Solubility Data of the Bioactive Compound Piperine in (Transcutol + Water) Mixtures: Computational Modeling, Hansen Solubility Parameters and Mixing Thermodynamic Parameters
Abstract
1. Introduction
2. Results and Discussion
2.1. Experimental Solubility Values of PPN and Literature Comparison
2.2. Hansen Solubility Parameters (HSPs)
2.3. Mixing Thermodynamic Parameters of PPN Solution
2.4. Solute–Solvent Molecular Interactions
2.5. Modeling of PPN Solubility
3. Experimental
3.1. Materials
3.2. PPN Solubility Measurement
3.3. Computation of HSPs
3.4. Mixing Thermodynamics Parameters of PPN Solution
3.5. Solute–Solvent Molecular Interactions
3.6. Thermodynamics-Based Computational Models
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vasavirama, K.; Upender, M. Piperine: A valuable alkaloid from piper species. Int. J. Pharm. Pharm. Sci. 2014, 6, 34–38. [Google Scholar]
- Gorgani, G.; Mohammadi, M.; Najafpour, G.D.; Nikzad, M. Piperine-the bioactive compound of black pepper: From isolation to medicinal formulations. Compr. Rev. Food Sci. Food Saf. 2017, 16, 124–140. [Google Scholar] [CrossRef]
- Pradeep, C.R.; Kuttan, G. Effect of piperine on the inhibition of lung metastatis induced B16F-10 melanoma cells in mice. Clin. Exp. Metast. 2002, 19, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Platel, K.; Srinivasan, K. Influence of dietary spices and their active principles on pancreatic digestive enzymes in albino rats. Food/Nahrung. 2000, 44, 42–46. [Google Scholar] [CrossRef]
- Yang, Y.C.; Lee, S.G.; Lee, H.K.; Kim, M.K.; Lee, S.H.; Lee, H.S. A piperidine amide extracted from piper longum L. fruit shows activity against Aedes aegypti mosquito larvae. J. Agric. Food Chem. 2002, 50, 3765–3767. [Google Scholar] [CrossRef]
- Piyachaturawat, P.; Glinsukon, T.; Peugvicha, P. Postcoital antifertility effect of piperine. Contraception 1982, 26, 625–633. [Google Scholar] [CrossRef]
- Sethiya, N.K.; Shah, P.; Rajpara, A.; Nagar, P.A.; Mishra, S.H. Antioxidants and hepatoprotective effects of mixed micellar lipid formulation of phyllanthin and piperine in carbon tetrachloride-induced liver injury in rodents. Food Funct. 2015, 6, 3593–3603. [Google Scholar] [CrossRef]
- Bai, Y.F.; Xu, H. Protective action of piperine against experimental gastric ulcer. Acta Pharmacol. Sin. 2000, 21, 357–359. [Google Scholar]
- Meghwal, M.; Goswami, T.K. Piper nigrum and piperine: An update. Compr. Phytother. Res. 2013, 27, 1121–1130. [Google Scholar] [CrossRef]
- Shamkuwar, P.B.; Shahi, S.R. Study of antidiarrhoeal activity of piperine. Der Pharm. Lett. 2012, 4, 217–221. [Google Scholar]
- Ma, Z.G.; Yuan, Y.P.; Zhang, X.; Xu, S.C.; Wang, S.S.; Tang, Q.Z. Piperine attenuates pathological cardiac fibrosis via PPAR-γ/AKT pathways. EBioMedicine 2017, 18, 179–187. [Google Scholar] [CrossRef]
