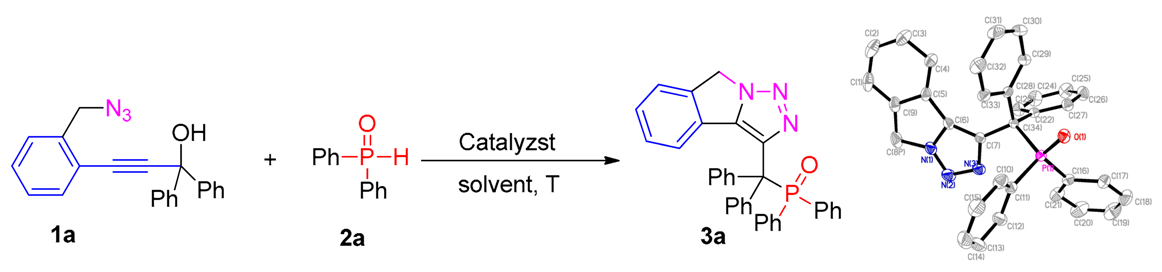
Abstract
An efficient approach for the synthesis of phosphorylated isoindoline fused with triazoles via Zn(OTf)2-catalyzed cascade cyclization of easily prepared ortho–propynol benzyl azides and diarylphosphine oxides is developed. The transformation occurred smoothly in moderate to excellent yields and tolerated various propargylic alcohol substrates.
1. Introduction
Isoindolines are important scaffolds that are widely found in various drugs, bioactive compounds, and natural products [1,2]. On the other hand, fused 1,2,3–triazoles are also interesting skeletons found to exhibit a wide range of biological activities. Figure 1 shows the representative examples of some bioactive heterocyclic compounds fused with triazoles. In particular, isoindolines fused with 1,2,3–triazoles have attracted significant attention due to their distinct synthetic applications and pharmacological characteristics [3,4,5,6,7]. In the past several decades, some conventional approaches for construction of such compounds have been developed [8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28]. These approaches mainly include: (1) the reaction of azide and alkyne functionalities via intramolecular cycloaddition [9]; (2) Metal-catalyzed cyclization of terminal alkynes and azidoaryl halides [10,11,12,13]; and (3) Organocatalyzed the cyclization of organic azides and carbonyl compounds [14,15,16,17,18,19,20,21,22,23,24,25,26,27,28]. Despite these advances, new approaches for the construction of diverse functionalized isoindolines fused with triazoles under mild conditions is still attractive and desirable.
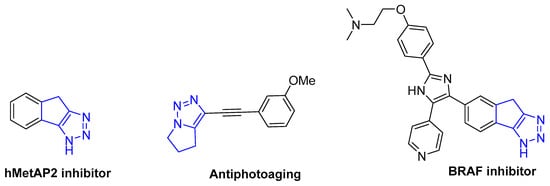
Figure 1.
Representative molecules of bioactive fused 1,2,3–triazoles.
Meanwhile, organophosphorus compounds have been widely applied in material science and organic and pharmaceutical chemistry [29,30,31]. It is well established that the introduction of organophosphorus functionalities into heterocycles could potentially introduce new biological and physical properties into the parent molecule [32,33,34]. If both the isoindoline–triazole and phosphinoyl group can be simultaneously installed in organic frameworks, such compounds could be used to modulate the bioactivity of original pharmaceutical molecule or lead compounds. Therefore, it is important to develop an efficient strategy for the one-pot construction isoindolines fused with triazoles and C–P bond.
In recent years, tandem reactions of propynols with nucleophiles have emerged as powerful tools for the construction of various carbo- and heterocycle compounds [35,36,37,38]. In 2013, the Tanimoto group developed an efficient approach to prepare fully substituted 1H–1,2,3–triazoles through TMSOTf-promoted cyclization of alkynols with organic azides [39,40]. Mechanistically, the reaction takes place by the nucleophilic attack of the organic azides to the allenyl cation intermediate, followed by another nucleophilic attack on intermediate II. In their creative work, a series of nucleophilies, including indoles, azides, alcohols, amines, etc., were successfully added to intermediate II. Notably, phosphine nucleophiles have not been reported to capture intermediate II. In a continuation of our work on propargylic alcohols [41,42,43,44,45,46] and the synthesis of organophosphorus compounds, we herein report our detailed findings (Scheme 1).
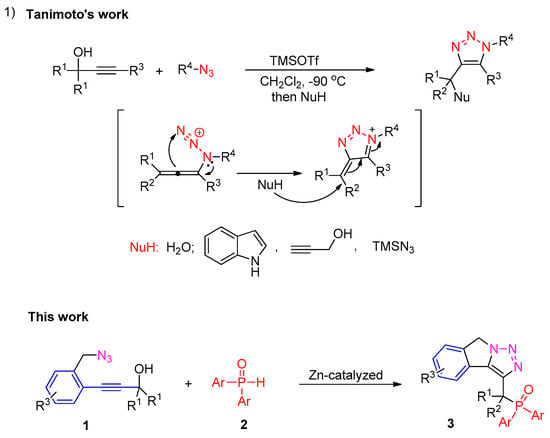
Scheme 1.
Previous work and our strategy.
2. Results and Discussion
To begin, the reaction between 2–propynol benzyl azides 1a and diphenylphosphine oxide 2a was selected as the mock-up reaction to examine the optimal conditions, as shown in Table 1. In an initial experiment, the reaction was performed in DCE at 100 ºC catalyzed by Sc (OTf)3 under open air (entry 1). Gratifyingly, our expected product ((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)diphenylmethyl)diphenylphosphine oxide 3a was isolated in 42% yield after 1.0 h. The 3a was unambiguously confirmed by X-ray structure analysis [47]. On the basis of this result, a subsequent brief screening of some representative Lewis acid, including Zn(OTf)2, Cu(OAc)2, Cu(OTf)2, CuCl2, and AgOTf revealed that Zn(OTf)2 performed most efficiently and could increase the yield of desired product 3a to 53% (entries 2−6). Subsequently, among the reaction solvents examined, it turned out that the reaction in CH3CN gave the best yields (entries 7–10). No better results were obtained when decreasing or increasing the reaction temperature (entries 11–12). Then, the loading of catalyst and diphenylphosphine oxide 2a was also investigated, and the results indicated that 20 mol% of Zn(OTf)2 and 2.5 equiv of 2a gave the better yield for this transformation (entries 13–16). Therefore, we selected the following optimum conditions: 0.1 mmol of 2–propynol benzyl azides 1, 20 mol% of Zn(OTf)2, 2.5 equiv of diphenylphosphine oxide 2a in CH3CN at 100 °C for 1.0 h.

