Abstract
Structural studies of ternary complexes of CD1d/glycosyl ceramides/iNKT cells and CD1d/sulfatide/sulfatide reactive Type II NKT cells have shown how the polar moieties on the glycolipids interact with both the antigen presenting protein (CD1d) and the T cell receptors. However, these structures alone do not reveal the relative importance of these interactions. This study focuses on the synthesis of the previously unknown 2''-deoxy-β-galactosyl ceramide 2. This glycolipid is also evaluated for its ability to stimulate iNKT cells and sulfatide-reactive Type II NKT cells.
1. Introduction
α-Galactosyl ceramide (α-GalCer) and related compounds have garnered increasing attention in the last decade and a half because of their ability to elicit immune responses via CD1d mediated activation of natural killer T (NKT) cells [1,2,3,4,5]. NKT cells are a heterogeneous group of T lymphocytes that express a T cell antigen receptor (TCR) and that perform functions related to both innate and adaptive immunity, including defense against microbes, maintenance of immune tolerance and anti-tumor responses. While most T lymphocytes respond to peptides bound to antigen presenting molecules encoded in the Major Histocompatibility Complex (MHC), a defining characteristic of NKT cells is their recognition of lipid/glycolipid antigens presented by CD1d, which is related to the MHC antigen presenting proteins. CD1d-reactive NKT cells are split into two classes, Type I and Type II. Type I NKT (or iNKT) cells are characterized by the expression of an invariant (i) TCRα chain paired with a limited number of TCRβ chains, and these cells have been extensively studied [6,7,8,9,10,11]. They recognize various lipid containing antigens, particularly glycosphingolipids. On the other hand, the Type II NKT cells have diverse TCRs and are not as well characterized, although recent attention has focused on the role of sulfatide-reactive cells that belong to this group [12,13,14,15,16,17].
The understanding of the role of the sugar and the ceramide in influencing iNKT cell responses has been greatly enhanced by both structure activity relationship (SAR) studies [7,9,10,18,19] and by the availability of an ever increasing number of binary (glycolipid/CD1d) [20,21,22] and ternary (NKT cell TCR/glycolipid/CD1d) crystal structures [23,24,25,26,27,28]. Again, SAR and structural data [29,30,31] for Type II NKT cells lag those for Type I cells. Structural studies show that the lipid moiety of the glycolipids is buried in two pockets of CD1d and that it is predominantly the sugar that interacts with the TCR, which recognizes a composite of the sugar and the surface of the CD1d protein. Although the hydrogen bonding interactions of both CD1d and the NKT cell TCR with the CD1d-exposed sugar moiety are well-characterized, access to deoxysugar analogs has provided a nuanced understanding of the relative influence of each carbohydrate OH and the possibility for fine specificity in interactions [26]. Ternary Type I and Type II structures show that the OH on C2'' has hydrogen bonding interaction with both CD1d and the NKT cell TCR for α-galactosyl ceramides (α-GalCers) and sulfatides (which have β-linked sugars). Indeed, 2''-deoxy-α-GalCer 1 (Figure 1) does not activate Type I NKT cells [32]. Interestingly, however, both β-glucosyl ceramide (β-GluCer) [33] and β-mannosyl ceramide (β-ManCer) [34] have been recently reported to stimulate Type I NKT cells. To our knowledge, no 2''-deoxy-β-glycosyl ceramide has been prepared or evaluated. Herein we report the preparation of 2''-deoxy-β-GalCer 2 as a tool for further developing an understanding of NKT cell activation.
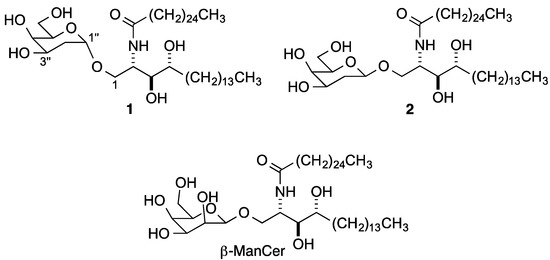
Figure 1.
2''-Deoxy galactosyl ceramides 1 and 2 and β-ManCer.
The synthesis of 2''-deoxy-β-glycosides is challenging. If glycosylations targeting such compounds are conducted on already deoxygenated sugar donors, which lack the functionality needed to exploit anchimeric assistance, the stereoselectivity can be low. Moreover, the glycosidic bond is more labile due to the lack of an electron withdrawing C2'' substituent [35]. The most common approach involves the use of a heteroatom substituent at C2, followed by its reductive removal subsequent to glycosylation [36,37,38,39,40,41,42,43,44,45]. Other methods include the use of α-glycosyl phosphites [46,47], SN2 reactions of α-glycosyl halides [48,49,50], Pd-catalyzed glycosylation reactions [51], the use of glycosyl imidates under oxidative conditions [52], and anomeric O-alkylation/arylation [53]. Although many of these approaches provided good yields and stereoselectivities with some acceptors, our biggest concern centered on the well-known decreased reactivity of either ceramide or sphingoid base acceptors in comparison to coupling partners used in the majority of these studies. Danishefsky and coworkers had used azidosphingosine 3 in a reaction with 1α,2α-oxirane 4 (Figure 2) derived from galactal, and the glycosylation gave 60% isolated yield of the β-anomer [54]. Although there was not a subsequent deoxygenation in this case, the group has used tin mediated deoxygenations in many other cases. However, our previous experience with difficult purifications in tin-mediated deoxygenations of glycolipids led us to consider an alternative approach.
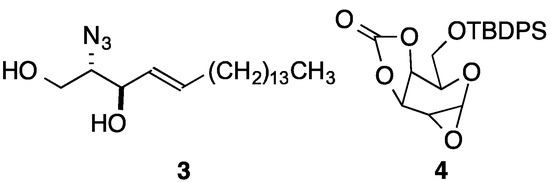
Figure 2.
