Abstract
An efficient and practical synthetic route toward chiral matsutakeol and analogs was developed by asymmetric addition of terminal alkyne to aldehydes. (R)-matsutakeol and other flavored substances were feasibly synthesized from various alkylaldehydes in high yield (up to 49.5%, in three steps) and excellent enantiomeric excess (up to >99%). The protocols may serve as an alternative asymmetric synthetic method for active small-molecule library of natural fatty acid metabolites and analogs. These chiral allyl alcohols are prepared for food analysis and screening insect attractants.
1. Introduction
Many natural unsaturated alcohols 1–6 (Figure 1) are very important fatty acid metabolites derived from fungi [1] and plants [2]. These substances have been found in nutrient, pharmaceutical, and agricultural use, as their structural diversity and variety of biological activities. For examples, the chiral allyl alcohols 6a–c (Figure 1) are a class of important flavor substances and dietary supplements which are widely used in the food industry [3,4,5]. (R)-matsutakeol 6a isolated from matsutake [6], has been found to possess antitumor properties [3]. Effects of inhibition on fungal spore germination and mushroom and plant development have also been discovered [7]. Recently, (R)-matsutakeol 6a and its analogs are widely used in insecticidal compositions as effective attractants for some harmful hematophagous insects [8,9]. In particular, the enantioselectivity and chiral configuration of the compounds directly determined biological activities such as smell and taste [10].
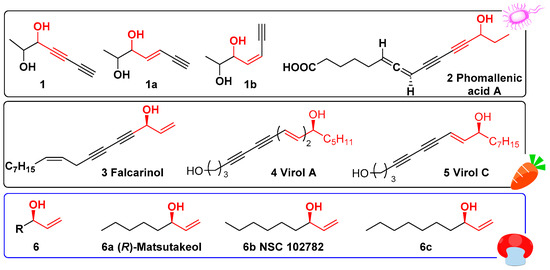
Figure 1.
Natural unsaturated alcohols derived from fungi and plant.
Inspired by potential applications above, great attention has been paid to preparation of compounds 6 with high enantioselectivity and structural diversity [11,12,13,14,15,16,17,18,19]. To take (R)-matsutakeol 6a as an example, in 1987, 6a and its enantiomer were obtained by Helmchen through the asymmetric retro Diels-Alder reaction [11]. In the same year, Takano prepared the (R)-matsutakeol by using the optically active starting material of (R)-epichlorohydrin [12]. In 1988, Kitamura obtained (R)-matsutakeol (21% ee) via kinetic resolution of racemic allylic alcohols [13]. Oppolzer developed an enantioseletive synthesis of (R)-matsutakeol (96% ee) by catalytic asymmetric addition of divinylzinc to hexanal [14]. In 2005, a chiral resolution of racemic matsutakeol was conducted using (S)-(+)-2-methoxy-2-(1-naphthyl) propionic acid (MαNP acid) by Kusuda [15]. In addition, various enzymatic kinetic resolution and enzyme-catalyzed reactions have also been used in preparation of chiral matsutakeol. In 2010, Bisogno developed an efficient oxidoreductase-catalyzed system to obtain (R)-matsutakeol in 86% ee [16]. Recently, Rej prepared (S)-matsutakeol as an important intermediate, by ME-DKR (metal enzyme combined dynamic kinetic resolution) of (±)-oct-1-en-3-ol [17,18]. In 2016, Lee developed a synthetic strategy towards (S)-matsutakeol in 10 steps from inexpensive, natural (2S,3S)-d-tartaric acid with 32% yield [19].
In summary, matsutakeol could be prepared by chiral pool synthesis [19], enzymatic kinetic resolution [17,18], and asymmetric synthesis [14] (Scheme 1). Although, several synthetic routes have been developed, there is still a lack of efficient method for obtaining a highly enantioselective allyl alcohols library in screening for flavors or attractants. Compared to asymmetric synthesis (Scheme 1, method 3), the structural diversity of the reaction products is limited by chiral sources (Scheme 1, method 1) [19], and the effect of enzymatic reaction (Scheme 1, method 2) is constrained by stability and activity of enzymes [17,18]. By retro-synthetic analysis, the chiral olefinic alcohol 6 could be formed from propargyl alcohol 12, and chiral propargyl alcohol 12 can be synthesized from the corresponding methyl propiolate 13 (Scheme 1, method 3). Highly enantioselective methyl propiolate (S)-13 is obtained by Zn-catalyzed asymmetric addition of terminal alkynes 15 to various aldehydes 14. This reaction has been widely studied (Scheme 2) [20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50], in which numerous chiral ligands are applied, such as (R,R)-ProPhenol 16a [24,39], (R)-BINOL 17a, and its derivatives [28,30], β-Sulfonamide Alcohol 18 [31,32], Wolf’s ligand 19 [41], and Wang’s ligand 20 (our previous group research) [33]. Among the numerous catalysts, (R,R)-ProPhenol 16a and (R)-BINOL 17a are readily available and well-established [20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50].
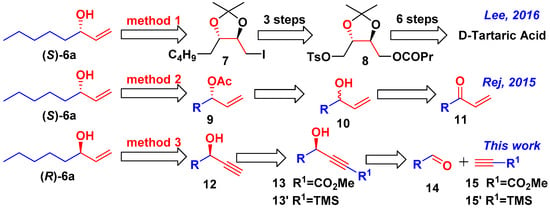
Scheme 1.
Retro-synthetic analysis of matsutakeol and its natural analogs reported in the latest literature.
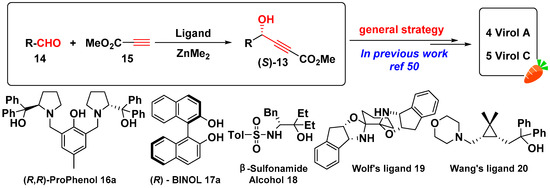
Scheme 2.
Zn-catalyzed asymmetric direct addition of terminal alkynes to aldehydes.
2. Results and Discussion
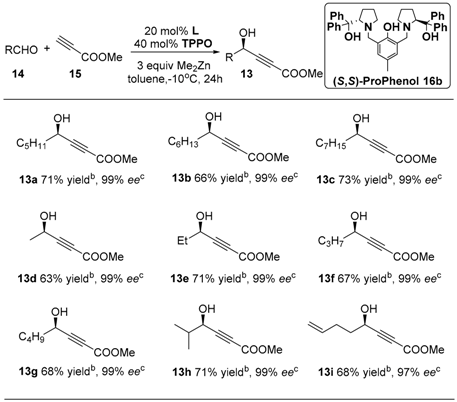
Herein we report the asymmetric synthesis of (R)-matsutakeol and its natural analogs. In our previous work (Scheme 2) [50], the reaction conditions were optimized for the stereoselective addition of methyl propiolate to aliphatic aldehydes, and the highest enantiomeric excess values (97%–99% ee) of (S)-alkynol product 13 were afforded by (R,R)-ProPhenol/Zn complex. It is developed in a general strategy toward the total synthesis of C17 polyacetylenes such as virol A and virol C [50].
On the basis of previous work [24,25,39,50], we expanded substrate scope of the (S,S)-ProPhenol- catalyzed direct asymmetric addition, affording the corresponding (R)-configured propargyl alcohols 13 in moderated yields (63%–73%) and with excellent enantioselectivities (97%–99% ee) (Table 1). Thus, we developed a general method with two steps to obtain highly enantioselective allyl alcohol flavors.

Table 1.
Synthesis of chiral alkynol units 13a–e of C17 polyacetylenes via the asymmetric addition of methyl propiolate to aliphatic aldehydes a.
