Characterization of the Aroma Profile and Main Key Odorants of Espresso Coffee
Abstract
1. Introduction
1.1. History
1.2. Main Coffee Species and Uses
1.3. Espresso Coffee
2. An Overview of EC Aroma: Arabica and Robusta
2.1. Aroma Profile of Espresso Coffee (EC)
2.2. Espresso Coffee (EC) Key Odorant Compounds
2.3. Sensory Attributes of Espresso Coffee and Their Relation to the Volatile Profile
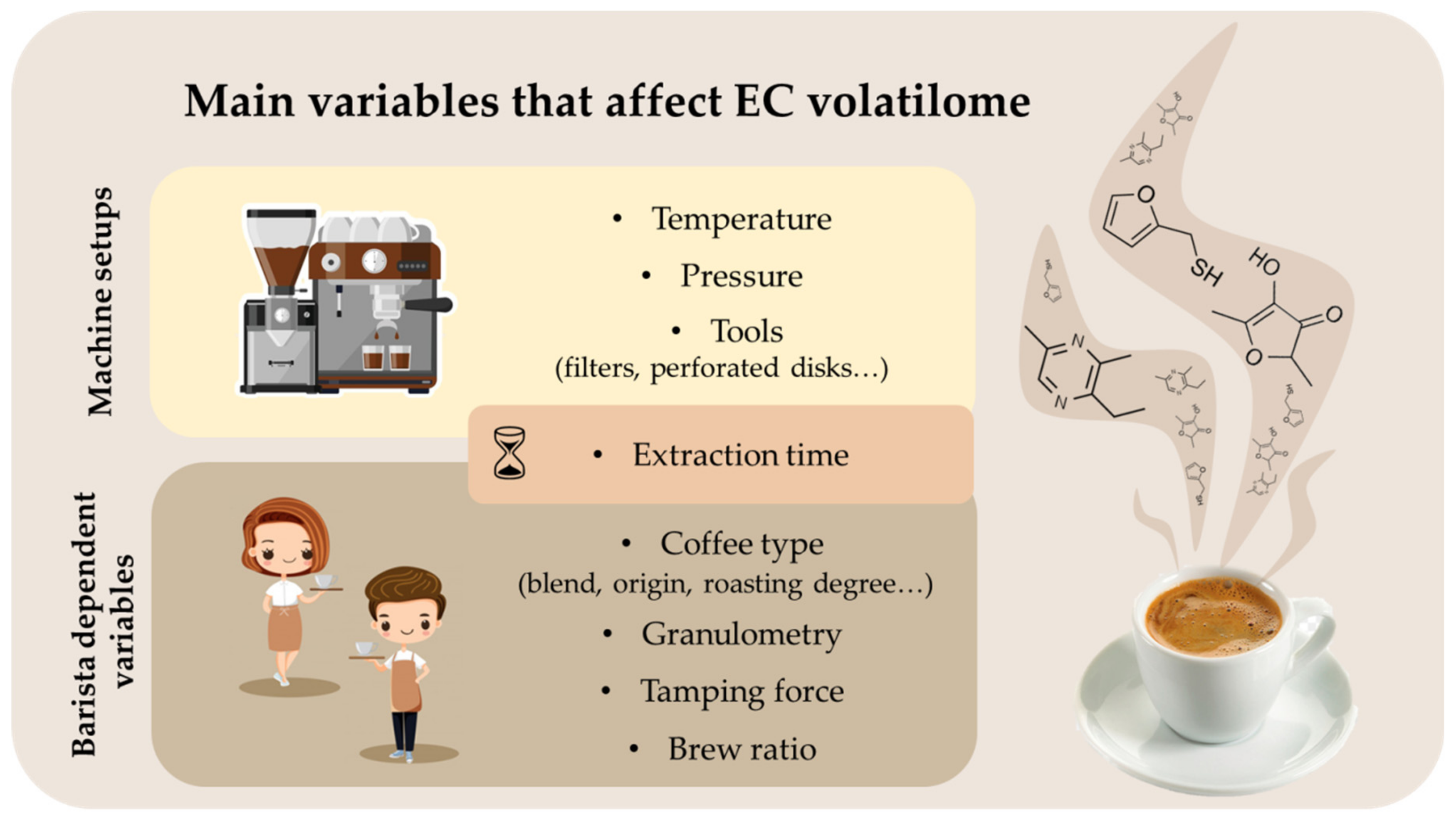
3. EC Machine Parameters and Their Influence on the EC Flavor
3.1. Extraction Time
3.2. Water Temperature
3.3. Pressure
3.4. Particle Sizes
4. Analytical Methods for the Determination of VOC in Espresso Coffee
4.1. Gas Chromatography Methods for the Analysis of VOC in EC
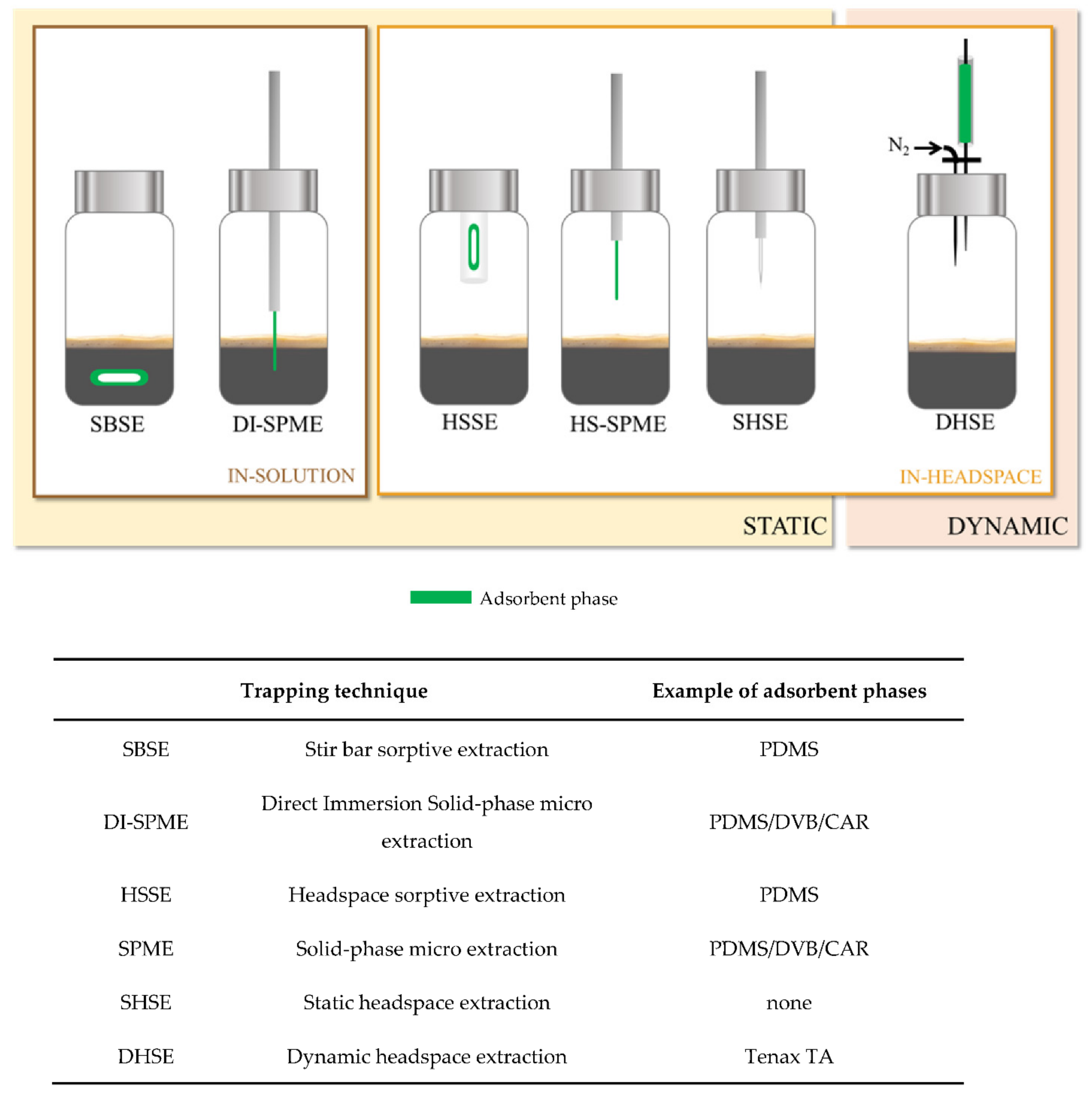
4.1.1. Sample Preparation Techniques
Solvent Extraction Method
Static Headspace Extraction
HS-Solid Phase Microextraction (HS-SPME) Technique
Dynamic Headspace (DHS) Technique
Other HS Sampling Techniques
4.1.2. Gas Chromatography (GC) Separation
4.1.3. GC Detecting Systems in EC Analysis
4.2. Electronic Nose (EN) Sensing in the VOCs Analysis of EC
4.3. Proton Transfer Reaction-MS (PTR-MS) Techniques
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lannigan, J. Making a space for taste: Context and discourse in the specialty coffee scene. Int. J. Inf. Manag. 2019. [Google Scholar] [CrossRef]
- Pham, Y.; Reardon-Smith, K.; Mushtaq, S.; Cockfield, G. The impact of climate change and variability on coffee production: A systematic review. Clim. Change 2019. [Google Scholar] [CrossRef]
- Gloess, A.N.; Schönbächler, B.; Klopprogge, B.; D’Ambrosio, L.; Chatelain, K.; Bongartz, A.; Strittmatter, A.; Rast, M.; Yeretzian, C. Comparison of nine common coffee extraction methods: Instrumental and sensory analysis. Eur. Food Res. Technol. 2013, 236, 607–627. [Google Scholar] [CrossRef]
- Buratti, S.; Benedetti, S.; Giovanelli, G. Application of electronic senses to characterize espresso coffees brewed with different thermal profiles. Eur. Food Res. Technol. 2017, 243, 511–520. [Google Scholar] [CrossRef]
- Illy, A.; Viani, R.; Liverani, F.S.; Petracco, M. Il Caffè Espresso: La Scienza della Qualità; Egea: Milan, Italy, 2009. [Google Scholar]
- Angeloni, S.; Navarini, L.; Sagratini, G.; Torregiani, E.; Vittori, S.; Caprioli, G. Development of an extraction method for the quantification of lignans in espresso coffee by using HPLC-MS/MS triple quadrupole. J. Mass Spectrom. 2018, 53. [Google Scholar] [CrossRef]