Table 1.
Optimization of the reaction for the synthesis of 3a a.
With optimal reaction conditions for the transformation of 2–propynol benzyl azides to P-containing isoindoline fused with triazoles in hand, we investigated the generality of the cascade reaction, and the corresponding results are summarized in Figure 2. The phosphorylated isoindoline fused with triazoles 3 were formed in moderate to excellent yields under optimal conditions. Both electron-donating (Me, OMe) and electron-withdrawing groups (F, Cl) on two aromatic rings were tolerated well to generate the target compounds in acceptable yields. In general, substrates containing electron-poor substituents gave lower yields than electron-rich ones (3a–3c vs 3d–3e). Furthermore, the steric effect of substituents showed little effect on this transformation; substrate bearing the ortho-position substituents on aryl groups generates a good yield of desired product (3j). Notably, the substrate with polycyclic aryl ring still gave a moderate yield of 49% (3k), which might be due to the ring strain. Moreover, no desired products (3n and 3o) were observed using substrates with dibenzyl and dimethyl instead of aryl groups under the optimal conditions. This might be due to the fact that it is difficult to the formation of allenic intermediate. Subsequently, the substituents (R2) on another benzene ring were also examined under the optimal conditions. Both electron-donating and withdrawing substituents on the para-position of aryl ring were found to be compatible. Additionally, diarylphosphine oxides with representative substituents at the 3- and 4-positions (1p–1q) were tolerated well for this reaction (3p–3q). However, no desired product was obtained when diethyl phosphite 1r was employed under the optimal conditions. This might be due to the low nucleophilicity and thermostability of the diethyl phosphite. The final concern was that the reaction system still worked smoothly with secondary propynols; the corresponding products (3s–3t) were obtained in moderate yield. Compared to tertiary propynols substrates, lower yields were obtained because of the formation of unstable intermediate B.
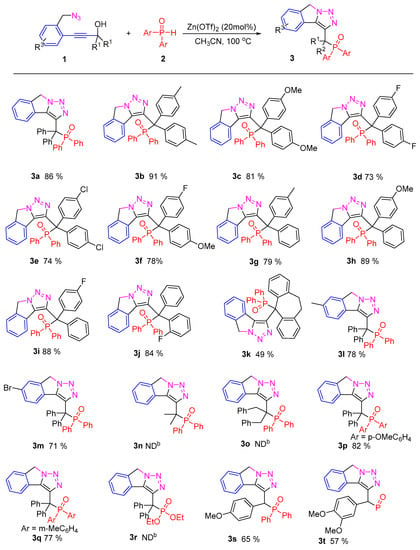
Figure 2.
Transformation of propargylic alcohols 1 to phosphorylated isoindoine fused with trizoles 3. b The corresponding product cannot be detected by TLC.
Furthermore, a gram-scale reaction of ortho–propynol benzyl azides 1a and diphenylphosphine oxide 2a could be conducted smoothly to demonstrate the scalability of this reaction under the standard conditions (Scheme 2). The target compound phosphorylated isoindoline 3a was generated in 75% yield, which may offer potential application in organic synthesis and medicinal chemistry.
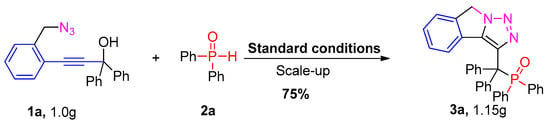
Scheme 2.
Scale-up experiment.
According to our experimental results and reported literatures [39,40,48,49], a plausible reaction mechanism was proposed in Scheme 3. Firstly, the coordination of the hydroxyl and alkynyl group of 2–propynol benzyl azides 1 with Zn(OTf)2 afforded complex A. Intermediate A was then intramolecularly attacked by the azides group to generate allenylaminodiazonium intermediate B. Subsequently, intermediate B undergoes intramolecular cyclization to generate the intermediate C. Finally, the nucleophilic attack on intermediate C by diphenylphosphine oxide 2a followed by the loss of a proton produced the desired product 3.
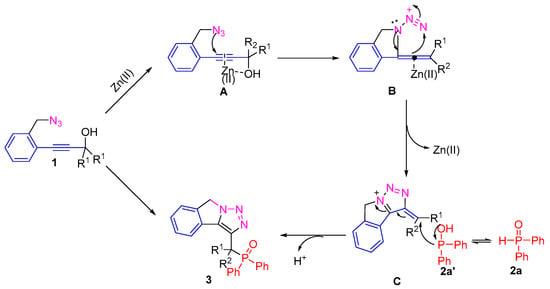
Scheme 3.
Proposed reaction mechanism.
3. Materials and Methods
General Remarks: Column chromatography was carried out on silica gel. 1H–NMR spectra were recorded on 400 MHz in CDCl3 and 13C–NMR spectra were recorded on 100 MHz in CDCl3. Chemical shifts (ppm) were recorded with tetramethylsilane (TMS) as the internal reference standard. Multiplicities are given as: s (singlet), d (doublet), t (triplet), dd (doublet of doublets), q (quartet), or m (multiplet). High-resolution mass spectrometry (HRMS) was performed on a TOF/Q–TOF mass spectrometer. Melting points were determined on a microscopic apparatus and were uncorrected. Copies of the 1H–NMR and 13C–NMR spectra are provided in the Supplementary Materials. Commercially available reagents were used without further purification. All solvents were dried under standard method.
