Plasma-Based Proteomics Profiling of Patients with Hyperthyroidism after Antithyroid Treatment
Abstract
1. Introduction
2. Results
2.1. Biochemical Parameters of the Study Subjects
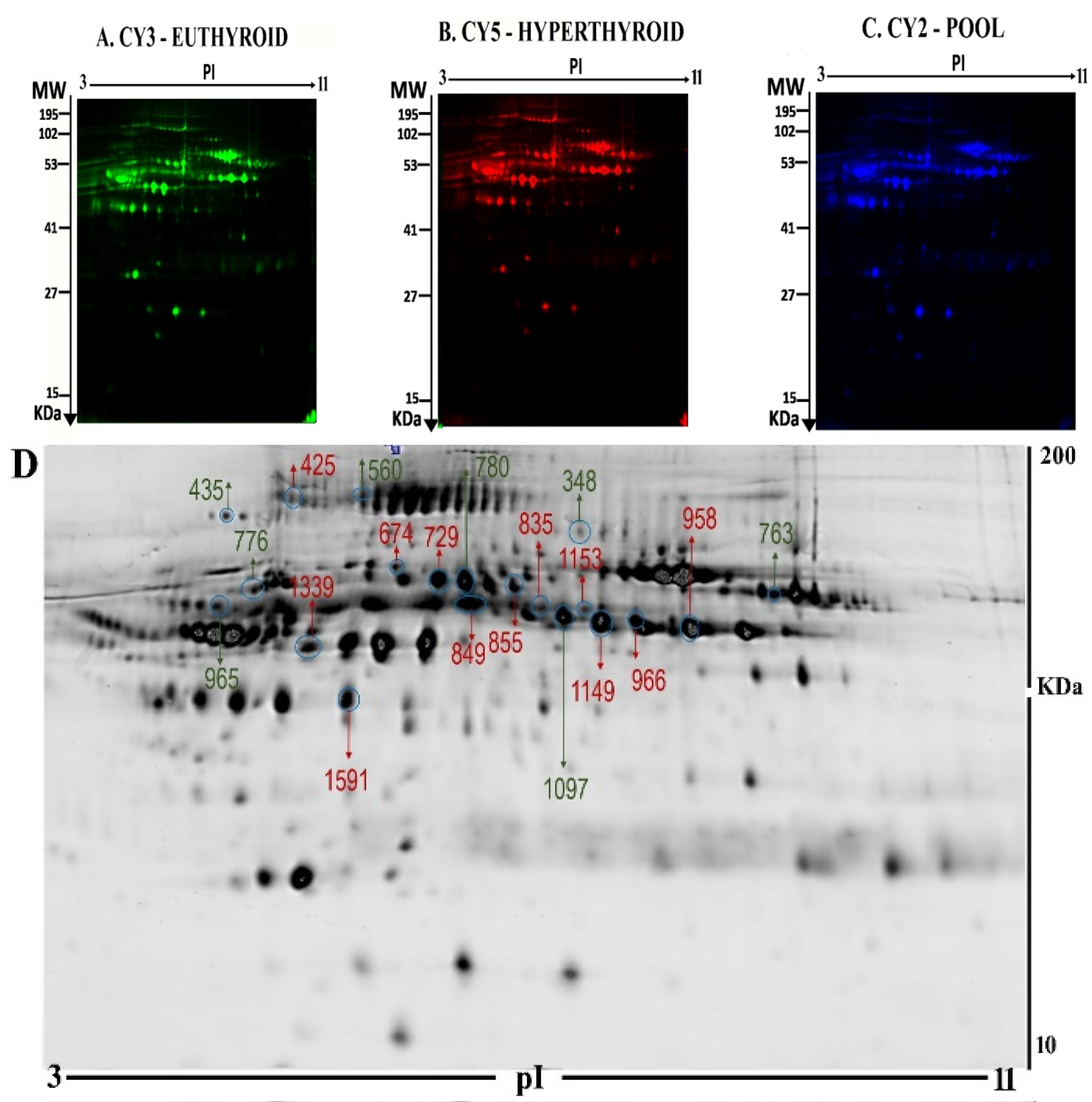
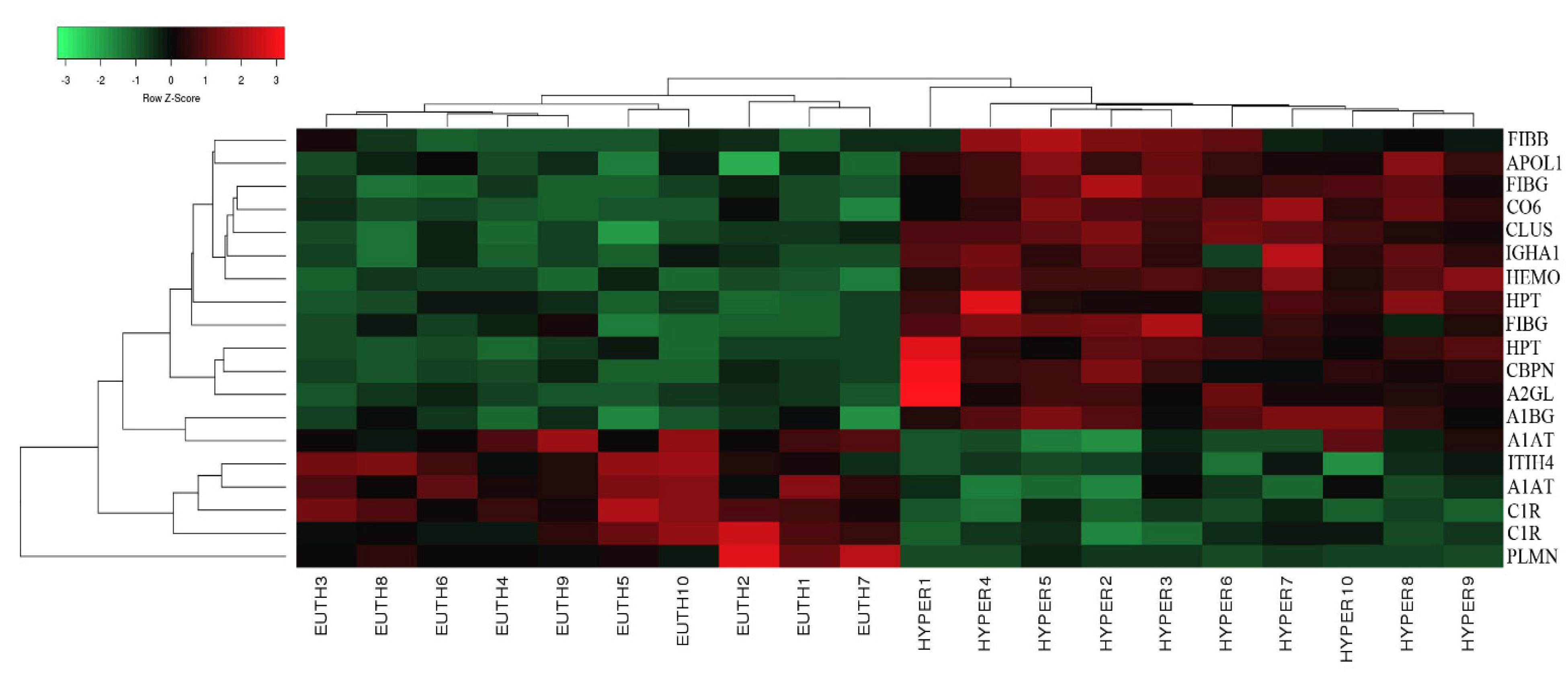
2.2. Mass Spectrometry Identification of Differentially Expressed Proteins on 2D-DIGE
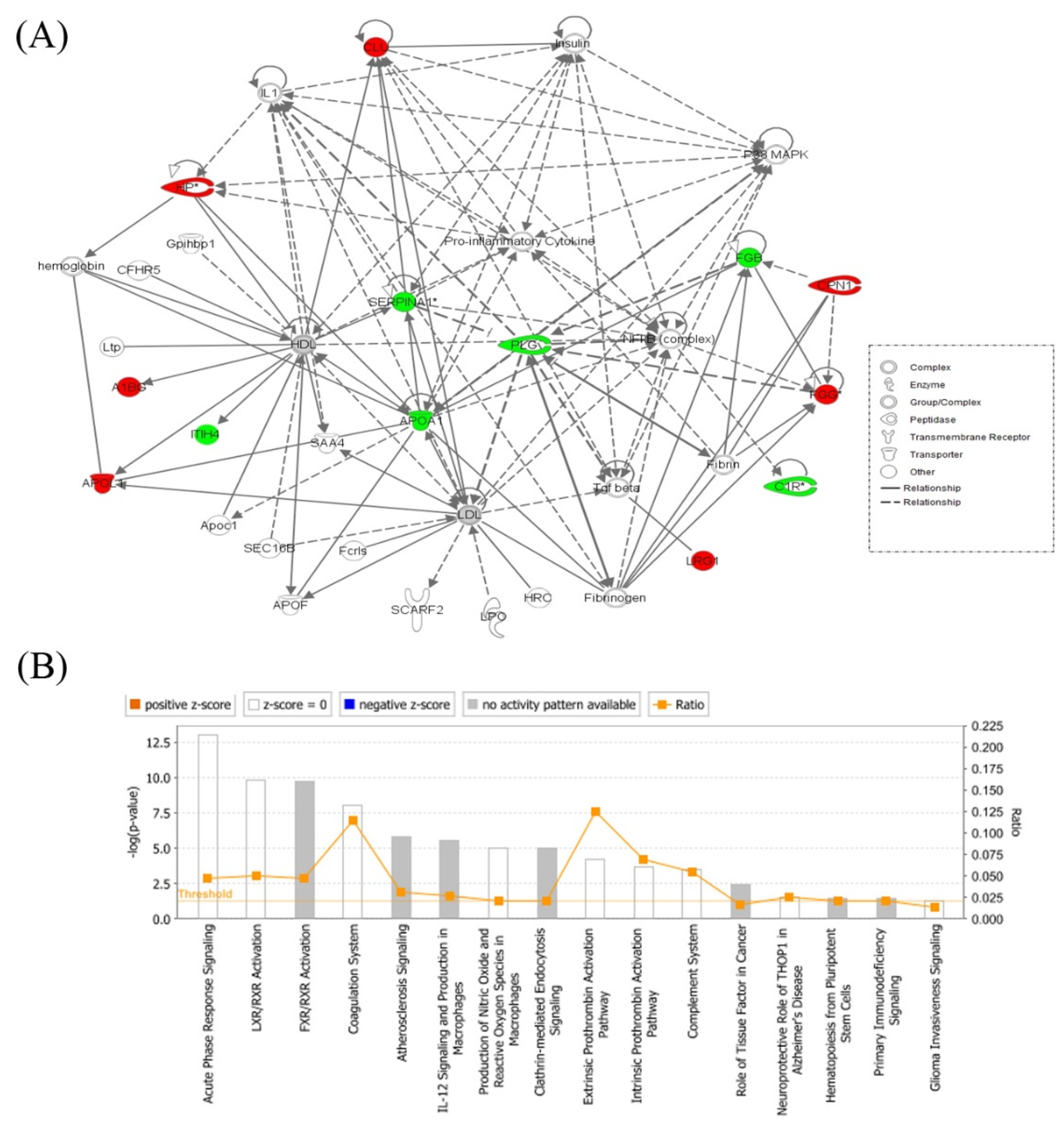
2.3. Interactions of Identified Proteins and Network Connectivity Mapping Using Ingenuity Pathway Analysis (IPA)
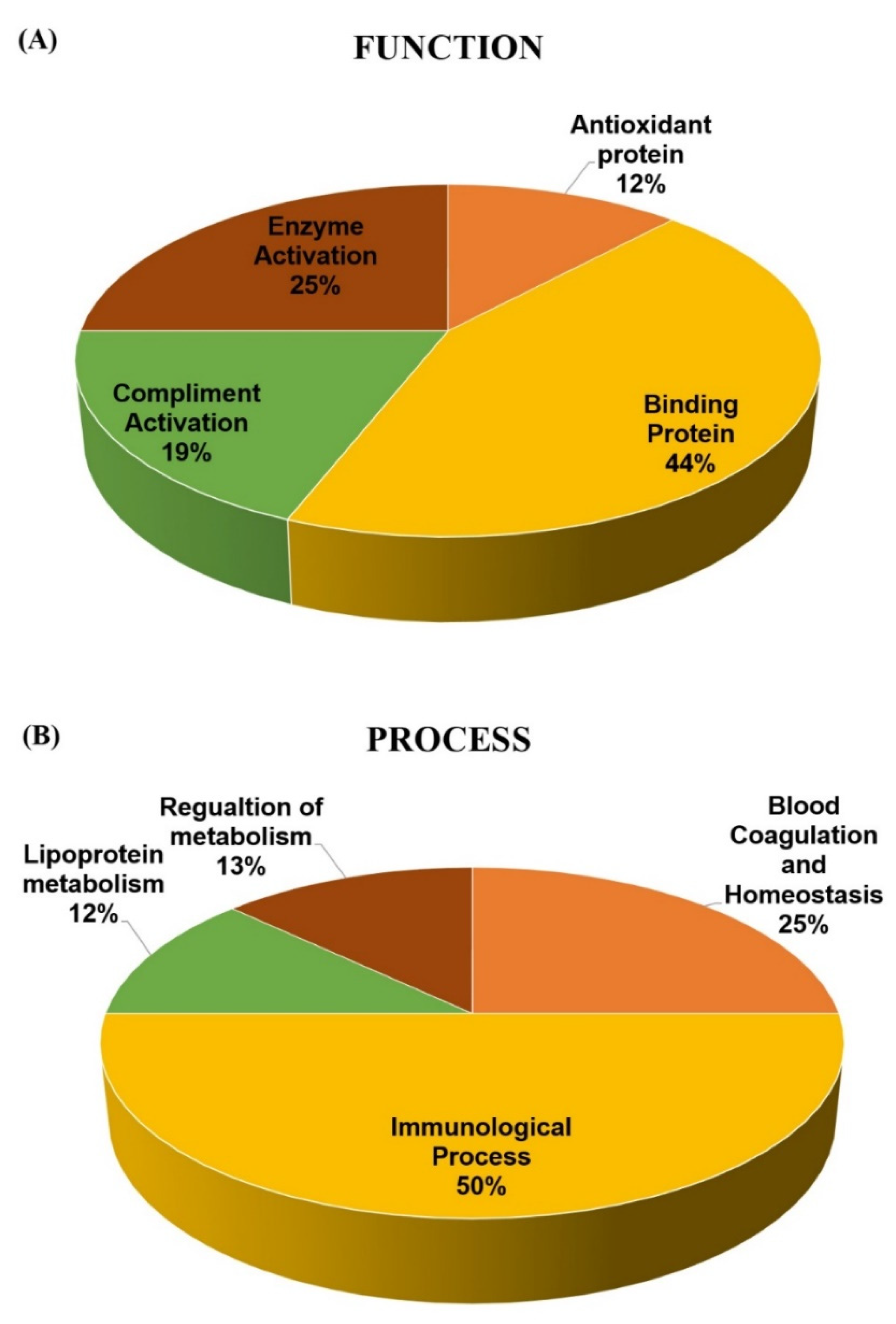
2.4. Classification of Key Proteins ased on Function
3. Discussion and Conclusions
3.1. Differential Regulation of Proteins Involved in Lipid Metabolism
3.2. Differential Regulation of Proteins Involved in the Acute Phase Immune Response
3.3. Differential Regulation of Proteins Involved in Vascular Homeostasis and Coagulation
3.4. Network Pathway Analysis of the Significant Differentially Abundant Proteins
4. Materials and Methods
4.1. Ethical Considerations and Informed Consent
4.2. Study Design and Subjects
4.3. Biochemical Analysis
4.4. Sample Processing and Protein Extraction
4.5. CyDye labeling, 2D-DIGE and Imaging
4.6. Colloidal Coomassie Blue Staining of the Preparative Gel
4.7. Protein Identification by MALDO-TOF MS
4.8. Bionformatic Analysis
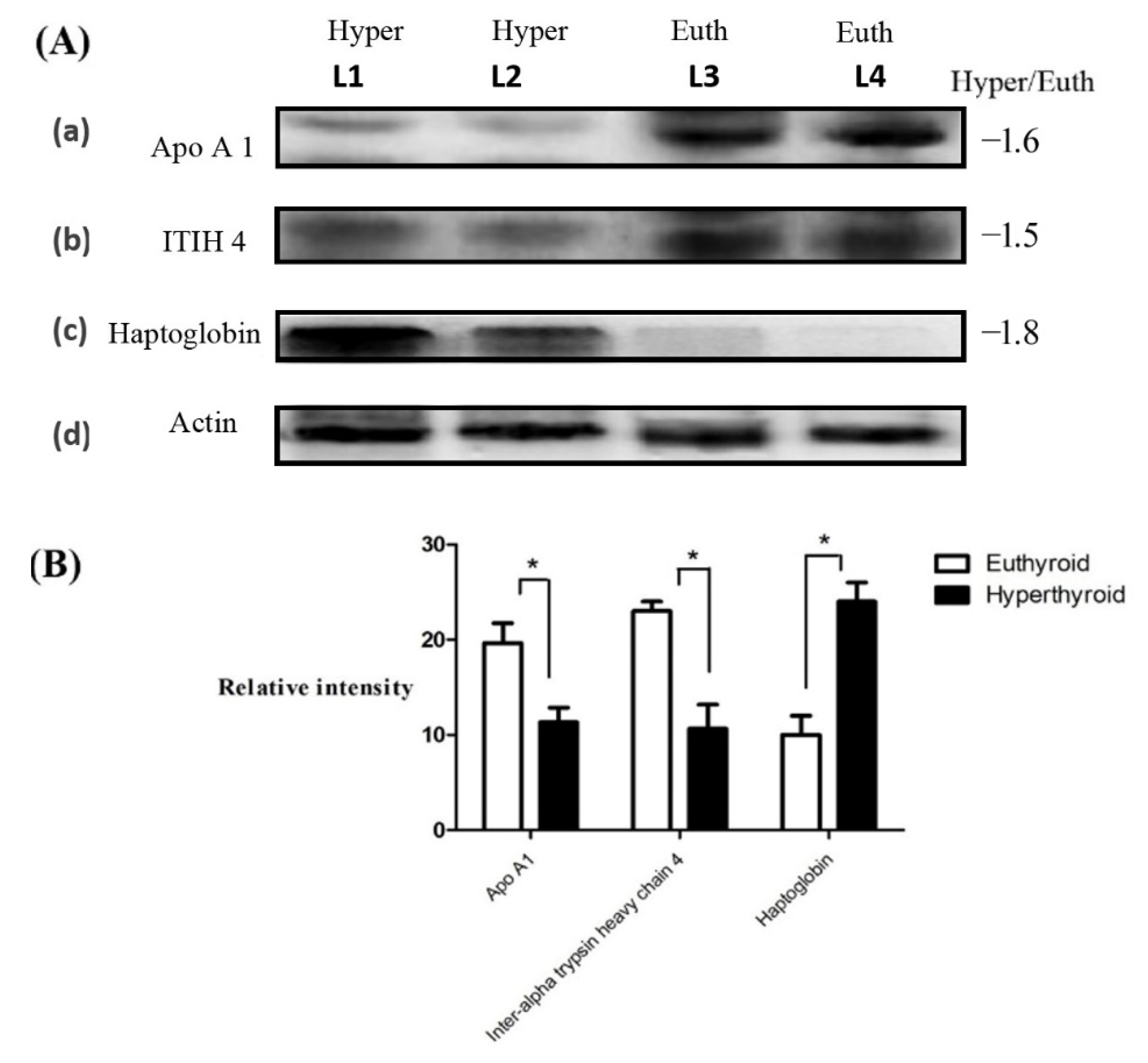
4.9. Immunoblotting
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brent, G.A. Mechanisms of thyroid hormone action. J. Clin. Investig. 2012, 122, 3035–3043. [Google Scholar] [CrossRef]
- Yen, P.M. Physiological and molecular basis of thyroid hormone action. Physiol. Rev. 2001, 81, 1097–1142. [Google Scholar] [CrossRef]
- Kawa, M.P.; Grymula, K.; Paczkowska, E.; Baskiewicz-Masiuk, M.; Dabkowska, E.; Koziolek, M.; Tarnowski, M.; Klos, P.; Dziedziejko, V.; Kucia, M.; et al. Clinical relevance of thyroid dysfunction in human haematopoiesis: Biochemical and molecular studies. Eur. J. Endocrinol. 2010, 162, 295–305. [Google Scholar] [CrossRef]
- Lopez, M.; Alvarez, C.V.; Nogueiras, R.; Dieguez, C. Energy balance regulation by thyroid hormones at central level. Trends Mol. Med. 2013, 19, 418–427. [Google Scholar] [CrossRef]
