Cytotoxic Effects of Compounds Isolated from Ricinodendron heudelotii
Abstract
1. Introduction
2. Results
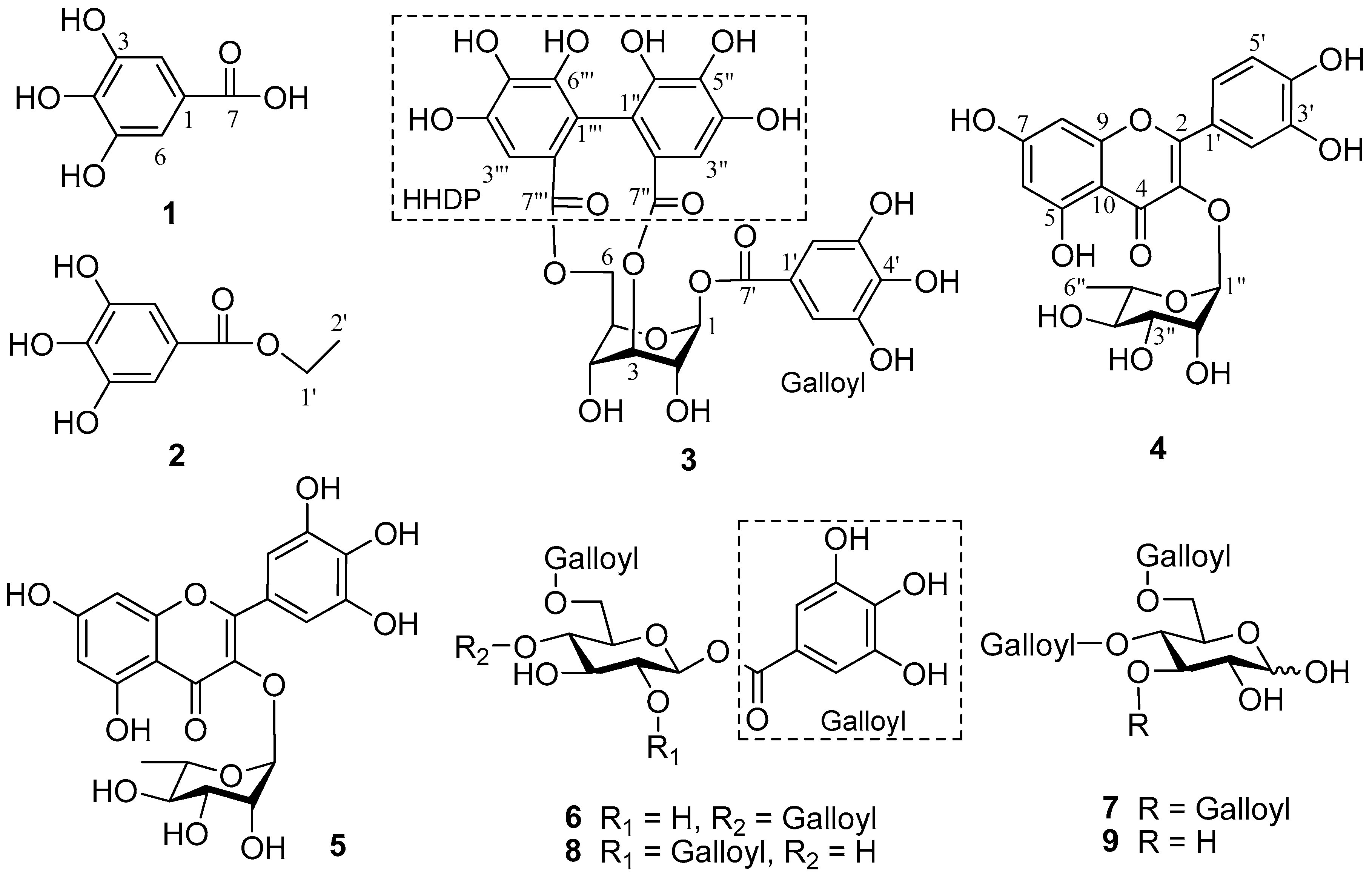
2.1. Structural Elucidation of Isolated Compounds
2.2. Spectroscopic Data of Compounds 1–9
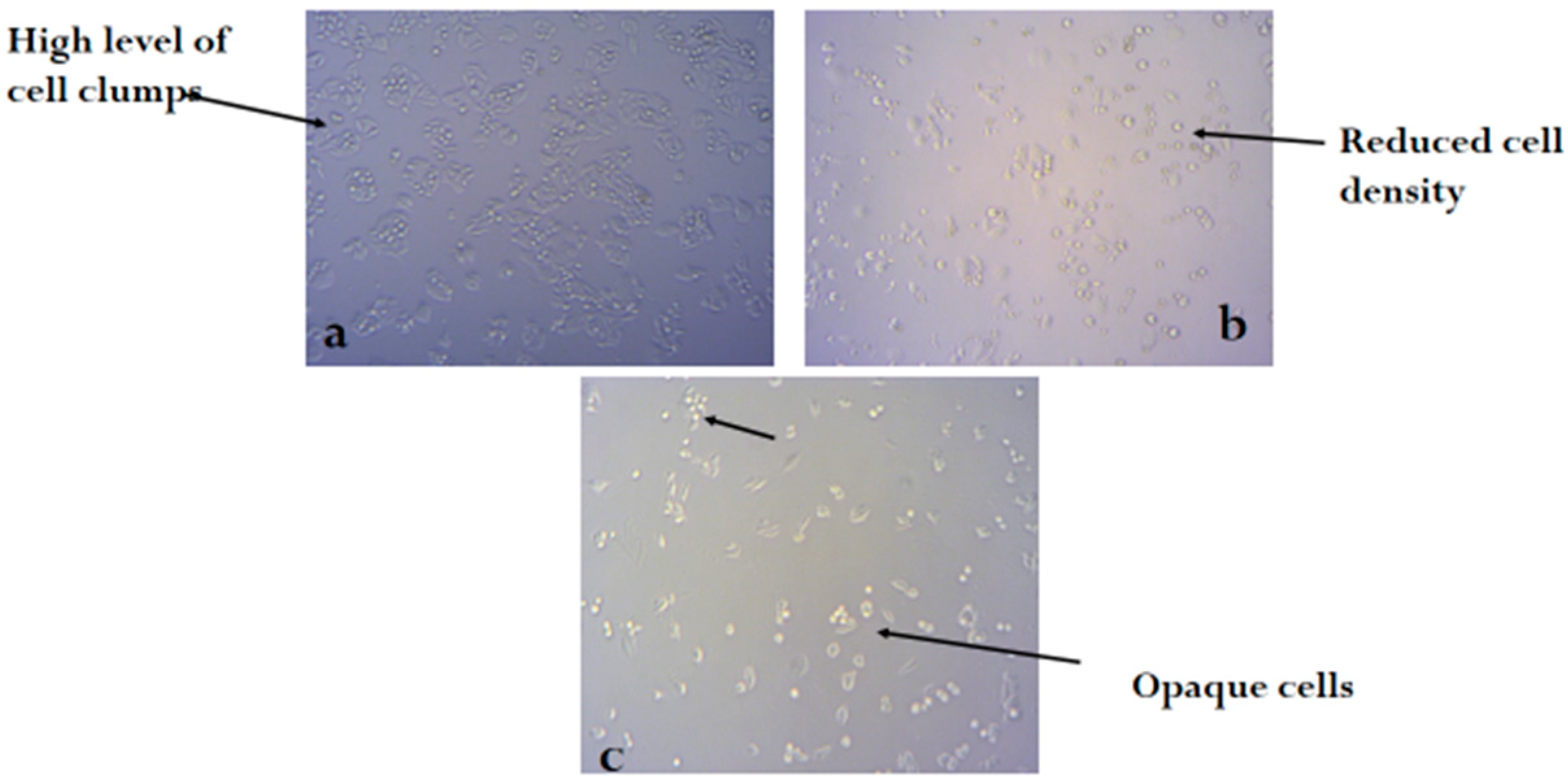
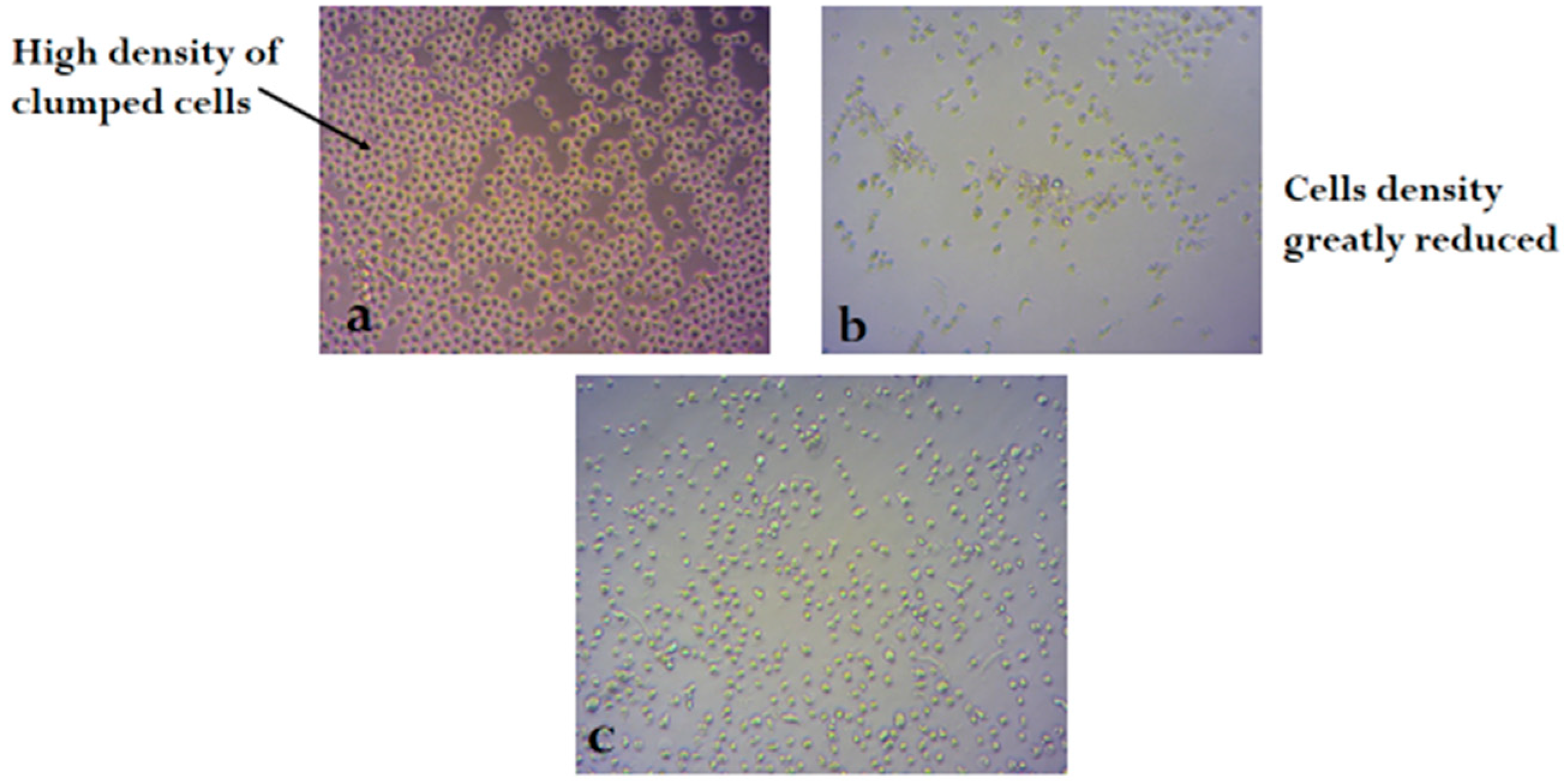
2.3. Bioassay
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Sample Collection
4.3. Extraction and Solvent Partitioning ofthe Crude Extract
4.4. Isolation of Compounds fromthe Partitioned Fraction
4.5. Bioassay
4.5.1. Cell Culture
4.5.2. Sample Preparation
4.5.3. Assay Procedures
4.6. Selectivity Index
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sylla, B.S.; Wild, C.P. A million Africans a year dying from cancer by 2030: What can cancer research and control offer to the continent? Int. J. Cancer 2012, 130, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global Cancer Statistics, 2012. Cancer J. Clin. 2012, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- López-Abente, G.; Aragonés, N.; Pérez-Gómez, B.; Pollan, M.; García-Pérez, J.; Ramis, R.; Fernández-Navarro, P. Time trends in municipal distribution patterns of cancer mortality in Spain. BMC Cancer 2014, 14, 535. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Newman, D.J.; Snader, K.M. Natural products in drug discovery and development. J. Nat. Prod. 1997, 60, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Momeni, J.; Djoulde, R.D.; Akam, M.T.; Kimbu, S.F. Chemical constituents and antibacterial activities of the stem bark extracts of Ricinodendron heudelotii (Euphorbiaceae). Indian J. Pharm. Sci. 2005, 67, 386–389. [Google Scholar]
- Yakubu, O.F.; Adebayo, A.H.; Famakinwa, T.O.; Adegbite, O.S.; Ishola, T.A.; Imonikhe, L.O.; Adeyemi, O.A.; Awotoye, O.A.; Iweala, E.E.J. Antimicrobial and toxicological studies of Ricinodendron heudelotii (Baill.). Asian J. Pharm. Clin. Res. 2018, 11, 299–305. [Google Scholar] [CrossRef]