- Morris, J. Making Italian Espresso, Making Espresso Italian. Food Hist. 2010, 8, 155–183. [Google Scholar] [CrossRef]
- Khamitova, G.; Angeloni, S.; Borsetta, G.; Xiao, J.; Maggi, F.; Sagratini, G.; Vittori, S.; Caprioli, G. Optimization of espresso coffee extraction through variation of particle sizes, perforated disk height and filter basket aimed at lowering the amount of ground coffee used. Food Chem. 2020, 314. [Google Scholar] [CrossRef]
- Khamitova, G.; Angeloni, S.; Fioretti, L.; Ricciutelli, M.; Sagratini, G.; Torregiani, E.; Vittori, S.; Caprioli, G. The impact of different filter baskets, heights of perforated disc and amount of ground coffee on the extraction of organics acids and the main bioactive compounds in espresso coffee. Food Res. Int. 2020, 133, 109220. [Google Scholar] [CrossRef]
- Andueza, S.; Maeztu, L.; Dean, B.; de Pena, M.P.; Bello, J.; Cid, C.; De Peña, M.P.; Bello, J.; Cid, C. Influence of water pressure on the final quality of arabica espresso coffee. Application of multivariate analysis. J. Agric. Food Chem. 2002, 50, 7426–7431. [Google Scholar] [CrossRef]
- Severini, C.; Ricci, I.; Marone, M.; Derossi, A.; De Pilli, T. Changes in the aromatic profile of espresso coffee as a function of the grinding grade and extraction time: A study by the electronic nose system. J. Agric. Food Chem. 2015, 63, 2321–2327. [Google Scholar] [CrossRef]
- Online Etymology Dictionary. Coffee—Search Online Etymology Dictionary. Available online: https://www.etymonline.com/search?q=coffee (accessed on 25 April 2021).
- Yılmaz, B.; Acar-Tek, N.; Sözlü, S. Turkish cultural heritage: A cup of coffee. J. Ethn. Foods 2017, 4, 213–220. [Google Scholar] [CrossRef]
- Bond, T.J. The Origins of Tea, Coffee and Cocoa as Beverages. In Teas, Cocoa and Coffee: Plant Secondary Metabolites and Health; Wiley-Blackwell: Oxford, UK, 2011; pp. 1–24. [Google Scholar]
- Folmer, B. The Craft and Science of Coffee, 1st ed.; Elsevier: London, UK, 2017; ISBN 9780128035580. [Google Scholar]
- Teketay, D. History, botany and ecological requirements of coffee. Walia 1999, 20, 28–50. [Google Scholar]
- Locatelli, E. La Pavoni. 1905–2005; La Pavioni S.p.A.: Milan, Italy, 2005. [Google Scholar]
- Morris, J. Why espresso? Explaining changes in European coffee preferences from a production of culture perspective. Eur. Rev. Hist. Rev. Eur. d’Hist. 2013, 20, 881–901. [Google Scholar] [CrossRef]
- The Plant List. 2013 Coffea—The Plant List. 2013. Available online: http://www.theplantlist.org/1.1/browse/A/Rubiaceae/Coffea/ (accessed on 12 November 2019).