β-Glycosylation partners.
We elected to employ Magnusson’s NIS promoted glycosylation [55] in combination with Danishefsky’s observation that beta-selectivity in 1,2-anhydrosugars was sometimes enhanced with a carbonate (as compared to benzyl groups or an isopropylidene) [56]. Most approaches to glycosyl ceramides involve coupling of a sugar and ceramide or of a sugar and sphingoid base, followed by acylation. The latter is the pathway we chose (Scheme 1).
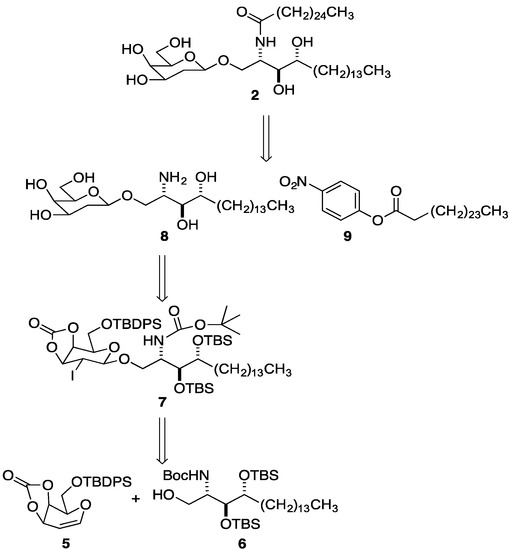
Scheme 1.
Strategy for the synthesis of 2''-deoxy-β-GalCer 2.
Thus, the strategy was to glycosylate via iodoetherification of galactal 5 with phytosphingosine alcohol 6. Our initial plan was to do a multi-stage deprotection/removal of the iodide of 7 to provide fully deprotected glycosylated phytosphingsine 8. Final acylation with activated cerotic acid ester 9 would achieve the synthesis of 2''-deoxy-β-GalCer 2.
2. Results and Discussion
Glycosyl donor 5 was prepared in two steps from d-galactal as described by Danishefsky and coworkers [54]. Glycosyl acceptor 6 was synthesized in three steps from phytosphingosine as previously reported [57]. With donor 5 (1 equiv.) and acceptor 6 (2 equiv.) in hand, NIS-induced glycosylation in CH2Cl2 at room temperature was attempted. The reaction was stirred overnight, and TLC analysis showed consumption of 5. However, separation of the desired product 7 from the excess of acceptor 6 using column chromatography was extremely difficult, resulting in a poor isolated yield of 7. A reaction with acceptor 6 as the limiting reactant was sluggish, not going to completion even after 7 days.
In order to have an acceptor that was more easily separated from the product, NIS-induced glycosylation reaction using acceptor 10 was tried. The reaction was complete in 48 h, and β-glycosylated phytosphingosine 11 was isolated in 48% yield. Compound 11 was then treated with catalytic NaOMe in MeOH, followed by deiodohydrogenation using palladium on carbon (10 wt %) in one pot, to give compound 12 in 67% yield (Scheme 2). However due to the absence of a functionalizable group at C2, cleavage of the anomeric bond was observed in the subsequent Boc deprotection reaction using HCl (4 M in dioxane). Hence the strategy needed to be modified.
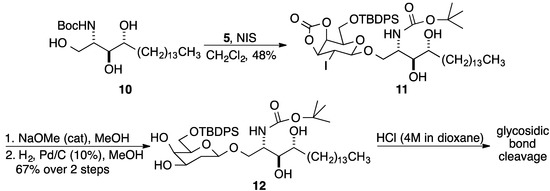
Scheme 2.
Initial attempt to prepare 2.
Glycosylated product 11 was subjected to a one pot, two-stage deprotection (Scheme 3). First, the cyclic carbonate group was removed using cat NaOMe in MeOH; removal of the TBPDS group using TBAF (1.0 M solution in THF), then gave 13 in 56% yield over two steps. Boc cleavage, followed by N-acylation using the para-nitrophenol ester 9 of cerotic acid in pyridine, afforded 14 in 42% yield. Deiodohydrogenation using palladium on carbon (10 wt %) concluded the synthesis of 2. A substantial quantity of ceramide 15 was recovered due to the cleavage of the anomeric bond. Nevertheless, there was sufficient material to evaluate 2''-deoxy-β-GalCer 2 for its ability to stimulate both Type I and sulfatide-active Type II NKT cells.
2''-Deoxy-β-GalCer 2 was assessed for its ability to stimulate iNKT cells using well-characterized immortalized cells or hybridomas. Hybridoma 1.2 is a well characterized iNKT cell hybridoma [58] that has the common Vα14 chain characteristic of this population, paired with Vβ8.2, the most common TCR β chain expressed by iNKT cells. The type II NKT cell hybridomas XV19 (19) and VIII24 (24) were also tested. Hybridoma 19 is reactive to sulfatide, lyso-sulfatide and other antigens [14,59], and it has a TCR containing Vα1 and Vβ16. Hybridoma 24 has a TCR containing Vα3 and Vβ9 and it is CD1d autoreactive for antigens that have not been characterized.
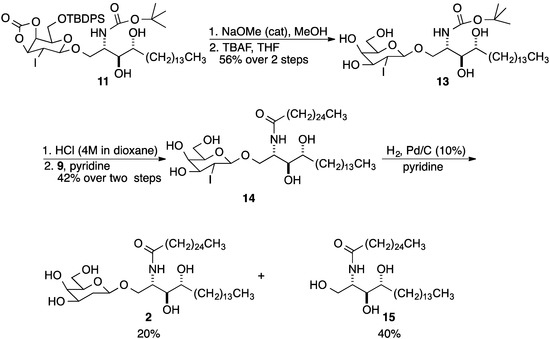
Scheme 3.