- Moon, Y.S.; Choi, W.S.; Park, E.S.; Bae, I.K.; Choi, S.D.; Paek, O.; Kim, S.H.; Chun, H.S.; Lee, S.E. Antifungal and antiaflatoxigenic methylene-dioxy containing compounds and piperine-like synthetic compounds. Toxins 2016, 8, 240. [Google Scholar] [CrossRef]
- Singh, N.K.; Saini, S.P.S.; Singh, H.; Jyoti; Sharma, S.K.; Rath, S.S. In vitro assessment of the acaricidal activity of piper longum, piper nigrum, and zingiber officinale extracts against hyalomma anatolicum ticks. Exp. Appl. Acarol. 2017, 71, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Sosa, S.; Balick, M.; Arvigo, R.; Esposito, R.; Pizza, C.; Altinier, G.; Tubaro, A. Screening of the topical anti-inflammatory activity of some Central American plants. J. Ethnopharmacol. 2002, 81, 211–215. [Google Scholar] [CrossRef]
- Bang, J.S.; Choi, H.M.; Sur, B.J.; Lim, S.J.; Kim, J.Y.; Yang, H.I.; Yoo, M.C.; Hahm, D.H.; Kim, K.S. Anti-inflammatory and antiarthritic effects of piperine in human interleukin 1β-stimulated fibroblast-like synoviocytes and in rat arthritis models. Arthritis Res. Ther. 2009, 11, 1–9. [Google Scholar] [CrossRef]
- Kaul, I.; Kapil, A. Evaluation of liver protective potential of piperine: An active principle of black pepper. Planta Med. 1993, 59, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Sabina, E.P.; Souriyan, A.D.H.; Jackline, D.; Rasool, M.K. Piperine, an active ingredient of black pepper attenuates acetaminophen-induced hepatotoxicity in mice. Asian Pac. J. Trop. Dis. 2010, 3, 971–976. [Google Scholar] [CrossRef]
- Raman, G.; Gaikar, V.G. Microwave-assisted extraction of piperine from Piper nigrum. Ind. Eng. Chem. Res. 2002, 41, 2521–2528. [Google Scholar] [CrossRef]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef]
- Mujumdar, A.M.; Dhuley, J.N.; Deshmukh, V.K.; Naik, S.R. Effect of piperine on bioavailability oxyphenylbutazone in rats. Indian Drugs 1999, 36, 123–126. [Google Scholar]
- Johnson, J.J.; Nihal, M.; Siddiqui, I.A.; Scarlett, C.O.; Bailey, H.H.; Mukhtar, H.; Ahmad, N. Enhancing the bioavailability of resveratrol by combining it with piperine. Mol. Nutr. Food Res. 2011, 55, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Shakeel, F.; Haq, N.; Salem-Bekhit, M.M.; Raish, M. Solubility and dissolution thermodynamics of sinapic acid in (DMSO + water) binary solvent mixtures at different temperatures. J. Mol. Liq. 2017, 225, 833–839. [Google Scholar] [CrossRef]
- Shakeel, F.; Haq, N.; Siddiqui, N.A.; Alanazi, F.K.; Alsarra, I.A. Solubility and thermodynamic behavior of vanillin in propane-1,2-diol + water cosolvent at different temperatures. Food Chem. 2015, 188, 57–61. [Google Scholar] [CrossRef]