General Procedure for the Construction of Phosphorylated Isoindoline Fused with Triazoles 3: To a seal tube was added Zn(OTf)2 (0.04 mmol), ortho–propynol benzyl azides (1) (0.2 mmol), diphenylphosphine oxide 2a (0.5 mmol), in CH3CN at 100°C under air atmosphere. After 1.0 h, as monitored by TLC, the reaction mixture was concentrated in vacuum and purified by column chromatography to generate 3.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)diphenylmethyl)diphenylphosphine oxide (3a): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3a (86%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); white solid; mp: 172–174 °C. 1H–NMR (400 MHz, CDCl3): δ ppm 5.30–5.32 (m, 3 H), 6.89–6.90 (m, 1 H), 7.15–7.28 (m, 11 H), 7.31–7.36 (m, 3 H), 7.42–7.44 (m, 4 H), 7.80–7.84 (m, 4 H). 13C-NMR (100 MHz, CDCl3): δ ppm 50.8, 59.5, 60.1, 123.2, 127.3, 127.4, 127.5, 128.0, 128.1, 128.2, 128.3, 130.6, 130.6, 131.0, 131.0, 132.6, 133.5, 134.3, 134.3, 139.7, 139.7, 140.5, 141.1, 143.1, 143.2. 31P NMR (162 MHz, CDCl3): δ ppm 40.31. HRMS (ESI, m/z): calcd for C34H26N3OP: M + H = 524.1886; found: 524.1883.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3-yl)di–p–tolylmethyl)diphenylphosphine oxide (3b): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3b (91%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 2.21 (s, 6 H), 5.19 (s, 2 H), 5.30 (d, J = 8.0 Hz, 1 H), 6.80 (t, J = 7.6 Hz, 1 H), 6.91–6.93 (m, 4 H), 7.06–7.09 (m, 1 H), 7.12–7.17 (m, 4 H), 7.19–7.26 (m, 7 H), 7.69–7.74 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 20.9, 50.7, 58.7, 59.3, 123.0, 123.3, 127.1, 127.2, 127.8, 128.0, 128.1, 128.8, 128.8, 130.3, 130.3, 130.8, 132.7, 133.6, 134.1, 134.2, 136.6, 136.7, 136.9, 137.0, 140.4, 141.3, 141.3, 143.0, 143.0. 31P NMR (162 MHz, CDCl3): δ ppm 40.13. HRMS (ESI, m/z): calcd for C36H30N3OP: M + H = 552.2199; found: 552.2197.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)bis(4–methoxyphenyl)methyl)diphenylphosphine oxide (3c): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3c (81%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); white solid; mp: 133–135 °C. 1H–NMR (400 MHz, CDCl3): δ ppm 3.77 (s, 6 H), 5.31 (s, 2 H), 5.56 (d, J = 8.0 Hz, 1 H), 6.74 (d, J = 8.8 Hz, 4 H), 6.95 (t, J = 7.6 Hz, 1 H), 7.17–7.26 (m, 5 H), 7.30–7.37 (m, 7 H), 7.73–7.78 (m, 4 H). 13C-NMR (100 MHz, CDCl3): δ ppm 50.8, 55.2, 57.9, 58.5, 113.4, 123.1, 123.3, 127.2, 127.3, 127.9, 128.1, 128.4, 130.9, 131.0, 131.7, 131.7, 131.8, 131.8, 132.6, 133.6, 134.1, 134.2, 140.5, 141.4, 143.0, 143.0, 158.7. 31P NMR (162 MHz, CDCl3): δ ppm 39.72. HRMS (ESI, m/z): calcd for C36H30N3O3P: M + H = 584.2098; found: 584.2096.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3-yl)bis(4–fluorophenyl)methyl)diphenylphosphine oxide (3d): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3d (73%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow solid; mp: 105–107 °C. 1H–NMR (400 MHz, CDCl3): δ ppm 5.32 (s, 2 H), 5.45 (d, J = 7.6 Hz, 1 H), 6.89–6.99 (m, 5 H), 7.20–7.24 (m, 3 H), 7.26–7.28 (m, 2 H), 7.34–7.40 (m, 7 H), 7.79–7.84 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 50.9, 58.2, 58.8, 115.0, 115.2, 122.7, 123.4, 127.4, 127.5, 127.7, 128.3, 128.5, 131.3, 131.3, 131.9, 132.1, 132.1, 132.1, 132.2, 132.8, 134.1, 134.2, 135.3, 140.5, 140.7, 143.0, 143.0, 160.7, 163.2. 31P NMR (162 MHz, CDCl3): δ ppm 35.74. HRMS (ESI, m/z): calcd for C34H24F2N3OP: M + H = 560.1698; found: 560.1698.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)bis(4–chlorophenyl)methyl)diphenylphosphine oxide (3e): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3e (74%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 5.33 (s, 2 H), 5.45 (d, J = 8.0 Hz, 1 H), 6.99 (t, J = 8.0 Hz, 1 H), 7.18–7.22 (m, 4 H), 7.24–7.29 (m, 5 H), 7.32–7.40 (m, 7 H), 7.82–7.87 (m, 4 H). 13C-NMR (100 MHz, CDCl3): δ ppm 50.9, 58.6, 59.2, 122.8, 123.4, 127.5, 127.6, 128.4, 128.4, 128.6, 131.4, 131.4, 131.7, 131.8, 132.7, 133.7, 133.8, 134.2, 138.0, 138.0, 140.2, 140.6, 143.0, 143.1. 31P NMR (162 MHz, CDCl3): δ ppm 40.45. HRMS (ESI, m/z): calcd for C34H24Cl2N3OP: M + H = 592.1107; found: 592.1105.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)(4–fluorophenyl)(4 methoxyphenyl)methyl)diphenylphosphine oxide (3f): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3f (78%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); red liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 3.77 (s, 3 H), 5.31 (s, 2 H), 5.50 (d, J = 7.6 Hz, 1 H), 6.75 (d, J = 8.8 Hz, 2 H), 6.88–6.89 (m, 3 H), 7.19–7.27 (m, 4 H), 7.30 (s, 1 H), 7.31–7.41 (m, 7 H), 7.76–7.82 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 50.8, 55.2, 58.0, 58.6, 113.6, 114.8, 115.0, 123.0, 123.3, 127.3, 127.4, 127.8, 128.1, 128.4, 131.1, 131.5, 131.5, 131.6, 132.2, 132.2, 132.2, 132.3, 133.2, 133.2, 134.1, 134.2, 135.7, 140.5, 141.0, 143.0, 143.0, 158.8, 160.7, 163.2. 31P NMR (162 MHz, CDCl3): δ ppm 35.36. HRMS (ESI, m/z): calcd for C35H27FN3O2P: M + H = 572.1898; found: 572.1894.