Preparation of 2.
To assay for antigenic potency, a cell-free antigen presentation assay was employed [60]. The assay measures the secretion of interleukin 2 (IL-2) by T cell hybridomas that have been cultured in microwells coated with complexes of CD1d with glycolipids. IL-2 production correlates directly with the strength of TCR stimulation, and because of the high density of lipid-CD1d complexes that can be achieved on the coated plate, this test provides the most sensitive assay for measuring iNKT cell responses.
Figure 3 shows that hybridoma 1.2 weakly reacted to CD1d coated plates loaded with 2''-deoxy-β-GalCer 2. There is a background reactivity of 127 units/mL of IL-2 released to CD1d molecules obtained from insect cells, which are loaded primarily with phosphatidyl choline [61], but this is more than doubled when the cells were cultured with CD1d molecules loaded with 100 ng/mL of 2''-deoxy-β-GalCer 2. By contrast, α-GalCer stimulated approximately 10-fold more IL-2 release at a similar concentration. In some assays, however, this 2''-deoxy-β-GalCer 2 reactivity could not be detected, and a weak and somewhat inconsistent stimulation was observed when using antigen presenting cells expressing very high amounts of CD1d or when 1.4, a different iNKT cell hybridoma, was tested (data not shown). The pattern was very different with the Type II NKT cell hybridomas (19 and 24), which were completely unreactive to plates coated with CD1d that was not loaded, or loaded with 2''-deoxy-β-GalCer 2.
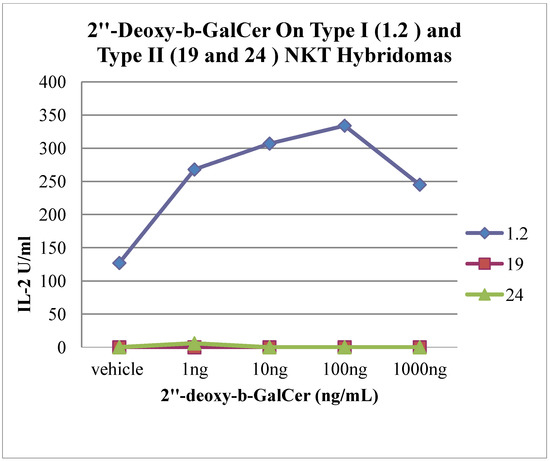
Figure 3.
Type I and Type II NKT cell hybridoma responses to 2''-deoxy-β-GalCer 2.
3. Experimental
3.1. General Information
Melting points were determined in open Pyrex capillary tubes and are uncorrected. Specific rotations [α]D were recorded on a JASCO DIP-1000 polarimeter using the sodium D-line as a source, and the concentration (c) is expressed in units of g per 100 mL. IR spectra were recorded on a FT-IR spectrometer using KBr. NMR spectra were recorded on a Bruker Avance DRX-400 (400 MHz 1H, 100 MHz 13C), Bruker Avance (500 MHz 1H, 125 MHz 13C), or Bruker Avance (300 MHz 1H, 75 MHz 13C) spectrometer. The 1H and 13C chemical shifts are reported in parts per million (ppm) from TMS. The following abbreviations are used for peak multiplicities s (singlet); d (doublet); t (triplet); q (quartet); m (multiplet); dd (doublet of doublets); br s (broad singlet). Coupling constants, J, are reported in hertz (Hz). Low-resolution mass spectra were obtained on a HP 5970 series GC-MSD system and are reported in units of mass/charge. HRMS were obtained on a JMS-AX505HA spectrometer at the University of Notre Dame.
p-Nitrophenyl hexacosanoate (9). A mixture of hexacosanoic acid (1.0 g, 2.5 mmol) and thionyl chloride (12.5 mL) was heated at 90 °C under N2 for 12 h. The excess thionyl chloride was distilled off, and traces were removed under high vacuum. The residue was dissolved in dry THF (25 mL), and the solution was added drop-wise to a stirred solution of 4-nitrophenol (0.76 g, 5.5 mmol) and a catalytic amount of DMAP in freshly distilled pyridine (25 mL) under N2 at −5 °C. After 1 h at −5 °C the cooling bath was removed, and the mixture was stirred for 12 h at rt. The reaction mixture was then concentrated, and the residue was taken up in CH2Cl2 and preloaded on silica gel. Purification by flash chromatography on silica gel (petroleum ether/EtOAc 98:2 to 95:5) provided p-nitrophenyl hexacosanoate (9) as a white powder (0.80 g, 61%): mp 87–88 °C; IR (KBr) 2916, 2849, 1743, 1620, 1593, 1535, 1472, 1348, 1206, 1138 cm−1; 1H-NMR (CDCl3) δ 8.29 (d, J = 9.1 Hz, 2H), 7.30 (d, J = 9.3 Hz, 2H), 2.62 (t, J = 7.5 Hz, 2H), 1.78 (m, 2H), 1.28 (m, 44H), 0.90 (t, J = 6.8 Hz, 3H); 13C-NMR (CDCl3) δ 171.3, 155.5, 145.2, 125.2, 122.4, 34.3, 31.9, 29.7, 29.7, 29.6, 29.6, 29.4, 29.4, 29.4, 29.2, 29.0, 24.7, 22.7, 14.1; MS (EI) m/z: 279, 167, 149, 104, 70, 57; Anal. Calcd for C32H55NO4: C, 74.23; H, 10.71; N, 2.71, found: C, 74.22; H, 10.46; N, 2.64.