- Shakeel, F.; Haq, N.; Siddiqui, N.A.; Alanazi, F.K.; Alsarra, I.A. Solubility and thermodynamics of vanillin in Carbitol-water mixtures at different temperatures. LWT Food Sci. Technol. 2015, 64, 1278–1282. [Google Scholar] [CrossRef]
- Shakeel, F.; Haq, N.; Siddiqui, N.A.; Alanazi, F.K.; Alsarra, I.A. Thermodynamics of the solubility of reserpine in {{2-(2-ethoxyethoxy)ethanol + water}} mixed solvent systems at different temperatures. J. Chem. Thermodyn. 2015, 82, 57–60. [Google Scholar] [CrossRef]
- Shakeel, F.; Haq, N.; Alanazi, F.K.; Alanazi, S.A.; Alsarra, I.A. Solubility of sinapic acid in various (Carbitol + water) systems: Computational modeling and solution thermodynamics. J. Therm. Anal. Calorim. 2020, in press. [Google Scholar] [CrossRef]
- Shakeel, F.; Alshehri, S.; Haq, N.; Elzayat, E.; Ibrahim, M.; Altamimi, M.A.; Mohsin, K.; Alanazi, F.K.; Alsarra, I.A. Solubility determination and thermodynamic data apigenin in binary {Transcutol® + water} mixtures. Ind. Crops Prod. 2018, 116, 56–63. [Google Scholar] [CrossRef]
- Ezawa, T.; Inoue, Y.; Tunvichien, S.; Suzuki, R.; Kanamoto, I. Changes in the physicochemical properties of piperine/β-cyclodextrin due to the formation of inclusion complexes. Int. J. Med. Chem. 2016, 2016, E8723139. [Google Scholar] [CrossRef]
- Khatri, S.; Awasthi, R. Piperine containing floating microspheres: An approach for drug targeting to the upper gastrointestinal tract. Drug Deliv. Transl. Res. 2016, 6, 299–303. [Google Scholar] [CrossRef]
- Veerareddy, P.R.; Vobalaboina, V.; Nahid, A. Formulation and evaluation of oil-in-water emulsions of piperine in visceral leishmaniasis. Pharmazie 2004, 59, 194–197. [Google Scholar]
- Shao, B.; Cui, C.; Ji, H.; Tang, J.; Wang, Z.; Liu, H.; Qin, M.; Li, X.; Wu, L. Enhanced oral delivery of piperine by self-emulsifying drug delivery systems: In vitro, in vivo and in situ intestinal permeability studies. Drug Deliv. 2015, 22, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhai, W.; Jiang, Q.; Huang, R.; Liu, L.; Dai, J.; Gong, W.; Du, S.; Wu, Q. Curcumin-piperine mixtures in self-microemulsifying drug delivery system for ulcerative colitis therapy. Int. J. Pharm. 2015, 490, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Pentak, D. In vitro spectroscopic study of piperine-encapsulated nanosize liposomes. Eur. Biophys. J. 2016, 45, 175–186. [Google Scholar] [CrossRef]
- Cherniakov, I.; Izgelov, D.; Barasch, D.; Davidson, E.; Domb, A.J.; Hoffman, A. Piperine-pro-nanolipospheres as a novel oral delivery system of cannabinoids: Pharmacokinetic evaluation in healthy volunteers in comparison to buccal spray administration. J. Control. Release 2017, 266, 1–7. [Google Scholar] [CrossRef]