- Mullur, R.; Liu, Y.Y.; Brent, G.A. Thyroid hormone regulation of metabolism. Physiol. Rev. 2014, 94, 355–382. [Google Scholar] [CrossRef]
- Clausen, T.; Van Hardeveld, C.; Everts, M.E. Significance of cation transport in control of energy metabolism and thermogenesis. Physiol. Rev. 1991, 71, 733–774. [Google Scholar] [CrossRef]
- Lin, K.H.; Lee, H.Y.; Shih, C.H.; Yen, C.C.; Chen, S.L.; Yang, R.C.; Wang, C.S. Plasma protein regulation by thyroid hormone. J. Endocrinol. 2003, 179, 367–377. [Google Scholar] [CrossRef]
- Mendoza, A.; Hollenberg, A.N. New insights into thyroid hormone action. Pharmacol. Ther. 2017, 173, 135–145. [Google Scholar] [CrossRef]
- Pietzner, M.; Engelmann, B.; Kacprowski, T.; Golchert, J.; Dirk, A.L.; Hammer, E.; Iwen, K.A.; Nauck, M.; Wallaschofski, H.; Fuhrer, D.; et al. Plasma proteome and metabolome characterization of an experimental human thyrotoxicosis model. BMC Med. 2017, 15, 6. [Google Scholar] [CrossRef]
- Engelmann, B.; Bischof, J.; Dirk, A.L.; Friedrich, N.; Hammer, E.; Thiele, T.; Fuhrer, D.; Homuth, G.; Brabant, G.; Volker, U. Effect of Experimental Thyrotoxicosis onto Blood Coagulation: A Proteomics Study. Eur. Thyroid. J. 2015, 4, 119–124. [Google Scholar] [CrossRef]
- Alfadda, A.A.; Benabdelkamel, H.; Masood, A.; Jammah, A.A.; Ekhzaimy, A.A. Differences in the Plasma Proteome of Patients with Hypothyroidism before and after Thyroid Hormone Replacement: A Proteomic Analysis. Int. J. Mol. Sci. 2018, 19, 88. [Google Scholar] [CrossRef]
- Duntas, L.H.; Brenta, G. A Renewed Focus on the Association Between Thyroid Hormones and Lipid Metabolism. Front. Endocrinol. 2018, 9, 511. [Google Scholar] [CrossRef]
- Muls, E.; Blaton, V.; Rosseneu, M.; Lesaffre, E.; Lamberigts, G.; De Moor, P. Serum lipids and apolipoproteins A-I, A-II, and B in hyperthyroidism before and after treatment. J. Clin. Endocrinol. Metab. 1982, 55, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Benvenga, S.; Alesci, S.; Trimarchi, F. High-density lipoprotein-facilitated entry of thyroid hormones into cells: A mechanism different from the low-density lipoprotein-facilitated entry. Thyroid. Off. J. Am. Thyroid Assoc. 2002, 12, 547–556. [Google Scholar] [CrossRef]