(2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-2-iodo-3,4-oxacarbonyl-β-d-galactopyranosyl]-1,3,4-octadecantriol (11). 1,5-Anhydro-6-O-(tert-butyl-diphenylsilyl)-2-deoxy-d-lyxohex-1-enopyranose-3,4-carbonate (5) [54] (0.29 g, 0.71 mmol) and N-(tert-butoxycarbonyl) phytosphingosine (0.59 g, 1.4 mmol) (10) [62] were added to dry CH2Cl2 (12 mL) under N2 in a foil covered flask. N-Iodosuccinimide (0.35 g, 1.6 mmol) was added to the mixture, which was then stirred at rt for 48 h. Then, 10% aq. Na2S2O3 (20 mL) was added, and the mixture was stirred vigorously for 5 min. The mixture was then diluted with EtOAc (100 mL) and transferred to a separatory funnel. The aqueous layer was discarded, and the organic layer was washed with 10% aq. Na2S2O3 (10 mL) and brine (10 mL) and dried (MgSO4). The solvents were removed, and the residue was then subjected to gravity column chromatography (petroleum ether/EtOAc 80:20), giving (2S,3R,4R)-2-(N-tert-butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-2-iodo-3,4-oxacarbonyl-β-d-galactopyranosyl]-1,3,4-octadecantriol (11) as a white solid (0.32 g, 48%): mp 55–57 °C; [α]25D −4.49 (c 1.5, CH2Cl2); IR (KBr) 3419 (br), 3072, 2926, 2854, 1818, 1699, 1174, 1114, 702 cm−1; 1H-NMR (CDCl3) δ 7.65 (m, 4H), 7.45 (m, 6H), 5.17 (dd, J = 7.4, 5.6 Hz, 1H), 5.14 (m, 1H), 4.80 (dd, J = 9.0, 9.0 Hz, 2H), 4.16 (m, 2H), 4.03 (dd, J = 5.8, 5.8 Hz, 1H), 3.91 (m, 2H), 3.82 (br s, 1H), 3.59 (m, 3H), 2.85 (br s, 1H), 2.14 (br s, 1H), 1.65 (m, 1H) 1.43 (s, 10H), 1.26 (s, 24H), 1.08 (s, 9H), 0.89 (t, J = 6.7 Hz, 3H); 13C-NMR (CDCl3) 156.2, 153.4, 135.5, 135.5, 132.8, 132.5 130.1, 128.1, 101.5, 80.1, 79.7, 74.6, 73.4, 73.2, 71.8, 70.0, 62.2, 51.5, 32.5, 32.1, 29.8, 29.5, 28.6, 27.0, 26.1, 22.9, 19.4, 14.3; HRMS (ESI) m/z calcd for C46H73INO10Si [M+H]+ 954.4043, found 954.4051.
(2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-β-d-galactopyranosyl]-1,3,4-octadecantriol (12). (2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-2-iodo-3,4-oxacarbonyl-β-d-galactopyranosyl]-1,3,4-octadecan-triol (11) (0.21 g, 0.22 mmol) was dissolved in MeOH (1.1 mL) at rt under N2, followed by the addition of a catalytic amount of NaOMe (0.5 M in MeOH) (0.030 mL, 0.013 mmol). The reaction mixture was stirred at rt for 0.5 h. The reaction mixture was concentrated, and the residue was dissolved in MeOH/EtOAc (5:1, 6.5 mL), followed by the addition of Et3N (0.04 mL) and Pd/C (10%, 86 mg). The mixture was stirred vigorously overnight under H2 (1 atm) and then filtered through celite and concentrated. The crude reaction mixture was purified by gravity column chromatography (CH2Cl2/MeOH 98:2), giving (2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-2-iodo-3,4-oxacarbonyl-β-d-galactopyranosyl]-1,3,4-octadecantriol (12) as a clear oil (0.12 g, 67%): IR (KBr) 3419 (br), 2926, 1699, 1174, 1114 cm−1; 1H-NMR (CDCl3) δ 7.63 (m, 4H), 7.37 (m, 6H), 5.20 (d, J = 8.6 Hz, 1H), 4.33 (d, J = 9.2 Hz, 1H), 4.14 (d, J = 8.4 Hz, 1H), 3.82 (m, 4H), 3.57 (m, 5H), 3.35 (dd, J = 5.7, 5.7 Hz, 1H), 3.13 (dd, J = 14.6, 7.3 Hz, 1H), 3.07 (m, 1H), 2.60 (br s, 1H), 1.95 (m, 1H), 1.65 (m, 2H), 1.42 (m, 3H), 1.39 (s, 9H), 1.22 (br s, 22H), 1.01 (s, 9H), 0.84 (t, J = 6.6 Hz, 3H); 13C-NMR (CDCl3) δ 155.9, 135.8, 135.7, 133.2, 133.2, 130.1, 128.1, 100.7, 79.9, 75.2, 74.7, 73.0, 69.1, 68.7, 67.6, 63.3, 53.5, 51.4, 46.5, 35.2, 32.3, 32.1, 29.9, 29.6, 28.6, 27.0, 26.2, 22.9, 19.4, 14.3, 8.8; HRMS (ESI) m/z calcd for C45H76NO9Si [M+H]+ 802.5284, found 802.5300.