- Ghosh, P.; Venkatachalapathy, N. Processing and Drying of Coffee—A review. Int. J. Eng. Res. Technol. 2014, 3, 784–794. [Google Scholar]
- Murthy, P.S.; Madhava Naidu, M. Sustainable management of coffee industry by-products and value addition—A review. Resour. Conserv. Recycl. 2012, 66, 45–58. [Google Scholar] [CrossRef]
- Dórea, J.G.; da Costa, T.H.M. Is coffee a functional food? Br. J. Nutr. 2005, 93, 773–782. [Google Scholar] [CrossRef]
- Brien, D.L.; Adams, J. Coffee: A Cultural and Media Focussed Approach. M/C J. 2012, 15. [Google Scholar] [CrossRef]
- Homan, D.J.; Mobarhan, S. Coffee: Good, Bad, or Just Fun? A Critical Review of Coffee’s Effects on Liver Enzymes. Nutr. Rev. 2006, 64, 43–46. [Google Scholar] [CrossRef]
- Sunarharum, W.B.; Williams, D.J.; Smyth, H.E. Complexity of coffee flavor: A compositional and sensory perspective. Food Res. Int. 2014, 62, 315–325. [Google Scholar] [CrossRef]
- Caprioli, G.; Cortese, M.; Sagratini, G.; Vittori, S. The influence of different types of preparation (espresso and brew) on coffee aroma and main bioactive constituents. Int. J. Food Sci. Nutr. 2015, 66, 505–513. [Google Scholar] [CrossRef]
- Lane, S.; Palmer, J.; Christie, B.; Ehlting, J.; Le, C. Can Cold Brew Coffee Be Convenient? A Pilot Study For Caffeine Content in Cold Brew Coffee Concentrate Using High Performance Liquid Chromatography. Arbutus Rev. 2017, 8, 15–23. [Google Scholar] [CrossRef]
- Fuller, M.; Rao, N.Z. The Effect of Time, Roasting Temperature, and Grind Size on Caffeine and Chlorogenic Acid Concentrations in Cold Brew Coffee. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Odello, L.; Odello, C. Espresso Italiano Tasting; Centro Studi Assaggiatori: Brescia, Italy, 2006. [Google Scholar]
- Zhang, C.; Linforth, R.; Fisk, I.D. Cafestol extraction yield from different coffee brew mechanisms. Food Res. Int. 2012, 49, 27–31. [Google Scholar] [CrossRef]
- Jeszka-Skowron, M.; Zgoła-Grześkowiak, A.; Grześkowiak, T. Analytical methods applied for the characterization and the determination of bioactive compounds in coffee. Eur. Food Res. Technol. 2014, 240, 19–31. [Google Scholar] [CrossRef]
- Salamanca, C.A.; Fiol, N.; González, C.; Saez, M.; Villaescusa, I. Extraction of espresso coffee by using gradient of temperature. Effect on physicochemical and sensorial characteristics of espresso. Food Chem. 2017, 214, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.W.; Cheong, M.W.; Curran, P.; Yu, B.; Liu, S.Q. Coffee fermentation and flavor—An intricate and delicate relationship. Food Chem. 2015, 185, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Parenti, A.; Guerrini, L.; Masella, P.; Spinelli, S.; Calamai, L.; Spugnoli, P. Comparison of espresso coffee brewing techniques. J. Food Eng. 2014, 121, 112–117. [Google Scholar] [CrossRef]
- Dold, S.; Lindinger, C.; Kolodziejczyk, E.; Pollien, P.; Ali, S.; Germain, J.C.; Perin, S.G.; Pineau, N.; Folmer, B.; Engel, K.H.; et al. Influence of foam structure on the release kinetics of volatiles from espresso coffee prior to consumption. J. Agric. Food Chem. 2011, 59, 11196–11203. [Google Scholar] [CrossRef]
- Buffo, R.A.; Cardelli-Freire, C. Coffee flavour: An overview. Flavour Fragr. J. 2004, 19, 99–104. [Google Scholar] [CrossRef]
- Lolli, V.; Acharjee, A.; Angelino, D.; Tassotti, M.; Del Rio, D.; Mena, P.; Caligiani, A. Chemical Characterization of Capsule-Brewed Espresso Coffee Aroma from the Most Widespread Italian Brands by HS-SPME/GC-MS. Molecules 2020, 25, 1166. [Google Scholar] [CrossRef]
- Cordoba, N.; Fernandez-Alduenda, M.; Moreno, F.L.; Ruiz, Y. Coffee extraction: A review of parameters and their influence on the physicochemical characteristics and flavour of coffee brews. Trends Food Sci. Technol. 2020, 96, 45–60. [Google Scholar] [CrossRef]
- Liu, C.; Yang, N.; Yang, Q.; Ayed, C.; Linforth, R.; Fisk, I.D. Enhancing Robusta coffee aroma by modifying flavour precursors in the green coffee bean. Food Chem. 2019, 281, 8–17. [Google Scholar] [CrossRef]
- Caprioli, G.; Cortese, M.; Cristalli, G.; Maggi, F.; Odello, L.; Ricciutelli, M.; Sagratini, G.; Sirocchi, V.; Tomassoni, G.; Vittori, S. Optimization of espresso machine parameters through the analysis of coffee odorants by HS-SPME-GC/MS. Food Chem. 2012, 135, 1127–1133. [Google Scholar] [CrossRef]
- Blank, I.; Sen, A.; Grosch, W. Aroma impact compounds of Arabica and Robusta coffee. Qualitative and quantitative investigations. In Proceedings of the 14th International Scientific Colloquium on Coffee, San Francisco, CA, USA, 14–19 July 1991; pp. 117–129. [Google Scholar]
- Czerny, M.; Grosch, W. Potent Odorants of Raw Arabica Coffee. Their Changes during Roasting. J. Agric. Food Chem. 2000. [Google Scholar] [CrossRef]
- Czerny, M.; Mayer, F.; Grosch, W. Sensory Study on the Character Impact Odorants of Roasted Arabica Coffee. J. Agric. Food Chem. 1999. [Google Scholar] [CrossRef]
- Grosch, W. Specificity of the human nose in perceiving food odorants. In Frontiers of Flavour Science, Proceedings of the Ninth Weurman Flavour: Research Symposium, Freising, Germany, 22–25 June 1999; Deutsche Forschungsanstalt für Lebensmittelchemie: Freising, Germany, 2000; pp. 213–219. [Google Scholar]
- Grosch, W. Evaluation of the key odorants of foods by dilution experiments, aroma models and omission. Chem. Senses 2001, 26, 533–545. [Google Scholar] [CrossRef]
- Risticevic, S.; Carasek, E.; Pawliszyn, J. Headspace solid-phase microextraction-gas chromatographic-time-of-flight mass spectrometric methodology for geographical origin verification of coffee. Anal. Chim. Acta 2008, 617, 72–84. [Google Scholar] [CrossRef]
- Caporaso, N.; Genovese, A.; Canela, M.D.; Civitella, A.; Sacchi, R. Neapolitan coffee brew chemical analysis in comparison to espresso, moka and American brews. Food Res. Int. 2014, 61, 152–160. [Google Scholar] [CrossRef]
- Petisca, C.; Pérez-Palacios, T.; Pinho, O.; Ferreira, I.M.P.L.V.O. Optimization and Application of a HS-SPME-GC-MS Methodology for Quantification of Furanic Compounds in Espresso Coffee. Food Anal. Methods 2014, 7, 81–88. [Google Scholar] [CrossRef]
- Andueza, S.; Maeztu, L.; Pascual, L.; Ibáñez, C.; de Peña, M.P.; Cid, C. Influence of extraction temperature on the final quality of espresso coffee. J. Sci. Food Agric. 2003, 83, 240–248. [Google Scholar] [CrossRef]