- Pamir, N.; Hutchins, P.M.; Ronsein, G.E.; Wei, H.; Tang, C.; Das, R.; Vaisar, T.; Plow, E.; Schuster, V.; Koschinsky, M.L.; et al. Plasminogen promotes cholesterol efflux by the ABCA1 pathway. JCI Insight 2017, 2, e92176. [Google Scholar] [CrossRef]
- Wan, G.; Zhaorigetu, S.; Liu, Z.; Kaini, R.; Jiang, Z.; Hu, C.A. Apolipoprotein L1, a novel Bcl-2 homology domain 3-only lipid-binding protein, induces autophagic cell death. J. Biol. Chem. 2008, 283, 21540–21549. [Google Scholar] [CrossRef]
- Cubedo, J.; Padro, T.; Alonso, R.; Mata, P.; Badimon, L. ApoL1 levels in high density lipoprotein and cardiovascular event presentation in patients with familial hypercholesterolemia. J. Lipid Res. 2016, 57, 1059–1073. [Google Scholar] [CrossRef]
- Hoofnagle, A.N.; Wu, M.; Gosmanova, A.K.; Becker, J.O.; Wijsman, E.M.; Brunzell, J.D.; Kahn, S.E.; Knopp, R.H.; Lyons, T.J.; Heinecke, J.W. Low clusterin levels in high-density lipoprotein associate with insulin resistance, obesity, and dyslipoproteinemia. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 2528–2534. [Google Scholar] [CrossRef] [PubMed]
- Trougakos, I.P.; Lourda, M.; Agiostratidou, G.; Kletsas, D.; Gonos, E.S. Differential effects of clusterin/apolipoprotein J on cellular growth and survival. Free Radic. Biol. Med. 2005, 38, 436–449. [Google Scholar] [CrossRef]
- Tapia, G.; Fernandez, V.; Varela, P.; Cornejo, P.; Guerrero, J.; Videla, L.A. Thyroid hormone-induced oxidative stress triggers nuclear factor-kappaB activation and cytokine gene expression in rat liver. Free Radic. Biol. Med. 2003, 35, 257–265. [Google Scholar] [CrossRef]
- Griffin, E.E.; Miller, L.L. Effects of hypothyroidism, hyperthyroidism, and thyroxine on net synthesis of plasma proteins by the isolated perfused rat liver. Modulation of the response to insulin plus cortisol in the net synthesis of albumin, fibrinogen, 1-acid glycorprotein, 2-(acute phase) globulin, and haptoglobin. J. Biol. Chem. 1973, 248, 4716–4723. [Google Scholar]
- Salvatore, A.; Cigliano, L.; Bucci, E.M.; Corpillo, D.; Velasco, S.; Carlucci, A.; Pedone, C.; Abrescia, P. Haptoglobin binding to apolipoprotein A-I prevents damage from hydroxyl radicals on its stimulatory activity of the enzyme lecithin-cholesterol acyl-transferase. Biochemistry 2007, 46, 11158–11168. [Google Scholar] [CrossRef]