(2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-β-d-galactopyranosyl]-1,3,4-octadecantriol (13). (2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-2-iodo-3,4-oxacarbonyl-β-d-galactopyranosyl]-1,3,4-octadecantriol (11) (0.10 g, 0.1 mmol) was dissolved in MeOH (0.5 mL) at rt under N2, followed by the addition of a catalytic amount of NaOMe (0.5 M in MeOH) (0.01 mL, 0.006 mmol). The reaction mixture was stirred at rt for 0.5 h. The reaction mixture was concentrated, and the residue was dissolved in dry THF (0.5 mL). The reaction mixture was cooled to 0 °C under N2, and TBAF (1.0 M, THF) (0.2 mL, 0.2 mmol) was added dropwise. The cooling bath was removed, and the reaction mixture was allowed to stir at rt for 1 h. The mixture was then concentrated and purified by gravity column chromatography (CH2Cl2/MeOH 95:5) giving (2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[2-deoxy-2-iodo-β-d-galactopyranosyl]-1,3,4-octa-decantriol (13) as a white solid (0.040 g, 56%): mp 140–142 °C; [α]25D −26.1 (c 0.64, CH2Cl2/MeOH, 90:10); IR (KBr) 3430, 2924, 2854, 1716, 1698, 1685 cm-1; 1H-NMR (CDCl3) δ 5.49 (d, J = 9.7 Hz, 1H), 4.61 (m, 2H), 4.40 (m, 2H), 4.26 (m, 1H), 4.19 (br s, 1H), 4.09 (dd, J = 9.7, 9.7 Hz, 1H), 3.93–3.74 (m, 8H), 3.64 (br s, 1H), 3.54 (m, 1H), 2.57 (br s, 1H), 1.54 (m, 2H), 1.46 (br s, 10H), 1.28 (s, 22H), 0.90 (t, J = 6.0 Hz, 3H); 13C-NMR (CDCl3) 156.0, 104.1, 80.1, 77.4, 75.5, 75.4, 74.0, 73.1, 69.7, 61.9, 51.2, 35.5, 32.2, 30.0, 30.0, 29.9, 29.6, 28.7, 26.5, 22.9, 14.3; HRMS (ESI) m/z calcd for C29H56INNaO9 [M+Na]+ 712.2892, found 712.2914.
(2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-β-d-galactopyranosyl]-1,3,4-octadecantriol (14). (2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[2-deoxy-2-iodo-β-d-galactopyranosyl]-1,3,4-octa-decantriol (13) (0.037 g, 0.054 mmol) was dissolved in dry 1,4-dioxane (0.5 mL) at rt under N2. HCl (4 M, dioxane) (0.5 mL) was added, and the reaction mixture was stirred at rt for 40 min. The reaction mixture was then brought to pH 8 by adding saturated aqueous NaHCO3 and was stirred for a further 30 min. A solid appeared and was subsequently filtered, dissolved in pyridine, then concentrated. The crude solid was dissolved in a minimum quantity of dry pyridine (~1.0 mL) at rt under N2. The p-nitrophenol ester (9) of cerotic acid (0.026 g, 0.050 mmol), dissolved in a minimum quantity of pyridine (~0.5 mL), was added to the reaction mixture, and the solution was stirred for 20 h at rt under N2. The reaction mixture was concentrated, and the residue was purified by gravity column chromatography (CH2Cl2/MeOH 93:7) giving (2S,3R,4R)-1-O-[2-deoxy-2-iodo-β-d-galactopyranosyl]-1,3,4-octadecantriol (14) as a white solid (0.20 g, 42%): mp 120 °C; [α]25D 80.2 (c 0.05, pyridine); IR (KBr) 3430, 2957, 2851, 1651, 1114, 1067 cm−1; 1H-NMR (pyridine-d5) δ 8.22 (d, J = 8.1 Hz, 1H), 6.05 (br s, 5H), 5.12 (m, 2H), 4.82 (m, 2H), 4.50−4.35 (m, 5H), 4.23 (m, 2H), 4.09 (dd, J = 6.9, 6.9 Hz, 1H), 2.50 (m, 2H), 2.24 (m, 1H), 1.95−1.85 (m, 4H), 1.71 (m, 1H), 1.30 (m, 66H), 0.87 (m, 6H); 13C-NMR (pyridine-d5) 173.0, 104.4, 77.1, 73.3, 75.1, 72.8, 70.5, 61.6, 51.9, 39.7, 36.7, 33.0, 31.8, 30.1, 29.9, 29.6, 29.3, 26.4, 26.1, 22.7, 14.0; HRMS (ESI) m/z calcd for C50H99INO8 [M+H]+ 968.6410, found 968.6434.
(2S,3R,4R)-2-(N-tert-Butoxycarbonyl)amino-1-O-[6-O-(tert-butyldiphenylsilyl)-2-deoxy-β-d-galactopyranosyl]-1,3,4-octadecantriol (2). Pd/C (10%, 29 mg) was added to a stirred solution of (2S,3R,4R)-1-O-[2-deoxy-2-iodo-β-d-galactopyranosyl]-2-hexacosanoylamino-1,3,4-octadecantriol (14) (0.05 g, 0.05 mmol) in pyridine (1.5 mL). The mixture was stirred vigorously overnight under H2 (1 atm) and then filtered through celite and concentrated. The crude reaction mixture was purified by gravity column chromatography (CH2Cl2/MeOH, 93:7), giving (2S,3R,4R)-1-O-[2-deoxy-β-d-galactopyranosyl]-2-hexacosanoylamino-1,3,4-octadecane-triol (2) as a white solid (9.0 mg, 20%): mp 175 °C; [α]25D −0.74 (c 0.37, MeOH/CH2Cl2, 20:80); IR (KBr) 3423, 2919, 2851, 2361, 2337, 1652, cm−1; 1H-NMR (CDCl3:MeOH; 95:5) δ 4.41 (d, J = 9.4 Hz, 1H), 4.16 (m, 1H), 3.97 (dd, J = 10.8, 4.0 Hz, 1H), 3.82 (dd, J = 11.6, 5.8 Hz, 1H), 3.71 (m, 3H), 3.61 (m, 1H), 3.51 (m, 2H), 3.33 (m, 2H), 3.06 (dd, J = 5.20, 5.20 Hz, 1H), 1,89 (m, 1H), 1.78 (m, 1H), 1.68–1.36 (m, 6H), 1.18 (s, 66H), 0.80 (t, J = 6.60 Hz, 6H); 13C-NMR (CDCl3:MeOH; 95:5) δ 174.2, 100.9, 75.3, 75.0, 72.9, 68.2, 68.6, 62.9, 50.7, 36.8, 34.8, 33.1, 32.1, 26.1, 25.9, 22.8, 22.7, 22.4, 14.1; HRMS (ESI) m/z calcd for C50H99NNaO8 [M+Na]+ 864.7263, found 864.7250.