((8H–[1,2,3]triazolo[5,1–a]isoindol-3–yl)(phenyl)(p–tolyl)methyl)diphenylphosphine oxide (3g): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3g (79%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 2.30 (s, 3 H), 5.29–5.36 (m, 3 H), 6.89 (t, J = 8.0 Hz, 1 H), 7.01–7.03 (m, 2 H), 7.16–7.26 (m, 8 H), 7.28–7.36 (m, 5 H), 7.40–7.42 (m, 2 H), 7.78–7.83 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 20.9, 50.8, 59.1, 59.7, 123.1, 123.2, 127.2, 127.2, 127.3, 127.3, 127.9, 128.0, 128.1, 128.2, 128.9, 130.3, 130.4, 130.5, 130.5, 130.9, 130.9, 132.5, 132.6, 133.5, 133.6, 134.2, 134.3, 136.5, 137.1, 139.8, 140.5, 141.2, 143.0, 143.1. 31P NMR (162 MHz, CDCl3): δ ppm 40.26. HRMS (ESI, m/z): calcd for C35H28N3OP: M + H = 538.2043; found: 538.2041.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)(4–methoxyphenyl)(phenyl)methyl)diphenylphosphine oxide (3h): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3h (89%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 3.68 (s, 3 H), 5.22 (s, 2 H), 5.34 (d, J = 7.6 Hz, 1 H), 6.67 (d, J = 9.2 Hz, 1 H), 6.83 (t, J = 8.0 Hz, 1 H), 7.10–7.18 (m, 9 H), 7.24–7.28 (m, 4 H), 7.32 (d, J = 8.0 Hz, 2 H), 7.68–7.74 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 50.8, 55.2, 58.7, 59.3, 113.4, 123.1, 123.2, 127.2, 127.2, 127.3, 127.3, 127.9, 128.0, 128.1, 128.3, 130.5, 130.5, 130.9, 131.4, 131.5, 131.7, 131.7, 132.5, 132.6, 133.4, 133.5, 134.1, 134.2, 134.2, 134.3, 139.9, 140.0, 140.5, 141.2, 143.0, 143.1, 158.7, 158.8. 31P NMR (162 MHz, CDCl3): δ ppm 40.17. HRMS (ESI, m/z): calcd for C35H28N3O2P: M + H = 554.1992; found: 554.1988.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)(4–fluorophenyl)(phenyl)methyl)diphenylphosphine oxide (3i): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3i (88%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 5.23 (s, 2 H), 5.29 (d, J = 7.6 Hz, 1 H), 6.80–6.86 (m, 3 H), 7.12–7.18 (m, 8 H), 7.24–7.29 (m, 2 H), 7.30–7.35(m, 5 H), 7.70–7.77 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 50.9, 58.9, 59.5, 114.9, 115.1, 123.0, 123.3, 127.3, 127.4, 127.4, 127.5, 127.6, 127.9, 128.2, 128.4, 128.4, 128.7, 130.4, 130.4, 131.2, 132.2, 132.3, 132.3, 132.4, 132.4, 133.1, 133.2, 134.2, 134.3, 134.3, 135.4, 139.5, 139.7, 140.6, 140.9, 143.1, 143.2, 160.8, 163.3. 31P NMR (162 MHz, CDCl3): δ ppm 40.61. HRMS (ESI, m/z): calcd for C34H25FN3OP: M + H = 542.1792; found: 542.1791.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)(2–fluorophenyl)(phenyl)methyl)diphenylphosphine oxide (3j): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3j (84%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 5.20 (s, 2 H), 5.66 (d, J = 8.0 Hz, 1 H), 6.71–6.76 (m, 1 H), 6.87 (t, J = 8.0 Hz, 1 H), 7.09–7.19 (m, 8 H), 7.19–7.29 (m, 5 H), 7.45–7.53 (m, 6 H), 8.24 (t, J = 8.0 Hz, 1 H). 13C–NMR (100 MHz, CDCl3): δ ppm 50.6, 56.6, 57.2, 117.0, 117.2, 123.0, 123.1, 123.9, 123.9, 127.1, 127.3, 127.5, 127.6, 127.7, 127.7, 127.7, 128.2, 128.2, 129.3, 129.4, 129.7, 129.8, 131.0, 131.1, 131.1, 131.2, 131.2, 131.3, 131.6, 131.8, 132.1, 132.2, 132.6, 132.7, 133.4, 133.5, 133.7, 133.8, 137.7, 138.7, 140.4, 142.8, 160.2, 160.3, 162.7, 162.8. 31P–NMR (162 MHz, CDCl3): δ ppm 38.49. HRMS (ESI, m/z): calcd for C34H25FN3OP: M + H = 542.1792; found: 542.1791.
(5–(8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)–10,11–dihydro–5H–dibenzo[a,d][7]annulen–5–yl)diphenylphosphine oxide (3k): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3k (49%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow solid; mp: 276–278 °C. 1H–NMR (400 MHz, CDCl3): δ ppm 2.98–3.04 (m, 2 H), 4.01–4.07 (m, 2 H), 5.19 (s, 2 H), 6.02 (d, J = 7.6 Hz, 1 H), 6.33–6.35 (m, 2 H), 6.41–6.44 (m, 2 H), 6.87–6.90 (m, 3 H), 7.03–7.07 (m, 1 H), 7.08–7.10 (m, 2 H), 7.17–7.23 (m, 6 H), 7.25–7.29 (m, 1 H), 8.10–8.14 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 37.1, 51.1, 121.8, 123.3, 124.9, 127.1, 127.2, 127.3, 127.7, 127.8, 128.5, 130.5, 130.9, 131.9, 132.8, 134.2, 134.4, 134.5, 136.2, 140.3, 145.2, 146.5. 31P–NMR (162 MHz, CDCl3): δ ppm 41.14. HRMS (ESI, m/z): calcd for C36H28N3OP: M + H = 550.2043; found: 550.2041.