- Cherniakov, I.; Izgelov, D.; Domb, A.J.; Hoffman, A. The effect of pro nanolipospheres (PNL) formulation containing natural enhancers on the oral bioavailability of delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD) in a rat model. Eur. J. Pharm. Sci. 2017, 109, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, M.; Khan, M.; Khan, R.A.; Ahmed, B. Preparation, characterization, in vivo and biochemical evaluation of brain targeted piperine solid lipid nanoparticles in an experimentally induced Alzheimer’s disease model. J. Drug Target. 2013, 21, 300–311. [Google Scholar] [CrossRef]
- Thenmozhi, K.; Yoo, Y.J. Enhanced solubility of piperine using hydrophilic carrier-based potent solid dispersion systems. Drug Dev. Ind. Pharm. 2017, 43, 1501–1509. [Google Scholar] [CrossRef] [PubMed]
- Alshehri, S.; Haq, N.; Shakeel, F. Solubility, molecular interactions and mixing thermodynamic properties of piperine in various pure solvents at different temperatures. J. Mol. Liq. 2018, 250, 63–70. [Google Scholar] [CrossRef]
- Raman, G.; Gaikar, V.G. Extraction of piperine from piper nigrum (black pepper) by hydrotropic solubilization. Ind. Eng. Chem. Res. 2002, 41, 2966–2976. [Google Scholar] [CrossRef]
- Padalkar, K.V.; Gaikar, V.G. Extraction of piperine from piper nigrum (black pepper) by aqueous solutions of surfactant and surfactant + hydrotrope mixtures. Sep. Sci. Technol. 2008, 43, 3097–3118. [Google Scholar] [CrossRef]
- Huang, X.; Lin, X.; Guo, M.; Zou, Y. Characteristics of piperine solubility in multiple solvent. Adv. Mater. Res. 2011, 236–238, 2495–2498. [Google Scholar] [CrossRef]
- Kumoro, A.C.; Singh, H.; Hasan, M. Solubility of piperine in supercritical and near critical carbon dioxide. Chin. J. Chem. Eng. 2009, 17, 1014–1020. [Google Scholar] [CrossRef]
- Apelblat, A.; Manzurola, E. Solubilities of o-acetylsalicylic, 4-aminosalicylic, 3,5-dinitrosalicylic and p-toluic acid and magnesium-DL-aspartate in water from T = (278–348) K. J. Chem. Thermodyn. 1999, 31, 85–91. [Google Scholar] [CrossRef]
- Manzurola, E.; Apelblat, A. Solubilities of L-glutamic acid, 3-nitrobenzoic acid, acetylsalicylic, p-toluic acid, calcium-L-lactate, calcium gluconate, magnesium-DL-aspartate, and magnesium-L-lactate in water. J. Chem. Thermodyn. 2002, 34, 1127–1136. [Google Scholar] [CrossRef]
- Yalkowsky, S.H.; Roseman, T.J. Solubilization of drugs by cosolvents. In Techniques of Solubilization of Drugs; Yalkowsky, S.H., Ed.; Marcel Dekker Inc.: New York, NY, USA, 1981; pp. 91–134. [Google Scholar]
- Sotomayor, R.G.; Holguín, A.R.; Romdhani, A.; Martínez, F.; Jouyban, A. Solution thermodynamics of piroxicam in some ethanol + water mixtures and correlation with the Jouyban–Acree model. J. Sol. Chem. 2013, 42, 358–371. [Google Scholar] [CrossRef]
