methylpropylacetylene (2h) nor phenylpropylacetylene (2i) reacted with p-anisidine (1i), even when the mixture was heated to 80 ºC for 53 h, probably because both electronic and steric effects impede the addition of the aniline or do not allow for the formation of the σ-Hg(I)-acetylene complex (see below).
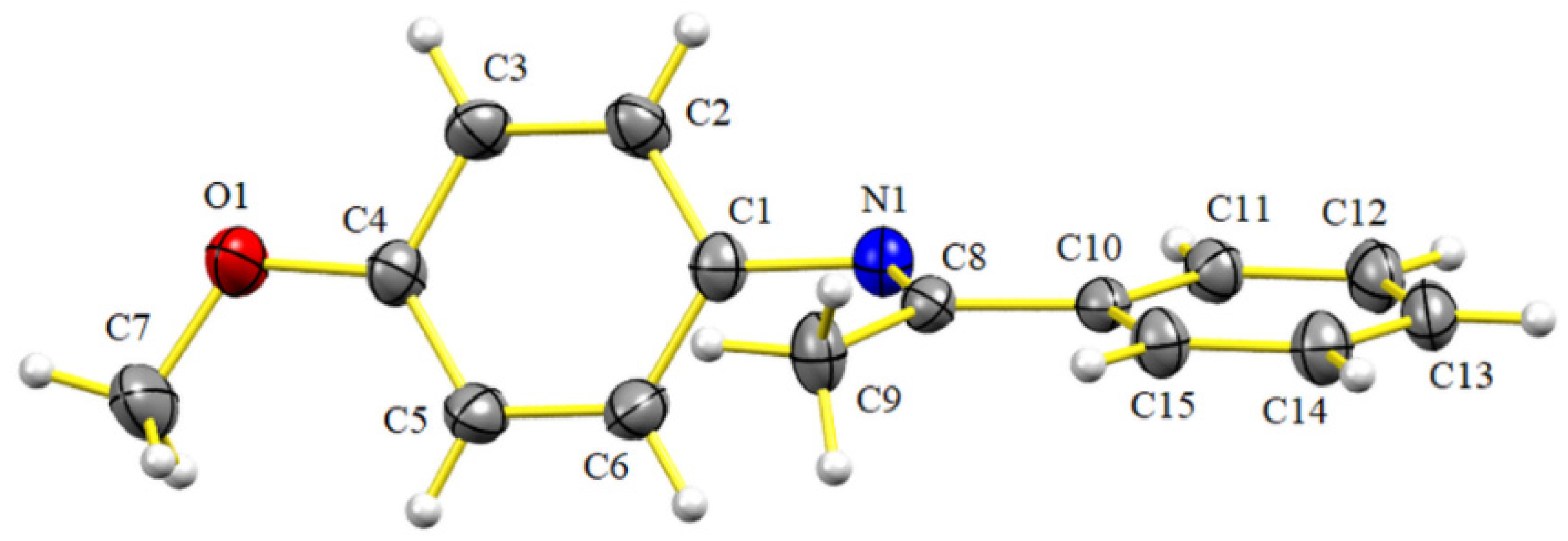
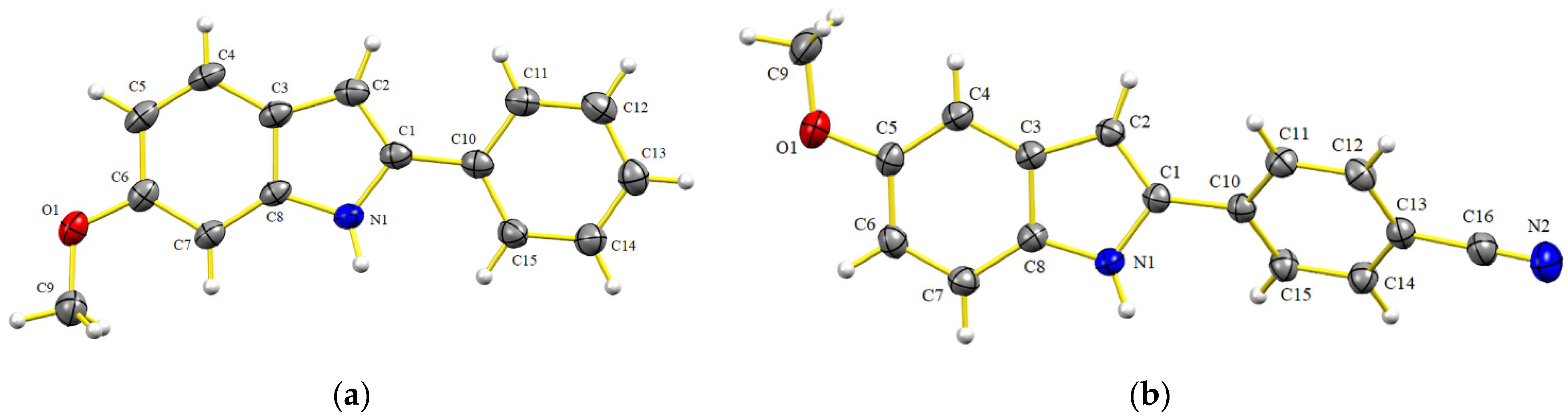
All the imines prepared by this method were characterized by IR and NMR (1D and 2D), and in some cases, HRMS. The latter technique was performed in case of finding a discrepancy between the data reported in the literature and the melting point or physical state of the compound in the current effort. Imine 5i was crystallized in order to be subjected to X-ray crystallographic analysis (Figure 2), which demonstrated the expected E configuration of the double bond. However, the N-anisyl ring was not coplanar to the imine double bond, instead adopting a quasi-orthogonal conformation (torsion angle, C(8)-N(1)-C(1)-C(2) = 115.31(17), see Supplementary Materials), which indicates that the π-system is not conjugated. In contrast, the phenyl ring attached to the imine double bond displayed a planar conformation (torsion angle, C(11)-C(10)-C(8)-N(1) = –4.7(2), see Supplementary Materials).
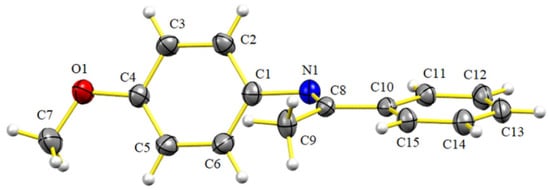
Figure 2.
Structure of 5i, as determined by single X-ray diffraction crystallography (ellipsoids at the 30% probability level).
2.2. One-Pot Synthesis of Indoles 6a–p by Subsequent Hg(I)- and Pd(II)-Catalyzed Conversion of Anilines 1a–g and 1i–k with Alkynes 2a–g
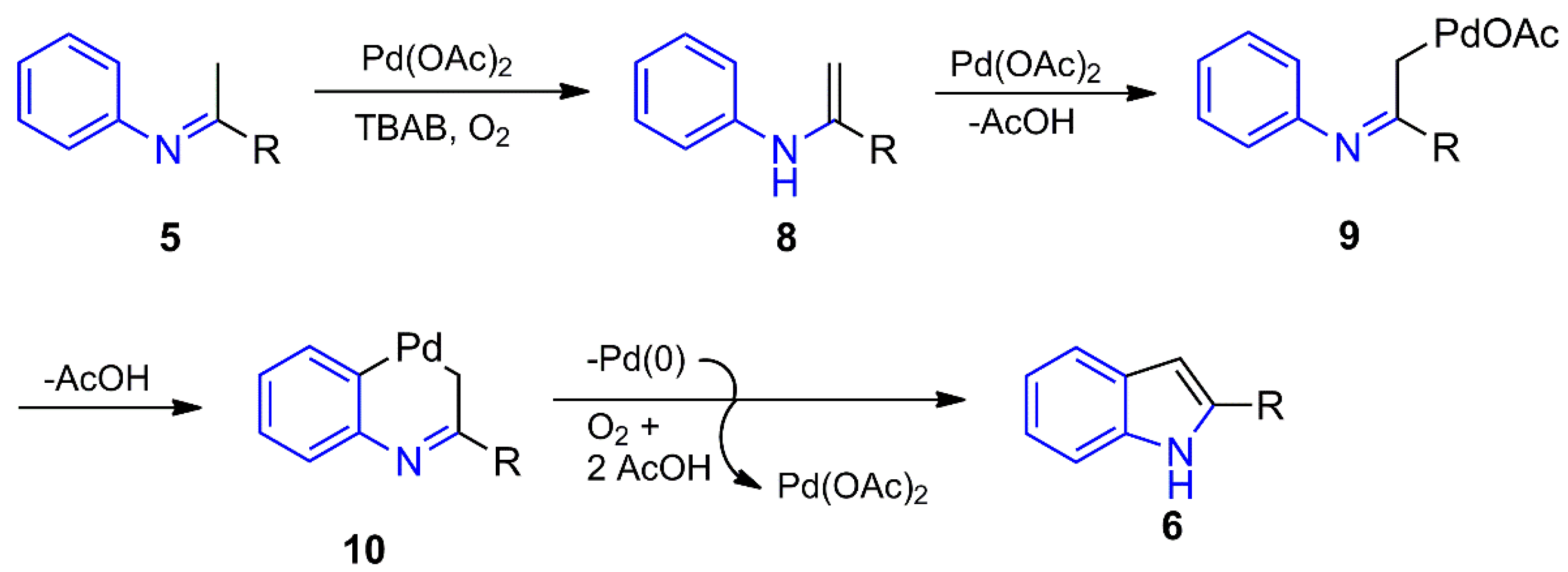
The Pd(II)-catalyzed conversion of imines/enamines into indoles has been performed by fairly similar methods [89,90,91], previously forming imine 5 or generating it in situ as the intermediate [66,67,68] (Scheme 1b). In all the approaches, the mechanism seems to start from the tautomerization of imine 5 to enamine 8, which undergoes a dehydrogenative reaction by the metal to furnish the σ-Pd(II) complex 9 (Scheme 2). An aryl C–H activation affords palladacycle 10, bridging the aryl and the methylene moieties. By accomplishing the reductive elimination step, the internal oxidative cross-coupling reaction of 10 promotes the formation of the C-3/C-3a sigma bond of the corresponding indole 6 and Pd(0). The latter is oxidized back to Pd(II) by the oxidizing agent to renew the catalytic cycle. This aerobic oxidative cyclization reaction is only modestly efficient and regioselective in a one-pot reaction beginning from the aniline and the ketone [67]. With an excess of Cu(OAc)2, on the other hand, the reaction produces a couple of indoles in moderate yields (41–55%) [66].
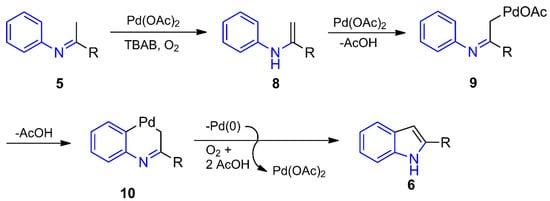
Scheme 2.
Indole synthesis from imines through a Pd(II)-catalyzed oxidative cross-coupling.
Considering the feasibility of the oxidative cyclization reaction from imines 5 to indoles 6, a one-pot reaction was explored that started from aniline 1i and phenylacetylene (2a) and progressed by a sequential Hg(I)/Pd(II)-catalyzed process. Diverse reagents were tested under aerobic conditions to find the proper oxidizing agent (Table 3). The reaction conditions were standardized and carried out in two steps. The first step consisted of applying the optimal reaction conditions found for the preparation of imines 5, thus fixing the temperature and reaction time at 40 ºC and 2 h. The second step involved the addition of Pd(OAc)2 along with the oxidizing agent in DMSO as the solvent, and then stirring at 60 ºC for 4.5 h.

Table 3.
Selection of the oxidizing agent and optimization of the reaction conditions for the one-pot conversion of 1i and 2a into indole 6h a.
The synergic action of Cu(OAc)2 and oxygen turned out to be the best oxidizing agent (Table 3, entries 8 and 9), although no significant conversion was shown when reacting each separately (entries 1 and 2). The addition of 0.2 mol equivalents of Pd(OAc)2 was critical for improving the yield (entry 9). Interestingly, mixing all the catalysts with the oxidant and solvent (DMSO) under similar reaction conditions and as a single step did not provide any trace of the imine or the indole. A possible explanation is that the Hg(I) catalyst was inactivated by DMSO. Indeed, the first step was always inefficient with the use of this solvent (Table 1).
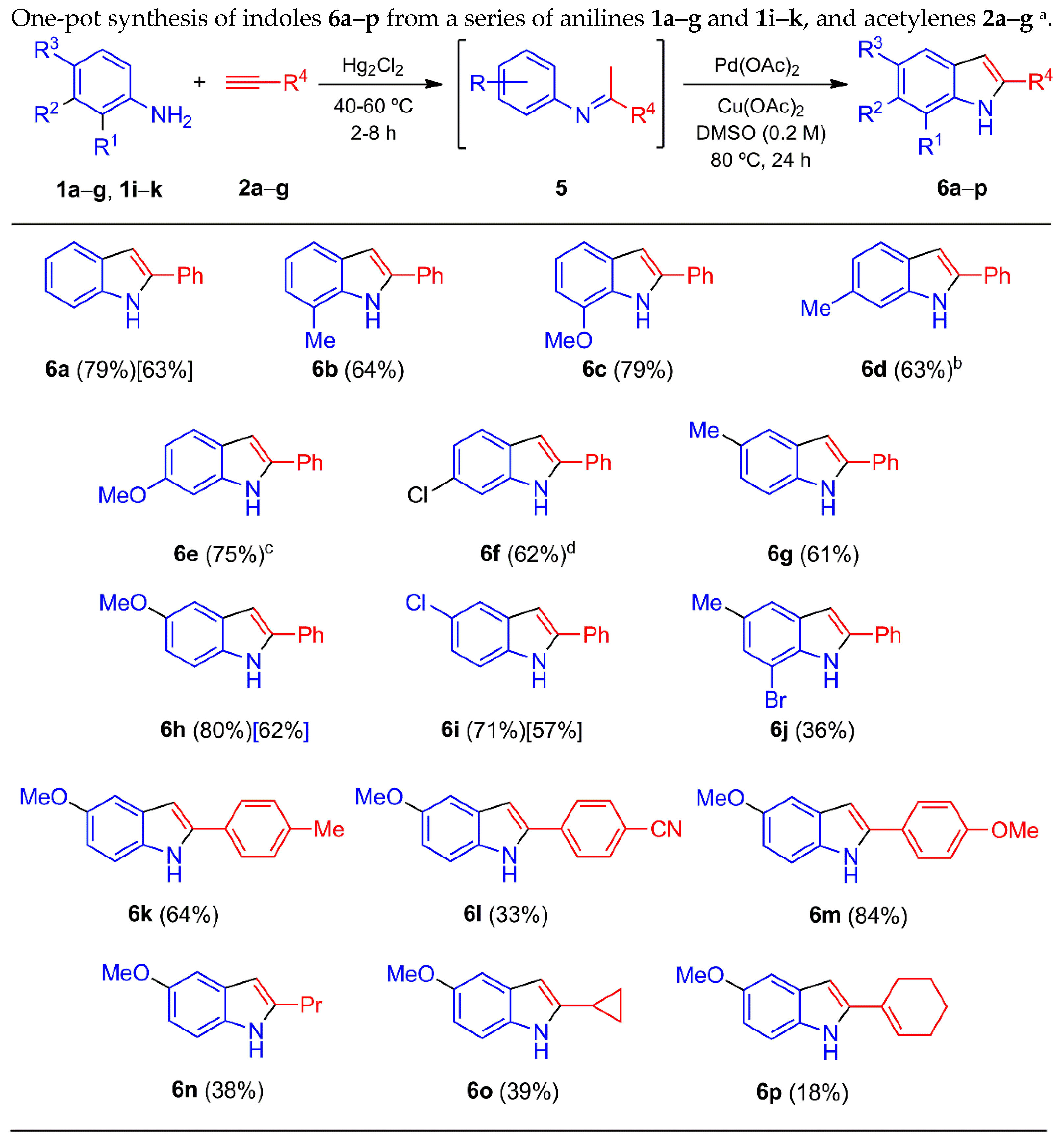
With the aim of optimizing the process, the additive was changed to K2CO3 and Cs2CO3 rather than Li2CO3, but without success (not shown in Table 3). Hence, the conversion depicted in Table 3 was carried out with or without the addition of Li2CO3 in the second step. The addition of an acid instead of Li2CO3 (entries 10–12) caused a non-significant decrease in the yield. A similar outcome was found without the presence of oxygen (entry 13). However, when both the additive and oxygen were removed from the whole process, the yield sharply increased (entry 14). The reaction temperatures and times with the best efficiency were determined, and the optimized method for the elaboration of 6h was applied to the entire series, mixing anilines 1a–g and 1i–k with acetylenes 2a–g, which gave the corresponding indoles 6a–p (Scheme 3). Most of the 2-arylindoles were obtained in good yields, regardless of the position of the substituent in the benzene ring.