- Krisp, C.; Randall, S.A.; McKay, M.J.; Molloy, M.P. Towards clinical applications of selected reaction monitoring for plasma protein biomarker studies. Proteomics. Clin. Appl. 2012, 6, 42–59. [Google Scholar] [CrossRef]
- Ferrari, E.; Wittig, A.; Basilico, F.; Rossi, R.; De Palma, A.; Di Silvestre, D.; Sauerwein, W.A.G.; Mauri, P.L. Urinary Proteomics Profiles Are Useful for Detection of Cancer Biomarkers and Changes Induced by Therapeutic Procedures. Molecules 2019, 24, 794. [Google Scholar] [CrossRef]
- Guttman, O.; Baranovski, B.M.; Schuster, R.; Kaner, Z.; Freixo-Lima, G.S.; Bahar, N.; Kalay, N.; Mizrahi, M.I.; Brami, I.; Ochayon, D.E.; et al. Acute-phase protein alpha1-anti-trypsin: Diverting injurious innate and adaptive immune responses from non-authentic threats. Clin. Exp. Immunol. 2015, 179, 161–172. [Google Scholar] [CrossRef]
- Poblete, M.T.; Nualart, F.; del Pozo, M.; Perez, J.A.; Figueroa, C.D. Alpha 1-antitrypsin expression in human thyroid papillary carcinoma. Am. J. Surg. Pathol. 1996, 20, 956–963. [Google Scholar] [CrossRef]
- El-Akawi, Z.J.; Abu-Awad, A.M.; Khouri, N.A. Alpha-1 Antitrypsin Blood Levels as Indicator for the Efficacy of Cancer Treatment. World J. Oncol. 2013, 4, 83–86. [Google Scholar] [CrossRef][Green Version]
- Kim, H.J.; Yoo, E.K.; Kim, J.Y.; Choi, Y.K.; Lee, H.J.; Kim, J.K.; Jeoung, N.H.; Lee, K.U.; Park, I.S.; Min, B.H.; et al. Protective role of clusterin/apolipoprotein J against neointimal hyperplasia via antiproliferative effect on vascular smooth muscle cells and cytoprotective effect on endothelial cells. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 1558–1564. [Google Scholar] [CrossRef]
- Rosenberg, M.E.; Silkensen, J. Clusterin: Physiologic and pathophysiologic considerations. Int. J. Biochem. Cell Biol. 1995, 27, 633–645. [Google Scholar] [CrossRef]
- Seo, J.A.; Kang, M.C.; Ciaraldi, T.P.; Kim, S.S.; Park, K.S.; Choe, C.; Hwang, W.M.; Lim, D.M.; Farr, O.; Mantzoros, C.; et al. Circulating ApoJ is closely associated with insulin resistance in human subjects. Metab. Clin. Exp. 2018, 78, 155–166. [Google Scholar] [CrossRef]