((6–methyl–8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)diphenylmethyl)diphenylphosphine oxide (3l): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3l (78%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow solid; mp: 252–254 °C. 1H–NMR (400 MHz, CDCl3): δ ppm 2.19 (s, 3 H), 5.08 (d, J = 8.0 Hz, 1 H), 5.17 (s, 2 H), 6.61 (d, J = 8.0 Hz, 1 H), 7.08–7.18 (m, 11 H), 7.23–7.27 (m, 2 H), 7.33–7.35 (m, 4 H), 7.73–7.78 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 21.4, 50.7, 59.4, 60.0, 122.7, 123.8, 125.3, 127.2, 127.3, 127.4, 128.1, 129.0, 130.5, 130.5, 130.9, 130.9, 132.5, 133.5, 134.2, 134.3, 138.3, 139.6, 139.6, 140.5, 140.8, 143.1, 143.2. 31P–NMR (162 MHz, CDCl3): δ ppm 40.64. HRMS (ESI, m/z): calcd for C35H28N3OP: M + H = 538.2043; found: 538.2041.
((6–bromo–8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)diphenylmethyl)diphenylphosphine oxide (3m): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3q (71%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); red solid; mp: 274–276 °C. 1H–NMR (400 MHz, CDCl3): δ ppm 5.13 (d, J = 8.4 Hz, 1 H), 5.22 (s, 2 H), 6.96 (dd, J = 1.6, 8.4 Hz, 1 H), 7.12–7.19 (m, 10 H), 7.25–7.28 (m, 2 H), 7.29–7.33 (m, 4 H), 7.42 (d, J = 0.8 Hz, 1 H), 7.64–7.69 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 50.4, 59.4, 60.0, 122.1, 124.5, 126.5, 127.0, 127.3, 127.4, 127.5, 128.3, 130.5, 130.6, 131.1, 131.1, 131.6, 132.2, 133.1, 134.0, 134.1, 139.5, 139.5, 141.3, 142.2, 142.4, 142.5. 31P–NMR (162 MHz, CDCl3): δ ppm 39.94. HRMS (ESI, m/z): calcd for C34H25BrN3OP: M + H = 602.0991; found: 602.0990.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)diphenylmethyl)bis(4–methoxyphenyl)phosphine oxide (3p): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3p (82%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow solid; mp: 118–120 °C. 1H–NMR (400 MHz, CDCl3): δ ppm 3.74 (s, 6 H), 5.30–5.34 (m, 3 H), 6.73–6.79 (m, 4 H), 6.88 (t, J = 8.0 Hz, 1 H), 7.14–7.25 (m, 7 H), 7.34 (d, J = 7.6 Hz, 1 H), 7.41–7.43 (m, 4 H), 7.66–7.71 (m, 4 H). 13C–NMR (100 MHz, CDCl3): δ ppm 50.7, 55.0, 59.3, 60.0, 112.7, 112.8, 123.1, 123.2, 123.8, 124.8, 127.3, 127.9, 128.0, 128.1, 128.2, 130.5, 130.6, 135.9, 136.0, 139.9, 139.9, 140.4, 141.3, 143.1, 143.1. 31P–NMR (162 MHz, CDCl3): δ ppm 40.00. HRMS (ESI, m/z): calcd for C36H30N3O3P: M+H = 584.2098; found: 584.2096.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)diphenylmethyl)di–m–tolylphosphine oxide (3q): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3q (77%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 2.13 (s, 6 H), 5.22 (s, 2 H), 5.36 (d, J = 8.0 Hz, 1 H), 6.82 (t, J = 7.6 Hz, 1 H), 7.01–7.10 (m, 5 H), 7.13–7.17 (m, 6 H), 7.26–7.28 (m, 1 H), 7.32 – 7.43 (m, 8 H). 13C–NMR (100 MHz, CDCl3): δ ppm 21.3, 50.8, 59.5, 60.1, 123.1, 123.4, 127.1, 127.2, 127.4, 127.9, 128.1, 128.2, 128.3, 130.9, 130.9, 131.2, 131.2, 131.9, 131.9, 132.1, 133.0, 134.5, 134.6, 136.9, 137.0, 139.8, 140.5, 141.0, 143.2, 143.2. 31P–NMR (162 MHz, CDCl3): δ ppm 39.96. HRMS (ESI, m/z): calcd for C36H30N3OP: M + H = 552.2199; found: 552.2197.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)(4–methoxyphenyl)methyl)diphenylphosphine oxide (3s): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3s (65%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow liquid. 1H–NMR (400 MHz, CDCl3): δ ppm 3.62 (s, 3 H), 5.10 (s, 2 H), 5.45 (d, J = 12.0 Hz, 1 H), 6.62 (d, J = 8.8 Hz, 2 H), 7.22–7.35 (m, 10 H), 7.42–7.46(m, 1 H), 7.59–7.64 (m, 2 H), 7.72–7.77 (m, 2 H), 8.61 (d, J = 8.0 Hz, 1 H). 13C–NMR (100 MHz, CDCl3): δ ppm 45.7, 46.3, 50.8, 55.2, 113.9, 123.4, 124.3, 126.9, 126.9, 128.2, 128.2, 128.3, 128.5, 129.1, 130.8, 130.9, 131.1, 131.2, 131.3, 131.4, 131.5, 131.6, 131.6, 132.0, 132.1, 133.0, 135.0, 135.1, 140.5, 140.9, 140.9, 158.7. 31P–NMR (162 MHz, CDCl3): δ ppm 30.54. HRMS (ESI, m/z): calcd for C29H24N3O2P: M + H = 478.1679; found: 478.1673.