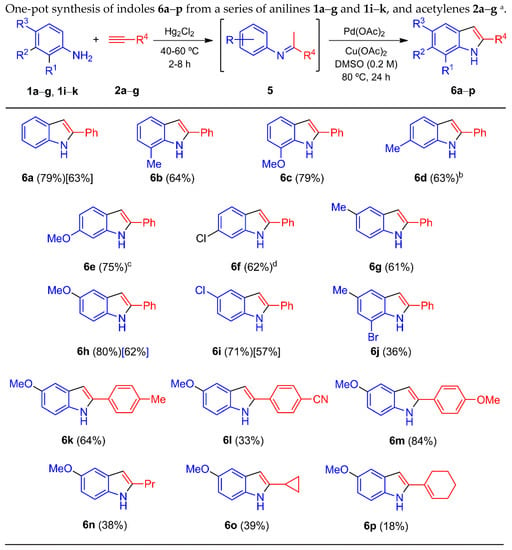
Scheme 3.
a Yields in square brackets were obtained by conversion of imines 5a and 5i–j into indoles 6a and 6h–i (see Experimental Section). b Minor isomer 4-methyl-2-phenyl-1H-indole (6d’) was detected (12% ratio) by GC/MS of the crude mixture, but could not be isolated. c Minor isomer 4-methoxy-2-phenyl-1H-indole (6e’) was detected (7% ratio) by GC/MS of the crude mixture, but could not be isolated. d Minor isomer 4-chloro-2-phenyl-1H-indole (6f’) was detected (19% ratio) by GC/MS of the crude mixture, but could not be isolated.
On the other hand, the indoles produced in low yields showed a certain correlation with the imines afforded in fairly modest yields (Table 2). For example, the in-situ formation of imine 5k delivered indole 6j in a low yield. The unstable and non-isolated imines 5m and 5o–q, generated by acetylenes 2c and 2e–g respectively, provided low yields of their corresponding indoles 6l and 6n–p. It is likely that the low yield of indole 6l was due to the low stability of the imine under the two-step reaction conditions, since the corresponding p-cyanoacetophenone (7b) was isolated in 20% yield. The reaction of aniline 1h and phenylacetylene (2a) resulted in a complex mixture of products. With the use of meta-substituted anilines 1d–f with 2a, a mixture of regioisomers 6d–f/6d’–f’ was found, respectively. The 6-substituted indoles 6d–f were the major isomers, and the 4-substituted indoles 6d’–f’ the minor ones. The latter could not be isolated (see caption, Scheme 3).
Analogous second-step reaction conditions (Pd(OAc)2/Cu(OAc)2, DMSO, 80 ºC, 24 h) were evaluated for the preparation of indoles 6a and 6h–i from imines 5a and 5i–j, leading to lower yields than those obtained from the one-pot procedure (Scheme 3, square brackets). This suggests that a possible Hg/Pd transmetalation reaction during the one-pot process may foster the necessary catalytic activity to promote the oxidative cyclization of the in-situ formed imine and provide the indole. Actually, there are many examples of satisfactory palladium-catalyzed cross-coupling reactions involving a transmetalation process with organomercury substrates [80,92,93].
The series of indoles 6a–p was characterized by IR, 1H and 13C NMR (the signals were attributed by HMQC and HMBC experiments), and HRMS. Indoles 6e and 6l were isolated as colorless and pale reddish crystals respectively, and their structures were determined by single X-ray diffraction crystallography (Figure 3). Regarding 6e, the C-2-substituted benzene ring adopted a coplanar conformation in relation to the heterocycle (torsion angle: N(1)-C(1)-C(10)-C(15) = 1.8(3), see Supplementary Materials). A larger torsion angle can be appreciated for 6l (torsion angle: C(15)-C(10)-C(1)-N(1) = 32.41(18), see Supplementary Materials). Thus, it is likely that the π-system of the aromatic rings in 6e is stabilized by conjugation, despite the possible van der Waals repulsions triggered by the eclipsed conformation of the ortho C-H protons (C2-H and C11-H). Meanwhile, the coplanarity between the aromatic rings is lost for 6l, leading to the rotation of the sigma bond C1–C10. This is probably due to the presence of the cyano group, which compensates for the loss of stability caused by the conjugation of the aromatic rings. The latter loss is due to the instability caused by such ortho C-H and N-H repulsions.
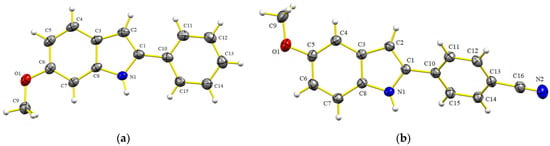
Figure 3.
Structures of 6e (a) and 6l (b) as determined by single X-ray diffraction crystallography (ellipsoids at the 30% probability level).
2.3. Mechanism of the Formation of Indoles 6a–p
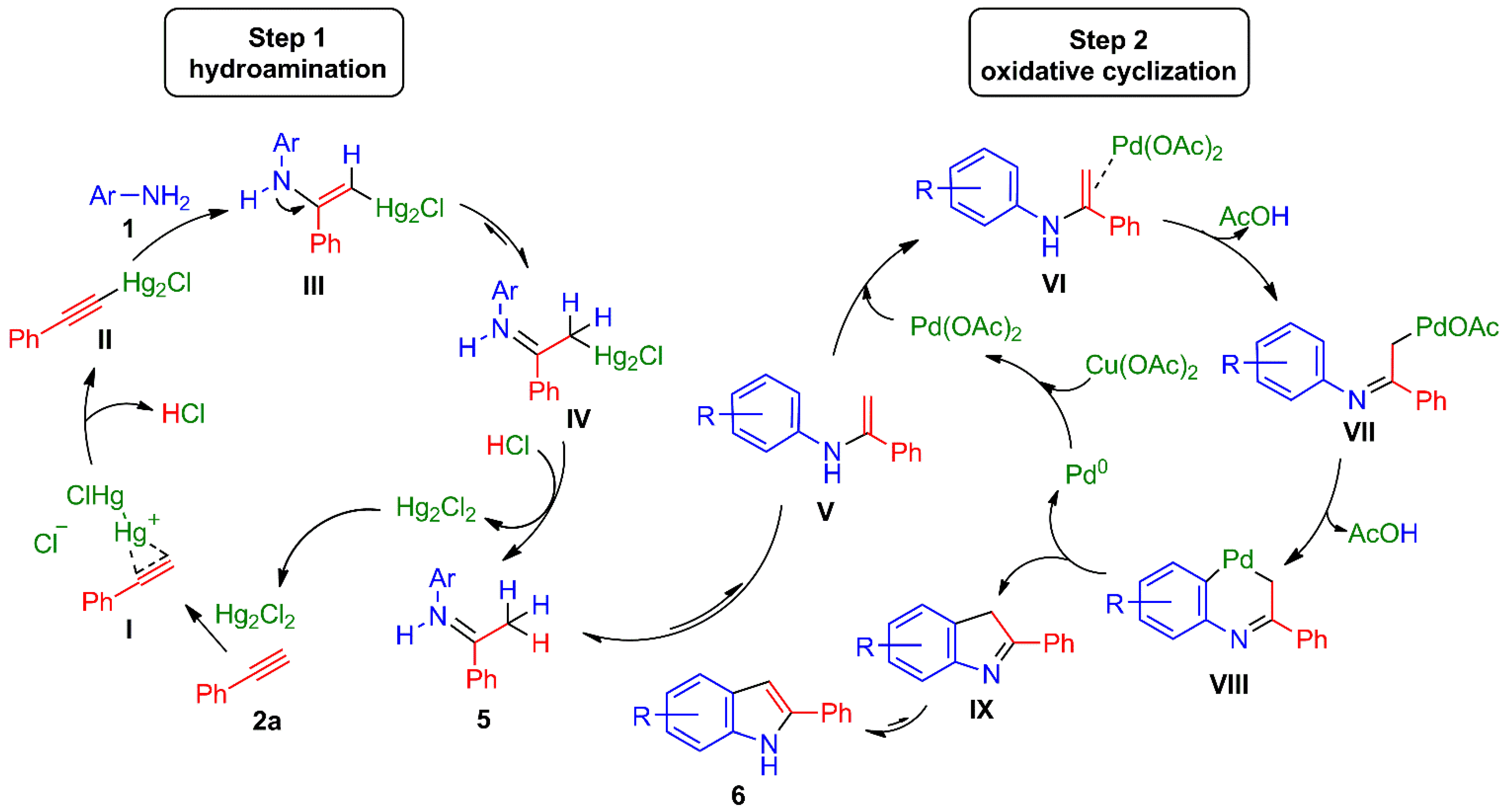
Taking into account the aforementioned results as well as the role played by the catalysts through each step [54,66,67,68] in the one-pot process for 2a, two consecutive catalytic cycles can be suggested (Scheme 4). For the first event, the triple bond of 2a would be activated by a π-Hg(I) mercurinium cation complex I, and then probably stabilized by the formation of the alkynylmercury(I) II [83]. The addition of aniline 1 to either of the two possible complexes (I or II) would generate enamino-vinylmercury(I) III, followed by tautomerization to obtain imine IV. The protonolysis of the latter [80] would afford imine 5 along with the regeneration of catalyst Hg2Cl2 to initiate a new catalytic cycle.
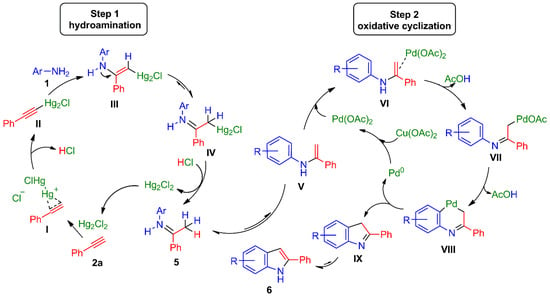
Scheme 4.
Possible consecutive Hg(I)/Pd(II) catalytic cycles for the one-pot synthesis of indoles 6. Firstly, hydroamination provides imines 5 (Step 1), and then oxidative cyclization (Step 2) furnishes the indole frame.
The second step would start with the tautomerization of imine 5 to enamine V, leading to the π-Pd(II) complex VI and the subsequent elimination of AcOH to deliver σ-Pd(II) complex VII (Scheme 4) [66,67,68]. Intramolecular aryl C–H activation would furnish palladacycle VIII, which would undergo a coupling reaction to give rise to the new C3-C3a sigma bond of heterocycle IX. The latter process would be associated with the reductive elimination of Pd(II) to form Pd(0), which would again be oxidized by the action of Cu(OAc)2. A final aromatization of IX would complete the synthesis of indoles 6.
Considering the possibility that an Hg/Pd transmetalation reaction takes place to improve the one-pot process with respect to the conversion of imines 5 to indoles 6, a plausible alternative reaction mechanism can be postulated. The Pd(II)-catalyzed transmetalation reaction may occur either with the vinylmercurial complex III [92,93] or the alkylmercurial complex IV to directly produce the σ-Pd(II) complex VII, which implies a shortcut capable of improving the process and avoiding the in-situ formation of imines 5.
Since modest efficiency of the mixture of the catalyst and oxidant agent Pd(OAc)2/Cu(OAc)2 was achieved for the one-pot conversion of anilines and ketones to indoles in only two cases [66], the Hg(I)-free reaction of aniline 1i with 2a in the presence of the Pd(OAc)2/Cu(OAc)2 couple was also tested. However, no indole was found [51]. Indeed, the alkyne was consumed only to generate 1,4-diphenylbuta-1,3-diyne, being the well-known, palladium-catalyzed, self-coupling product of terminal alkynes [94].
Given that Hg(II) was an active catalyst for promoting the formation of imine 5i (Table 1, entry 9), analogous alkynyl- and vinyl-mercury(II) (i.e., species I–III) could not be discarded as a competitive complex species [54]. Consequently, an assay was carried out with an HgCl2-catalyzed reaction of aniline 1i with 2a, followed by the Pd(OAc)2/Cu(OAc)2 couple-catalyzed cyclization in DMSO, which led to indole 6h in low yield (12%). This indicates that the mechanism of the reaction described in Scheme 4 is mainly driven by Hg(I) intermediates (i.e., species I–IV).