- Flament, I. Coffee Flavor Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2001; ISBN 0471720380. [Google Scholar]
- Hovell, A.M.C.; Pereira, E.J.; Arruda, N.P.; Rezende, C.M. Evaluation of alignment methods and data pretreatments on the determination of the most important peaks for the discrimination of coffee varieties Arabica and Robusta using gas chromatography–mass spectroscopy. Anal. Chim. Acta 2010, 678, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Petisca, C.; Pérez-Palacios, T.; Farah, A.; Pinho, O.; Ferreira, I.M. Furans and other volatile compounds in ground roasted and espresso coffee using headspace solid-phase microextraction: Effect of roasting speed. Food Bioprod. Process. 2013, 91, 233–241. [Google Scholar] [CrossRef]
- Joint FAO/WHO Expert Committee on Food Additives; World Health Organization; Food and Agriculture Organization of the United Nations. Evaluation of Certain Veterinary Drug Residues in Food: Fifty-Fourth Report of the Joint FAO/WHO Expert Committee on Food Additives; WHO: Geneva, Switzerland, 2001. [Google Scholar]
- Kuballa, T.; Stier, S.; Strichow, N. Furan concentrations in coffee and coffee beverages. Dtsch. Leb. 2005, 101, 229–235. [Google Scholar]
- Zoller, O.; Sager, F.; Reinhard, H. Furan in food: Headspace method and product survey. Food Addit. Contam. 2007, 24, 91–107. [Google Scholar] [CrossRef]
- Altaki, M.S.; Santos, F.J.; Galceran, M.T. Occurrence of furan in coffee from Spanish market: Contribution of brewing and roasting. Food Chem. 2011, 126, 1527–1532. [Google Scholar] [CrossRef]
- Rahn, A.; Yeretzian, C. Impact of consumer behavior on furan and furan-derivative exposure during coffee consumption. A comparison between brewing methods and drinking preferences. Food Chem. 2019, 272, 514–522. [Google Scholar] [CrossRef]
- Authority, E.F.S. Update on furan levels in food from monitoring years 2004–2010 and exposure assessment. EFSA J. 2011, 9, 2347. [Google Scholar]
- Ribeiro, J.S.; Augusto, F.; Salva, T.J.G.; Thomaziello, R.A.; Ferreira, M.M.C. Prediction of sensory properties of Brazilian Arabica roasted coffees by headspace solid phase microextraction-gas chromatography and partial least squares. Anal. Chim. Acta 2009, 634, 172–179. [Google Scholar] [CrossRef]
- Cheong, M.W.; Tong, K.H.; Ong, J.J.M.; Liu, S.Q.; Curran, P.; Yu, B. Volatile composition and antioxidant capacity of Arabica coffee. Food Res. Int. 2013, 51, 388–396. [Google Scholar] [CrossRef]
- Bicho, N.C.; Leitão, A.E.; Ramalho, J.C.; de Alvarenga, N.B.; Lidon, F.C. Impact of roasting time on the sensory profile of Arabica and Robusta coffee. Ecol. Food Nutr. 2013, 52, 163–177. [Google Scholar] [CrossRef]
- Sanz, C.; Maeztu, L.; Jose Zapelena, M.; Bello, J.; Cid, C. Profiles of volatile compounds and sensory analysis of three blends of coffee: Influence of different proportions of Arabica and Robusta and influence of roasting coffee with sugar. J. Sci. Food Agric. 2002, 82, 840–847. [Google Scholar] [CrossRef]
- Toci, A.T.; Farah, A. Volatile fingerprint of Brazilian defective coffee seeds: Corroboration of potential marker compounds and identification of new low quality indicators. Food Chem. 2014, 153, 298–314. [Google Scholar] [CrossRef]
- Pickard, S.; Becker, I.; Merz, K.-H.; Richling, E. Determination of the alkylpyrazine composition of coffee using stable isotope dilution–gas chromatography–mass spectrometry (SIDA-GC-MS). J. Agric. Food Chem. 2013, 61, 6274–6281. [Google Scholar] [CrossRef]
- Toledo, P.R.A.B.; Pezza, L.; Pezza, H.R.; Toci, A.T. Relationship between the different aspects related to coffee quality and their volatile compounds. Compr. Rev. Food Sci. Food Saf. 2016, 15, 705–719. [Google Scholar] [CrossRef]
- Bressanello, D.; Liberto, E.; Cordero, C.; Rubiolo, P.; Pellegrino, G.; Ruosi, M.R.; Bicchi, C. Coffee aroma: Chemometric comparison of the chemical information provided by three different samplings combined with GC–MS to describe the sensory properties in cup. Food Chem. 2017, 214, 218–226. [Google Scholar] [CrossRef]
- Andueza, S.; De Peña, M.P.; Cid, C. Chemical and sensorial characteristics of espresso coffee as affected by grinding and torrefacto roast. J. Agric. Food Chem. 2003, 51, 7034–7039. [Google Scholar] [CrossRef]
- Andueza, S.; Vila, M.A.; Paz de Peña, M.; Cid, C. Influence of coffee/water ratio on the final quality of espresso coffee. J. Sci. Food Agric. 2007, 87, 586–592. [Google Scholar] [CrossRef]
- Akiyama, M.; Murakami, K.; Ikeda, M.; Iwatsuki, K.; Wada, A.; Tokuno, K.; Onishi, M.; Iwabuchi, H.; Sagara, Y. Analysis of freshly brewed espresso using a retronasal aroma simulator and influence of milk addition. Food Sci. Technol. Res. 2009, 15, 233–244. [Google Scholar] [CrossRef]
- Piccone, P.; Lonzarich, V.; Navarini, L.; Fusella, G.; Pittia, P. Effect of sugars on liquid–vapour partition of volatile compounds in ready-to-drink coffee beverages. J. Mass Spectrom. 2012, 47, 1120–1131. [Google Scholar] [CrossRef]
- López-Galilea, I.; Fournier, N.; Cid, C.; Guichard, E. Changes in headspace volatile concentrations of coffee brews caused by the roasting process and the brewing procedure. J. Agric. Food Chem. 2006, 54, 8560–8566. [Google Scholar] [CrossRef]
- Tressl, R.; Silwar, R. Investigation of sulfur-containing components in roasted coffee. J. Agric. Food Chem. 1981, 29, 1078–1082. [Google Scholar] [CrossRef]
- Rocha, S.; Maeztu, L.; Barros, A.; Cid, C.; Coimbra, M.A. Screening and distinction of coffee brews based on headspace solid phase microextraction/gas chromatography/principal component analysis. J. Sci. Food Agric. 2004, 84, 43–51. [Google Scholar] [CrossRef]
- Semmelroch, P.; Grosch, W. Studies on character impact odorants of coffee brews. J. Agric. Food Chem. 1996, 44, 537–543. [Google Scholar] [CrossRef]
- Maeztu, L.; Sanz, C.; Andueza, S.; Paz de Pena, M.; Bello, J.; Cid, C. Characterization of espresso coffee aroma by static headspace GC− MS and sensory flavor profile. J. Agric. Food Chem. 2001, 49, 5437–5444. [Google Scholar] [CrossRef] [PubMed]
- Michishita, T.; Akiyama, M.; Hirano, Y.; Ikeda, M.; Sagara, Y.; Araki, T. Gas chromatography/olfactometry and electronic nose analyses of retronasal aroma of espresso and correlation with sensory evaluation by an artificial neural network. J. Food Sci. 2010, 75, S477–S489. [Google Scholar] [CrossRef]