- Jouyban, A. Review of the cosolvency models for predicting solubility of drugs in water-cosolvent mixtures. J. Pharm. Pharm. Sci. 2008, 11, 32–58. [Google Scholar] [CrossRef]
- Babaei, M.; Shayanfar, A.; Rahimpour, E.; Barzegar-Jalali, M.; Martínez, F.; Jouyban, A. Solubility of bosentan in {propylene glycol + water} mixtures at various temperatures: Experimental data and mathematical modeling. Phys. Chem. Liq. 2019, 57, 338–348. [Google Scholar] [CrossRef]
- Alshahrani, S.M.; Shakeel, F. Solubility data and computational modeling of baricitinib in various (DMSO + water) mixtures. Molecules 2020, 25, 2124. [Google Scholar] [CrossRef]
- Higuchi, T.; Connors, K.A. Phase-solubility techniques. Adv. Anal. Chem. Inst. 1965, 4, 117–122. [Google Scholar]
- Zhu, Q.N.; Wang, Q.; Hu, Y.B.; Abliz, X. Practical determination of the solubility parameters of 1-alkyl-3-methylimidazolium bromide ([CnC1im]Br, n = 5, 6, 7, 8) ionic liquids by inverse gas chromatography and the Hansen solubility parameter. Molecules 2019, 24, 1346. [Google Scholar] [CrossRef]
- Shakeel, F.; Haq, N.; Alsarra, I.A.; Alshehri, S. Solubility, Hansen solubility parameters and thermodynamic behavior of emtricitabine in various (polyethylene glycol-400 + water) mixtures: Computational modeling and thermodynamics. Molecules 2020, 25, 1559. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; He, H.; Huang, Z.; Zhang, P.; Sha, J.; Li, T.; Ren, B. Solubility, thermodynamic modeling and Hansen solubility parameter of 5-norbornene-2,3-dicarboximide in three binary solvents (methanol, ethanol, ethyl acetate + DMF) from 278.15 K to 323.15 K. J. Mol. Liq. 2020, 300, E112097. [Google Scholar] [CrossRef]
- Smith, J.M.; Ness, H.C.V.; Abbott, M.M. Introduction to Chemical Engineering Thermodynamics; McGraw-Hill: New York, NY, USA, 2001. [Google Scholar]
- Li, X.; Cong, Y.; Du, C.; Zhao, H. Solubility and solution thermodynamics of 2-methyl-4-nitroaniline in eleven organic solvents at elevated temperatures. J. Chem. Thermodyn. 2017, 105, 276–288. [Google Scholar] [CrossRef]
- Zhao, K.; Yang, P.; Du, S.; Li, K.; Li, X.; Li, Z.; Liu, Y.; Lin, L.; Hou, B.; Gong, J. Determination and correlation of solubility and thermodynamics of mixing of 4-aminobutyric acid in mono-solvents and binary solvent mixtures. J. Chem. Thermodyn. 2016, 102, 276–286. [Google Scholar] [CrossRef]
- Vanderbilt, B.M.; Clayton, R.E. Bonding of fibrous glass to elastomers. Ind. Eng. Chem. Prod. Res. Dev. 1965, 4, 18–22. [Google Scholar] [CrossRef]
- Ruidiaz, M.A.; Delgado, D.R.; Martínez, F.; Marcus, Y. Solubility and preferential solvation of indomethacin in 1,4-dioxane + water solvent mixtures. Fluid Phase Equilib. 2010, 299, 259–265. [Google Scholar] [CrossRef]