As shown in Scheme 3, treatment of compounds 13a–c with LiOH in THF gave the corresponding carboxylic acid intermediates [38], which were then subjected to the CuCl-catalyzed decarboxylation directly, producing the chiral propargyl alcohols 12a–c in good yields (83%–86%) and without loss of enantio-selectivity (99% ee). Reduction of compounds 12a–c with NaBH4 in the presence of Ni(OAc)2 gave (R)-matsutakeol 6a and its natural analogs 6b and 6c in good yields (82%–85%) [51]. However, the ee values of (R)-matsutakeol and analogs could not be directly resolved by HPLC on a chiral column conveniently. Therefore, derivatization of compounds 6a–c through introducing 3,5-dinitrobenzoyl moiety to the natural molecules, directly afforded compounds 21a–c in nearly quantitative yields (93%–95%) and 99% ee.
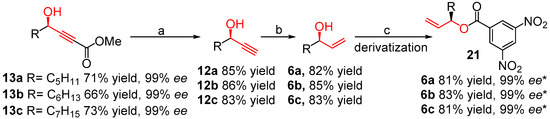
Scheme 3.
Total synthesis of (R)-matsutakeol and its natural analogs 6a–c by using (S,S)-ProPhenol (Scheme 1, Method 3). Reagents and conditions: (a) (i) LiOH, THF, 0~25 °C, 1 h; (ii) CuCl, CH3CN,13 h; (b) Ni(OAc)2, NaBH4, EDA, EtOH, 0~25 °C; (c) 3,5-Dinitrobenzoyl chloride, Et3N, 0~25 °C, overnight; * The ee value is determined by HPLC on a chiral column after derivatization.
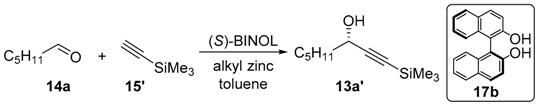
The BINOL-ZnEt2-Ti (IV) complex catalyzed asymmetric addition of trimethylsilylacetylene to the aliphatic aldehydes was well developed by Pu’s group [20,22,34]. It is also a practical approach for providing chiral acetylene alcohols. In the optimization procedure, to increase the catalyst loading of (R,R)-ProPhenol 16b (entry 1 and 2) and to reduce the temperature (entry 2 and 3) could slightly improve the ee value, of product 13a’ (Table 2, entry 2, 72% yield, 78% ee). Compared to addition catalyzed by (R,R)-ProPhenol 16b, almost no product was detected at −10 °C (Table 2, entry 4). The reaction yields and optical yields were increased on increasing the amount of BINOL (Table 2, entry 5–7). When the amount of BINOL was increased to 60%, there was no obvious improvement in yield and ee value (entry 7, 67% yield, 81 % ee). Compared to the reaction catalyzed by (R,R)-ProPhenol 16b (entry 2, 72% yield, 78% ee and entry 3, 74% yield, 76% ee), the asymmetric addition of trimethylsilylacetylene by using BINOL as chiral ligand could be smoothly carried out at room temperature, affording higher ee value and almost the same yield (entry 6, 71% yield, 80% ee).

Table 2.
Screening of reaction conditions and ligands of asymmetric addition of 15’ with 14a.
Herein, we attempted to synthesize the above natural products 6a–c on gram scale, by employing the (S)-BINOL 17b as the catalyst. As shown in Scheme 4, (S)-BINOL efficiently catalyzed the asymmetric addition of trimethylsilylacetylene with the corresponding aliphatic aldehydes 14a–c, affording compounds 13a’–c’ in moderate yields (71%–72%) and enantioselectivities (80%–82% ee), slightly lower than those of compounds 13a–c. K2CO3-promoted deprotection of trimethyl group of compounds 13a’–c’ under mild conditions gave compounds 12a–c in good yields (87%–89%) [52]. Following the same procedures as shown in Scheme 3, compounds 6a–c were obtained in moderate yields (81%–83%) and nearly without loss of enantioselectivities (79%–82% ee, the ee values were measured by HPLC after derived by 3,5-dinitrobenzoyl chloride). The enantiomeric purity of 21a–c could be improved to over 99% ee by slow recrystallization from diethylether/n-hexane (1:5) at a low temperature. Subsequently, the esters 21a–c could be quantitatively hydrolyzed to the corresponding chiral alcohols 6a–c followed in our previous group research [22].
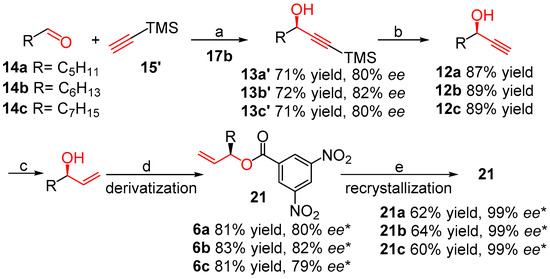
Scheme 4.
Total synthesis of (R)-matsutakeol and its natural analogs 6a–c by using (S)-BINOL (Scheme 1, Method 3). Reagents and conditions: (a) (i) ZnEt2, THF, N2, rt; (ii) Ti(iPrO)4, THF, N2, rt; (b) K2CO3, MeOH, 0~25 °C; (c) Ni(OAc)2, NaBH4, EDA, EtOH, 0~25 °C; (d) 3,5-Dinitrobenzoyl chloride, Et3N, 0~25 °C, overnight; (e) Recrystallization from diethylether/n-hexane (1/5) at 0~25 °C, * The ee value is determined by HPLC on a chiral column after derivatization.
3. Materials and Methods
All reactions were performed under an argon atmosphere. Solvents were dried according to standard procedures and distilled before use. All reagents were purchased commercially and used without further purification, unless stated otherwise. 1H- and 13C-NMR spectra were recorded at 300 and 75 MHz, respectively. High-resolution mass spectra were recorded on an agilent instrument by the TOF MS technique. Enantiomeric excesses (ee) were determined by chiral HPLC analyses using a chiral column (Chiralpak OD-H, AD-H, OJ-H), and elution with isopropanol-hexane. The optical rotations were measured on PERKIN ELMER 341 Polarimeter. 1H-, 13C-NMR spectra and HPLC chromatography of the chiral products are in the Supplementary Materials.
3.1. General Procedure of Asymmetric Addition of Methyl Propiolate to Aliphatic Aldehydes (Table 1)
To a stirred solution of methyl propiolate (84 mg, 1 mmol), (S,S)-ProPhenol (128 mg, 0.2 mmol), triphenylphosphine oxide (111 mg, 0.4 mmol) in toluene (1 mL), dimethylzinc (2.5 mL, 1.2 M in toluene, 3 mmol) was added slowly at −10 °C. After stirring for 1.5 h at −10 °C, aldehyde (1.5 mmol) in toluene (3 mL) was added via syringe at a slow rate in 24 h at −10 °C, and quenched with water (10 mL). The mixture was filtered through a celite pad. The aqueous phase was extracted with ether. The combined organic phases were washed with saturated brine solution, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel chromatography to get the product [25,36].
3.1.1. Synthesis of (R)-Methyl-4-hydroxynon-2-ynoate: (R)-13a
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13a (131 mg, 71% yield, 99% ee) as colorless oil. [α] = + 5.7 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.57–4.44 (m, 1H), 3.76 (s, 3H), 2.66 (d, J = 4.7 Hz, 1H), 1.80–1.67 (m, 2H), 1.52–1.38 (m, 2H), 1.35–1.25 (m, 4H), 0.88 (t, J = 6.7 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.57, 88.19, 75.78, 61.71, 52.40, 36.46, 30.95, 24.23, 22.08, 13.54. HRMS ESI [M + Na]+ calcd for C10H16NaO3+ 207.0992, found 207.0992. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 14.93 min, minor (S)-enantiomer tr = 16.73 min.