- Curioni, P.M.G.; Bosset, J.O. Key odorants in various cheese types as determined by gas chromatography-olfactometry. Int. Dairy J. 2002, 12, 959–984. [Google Scholar] [CrossRef]
- Marin, K.; Požrl, T.; Zlatić, E.; Plestenjak, A. A new aroma index to determine the aroma quality of roasted and ground coffee during storage. Food Technol. Biotechnol. 2008, 46, 442–447. [Google Scholar]
- Blank, I.; Sen, A.; Grosch, W. Potent odorants of the roasted powder and brew of Arabica coffee. Z. Leb. Forsch. 1992, 195, 239–245. [Google Scholar] [CrossRef]
- Akiyama, M.; Murakami, K.; Ikeda, M.; Iwatsuki, K.; Wada, A.; Tokuno, K.; Onishi, M.; Iwabuchi, H. Analysis of the headspace volatiles of freshly brewed Arabica coffee using solid-phase microextraction. J. Food Sci. 2007, 72, C388–C396. [Google Scholar] [CrossRef] [PubMed]
- Mayer, F.; Czerny, M.; Grosch, W. Influence of provenance and roast degree on the composition of potent odorants in Arabica coffees. Eur. Food Res. Technol. 1999, 209, 242–250. [Google Scholar] [CrossRef]
- Mayer, F.; Czerny, M.; Grosch, W. Sensory study of the character impact aroma compounds of a coffee beverage. Eur. Food Res. Technol. 2000, 211, 272–276. [Google Scholar] [CrossRef]
- Holscher, W.; Vitzthum, O.G.; Steinhart, H. Identification and sensorial evaluation of aroma-impact-compounds in roasted Colombian coffee. Cafe-Cacao-thè 1990, 34, 205–212. [Google Scholar]
- Mayer, F.; Grosch, W. Aroma simulation on the basis of the odourant composition of roasted coffee headspace. Flavour Fragr. J. 2001, 16, 180–190. [Google Scholar] [CrossRef]
- Ribeiro, J.S.; Augusto, F.; Salva, T.J.G.; Ferreira, M.M.C. Prediction models for Arabica coffee beverage quality based on aroma analyses and chemometrics. Talanta 2012, 101, 253–260. [Google Scholar] [CrossRef]
- Bicchi, C.; Ruosi, M.R.; Cagliero, C.; Cordero, C.; Liberto, E.; Rubiolo, P.; Sgorbini, B. Quantitative analysis of volatiles from solid matrices of vegetable origin by high concentration capacity headspace techniques: Determination of furan in roasted coffee. J. Chromatogr. A 2011, 1218, 753–762. [Google Scholar] [CrossRef]
- Burdock, G.A. Alphabetical listing. Fenaroli’s Handbook. Flavor Ingredients, 6th ed.; CRC Press Taylor Francis Group: Boca Raton, FL, USA, 2010; pp. 1–2035. [Google Scholar]
- Semmelroch, P.; Grosch, W. Analysis of roasted coffee powders and brews by gas chromatography-olfactometry of headspace samples. LWT Food Sci. Technol. 1995, 28, 310–313. [Google Scholar] [CrossRef]
- Clarke, R.; Vitzthum, O.G. Coffee: Recent Developments; John Wiley & Sons: Hoboken, NJ, USA, 2008; ISBN 0470680210. [Google Scholar]
- Wold, S.; Sjöström, M. Chemometrics, present and future success. Chemom. Intell. Lab. Syst. 1998, 44, 3–14. [Google Scholar] [CrossRef]
- Ludwig, I.A.; Sanchez, L.; Caemmerer, B.; Kroh, L.W.; De Peña, M.P.; Cid, C. Extraction of coffee antioxidants: Impact of brewing time and method. Food Res. Int. 2012, 48, 57–64. [Google Scholar] [CrossRef]
- Sánchez-López, J.A.; Zimmermann, R.; Yeretzian, C. Insight into the time-resolved extraction of aroma compounds during espresso coffee preparation: Online monitoring by PTR-ToF-MS. Anal. Chem. 2014, 86, 11696–11704. [Google Scholar] [CrossRef]
- Mestdagh, F.; Glabasnia, A.; Giuliano, P. The Brew-Extracting for Excellence. In The Craft and Science of Coffee; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 355–380. ISBN 9780128035580. [Google Scholar]
- Albanese, D.; Di Matteo, M.; Poiana, M.; Spagnamusso, S. Espresso coffee (EC) by POD: Study of thermal profile during extraction process and influence of water temperature on chemical-physical and sensorial properties. Food Res. Int. 2009, 42, 727–732. [Google Scholar] [CrossRef]
- López, J.A.S.; Wellinger, M.; Gloess, A.N.; Zimmermann, R.; Yeretzian, C. Extraction kinetics of coffee aroma compounds using a semi-automatic machine: On-line analysis by PTR-ToF-MS. Int. J. Mass Spectrom. 2016, 401, 22–30. [Google Scholar] [CrossRef]
- Corrochano, B.R.; Melrose, J.R.; Bentley, A.C.; Fryer, P.J.; Bakalis, S. A new methodology to estimate the steady-state permeability of roast and ground coffee in packed beds. J. Food Eng. 2015, 150, 106–116. [Google Scholar] [CrossRef]
- Caprioli, G.; Cortese, M.; Maggi, F.; Minnetti, C.; Odello, L.; Sagratini, G.; Vittori, S. Quantification of caffeine, trigonelline and nicotinic acid in espresso coffee: The influence of espresso machines and coffee cultivars. Int. J. Food Sci. Nutr. 2014, 65, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Angeloni, G.; Guerrini, L.; Masella, P.; Bellumori, M.; Daluiso, S.; Parenti, A.; Innocenti, M. What kind of coffee do you drink? An investigation on effects of eight different extraction methods. Food Res. Int. 2019, 116, 1327–1335. [Google Scholar] [CrossRef]
- Cappuccio, R.; Liverani, F.S. Computer simulation as a tool to model coffee brewing cellular automata for percolation processes; 2D and 3D techniques for fluid-dynamic simulations. In Proceedings of the Colloque Scientifique International sur le Cafe, Helsinki, Finland, 2–6 August 1999; pp. 173–178. [Google Scholar]
- Baggenstoss, J.; Perren, R.; Escher, F. Water content of roasted coffee: Impact on grinding behaviour, extraction, and aroma retention. Eur. Food Res. Technol. 2008, 227, 1357–1365. [Google Scholar] [CrossRef][Green Version]
- Moroney, K.M.; Lee, W.T.; O’Brien, S.B.G.; Suijver, F.; Marra, J. Modelling of coffee extraction during brewing using multiscale methods: An experimentally validated model. Chem. Eng. Sci. 2015, 137, 216–234. [Google Scholar] [CrossRef]
- Moroney, K.M.; Lee, W.T.; O’Brien, S.B.G.; Suijver, F.; Marra, J. Asymptotic analysis of the dominant mechanisms in the coffee extraction process. SIAM J. Appl. Math. 2016, 76, 2196–2217. [Google Scholar] [CrossRef]
- Moroney, K.M.; Lee, W.T.; O’Brien, S.B.G.; Suijver, F.; Marra, J. Coffee extraction kinetics in a well mixed system. J. Math. Ind. 2016, 7, 1–19. [Google Scholar] [CrossRef]
- Majchrzak, T.; Wojnowski, W.; Lubinska-Szczygeł, M.; Różańska, A.; Namieśnik, J.; Dymerski, T. PTR-MS and GC-MS as complementary techniques for analysis of volatiles: A tutorial review. Anal. Chim. Acta 2018, 1035, 1–13. [Google Scholar] [CrossRef]
- De Vivo, A.; Tricarico, M.C.; Sarghini, F. Espresso coffee design based on non-monotonic granulometric distribution of aromatic profile. Food Res. Int. 2019, 123, 650–661. [Google Scholar] [CrossRef]