- Ricklin, D.; Hajishengallis, G.; Yang, K.; Lambris, J.D. Complement: A key system for immune surveillance and homeostasis. Nat. Immunol. 2010, 11, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Weetman, A.P.; Tandon, N.; Morgan, B.P. Antithyroid drugs and release of inflammatory mediators by complement-attacked thyroid cells. Lancet 1992, 340, 633–636. [Google Scholar] [CrossRef]
- Wijeyewickrema, L.C.; Yongqing, T.; Tran, T.P.; Thompson, P.E.; Viljoen, J.E.; Coetzer, T.H.; Duncan, R.C.; Kass, I.; Buckle, A.M.; Pike, R.N. Molecular determinants of the substrate specificity of the complement-initiating protease, C1r. J. Biol. Chem. 2013, 288, 15571–15580. [Google Scholar] [CrossRef]
- Erem, C. Blood coagulation, fibrinolytic activity and lipid profile in subclinical thyroid disease: Subclinical hyperthyroidism increases plasma factor X activity. Clin. Endocrinol. 2006, 64, 323–329. [Google Scholar] [CrossRef]
- Bensalah, M.; Squizzato, A.; Ould Kablia, S.; Menia, H.; Kemali, Z. Cerebral vein and sinus thrombosis and hyperthyrodism: A case report and a systematic review of the literature. Thromb. Res. 2011, 128, 98–100. [Google Scholar] [CrossRef]
- Poplawska-Kita, A.; Siewko, K.; Telejko, B.; Modzelewska, A.; Mysliwiec, J.; Milewski, R.; Gorska, M.; Szelachowska, M. The changes in the endothelial function and haemostatic and inflammatory parameters in subclinical and overt hyperthyroidism. Int. J. Endocrinol. 2013, 2013, 981638. [Google Scholar] [CrossRef]
- Duckers, J.M.; Shale, D.J.; Stockley, R.A.; Gale, N.S.; Evans, B.A.; Cockcroft, J.R.; Bolton, C.E. Cardiovascular and musculskeletal co-morbidities in patients with alpha 1 antitrypsin deficiency. Respir. Res. 2010, 11, 173. [Google Scholar] [CrossRef]
- Mousa, S.S.; Davis, F.B.; Davis, P.J.; Mousa, S.A. Human platelet aggregation and degranulation is induced in vitro by L-thyroxine, but not by 3,5,3’-triiodo-L-thyronine or diiodothyropropionic acid (DITPA). Clin. Appl. Thromb. Hemost. Off. J. Int. Acad. Clin. Appl. Thromb. Hemost. 2010, 16, 288–293. [Google Scholar] [CrossRef]
- Davis, P.J.; Mousa, S.A.; Schechter, G.P. New Interfaces of Thyroid Hormone Actions With Blood Coagulation and Thrombosis. Clin. Appl. Thromb. Hemost. Off. J. Int. Acad. Clin. Appl. Thromb. Hemost. 2018, 24, 1014–1019. [Google Scholar] [CrossRef]