3.1.2. Synthesis of (R)-Methyl-4-hydroxydec-2-ynoate: (R)-13b
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13b (131 mg, 66% yield, 99% ee) as colorless oil. [α] = + 5.8 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.57–4.44 (m, 1H), 3.77 (s, 3H), 2.36 (d, J = 5.7 Hz, 1H), 1.84–1.71 (m, 2H), 1.51–1.24 (m, 8H), 0.87 (t, J = 6.7 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.51, 88.07, 75.85, 61.76, 52.40, 36.53, 31.25, 28.44, 24.51, 22.16, 13.62. HRMS ESI [M + Na]+ calcd for C11H18NaO3+ 221.1148, found 221.1150. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (99:1 n-hexanes:isopropanol, 0.7 mL/min, 210 nm), major (R)-enantiomer tr = 31.48 min, minor (S)-enantiomer tr = 37.63 min.
3.1.3. Synthesis of (R)-Methyl-4-hydroxyundec-2-ynoate: (R)-13c
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13c (155 mg, 73% yield, 99% ee) as colorless oil. [α] = +6.7 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.57–4.44 (m, 1H), 3.76 (s, 3H), 2.67 (d, J = 5.8 Hz, 1H), 1.76–1.70 (m, 2H), 1.46–1.42 (m, 2H), 1.35–1.23 (m, 8H), 0.92–0.82 (m, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.58, 88.22, 75.77, 61.70, 52.41, 36.50, 31.36, 28.75, 28.72, 24.56, 22.23, 13.66. HRMS ESI [M + Na]+ calcd for C12H20NaO3+ 235.1305, found 235.1305. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (99:1 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 13.97 min, minor (S)-enantiomer tr = 15.47 min.
3.1.4. Synthesis of (R)-Methyl-4-hydroxypent-2-ynoate: (R)-13d
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13d (81 mg, 63% yield, 99% ee) as colorless oil. [α] = +3.4 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.57–4.44 (m, 1H), 3.80 (s, 3H), 2.97 (d, J = 5.6 Hz, 1H), 1.53 (d, J = 6.7 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.61, 88.72, 74.99, 57.53, 52.48, 22.85. HRMS ESI [M + Na]+ calcd for C6H8NaO3+ 151.0366, found 151.0365. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 220 nm), major (R)-enantiomer tr = 23.06 min, minor (S)-enantiomer tr = 26.75 min.
3.1.5. Synthesis of Methyl (R)-Methyl-4-hydroxyhex-2-ynoate: (R)-13e
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13e (101 mg, 71% yield, 99% ee) as colorless oil. [α] = +8.2 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.57–4.44 (m, 1H), 3.79 (s, 3H), 3.09 (d, J = 5.8 Hz, 1H), 1.87–1.75 (m, 2H), 1.04 (t, J = 7.4 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.64, 88.07, 75.76, 62.84, 52.45, 29.64, 8.88. HRMS ESI [M + Na]+ calcd for C7H10NaO3+ 165.0522, found 165.0522. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 220 nm), major (R)-enantiomer tr = 20.98 min, minor (S)-enantiomer tr = 23.86 min.
3.1.6. Synthesis of (R)-Methyl-4-hydroxyhept-2-ynoate: (R)-13f
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13f (105 mg, 67% yield, 99% ee) as colorless oil. [α] = +5.4 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.57–4.44 (m, 1H), 3.79 (s, 3H), 3.02 (d, J = 5.8 Hz, 1H), 1.83–1.66 (m, 2H), 1.58–1.41 (m, 2H), 0.96 (t, J = 7.3 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.64, 88.29, 75.69, 61.40, 52.45, 38.47, 17.88, 13.22. HRMS ESI [M + Na]+ calcd for C8H12NaO3+ 179.0679, found 179.0681. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 220 nm), major (R)-enantiomer tr = 24.14 min, minor (S)-enantiomer tr = 25.45 min.
3.1.7. Synthesis of (R)-Methyl-4-hydroxyoct-2-ynoate: (R)-13g
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13g (116 mg, 68% yield, 99% ee) as colorless oil. [α] = +2.3 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.55–4.44 (m, 1H), 3.77 (s, 3H), 2.39 (d, J = 5.8 Hz, 1H), 1.82–1.68 (m, 2H), 1.48–1.31 (m, 4H), 0.91 (t, J = 7.1 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.51, 88.06, 75.85, 61.75, 52.40, 36.23, 26.66, 21.88, 13.48. HRMS ESI [M + Na]+ calcd for C9H14NaO3+ 193.0835, found 193.0835. Enantiomeric excess was determined by HPLC with a Chiralcel OJ-H column (95:5 n-hexanes:isopropanol, 1.0 mL/min, 220 nm), major (R)-enantiomer tr = 7.45 min, minor (S)-enantiomer tr = 8.02 min.
3.1.8. Synthesis of (R)-Methyl-4-hydroxy-5-methylhex-2-ynoate: (R)-13h
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13h (111 mg, 71% yield, 99% ee) as colorless oil. [α] = +7.2 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.27 (t, J = 5.9 Hz, 1H), 3.76 (s, 3H), 3.11 (d, J = 5.9 Hz, 1H), 1.97–1.90 (m, 1H), 1.00 (dd, J = 6.8, 4.4 Hz, 6H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.64, 87.36, 67.05, 52.44, 33.79, 17.58, 17.09. HRMS ESI [M + Na]+ calcd for C8H12NaO3+ 179.0679, found 179.0680. Enantiomeric excess was determined by HPLC with a Chiralcel OJ-H column (95:5 n-hexanes:isopropanol, 1.0 mL/min, 220 nm), major (R)-enantiomer tr = 8.66 min, minor (S)-enantiomer tr = 10.71 min.
3.1.9. Synthesis of (R)-Methyl-4-hydroxyoct-7-en-2-ynoate: (R)-13i
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-13i (114 mg, 68% yield, 97% ee) as colorless oil. [α] = −10.1 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 5.89–5.75 (m, 1H), 5.12–4.96 (m, 2H), 4.52 (q, J = 6.4 Hz, 1H), 3.79 (s, 3H), 2.64 (d, J = 5.5 Hz, 1H), 2.26 (dd, J = 14.4, 6.8 Hz, 2H), 1.92–1.81 (m, 2H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 153.53, 136.64, 115.46, 87.75, 76.03, 61.08, 52.48, 35.48, 28.73. HRMS ESI [M + Na]+ calcd for C9H12NaO3+ 191.0679, found 191.0679. Enantiomeric excess was determined by HPLC with a Chiralcel OJ-H column (95:5 n-hexanes:isopropanol, 1.0 mL/min, 220 nm), major (R)-enantiomer tr = 9.01 min, minor (S)-enantiomer tr = 9.75 min.
3.2. General Procedure for the Synthesis of Chiral Alkynols 12 from Propargyl Alcohols 13
A solution of the chiral alkynol (5 mmol) and THF (60 mL) were cooled to 0 °C, 1 M aq LiOH (25 mmol, 5 eq) was added at a slow rate. The solution was warmed to rt and stirred for an additional 1 h before it was quenched with 1M aq NaHSO4 (50 mL). The aqueous phase was extracted by ethyl acetate. The combined organic phases were dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was dissolved in acetonitrile (12 mL), CuCl (0.5940 g, 6 mmol, 1.2 eq) was added in one portion to the mixture. The mixture was allowed to warm to r.t. and stirred for another 13 h. The aqueous phase was extracted by ether. The combined organic phases were dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel chromatography to get the product.
3.2.1. Synthesis of (R)-Oct-1-yn-3-ol: (R)-12a
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 15:1) to offer (R)-12a (536 mg, 85% yield) as colorless oil. [α] = +18.5 (c 1.0, ethyl ether), lit. [53] [α] = +19.3 (c 1.0, ethyl ether), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.33–4.27 (m, 1H), 3.10 (s, 1H), 2.40 (d, J = 2.1 Hz, 1H), 1.69–1.60 (m, 2H), 1.46–1.33 (m, 2H), 1.33–1.11 (m, 4H), 0.83 (t, J = 7.0 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 84.82, 72.31, 61.71, 37.15, 31.03, 24.33, 22.11, 13.53. HRMS ESI [M + Na]+ calcd for C8H14NaO+ 149.0937, found 149.0938.