3.2. Biological Assays
Stimulation of NKT cell hybridomas on microwell plates coated with soluble mouse CD1d was carried out according to published protocols [60,63,64]. Briefly, the amounts of compound indicated in the figure were incubated for 24 h in microwells that had been coated with 1.0 µg of mCD1d. After washing, 5 × 104 to 1 × 105 NKT hybridoma cells were cultured on the plate for 20 h, and IL-2 in the supernatant was measured by enzyme-linked immunoassay (ELISA).
4. Conclusions
The synthesis of 2''-deoxy-β-GalCer 2 and evaluation of its ability to stimulate two types of NKT cells have been described. Type II NKT cell hybridoma 19 reacts to sulfatide, as well as β-GalCer [14], and although iNKT cells prefer glycosphingolipids with α-linked sugars, it has been recently reported that they react to β-GluCer and β-ManCer. Therefore, it was conceivable that the hybridomas tested would have reactivity for this novel glycosphingolipid. However, only a marginal stimulation for iNKT cells was observed. For the sulfatide-reactive Type II NKT cells previously described structural data suggest that the 2''-OH group on the sugar plays a critical role through its interactions with CD1d and the NKT cell receptor. We have confirmed the crucial importance of the 2''-OH for sulfatide-reactive Type II NKT cell stimulation, as 2''-deoxy-β-GalCer did not stimulate the Type II NKT cell hybridomas.
Acknowledgments
NIH grant RO1 GM087136 (AH), NIH grant RO1 AI45153 (MK), to Dr. Susanna Cardell for providing the type II NKT cell hybridomas and to Kyowa Hakko Kirin for providing α-GalCer.
Author Contributions
AH and MK designed research; MT and AH planned and completed synthesis of 2; AK and MK conducted and analyzed biological assays; AH and MK wrote the paper; All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Rossjohn, J.; Pellicci, D.G.; Patel, O.; Gapin, L.; Godfrey, D.I. Recognition of CD1d-restricted Antigens by Natural Killer T Cells. Nat. Rev. Immunol. 2012, 12, 845–857. [Google Scholar] [CrossRef]
- Adams, E.J.; Luoma, A.M. The yin and yang of CD1d recognition. Nat. Immunol. 2012, 13, 814–815. [Google Scholar] [CrossRef]
- Girardi, E.; Zajonc, D.M. Molecular basis of lipid antigen presentation by CD1d and recognition by natural killer T cells. Immunol. Rev. 2012, 250, 167–179. [Google Scholar] [CrossRef]
- Godfrey, D.I.; Pellicci, D.G.; Patel, O.; Kjer-Nielsen, L.; McCluskey, J.; Rossjohn, J. Antigen recognition by CD1d-restricted NKT T cell receptors. Semin. Immunol. 2010, 22, 61–67. [Google Scholar] [CrossRef]
- Zajonc, D.M.; Kronenberg, M. Carbohydrate specificity of the recognition of diverse glycolipids by natural killer T cells. Immunol. Rev. 2009, 230, 188–200. [Google Scholar] [CrossRef]
- Lawson, V. Turned on by danger: Activation of CD1d-restricted invariant natural killer T cells. J. Immunol. 2012, 137, 20–27. [Google Scholar] [CrossRef]
- Tashiro, T. Structure-activity relationship studies of novel glycosphingolipids that stimulate natural killer T-cells. Biosci. Biotech. Bioch. 2012, 76, 1055–1067. [Google Scholar] [CrossRef]
- Mallevaey, T.; Selvanantham, T. Strategy of lipid recognition by invariant natural killer T cells: ‘One for all and all for one’. Immunology 2012, 136, 273–282. [Google Scholar] [CrossRef]
- Banchet-Cadeddu, A.; Henon, E.; Dauchez, M.; Renault, J.-H.; Monneaux, F.; Haudrechy, A. The stimlating adventure of KRN7000. Org. Biomol. Chem. 2011, 9, 3080–3104. [Google Scholar] [CrossRef]
- Venkataswamya, M.M.; Porcelli, S.A. Lipid and glycolipid antigens of CD1D-restricted natural killer T cells. Semin. Immunol. 2010, 22, 68–78. [Google Scholar]
- Laurent, X.; Bertin, B.; Renault, N.; Farce, A.; Speca, S.; Milhomme, O.; Millet, R.; Desreumaux, P.; Henon, E.; Chavatte, P. Switching invariant natural killer t (inkt) cell response from anticancerous to anti-inflammatory effect: Molecular bases. J. Med. Chem. 2014. In Print. [Google Scholar]
- Izhak, L.; Ambrosino, E.; Kato, S.; Parish, S.T.; O’Konek, J.J.; Weber, H.; Xia, Z.; Venzon, D.; Berzofsky, J.A.; Terabe, M. Delicate balance among three types of T cells in concurrent regulation of tumor immunity. Cancer Res. 2013, 73, 1514–1523. [Google Scholar] [CrossRef]