- Genovese, A.; Caporaso, N.; Civitella, A.; Sacchi, R. Effect of human saliva and sip volume of coffee brews on the release of key volatile compounds by a retronasal aroma simulator. Food Res. Int. 2014, 61, 100–111. [Google Scholar] [CrossRef]
- Pua, A.; Lau, H.; Liu, S.Q.; Tan, L.P.; Goh, R.M.V.; Lassabliere, B.; Leong, K.C.; Sun, J.; Cornuz, M.; Yu, B. Improved detection of key odourants in Arabica coffee using gas chromatography-olfactometry in combination with low energy electron ionisation gas chromatography-quadrupole time-of-flight mass spectrometry. Food Chem. 2020, 302, 125370. [Google Scholar] [CrossRef]
- Zanin, R.C.; Smrke, S.; Kurozawa, L.E.; Yamashita, F.; Yeretzian, C. Novel experimental approach to study aroma release upon reconstitution of instant coffee products. Food Chem. 2020, 317, 126455. [Google Scholar] [CrossRef]
- Zanin, R.C.; Smrke, S.; Kurozawa, L.E.; Yamashita, F.; Yeretzian, C. Modulation of aroma release of instant coffees through microparticles of roasted coffee oil. Food Chem. 2021, 341, 128193. [Google Scholar] [CrossRef]
- Amanpour, A.; Selli, S. Differentiation of Volatile Profiles and Odor Activity Values of Turkish Coffee and French Press Coffee. J. Food Process. Preserv. 2016, 40, 1116–1124. [Google Scholar] [CrossRef]
- Thammarat, P.; Kulsing, C.; Wongravee, K.; Leepipatpiboon, N.; Nhujak, T. Identification of Volatile Compounds and Selection of Discriminant Markers for Elephant Dung Coffee Using Static Headspace Gas Chromatography-Mass Spectrometry and Chemometrics. Molecules 2018, 23, 1910. [Google Scholar] [CrossRef]
- Sopelana, P.; Pérez-Martínez, M.; López-Galilea, I.; de Peña, M.P.; Cid, C. Effect of ultra high temperature (UHT) treatment on coffee brew stability. Food Res. Int. 2013, 50, 682–690. [Google Scholar] [CrossRef]
- Bröhan, M.; Huybrighs, T.; Wouters, C.; Van der Bruggen, B. Influence of storage conditions on aroma compounds in coffee pads using static headspace GC-MS. Food Chem. 2009, 116, 480–483. [Google Scholar] [CrossRef]
- Kamgang Nzekoue, F.; Angeloni, S.; Caprioli, G.; Cortese, M.; Maggi, F.; Marconi, U.M.B.; Perali, A.; Ricciutelli, M.; Sagratini, G.; Vittori, S. Fiber-Sample Distance, An Important Parameter to Be Considered in Headspace Solid-Phase Microextraction Applications. Anal. Chem. 2020, 92, 7478–7484. [Google Scholar] [CrossRef]
- Ongo, E.A.; Montevecchi, G.; Antonelli, A.; Sberveglieri, V.; Sevilla, F. Metabolomics fingerprint of Philippine coffee by SPME-GC-MS for geographical and varietal classification. Food Res. Int. 2020, 134, 109227. [Google Scholar] [CrossRef]
- Mustafa, A.M.; Angeloni, S.; Nzekoue, F.K.; Abouelenein, D.; Sagratini, G.; Caprioli, G.; Torregiani, E. An Overview on Truffle Aroma and Main Volatile Compounds. Molecules 2020, 25, 5948. [Google Scholar] [CrossRef]
- Steen, I.; Waehrens, S.S.; Petersen, M.A.; Münchow, M.; Bredie, W.L.P. Influence of serving temperature on flavour perception and release of Bourbon Caturra coffee. Food Chem. 2017, 219, 61–68. [Google Scholar] [CrossRef]
- Ochiai, N.; Tsunokawa, J.; Sasamoto, K.; Hoffmann, A. Multi-volatile method for aroma analysis using sequential dynamic headspace sampling with an application to brewed coffee. J. Chromatogr. A 2014, 1371, 65–73. [Google Scholar] [CrossRef]
- Diez-Simon, C.; Ammerlaan, B.; van den Berg, M.; van Duynhoven, J.; Jacobs, D.; Mumm, R.; Hall, R.D. Comparison of volatile trapping techniques for the comprehensive analysis of food flavourings by Gas Chromatography-Mass Spectrometry. J. Chromatogr. A 2020, 1624, 461191. [Google Scholar] [CrossRef]
- Bicchi, C.; Iori, C.; Rubiolo, P.; Sandra, P. Headspace sorptive extraction (HSSE), stir bar sorptive extraction (SBSE), and solid phase microextraction (SPME) applied to the analysis of roasted Arabica coffee and coffee brew. J. Agric. Food Chem. 2002, 50, 449–459. [Google Scholar] [CrossRef]
- Ochiai, N.; Sasamoto, K.; David, F.; Sandra, P. Recent Developments of Stir Bar Sorptive Extraction for Food Applications: Extension to Polar Solutes. J. Agric. Food Chem. 2018, 66, 7249–7255. [Google Scholar] [CrossRef]
- Pacheco-Fernández, I.; Rentero, M.; Ayala, J.H.; Pasán, J.; Pino, V. Green solid-phase microextraction fiber coating based on the metal-organic framework CIM-80(Al): Analytical performance evaluation in direct immersion and headspace using gas chromatography and mass spectrometry for the analysis of water, urine and brewed. Anal. Chim. Acta 2020, 1133, 137–149. [Google Scholar] [CrossRef]
- Zamanipoor, M.H.; Yakufu, B.; Tse, E.; Rezaeimotlagh, A.; Hook, J.M.; Bucknall, M.P.; Thomas, D.S.; Trujillo, F.J. Brewing coffee?—Ultra-sonication has clear beneficial effects on the extraction of key volatile aroma components and triglycerides. Ultrason. Sonochem. 2020, 60, 104796. [Google Scholar] [CrossRef]
- Alstrup, J.; Petersen, M.A.; Larsen, F.H.; Münchow, M. The effect of roast development time modulations on the sensory profile and chemical composition of the coffee brew as measured by nmr and dhs-gc–ms. Beverages 2020, 6, 70. [Google Scholar] [CrossRef]
- Fisk, I.D.; Kettle, A.; Hofmeister, S.; Virdie, A.; Kenny, J.S. Discrimination of roast and ground coffee aroma. Flavour 2012, 1, 1–9. [Google Scholar] [CrossRef]
- Majcher, M.A.; Klensporf-Pawlik, D.; Dziadas, M.; Jeleń, H.H. Identification of aroma active compounds of cereal coffee brew and its roasted ingredients. J. Agric. Food Chem. 2013, 61, 2648–2654. [Google Scholar] [CrossRef]
- Abdelwareth, A.; Zayed, A.; Farag, M.A. Chemometrics-based aroma profiling for revealing origin, roasting indices, and brewing method in coffee seeds and its commercial blends in the Middle East. Food Chem. 2021, 349, 129162. [Google Scholar] [CrossRef] [PubMed]
- Turan Ayseli, M.; Kelebek, H.; Selli, S. Elucidation of aroma-active compounds and chlorogenic acids of Turkish coffee brewed from medium and dark roasted Coffea arabica beans. Food Chem. 2021, 338, 127821. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Hong, S.J.; Cho, J.-J.; Boo, C.G.; Kim, D.-S.; Shin, E.-C. Peanut Coffee: Enhancement of Nutritional, Physicochemical, and Sensory Characteristics in Coffee Brewed with Conventional and High-Oleic Peanut Extracts. Foods 2020, 9, 1664. [Google Scholar] [CrossRef] [PubMed]
- Karakaya, D.; Ulucan, O.; Turkan, M. Electronic Nose and Its Applications: A Survey. Int. J. Autom. Comput. 2020, 17, 179–209. [Google Scholar] [CrossRef]