3.2.2. Synthesis of (R)-Non-1-yn-3-ol: (R)-12b
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 15:1) to offer (R)-12b (603 mg, 86% yield) as colorless oil. [α] = +5.3 (c 2.0, CHCl3), lit. [54] [α] = +4.6 (c 1.92, CHCl3), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.37 (t, J = 5.9 Hz, 1H), 2.47 (d, J = 5.9 Hz, 1H), 2.38 (s, 1H), 1.75–1.68 (m, 2H), 1.52–1.41 (m, 2H), 1.38–1.27 (m, 6H), 0.89 (t, J = 6.7 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 84.78, 72.38, 61.90, 37.28, 31.35, 28.53, 24.62, 22.19, 13.66. HRMS ESI [M + Na]+ calcd for C9H16NaO+ Exact Mass: 163.1093, found 163.1093.
3.2.3. Synthesis of (R)-Dec-1-yn-3-ol: (R)-12c
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 15:1) to offer (R)-12c (640 mg, 83% yield) as colorless oil. [α] = +6.2 (c 1.1, CHCl3), lit. [55] [α] = +4.2 (c 1.1, CHCl3), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.35 (dd, J = 6.5, 4.9 Hz, 1H), 2.44 (d, J = 2.1 Hz, 1H), 2.33 (s, 1H), 1.78–1.60 (m, 2H), 1.50–1.38 (m, 2H), 1.32–1.26 (m, 8H), 0.86 (t, J = 6.8 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 84.74, 72.41, 61.97, 37.31, 31.40, 29.34, 28.83, 24.66, 22.26, 13.69. HRMS ESI [M + Na]+ calcd for C10H18NaO+ 177.1250, found 177.1250.
3.3. General Procedure for the Selective Reduction of the Chiral Alkynols 12
To a stirred solution of nickel acetate tetrahydrate (352 mg, 2 mmol) in ethanol (5 mL) under hydrogen, sodium borohydride (76 mg, 2 mmol) in ethanol (2 mL) was added at 0 °C. After stirring for 1 h at 25 °C, ethylenediamine (481 mg, 8 mmol) was added. The reaction mixture was stirred for another 10 min before chiral alkynol (2 mmol) in ethanol (2 mL) was added slowly to the reaction mixture at 0 °C. The reaction was allowed to proceed at 25 °C under hydrogen for 6 h at 25 °C. The mixture was filtered through a celite pad, diluted with ether, and concentrated under reduced pressure. The residue was purified by silica gel chromatography to get the product.
3.3.1. Synthesis of (R)-Oct-1-en-3-ol: (R)-6a
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 10:1) to offer (R)-6a (210 mg, 82% yield) as colorless oil. [α] = −10.8 (c 1.1, CHCl3), lit. [56] [α] = −10.0 (c 1.67, CHCl3), 1H-NMR (300 MHz, CDCl3) δ (ppm): 5.95–5.83 (m, 1H), 5.27–5.21 (m, 1H), 5.14–5.10 (m, 1H), 4.12 (d, J = 4.8 Hz, 1H), 1.61 (d, J = 2.8 Hz, 1H), 1.58–1.48 (m, 2H), 1.44–1.25 (m, 6H), 0.91 (t, J = 6.7 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 141.02, 114.12, 72.92, 36.68, 31.40, 24.64, 22.23, 13.64. HRMS ESI [M + Na]+ calcd for C8H16NaO+ 151.1093, found 151.1093.
3.3.2. Synthesis of (R)-Non-1-en-3-ol: (R)-6b
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 10:1) to offer (R)-6b (242 mg, 85% yield) as colorless oil. [α] = −11.5 (c 1.1, ethanol), lit. [57] [α] = −13.4 (c 1.12, ethanol), 1H-NMR (300 MHz, CDCl3) δ (ppm): 5.94–5.83 (m, 1H), 5.27–5.20 (m, 1H), 5.14–5.09 (m, 1H), 4.12 (d, J = 5.9 Hz, 1H), 1.67 (d, J = 3.5 Hz, 1H), 1.60–1.49 (m, 2H), 1.40–1.29 (m, 8H), 0.90 (t, J = 6.7 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 141.03, 114.10, 72.91, 36.72, 31.43, 28.86, 24.93, 22.22, 13.68. HRMS ESI [M + Na]+ calcd for C9H18NaO+ 165.1250, found 165.1250.
3.3.3. Synthesis of (R)-Dec-1-en-3-ol: (R)-6c
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 10:1) to offer (R)-6c (259 mg, 83% yield) as colorless oil. [α] = −16.7 (c 1.1, CHCl3), lit. [58] [α] = −18.1 (c 1.22, CHCl3), 1H-NMR (300 MHz, CDCl3) δ (ppm): 5.92–5.81 (m, 1H), 5.32–5.17 (m, 1H), 5.16–5.06 (m, 1H), 4.15–4.05 (m, 1H), 1.91 (d, J = 3.7 Hz, 1H), 1.58–1.46 (m, 2H), 1.40–1.22 (m, 10H), 0.89 (t, J = 6.7 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 141.04, 114.04, 72.86, 36.70, 31.45, 29.16, 28.88, 24.97, 22.27, 13.68. HRMS ESI [M + Na]+ calcd for C10H20NaO+ 179.1406, found 179.1408.
3.4. General Procedure for the Esterification Reaction and Determination of Enantiomeric Excess by HPLC
Triethylamine (152 mg, 1.5 mmol) and 3,5-dinitrobenzoyl chloride (277 mg, 1.2 mmol) were added to a stirred solution of the chiral alcohol (1 mmol) in CH2Cl2 (6 mL) at −5 °C. The mixture was stirred for 5 h at 25 °C before water (2 mL) was poured into the mixture at 0 °C. The aqueous phase was extracted with ether, and combined organic phases were washed with saturated brine solution, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel chromatography to get the product.
3.4.1. Synthesis of (R)-Oct-1-en-3-yl-3,5-dinitrobenzoate: (R)-21a
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-21a (306 mg, 95% yield, 99% ee) as white solid. [α] = −16.2 (c 1.1, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 9.22 (t, J = 2.1 Hz, 1H), 9.16 (d, J = 2.1 Hz, 2H), 5.92 (m 1H), 5.56 (q, J = 6.9 Hz, 1H), 5.36 (m, 2H), 1.96–1.72 (m, 2H), 1.48–1.26 (m, 6H), 0.89 (t, J = 6.9 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 161.45, 148.34, 135.09, 134.00, 129.02, 121.92, 118.05, 77.82, 33.69, 31.09, 24.40, 22.10, 13.57. HRMS ESI [M + Na]+ calcd for C15H18N2NaO6+ 345.1057, found 345.1057. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (99:1 n-hexanes: isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 17.74 min, minor (S)-enantiomer tr = 14.46 min.
3.4.2. Synthesis of (R)-Non-1-en-3-yl-3,5-dinitrobenzoate: (R)-21b
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-21b (312 mg, 93% yield, 99% ee) as colorless oil. [α] = −18.3 (c 1.1, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 9.21 (t, J = 2.1 Hz, 1H), 9.15 (d, J = 2.1 Hz, 2H), 6.01–5.84 (m, 1H), 5.56 (d, J = 6.6 Hz, 1H), 5.34 (dd, J = 26.1, 13.8 Hz, 2H), 1.95–1.70 (m, 2H), 1.45–1.26 (m, 8H), 0.87 (t, J = 6.7 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 161.45, 148.33, 135.11, 133.99, 129.02, 121.92, 118.01, 77.81, 33.73, 31.27, 28.59, 24.70, 22.17, 13.63. HRMS ESI [M + Na]+ calcd for C16H20N2NaO6+ 359.1214, found 359.1214. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 15.27 min, minor (S)-enantiomer tr = 12.67 min.