3. Materials and Methods
3.1. General
Melting points were determined on a Krüss KSP 1N (KRÜSS GmbH, Hamburg, Germany) capillary melting point apparatus. IR spectra were recorded on Perkin-Elmer 2000 (PerkinElmer, Waltham, MA, USA) and Bruker Vertex 70 (ATR-FT) (Bruker Corporation, Billerica, MA, USA) spectrophotometers. 1H and 13C NMR spectra were captured on Varian Mercury (300 MHz) (Varian, Inc., Palo Alto, CA, USA), Varian VNMR (500 MHz) (Varian, Inc., Palo Alto, CA, USA), and Bruker 600 AVANCE III (600 MHz) (Bruker Corporation, Billerica, MA, USA) instruments, with CDCl3 as the solvent and TMS as the internal standard. Signal assignments were based on 2D NMR spectra (HMQC, HMBC, and/or ROESY). Mass spectra (MS) were recorded on Thermo Polaris Q-Trace GC Ultra (Finnigan Co., Waltham, MA, USA) and Hewlett-Packard 5971 A (Hewlett Packard Co., Houston, TX, USA) spectrometers. High-resolution mass spectra (HRMS) were obtained (in electron impact mode) on a Jeol JSM-GCMateII (JEOL, Ltd., Tokyo, Japan) spectrometer. X-Ray crystallographic measurements were collected on an Oxford XcaliburS diffractometer (Rigaku Co., Tokyo, Japan). Analytical thin-layer chromatography was performed with 0.25 plates coated with E. Merck (Darmstadt, Germany) silica gel 60 F254 and visualized with a short/long-wavelength UV lamp. Flash column chromatography was conducted over Natland International Co. (Morrisville, NC, USA) silica gel (230–400 and 230–400 mesh). All air moisture-sensitive reactions were conducted under N2 in oven-dried glassware. DMSO was distilled over CaH2, and Li2CO3 and K2CO3 were dried overnight at 250 °C prior to use. All other reagents were utilized without further purification. Warning: The hazardous wastes of mercury-containing silica gel and reaction residues were treated with sulfur and placed in high-density plastic containers for disposal, then handled by an authorized hazardous waste company.
3.2. General Method A for the Preparation of Imines 5a–n
To a round-bottomed flask (at room temperature and in the dark), the corresponding arylacetylene 2a–d (1.0 mol equiv.), Li2CO3 (0.20 mol equiv.), the respective aniline 1a–k (1.05 mol equiv.), and mercury(I) chloride (0.05 mol equiv.) were successively added. The mixture was vigorously stirred at 60 ºC for 2–8 h. The organic layer was extracted with EtOAc (3 × 5 mL) and dried with Na2SO4. The solvent was removed under vacuum and the residue was purified by column chromatography over silica gel treated with Et3N (10% w/w) (40 g/g crude, hexane/EtOAc, 98:2) to afford imines 5a–n.
3.3. General Method B for the Preparation of Indoles 6a–p
To a round-bottomed flask (at room temperature and in the dark), the corresponding alkyl and arylacetylenes 2a–g (1.0 mol equiv.), the respective aniline 1a–g and 1i–k (1.05 mol equiv.), and mercury(I) chloride (0.05 mol equiv.) were successively added. The mixture was vigorously stirred at 40–60 ºC for 2–8 h, and anhydrous DMSO (0.2 M), Cu(OAc)2 (1.5 mol equiv.), and Pd(OAc)2 (0.20 mol equiv.) were successively added. The mixture was stirred at 80 ºC for 24 h, filtered over celite, and washed with EtOAc (4 × 20 mL). The organic layer was washed with brine (40 mL) and dried with Na2SO4. The solvent was removed under vacuum and the residue was purified by column chromatography over silica gel (30 g/g crude, hexane/EtOAc, 98:2) to provide indoles 6a–p.
3.4. General Method C for the Preparation of Indoles 6a and 6h–i Starting from Imines 5a and 5i–j
In a round-bottomed flask (at room temperature), the respective imine 5a and 5i–j, anhydrous DMSO (0.2 M), Cu(OAc)2 (1.5 mol equiv.), and Pd(OAc)2 (0.20 mol equiv.) were mixed and stirred at 80 ºC for 24 h. The crude reaction was filtered over celite and washed with EtOAc (4 × 20 mL). The organic layer was washed with brine (40 mL) and dried with Na2SO4. The solvent was removed under vacuum and the residue was purified by column chromatography over silica gel (50 g/g crude, hexane/EtOAc, 98:2) to obtain indoles 6a and 6h–i.
3.5. Preparation and Characterization of Imines 5a–n
3.5.1. (E)-N-(1-Phenylethylidene)aniline (5a)
Following general method A, a mixture of 2a (0.094 g, 0.92 mmol), Li2CO3 (0.014 g, 0.19 mmol), 1a (0.090 g, 0.97 mmol), and Hg2Cl2 (0.022 g, 0.047 mmol) was reacted for 4 h to furnish 5a (0.159 g, 88%) as a yellow solid. Rf 0.72 (hexane/EtOAc, 7:3); mp 35–36 °C (Lit. 37 °C [10]; 42–43 °C [95]). IR (film): ῡ 3058, 3027, 1638, 1593, 1482, 1447, 1366, 1287, 1214, 1072, 1026, 783, 762, 730, 694 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.23 (s, 3H, CH3-2’), 6.78–6.82 (m, 2H, H-2), 7.06–7.11 (m, 1H, H-4), 7.32–7.38 (m, 2H, H-3), 7.41–7.49 (m, 3H, H-3”, H-4”), 7.95–7.80 (m, 2H, H-2”). 13C NMR (125 MHz, CDCl3): δ 17.4 (CH3-2’), 119.3 (C-2), 123.2 (C-4), 127.1 (C-2”), 128.3 (C-3”), 128.9 (C-3), 130.4 (C-4”), 139.5 (C-1”), 151.7 (C-1), 165.4 (C-1′). HRMS (EI): m/z [M+] calcd for C14H13N: 195.1048; found: 195.1046.
3.5.2. (E)-2-Methyl-N-(1-phenylethylidene)aniline (5b)
Following general method A, a mixture of 2a (0.101 g, 0.99 mmol), Li2CO3 (0.015 g, 0.20 mmol), 1b (0.111 g, 1.04 mmol), and Hg2Cl2 (0.023 g, 0.049 mmol) was reacted for 4 h to give 5b (0.176 g, 85%) as a yellow solid. Rf 0.77 (hexane/EtOAc, 7:3); mp 56–58 °C (Lit. 61 °C [10]). IR (film): ῡ 3063, 3016, 1637, 1597, 1578, 1482, 1447, 1366, 1287, 1221, 1114, 1026, 787, 765, 737, 692 cm−1. 1H NMR (600 MHz, CDCl3): δ 2.10 (s, 3H, CH3Ar), 2.16 (s, 3H, CH3-2’), 6.65 (br d, J = 7.8 Hz, 2H, H-6), 7.00 (br dd, J = 7.8, 7.2 Hz, 1H, H-4), 7.17 (br dd, J = 7.8, 7.2 Hz, 1H, H-5), 7.21 (br d, J = 7.2 Hz, 1H, H-3), 7.43–7.49 (m, 3H, H-3”, H-4”), 7.99–8.02 (m, 2H, H-2”). 13C NMR (150 MHz, CDCl3): δ 17.4 (CH3-2’), 17.8 (CH3Ar), 118.4 (C-6), 123.2 (C-4), 126.3 (C-5), 127.1 (C-2, C-2”), 128.4 (C-3”), 130.3 (C-3), 130.4 (C-4”), 139.4 (C-1”), 150.3 (C-1), 164.9 (C-1’).
3.5.3. (E)-2-Methoxy-N-(1-phenylethylidene)aniline (5c)
Following general method A, a mixture of 2a (0.107 g, 1.05 mmol), Li2CO3 (0.015 g, 0.20 mmol), 1c (0.135 g, 1.10 mmol), and Hg2Cl2 (0.025 g, 0.053 mmol) was reacted for 4 h to afford 5c (0.205 g, 87%) as a yellow oil. Rf 0.70 (hexane/EtOAc, 7:3). IR (film): ῡ 3059, 3001, 2957, 2834, 1684, 1635, 1590, 1489, 1448, 1366, 1242, 1208, 1180, 1115, 1046, 1026, 765, 746, 693 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.18 (s, 3H, CH3-2’), 3.79 (s, 3H, CH3O), 6.78 (dd, J = 7.5, 1.7 Hz, 2H, H-6), 6.94 (dd, J = 7.5, 1.5 Hz, 1H, H-3), 6.96 (td, J = 7.5, 1.5 Hz, 1H, H-5), 7.08 (td, J = 7.5, 1.7 Hz, 1H, H-4), 7.42–7.48 (m, 3H, H-3”, H-4”), 7.97–8.03 (m, 2H, H-2”). 13C NMR (125 MHz, CDCl3): δ 17.8 (CH3-2’), 55.6 (CH3O), 111.5 (C-3), 120.5 (C-5), 120.8 (C-6), 124.1 (C-4), 127.3 (C-2”), 128.3 (C-3”), 130.4 (C-4”), 139.3 (C-1”), 140.5 (C-1), 148.9 (C-2), 167.1 (C-1’) [96].
3.5.4. (E)-3-Methyl-N-(1-phenylethylidene)aniline (5d)
Following general method A, a mixture of 2a (0.096 g, 0.94 mmol), Li2CO3 (0.014 g, 0.19 mmol), 1d (0.106 g, 0.99 mmol), and Hg2Cl2 (0.022 g, 0.047 mmol) was reacted for 4 h to give 5d (0.163 g, 83%) as a yellow oil. Rf 0.67 (hexane/EtOAc, 7:3); (Lit. mp 34 °C [10]). IR (film): ῡ 3059, 3001, 2958, 2834, 1684, 1634, 1594, 1485, 1447, 1367, 1313, 1288, 1261, 1193, 1148, 1045, 913, 852, 784, 765, 694 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.23 (s, 3H, CH3-2’), 2.35 (s, 3H, CH3Ar), 6.60 (dm, J = 7.5 Hz, 1H, H-6), 6.61–6.64 (m, 1H, H-2), 6.90 (dm, J = 7.5 Hz, 1H, H-4), 7.23 (t, J = 7.5 Hz, 1H, H-5), 7.41–7.48 (m, 3H, H-3”, H-4”), 7.94–7.98 (m, 2H, H-2”). 13C NMR (125 MHz, CDCl3): δ 17.3 (CH3-2’), 21.5 (CH3Ar), 116.3 (C-6), 120.0 (C-2), 123.9 (C-4), 127.1 (C-2”), 128.3 (C-3”), 128.8 (C-5), 130.4 (C-4”), 138.7 (C-3), 139.5 (C-1”), 151.7 (C-1), 165.2 (C-1’). HRMS (EI): m/z [M+] calcd for C15H15N: 209.1204; found: 209.1205.
3.5.5. (E)-3-Methoxy-N-(1-phenylethylidene)aniline (5e)
Following general method A, a mixture of 2a (0.096 g, 0.94 mmol), Li2CO3 (0.014 g, 0.19 mmol), 1e (0.122 g, 0.99 mmol), and Hg2Cl2 (0.022 g, 0.047 mmol) was reacted for 4 h to form 5e (0.178 g, 84%) as a yellow oil. Rf 0.70 (hexane/EtOAc, 7:3). IR (film): ῡ 3452, 3369, 2929, 2837, 1602, 1496, 1464, 1292, 1206, 1158, 1038, 835, 764, 688. 1H NMR (500 MHz, CDCl3): δ 2.25 (s, 3H, CH3-2’), 3.81 (s, 3H, CH3O), 6.35–6.40 (m, 2H, H-2, H-6), 6.64 (ddd, J = 8.0, 2.0, 1.0 Hz, 1H, H-4), 7.25 (t, J = 8.0 Hz, 1H, H-5), 7.42–7.49 (m, 3H, H-3”, H-4”), 7.95–7.98 (m, 2H, H-2”). 13C NMR (125 MHz, CDCl3): δ 17.4 (CH3-2’), 55.2 (CH3O), 105.0 (C-2), 108.8 (C-4), 111.7 (C-6), 127.1 (C-2”), 128.3 (C-3”), 129.8 (C-5), 130.5 (C-4”), 139.4 (C-1”), 153.1 (C-1), 160.3 (C-3), 165.5 (C-1’) [97].
3.5.6. (E)-3-Chloro-N-(1-phenylethylidene)aniline (5f)
Following general method A, a mixture of 2a (0.111 g, 1.09 mmol), Li2CO3 (0.016 g, 0.22 mmol), 1f (0.145 g, 1.14 mmol), and Hg2Cl2 (0.026 g, 0.055 mmol) was reacted for 4 h to provide 5f (0.132 g, 53%) as a brown oil. Rf 0.76 (hexane/EtOAc, 7:3). IR (film): ῡ 3060, 2920, 1635, 1588, 1470, 1448, 1367, 1288, 1212, 1072, 864, 788, 763, 692 cm−1. 1H NMR (600 MHz, CDCl3): δ 2.25 (s, 3H, CH3-2’), 6.68 (dm, J = 8.1 Hz, 1H, H-6), 6.81 (t, J = 1.8 Hz, 1H, H-2), 7.07 (dm, J = 8.1 Hz, 1H, H-4), 7.27 (t, J = 8.1 Hz, 1H, H-5), 7.43–7.51 (m, 3H, H-3”, H-4”), 7.94–7.97 (m, 2H, H-2”). 13C NMR (150 MHz, CDCl3): δ 17.9 (CH3-2’), 118.0 (C-6), 119.8 (C-2), 123.5 (C-4), 127.5 (C-2”), 128.7 (C-3”), 130.4 (C-5), 131.1 (C-4”), 134.8 (C-3), 139.3 (C-1”), 152.2 (C-1), 166.6 (C-1’). HRMS (EI): m/z [M+] calcd for C14H12N: 229.0658; found: 209.0652 [13].
3.5.7. (E)-4-Methyl-N-(1-phenylethylidene)aniline (5g)
Following general method A, a mixture of 2a (0.096, 0.94 mmol), Li2CO3 (0.014 g, 0.19 mmol), 1g (0.105 g, 0.98 mmol), and Hg2Cl2 (0.022 g, 0.047 mmol) was reacted for 2 h to afford 5g (0.179 g, 91%) as a yellow oil. Rf 0.76 (hexane/EtOAc, 7:3); (Lit. 62 °C [10]). IR (film): ῡ 3449, 3363, 3023, 2920, 1683, 1633, 1516, 1505, 1447, 1364, 1288, 1266, 1219, 1026, 842, 827, 763, 692 cm−1. 1H NMR (600 MHz, CDCl3): δ 2.24 (s, 3H, CH3-2’), 2.35 (s, 3H, CH3Ar), 6.70 (d, J = 7.8 Hz, 2H, H-2), 7.16 (d, J = 7.8 Hz, 2H, H-3), 7.41–7.49 (m, 3H, H-3”, H-4”), 7.94–7.99 (m, 2H, H-2”). 13C NMR (150 MHz, CDCl3): δ 17.3 (CH3
-2’), 20.9 (CH3Ar), 119.4 (C-2), 127.1 (C-2”), 128.3 (C-3”), 129.5 (C-3), 130.3 (C-4”), 132.6 (C-4), 139.7 (C-1”), 149.0 (C-1), 165.5 (C-1’). HRMS (EI): m/z [M+] calcd for C15H15N: 209.1204; found: 209.1210 [51].