- Kong, F.; Singh, R.P. Advances in instrumental methods to determine food quality deterioration. In Food and Beverage Stability and Shelf Life; Elsevier: Amsterdam, The Netherlands, 2011; pp. 381–404. [Google Scholar]
- Romano, A.; Cappellin, L.; Ting, V.; Aprea, E.; Navarini, L.; Gasperi, F.; Biasioli, F. Nosespace analysis by PTR-ToF-MS for the characterization of food and tasters: The case study of coffee. Int. J. Mass Spectrom. 2014, 365–366, 20–27. [Google Scholar] [CrossRef]
| No. | Compound Name and Class | Arabica (% Area) | Blend (Robusta-Arabica (80:20)) (% Area) | References |
|---|---|---|---|---|
| Aldehydes | ||||
| 1 | Acetaldehyde | 0.17–0.49 | 0.34–0.40 | [10,47,49,67,68] |
| 2 | 2-Methylpropanal | 0.65–2.46 | 2.00-2.83 | [10,47,49,67,68] |
| 3 | Butanal | 0.08–1.16 | [47,70] | |
| 4 | 2-Methylbutanal | 1.25–2.35 | 1.26–2.01 | [10,47,49,67,68,69,70] |
| 5 | 3-Methylbutanal | 0.59–3.78 | 2.35–3.38 | [10,47,49,68,69,70] |
| 6 | Hexanal | 0.02–0.08 | 0.05–0.1 | [10,47,49,67,68] |
| 7 | Propanal | 0.54–0.71 | 0.47–0.55 | [10,49,67,68] |
| Pyrazines | ||||
| 8 | 2-Ethyl-6-methylpyrazine | 0.02–1.73 | 0.04–0.08 | [10,47,49,67,68,69,70] |
| 9 | 2-Ethyl-5-methylpyrazine | 1.03–2.9 | [47,69,70] | |
| 10 | 2-Propylpyrazine | 0.13–0.48 | [47,70] | |
| 11 | 2,6-Diethylpyrazine | 0.34–0.48 | [47,70] | |
| 12 | 2-Ethyl-3,5-dimethylpyrazine | 0.01–2.30 | 0.04–0.08 | [10,47,49,67,68] |
| 13 | 2-Methyl-3-trans-propenylpyrazine | 0.38 | [47] | |
| 14 | Pyrazine | 0.24–0.44 | [69,70] | |
| 15 | 2-Methylpyrazine | 2.45–4.08 | [69,70] | |
| 16 | 2,5-Dimethylpyrazine | 1.43–1.45 | [69,70] | |
| 17 | 2,6-Dimethylpyrazine | 1.61–1.66 | [69,70] | |
| 18 | 2-Ethylpyrazine | 0.06–1.61 | 0.11–0.17 | [10,49,67,68,69,70] |
| 19 | Trimethylpyrazine | 0.67-1.11 | [69,70] | |
| 20 | 3-Ethyl-2,5-dimethylpyrazine | 1.04–1.38 | [69,70] | |
| 21 | 2,3-Dimethylpyrazine | 0.28 | [70] | |
| 22 | 2-ethyl-3-methyl pyrazine | 1.12 | [70] | |
| 23 | 5H-5-Methyl-6,7- Dihydrocyclopentapyrazine | 0.26 | [70] | |
| 24 | Acetyl pyrazine | 0.06 | [70] | |
| 25 | 1-(6-Methyl-2-pyrazinyl)-1-ethanone | 0.18 | [70] | |
| Furans | ||||
| 26 | 2,5-Dimethylfuran | 0.21–0.32 | [47,69,70] | |
| 27 | Furfuryl methyl ether | 0.22–1.41 | [47,70] | |
| 28 | Furfurylmethyl sulfide | 0.61–7.47 | [47,70] | |
| 29 | 2-Furanmethanol acetate | 9.46–37.35 | 0.17 | [47,48,70] |
| 30 | 2-Furanmethanol (furfuryl alcohol) | 7.84–18.00 | 21.00 | [47,48,69,70] |
| 31 | 2-Methylfuran | 1.15–2.10 | [69,70] | |
| 32 | 3-Methylfuran | 0.15 | [69] | |
| 33 | 2-Ethenyl-5-methylfuran | 0.29 | [69] | |
| 34 | 2-(Methoxymethyl)furan | 0.64 | [69] | |
| 35 | Dihydro-2-methyl-3(2H)-furanone | 1.29 | [69] | |
| 36 | 2-Furancarboxaldehyde | 7.65 | [69] | |
| 37 | 2-[(Methylthio)methyl]furan | 1.27 | [69] | |
| 38 | Furfuryl formate | 0.29–3.59 | [69] | |
| 39 | 1-(2-Furanyl)-ethanone | 2.33 | [69] | |
| 40 | Furfuryl acetate | 15.34 | [69] | |
| 41 | 5-Methyl-2-furancarboxaldehyde (5-Methylfurfural) | 4.77–7.90 | 8.20 | [48,69,70] |
| 42 | 2,2’-Methylenebisfuranc | 0.82 | [69] | |
| 43 | 2-(2-Furanylmethyl)-5-methylfuran | 0.40 | [69] | |
| 44 | 2,3-Dihydro-6-methylthieno[2,3c]furan | 0.91 | [69] | |
| 45 | Furan | 0.21–0.40 | 0.03 | [48,70] |
| 46 | 2-vinylfuran | 0.24 | [70] | |
| 47 | cis-2-Methyl-5-npropenylfuran | 0.13 | [70] | |
| 48 | Furfural | 2.15–10.00 | 11.00 | [48,70] |
| 49 | Furaneol | 0.15 | [70] | |
| 50 | 2-Acetylfuran/2-furfuryl methyl ketone | 0.40 | [70] | |
| 51 | 1-(2-Furyl)-2-propanone | 0.54 | [70] | |
| 52 | Furanmethanol acetate | 9.46 | [70] | |
| 53 | 2-Furanmethanol propanoate | 0.90 | [70] | |
| 54 | 2-furfuryl furan | 0.76 | [70] | |
| 55 | Dihydro-2(3H)-furanone | 0.59 | [70] | |
| 56 | 1-(2-Furyl)-butan-3-one | 0.29 | [70] | |
| 57 | 5-Methyl-2-furfurylfuran | 0.61 | [70] | |
| 58 | 3,4-dimethyl 2,5-furandione | 0.09 | [70] | |
| 59 | 2,3-dihydro-6-methylthylthieno furan | 0.62 | [70] | |
| 60 | 2-Acetyl-5-methylfuran | 1.48 | [70] | |
| 61 | Difurfuryl ether | 2.59 | [70] | |
| 62 | 2-Vinyl-5-methylfuran | 0.23 | [70] | |
| Ketones | ||||
| 63 | 2,3-Pentanedione | 0.59–2.37 | 0.42–0.53 | [10,47,49,67,68,69,70] |
| 64 | β-Damascenone | 0.06 | [47] | |
| 65 | 2-Butanone | 0.50–0.86 | [69,70] | |
| 66 | 2,3-Butanedione | 0.46–1.11 | 0.32–0.34 | [10,49,67,68,69] |
| 67 | 2,3-Hexanedione | 0.69 | [69] | |
| 68 | 2,3-Butanedione (diacetyl) | 0.32 | [70] | |
| 69 | 2,4-Dimethyl-3-pentanone | 0.23 | [70] | |
| 70 | 3,4-Hexanedione | 0.17 | [70] | |
| 71 | 3-Hydroxy-2-butanone | 0.13 | [70] | |
| 72 | 1-Hydroxy-2-propanone | 0.50 | [70] | |
| 73 | 2-Methyl 2-cyclopenten-1-one | 0.09 | [70] | |
| 74 | 1-Hydroxy-2-butanone | 0.08 | [70] | |
| 75 | 3,3-Dimethyl 2-butanone | 0.71 | [70] | |
| 76 | 1-(Acetyloxy) 2-butanone | 0.71 | [70] | |
| 77 | Ethylcyclopentenolone | 0.37 | [70] | |
| 78 | 3,5-Dimethyl cyclopentenolone | 0.12 | [70] | |
| 79 | 2-Cyclopenten-1-one, 2-Hydroxy-3-methyl | 0.24 | [70] | |
| 80 | 3-Ethyl-2-hydroxy-2-cyclopenten-1-one | 0.23 | [70] | |
| 81 | 3-Hydroxy-2-methyl-4H-pyran-4-one | 1.35 | [70] | |
| Alcohols | ||||
| 82 | 2-Methyl-3-Pentanol | 0.07 | [70] | |
| 83 | 3-Penten-2-ol | 0.07 | [70] | |
| 84 | Phenylethyl alcohol | 0.21 | [70] | |
| Acids | ||||
| 85 | Acetic acid | 2.30 | [70] | |
| 86 | Propanoic acid | 0.10 | [70] | |
| 87 | iso-Valeric acid | 0.72 | [70] | |
| 88 | 3-methyl-2-butenoic acid | 0.12 | [70] | |
| Esters | ||||
| 89 | Methyl acetate | 0.87 | [69] | |
| 90 | Acetol acetate | 2.01 | [70] | |
| Pyrroles | ||||
| 91 | 1-Methyl-(1H-pyrrol-2-yl)-1-ethanone | 1.88 | [47] | |
| 92 | 1-Methyl-1H-pyrrole | 0.47–1.73 | [69,70] | |
| 93 | 1-Ethyl-1H-pyrrole | 0.25 | [69] | |
| 94 | 1H-Pyrrole | 0.40–1.51 | [69,70] | |
| 95 | 1-Methyl-1H-pyrrole-2-carboxaldehyde | 1.14 | [69] | |
| 96 | 1-(2-Furanylmethyl)-1H-pyrrole | 1.61 | [69] | |
| 97 | 2,5-Dimethyl-1H-pyrrole | 2.42 | [70] | |
| 98 | 2-Formyl-1-methylpyrrole | 1.31 | [70] | |
| 99 | 2-Formyl-4,5-dimethylpyrrole | 0.39 | [70] | |
| 100 | N-furfuryl pyrrole | 3.45 | [70] | |
| 101 | Acetyl pyrrole | 1.97 | [70] | |
| 102 | 1H-Pyrrole-2-carboxaldehyde | 1.24 | [70] | |
| 103 | 2-formyl-1-methylpyrrole | 0.71 | [70] | |
| Sulfur compounds | ||||
| 104 | Methanethiol | 0.11–0.16 | 0.10–0.12 | [10,47,49,67,68] |
| 105 | 2-Propyl-thiophene | 0.11 | [70] | |
| Phenolic compounds | ||||