3.4.3. Synthesis of (R)-Dec-1-en-3-yl-3,5-dinitrobenzoate: (R)-21c
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-6c (332 mg, 95% yield, 99% ee) as white solid. [α] = −21.2 (c 1.05, CH2Cl2), 1H-NMR (300 MHz, CDCl3) δ (ppm): 9.23 (t, J = 2.1 Hz, 1H), 9.16 (d, J = 2.1 Hz, 2H), 5.98–5.87 (m, 1H), 5.57 (q, J = 6.8 Hz, 1H), 5.35 (dd, J = 25.1, 13.8 Hz, 2H), 1.99–1.71 (m, 2H), 1.40–1.28 (m, 10H), 0.88 (t, J = 6.6 Hz, 3H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 161.45, 148.35, 135.09, 134.02, 129.03, 121.92, 118.08, 77.85, 33.74, 31.37, 28.90, 28.75, 24.76, 22.24, 13.68. HRMS ESI [M + Na]+ calcd for C17H22N2NaO6+ 373.1370, found 373.1370. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 16.89 min, minor (S)-enantiomer tr = 13.50 min.
3.5. General Procedure of Asymmetric Addition of Ethynyltrimethylsilane to Aliphatic Aldehydes
To a stirred solution of ethynyltrimethylsilane (3922 mg, 40 mmol), (S)-BINOL (1144 mg, 4 mmol), HMPA (3584 mg, 20 mmol) in methylene chloride (120 mL), diethylzinc (40 mL, 40 mmol) was added slowly at 0 °C. After stirring for 16 h at 25 °C, Titanium(IV) isopropoxide (2842 mg, 10 mmol) was added and the stirring was continued for another 1 h at 25 °C. Then an aldehyde (10 mmol) was added and the reaction was allowed to proceed at 25 °C for another 6 h before being quenched with water (20 mL). The mixture was filtered through a celite pad. The aqueous phase was extracted with ether. The combined organic phases were washed with saturated brine, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel chromatography to get the product [34,40].
3.5.1. Synthesis of (R)-1-(Trimethylsilyl)oct-1-yn-3-ol: (R)-13a’
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 20:1) to offer (R)-13a’ (1408 mg, 71% yield, 80% ee) as colorless oil. [α] = +8.2 (c 1.1, CHCl3), lit. [56] [α] = +13.6 (c 1.1, CHCl3), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.38 (dd, J = 12.2, 6.4 Hz, 1H), 1.80 (d, J = 5.5 Hz, 1H), 1.76–1.68 (m, 2H), 1.47 (d, J = 7.3 Hz, 2H), 1.35 (dd, J = 7.1, 3.5 Hz, 4H), 0.92 (t, J = 6.7 Hz, 3H), 0.20 (s, 9H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 106.61, 88.96, 62.61, 37.35, 31.04, 24.41, 22.16, 13.59, −0.47. HRMS ESI [M + Na]+ calcd for C11H22NaOSi+ 221.1332, found 221.1332. Enantiomeric excess was determined by HPLC with a Chiralcel AD-H column (99:1 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 14.72 min, minor (S)-enantiomer tr = 13.52 min.
3.5.2. Synthesis of (R)-1-(Trimethylsilyl)non-1-yn-3-ol: (R)-13b’
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 20:1) to offer (R)-13b’ (1529 mg, 72% yield, 82% ee) as colorless oil. [α] = +2.3 (c 1.1, CHCl3), 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.38 (dd, J = 12.3, 6.5 Hz, 1H), 1.78–1.68 (m, 3H), 1.46 (d, J = 6.5 Hz, 2H), 1.38–1.27 (m, 6H), 0.92 (t, J = 6.7 Hz, 3H), 0.20 (s, 9H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 106.68, 88.89, 62.54, 37.36, 31.34, 28.52, 24.70, 22.17, 13.67, −0.48. HRMS ESI [M + Na]+ calcd for C12H24NaOSi+ 235.1489, 235.1490. Enantiomeric excess was determined by HPLC with a Chiralcel AD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 13.68 min, minor (S)-enantiomer tr = 12.42 min.
3.5.3. Synthesis of (R)-1-(Trimethylsilyl)dec-1-yn-3-ol: (R)-13c’
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 20:1) to offer (R)-13c’ (1608 mg, 71% yield, 80% ee) as colorless oil. [α] = +3.2 (c 1.1, CHCl3). 1H-NMR (300 MHz, CDCl3) δ (ppm): 4.36 (dd, J = 12.3, 6.5 Hz, 1H), 2.00 (m, 1H), 1.83–1.61 (m, 2H), 1.53–1.40 (m, 2H), 1.40–1.31 (m, 8H), 0.90 (t, J = 6.7 Hz, 3H), 0.18 (m, 9H). 13C-NMR (75 MHz, CDCl3) δ (ppm): 106.70, 88.87, 62.54, 37.36, 31.39, 28.81, 28.79, 24.74, 22.26, 13.70, −0.48. HRMS ESI [M + Na]+ calcd for C13H26NaOSi+ 249.1645, found 249.1645. Enantiomeric excess was determined by HPLC with a Chiralcel AD-H column (99:1 n-hexanes:isopropanol, 0.9 mL/min, 210 nm), major (R)-enantiomer tr = 18.90 min, minor (S)-enantiomer tr = 16.94 min.
3.6. General Procedure for the Synthesis of chIral Alkynols 12 from Propargyl Alcohols 13’
To a stirred solution of chiral alkynol (10 mmol) in methanol (20 mL), potassium carbonate (2764 mg, 20 mmol) was added slowly at 0 °C. After stirring for 20 h at 25 °C, water (20 mL) was added slowly at 0 °C. The reaction mixture was concentrated under reduced pressure. The aqueous phase was extracted with ether. The combined organic phases were washed with saturated brine solution, dried over anhydrous Na2SO4, concentrated under reduced pressure. The residue was purified by silica gel chromatography to get the product.
3.6.1. Synthesis of (R)-Oct-1-yn-3-ol: (R)-12a
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 15:1) to offer (R)-12a (1098 mg, 87% yield) as colorless oil. [α] = +16.2 (c 1.0, ethyl ether), lit. [53] [α] = +19.3 (c 1.0, ethyl ether). HRMS ESI [M + Na]+ calcd for C8H14NaO+ 149.0937, found 149.0937.
3.6.2. Synthesis of (R)-Non-1-yn-3-ol: (R)-12b
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 15:1) to offer (R)-12b (1248 mg, 89% yield) as colorless oil. [α] = +4.1 (c 2.0, CHCl3), lit. [54] [α] = +4.6 (c 1.92, CHCl3). HRMS ESI [M + Na]+ calcd for C9H16NaO+ Exact mass: 163.1093, found 163.1093.
3.6.3. Synthesis of (R)-Dec-1-yn-3-ol: (R)-12c
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 15:1) to offer (R)-12c (1373 mg, 89% yield) as colorless oil. [α] = +5.1 (c 1.1, CHCl3), lit. [55] [α] = +4.2 (c 1.1, CHCl3). HRMS ESI [M + Na]+ calcd. for C10H18NaO+ 177.1250, found 177.1250.
3.7. General Procedure for the Selective Reduction of the Chiral Alkynols 12
To a stirred solution of nickel acetate tetrahydrate (1408 mg, 8 mmol) in ethanol (20 mL) under hydrogen, sodium borohydride (303 mg, 8 mmol) in ethanol (8 mL) was added at 0 °C. After stirring for 1 h at 25 °C, ethylenediamine (481 mg, 8 mmol) was added. The reaction mixture was stirred for another 10 min before chiral alkynol (8 mmol) in ethanol (8 mL) was added slowly to the reaction mixture at 0 °C. The reaction was allowed to proceed at 25 °C under hydrogen for 6 h at 25 °C. The mixture was filtered through a celite pad, diluted with ether, and concentrated under reduced pressure. The residue was purified by silica gel chromatography to get the product.