- Kimbu, S.F.; Keumedjio, F.; Sondengam, L.B.; Connolly, J.D. Two dinorditerpenoids from Ricinodendron heudelotii. Phytochemistry 1991, 30, 619–621. [Google Scholar] [CrossRef]
- Yu, J.H.; Shen, Y.; Wu, Y.; Leng, Y.; Zhang, H.; Yue, J.M. Ricinodols A—G: New tetracyclic triterpenoids as 11β-HSD1 inhibitors from Ricinodendron heudelotii. RSC Adv. 2015, 5, 26777–26784. [Google Scholar] [CrossRef]
- Yakubu, O.F.; Adebayo, A.H.; Okechukwu, E.S.; Adeyemi, O.A.; Iweala, E.J.; Zhang, Y. Co-administration of artemisinin and Ricinodendron heudelotii leaf extract—Effects on selected antioxidants and liver parameters in male Wistar rats. Comp. Clin. Path. 2018, 27, 765–772. [Google Scholar] [CrossRef]
- Eldahshan, O.A. Isolation and structure elucidation of phenolic compounds of carob leaves grown in Egypt. J. Biol. Sci. 2011, 3, 52–55. [Google Scholar]
- Soro, Y.; Kassi, A.B.B.; Bamba, F.; Siaka, S.; Toure, S.A.; Coustard, J.M. Flavonons and gallic acid from leaves of Santaloides afzelii (Connaraceae). Rasayan J. Chem. 2012, 5, 332–337. [Google Scholar]
- Mori, T.; Chang, C.; Maurtua, D.; Hammond, G.B. Isolation of the active compound in Mauria heterophylla, a peruvian plant with antibacterial activity. Phytother. Res. 2006, 20, 160–161. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Chen, L.; Liu, S.; Deng, Y.; Zheng, G.; Gu, Y.; Ming, Y. Bioguided fraction and isolation of the antitumor components from Phyllanthus niruri L. BioMed Res. Int. 2016. [Google Scholar] [CrossRef]
- Bose, S.; Maji, S.; Chakraborty, P. Quercitrin from Ixora coccinea leaves and its anti-oxidant activity. J. Pharmacol. Sci. Technol. 2013, 2, 72–74. [Google Scholar]