3.5.8. (E)-4-(N,N-dimethylamino)-N-(1-phenylethylidene)aniline (5h)
Following general method A, a mixture of 2a (0.099 g, 0.97 mmol), Li2CO3 (0.014 g, 0.19 mmol), 1h (0.139 g, 1.02 mmol) and Hg2Cl2 (0.023 g, 0.049 mmol) was reacted for 2 h to obtain 5h (0.158 g, 68%) as a brown solid. Rf 0.53 (hexane/EtOAc, 7:3); mp 90–92 °C. IR (film): ῡ 2918, 2803, 1622, 1510, 1446, 1364, 1222, 830, 760, 690 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.28 (s, 3H, CH3-2’), 2.95 (s, 6H, (CH3)2N), 6.75–6.82 (m, 4H, H-2, H-3), 7.41–7.45 (m, 3H, H-3”, H4”), 7.95–7.97 (m, 2H, H-2”). 13C NMR (125 MHz, CDCl3): δ 17.3 (CH3-2’), 41.2 ((CH3)2N), 113.5 (C-3), 121.0 (C-2), 127.0 (C-2”), 128.3 (C-3”), 130.1 (C-4”), 140.1 (C-1”), 141.7 (C-1), 147.4 (C-4), 165.0 (C-1’). HRMS (EI): m/z [M+] calcd for C16H18N2: 238.1470; found: 238.1471 [98].
3.5.9. (E)-4-Methoxy-N-(1-phenylethylidene)aniline (5i)
Following general method A, a mixture of 2a (0.125 g, 1.23 mmol), Li2CO3 (0.018 g, 0.24 mmol), 1i (0.158 g, 1.28 mmol), and Hg2Cl2 (0.029 g, 0.06 mmol) was reacted for 2 h to furnish 5i (0.252 g, 91%) as a pale yellow crystalline solid. Rf 0.70 (hexane/EtOAc, 7:3); mp 93–94 °C (Lit. 86 °C [10,95]; Lit. 85–86 °C [50]). IR (KBr): ῡ 2954, 2929, 1616, 1576, 1510, 1443, 1365, 1284, 1240, 1210, 1032, 846, 771, 752, 699 cm−1. 1H NMR (600 MHz, CDCl3): δ 2.26 (s, 3H, CH3-2’), 3.82 (s, 3H, CH3O), 6.73–6.79 (m, 2H, H-2), 6.89–6.94 (m, 2H, H-3), 7.41–7.49 (m, 3H, H-3”, H-4”), 7.95–7.98 (m, 2H, H-2”). 13C NMR (150 MHz, CDCl3): δ 17.3 (C-2’), 55.5 (CH3O), 114.2 (C-3), 120.7 (C-2), 127.1 (C-2”), 128.3 (C-3”), 130.3 (C-4”), 139.8 (C-1”), 144.8 (C-1), 155.9 (C-4), 165.7 (C-1’). HRMS (EI): m/z [M+] calcd for C15H15NO: 225.1154; found: 225.1164 [51,99].
3.5.10. (E)-4-Chloro-N-(1-phenylethylidene)aniline (5j)
Following general method A, a mixture of 2a (0.119 g, 1.17 mmol), Li2CO3 (0.017 g, 0.23 mmol), 1j (0.155 g, 1.22 mmol), and Hg2Cl2 (0.027 g, 0.057 mmol) was reacted for 2 h to give 5j (0.209 g, 78%) as a yellow solid. Rf 0.76 (hexane/EtOAc, 7:3); mp 68–70 °C (Lit. 73–74 °C [99]). IR (film): ῡ 3061, 2955, 2854, 1634, 1579, 1483, 1447, 1367, 1288, 1214, 1091, 1010, 844, 762, 692, 674 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.23 (s, 3H, CH3-2’), 6.71–6.76 (m, 2H, H-2), 7.28–7.33 (m, 2H, H-3), 7.42–7.50 (m, 3H, H-3”, H-4”), 7.93–7.98 (m, 2H, H-2”). 13C NMR (125 MHz, CDCl3): δ 17.4 (CH3-2’), 120.8 (C-2), 127.2 (C-2”), 128.4 (C-3”), 128.5 (C-4), 129.0 (C-3), 130.7 (C-4”), 139.2 (C-1”), 150.2 (C-1), 166.2 (C-1’) [13,51].
3.5.11. (E)-2-Bromo-4-methoxy-N-(1-phenylethylidene)aniline (5k)
Following general method A, a mixture of 2a (0.093 g, 0.91 mmol), Li2CO3 (0.013 g, 0.182 mmol), 1k (0.177 g, 0.95 mmol), and Hg2Cl2 (0.021 g, 0.044 mmol) was reacted for 8 h to produce 5k (0.110 g, 42%) as a yellow oil. Rf 0.73 (hexane/EtOAc, 7:3). IR (film): ῡ 3057, 3026, 2921, 1645, 1634, 1578, 1480, 1447, 1366, 1293, 1221, 1044, 865, 828, 761, 692 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.19 (s, 3H, CH3-2’), 2.33 (s, 3H, CH3Ar), 6.68 (d, J = 8.0 Hz, 1H, H-6), 7.09 (dd, J = 8.0, 2.0 Hz, 1H, H-5), 6.43 (d, J = 2.0 Hz, 1H, H-3), 7.44–7.50 (m, 3H, H-3”, H-4”), 7.98–8.02 (m, 2H, H-2’). 13C NMR (125 MHz, CDCl3): δ 18.0 (CH3-2’), 20.5 (CH3Ar), 113.5 (C-2), 120.1 (C-6), 127.4 (C-2”), 128.4 (C-3”), 128.7 (C-5), 130.7 (C-4”), 133.1 (C-3), 134.2 (C-4), 139.1 (C-1”), 147.4 (C-1), 167.6 (C-1’). HRMS (EI): m/z [M+] calcd for C15H14BrN: 287.0310; found: 287.0318.
3.5.12. (E)-4-Methoxy-N-(1-(p-tolyl)ethylidene)aniline (5l)
Following general method A, a mixture of 2b (0.118 g, 1.02 mmol), Li2CO3 (0.015 g, 0.20 mmol), 1i (0.132 g, 1.07 mmol), and Hg2Cl2 (0.024 g, 0.051 mmol) was reacted for 2 h to generate 5l (0.221 g, 91%) as a yellow solid. Rf 0.73 (hexane/EtOAc, 7:3); mp 78–80 °C (Lit. 83 °C [10]). IR (film): ῡ 2996, 2954, 2930, 2835, 1621, 1576, 1505, 1445, 1366, 1286, 1242, 1211, 1181, 1032, 846, 761, 699 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.22 (s, 3H, CH3-2’), 2.40 (s, 3H, CH3Ar), 3.81 (s, 3H, CH3O), 6.72–6.76 (m, 2H, H-2), 6.88–6.92 (m, 2H, H-3), 7.22–7.25 (m, 2H, H-3”), 7.84–7.87 (m, 2H, H-2”). 13C NMR (125 MHz, CDCl3): δ 17.2 (CH3-2’), 21.3 (CH3Ar), 55.5 (CH3O), 114.2 (C-3), 120.8 (C-2), 127.1 (C-2”), 129.0 (C-3”), 137.1 (C-1”), 140.5 (C-4”), 145.0 (C-1), 155.8 (C-4), 165.5 (C-1’) [100].
3.5.13. (E)-4-(1-((4-methoxyphenyl)imino)ethyl)benzonitrile (5m)
Following general method A, a mixture of 2c (0.083 g, 0.65 mmol), Li2CO3 (0.010 g, 0.13 mmol), 1i (0.084 g, 0.68 mmol), and Hg2Cl2 (0.014 g, 0.03 mmol) was reacted for 2 h, resulting in an inseparable mixture of 5m/7b (95:5) as an instable brown solid. The yield of compound 5m (0.147 g, 90%) was estimated by 1H NMR and GC/MS. Rf 0.48 (hexano/AcOEt, 7:3). 1H NMR (500 MHz, CDCl3): δ 2.28 (s, 3H, CH3-2’), 3.83 (s, 3H, CH3O), 6.73–6.78 (m, 2H, H-2), 6.91–6.95 (m, 2H, H-3), 7.72–7.75 (m, 2H, H-3”), 8.04–8.09 (m, 2H, H-2”). Signals attributed to 7b: 2.65 (s, CH3-2’), 3.74 (s, CH3O), 7.74–7.80 (m, H-3”) [100].
3.5.14. (E)-4-Methoxy-N-(1-(4-methoxyphenyl)ethylidene)aniline (5n)
Following general method A, a mixture of 2d (0.095 g, 0.72 mmol), Li2CO3 (0.011 g, 0.15 mmol), 1i (0.093 g, 0.76 mmol), and Hg2Cl2 (0.017 g, 0.036 mmol) was reacted for 2 h to form 5n (0.152 g, 83%) as a pale yellow solid. Rf 0.57 (hexane/EtOAc, 7:3); mp 130–132 °C (Lit. 132–134 °C [95]). IR (film): ῡ 2968, 2837, 1626, 1601, 1502, 1379, 1286, 1258, 1239, 1207, 1172, 1028, 833 cm−1. 1H NMR (600 MHz, CDCl3): δ 2.22 (s, 3H, CH3-2’), 3.82 (s, 3H, CH3O-4), 3.86 (s, 3H, CH3O-4”), 6.72–6.76 (m, 2H, H-2), 6.88–6.92 (m, 2H, H-3), 6.93–6.96 (m, 2H, H-3”), 7.91–7.95 (m, 2H, H-2”). 13C NMR (150 MHz, CDCl3): δ 17.1 (C-2’), 55.4 (CH3O-C4”), 55.5 (CH3O-C4), 113.6 (C-3”), 114.2 (C-3), 120.9 (C-2), 128.7 (C-2”), 132.5 (C-1”), 145.0 (C-1), 155.8 (C-4), 161.4 (C4”), 164.8 (C-1’) [99,100].
3.6. Preparation and Characterization of Indoles 6a–p
3.6.1. 2-Phenyl-1H-indole (6a)
Following general method B, a mixture of 2a (0.080 g, 0.79 mmol), 1a (0.077 g, 0.83 mmol), and Hg2Cl2 (0.019 g, 0.04 mmol) was stirred at 60 °C for 4 h. It was then reacted with DMSO (3.9 mL), Cu(OAc)2 (0.215 g, 1.18 mmol), and Pd(OAc)2 (0.035 g, 0.16 mmol) to yield 6a (0.120 g, 79%) as a brown solid. Rf 0.77 (hexane/EtOAc, 7:3).
Following general method C, a mixture of 5a (0.088 g, 0.45 mmol), DMSO (2.2 mL), Cu(OAc)2 (0.123 g, 0.68 mmol), and Pd(OAc)2 (0.020 g, 0.09 mmol) was reacted to yield 6a (0.054 g, 63%) as a pale brownish solid. Rf 0.77 (hexane/EtOAc, 7:3); mp 189–190 °C (Lit. 189–190 ºC [66], 188–189 ºC [101], 187–188 ºC [102]). IR (ATR): ῡ 3439, 2919, 2853, 1455, 1230, 794, 770, 742, 690 cm−1. 1H NMR (600 MHz, CDCl3): δ 6.83 (dd, J = 2.1, 0.9 Hz, 1H, H-3), 7.12 (td, J = 7.8, 0.6 Hz, 1H, H-5), 7.19 (td, J = 7.8, 1.2 Hz, 1H, H-6), 7.30–7.33 (m, 1H, H-4’), 7.39 (dd, J = 7.8, 0.9 Hz, 1H, H-7), 7.41–7.46 (m, 2H, H-3’), 7.63 (dd, J = 7.8, 0.9 Hz, 1H, H-4), 7.64–7.67 (m, 2H, H-2’), 8.32 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 100.0 (C-3), 110.9 (C-7), 120.3 (C-5), 120.7 (C-4), 122.4 (C-6), 125.2 (C-2’), 127.7 (C-4’), 129.0 (C-3’), 129.3 (C-3a), 132.4 (C-1’), 136.8 (C-7a), 137.9 (C-2).
3.6.2. 7-Methyl-2-phenyl-1H-indole (6b)
Following general method B, a mixture of 2a (0.077 g, 0.57 mmol), 1b (0.085 g, 0.79 mmol), and Hg2Cl2 (0.018 g, 0.04 mmol) was stirred at 60 °C for 4 h. It was then reacted with DMSO (3.8 mL), Cu(OAc)2 (0.205 g, 1.13 mmol), and Pd(OAc)2 (0.034 g, 0.15 mmol) to provide 6b (0.10 g, 64%) as a brown solid. Rf 0.67 (hexane/EtOAc, 7:3); mp 114–116 °C (Lit. 112–118 °C [103]). IR (ATR): ῡ 3450, 1601, 1486, 1449, 1335, 1330, 1299, 804, 770, 737, 690 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.52 (s, 3H, CH3Ar), 6.81 (d, J = 2.5 Hz, 1H, H-3), 6.98 (dm, J = 7.5 Hz, 1H, H-6), 7.04 (t, J = 7.5 Hz, 1H, H-5), 7.30 (tm, J = 7.5 Hz, 1H, H-4’), 7.40–7.44 (m, 2H, H-3’), 7.47 (dm, J = 7.5 Hz, 1H, H-4), 7.64–7.68 (m, 2H, H-2’), 8.15 (br s, 1H, NH). 13C NMR (125 MHz, CDCl3): δ 16.7 (CH3Ar), 100.6 (C-3), 118.4 (C-4), 120.0 (C-7), 120.4 (C-5), 122.9 (C-6), 125.2 (C-2’), 127.6 (C-4’), 128.8 (C-3a), 129.0 (C-3’), 132.5 (C-1’), 136.4 (C-7a), 137.6 (C-2).
3.6.3. 7-Methoxy-2-phenyl-1H-indole (6c)
Following general method B, a mixture of 2a (0.086 g, 0.84 mmol), 1c (0.110 g, 0.89 mmol), and Hg2Cl2 (0.020 g, 0.04 mmol) was stirred at 60 °C for 4 h. It was then reacted with DMSO (4.2 mL), Cu(OAc)2 (0.230 g, 1.27 mmol), and Pd(OAc)2 (0.038 g, 0.17 mmol) to furnish 6c (0.149 g, 79%) as an amber solid. Rf 0.67 (hexane/EtOAc, 7:3); mp 63–64 °C (Lit. 94–95 ºC [66]). IR (ATR): ῡ 3429, 1608, 1581, 1505, 1485, 1453, 1401, 1332, 1316, 1258, 1225, 1097, 798, 772, 753, 730 cm−1. 1H NMR (600 MHz, CDCl3): δ 4.00 (s, 3H, CH3O), 6.66 (d, J = 7.8 Hz, 1H, H-6), 6.81 (d, J = 2.4, 1H, H-3), 7.04 (t, J = 7.8 Hz, 1H, H-5), 7.24 (d, J = 7.8 Hz, 1H, H-4), 7.31–7.34 (m, 1H, H-4’), 7.42–7.46 (m, 2H, H-3’), 7.68–7.70 (m, 2H, H-2’), 8.56 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 55.3 (CH3O), 100.2 (C-3), 102.1 (C-6), 113.3 (C-4), 120.5 (C-5), 125.1 (C-2’), 127.3 (C-7a), 127.6 (C-4’), 129.0 (C-3’), 130.4 (C-3a), 132.4 (C-1’), 137.5 (C-2), 145.9 (C-7). HRMS (EI): m/z [M+] calcd for C15H13NO: 223.0997; found: 223.0994.