3.7.1. Synthesis of (R)-Oct-1-en-3-ol: (R)-6a
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 10:1) to offer (R)-6a (831 mg, 81% yield) as colorless oil. [α] = −7.3 (c 1.1, CHCl3), lit. [56] [α] = −10.0 (c 1.67, CHCl3). HRMS ESI [M + Na]+ calcd. for C8H16NaO+ 151.1093, found 151.1093.
3.7.2. Synthesis of (R)-Non-1-en-3-ol: (R)-6b
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 10:1) to offer (R)-6b (944 mg, 83% yield) as colorless oil. [α] = −10.5 (c 1.1, ethanol), lit. [57] [α] = −13.4 (c 1.12, ethanol). HRMS ESI [M + Na]+ calcd. for C9H18NaO+ 165.1250, found 165.1250.
3.7.3. Synthesis of (R)-Dec-1-en-3-ol: (R)-6c
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 10:1) to offer (R)-6c (1013 mg, 81% yield) as colorless oil. [α] = −14.2 (c 1.1, CHCl3), lit. [58] [α] = −18.1 (c 1.22, CHCl3). HRMS ESI [M + Na]+ calcd. for C10H20NaO+ 179.1406, found 179.1406.
3.8. General Procedure for the Esterification Reaction and Determination of Enantiomeric Excess by HPLC
Triethylamine (909 mg, 9 mmol) and 3,5-dinitrobenzoyl chloride (1660 mg, 7.2 mmol) were added to a stirred solution of the chiral alcohol (6 mmol) in CH2Cl2 (40 mL) at −5 °C. The mixture was stirred for 5 h at 25 °C before water (10 mL) was poured into the mixture at 0 °C. The aqueous phase was extracted with ether and combined organic phases were washed with saturated brine solution, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel chromatography to get the product.
3.8.1. Synthesis of (R)-Oct-1-en-3-yl-3,5-dinitrobenzoate: (R)-21a
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-21a (1837 mg, 95% yield, 80% ee) as white solid. [α] = −14.2 (c 1.1, CH2Cl2). Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (99:1 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 17.80 min, minor (S)-enantiomer tr = 14.65 min.
3.8.2. Synthesis of (R)-Non-1-en-3-yl-3,5-dinitrobenzoate: (R)-21b
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-21b (1987 mg, 98% yield, 82% ee) as colorless oil. [α] = −15.2 (c 1.1, CH2Cl2). Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 15.76 min, minor (S)-enantiomer tr = 12.93 min.
3.8.3. Synthesis of (R)-Dec-1-en-3-yl-3,5-dinitrobenzoate: (R)-21c
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-21c (2018 mg, 96% yield, 79% ee) as white solid. [α] = −18.2 (c 1.05, CH2Cl2). Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 16.58 min, minor (S)-enantiomer tr = 13.66 min.
3.9. General Procedure for the Recrystallization to Improve Optical Purity
Dinitrobenzoates (5 mmol) were dissolved in diethyl ether at room temperature, then n-hexane was added slowly to the mixture until a white precipitate occurred. A small portion of diethyl ether was added and the white precipitate was dissolved. The mixture was cooled to −30 °C and stayed for 48 h to get a white crystal of the dinitrobenzoate.
3.9.1. Recrystallization of (R)-Oct-1-en-3-yl-3,5-dinitrobenzoate: (R)-21a
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-21a (999 mg, 62% yield, 99% ee) as white solid. [α] = −16.1 (c 1.1, CH2Cl2). HRMS ESI [M + Na]+ calcd for C15H18N2NaO6+ 345.1057, found 345.1057. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (99:1 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 18.20 min, minor (S)-enantiomer tr = 14.46 min.
3.9.2. Recrystallization of (R)-Non-1-en-3-yl-3,5-dinitrobenzoate: (R)-21b
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-21b (1076 mg, 64% yield, 99% ee) as colorless oil. [α] = −18.7 (c 1.1, CH2Cl2). HRMS ESI [M + Na]+ calcd for C16H20N2NaO6+ 359.1214, found 359.1214. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 15.60 min, minor (S)-enantiomer tr = 12.67 min.
3.9.3. Recrystallization of (R)-Dec-1-en-3-yl-3,5-dinitrobenzoate: (R)-21c
Following the general procedure, the residue was purified by silica gel chromatography (hexanes/ethyl acetate 8:1) to offer (R)-6c (1051 mg, 60% yield, 99% ee) as white solid. [α] = −21.5 (c 1.05, CH2Cl2). HRMS ESI [M + Na]+ calcd for C17H22N2NaO6+ 373.1370, found 373.1370. Enantiomeric excess was determined by HPLC with a Chiralcel OD-H column (98:2 n-hexanes:isopropanol, 1.0 mL/min, 210 nm), major (R)-enantiomer tr = 16.46 min, minor (S)-enantiomer tr = 13.50 min.
4. Conclusions
In summary, we developed a general synthetic route toward chiral matsutakeol and its analogs via the asymmetric catalytic alkynylation. A practical and efficient access was provided to prepare the (R)-matsutakeol (99% ee, total yield up to 50.2%, in three steps) and its highly enantioselective analogs, by utilizing the (S,S)-ProPhenol as chiral catalyst. In addition, the method may allow for gram-scale synthesis of (R)-matsutakeol and its analogs by using (S)-BINOL as chiral catalyst, thus facilitating their potential applications. Besides, this strategy has been proven to be practical for obtaining flavored allyl alcohols with high enantioselectivity in food analysis and screening insect attractants. Biological evaluation of target molecules is in progress and will be reported in due course.
Supplementary Materials
The following are available online: Figure S1–S43.
Acknowledgments
We thank the Special Fund for Agroscientific Research in the Public Interest (No.201503112), National Natural Science Foundation of China (No.21602043 and 31601657), the Agricultural Technology System of Henan (No.S2014-11-G03), Science and Technology Planning Project of Henan Province of China (No.142102110050) and (No.141PPTGG424), the Educational Commission of Henan Province of China (No.14A210027), Henan Key Laboratory of the Innovation and Application of Novel Pesticide, Collaborative Innovation Center of Henan Grain Crops, National Key Laboratory of Wheat and Maize Crop Science for the financial support of this study.