- Ibrahim, M.T.; Abu, S.A.; Sleem, A.A. Phytochemical and biological investigation of Thunbergia grandiflora. J. Pharmacol. Phytochem. 2017, 6, 43–51. [Google Scholar]
- Zhong, X.N.; Otsuka, H.; Ide, T.; Hirata, E.; Takushi, A.; Takeda, Y. Three flavonol glycosides from leaves of Myrsine seguinii. Phytochemical 1997, 46, 943–946. [Google Scholar] [CrossRef]
- Hassan, M.; Kubmarawaı, D.; Oladosu, P. Antiallergic polyphenols from Citharexylum spinosum. Trends Phytochem. Res. 2017, 1, 129–134. [Google Scholar]
- Si, C.L.; Xu, J.; Lu, Y.Y.; Su, Z.; Su, Y.F.; Bae, Y.S. Hydrolysable tannins from Juglans sigillata stem barks. Biochem. Syst. Ecol. 2011, 39, 225–227. [Google Scholar] [CrossRef]
- Bag, A.; Bhattacharyya, S.K.; Chattopadhyay, R.R. Isolation and identification of a gallotannin 1,2,6-tri-O-galloyl-β-d-glucopyranose from hydroalcoholic extract of Terminaliachebula fruits effective against multidrug-resistant uropathogens. J. Appl. Microbiol. 2013, 115, 390–397. [Google Scholar] [CrossRef]
- Nawwar, M.A.M.; Hussein, S.A.M.; Merfort, I. NMR spectral analysis of polyphenols from Punica granatum. Phytochemical 1994, 36, 793–798. [Google Scholar] [CrossRef]
- Okuda, T.; Ito, H. Tannins of constant structure in medicinal and food plants-hydrolyzable tannins and polyphenols related to tannins. Molecules 2011, 16, 2191–2217. [Google Scholar] [CrossRef]
- Saijo, R.; Nonaka, G.; Nishioka, I. Tannins and related compounds. LXXXIV. Isolation and characterization of five new hydrolyzable tannins from the bark of Mallotus japonicus. Chem. Pharm. Bull. 1989, 37, 2063–2070. [Google Scholar] [PubMed]
- Rang, H.P. Pharmacology; Churchill Livingstone: London, UK, 2003; ISBN 0-4430-7145-4. [Google Scholar]