((8H–[1,2,3]triazolo[5,1–a]isoindol–3–yl)(3,4–dimethoxyphenyl)methyl)diphenylphosphine oxide (3t): The title compound was prepared according to the general procedure and purified by column chromatography (silica gel, petroleum ether/ethyl acetate) to give a product 3t (57%); Rf = 0.47 (petroleum ether/ethyl acetate 50:20); yellow solid; mp: 225–227 °C. 1H–NMR (400 MHz, CDCl3): δ ppm 3.52 (s, 3 H), 3.69 (s, 3 H), 5.12 (s, 2 H), 5.45 (d, J = 11.6 Hz, 1 H), 6.58 (d, J = 8.4 Hz, 1 H), 6.87–6.89 (m, 2 H), 7.24–7.38 (m, 8 H), 7.45 (t, J = 7.2 Hz, 1 H), 7.60–7.64 (m, 2 H), 7.75–7.80 (m, 2 H), 8.72 (d, J = 8.0 Hz, 1 H). 13C–NMR (100 MHz, CDCl3): δ ppm 46.0, 46.7, 50.8, 55.6, 55.7, 110.8, 112.8, 112.8, 122.0, 122.0, 123.4, 124.3, 127.1, 127.2, 128.1, 128.3, 128.3, 128.4, 128.4, 128.9, 130.9, 131.1, 131.2, 131.3, 131.4, 131.5, 131.6, 131.6, 131.6, 131.9, 132.0, 133.0, 134.8, 134.8, 140.5, 140.8, 140.9, 148.0, 148.5. 31P–NMR (162 MHz, CDCl3): δ ppm 30.51. HRMS (ESI, m/z): calcd for C30H26N3O3P: M + H = 508.1785; found: 508.1783.
4. Conclusions
We have successfully described a general and novel Zn(OTf)2-catalyzed cascade cyclization of ortho–propynol benzyl azides and diphenylphosphine oxides. In this transformation, a series of alkynols substrates with various functional groups could be tolerated to form the corresponding phosphorylated isoindolines fused with triazoles in moderate to excellent yields. This reaction likely proceeds via the formation of allenylaminodiazonium intermediate followed by cyclization and nuclophilic attack of diphenylphosphine oxide. Moreover, this reaction can be performed in gram-scale with good yield, which could lead to potential application in organic synthesis. Further studies into the use of this novel strategy for the construction of other functionalized heterocyclics fused with triazoles are ongoing in our group.
Supplementary Materials
The following are available online at https://www.mdpi.com/1420-3049/24/19/3526/s1.
Author Contributions
Conceptualization, X.-R.S. and Q.X.; data curation, J.B.; formal analysis, T.Y.; writing—original draft preparation, T.Y.; writing—review and editing, R.Y. and H.D.
Funding
This work was funded by the National Science Foundation of China (No. 21676131 and No. 21462019), the Science Foundation of Jiangxi Province (20181BAB203005 and 20143ACB20012), the Education Department of Jiangxi Province (GJJ180616), Jiangxi Science & Technology Normal University (2017QNBJRC004).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Berger, D.; Citarella, R.; Dutia, M.; Greenberger, L.; Hallett, W.; Paul, R.; Powell, D. Novel Multidrug Resistance Reversal Agents. J. Med. Chem. 1999, 42, 2145–2161. [Google Scholar] [CrossRef]
- Goethem, S.V.; Matheeussen, V.; Joossens, J.; Lambeir, A.-M.; Chen, X.; Meester, I.D.; Haemers, A.; Augustyns, K.; der Veken, P.V. Structure-Activity Relationship Studies on Isoindoline Inhibitors of Dipeptidyl Peptidases 8 and 9 (DPP8, DPP9): Is DPP8-Selectivity an Attainable Goal? J. Med. Chem. 2011, 54, 5737–5746. [Google Scholar] [CrossRef] [PubMed]
- Das Adhikary, N.; Chattopadhyay, P. Design and Synthesis of 1,2,3-Triazole-Fused Chiral Medium-Ring Benzo-Heterocycles, Scaffolds Mimicking Benzolactams. J. Org. Chem. 2012, 77, 5399–5405. [Google Scholar] [CrossRef] [PubMed]
- Kallander, L.S.; Lu, Q.; Chen, W.; Tomaszek, T.; Yang, G.; Tew, D.; Meek, T.D.; Hofmann, G.A.; Schulz-Pritchard, C.K.; Smith, W.W.; et al. 4-Aryl-1,2,3-triazole: A Novel Template for a Reversible Methionine Aminopeptidase 2 Inhibitor, Optimized To Inhibit Angiogenesis in Vivo. J. Med. Chem. 2005, 48, 5644–5647. [Google Scholar] [CrossRef]
- Greenblatt, D.J.; Harmatz, J.S.; Shapiro, L.; Engelhardt, N.; Gouthro, T.A.; Shader, R.I. Sensitivity to Triazolam in the Elderly. N. Engl. J. Med. 1991, 324, 1691–1698. [Google Scholar] [CrossRef]
- Tatsuta, K.; Ikeda, Y.; Miura, S. Synthesis and Glycosidase Inhibitory Activities of Nagstatin Triazole Analogs. J. Antibiot. 1996, 49, 836. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hseih, H.-Y.; Lee, W.-C.; Chandru, G.C.; Hu, W.-P.; Liang, J.-J.; Tsai, T.-R.; Chou, Y.-W.; Kuo, K.-K.; Chen, C.-Y.; Wang, J.J.; et al. Discovery, Synthetic Methodology, and Biological Evaluation for Antiphotoaging Activity of Bicyclic[1,2,3]triazoles: In Vitro and in Vivo Studies. J. Med. Chem. 2013, 56, 5422–5435. [Google Scholar] [CrossRef]
- Shafran, E.A.; Bakulev, V.A.; Rozin, Y.A.; Shafran, Y.M. Condensed 1,2,3-triazoles. Chem. Heterocycl. Compd. 2008, 44, 1040–1069. [Google Scholar] [CrossRef]
- Majumdar, K.C.; Ray, K. Synthesis of 1,2,3-Triazole-Fused Heterocycles via Intramolecular Azide-Alkyne Cycloaddition Reactions. Synthesis 2011, 3767–3783. [Google Scholar] [CrossRef]