- Manrique, Y.J.; Pacheco, D.P.; Martínez, F. Thermodynamics of mixing and solvation of ibuprofen and naproxen in propylene glycol + water cosolvent mixtures. J. Sol. Chem. 2008, 37, 165–181. [Google Scholar] [CrossRef]
- Shakeel, F.; Alshehri, S.; Imran, M.; Haq, N.; Alanazi, A.; Anwer, M.K. Experimental and computational approaches for solubility measurement of pyridazinone derivative in binary (DMSO + water) systems. Molecules 2020, 25, 171. [Google Scholar] [CrossRef]
- Hildebrand, J.H.; Prausnitz, J.M.; Scott, R.L. Regular and Related Solutions; Van Nostrand Reinhold: New York, NY, USA, 1970. [Google Scholar]
- Jouyban, A.; Chan, H.K.; Chew, N.Y.; Khoubnasabiafari, N.; Acree, W.E., Jr. Solubility prediction of paracetamol in binary and ternary solvent mixtures using Jouyban-Acree model. Chem. Pharm. Bull. 2006, 54, 428–431. [Google Scholar] [CrossRef]
- Jouyban, A.; Acree, W.E., Jr. In silico prediction of drug solubility in water-ethanol mixtures using Jouyban-Acree model. J. Pharm. Pharm. Sci. 2006, 9, 262–269. [Google Scholar]
- Khoubnasabjafari, M.; Shayanfar, A.; Martínez, F.; Acree, W.E., Jr.; Jouyban, A. Generally trained models to predict solubility of drugs in carbitol + water mixtures at various temperatures. J. Mol. Liq. 2016, 219, 435–438. [Google Scholar] [CrossRef]
- Jouyban, A.; Fakhree, M.A.A.; Acree, W.E., Jr. Comment on “Measurement and correlation of solubilities of (Z)-2-(2-aminothiazol-4-yl)-2-methoxyiminoacetic acid in different pure solvents and binary mixtures of water + (ethanol, methanol, or glycol)”. J. Chem. Eng. Data 2012, 57, 1344–1346. [Google Scholar] [CrossRef]
- Jouyban-Gharamaleki, A.; Hanaee, J. A novel method for improvement of predictability of the CNIBS/R-K equation. Int. J. Pharm. 1997, 154, 245–247. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compound PPN are available from the authors. |
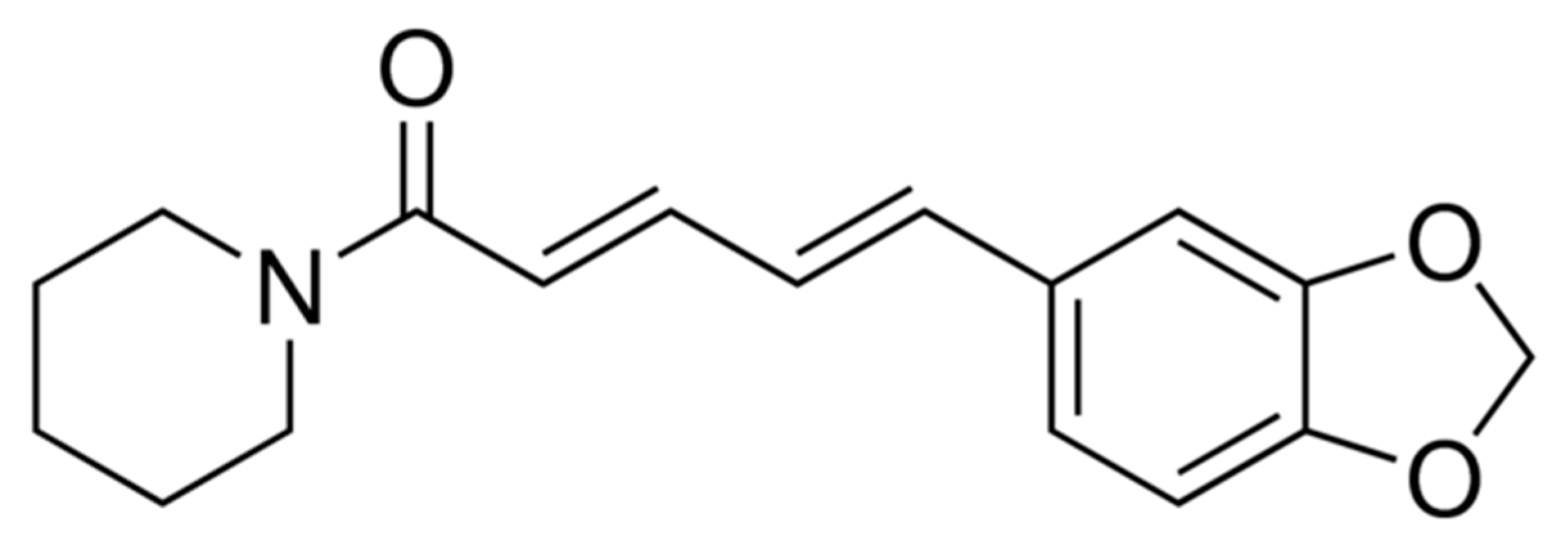
 shows the experimental mole fraction solubility of PPN in (A) pure water and (B) pure THP, and the symbol
shows the experimental mole fraction solubility of PPN in (A) pure water and (B) pure THP, and the symbol  shows the reported solubilities of PPN in (A) pure water and (B) pure THP taken from reference [38].
shows the reported solubilities of PPN in (A) pure water and (B) pure THP taken from reference [38].
 shows the experimental mole fraction solubility of PPN in (A) pure water and (B) pure THP, and the symbol
shows the experimental mole fraction solubility of PPN in (A) pure water and (B) pure THP, and the symbol  shows the reported solubilities of PPN in (A) pure water and (B) pure THP taken from reference [38].
shows the reported solubilities of PPN in (A) pure water and (B) pure THP taken from reference [38].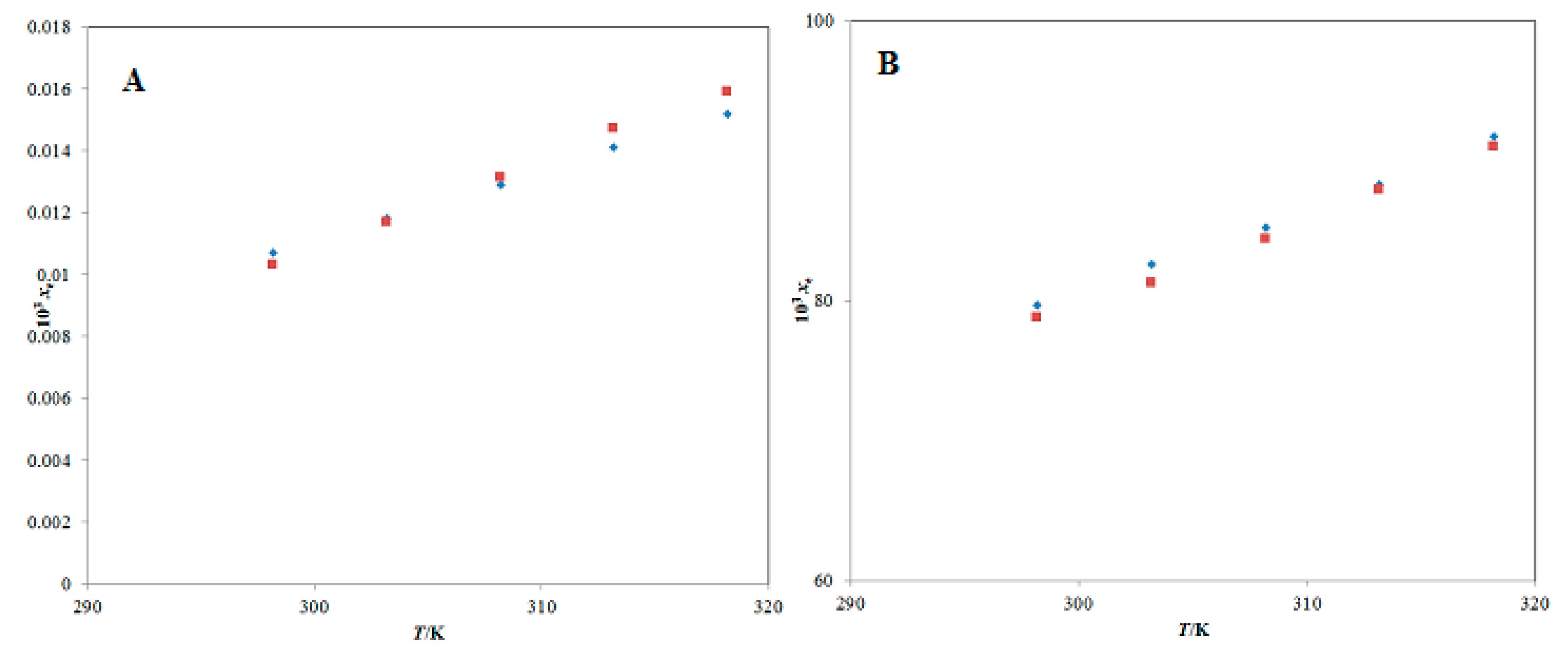
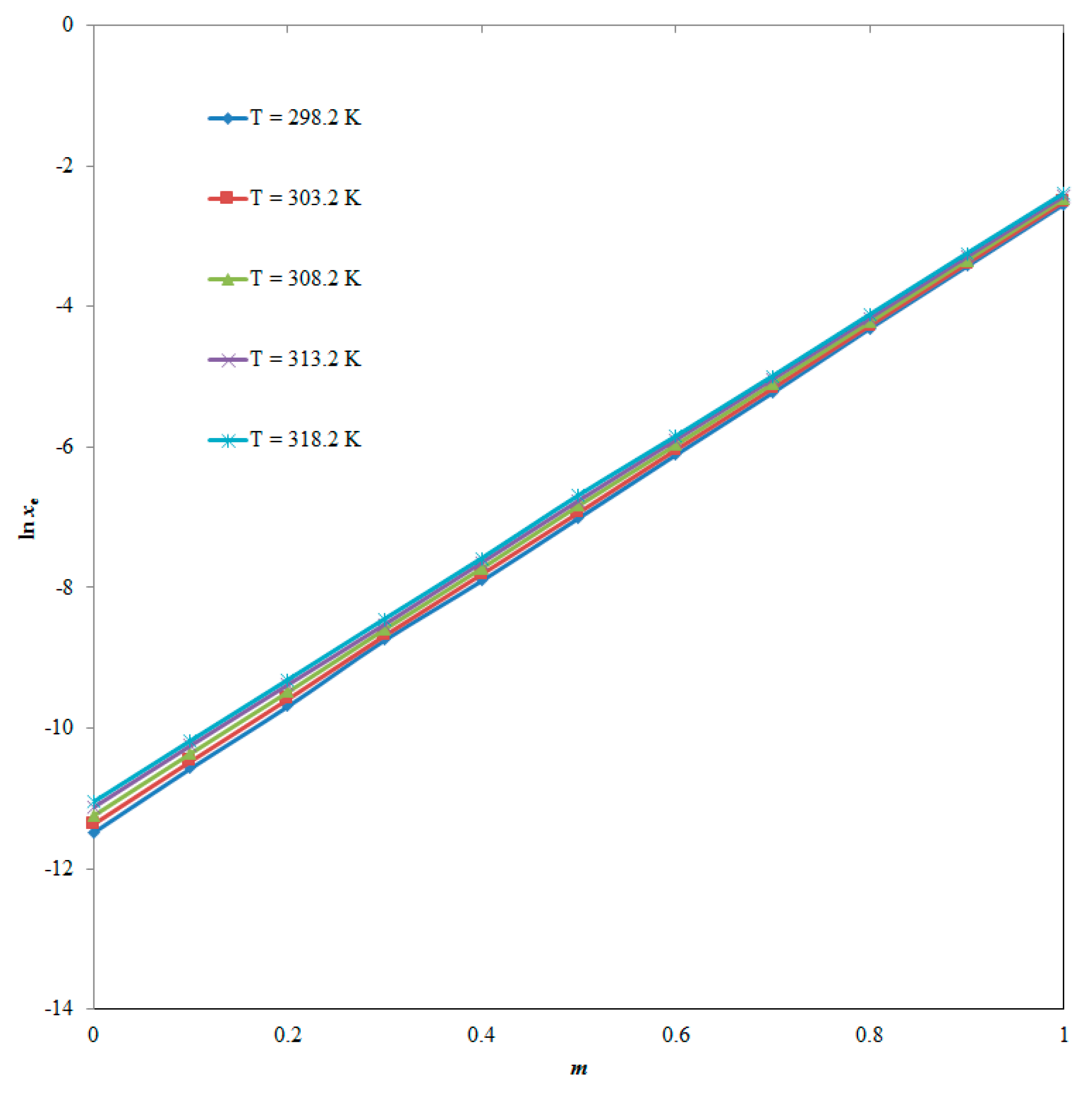