- Abd-Rabou, A.A.; Zoheir, K.M.A.; Ahmed, H.H. Potential impact of curcumin and taurine on human hepatoma cells using Huh-7 cell line. Clin. Biochem. 2012, 45, 1519–1521. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, O.T.; Lademann, R. Corilagin, ein weiterer kristallisierter Gerbstoff aus Dividivi. X. Mitteilung über natürliche Gerbstoffe. Justus Liebigs Annalen Der. Chemie 1951, 571, 232–237. [Google Scholar] [CrossRef]
- Satomi, H.; Umemura, K.; Ueno, A.; Hatano, T.; Okuda, T.; Noro, T. Carbonic anhydrase inhibitors from the pericarps of Punica granatum L. Biol. Pharm. Bull. 1993, 16, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, S.; Inoue, Y.; Nakama, S.; Ichiba, T.; Aniya, Y. Antioxidant and hepatoprotective actions of medicinal herb, Terminalia catappa L. from Okinawa Island and its tannin corilagin. Phytomedicine 2007, 14, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Li, H.R.; Liu, J.; Zhang, S.L.; Luo, T.; Wu, F.; Dong, J.H.; Guo, Y.J.; Zhao, L. Corilagin ameliorates the extreme inflammatory status in sepsis through TLR4 signaling pathways. BMC Complement. Altern. Med. 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.T.; Lin, T.C.; Hsu, F.L. Antihypertensive effect of corilagin in the rat. Can. J. Physiol. Pharmacol. 1995, 73, 1425–1429. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Jin, H.; Zhou, J.; Chen, L.; Lu, Y.; Ming, Y.; Yu, Y. A potential anti-tumor herbal medicine, Corilagin, inhibits ovarian cancer cell growth through blocking the TGF-β signaling pathways. BMC Complement. Altern. Med. 2013, 13, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.T.; Li, G.H.; Li, Z.Y.; Feng, S.; Liu, X.Q.; Han, G.K.; Zhang, H.; Qin, X.; Zhang, R.; Nie, Q.; et al. Effect of corilagin on the proliferation and NF-κB in U251 glioblastoma cells and U251 glioblastoma stem-like cells. Evid. Based Complement. Altern. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Park, K.M.; Kim, N.; Yeom, Y.L. Doxorubicin prevents endoplasmic reticulum stress-induced apoptosis. Biochem. Biophys. Res. Commun. 