- Choudhury, C.; Mandal, S.B.; Achari, B. Palladium-copper catalysed heteroannulation of acetylenic compounds: An expeditious synthesis of isoindoline fused with triazoles. Tetrahedron Lett. 2005, 46, 8531–8534. [Google Scholar] [CrossRef]
- Fiandanese, V.; Marchese, G.; Punzi, A.; Iannone, F.; Rafaschieri, G.C. An easy synthetic approach to 1,2,3-triazole-fused heterocycles. Tetrahedron 2010, 66, 8846–8853. [Google Scholar] [CrossRef]
- Fiandanese, V.; Marino, I.; Punzi, A. An easy access to 4-(1,2,3-triazolylalkyl)-1,2,3-triazole-fused dihydroisoquinolines and dihydroisoindoles. Tetrahedron 2012, 68, 10310–10317. [Google Scholar] [CrossRef]
- Brahma, K.; Achari, B.; Choudhury, C. Facile Synthesis of [1,2,3]-Triazole-Fused Isoindolines, Tetrahydroisoquinolines, Benzoazepines and Benzoazocines by Palladium-Copper Catalysed Heterocyclisation. Synthesis 2013, 45, 545–555. [Google Scholar]
- Ramachary, D.B.; Ramakumar, K.; Narayana, V.V. Amino Acid-Catalyzed Cascade [3+2]-Cycloaddition/Hydrolysis Reactions Based on the Push–Pull Dienamine Platform: Synthesis of Highly Functionalized NH-1,2,3-Triazoles. Chem. Eur. J. 2008, 14, 9143–9147. [Google Scholar] [CrossRef] [PubMed]
- Ramachary, D.B.; Shashank, A.B. Organocatalytic Triazole Formation, Followed by Oxidative Aromatization: Regioselective Metal-Free Synthesis of Benzotriazoles. Chem. Eur. J. 2013, 19, 13175–13181. [Google Scholar] [CrossRef] [PubMed]
- Belkheira, M.; Abed, D.E.; Pons, J.-M.; Bressy, C. Organocatalytic Synthesis of 1,2,3-Triazoles from Unactivated Ketones and Arylazides. Chem. Eur. J. 2011, 17, 12917–12921. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Peng, S.Y.; Danence, L.J.T.; Gao, Y.; Wang, J. Amine-Catalyzed [3+2] Huisgen Cycloaddition Strategy for the Efficient Assembly of Highly Substituted 1,2,3-Triazoles. Chem. Eur. J. 2012, 18, 6088–6093. [Google Scholar] [CrossRef]
- Seus, N.; Goncalves, L.C.; Deobald, A.M.; Savegnago, L.; Alves, D.; Paixao, M.W. Synthesis of arylselanyl-1H-1,2,3-triazole-4-carboxylates by organocatalytic cycloaddition of azidophenyl arylselenides with β-keto-esters. Tetrahedron 2012, 68, 10456–10463. [Google Scholar] [CrossRef]
- Li, W.; Jia, Q.; Du, Z.; Wang, J. Direct access to triazole-olefins through catalytic cycloaddition of azides to unsaturated aldehydes. Chem. Commun. 2013, 49, 10187–10189. [Google Scholar]
- Yeung, D.K.J.; Gao, T.; Huang, J.; Sun, S.; Guo, H.; Wang, J. Organocatalytic 1,3-dipolar cycloaddition reactions of ketones andazides with water as a solvent. Green Chem. 2013, 15, 2384–2388. [Google Scholar] [CrossRef]
- Seus, N.; Goldani, B.; Lenardão, E.J.; Savegnago, L.; Paixão, M.W.; Alves, D. Organocatalytic Synthesis of (Arylselanyl)phenyl-1H-1,2,3-triazole-4-carboxamides by Cycloaddition between Azidophenyl Arylselenides and β-Oxo-amides. Eur. J. Org. Chem. 2013, 2014, 1059–1065. [Google Scholar] [CrossRef]
- Li, W.; Du, Z.; Huang, J.; Jia, Q.; Zhang, K.; Wang, J. Direct access to 1,2,3-triazoles through organocatalytic 1,3-dipolar cycloaddition reaction of allyl ketones with azides. Green Chem. 2014, 16, 3003–3006. [Google Scholar] [CrossRef]
- Li, W.; Du, Z.; Zhang, K.; Wang, J. Organocatalytic 1,3-dipolar cycloaddition reaction of α,β-unsaturated ketones with azides through iminium catalysis. Green Chem. 2015, 17, 781–784. [Google Scholar] [CrossRef]
- Ramachary, D.B.; Shashank, A.B.; Karthik, S. An Organocatalytic Azide–Aldehyde [3+2] Cycloaddition: High-Yielding Regioselective Synthesis of 1,4-Disubstituted 1,2,3-Triazoles. Angew. Chem. Int. Ed. 2014, 53, 10420–10424. [Google Scholar] [CrossRef] [PubMed]
- Shashank, A.B.; Karthik, S.; Madhavachary, R.; Ramachary, D.B. An Enolate-Mediated Organocatalytic Azide–Ketone [3+2]-Cycloaddition Reaction: Regioselective High-Yielding Synthesis of Fully Decorated 1,2,3-Triazoles. Chem. Eur. J. 2014, 20, 16877–16881. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, J. Lewis Base Catalyzed Aerobic Oxidative Intermolecular Azide–Zwitterion Cycloaddition. Angew. Chem. Int. Ed. 2014, 53, 14186–14190. [Google Scholar] [CrossRef] [PubMed]
- Ramasastry, S.S.V. Enamine/Enolate-Mediated Organocatalytic Azide–Carbonyl [3+2] Cycloaddition Reactions for the Synthesis of Densely Functionalized 1,2,3-Triazoles. Angew. Chem. Int. Ed. 2014, 53, 14310–14312. [Google Scholar] [CrossRef]
- Ali, A.; Corrêa, A.G.; Alves, D.; Zukerman-Schpector, J.; Westermann, B.; Ferreira, M.A.B.; Paixao, M.W. An efficient one-pot strategy for the highly regioselective metal-free synthesis of 1,4-disubstituted-1,2,3-triazoles. Chem. Commun. 2014, 50, 11926–11929. [Google Scholar] [CrossRef]
- Tang, W.; Zhang, X. New Chiral Phosphorus Ligands for Enantioselective Hydrogenation. Chem. Rev. 2003, 103, 3029–3070. [Google Scholar] [CrossRef]
- George, A.; Veis, A. Phosphorylated Proteins and Control over Apatite Nucleation, Crystal Growth, and Inhibition. Chem. Rev. 2008, 108, 4670–4693. [Google Scholar] [CrossRef]