| 106 | 2-Methoxyphenol (guaiacol) | 0.02–9.12 | 0.01–0.04 | [10,47,49,67,68,69,70] |
| 107 | 4-Ethylguaiacol | 1.81–4.85 | [47,70] | |
| 108 | 4-Vinylguaiacol | 3.24 | [47] | |
| 109 | 4-Ethenyl-2-methoxyphenol | 0.45 | [69] | |
| 110 | Phenol | 1.00 | [70] | |
| 111 | 4-Vinyl-2-methoxy-phenol | 4.19 | [70] | |
| others | ||||
| 112 | Pyridine | 5.79–11.90 | [69,70] | |
| 113 | 3-Ethyl-3-methyl maleic anhydride | 0.09 | [70] | |
| 114 | Trimethyl oxazole | 0.06 | [70] | |
| 115 | 3-methyl 2(1H)-quinolinone | 0.30 | [70] | |
| 116 | 2-Methyl pyridine | 0.03 | [70] | |
| 117 | 3-Ethylpyridine | 0.18 | [70] | |
| 118 | Linalool oxide | 0.16 | [70] | |
| 119 | Linalool | 0.08 | [70] | |
| 120 | β-Myrcene | 0.06 | [70] | |
| 121 | D-Limonene | 0.05 | [70] |
| Key Odorants Identified in EC | Odor Description | |
|---|---|---|
| ALDEHYDES | ||
| 2-Methylpropanal | Grassy, fermented/Buttery–oily | [67,73,78,79,80] |
| 2-Methylbutanal | Malty, fermented/Buttery–oily | [73,78,79,80] |
| 3-Methylbutanal | Almond, fruity/Buttery–oily | [73,78,79,80] |
| Hexanal | Fruity | [67,79] |
| (E)-2-Nonenal | Buttery–oily | [78,80] |
| Acetaldehyde | Fruity | [67,79] |
| Benzeneacetaldehyde | Sweet–fruity | [78] |
| Propanal | Fruity | [67,79] |
| KETONES | ||
| 2,3-Pentanedione | Buttery–oily, caramel-like | [73,78,79,80] |
| 2,3-Butanedione | Buttery–oily | [78,79,80] |
| (E)-β-Damascenone | Sweet–fruity | [78,80] |
| 1-Octen-3-one | Mushroom-like | [78,80] |
| ACIDS | ||
| 2-Methylbutyric acid | Acidic | [80] |
| 3-Methylbutyric acid | Acidic | [78,80] |
| FURANES | ||
| 2-((Methylthio)methyl)furan | Smoke roast | [78,80] |
| 2-Furfurylthiol (2-furanmethanethiol) | Smoke roast | [78,80] |
| SULFUR COMPOUNDS | ||
| Dimethyl trisulfide | - | [78,80] |
| Methanethiol | Freshness | [67,79] |
| 3-(Methylthio)propionaldehyde | Soy sauce | [78,80] |
| 3-Methyl-2-butene-1-thiol | Smoke roast | [78,80] |
| 3-Mercapto-3-methylbutanol | Smoke roast | [78,80] |
| 3-Mercapto-3-methylbutylformate | green–blackcurrant | [78,80] |
| FURANONES | ||
| 4-Hydroxy-2,5-dimethyl-3(2H)-furanone | Sweet–caramel | [80] |
| 4,5-Dimethyl-3-hydroxy-2(5H)-furanone | Sweet–caramel | [78,80] |
| 2-Hydroxy-3-methyl-2-cyclopenten-1-one | Sweet–caramel | [78] |
| 2-Ethyl-4-hydroxy-5-methyl-3(2H)-furanone | Sweet–caramel | [78] |
| PHENOLIC COMPUNDS | ||
| 2-Methoxyphenol (guaiacol) | Phenolic, spicy | [67,73,78,79,80] |
| 4-Ethyl-2-methoxyphenol (4-ethylguaiacol) | Phenolic | [78,80] |
| 4-Ethenyl-2-methoxyphenol (4-vinylguaiacol) | Phenolic | [78,80] |
| PYRAZINE | ||
| 2-Ethylpyrazine | Earthy, musty | [73,79] |
| 2-Ethyl-6-methylpyrazine | Earthy, musty/Earth, mould | [67,73,79] |
| 2-Ethyl-3,5-dimethylpyrazine | Woody,papery/burned/nutty roast | [67,73,78,79,80] |
| 2,3-Diethyl-5-methylpyrazine | Nutty roast | [78,80] |
| 2-Methoxy-3- isopropylpyrazine | Green–earthy | [78,80] |
| 2-Methoxy-3-(1- methylpropyl)pyrazine | Green–earthy | [78,80] |
| TERPENE | ||
| Linalool | Sweet–fruity | [78,80] |
| OTHERS | ||
| (3,4-Dihydro-2H-pyrrol-5-yl)-ethanone | Nutty roast | [78] |
| 3-Methyl-1H-indole | Phenolic | [78] |
| Variables | Constant Conditions | Chemical Analysis | Flavor Profile | Reference |
|---|---|---|---|---|
| Time (s) | ||||
|
| • Caffeine ↓ when the extraction time increases | • Compounds responsible for the aromatic profile ↓ when the ex-traction time increases. Their concentration is higher in samples with the finest grinding grade suggesting that by reducing the particle size, the extraction increases | [11] |
|
|
| • Not given | [92] |
|
|
|
| [3] |
|
|
| Not given | [91] |
| T (°C) | ||||
|
|
|
| [49] |
|
|
|
| [32] |
|
| • Increasing temperature: ↑caffeine and foam index | Data not shown | [94] |
| P (bar) | ||||
|
|
|
| [10] |
| PS (μm) b | ||||
|
|
|
| [49] |
| P (bar) + T (°C) | ||||
|
|
|
| [40] |
|
|
| • Least polar com-pounds are the most affected, impacting the aroma balance in the last stage of the extraction and the cup. | [95] |
| P (bar) + T (s) | ||||
| • T° (°C): 92–93 °C |
| Not given | [98] |
| Sampling | Analyte Separation | Detecting System | References | |
|---|---|---|---|---|
| GC-based method | 1. Solvent-assisted extraction | 1. Monodimentional (ZB-FFAP, HP-WAX, DB-WAX, DB-5) | 1. Mass Spectrometer (GC-MS) - Quadrupole MS - Q-ToF | [8,10,11,40,67,69,75,76,104,105,106] |
| 2. Static Headspace (SHS) | ||||
| 3. Headspace solid-phase microextraction (HS-SPME) | 2. Multidimentional GC x GC (DB-5 x Supelcowax 10) | 2. Flame Ionization Detector (GC-FID) | [107] | |
| 4. Dynamic HS | ||||
| 5. Headspace sorptive extraction (HSSE) and stir bar sorptive extraction (SBSE) | 3. Olfactometry detector (GC-O) - Aroma Extract Dilution Analysis (AEDA) - Odor activity values (OAVs)Odor spectrum values (OSV) | [25,79] | ||
| Proton Transfer Reaction-MS (PTR-MS) | Direct Injection of VOCs from HS | Not applicable | PTR-ToF-MS | [92,108,109] |
| Electronic Nose (EN) sensing | HS | Not applicable | Metal-oxide Sensors (MOS) | [11,76] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Angeloni, S.; Mustafa, A.M.; Abouelenein, D.; Alessandroni, L.; Acquaticci, L.; Nzekoue, F.K.; Petrelli, R.; Sagratini, G.; Vittori, S.; Torregiani, E.; et al. Characterization of the Aroma Profile and Main Key Odorants of Espresso Coffee. Molecules 2021, 26, 3856. https://doi.org/10.3390/molecules26133856
Angeloni S, Mustafa AM, Abouelenein D, Alessandroni L, Acquaticci L, Nzekoue FK, Petrelli R, Sagratini G, Vittori S, Torregiani E, et al. Characterization of the Aroma Profile and Main Key Odorants of Espresso Coffee. Molecules. 2021; 26(13):3856. https://doi.org/10.3390/molecules26133856
Chicago/Turabian StyleAngeloni, Simone, Ahmed M. Mustafa, Doaa Abouelenein, Laura Alessandroni, Laura Acquaticci, Franks Kamgang Nzekoue, Riccardo Petrelli, Gianni Sagratini, Sauro Vittori, Elisabetta Torregiani, and et al. 2021. "Characterization of the Aroma Profile and Main Key Odorants of Espresso Coffee" Molecules 26, no. 13: 3856. https://doi.org/10.3390/molecules26133856
APA StyleAngeloni, S., Mustafa, A. M., Abouelenein, D., Alessandroni, L., Acquaticci, L., Nzekoue, F. K., Petrelli, R., Sagratini, G., Vittori, S., Torregiani, E., & Caprioli, G. (2021). Characterization of the Aroma Profile and Main Key Odorants of Espresso Coffee. Molecules, 26(13), 3856. https://doi.org/10.3390/molecules26133856