3.6.4. 6-Methyl-2-phenyl-1H-indole (6d). 4-Methyl-2-phenyl-1H-indole (6d’)
Following general method B, a mixture of 2a (0.076 g, 0.75 mmol), 1d (0.084 g, 0.79 mmol), and Hg2Cl2 (0.019 g, 0.04 mmol) was stirred at 60 °C for 4 h. It was then reacted with DMSO (3.7 mL), Cu(OAc)2 (0.204 g, 1.12 mmol), and Pd(OAc)2 (0.034 g, 0.15 mmol) to afford a mixture of 6d/6d’ (88:12), which was separated to provide 6d (0.097 g, 63%) as a pale yellow solid. Rf 0.70 (hexane/EtOAc, 7:3); mp 186–187 °C (Lit. 189–190 °C [102], 190–192 °C [103]). IR (ATR): ῡ 3429, 1454, 1384, 1350, 1232, 814, 760, 741, 687 cm−1. 1H NMR (600 MHz, CDCl3): δ 2.47 (s, 3H, CH3Ar), 6.78 (dd, J = 2.4, 0.9 Hz, 1H, H-3), 6.96 (dd, J = 8.4, 0.9 Hz, 1H, H-5), 7.20 (d, J = 0.9 Hz, 1H, H-7), 7.29–7.32 (m, 1H, H-4’), 7.41–7.45 (m, 2H, H-3’), 7.51 (d, J = 8.4, 1H, H-4), 7.63–7.66 (m, 2H, H-2’), 8.21 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 21.8 (CH3Ar), 99.8 (C-3), 110.8 (C-7), 120.3 (C-4), 122.0 (C-5), 125.0 (C-2’), 127.1 (C-3a), 127.5 (C-4’), 129.0 (C-3’), 132.3 (C-6), 132.6 (C-1’), 137.2 (C-7a), 137.3 (C-2). Data for 6d: MS (70 eV): m/z 207 (M+, 100), 206 (60), 178 (5), 130 (7), 77 (18), 44 (15). Data for 6d’: MS (70 eV): m/z 207 (M+, 10), 177 (13), 169 (11), 143 (7), 119 (14), 77 (30), 44 (100).
3.6.5. 6-Methoxy-2-phenyl-1H-indole (6e). 4-Methoxy-2-phenyl-1H-indole (6e’)
Following general method B, a mixture of 2a (0.085 g, 0.83 mmol), 1e (0.107 g, 0.87 mmol), and Hg2Cl2 (0.019 g, 0.04 mmol) was stirred at 60 °C for 4 h. It was then reacted with DMSO (4.4 mL), Cu(OAc)2 (0.225 g, 1.24 mmol), and Pd(OAc)2 (0.038 g, 0.17 mmol), leading to a mixture of 6e/6e’ (93:7) [66], which was separated by column chromatography over silica gel (30 g/g crude, hexane/EtOAc, 98:2) to give 6e (0.138 g, 75%) as a brown solid. Rf 0.60 (hexane/EtOAc, 7:3); mp 160–161 °C (Lit. 177–178 °C [66]; 173–176 °C [102]). IR (ATR): ῡ 3396, 1625, 1446, 1257, 1200, 1158, 1016, 823, 756 cm−1. 1H NMR (600 MHz, CDCl3): δ 3.86 (s, 3H, CH3O), 6.76 (d, J = 1.8, 1H, H-3), 6.80 (dd, J = 8.4, 2.4 Hz, 1H, H-5), 6.90 (d, J = 2.4, 1H, H-7), 7.27–7.31 (m, 1H, H-4’), 7.41–7.44 (m, 2H, H-3’), 7.50 (d, J = 8.4, 1H, H-4), 7.60–7.63 (m, 2H, H-2’), 8.24 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 55.6 (CH3O), 94.4 (C-7), 99.8, (C-3), 110.2 (C-5), 121.3 (C-4), 123.5 (C-3a), 124.7 (C-2’), 127.2 (C-4’), 129.0 (C-3’), 132.5 (C-1’), 136.8 (C-2), 137.6 (C-7a), 156.7 (C-6). Data for 6e: MS (70 eV): m/z 223 (M+, 77), 208 (100), 180 (23), 152 (17), 77 (3). Data for 6e’: MS (70 eV): m/z 223 (M+, 100), 208 (87), 180 (17), 127 (15), 89 (23), 77 (13). HRMS (EI): m/z [M+] calcd for C15H13NO: 223.0997; found: 223.0991.
3.6.6. 6-Chloro-2-phenyl-1H-indole (6f). 4-Chloro-2-phenyl-1H-indole (6f’)
Following general method B, a mixture of 2a (0.079 g, 0.77 mmol), 1f (0.103 g, 0.81 mmol), and Hg2Cl2 (0.019 g, 0.04 mmol) was stirred at 60 °C for 4 h. It was then reacted with DMSO (4.3 mL), Cu(OAc)2 (0.211 g, 1.16 mmol), and Pd(OAc)2 (0.034 g, 0.15 mmol), resulting in a mixture of 6f/6f’ (81:19), which was separated by column chromatography over silica gel (30 g/g crude, hexane/EtOAc, 98:2) to obtain 6f (0.109 g, 62%) as a yellow solid. Rf 0.70 (hexane/EtOAc, 7:3); mp 181–182 °C (Lit. 171–173 °C [104]; 182–183 °C [105]). IR (ATR): ῡ 3429, 1453, 1310, 1067, 876, 805, 758, 736, 688 cm−1. 1H NMR (500 MHz, CDCl3): δ 6.79 (d, J = 2.0 Hz, 1H, H-3), 7.09 (dd, J = 8.5, 1.5 Hz, 1H, H-5), 7.34 (br t, J = 7.5 Hz, 1H, H-4’), 7.39 (d, J = 1.5 Hz, 1H, H-7), 7.45 (t, J = 7.5 Hz, 2H, H-3’), 7.52 (d, J = 8.5 Hz, 1H, H-4), 7.65 (d, J = 7.5 Hz, 2H, H-2’), 8.32 (br s, 1H, NH). 13C NMR (125 MHz, CDCl3): δ 100.0 (C-3), 110.8 (C-7), 121.0 (C-5), 121.5 (C-4), 125.2 (C-2’), 127.8 (C-3a), 128.0 (C-4’), 128.1 (C-6), 129.1 (C-3’), 131.9 (C-1’), 137.1 (C-7a), 138.6 (C-2). Data for 6f: MS (70 eV): m/z 229 (M++2, 37), 227 (M+, 100), 190 (8), 165 (23), 114 (5), 89 (13), 77 (3). Data for 6f’: MS (70 eV): m/z 229 (M++2, 30), 227 (M+, 100), 190 (8), 165 (17), 89 (15).
3.6.7. 5-Methyl-2-phenyl-1H-indole (6g)
Following general method B, a mixture of 2a (0.083 g, 0.81 mmol), 1g (0.092 g, 0.86 mmol), and Hg2Cl2 (0.019 g, 0.04 mmol) was stirred at 60 °C for 2 h. It was then reacted with DMSO (4.1 mL), Cu(OAc)2 (0.222 g, 1.22 mmol), and Pd(OAc)2 (0.036 g, 0.16 mmol) to produce 6g (0.103 g, 61%) as a pale-yellow solid. Rf 0.70 (hexane/EtOAc, 7:3); mp 214–215 °C (Lit. 216–218 °C [66]; 211–213 °C [102]; 218–220 °C [103]). IR (ATR): ῡ 3405, 1603, 1449, 1406, 1299, 1202, 1079, 804, 799, 737, 761, 686 cm−1. 1H NMR (600 MHz, CDCl3): δ 2.45 (s, 3H, CH3), 6.75 (dd, J = 2.4, 0.6 Hz, 1H, H-3), 7.02 (dd, J = 8.4, 1.2 Hz, 1H, H-6), 7.28 (d, J = 8.4 Hz, 1H, H-7), 7.29–7.32 (m, 1H, H-4’), 7.41 (dd, J = 1.2, 0.6 Hz, 1H, H-4), 7.41–7.44 (m, 2H, H-3’), 7.63–7.66 (m, 2H, H-2’), 8.23 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 21.5 (CH3), 99.5 (C-3), 110.5 (C-7), 120.3 (C-4), 124.0 (C-6), 125.0 (C-2’), 127.6 (C-4’), 129.0 (C-3’), 129.46 (C-5), 129.54 (C-3a), 132.5 (C-1’), 135.2 (C-7a), 137.9 (C-2).
3.6.8. 5-Methoxy-2-phenyl-1H-indole (6h)
Following general method B, a mixture of 2a (0.083 g, 0.82 mmol), 1i (0.106 g, 0.86 mmol), and Hg2Cl2 (0.019 g, 0.04 mmol) was stirred at 60 °C for 2 h. It was then reacted with DMSO (4.1 mL), Cu(OAc)2 (0.222 g, 1.22 mmol), and Pd(OAc)2 (0.036 g, 0.16 mmol) to generate 6h (0.146 g, 80%) as a yellow solid. Rf 0.60 (hexane/EtOAc, 7:3).
Following general method C, a mixture of 5i (0.060 g, 0.27 mmol), DMSO (1.33 mL), Cu(OAc)2 (0.073 g, 0.40 mmol), and Pd(OAc)2 (0.012 g, 0.054 mmol) was reacted to afford 6h (0.037 g, 62%) as a pale brownish solid. Rf 0.60 (hexane/EtOAc, 7:3). mp 168–169 °C (Lit. 169–170 °C [66], 166–169 °C [101], 160–162 °C [102]). IR (KBr): ῡ 3415, 1615, 1479, 1208, 1148, 1030, 765 cm−1. 1H NMR (600 MHz, CDCl3): δ 3.87 (s, 3H, CH3O), 6.76 (dd, J = 2.4, 1.2 Hz, 1H, H-3), 6.86 (dd, J = 9.0, 2.4 Hz, 1H, H-6), 7.09 (d, J = 2.4 Hz, 1H, H-4), 7.29 (d, J = 9.0 Hz, 1H, H-7), 7.30–7.33 (m, 1H, H-4’), 7.41–7.46 (m, 2H, H-3’), 7.63–7.66 (m, 2H, H-2’), 8.21 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 55.8 (CH3O), 99.9 (C-3), 102.3 (C-4), 111.6 (C-7), 112.6 (C-6), 125.1 (C-2’), 127.6 (C-4’), 129.0 (C-3’), 129.7 (C-3a), 132.0 (C-7a), 132.5 (C-1’), 138.6 (C-2), 154.5 (C-5).
3.6.9. 5-Chloro-2-phenyl-1H-indole (6i)
Following general method B, a mixture of 2a (0.085 g, 0.83 mmol), 1j (0.111 g, 0.87 mmol), and Hg2Cl2 (0.019 g, 0.04 mmol) was stirred at 60 °C for 2 h. It was then reacted with DMSO (4.1 mL), Cu(OAc)2 (0.225 g, 1.24 mmol), and Pd(OAc)2 (0.037 g, 0.17 mmol) to give 6i (0.134 g, 71%) as a yellow solid. Rf 0.60 (hexane/EtOAc, 7:3).
Following general method C, a mixture of 5j (0.081 g, 0.35 mmol), DMSO (1.77 mL), Cu(OAc)2 (0.073 g, 0.53 mmol), and Pd(OAc)2 (0.016 g, 0.07 mmol) was reacted to provide 6i (0.046 g, 57%) as a yellow solid. Rf 0.60 (hexane/EtOAc, 7:3); mp 195–196 °C (Lit. 196–197 °C [66], 198–200 °C [103]). IR (ATR): ῡ 3429, 1452, 1313, 1065, 876, 803, 754, 736, 687 cm−1. 1H NMR (600 MHz, CDCl3): δ 6.76 (d, J = 1.8 Hz, 1H, H-3), 7.14 (dd, J = 8.4, 1.8 Hz, 1H, H-6), 7.31 (d, J = 8.4 Hz, 1H, H-7), 7.35 (t, J = 7.5 Hz, 1H, H-4’), 7.45 (t, J = 7.5 Hz, 2H, H-3’), 7.59 (d, J = 1.8 Hz, 1H, H-4), 7.65 (d, J = 7.5 Hz, 2H, H-2’), 8.35 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 99.6 (C-3), 111.8 (C-7), 120.0 (C-4), 122.6 (C-6), 125.2 (C-2’), 125.9 (C-5), 128.1 (C-4’), 129.1 (C-3’), 130.3 (C-3a), 131.9 (C-1’), 135.1 (C-7a), 139.3 (C-2).
3.6.10. 7-Bromo-5-methyl-2-phenyl-1H-indole (6j)
Following general method B, a mixture of 2a (0.085 g, 0.83 mmol), 1k (0.163 g, 0.88 mmol), and Hg2Cl2 (0.020 g, 0.04 mmol) was stirred at 60 °C for 8 h. It was then reacted with DMSO (4.2 mL), Cu(OAc)2 (0.227 g, 1.25 mmol), and Pd(OAc)2 (0.037 g, 0.17 mmol) to obtain 6j (0.085 g, 36%) as a brown solid. Rf 0.73 (hexane/EtOAc, 7:3); mp 75–77 °C (Lit. 130–131 °C [106]). IR (film): ῡ 3446, 3036, 2918, 1605, 1568, 1477, 1449, 1395, 1310, 1214, 856, 840, 759, 734, 690 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.42 (s, 3H, CH3), 6.78 (dd, J = 2.5, 0.6 Hz, 1H, H-3), 7.18 (br s, 1H, H-6), 7.32–7.36 (m, 1H, H-4, H-4’), 7.42–7.47 (m, 2H, H-3’), 7.65–7.68 (m, 2H, H-2’), 8.32 (br s, 1H, NH). 13C NMR (125 MHz, CDCl3): δ 21.2 (CH3), 100.5 (C-3), 103.9 (C-7), 119.5 (C-4), 125.3 (C-2’), 125.9 (C-6), 128.0 (C-4’), 129.0 (C-3’), 130.4 (C-3a), 131.1 (C-5), 131.9 (C-1’), 133.8 (C-7a), 138.6 (C-2). HRMS (EI): m/z [M+] calcd for C15H12NBr: 285.0153; found: 285.0160.