- Viale, R.; Ware, R.; Maricic, I.; Chaturvedi, V.; Kumar, V. NKT cell subsets can exert opposing effects in autoimmunity, tumor surveillance and inflammation. Curr. Immunol. Rev. 2012, 8, 287–296. [Google Scholar] [CrossRef]
- Rhost, S.; Sedimbi, S.; Kadri, N.; Cardell, S.L. Immunomodulatory type II natural killer T lymphocytes in health and disease. Scand. J. Immunol. 2012, 76, 246–255. [Google Scholar] [CrossRef]
- Arrenberg, P.; Maricic, I.; Kumar, V. Sulfatide-mediated activation of type II natural killer T cells prevents hepatic ischemic reperfusion injury in mice. Gastroenterology 2011, 140, 646–655. [Google Scholar]
- Sundell, I.B.; Halder, R.; Zhang, M.; Maricic, I.; Koka, P.S.; Kumar, V. Sulfatide administration leads to inhibition of HIV-1 replication and enhanced hematopoeisis. J. Stem Cells 2010, 5, 33–42. [Google Scholar]
- Berzofsky, J.A.; Terabe, M. The contrasting roles of NKT cells in tumor immunity. Curr. Mol. Med. 2009, 9, 667–672. [Google Scholar] [CrossRef]
- Wu, D.; Fujio, M.; Wong, C.-H. Glycolipids as immunostimulating agents. Bioorg. Med. Chem. 2008, 16, 1073–1083. [Google Scholar]
- Savage, P.A.; Teyton, L.; Bendelac, A. Glycolipids for natural killer T cells. Chem. Soc. Rev. 2006, 35, 771–779. [Google Scholar] [CrossRef]
- Tyznik, A.J.; Farber, E.; Girardi, E.; Birkholz, A.; Li, Y.; Chitale, S.; So, R.; Arora, P.; Khurana, A.; Wang, J.; et al. Novel glycolipids that elicit IFN-γ responses from natural killer T cells. Chem. Biol. 2011, 18, 1620–1630. [Google Scholar] [CrossRef]
- Wu, D.; Zajonc, D.M.; Fujio, M.; Sullivan, B.; Kinjo, Y.; Kronenberg, M.; Wilson, I.A.; Wong, C.-H. Design of natural killer T cell activators: Structure and function of a microbial glycosphingolipid bound to mouse CD1d. Proc. Natl. Acad. Sci. USA 2006, 103, 3972–3977. [Google Scholar]
- Koch, M.; Stronge, V.S.; Shepherd, D.; Gadola, S.D.; Matthew, B.; Ritter, G.; Fersht, A.R.; Besra, G.S.; Schmidt, R.R.; Jones, E.Y.; et al. The crystal structure of human CD1d with and without α-galactosylceramide. Nat. Immunol. 2005, 6, 819–826. [Google Scholar]
- Wun, K.S.; Cameron, G.; Patel, O.; Pang, S.S.; Pellicci, D.G.; Sullivan, L.C.; Keshipeddy, S.; Young, M.H.; Uldrich, A.P.; Thakur, M.S.; et al. A molecular basis for the exquisite CD1d-restricted antigen-specificity and functional responses of natural killer T cells. Immunity 2011, 34, 327–339. [Google Scholar] [CrossRef]
- Wun, K.S.; Ross, F.; Patel, O.; Besra, G.S.; Porcelli, S.A.; Richardson, S.K.; Keshipeddy, S.; Howell, A.R.; Godfrey, D.I.; Rossjohn, J. Human and mouse type I natural killer T cell antigen receptors exhibit different fine specificities for CD1d-antigen complex. J. Biol. Chem. 2012, 287, 39139–39148. [Google Scholar] [CrossRef]
- Zajonc, D.M.; Savage, P.B.; Bendelac, A.; Wilson, I.A.; Teyton, L. Crystal structures of mouse CD1d-iGb3 complex and its cognate V.alpha.14 T cell receptor suggest a model for dual recognition of foreign and self glycolipids. J. Mol. Biol. 2008, 377, 1104–1116. [Google Scholar] [CrossRef]
- Wun, K.S.; Borg, N.A.; Kjer-Nielsen, L.; Beddoe, T.; Koh, R.; Richardson, S.K.; Thakur, M.; Howell, A.R.; Scott-Browne, J.P.; Gapin, L.; et al. A minimal binding footprint on CD1d glycolipid is a basis for selection of the unique human NKT TCR. J. Exp. Med. 2008, 205, 939–949. [Google Scholar] [CrossRef]
- Scott-Browne, J.P.; Matsuda, J.L.; Mallevaey, T.; White, J.; Borg, N.A.; McCluskey, J.; Rossjohn, J.; Kappler, J.; Marrack, P.; Gapin, L. Germline-encoded recognition of diverse glycolipids by natural killer T cells. Nat. Immunol. 2007, 8, 1105–1113. [Google Scholar] [CrossRef]
- Borg, N.A.; Wun, K.S.; Kjer-Nielsen, L.; Wilce, M.C.J.; Pellicci, D.G.; Koh, R.; Besra, G.S.; Bharadwaj, M.; Godfrey, D.I.; McCluskey, J.; et al. CD1d-lipid-antigen recognition by the semi-invariant NKT T-cell receptor. Nature 2007, 448, 44–49. [Google Scholar] [CrossRef]
- Girardi, E.; Maricic, I.; Wang, J.; Mac, T.-T.; Iyer, P.; Kumar, V.; Zajonc, D.M. Type II natural killer T cells use features of both innate-like and conventional T cells to recognize sulfatide self antigens. Nat. Immunol. 2012, 13, 851–856. [Google Scholar] [CrossRef]