| m | xe | ||||
|---|---|---|---|---|---|
| T = 298.2 K | T = 303.2 K | T = 308.2 K | T = 313.2 K | T = 318.2 K | |
| 0.0 | 1.03 × 10−5 | 1.17 × 10−5 | 1.31 × 10−5 | 1.47 × 10−5 | 1.59 × 10−5 |
| 0.1 | 2.57 × 10−5 | 2.85 × 10−5 | 3.19 × 10−5 | 3.55 × 10−5 | 3.80 × 10−5 |
| 0.2 | 6.20 × 10−5 | 6.88 × 10−5 | 7.61 × 10−5 | 8.40 × 10−5 | 9.01 × 10−5 |
| 0.3 | 1.59 × 10−4 | 1.71 × 10−4 | 1.86 × 10−4 | 1.99 × 10−4 | 2.15 × 10−4 |
| 0.4 | 3.71 × 10−4 | 4.07 × 10−4 | 4.42 × 10−4 | 4.79 × 10−4 | 5.09 × 10−4 |
| 0.5 | 9.06 × 10−4 | 9.80 × 10−4 | 1.08 × 10−3 | 1.16 × 10−3 | 1.25 × 10−3 |
| 0.6 | 2.23 × 10−3 | 2.39 × 10−3 | 2.56 × 10−3 | 2.74 × 10−3 | 2.88 × 10−3 |
| 0.7 | 5.40 × 10−3 | 5.74 × 10−3 | 6.10 × 10−3 | 6.51 × 10−3 | 6.80 × 10−3 |
| 0.8 | 1.35 × 10−2 | 1.40 × 10−2 | 1.47 × 10−2 | 1.55 × 10−2 | 1.63 × 10−2 |
| 0.9 | 3.26 × 10−2 | 3.37 × 10−2 | 3.53 × 10−2 | 3.70 × 10−2 | 3.87 × 10−2 |
| 1.0 | 7.88 × 10−2 | 8.12 × 10−2 | 8.44 × 10−2 | 8.79 × 10−2 | 9.10 × 10−2 |
| xidl | 5.13 × 10−2 | 6.02 × 10−2 | 7.06 × 10−2 | 8.26 × 10−2 | 9.63 × 10−2 |
| m | γi | ||||
|---|---|---|---|---|---|
| T = 298.2 K | T = 303.2 K | T = 308.2 K | T = 313.2 K | T = 318.2 K | |
| 0.0 | 4980.00 | 5150.00 | 5380.00 | 5620.00 | 6050.00 |
| 0.1 | 1995.20 | 2108.92 | 2215.74 | 2339.59 | 2533.27 |
| 0.2 | 827.00 | 875.00 | 927.00 | 984.00 | 1070.00 |
| 0.3 | 322.00 | 353.00 | 380.00 | 416.00 | 448.00 |
| 0.4 | 138.00 | 148.00 | 160.00 | 173.00 | 189.00 |
| 0.5 | 56.60 | 61.40 | 65.50 | 71.40 | 77.30 |
| 0.6 | 23.00 | 25.20 | 27.60 | 30.20 | 33.40 |
| 0.7 | 5.40 | 5.74 | 6.10 | 6.51 | 6.80 |
| 0.8 | 3.81 | 4.31 | 4.82 | 5.33 | 5.92 |
| 0.9 | 1.57 | 1.79 | 2.00 | 2.23 | 2.49 |
| 1.0 | 0.65 | 0.74 | 0.83 | 0.94 | 1.06 |
| m | a | b | R2 | RMSD (%) | Overall RMSD (%) |
|---|---|---|---|---|---|
| 0.0 | −4.45 | −2093.60 | 0.9960 | 1.11 | |
| 0.1 | −4.20 | −1897.30 | 0.9963 | 0.91 | |
| 0.2 | −3.65 | −1799.00 | 0.9973 | 0.70 | |
| 0.3 | −3.98 | −1421.50 | 0.9982 | 0.33 | |
| 0.4 | −2.83 | −1509.30 | 0.9968 | 0.62 | |
| 0.5 | −1.90 | −1520.50 | 0.9981 | 0.77 | 0.65 |
| 0.6 | −1.95 | −1238.70 | 0.9985 | 0.42 | |
| 0.7 | −1.49 | −1112.00 | 0.9973 | 0.42 | |
| 0.8 | −1.24 | −916.75 | 0.9935 | 0.56 | |
| 0.9 | −0.64 | −829.34 | 0.9932 | 1.01 | |
| 1.0 | −0.21 | −696.21 | 0.9960 | 0.31 |
| m | A | B | C | R2 | RMSD (%) | Overall RMSD (%) |
|---|---|---|---|---|---|---|
| 0.0 | 331.19 | −17,505.00 | −49.84 | 0.9995 | 0.78 | |
| 0.1 | 224.66 | −12,407.50 | −33.98 | 0.9982 | 0.73 | |
| 0.2 | 217.93 | −11,974.50 | −32.90 | 0.9993 | 0.58 | |
| 0.3 | −105.09 | 3214.87 | 15.01 | 0.9988 | 0.57 | |
| 0.4 | 228.14 | −12,114.70 | −34.29 | 0.9999 | 0.60 | |
| 0.5 | 45.42 | −3697.43 | −7.02 | 0.9981 | 0.45 | 0.54 |
| 0.6 | 87.58 | −5351.77 | −13.29 | 0.9991 | 0.34 | |