2006, 339, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Puthalakath, H.; O’Reilly, L.A.; Gunn, P.; Lee, L.; Kelly, P.N.; Huntington, N.D.; Hughes, P.D.; Michalak, E.M.; MicKimm-Breschkin, J.; Motoyama, N.; et al. ER stress triggers apoptosis by activating BH3-only protein bim. Cell 2007, 129, 1337–1349. [Google Scholar] [CrossRef] [PubMed]
- Ismail, T.; Calcabrini, C.; Diaz, A.R.; Fimognari, C.; Turrini, E.; Catanzaro, E.; Akhtar, S.; Sestili, P. Ellagitannins in cancer chemoprevention and therapy. Toxins 2016, 8, 151. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.L.; Hsu, Y.L.; Lin, T.C.; Lin, L.T.; Chang, J.K.; Lin, C.C. Casuarinin from the bark of Terminalia arjuna induces apoptosis and cell cycle arrest in human breast adenocarcinoma MCF-7 cells. Planta Med. 2005, 71, 237–743. [Google Scholar] [CrossRef] [PubMed]
- Naus, P.J.; Henson, R.; Bleeker, G.; Wehbe, H.; Meng, F.; Patel, T. Tannic acid synergizes the cytotoxicity of chemotherapeutic drugs in human cholangiocarcinoma by modulating drug efflux pathways. J. Hepatol. 2007, 46, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Le, V.; Esposito, D.; Grace, M.H.; Ha, D.; Pham, A.; Bortolazzo, A.; Bevens, Z.; Kim, J.; Komarnytsky, S.; Lila, M.A.; et al. Cytotoxic effects of ellagitannins isolated from walnuts in human cancer cells. Nutr. Cancer 2014, 66, 1304–1314. [Google Scholar] [CrossRef]
- Gambari, R.; Borgatti, M.; Lampronti, I.; Fabbri, E.; Brognara, E.; Bianchi, N.; Piccagli, L.; Yuen, M.C.W.; Kan, C.W.; Hau, D.K.; et al. Corilagin is a potent inhibitor of NF-κB activity and downregulates TNF-alpha induced expression of IL-8 gene in cystic fibrosis IB3-1 cells. Int. Immunopharmacol. 2012, 13, 308–315. [Google Scholar] [CrossRef]