3.6.11. 5-Methoxy-2-(p-tolyl)-1H-indole (6k)
Following general method B, a mixture of 2b (0.094 g, 0.81 mmol), 1i (0.105 g, 0.85 mmol), and Hg2Cl2 (0.019 g, 0.04 mmol) was stirred at 60 °C for 2 h. It was then reacted with DMSO (4.1 mL), Cu(OAc)2 (0.222 g, 1.22 mmol), and Pd(OAc)2 (0.036 g, 0.16 mmol) to give 6k (0.122 g, 64%) as a yellow solid. Rf 0.70 (hexane/EtOAc, 7:3); mp 184–186 °C (Lit. 186–187 °C [66]). IR (ATR): ῡ 3424, 1621, 1543, 1479, 1452, 1301, 1217, 1153, 1115, 1029, 823, 789 cm−1. 1H NMR (500 MHz, CDCl3): δ 2.39 (s, 3H, CH3Ar), 3.86 (s, 3H, CH3O), 6.71 (dd, J = 2.5, 1.0 Hz, 1H, H-3), 6.84 (dd, J = 8.5, 2.5 Hz, 1H, H-6), 7.08 (d, J = 2.5 Hz, 1H, H-4), 7.21–7.25 (m, 2H, H-3’), 7.26 (d, J = 8.5 Hz, 1H, H-7), 7.51–7.55 (m, 2H, H-2’), 8.17 (br s, 1H, NH). 13C NMR (125 MHz, CDCl3): δ 21.2 (CH3Ar), 55.8 (CH3O), 99.3 (C-3), 102.2 (C-4), 111.5 (C-7), 112.3 (C-6), 125.0 (C-2’), 129.6 (C-1’ or C-3a), 129.7 (C-3’), 129.8 (C-3a or C-1’), 131.9 (C-7a), 137.6 (C-4’), 138.8 (C-2), 154.5 (C-5).
3.6.12. 2-(4-Cyanophenyl)-5-methoxy-1H-indole (6l)
Following general method B, a mixture of 2c (0.078 g, 0.61 mmol), 1i (0.079 g, 0.64 mmol), and Hg2Cl2 (0.014 g, 0.03 mmol) was stirred at 60 °C for 2 h. It was then reacted with DMSO (3.1 mL), Cu(OAc)2 (0.167 g, 0.92 mmol), and Pd(OAc)2 (0.027 g, 0.12 mmol) to produce 6l (0.050 g, 33%) as yellow crystals. Rf 0.47 (hexane/EtOAc, 7:3); mp 191–192 °C (Lit. 195–196 °C [66]). IR (KBr): ῡ 3377, 2219, 1606, 1445, 1209, 1030, 836, 789 cm−1. 1H NMR (500 MHz, CDCl3): δ 3.87 (s, 3H, CH3O), 6.88 (dd, J = 1.5, 0.5 Hz, 1H, H-3), 6.92 (dd, J = 8.7, 2.5 Hz, 1H, H-6), 7.09 (d, J = 2.5 Hz, 1H, H-4), 7.31 (dd, J = 8.7, 0.5 Hz, 1H, H-7), 7.69–7.73 (m, 4H, H-2’, H-3’), 8.30 (br s, 1H, NH). 13C NMR (125 MHz, CDCl3): δ 55.8 (CH3O) 102.3 (C-4), 102.4 (C-3), 110.5 (C-4’), 112.0 (C-7), 114.3 (C-6), 118.8 (CN), 125.1 (C-2’), 129.4 (C-3a), 132.6 (C-7a), 132.8 (C-3’), 136.1 (C-2), 136.6 (C-1’), 154.8 (C-5). HRMS (EI): m/z [M+] calcd for C16H12N2O: 248.0950; found: 248.0943.
3.6.13. 5-Methoxy-2-(4-methoxyphenyl)-1H-indole (6m)
Following general method B, a mixture of 2d (0.075 g, 0.57 mmol), 1i (0.074 g, 0.60 mmol), and Hg2Cl2 (0.014 g, 0.03 mmol) was stirred at 60 °C for 2 h. It was then reacted with DMSO (3.1 mL), Cu(OAc)2 (0.156 g, 0.86 mmol), and Pd(OAc)2 (0.025 g, 0.11 mmol) to generate 6m (0.122 g, 84%) as a colorless crystalline solid. Rf 0.53 (hexane/EtOAc, 7:3); mp 218–219 °C (Lit. 218–219 °C [66]). IR (ATR): ῡ 3430, 1543, 1480, 1456, 1254, 1217, 1155, 1018, 829, 777 cm−1. 1H NMR (600 MHz, CDCl3): δ 3.85 (s, 3H, CH3O-C4’), 3.86 (s, 3H, CH3O-C5), 6.64 (d, J = 1.2 Hz, 1H, H-3), 6.83 (dd, J = 8.4, 2.4 Hz, 1H, H-6), 6.96–6.99 (m, 2H, H-3’), 7.10 (d, J = 2.4 Hz, 1H, H-4), 7.27 (d, J = 8.4 Hz, 1H, H-7), 7.56–7.59 (m, 2H, H-2’), 8.13 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 55.4 (CH3O), 55.9 (CH3O), 98.7 (C-3), 102.2 (C-4), 111.4 (C-7), 112.0 (C-6), 114.5 (C-3’), 125.3 (C-1’), 126.4 (C-2’), 129.9 (C-3a), 131.8 (C-7a), 138.7 (C-2), 154.5 (C-5), 159.3 (C-4’).
3.6.14. 5-Methoxy-2-propyl-1H-indole (6n)
Following general method B, a mixture of 2e (0.088 g, 1.29 mmol), 1i (0.166 g, 1.35 mmol), and Hg2Cl2 (0.028 g, 0.06 mmol) was stirred at 60 °C for 2 h. It was then reacted with DMSO (6.5 mL), Cu(OAc)2 (0.352 g, 1.94 mmol), and Pd(OAc)2 (0.058 g, 0.26 mmol) to form 6n (0.093 g, 38%) as an amber solid. Rf 0.67 (hexane/EtOAc, 7:3); p.f. 55–56 °C (Lit. 64–65 °C [107]). IR (ATR): ῡ 3381, 2961, 1620, 1582, 1478, 1450, 1195, 1166, 1034, 831 cm−1. 1H NMR (500 MHz, CDCl3): δ 1.00 (t, J = 7.5 Hz, 3H, H-3’), 1.73 (hex, J = 7.5 Hz, 2H, H-2’), 2.70 (t, J = 7.5 Hz, 2H, H-1’), 3.84 (s, 3H, CH3O), 6.17 (dd, J = 2.0, 1.0 Hz, 1H, H-3), 6.77 (dd, J = 8.5, 2.5 Hz, 1H, H-6), 7.00 (d, J = 2.5 Hz, H-4), 7.18 (d, J = 8.5 Hz, 1H, H-7), 7.74 (br s, 1H, NH). 13C NMR (125 MHz, CDCl3): δ 13.9 (C-3’), 22.5 (C-2’), 30.4 (C-1’), 55.9 (CH3O), 99.5 (C-3), 102.0 (C-4), 110.7 (C-6), 110.9 (C-7), 129.3 (C-3a), 130.9 (C-7a), 140.7 (C-2), 154.1 (C-5). HRMS (EI): m/z [M+] calcd for C12H15NO: 189.1154; found: 189.1153.
3.6.15. 2-Cyclopropyl-5-methoxy-1H-indole (6o)
Following general method B, a mixture of 2f (0.097 g, 1.47 mmol), 1i (0.189 g, 1.54 mmol), and Hg2Cl2 (0.033 g, 0.07 mmol) was stirred at 40 °C for 2 h. It was then reacted with DMSO (7.3 mL), Cu(OAc)2 (0.401 g, 2.21 mmol), and Pd(OAc)2 (0.065 g, 0.29 mmol) to provide 6o (0.107 g, 39%) as a yellow solid. Rf 0.67 (hexane/EtOAc, 7:3); mp 57–58 °C. IR (ATR): ῡ 3382, 2914, 1621, 1583, 1482, 1451, 1332, 1214, 1163, 1030, 837, 799, 770, 676 cm−1. 1H NMR (600 MHz, CDCl3): δ 0.73–0.76 (m, 2H, CH2), 0.91–0.95 (m, 2H, CH2), 1.87–1.93 (m, 1H, H-1’), 6.07 (dd, J = 1.2, 0.6 Hz, 1H, H-3), 6.76 (dd, J = 9.0, 2.4 Hz, 1H, H-6), 6.98 (d, J = 2.4 Hz, 1H, H-4), 7.13 (d, J = 9.0 Hz, 1H, H-7), 7.83 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 7.3 (2CH2), 8.9 (C-1’), 55.8 (CH3O), 97.5 (C-3), 101.9 (C-4), 110.7 (C-6), 110.8 (C-7), 129.1 (C-3a), 130.8 (C-7a), 142.6 (C-2), 154.1 (C-5). HRMS (EI): m/z [M+] calcd for C12H13NO: 187.0997; found: 187.0997 [66].
3.6.16. 2-(Cyclohex-1-en-1-yl)-5-methoxy-1H-indole (6p)
Following general method B, a mixture of 2g (0.096 g, 0.91 mmol), 1i (0.117 g, 0.95 mmol), and Hg2Cl2 (0.024 g, 0.05 mmol) was stirred at 40 °C for 2 h. It was then reacted with DMSO (4.5 mL), Cu(OAc)2 (0.247 g, 1.36 mmol), and Pd(OAc)2 (0.040 g, 0.18 mmol) to yield 6p (0.037 g 18%) as a brown solid. Rf 0.70 (hexane/EtOAc, 7:3); mp 108–110 °C. IR (KBr): ῡ 3429, 2929, 1620, 1586, 1483, 1452, 1414, 1210, 1147, 1033, 837, 802, 782 cm−1. 1H NMR (600 MHz, CDCl3): δ 1.66–1.71 (m, 2H, H-4’), 1.76–1.81 (m, 2H, H-5’), 2.21–2.25 (m, 2H, H-3’), 2.42–2.46 (m, 2H, H-6’), 6.09–6.11 (m, 1H, H-2’), 6.37 (d, J = 1.8 Hz, H-3), 6.80 (dd, J = 8.6, 2.4 Hz, 1H, H-6), 7.02 (d, J = 2.4 Hz, 1H, H-4), 7.18 (d, J = 8.6 Hz, 1H, H-7), 7.99 (br s, 1H, NH). 13C NMR (150 MHz, CDCl3): δ 22.2 (C-4’), 22.5 (C-5’), 25.5 (C-3’), 26.0 (C-6’), 55.8 (CH3O), 98.5 (C-3), 102.2 (C-4), 111.0 (C-7), 111.9 (C-6), 122.5 (C-2’), 129.1 (C-1’), 129.3 (C-3a), 131.3 (C-7a), 140.3 (C-2), 154.1 (C-5). HRMS (EI): m/z [M+] calcd for C15H17NO: 227.1310; found: 227.1306.
3.7. Single-Crystal X-ray Crystallography
Compounds 5i (yellow crystals, hexane/EtOAc, 98:2), 6e (colorless crystals, hexane/EtOAc, 98:2), and 6l (pale reddish crystals, hexane/EtOAc, 94:6) were prepared and mounted on glass fibers. Crystallographic measurements were performed with an area-detector with Mo Kα diffraction radiation (λ = 71,073 Å; graphite monochromator) at room temperature (Table 4). Unit cell parameters were obtained from a least-squares refinement. Intensities were corrected for Lorentz and polarization effects. Multi-scan absorption correction was applied. Anisotropic temperature factors were introduced for all non-hydrogen atoms. Hydrogen atoms were placed in accordance with electron density maps and idealized positions, and their atomic coordinates were refined by utilizing unit weights. After the structure was solved using SHELXT [108,109], it was implemented in WinGX [110], refined with SHELXL [111], and then visualized and plotted with the MERCURY program package [112].

Table 4.
Crystal data and structure refinement for 5i, 6e, and 6l.
4. Conclusions
A new Hg(I)-catalyzed hydroamination of terminal acetylenes 2a–d in the presence of anilines 1a–k generated a series of ketimines 5a–n. An efficient consecutive Hg(I)/Pd(II)-catalyzed one-pot process led to the direct conversion of 1 and 2 into a series of 2-substituted indoles 6a–p. Although good yields were found for most of the 2-arylindoles, the yields were fairly modest for halogenated, 2-alkyl, and 2-vinyl indoles. The latter limitation was possibly due to the low stability of the imine intermediate under these reaction conditions. Likewise, the direct Hg(I)-catalyzed preparation of the imines either provided low yields or made it impossible to avoid decomposition or hydrolysis to their corresponding acetophenones upon isolation from the reaction mixture. The reaction mechanism of this transformation was explored, finding that an Hg(I)–Pd(II) transmetalation step plausibly connects the consecutive cycles of Hg(I)-catalyzed enamine formation and Pd(II)-catalyzed oxidative cyclization.
Supplementary Materials
The following are available online. 1H- and 13C-NMR spectra are available online. The CIF files of crystal structures have been deposited with the CCDC, and the crystal data and torsion angles for 5i, 6e, and 6l are also provided.
Author Contributions
R.U.G. contributed by developing the method, synthesizing compounds, performing NMR and HRMS spectra, and writing the experimental section; M.H.-M. carried out compound synthesis; A.M.-M. performed IR spectra and analyzed the results; F.D. contributed by doing the literature research, analyzing the results, and reviewing the manuscript; J.T. designed the project, provided tutorial guidance, discussed the results, acquired the financial resources, and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The research was supported by the Consejo Nacional de Ciencia y Tecnología (CONACYT, Mexico) (Grants 178319, A1-S-17131, and 300520) and SIP/IPN (Grants 20170902, 20180198, 20195228, 20200227, and 20210700).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We thank M. Fernanda Ramos and Ailyn N. García and CNMN-IPN for their help in spectrometric measurements, and Bruce A. Larsen for proofreading. R.U.G. and M.H.-M. are grateful to CONACYT (SNI) for awarding them graduate and research stay scholarships, and also thank SIP/IPN (BEIFI) and the Ludwig K. Hellweg Foundation for scholarship complements. A.M.-M., F.D., and J.T. are fellows of the EDI-IPN and COFAA-IPN programs.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Not available.