Author Contributions
This research was carried on by all the authors. Chao Zheng, Shichao Lu, Jia Liu, Risong Na, and Bin Yu performed the experiments; Honglian Li, Xianru Guo, Xinming Yin, and Bin Yu participated in bio-activity experiment; Jia Liu and Min Wang wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Negri, R. Polyacetylenes from terrestrial plants and fungi: Recent phytochemical and biological advances. Fitoterapia 2015, 106, 92–109. [Google Scholar] [CrossRef] [PubMed]
- Konovalov, D.A. Polyacetylene compounds of plants of the Asteraceae Family (Review). Pharm. Chem. J. 2014, 48, 613–631. [Google Scholar] [CrossRef]
- Finimundy, T.C.; Dillon, A.J.P.; Henriques, J.A.P.; Ely, M.R. A review on general nutritional compounds and pharmacological properties of the Lentinula edodes mushroom. Food Nutr. Sci. 2014, 5, 1095–1105. [Google Scholar] [CrossRef]
- Swift, K.A.D. Current Topics in Flavors and Fragrances: Towards a New Millennium of Discovery; Springer Science + Business Media B.V.: GX Dordrecht, The Netherlands, 1999; p. 235. [Google Scholar]
- Mizuno, T. Bioactive biomolecules of mushrooms: Food function and medicinal effect of mushroom fungi. Food Rev. Int. 1995, 11, 7–21. [Google Scholar] [CrossRef]
- Murahashi, S. The odor of matsutake (Armillaria matsutake Ito et Imai Agaricaceae). II. Sci. Papers Inst. Phys. Chem. Res. 1938, 34, 155–172. [Google Scholar]
- Hung, R.; Lee, S.; Bennett, J.W. The effects of low concentrations of the enantiomers of mushroom alcohol (1-octen-3-ol) on Arabidopsis thaliana. Mycology 2014, 5, 73–80. [Google Scholar] [CrossRef] [PubMed]
- McMeniman, C.J.; Corfas, R.A.; Matthews, B.J.; Ritchie, S.A.; Vosshall, L.B. Multimodal Integration of Carbon Dioxide and Other Sensory Cues Drives Mosquito Attraction to Humans. Cell 2014, 156, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Sikuljak, T.; Mazuir, F.; Arevalo, A.; Menon, A. Pesticidal Mixture Comprising a Carboxamide Compound and a Biopesticide. WO2016142456 A1, 10 March 2016. [Google Scholar]
- Combet, E.; Henderson, J.; Eastwood, D.C.; Burton, K.S. Eight-carbon volatiles in mushrooms and fungi: Properties, analysis, and biosynthesis. Mycoscience 2006, 47, 317–326. [Google Scholar] [CrossRef]
- Helmchen, G.; Ihrig, K.; Schindler, H. EPC-syntheses via asymmetric Diels-Alder reactions/retro Diels-Alder reactions. I. (R)- and (S)-Matsutake alcohol, (R)- and (S)-sarcomycin methyl ester. Tetrahedron Lett. 1987, 28, 183–186. [Google Scholar] [CrossRef]
- Takano, S.; Yanase, M.; Takahashi, M.; Ogasawara, K. Enantiodivergent synthesis of both enantiomers of sulcatol and matsutake alcohol from (R)-epichlorohydrin. Chem. Lett. 1987, 16, 2017–2020. [Google Scholar] [CrossRef]
- Kitamura, M.; Kasahara, I.; Manabe, K.; Noyori, R.; Takaya, H. Kinetic resolution of racemic allylic alcohols by BINAP-ruthenium(II) catalyzed hydrogenation. J. Org. Chem. 1988, 53, 708–710. [Google Scholar] [CrossRef]
- Oppolzer, W.; Radinov, R.N. Enantioselective synthesis of sec-allyl alcohols by catalytic asymmetric addition of divinylzinc to aldehydes. Tetrahedron Lett. 1988, 29, 5645–5648. [Google Scholar] [CrossRef]
- Kusuda, S.; Ueno, Y.; Toru, T. Vinyl anion equivalent V. Asymmetric synthesis of allylic alcohols using chiral 2-(trialkylsilyl)ethyl sulfoxides. Tetrahedron 1994, 50, 1045–1062. [Google Scholar] [CrossRef]
- Bisogno, F.R.; Rioz-Martinez, A.; Rodriguez, C.; Lavandera, I.; de Gonzalo, G.; Torres Pazmino, D.E.; Fraaije, M.W.; Gotor, V. Oxidoreductases Working Together: Concurrent Obtaining of Valuable Derivatives by Employing the PIKAT Method. ChemCatChem 2010, 2, 946–949. [Google Scholar] [CrossRef]
- Rej, R.K.; Nanda, S. Chemoenzymic Asymmetric Total Synthesis of Nonanolide (Z)-Cytospolides D, E and Their Stereoisomers. Eur. J. Org. Chem. 2014, 2014, 860–871. [Google Scholar] [CrossRef]
- Rej, R.K.; Kumar, R.; Nanda, S. Asymmetric synthesis of cytospolide C and cytospolide D through successful exploration of stereoselective Julia-Kocienski olefination and Suzuki reaction followed by macrolactonization. Tetrahedron 2015, 71, 3185–3194. [Google Scholar] [CrossRef]
- Lee, W.H.; Bae, I.H.; Kim, B.M.; Seu, Y.-B. New Synthetic Approach to Optically Activate Matsutakeol, the Major Flavor Component of Tricholoma matsutake, from L-Tartaric Acid. Bull. Korean Chem. Soc. 2016, 37, 1910–1911. [Google Scholar] [CrossRef]
- Huang, J.; Wei, S.; Wang, L.; Zhang, C.; Li, S.; Liu, P.; Du, X.; Wang, Q. Highly enantioselective catalytic methyl propiolate addition to both aromatic and aliphatic aldehydes. Tetrahedron Asymmetry 2016, 27, 428–435. [Google Scholar] [CrossRef]
- Funes-Maldonado, M.; Sieng, B.; Amedjkouh, M. Enabling Asymmetric Alkynylation of Azaaryl Aldehydes with Soai Autocatalyst. Eur. J. Org. Chem. 2015, 2015, 4081–4086. [Google Scholar] [CrossRef]
- Yang, Y.-Q.; Li, S.-N.; Zhong, J.-C.; Zhou, Y.; Zeng, H.-Z.; Duan, H.-J.; Bian, Q.-H.; Wang, M. Total synthesis of each enantiomer of falcarinol and panaxjapyne A via asymmetric catalytic alkynylation of an aldehyde. Tetrahedron Asymmetry 2015, 26, 361–366. [Google Scholar] [CrossRef]
- Trost, B.M.; Quintard, A. Asymmetric Catalytic Alkynylation of Acetaldehyde: Application to the Synthesis of (+)-Tetrahydropyrenophorol. Angew. Chem. Int. Ed. 2012, 51, 6704–6708. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.M.; Burns, A.C.; Bartlett, M.J.; Tautz, T.; Weiss, A.H. Thionium Ion Initiated Medium-Sized Ring Formation: The Total Synthesis of Asteriscunolide D. J. Am. Chem. Soc. 2012, 134, 1474–1477. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.M.; Bartlett, M.J.; Weiss, A.H.; von Wangelin, A.J.; Chan, V.S. Development of Zn-ProPhenol-Catalyzed Asymmetric Alkyne Addition: Synthesis of Chiral Propargylic Alcohols. Chem. Eur. J. 2012, 18, 16498–16509. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.M.; Bartlett, M.J. Transition-Metal-Catalyzed Synthesis of Aspergillide B: An Alkyne Addition Strategy. Org. Lett. 2012, 14, 1322–1325. [Google Scholar] [CrossRef] [PubMed]
- Na, R.; Wang, B.; Liu, H.; Bian, Q.; Zhong, J.; Wang, M.; Guo, H. Synthesis and application of novel dithiane alcohol ligand based on chiral cyclopropane-backbone for asymmetric ethylation of aromatic aldehydes. Lett. Org. Chem. 2012, 9, 622–627. [Google Scholar] [CrossRef]
- Chen, X.; Chen, W.; Wang, L.; Yu, X.-Q.; Huang, D.-S.; Pu, L. Synthesis of a C1 symmetric BINOL-terpyridine ligand and highly enantioselective methyl propiolate addition to aromatic aldehydes. Tetrahedron 2010, 66, 1990–1993. [Google Scholar] [CrossRef]
- Ito, J.-I.; Asai, R.; Nishiyama, H. Asymmetric Direct Alkynylation Catalyzed by Chiral Ru-Bis(oxazolinyl)phenyl Complexes. Org. Lett. 2010, 12, 3860–3862. [Google Scholar] [CrossRef] [PubMed]