- Reddy, B.U.; Mullick, R.; Kumar, A.; Sudha, G.; Srinivasan, N.; Das, S. Small molecule inhibitors of HCV replication from Pomegranate. Sci. Rep. 2014, 4, 5411. [Google Scholar] [CrossRef]
- Singh, R.P.; Dhanalakshmi, S.; Agarwal, R. Phytochemicals as cell cycle modulators a less toxic approach in halting human cancers. Cell Cycle 2002, 1, 156–161. [Google Scholar] [CrossRef]
- Thakur, V.S.; Deb, G.; Babcook, M.A.; Gupta, S. Plant phytochemicals as epigenetic modulators: Role in cancer chemoprevention. AAPS J. 2014, 16, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Ming, Y.; Zheng, Z.; Chen, L.; Zheng, G.; Liu, S.; Yu, Y.; Tong, Q. Corilagin inhibits hepatocellular carcinoma cell proliferation by inducing G2/M phase arrest. Cell Biol. Int. 2013, 37, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Peña-Morán, O.A.; Villarreal, M.L.; Álvarez-Berber, L.; Meneses-Acosta, A.; Rodríguez-López, V. Cytotoxicity, post-treatment recovery, and selectivity analysis of naturally occurring podophyllotoxins from Bursera fagaroides var. fagaroides on Breast cancer cell lines. Molecules 2016, 21, 1013–1028. [Google Scholar]
- Iweala, E.E.J.; Liu, F.F.; Cheng, R.R.; Li, Y.; Omonhinmin, C.A.; Zhang, Y.J. Anti-cancer and free radical scavenging activity of some Nigerian food plants in vitro. Int. J. Cancer Res. 2015, 11, 41–51. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Adebayo, A.H.; Tan, N.H.; Akindahunsi, A.A.; Zeng, G.Z.; Zhang, Y.M. Anticancer and antiradical scavenging activity of Ageratum conyzoides L. (Asteraceae). Pharmacogn. Mag. 2010, 21, 62–66. [Google Scholar]
- Dahham, S.S.; Tabana, Y.M.; Iqbal, M.A.; Ahamed, M.B.K.; Ezzat, M.O.; Majid, A.S.A.; Majid, A.M.S.A. The anticancer, antioxidant and antimicrobial properties of the sesquiterpene β-Caryophyllene from the essential oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829. [Google Scholar] [CrossRef]