References
- Layer, R.W. The chemistry of imines. Chem. Rev. 1963, 63, 489–510. [Google Scholar] [CrossRef]
- Patil, R.D.; Adimurthy, S. Catalytic methods for imine synthesis. Asian J. Org. Chem. 2013, 2, 726–744. [Google Scholar] [CrossRef]
- Kim, D.; Kang, B.; Hong, S.H. Ruthenium-catalyzed selective imine synthesis from nitriles and secondary alcohols under hydrogen acceptor- and base-free conditions. Org. Chem. Front. 2016, 3, 475–479. [Google Scholar] [CrossRef]
- Amrutham, V.; Mameda, N.; Kodumuri, S.; Chevella, D.; Banothu, R.; Gajula, K.S.; Grigor’eva, N.G.; Nama, N. Hβ Catalyzed condensation reaction between aromatic ketones and anilines: To access ketimines (imines). Catal. Lett. 2017, 147, 2982–2986. [Google Scholar] [CrossRef]
- Zhang, M.; Wu, S.; Bian, L.; Cao, Q.; Fang, W. One-pot synthesis of Pd-promoted Ce-Ni mixed oxides as efficient catalysts for imine production from the direct N-alkylation of amine with alcohol. Catal. Sci. Technol. 2019, 9, 286–301. [Google Scholar] [CrossRef]
- Carey, A.R.E.; Fukata, G.; O’Ferrall, R.A.M.; Murphy, M.G. The mechanism of imine-enamine tautomerism of 2- and 4-phenacylquinolines. J. Chem. Soc. Perkin Trans. 1985, 1711–1722. [Google Scholar] [CrossRef]
- Capon, B.; Wu, Z.-P. Comparison of the tautomerization and hydrolysis of some secondary and tertiary enamines. J. Org. Chem. 1990, 55, 2317–2324. [Google Scholar] [CrossRef]
- Ciaccia, M.; Di Stefano, S. Mechanisms of imine exchange reactions in organic solvents. Org. Biomol. Chem. 2015, 13, 646–654. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Y.; Tan, H.; Wang, Y.; Han, P.; Wang, D.Z. Enantioselective organocatalytic Mannich reactions with autocatalysts and their mimics. J. Org. Chem. 2010, 75, 2403–2406. [Google Scholar] [CrossRef]
- Kutlescha, K.; Venkanna, G.T.; Kempe, R. The potassium hydride mediated trimerization of imines. Chem. Commun. 2011, 47, 4183–4185. [Google Scholar] [CrossRef]
- Yang, Z.; He, W.; Cheng, B.; Xu, J. Stereochemistry and mechanistic insight in the [2k+2i+2i] annulations of ketenes and imines. J. Org. Chem. 2016, 81, 4506–4515. [Google Scholar] [CrossRef]
- Wu, P.; Givskov, M.; Nielsen, T.E. Reactivity and synthetic applications of multicomponent Petasis reactions. Chem. Rev. 2019, 119, 11245–11290. [Google Scholar] [CrossRef] [Green Version]
- Mir, H.; Ahmed, D. Synthesis of Schiff bases of acetophenone with aniline and its different chloro-substituted derivatives, and study of their antioxidant, enzyme inhibitory and antimicrobial properties. J. Chem. Soc. Pak. 2016, 38, 981–989. [Google Scholar]
- da Silva, C.M.; da Silva, D.L.; Modolo, L.V.; Alves, R.B.; de Resende, M.A.; Martins, C.V.B.; de Fátima, A. Schiff bases: A short review of their antimicrobial activities. J. Adv. Res. 2011, 2, 1–8. [Google Scholar] [CrossRef] [Green Version]
- da Silva, E.T.; da Silva Araújo, A.; Moraes, A.M.; de Souza, L.A.; Silva Lourenço, M.C.; de Souza, M.V.N.; Wardell, J.L.; Wardell, M.S.V. Synthesis and biological activities of camphor hydrazone and imine derivatives. Sci. Pharm. 2016, 84, 467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hille, A.; Ott, I.; Kitanovic, A.; Kitanovic, I.; Alborzinia, H.; Lederer, E.; Wölfl, S.; Metzler-Nolte, N.; Schäfer, S.; Sheldrick, W.S.; et al. [N,N’-Bis(salicylidene)-1,2-phenylenediamine]metal complexes with cell death promoting properties. J. Biol. Inorg. Chem. 2009, 14, 711–725. [Google Scholar] [CrossRef]
- Hille, A.; Gust, R. Influence of methoxy groups on the antiproliferative effects of [FeIII(salophene-OMe)Cl] complexes. Eur. J. Med. Chem. 2010, 45, 5486–5492. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.K.; Singh, R.K.; Strongin, R.M.; Moore, R.G.; Brard, L.; Lange, T.S. Organometallic iron(III)-salophene exerts cytotoxic properties in neuroblastoma cells via MAPK activation and ROS generation. PLoS ONE 2011, 6, e19049. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.; Dandawate, P.; Rub, R.; Padhye, S.; Aphale, S.; Moghe, A.; Jagyasi, A.; Swamy, K.V.; Singh, B.; Chatterjee, A.; et al. Novel aza-resveratrol analogs: Synthesis, characterization and anticancer activity against breast cancer cell lines. Bioorg. Med. Chem. Lett. 2013, 23, 635–640. [Google Scholar] [CrossRef]
- Abdalla, F.; Nookala, A.; Padhye, S.B.; Kumar, A.; Bhat, H.K. 4-€-{(p-Tolylimino)-methylbenezene-1,2-diol} (TIMBD) suppresses HIV1-gp120 mediated production of IL6 and IL8 but not CCL5. Sci. Rep. 2017, 7, 8129. [Google Scholar] [CrossRef]
- Stivala, C.E.; Benoit, E.; Aráoz, R.; Servent, D.; Novikov, A.; Molgó, J.; Zakarian, A. Synthesis and biology of cyclic imine toxins, an emerging class of potent, globally distributed marine toxins. Nat. Prod. Rep. 2015, 32, 411–435. [Google Scholar] [CrossRef]
- Kochanowska-Karamyan, A.J.; Hamann, M.T. Marine indole alkaloids: Potential new drug leads for the control of depression and anxiety. Chem. Rev. 2010, 110, 4489–4497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, R.B.; Hu, J.-F.; Olson, K.M.; Norman, V.L.; Goering, M.G.; O’Neil-Johnson, M.; Eldridge, G.R.; Starks, C.M. Antibiotic indole sesquiterpene alkaloid from Greenwayodendron suaveolens with a new natural product framework. J. Nat. Prod. 2010, 73, 1008–1011. [Google Scholar] [CrossRef] [PubMed]
- Ciulla, M.G.; Kumar, K. The natural and synthetic indole weaponry against bacteria. Tetrahedron Lett. 2018, 59, 3223–3233. [Google Scholar] [CrossRef]
- Leboho, T.C.; Michael, J.P.; van Otterlo, W.A.L.; van Vuuren, S.F.; de Koning, C.B. The synthesis of 2- and 3-aryl indoles and 1,3,4,5-tetrahydropyrano [4,3-b]indoles and their antibacterial and antifungal activity. Bioorg. Med. Chem. Lett. 2009, 19, 4948–4951. [Google Scholar] [CrossRef]
- Kaushik, N.K.; Kaushik, N.; Attri, P.; Kumar, N.; Kim, C.H.; Verma, A.K.; Choi, E.H. Biomedical importance of indoles. Molecules 2013, 18, 6620–6662. [Google Scholar] [CrossRef]
- Melander, R.J.; Minvielle, M.J.; Melander, C. Controlling bacterial behavior with indole-containing natural products and derivatives. Tetrahedron 2014, 70, 6363–6372. [Google Scholar] [CrossRef] [Green Version]
- Han, X.; Wu, H.; Wang, W.; Dong, C.; Tien, P.; Wu, S.; Zhou, H.-B. Synthesis and SARs of indole-based α-amino acids as potent HIV-1 non-nucleoside reverse transcriptase inhibitors. Org. Biomol. Chem. 2014, 12, 8308–8317. [Google Scholar] [CrossRef]
- Sanphui, P.; Bolla, G.; Das, U.; Mukherjee, A.K.; Nangia, A. Acemetacin polymorphs: A rare case of carboxylic acid catemer and dimer synthons. CrystEngComm 2013, 15, 34–38. [Google Scholar] [CrossRef]
- Zhang, M.-Z.; Chen, Q.; Yang, G.-F. A review on recent developments of indole-containing antiviral agents. Eur. J. Med. Chem. 2015, 89, 421–441. [Google Scholar] [CrossRef]
- Wright, Z.V.F.; Wu, N.C.; Kadam, R.U.; Wilson, I.A.; Wolan, D.W. Structure-based optimization and synthesis of antiviral drug Arbidol analogues with significantly improved affinity to influenza hemagglutinin. Bioorg. Med. Chem. Lett. 2017, 27, 3744–3748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wishart, D.S.; Knox, C.; Guo, A.C.; Shrivastava, S.; Hassanali, M.; Stothard, P.; Chang, Z.; Woolsey, J. DrugBank: A comprehensive resource for in silico drug discovery and exploration. Nucleic Acids Res. 2006, 34, D668–D672. [Google Scholar] [CrossRef] [PubMed]
- Vitaku, E.; Smith, D.T.; Njardarson, J.T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 2014, 57, 10257–10274. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Cao, R.; Zhang, H.; Liu, J.; Xu, M.; Hu, H.; Li, Y.; Zhao, L.; Li, W.; Sun, X.; et al. The anti-influenza virus drug, arbidol is an efficient inhibitor of SARS-CoV-2 in vitro. Cell Discov. 2020, 6, 28–32. [Google Scholar] [CrossRef]
- Zhu, Z.; Lu, Z.; Xu, T.; Chen, C.; Yang, G.; Zha, T.; Lu, J.; Xue, Y. Arbidol monotherapy is superior to lopinavir/ritonavir in treating COVID-19. J. Infect. 2020, 81, e21–e23. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Tang, L.V.; Hu, Y. Treatment in the COVID-19 pandemic: An update on clinical trials. Expert Opin. Emerg. Drugs 2020, 25, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Ke, C.; Yue, D.; Li, W.; Hu, Z.; Liu, W.; Hu, S.; Wang, S.; Liu, J. Effectiveness of arbidol for COVID-19 prevention in health professionals. Front. Public Health 2020, 8, 249. [Google Scholar] [CrossRef] [PubMed]
- Barton, D.H.R.; Motherwell, W.B.; Simon, E.S.; Zard, S.Z. Reduction of oximes and aliphatic nitro compounds to imines for further in situ reactions: A novel synthesis of pyrroles and pyrrolin-2-ones. J. Chem. Soc. Perkin Trans. I 1986, 2243–2252. [Google Scholar] [CrossRef]
- Kitamura, M.; Zaman, S.; Narasaka, K. Synthesis of spiro imines from oximes by palladium-catalyzed cascade reaction. Synlett 2001, SI, 974–976. [Google Scholar] [CrossRef] [Green Version]
- Huang, F.; Zhang, S. Iminyl radicals by reductive cleavage of N–O bond in oxime ether promoted by SmI2: A straightforward synthesis of five-membered cyclic imines. Org. Lett. 2019, 21, 7430–7434. [Google Scholar] [CrossRef]
- Chakraborty, S.; Milstein, D. Selective hydrogenation of nitriles to secondary imines catalyzed by an iron pincer complex. ACS Catal. 2017, 7, 3968–3972. [Google Scholar] [CrossRef]
- Weiberth, F.J.; Hall, S.S. Copper(I)-activated addition of Grignard reagents to nitriles. Synthesis of ketimines, ketones, and amines. J. Org. Chem. 1987, 52, 3901–3904. [Google Scholar] [CrossRef]
- Feringa, B.L.; Jansen, J.F.G.A. A new synthesis of imines via Grignard- and cuprate additions to N-trimethylsilylformamides. Synthesis 1988, 3, 184–186. [Google Scholar] [CrossRef] [Green Version]
- Maggi, A.; Madsen, R. Dehydrogenative synthesis of imines from alcohols and amines catalyzed by a ruthenium N-heterocyclic carbene complex. Organometallics 2012, 31, 451–455. [Google Scholar] [CrossRef] [Green Version]
- Chen, B.; Wang, L.; Gao, S. Recent advances in aerobic oxidation of alcohols and amines to imines. ACS Catal. 2015, 5, 5851–5876. [Google Scholar] [CrossRef]
- Bennett, J.; Meldi, K.; Kimmell, C., II. Synthesis and analysis of a versatile imine for the undergraduate organic chemistry laboratory. J. Chem. Educ. 2006, 83, 1221–1224. [Google Scholar] [CrossRef]
- Look, G.C.; Murphy, M.M.; Campbell, D.A.; Gallop, M.A. Trimethylorthoformate: A mild and effective dehydrating reagent for solution and solid phase imine formation. Tetrahedron Lett. 1995, 36, 2937–2940. [Google Scholar] [CrossRef]
- Mirabdolbaghi, R.; Dudding, T. Expanding the forefront of strong organic Brønsted acids: Proton-catalyzed hydroamination of unactivated alkenes and activation of Au(I) for alkyne hydroamination. Org. Lett. 2015, 17, 1930–1933. [Google Scholar] [CrossRef]
- Mir, R.; Dudding, T.A. Au(I)-precatalyst with a cyclopropenium counterion: An unusual ion pair. J. Org. Chem. 2016, 81, 2675–2679. [Google Scholar] [CrossRef]
- Nagata, T.; Adachi, Y.; Obora, Y. Thiolate-protected Au25(SC2H4Ph)18 nanoclusters as a catalyst for intermolecular hydroamination of terminal alkynes. Synlett 2018, 29, 2655–2659. [Google Scholar]
- Casnati, A.; Voronov, A.; Ferrari, D.G.; Mancuso, R.; Gabriele, B.; Motti, E.; Della Ca’, N. PdI2 as a simple and efficient catalyst for the hydroamination of arylacetylenes with anilines. Catalysts 2020, 10, 176. [Google Scholar] [CrossRef] [Green Version]
- Barluenga, J.; Aznar, F.; Valdés, C.; Cabal, M.-P. Catalytic aminomercuration reactions of 3-alken-1-ynes: An improved method for the synthesis of 2-amino-1,3-butadienes and 1-aza-1,3-butadienes. J. Org. Chem. 1991, 56, 6166–6171. [Google Scholar] [CrossRef]
- Barluenga, J.; Aznar, F.; Liz, R.; Rodes, R. Catalytic and non-catalytic additions of aromatic amines to terminal acetylenes in the presence of mercury(II) chloride and acetate. J. Chem. Soc. Perkin Trans. I 1980, 2732–2737. [Google Scholar] [CrossRef]
- Barluenga, J.; Aznar, F.; Liz, R.; Rodes, R. Alkynylmercury chloride or acetate as intermediates in the mercury(II) salt-promoted addition of aliphatic and aromatic amines to terminal acetylenes. J. Chem. Soc. Perkin Trans. I 1983, 1087–1091. [Google Scholar] [CrossRef]
- Gribble, G.W. Indole Ring Synthesis from Natural Products to Drugs Discovery; John Wiley & Sons Ltd.: Chichester, UK, 2016. [Google Scholar]
- Wagaw, S.; Yang, B.H.; Buchwald, S.L. A palladium-catalyzed method for the preparation of indoles via the Fischer indole synthesis. J. Am. Chem. Soc. 1999, 121, 10251–10263. [Google Scholar] [CrossRef]