- Queffélec, C.; Petit, M.; Janvier, P.; Knight, D.A.; Bujoli, B. Surface Modification Using Phosphonic Acids and Esters. Chem. Rev. 2012, 112, 3777–3807. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.J.; Garner, R.C.; Nicholson, S.; Kissling, C.J.; Mayers, D. Microdose Pharmacokinetics of IDX899 and IDX989, Candidate HIV-1 Non-Nucleoside Reverse Transcriptase Inhibitors, Following Oral and Intravenous Administration in Healthy Male Subjects. J. Clin. Pharmacol. 2009, 49, 1408–1416. [Google Scholar] [CrossRef] [PubMed]
- La Regina, G.; Coluccia, A.; Silvestri, R. Antiviral. Chem. Chemother. 2010, 20, 213–237. [Google Scholar]
- Alexandre, F.R.; Amador, A.; Bot, S.; Caillet, C.; Convard, T.; Jakubik, J.; Musiu, C.; Poddesu, B.; Vargiu, L.; Liuzzi, M.; et al. Synthesis and Biological Evaluation of Aryl-phospho-indole as Novel HIV-1 Non-nucleoside Reverse Transcriptase Inhibitors. J. Med. Chem. 2011, 54, 392–395. [Google Scholar] [CrossRef]
- Muzart, J. Gold-catalysed reactions of alcohols: Isomerisation, inter- and intramolecular reactions leading to C–C and C–heteroatom bonds. Tetrahedron 2008, 64, 5815–5849. [Google Scholar] [CrossRef]
- Kabalka, G.W.; Yao, M.-L. Direct Propargylic Substitution of Hydroxyl Group in Propargylic Alcohols. Curr. Org. Synth. 2008, 5, 28–32. [Google Scholar] [CrossRef]
- Zhu, Y.; Sun, L.; Lu, P.; Wang, Y. Recent Advances on the Lewis Acid-Catalyzed Cascade Rearrangements of Propargylic Alcohols and Their Derivatives. ACS Catal. 2014, 4, 1911–1925. [Google Scholar] [CrossRef]
- Song, X.-R.; Qiu, Y.-F.; Liu, X.-Y.; Liang, Y.-M. Recent advances in the tandem reaction of azides with alkynes or alkynols. Org. Biomol. Chem. 2016, 14, 11317–11331. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tanimoto, H.; Morimoto, T.; Nishiyama, Y.; Kakiuchi, K. Regioselective Rapid Synthesis of Fully Substituted 1,2,3-Triazoles Mediated by Propargyl Cations. Org. Lett. 2013, 15, 5222–5225. [Google Scholar] [CrossRef]
- Zhang, H.; Tanimoto, H.; Morimoto, T.; Nishiyama, Y.; Kakiuchi, K. Acid-mediated synthesis of fully substituted 1,2,3-triazoles: Multicomponent coupling reactions, mechanistic study, synthesis of serine hydrolase inhibitor and its derivatives. Tetrahedron 2014, 70, 9828–9835. [Google Scholar] [CrossRef]
- Yang, T.; Ding, H.; Li, R.; Jin, F.; Song, X.-R.; Chen, X.; Bai, J.; Xiao, Q.; Liang, Y.-M. para-TsOH-Promoted Cascade Reaction of ortho-Propynol Phenyl Azides for the Synthesis of 4-Methoxy Quinolines and Propargyl Methyl Ethers: Insight on Mechanism of Propargylic Alcohols. Asian J. Org. Chem. 2019, 8, 391–398. [Google Scholar] [CrossRef]
- Li, R.; Jin, F.; Song, X.-R.; Yang, T.; Ding, H.; Yang, R.; Xiao, Q.; Liang, Y.-M. Acid-promoted cyclization of 2-propynolphenols leading to 4-tosyloxy-2H-chromenes. Tetrahedron Lett. 2019, 60, 331–334. [Google Scholar] [CrossRef]
- Li, R.; Song, X.-R.; Chen, X.; Ding, H.; Xiao, Q.; Liang, Y.-M. Copper-Catalyzed Cascade Cyclization of 2-Propynolphenols: Access to 4-Phosphorylated 2H-Chromenes. Adv. Synth. Catal. 2017, 359, 3962–3967. [Google Scholar] [CrossRef]
- Song, X.-R.; Li, R.; Ding, H.; Chen, X.; Yang, T.; Jiang, B.; Xiao, Q.; Liang, Y.-M. An efficient approach to 4-chloro quinolines via TMSCl-mediated cascade cyclization of ortho-propynol phenyl azides. Org. Chem. Front. 2018, 5, 1537–1541. [Google Scholar] [CrossRef]
- Song, X.-R.; Li, R.; Yang, T.; Chen, X.; Ding, H.; Xiao, Q.; Liang, Y.-M. Novel and Efficient Access to Flavones under Mild Conditions: Aqueous HI-Mediated Cascade Cyclization/Oxidative Radical Reaction of 2-Propynolphenols. Eur. J. Org. Chem. 2018, 40, 5548–5552. [Google Scholar] [CrossRef]
- Yang, T.; Kou, P.; Jin, F.; Song, X.-R.; Bai, J.; Ding, H.; Xiao, Q.; Liang, Y.-M. TFA-Promoted Sulfonation/Cascade Cyclization of 2-Propynolphenols with Sodium Sulfinates to 4-Sulfonyl 2H-Chromenes under Metal-free Conditions. Org. Chem. Front. 2019, 6, 3162–3166. [Google Scholar] [CrossRef]
- CCDC 1949212 (compound 3a) Contains the Supplementary Cystallographic Data for This Paper. Available online: www.ccdc.cam.ac.uk/data_request/cif (accessed on 26 August 2019).
- Song, X.-R.; Li, R.; Ding, H.; Yang, R.; Xiao, Q.; Liang, Y.-M. Highly efficient access to 4-chloro-2H-chromenes and 1,2-dihydroquinolines under mild conditions: TMSCl-mediated cyclization of 2-propynolphenols/anilines. Tetrahedron Lett. 2016, 57, 4519–4524. [Google Scholar] [CrossRef]
- Zhu, Y.; Yin, G.; Hong, D.; Lu, P.; Wang, Y. Tandem Reaction of Propargylic Alcohol, Sulfonamide, and N-Iodosuccinimide: Synthesis of N-(2-Iodoinden-1-yl)arenesulfonamide. Org. Lett. 2011, 13, 1024–1027. [Google Scholar] [CrossRef]
Sample Availability: Samples of the final compounds are available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).