Sample Availability: Not available. |

| Sample | HL-60 | MCF-7 | SW480 | BEAS-2B |
|---|---|---|---|---|
| Corilagin (3) | 25.81 ± 0.67 * | 33.18 ± 0.76 * | 37.04 ± 1.06 * | 16.32 ± 1.43 |
| Cisplatin | 2.32 ± 0.07 | 27.43 ± 1.26 | 10.19 ± 1.03 | --- |
| Compounds | HL-60 | A-549 | SMMC-7721 | MCF-7 | SW480 |
|---|---|---|---|---|---|
| 1 | 14.80 ± 5.00 | 13.67 ± 2.91 | 5.85 ± 1.16 | 18.41 ± 4.00 | 10.04 ± 3.51 |
| 2 | 12.26 ± 2.29 | 11.82 ± 0.14 | 5.19 ± 0.83 | 24.76 ± 3.30 | 1.15 ± 0.28 |
| 3 | 76.23 ± 0.51 | 29.19 ± 1.42 | 4.91 ± 1.43 | 57.93 ± 3.19 | 49.82 ± 3.80 |
| 4 | 4.56 ± 2.61 | 5.49 ± 2.01 | 1.12 ± 1.05 | 14.95 ± 0.82 | 2.44 ± 2.49 |
| 5 | 0.65 ± 2.87 | 8.32 ± 3.46 | 1.62 ± 1.05 | 10.67 ± 4.05 | 12.45 ± 3.66 |
| Sample | HL-60 | MCF-7 | SW480 |
|---|---|---|---|
| Corilagin (3) | 0.63 | 0.49 | 0.44 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yakubu, O.F.; Adebayo, A.H.; Dokunmu, T.M.; Zhang, Y.-J.; Iweala, E.E.J. Cytotoxic Effects of Compounds Isolated from Ricinodendron heudelotii. Molecules 2019, 24, 145. https://doi.org/10.3390/molecules24010145
Yakubu OF, Adebayo AH, Dokunmu TM, Zhang Y-J, Iweala EEJ. Cytotoxic Effects of Compounds Isolated from Ricinodendron heudelotii. Molecules. 2019; 24(1):145. https://doi.org/10.3390/molecules24010145
Chicago/Turabian StyleYakubu, Omolara F., Abiodun H. Adebayo, Titilope M. Dokunmu, Ying-Jun Zhang, and Emeka E.J. Iweala. 2019. "Cytotoxic Effects of Compounds Isolated from Ricinodendron heudelotii" Molecules 24, no. 1: 145. https://doi.org/10.3390/molecules24010145
APA StyleYakubu, O. F., Adebayo, A. H., Dokunmu, T. M., Zhang, Y.-J., & Iweala, E. E. J. (2019). Cytotoxic Effects of Compounds Isolated from Ricinodendron heudelotii. Molecules, 24(1), 145. https://doi.org/10.3390/molecules24010145