- Marongiu, F.; Conti, M.; Mameli, G.; Murtas, M.L.; Balzano, S.; Sorano, G.; Mamusa, A.M.; Martino, E. Fibrinogen and fibrinolytic activity in hyperthyroidism before and after antithyroid treatment. J. Endocrinol. Investig. 1988, 11, 723–725. [Google Scholar] [CrossRef]
- Lancellotti, S.; Rutella, S.; De Filippis, V.; Pozzi, N.; Rocca, B.; De Cristofaro, R. Fibrinogen-elongated gamma chain inhibits thrombin-induced platelet response, hindering the interaction with different receptors. J. Biol. Chem. 2008, 283, 30193–30204. [Google Scholar] [CrossRef] [PubMed]
- Reale, C.; Zotti, T.; Scudiero, I.; Vito, P.; Stilo, R. The NF-kappaB Family of Transcription Factors and Its Role in Thyroid Physiology. Vitam. Horm. 2018, 106, 195–210. [Google Scholar]
- Nandakumar, D.N.; Koner, B.C.; Vinayagamoorthi, R.; Nanda, N.; Negi, V.S.; Goswami, K.; Bobby, Z.; Hamide, A. Activation of NF-kappaB in lymphocytes and increase in serum immunoglobulin in hyperthyroidism: Possible role of oxidative stress. Immunobiology 2008, 213, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Moeller, L.C.; Broecker-Preuss, M. Transcriptional regulation by nonclassical action of thyroid hormone. Thyroid. Res. 2011, 4, S6. [Google Scholar] [CrossRef]
- Fliser, E.; Jerkovic, K.; Vidovic, T.; Gorenjak, M. Investigation of unusual high serum indices for lipemia in clear serum samples on siemens analysers dimension. Biochem. Med. 2012, 22, 352–362. [Google Scholar] [CrossRef]
- Tremblay, A.J.; Morrissette, H.; Gagne, J.M.; Bergeron, J.; Gagne, C.; Couture, P. Validation of the Friedewald formula for the determination of low-density lipoprotein cholesterol compared with beta-quantification in a large population. Clin. Biochem. 2004, 37, 785–790. [Google Scholar] [CrossRef]
- Rabilloud, T. Detecting proteins separated by 2-D gel electrophoresis. Anal. Chem. 2000, 72, 48A–55A. [Google Scholar] [CrossRef]
- Alfadda, A.A.; Benabdelkamel, H.; Masood, A.; Moustafa, A.; Sallam, R.; Bassas, A.; Duncan, M. Proteomic analysis of mature adipocytes from obese patients in relation to aging. Exp. Gerontol. 2013, 48, 1196–1203. [Google Scholar] [CrossRef]