| 0.7 | 84.34 | −5054.78 | −12.74 | 0.9981 | 0.44 | |
| 0.8 | −157.86 | 6268.79 | 23.26 | 0.9978 | 0.61 | |
| 0.9 | −157.97 | 6388.73 | 23.36 | 0.9985 | 0.45 | |
| 1.0 | −84.70 | 3179.61 | 12.54 | 0.9982 | 0.47 |
| m | Log xYal | RMSD (%) | Overall RMSD (%) | ||||
|---|---|---|---|---|---|---|---|
| T = 298.2 K | T = 303.2 K | T = 308.2 K | T = 313.2 K | T = 318.2 K | |||
| 0.1 | −4.59 | −4.54 | −4.50 | −4.45 | −4.42 | 1.21 | |
| 0.2 | −4.21 | −4.16 | −4.12 | −4.07 | −4.04 | 0.46 | |
| 0.3 | −3.82 | −3.77 | −3.74 | −3.69 | −3.67 | 2.81 | |
| 0.4 | −3.43 | −3.39 | −3.35 | −3.32 | −2.29 | 0.91 | |
| 0.5 | −3.04 | −3.01 | −2.97 | −2.94 | −2.91 | 2.27 | 1.24 |
| 0.6 | −2.65 | −2.62 | −2.59 | −2.56 | −2.54 | 1.11 | |
| 0.7 | −2.26 | −2.24 | −2.21 | −2.18 | −2.16 | 0.38 | |
| 0.8 | −1.88 | −1.85 | −1.83 | −1.81 | −1.79 | 1.31 | |
| 0.9 | −1.49 | −1.47 | −1.45 | −1.43 | −1.41 | 0.78 | |
| System | Jouyban–Acree | Jouyban–Acree–Van’t Hoff |
|---|---|---|
| A1–0.21 | ||
| PEG-400 + water | Ji–14.43 | B1–696.21 |
| A2–4.45 | ||
| B2–2093.60 | ||
| RMSD (%) | 0.42 | Ji–16.42 |
| 0.54 |
| Material | Molecular Formula | Molar Mass (g mol−1) | CAS Registry No. | Purification Method | Mass Fraction Purity | Analysis Method | Analysis Method | Source |
|---|---|---|---|---|---|---|---|---|
| PPN | C17H19NO3 | 285.34 | 94-62-2 | None | >0.99 | HPLC | HPLC | Sigma Aldrich |
| THP | C6H14O3 | 134.17 | 111-90-0 | None | >0.99 | GC | GC | Gattefosse |
| Water | H2O | 18.07 | 7732-18-5 | None | - | - | - | Milli-Q |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shakeel, F.; Haq, N.; Alshehri, S. Solubility Data of the Bioactive Compound Piperine in (Transcutol + Water) Mixtures: Computational Modeling, Hansen Solubility Parameters and Mixing Thermodynamic Parameters. Molecules 2020, 25, 2743. https://doi.org/10.3390/molecules25122743
Shakeel F, Haq N, Alshehri S. Solubility Data of the Bioactive Compound Piperine in (Transcutol + Water) Mixtures: Computational Modeling, Hansen Solubility Parameters and Mixing Thermodynamic Parameters. Molecules. 2020; 25(12):2743. https://doi.org/10.3390/molecules25122743
Chicago/Turabian StyleShakeel, Faiyaz, Nazrul Haq, and Sultan Alshehri. 2020. "Solubility Data of the Bioactive Compound Piperine in (Transcutol + Water) Mixtures: Computational Modeling, Hansen Solubility Parameters and Mixing Thermodynamic Parameters" Molecules 25, no. 12: 2743. https://doi.org/10.3390/molecules25122743
APA StyleShakeel, F., Haq, N., & Alshehri, S. (2020). Solubility Data of the Bioactive Compound Piperine in (Transcutol + Water) Mixtures: Computational Modeling, Hansen Solubility Parameters and Mixing Thermodynamic Parameters. Molecules, 25(12), 2743. https://doi.org/10.3390/molecules25122743