- Turlington, M.; DeBerardinis, A.M.; Pu, L. Highly Enantioselective Catalytic Alkyl Propiolate Addition to Aliphatic Aldehydes. Org. Lett. 2009, 11, 2441–2444. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Zhao, Q.; Li, A.N.; Ren, F.; Yang, F.; Wang, R. Enantioselective synthesis of Anomala osakana pheromone and Janus integer pheromone: A flexible approach to chiral γ-butyrolactones. Org. Biomol. Chem. 2009, 7, 3663–3665. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.; Wang, Q.; Lin, L.; Liu, X.; Jiang, X.; Zhao, Q.; Hu, G.; Wang, R. Highly enantioselective addition of terminal alkynes to aldehydes catalyzed by a new chiral β-sulfonamide alcohol/Ti(OiPr)4/Et2Zn/R3N catalyst system. Chirality 2009, 21, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.-C.; Hou, S.-C.; Bian, Q.-H.; Yin, M.-M.; Na, R.-S.; Zheng, B.; Li, Z.-Y.; Liu, S.-Z.; Wang, M. Highly enantioselective zinc/amino alcohol-catalyzed alkynylation of aldehydes. Chem. Eur. J. 2009, 15, 3069–3071. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Turlington, M.; Yu, X.-Q.; Pu, L. 3,3′-Anisyl-Substituted BINOL, H4BINOL, and H8BINOL Ligands: Asymmetric Synthesis of Diverse Propargylic Alcohols and Their Ring-Closing Metathesis to Chiral Cycloalkenes. J. Org. Chem. 2009, 74, 8681–8689. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wu, N.; Ding, Z.; Wang, T.; Mao, J.; Zhang, Y. L-Proline-derived tertiary amino alcohol as a new chiral ligand for enantioselective alkynylation of aldehydes. Tetrahedron Lett. 2009, 50, 926–929. [Google Scholar] [CrossRef]
- Yang, F.; Xi, P.; Yang, L.; Lan, J.; Xie, R.; You, J. Facile, Mild, and Highly Enantioselective Alkynylzinc Addition to Aromatic Aldehydes by BINOL/N-Methylimidazole Dual Catalysis. J. Org. Chem. 2007, 72, 5457–5460. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Jiang, X.; Liu, W.; Qiu, L.; Xu, Z.; Xu, J.; Chan, A.S.C.; Wang, R. Highly Enantioselective Synthesis of γ-Hydroxy-α,β-acetylenic Esters Catalyzed by a β-Sulfonamide Alcohol. Org. Lett. 2007, 9, 2329–2332. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.M.; Weiss, A.H. Catalytic Enantioselective Synthesis of Adociacetylene B. Org. Lett. 2006, 8, 4461–4464. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.M.; Weiss, A.H.; von Wangelin, A.J. Dinuclear Zn-Catalyzed Asymmetric Alkynylation of Unsaturated Aldehydes. J. Am. Chem. Soc. 2006, 128, 8–9. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Wang, Q.; Yu, X.-Q.; Xie, R.-G.; Pu, L. Highly enantioselective synthesis of γ-hydroxy-α,β-acetylenic esters by asymmetric alkyne addition to aldehydes. Angew. Chem. Int. Ed. 2006, 45, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Wolf, C.; Liu, S. Bisoxazolidine-catalyzed enantioselective alkynylation of aldehydes. J. Am. Chem. Soc. 2006, 128, 10996–10997. [Google Scholar] [CrossRef] [PubMed]
- Emmerson, D.P.G.; Hems, W.P.; Davis, B.G. Carbohydrate-Derived Amino-Alcohol Ligands for Asymmetric Alkynylation of Aldehydes. Org. Lett. 2006, 8, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Cozzi, P.G.; Rudolph, J.; Bolm, C.; Norrby, P.-O.; Tomasini, C. Me2Zn-Mediated Addition of Acetylenes to Aldehydes and Ketones. J. Org. Chem. 2005, 70, 5733–5736. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, M.; Yamada, K.-i.; Tomioka, K. Catalytic asymmetric addition of terminal alkynes to aldehydes mediated by (1R,2R)-2-(dimethylamino)-1,2-diphenylethanol. Adv. Synth. Catal. 2005, 347, 1649–1652. [Google Scholar] [CrossRef]
- Jiang, B.; Si, Y.-G. The first highly enantioselective alkynylation of chloral: A practical and efficient pathway to chiral trichloromethyl propargyl alcohols. Adv. Synth. Catal. 2004, 346, 669–674. [Google Scholar] [CrossRef]
- Dahmen, S. Enantioselective Alkynylation of Aldehydes Catalyzed by [2.2]Paracyclophane-Based Ligands. Org. Lett. 2004, 6, 2113–2116. [Google Scholar] [CrossRef] [PubMed]
- Stefani, H.A.; Menezes, P.H.; Costa, I.M.; Silva, D.O.; Petragnani, N. Use of chiral sulfoxide in the asymmetric synthesis of (+)-virol C. Synlett 2002, 8, 1335–1337. [Google Scholar] [CrossRef]
- Jiang, B.; Chen, Z.; Xiong, W. Highly enantioselective alkynylation of aldehydes catalyzed by a readily available chiral amino alcohol-based ligand. Chem. Commun. (Cambridge, UK) 2002, 14, 1524–1525. [Google Scholar] [CrossRef]
- Rotticci, D.; Haeffner, F.; Orrenius, C.; Norin, T.; Hult, K. Molecular recognition of sec-alcohol enantiomers by Candida antarctica lipase B. J. Mol. Catal. B Enzym. 1998, 5, 267–272. [Google Scholar] [CrossRef]
- Liu, J.; Li, H.-L.; Guo, X.-R.; Zhou, L.; Wang, Y.; Duan, Y.-N.; Wang, M.-Z.; Na, R.-S.; Yu, B. A general strategy toward the total synthesis of C17 polyacetylenes virols A and C. Tetrahedron 2016, 72, 6603–6610. [Google Scholar] [CrossRef]
- Morandi, S.; Palate, F.; Benvenuti, S.; Prati, F. Total synthesis of a dienynone from Echinacea pallida. Tetrahedron 2008, 64, 6324–6328. [Google Scholar] [CrossRef]
- Zheng, M.; Wu, F.; Chen, K.; Zhu, S. Styrene as 4π-Component in Zn (II)-Catalyzed Intermolecular Diels–Alder/Ene Tandem Reaction. Org. Lett. 2016, 18, 3554–3557. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.S.; Elliott, R.; Elliott, J.D. Asymmetric synthesis via acetal templates. 4. Reactions with silylacetylenic compounds. Formation of chiral propargylic alcohols. J. Am. Chem. Soc. 1983, 105, 2904–2905. [Google Scholar] [CrossRef]
- Jiang, X.; Fu, C.; Ma, S. Highly stereoselective iodolactonization of 4,5-allenoic acids—An efficient synthesis of 5-(1′-iodo-1′(Z)-alkenyl)-4,5-dihydro-2(3H)-furanones. Chemistry 2008, 14, 9656–9664. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Chen, J.; Guo, L.-C.; Ye, J.-L.; Ruan, Y.-P.; Huang, P.-Q. Asymmetric Total Synthesis of (−)-Awajanomycin. Org. Lett. 2009, 11, 5242–5245. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.; Nakayama, J.; Toru, T. Asymmetric Reduction of α-(Trimethylsilyl)methyl-β-ketosulfoxide with DIBAL under Basic Conditions. J. Org. Chem. 2003, 68, 5766–5768. [Google Scholar] [CrossRef] [PubMed]
- Bessodes, M.; Abushanab, E.; Antonakis, K. Enantiospecific synthesis of the immunopotentiators erythro-9-(2-hydroxy-3-nonyl)hypoxanthines and the threo-diastereomers. Tetrahedron Lett. 1984, 25, 5899–5902. [Google Scholar] [CrossRef]
- Gartner, M.; Mader, S.; Seehafer, K.; Helmchen, G. Enantio- and Regioselective Iridium-Catalyzed Allylic Hydroxylation. J. Am. Chem. Soc. 2011, 133, 2072–2075. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of (R)-Matsutakeol and compounds 13a–i are available from the authors.
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).