- Shevchenko, A.; Wilm, M.; Vorm, O.; Mann, M. Mass spectrometric sequencing of proteins silver-stained polyacrylamide gels. Anal. Chem. 1996, 68, 850–858. [Google Scholar] [CrossRef]
- Babicki, S.; Arndt, D.; Marcu, A.; Liang, Y.; Grant, J.R.; Maciejewski, A.; Wishart, D.S. Heatmapper: Web-enabled heat mapping for all. Nucleic Acids Res. 2016, 44, W147–W153. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
| Hyperthyroid | Euthyroid | p value | |
|---|---|---|---|
| N | 10 | ||
| Age(years) | 39.6 ± 10.6 | ||
| Glucose (mmol/L) | 5.3 ± 0.8 | 5.2 ± 0.5 | 0.19 |
| Urea (mmol/L) | 3.9 ± 0.9 | 4.6 ± 0.8 | 0.18 |
| Creatinine (umol/L) | 61.1 ± 12.7 | 66.3 ± 12.0 | 0.39 |
| Sodium (mmol/L) | 138.6 ± 2.1 | 138.3 ± 0.9 | 0.16 |
| Potassium (mmol/L) | 4.2 ± 0.2 | 4.3 ± 0.4 | 0.40 |
| Aspartate transaminase (IU/L) | 34.8 ± 9.1 | 31.8 ± 4.5 | 0.38 |
| Alanine transaminase (IU/L) | 17.5 ± 5 | 15.8 ± 2.0 | 0.40 |
| Alkaline phosphatase (IU/L) | 114.6 ± 53.7 | 120.2 ± 28.9 | 0.30 |
| FT4 (pmol/L) | 35.4 ± 9.9 | 17.0 ± 2.8 | 0.001 |
| TSH (mIU/L) | 0.014 ± 0.01 | 0.8 ± 0.4 | 0.00068 |
| Total cholesterol (mmol/L) | 4.6 ± 1.0 | 4.9 ± 0.8 | 0.10 |
| HDL cholesterol (mmol/L) | 1 ± 0.2 | 1.4 ± 0.2 | 0.02 |
| LDL cholesterol (mmol/L) | 2.8 ± 0.9 | 3.1 ± 0.7 | 0.09 |
| Triglycerides (mmol/L) | 1.1 ± 0.6 | 0.9 ± 0.2 | 0.14 |
| Spot No | Accession No a | Protein Name | MASCOT ID | Pi b | MW c | Cov% | Score d | p-Value (ANOVA) | Ratio Hyper/Control | EXP e |
|---|---|---|---|---|---|---|---|---|---|---|
| 1153 | P00738 | Haptoglobin | HPT_HUMAN | 6.13 | 45861 | 23 | 78 | 0.01 | 1.5 | UP |
| 729 | P01876 | Ig alpha-1 chain C region | IGHA1_HUMAN | 6.08 | 38486 | 42 | 58 | 0.01 | 1.5 | UP |
| 1149 | P00738 | Haptoglobin | HPT _HUMAN | 6.13 | 45861 | 27 | 69 | 0.01 | 1.7 | UP |
| 425 | P13671 | Complement component C6 | CO6_HUMAN | 6.39 | 108367 | 25 | 57 | 0.05 | 1.5 | UP |
| 966 | O14791 | Apolipoprotein L1 | APOL1_HUMAN | 5.60 | 44004 | 32 | 59 | 0.05 | 1.5 | UP |
| 855 | P02750 | Leucine-rich alpha-2-glycoprotein | A2GL_HUMAN | 6.45 | 38382 | 42 | 71 | 0.05 | 1.5 | UP |
| 1097 | P02647 | Apolipoprotein A-I | APOA1_HUMAN | 5.56 | 30759 | 72 | 238 | 0.05 | 1.5 | DOWN |
| 674 | P02679 | Fibrinogen gamma chain | FIBG_HUMAN | 5.37 | 52106 | 50 | 102 | 0.012 | 1.5 | UP |