- Heravi, M.M.; Rohani, S.; Zadsirjan, V.; Zahedi, N. Fischer indole synthesis applied to the total synthesis of natural products. RSC Adv. 2017, 7, 52852. [Google Scholar] [CrossRef] [Green Version]
- Baudin, J.-B.; Julia, S.A. Synthesis of indoles from N-aryl-1-alkenylsuphinamides. Tetrahedron Lett. 1986, 27, 837–840. [Google Scholar] [CrossRef]
- Bartoli, G.; Dalpozzo, R.; Nardi, M. Applications of Bartoli indole synthesis. Chem. Soc. Rev. 2014, 43, 4728–4750. [Google Scholar] [CrossRef]
- Gassman, P.G.; van Bergen, T.J.; Gruetzmacher, G. Use of halogen-sulfide complexes in the synthesis of indoles, oxindoles, and alkylated aromatic amines. J. Am. Chem. Soc. 1973, 95, 6508–6509. [Google Scholar] [CrossRef]
- Castro, C.E.; Gaughan, E.J.; Owsley, D.C. Indoles, benzofurans, phthalides, and tolanes via copper(I) acetylides. J. Org. Chem. 1966, 31, 4071–4078. [Google Scholar] [CrossRef]
- Larock, R.C.; Yum, E.K.; Refvik, M.D. Synthesis of 2,3-disubstituted indoles via palladium-catalyzed annulation of internal alkynes. J. Org. Chem. 1998, 63, 7652–7662. [Google Scholar] [CrossRef]
- Huestis, M.P.; Chan, L.; Stuart, D.R.; Fagnou, K. The vinyl moiety as a handle for regiocontrol in the preparation of unsymmetrical 2,3-aliphatic-susbtituted indoles and pyrroles. Angew. Chem. Int. Ed. 2011, 50, 1338–1341. [Google Scholar] [CrossRef] [PubMed]
- Ansari, N.H.; Dacko, C.A.; Akhmedov, N.G.; Söderberg, B.C.G. Double palladium catalyzed reductive cyclizations. Synthesis of 2,2’-, 2,3’, and 3,3’-bi-1H-indoles, indolo[3,2-b]indoles, and indolo[2,3-b]indoles. J. Org. Chem. 2016, 81, 9337–9349. [Google Scholar] [CrossRef] [PubMed]
- Ansari, N.H.; Banini, S.; Cummings, M.M.; Söderberg, B.C.G. Palladium catalyzed double reductive cyclization of 2,3-dinitro-1,4-dialkenylbenzenes. Synthesis of 1H,8H-pyrrolo[3,2-g]indoles. J. Org. Chem. 2020, 85, 4002–4010. [Google Scholar] [CrossRef]
- Wei, Y.; Deb, I.; Yoshikai, N. Palladium-catalyzed aerobic oxidative cyclization of N-aryl imines: Indoles synthesis from anilines and ketones. J. Am. Chem. Soc. 2012, 134, 9098–9101. [Google Scholar] [CrossRef] [PubMed]
- Ren, L.; Nan, G.; Wang, Y.; Xiao, Z. Carboxylic acid promoted single-step indole construction from simple anilines and ketones via aerobic cross-dehydrogenative coupling. J. Org. Chem. 2018, 83, 14472–14488. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.-S.; Dai, L.; Zhou, Y.; Zhang, X. Palladium-catalyzed tandem oxidative annulation of α-amino ketones leading to 2-aroylindoles. Tetrahedron 2020, 76, 130917. [Google Scholar] [CrossRef]
- Bautista, R.; Montoya, P.A.; Rebollar, A.; Burgueño, E.; Tamariz, J. Palladium-catalyzed synthesis of natural and unnatural 2-, 5-, and 7-oxygenated carbazole alkaloids from N-arylcyclohexane enaminones. Molecules 2013, 18, 10334–10351. [Google Scholar] [CrossRef] [Green Version]
- Labarrios, E.; Jerezano, A.; Jiménez, F.; Cruz, M.C.; Delgado, F.; Zepeda, L.G.; Tamariz, J. Efficient synthetic approach to substituted benzo[b]furans and benzo[b]thiophenes by iodine-promoted cyclization of enaminones. J. Heterocycl. Chem. 2014, 51, 954–971. [Google Scholar] [CrossRef]
- Espinoza-Hicks, C.; Montoya, P.; Bautista, R.; Jiménez-Vázquez, H.A.; Rodríguez-Valdez, L.M.; Camacho-Dávila, A.A.; Cossío, F.P.; Delgado, F.; Tamariz, J. Synthesis of exo-imidazolidin-2-one dienes, their isomerization, and selectivity in Diels-Alder cycloadditions. J. Org. Chem. 2018, 83, 5347–5364. [Google Scholar] [CrossRef] [PubMed]
- Cruz, M.C.; Jiménez, F.; Delgado, F.; Tamariz, J. Regioselective and versatile synthesis of indoles via intramolecular Friedel-Crafts heteroannulation of enaminones. Synlett 2006, 5, 749–755. [Google Scholar]
- Jerezano, A.V.; Labarrios, E.M.; Jiménez, F.E.; Cruz, M.C.; Pazos, D.C.; Gutiérrez, R.U.; Delgado, F.; Tamariz, J. Iodine-mediated one-pot synthesis of indoles and 3-dimethylaminoindoles via annulation of enaminones. Arkivoc 2014, iii, 18–53. [Google Scholar] [CrossRef] [Green Version]
- Hernández-Benitez, R.I.; Zárate-Zárate, D.; Delgado, F.; Tamariz, J. Palladium-catalyzed synthesis of diarylamines and 1- and 2-oxygenated carbazoles: Total syntheses of natural alkaloids clauraila A, clausenal, clausine P, and 7-methoxy-O-methylmukonal. Synthesis 2017, 49, 4357–4371. [Google Scholar]
- Escalante, C.H.; Martínez-Mora, E.I.; Espinoza-Hicks, C.; Camacho-Dávila, A.A.; Ramos-Morales, F.R.; Delgado, F.; Tamariz, J. Highly selective Diels–Alder and Heck arylation reactions in a divergent synthesis of isoindolo- and pyrrolo-fused polycyclic indoles from 2-formylpyrrole. Beilstein J. Org. Chem. 2020, 16, 1320–1334. [Google Scholar] [CrossRef]
- Gryff-Keller, A.; Kraska-Dziadecka, A.; Molchanov, S.; Wodyński, A. Shielding and indirect spin-spin coupling tensors in the presence of a heavy atom: An experimental and theoretical study of bis(phenylethynyl)mercury. J. Phys. Chem. A 2012, 116, 10615–10620. [Google Scholar] [CrossRef] [PubMed]
- Kadam, A.R.; Nair, G.B.; Dhoble, S.J. Insights into the extraction of mercury from fluorescent lamps: A review. J. Environ. Chem. Eng. 2019, 7, 103279. [Google Scholar] [CrossRef]
- Ozkan, S.A.; Uslu, B. From mercury to nanosensors: Past, present and the future perspective of electrochemistry in pharmaceutical biomedical analysis. J. Pharm. Biomed. Anal. 2016, 130, 126–140. [Google Scholar] [CrossRef]
- Hutchings, G.J.; Grady, D.T. Hydrochlorination of acetylene: The effect of mercuric chloride concentration on catalyst life. Appl. Catal. 1985, 17, 155–160. [Google Scholar] [CrossRef]
- Larock, R.C. Organomercurials in organic synthesis. Tetrahedron 1982, 38, 1713–1754. [Google Scholar] [CrossRef]
- Larock, R.C. Organomercury Compounds in Organic Synthesis. In Reactivity and Structure Concepts in Organic Chemistry; Hafner, K., Lehn, J.-M., Rees, C.W., von Ragué Schleyer, P., Trost, B.M., Zahradnik, R., Eds.; Springer: Berlin, Germany, 1985; Volume 22. [Google Scholar]
- Zheng, M.; Chen, K.; Zhu, S. One-pot synthesis of indole derivatives from the reaction of nitroalkynes and alkynes via mercury-carbene intermediate. Synthesis 2017, 49, 4173–4182. [Google Scholar]
- Pun-Hua, L.; Lei, W. Mercurous chloride catalyzed Mannich condensation of terminal alkynes with secondary amines and aldehydes. Chin. J. Chem. 2005, 23, 1076–1080. [Google Scholar] [CrossRef]
- Dehghanpour, S.; Jahani, K.; Mahmoudi, A.; Babakhodaverdi, M.; Notash, B. In situ hydrothermal synthesis of 2D mercury(I)-organic framework from 3-aminopyrazine-2-carboxylic acid and mercury(II) acetate. Inorg. Chem. Commun. 2012, 25, 79–82. [Google Scholar] [CrossRef]
- Gutiérrez, R.U.; Correa, H.C.; Bautista, R.; Vargas, J.L.; Jerezano, A.V.; Delgado, F.; Tamariz, J. Regioselective synthesis of 1,2-dihydroquinolines by a solvent-free MgBr2-catalyzed multicomponent reaction. J. Org. Chem. 2013, 78, 9614–9626. [Google Scholar] [CrossRef]
- Zangade, S.; Patil, P. A review on solvent-free methods in organic synthesis. Curr. Org. Chem. 2019, 23, 2295–2318. [Google Scholar] [CrossRef]
- Ol’dekop, Y.A.; Maier, N.A. Decarboxylation of mercury salts of organic acids under the action of peroxides and ultraviolet radiation. Russ. Chem. Bull. 1966, 15, 1127–1132. [Google Scholar] [CrossRef]
- Whitesides, G.M.; Hackett, M.; Brainard, R.L.; Lavalleye, J.-P.P.M.; Sowinski, A.F.; Izumi, A.N.; Moore, S.S.; Brown, D.W.; Satudt, E.M. Suppression of unwanted heterogeneous platinum(0)-catalyzed reactions by poisoning with mercury(0) in systems involving competing homogeneous reactions of soluble organoplatinum compounds: Thermal decomposition of bis(triethylphosphine)-3,3,4,4-tetramethylplatinacyclopentane. Organometallics 1985, 4, 1819–1830. [Google Scholar]
- Guan, Z.-H.; Yan, Z.-Y.; Ren, Z.-H.; Liu, X.-Y.; Liang, Y.-M. Preparation of indoles via iron catalyzed direct oxidative coupling. Chem. Commun. 2010, 46, 2823–2825. [Google Scholar] [CrossRef]
- Neumann, J.J.; Rakshit, S.; Dröge, T.; Würtz, S.; Glorius, F. Exploring the oxidative cyclization of substituted N-aryl enamines: Pd-catalyzed formation of indoles from anilines. Chem. Eur. J. 2011, 17, 7298–7303. [Google Scholar] [CrossRef]
- Shi, Z.; Zhang, C.; Li, S.; Pan, D.; Ding, S.; Cui, Y.; Jiao, N. Indoles from simple anilines and alkynes: Palladium-catalyzed C-H activation using dioxygen as the oxidant. Angew. Chem. Int. Ed. 2009, 48, 4572–4576. [Google Scholar] [CrossRef] [PubMed]
- Larock, R.C.; Harrison, L.W.; Hsu, M.H. Heteroannulation vis intramolecular (p-allyl)palladium displacement. J. Org. Chem. 1984, 49, 3662–3664. [Google Scholar] [CrossRef]
- Larock, R.C.; Riefling, B.; Fellows, C.A. Mercury in organic chemistry. 12. Synthesis of β-chloro-Δα,β-buetnolides via mercuration-carbonylation of propargylic alcohols. J. Org. Chem. 1984, 49, 131–137. [Google Scholar]
- Siemsen, P.; Livingston, R.C.; Diederich, F. Acetylenic coupling: A powerful tool in molecular construction. Angew. Chem. Int. Ed. 2000, 39, 2632–2657. [Google Scholar] [CrossRef]
- Gautier, F.-M.; Jones, S.; Martin, S.J. Asymmetric reduction of ketimines with trichlorosilane employing an imidazole derived organocatalyst. Org. Biomol. Chem. 2009, 7, 229–231. [Google Scholar] [CrossRef]
- Erken, C.; Hindemith, C.; Weyhermüller, T.; Hölscher, M.; Werlé, C.; Leitner, W. Hydroamination of aromatic alkynes to imines catalyzed by Pd(II)-anthraphos complexes. ACS Omega 2020, 5, 8912–8918. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.-Y.; Ding, P.; Huang, J.-S.; Che, C.-M. Synthesis of substituted 1,2-dihydroquinolines and quinolines from aromatic amines and alkynes by gold(I)-catalyzed tandem hydroamination-hydroarylation under microwave-assisted conditions. Org. Lett. 2007, 9, 2645–2648. [Google Scholar] [CrossRef]
- Yoshii, R.; Hirose, A.; Tanaka, K.; Chujo, Y. Functionalization of boron diiminates with unique optical properties: Multicolor tuning of crystallization-induced emission and introduction into the main chain of conjugated polymers. J. Am. Chem. Soc. 2014, 136, 18131–18139. [Google Scholar] [CrossRef]
- Imamoto, T.; Iwadate, N.; Yoshida, K.E. Enantioselective hydrogenation of acyclic aromatic N-aryl imines catalyzed by an iridium complex of (S,S)-1,2-bis(tert-butylmethylphosphino)ethane. Org. Lett. 2006, 8, 2289–2292. [Google Scholar] [CrossRef]
- Suslick, B.A.; Tilley, T.D. Olefin hydroarylation catalyzed by a single-component cobalt(-I) complex. Org. Let. 2021, 23, 1495–1499. [Google Scholar] [CrossRef]
- Deprez, N.R.; Kalyani, D.; Krause, A.; Sanford, M.S. Room temperature palladium-catalyzed 2-arylation of indoles. J. Am. Chem. Soc. 2006, 128, 4972–4973. [Google Scholar] [CrossRef]
- Shen, M.; Leslie, B.E.; Driver, T.G. Dirhodium-catalyzed intramolecular C-H amination of aryl azides. Angew. Chem. Int. Ed. 2008, 47, 5056–5059. [Google Scholar] [CrossRef]
- Xia, X.-D.; Xuan, J.; Wang, Q.; Lu, L.-Q.; Chen, J.-R.; Xiao, W.-J. Synthesis of 2-substituted indoles through visible light-induced photocatalytic cyclizations of styryl azides. Adv. Synth. Catal. 2014, 356, 2807–2812. [Google Scholar] [CrossRef]
- Liu, C.; Ding, D.; Guo, G.; Liu, W.; Yang, F.-L. Palladium-catalyzed direct arylation of indoles with arylsulfonyl hydrazides. Org. Biomol. Chem. 2016, 14, 2824–2827. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, Y.; Jiang, L.; Zhang, R.; Jin, K.; Zhao, D.; Duan, C. Ready synthesis of free N-H 2-arylindoles via the copper-catalyzed amination of 2-bromo-arylacetylenes with aqueous ammonia and sequential intramolecular cyclization. Org. Biomol. Chem. 2011, 9, 4983–4986. [Google Scholar] [CrossRef] [PubMed]
- Cacchi, S.; Fabrizi, G.; Goggiamani, A.; Iazetti, A.; Verdiglione, R. A facile palladium-catalyzed route to 2,5,7-trisubstituted indoles. Tetrahedron 2015, 71, 9346–9356. [Google Scholar] [CrossRef]
- Trabbic, C.J.; Dietsch, H.M.; Alexander, E.M.; Nagy, P.I.; Robinson, M.W.; Overmeyer, J.H.; Maltese, W.A.; Erhardt, P.W. Differential induction of cytoplasmic vacuolization and methuosis by novel 2-indolyl-substituted pyridinylpropenones. ACS Med. Chem. Lett. 2014, 5, 73–77. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [Green Version]
- Sheldrick, G.M. SHELXT–Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A 2015, 71, 3–8. [Google Scholar] [CrossRef] [Green Version]
- Farrugia, L.J. WinGX suite for small-molecule single-crystal crystallography. J. Appl. Crystallogr. 1999, 32, 837–838. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- Macrae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From visualization to analysis, design and prediction. J. Appl. Cryst. 2020, 53, 226–235. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).