| 348 | P00747 | Plasminogen | PLMN_HUMAN | 7.04 | 93247 | 30 | 77 | 0.016 | 1.8 | DOWN |
| 776 | P01009 | Alpha-1-antitrypsin | A1AT_HUMAN | 5.37 | 46878 | 40 | 78 | 0.039 | 1.7 | DOWN |
| 965 | P01009 | Alpha-1-antitrypsin | A1AT_HUMAN | 5.37 | 46878 | 36 | 68 | 0.047 | 1.5 | DOWN |
| 958 | P10909 | Clusterin | CLUS_HUMAN | 5.89 | 53031 | 26 | 78 | 0.053 | 1.5 | UP |
| 763 | P02675 | Fibrinogen beta chain | FIBB_HUMAN | 8.54 | 56577 | 55 | 205 | 0.050 | 1.5 | DOWN |
| 435 | P00736 | Complement C1r subcomponent | C1R_HUMAN | 5.82 | 81606 | 26 | 99 | 0.051 | 1.7 | DOWN |
| 780 | P00736 | Complement C1r subcomponent | C1R_HUMAN | 5.82 | 81606 | 33 | 115 | 0.0268 | 1.8 | DOWN |
| 560 | Q14624 | Inter-alpha-trypsin inhibitor heavy chain H4 | ITIH4_HUMAN | 6.51 | 103521 | 37 | 85 | 0.0386 | 1.5 | DOWN |
| 1339 | P04217 | Alpha-1B-glycoprotein | A1BG_HUMAN | 5.58 | 54809 | 37 | 90 | 0.016 | 1.5 | UP |
| 1591 | P02679 | Fibrinogen gamma chain | FIBG_HUMAN | 5.37 | 52106 | 50 | 129 | 0.0372 | 1.5 | UP |
| 849 | P15169 | Carboxypeptidase N catalytic chain | CBPN_HUMAN | 6.86 | 52538 | 33 | 62 | 0.021 | 1.5 | UP |
| 835 | P02790 | Hemopexin | HEMO_HUMAN | 6.55 | 52385 | 48 | 123 | 0.025 | 1.5 | UP |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masood, A.; Benabdelkamel, H.; Ekhzaimy, A.A.; Alfadda, A.A. Plasma-Based Proteomics Profiling of Patients with Hyperthyroidism after Antithyroid Treatment. Molecules 2020, 25, 2831. https://doi.org/10.3390/molecules25122831
Masood A, Benabdelkamel H, Ekhzaimy AA, Alfadda AA. Plasma-Based Proteomics Profiling of Patients with Hyperthyroidism after Antithyroid Treatment. Molecules. 2020; 25(12):2831. https://doi.org/10.3390/molecules25122831
Chicago/Turabian StyleMasood, Afshan, Hicham Benabdelkamel, Aishah A. Ekhzaimy, and Assim A. Alfadda. 2020. "Plasma-Based Proteomics Profiling of Patients with Hyperthyroidism after Antithyroid Treatment" Molecules 25, no. 12: 2831. https://doi.org/10.3390/molecules25122831
APA StyleMasood, A., Benabdelkamel, H., Ekhzaimy, A. A., & Alfadda, A. A. (2020). Plasma-Based Proteomics Profiling of Patients with Hyperthyroidism after Antithyroid Treatment. Molecules, 25(12), 2831. https://doi.org/10.3390/molecules25122831