- Patel, O.; Pellicci, D.G.; Gras, S.; Sandoval-Romero, M.L.; Uldrich, A.P.; Mallevaey, T.; Clarke, A.J.; le Nours, J.; Theodossis, A.; Cardell, S.L.; et al. Recognition of CD1d-sulfatide mediated by a type II natural killer T cell antigen receptor. Nat. Immunol. 2012, 13, 857–863. [Google Scholar] [CrossRef]
- Zajonc, D.M.; Maricic, I.; Wu, D.; Halder, R.; Keshab, R.; Wong, C.-H.; Kumar, V.; Wilson, I.A. Structural basis for CD1d presentation of a sulfatide derived from myelin and its implications for autoimmunity. J. Exp. Med. 2005, 202, 1517–1526. [Google Scholar] [CrossRef]
- Iijima, H.; Kimura, K.; Sakai, T.; Uchimura, A.; Shimizu, T.; Ueno, H.; Natori, T.; Koezuka, Y. Structure-activity relationship and conformational analysis of monoglycosylceramides on the syngeneic mixed leukocyte reaction. Bioorg. Med. Chem. 1998, 6, 1905–1910. [Google Scholar] [CrossRef]
- Brennan, P.J.; Tatituri, R. V.V.; Brigl, M.; Kim, E.Y.; Tuli, A.; Sanderson, J.P.; Gadola, S.D.; Hsu, F.-F.; Besra, G.S.; Brenner, M.B. Invariant natural killer T cells recognize lipid self antigen induced by microbial danger signals. Nat. Immunol. 2011, 12, 1202–1211. [Google Scholar] [CrossRef]
- O’Konek, J.J.; Illarionov, P.; Khursigara, D.S.; Ambrosino, E.; Izhak, L.; Castillo, B.F.I.; Raju, R.; Khalili, M.; Kim, H.-Y.; Howell, A.R.; et al. Mouse and human iNKT cell agonist β-mannosylceramide reveals a distinct mechanism of tumor immunity. J. Clin. Invest. 2011, 121, 683–694. [Google Scholar] [CrossRef]
- Toshima, K.; Tatsuta, K. Recent progress in O-glycosylation methods and its application to natural products synthesis. Chem. Rev. 1993, 93, 1503–1531. [Google Scholar]
- Takiura, K.; Honda, S. Hydroxy- and acetoxymercuration of D-glucal triacetate. Carbohyd. Res. 1972, 23, 369–377. [Google Scholar]
- Monneret, C.; Choay, P. A convenient synthesis of 2-deoxy-D-arabinohexose and its methyl and benzyl glycosides. Carbohyd. Res. 1981, 96, 299–305. [Google Scholar] [CrossRef]
- Giese, B.; Kopping, B.; Chatgilialoglu, C. Tris(trimethylsilyl)silane as mediator in organic synthesis via radicals. Tetrahedron Lett. 1989, 30, 681–684. [Google Scholar] [CrossRef]
- Thiem, J.; Klaffke, W. Syntheses of deoxyoligosaccharides. Top. Curr. Chem. 1990, 154, 285–332. [Google Scholar] [CrossRef]
- Gervay, J.; Danishefsky, S. A stereospecific route to 2-deoxy-β-glycosides. J. Org. Chem. 1991, 56, 5448–5451. [Google Scholar] [CrossRef]
- Durham, T.B.; Roush, W.R. Stereoselective synthesis of 2-deoxy-β-galactosides via 2-deoxy-2-bromo- and 2-deoxy-2-iodo-galactopyranosyl donors. Org. Lett. 2003, 5, 1871–1874. [Google Scholar] [CrossRef]
- Roush, W.R.; Bennett, C.E. A highly stereoselective synthesis of 2-deoxy-β-glycosides using 2-deoxy-2-iodo-glucopyranosyl acetate donors. J. Am. Chem. Soc. 1999, 121, 3541–3542. [Google Scholar] [CrossRef]
- Roush, W.R.; Briner, K.; Sebesta, D.P. Highly stereoselective synthesis of α-L-olivomycosides via trimethylsilyl triflate mediated glycosidations of 1-O-Acetyl-4-O-isobutyryl-2,6-dideoxy-2-iodo-3-C-methyl-α-L-mannopyranose. Synlett 1993, 264–266. [Google Scholar] [CrossRef]
- Roush, W.R.; Narayan, S. 2-Deoxy-2-iodo-α-mannopyranosyl and -talopyranosyl acetates: Highly stereoselective glycosyl donors for the synthesis of 2-deoxy-α-glycosides. Org. Lett. 1999, 1, 899–902. [Google Scholar] [CrossRef]
- Nicolaou, K.C.; Ladduwahetty, T.; Randall, J.L.; Chucholowski, A. Stereospecific 1,2-migrations in carbohydrates. Stereocontrolled synthesis of α- and β-2-deoxyglycosides. J. Am. Chem. Soc. 1986, 108, 2466–2467. [Google Scholar] [CrossRef]
- Pongdee, R.; Wu, B.; Sulikowski, G.A. One-pot synthesis of 2-deoxy-β-oligosaccharides. Org. Lett. 2001, 3, 3523–3525. [Google Scholar] [CrossRef]
- Hashimoto, S.; Yanagiya, Y.; Honda, T.; Ikegami, S. A stereocontrolled construction of 2-deoxy-β-glycosidic linkages via 1,2-trans-β-glycosidation of 2-deoxy-2-[(p-methoxyphenyl)thio]glycopyranosyl N,N,N',N'-tetramethylphosphoroamidates. Chem. Lett. 1992, 1511–1514. [Google Scholar]
- Binkley, R.W.; Koholic, D.J. Photoremovable hydroxyl group protection. Use of the p-tolylsulfonyl protecting group in β-disaccharide synthesis. J. Org. Chem. 1989, 54, 3577–3581. [Google Scholar] [CrossRef]
- Toshima, K.; Misawa, M.; Ohta, K.; Tatsuta, K.; Kinoshita, M. Enantiospecific synthesis of C20-C28 segment of concanamycin a: Application of diethylisopropylsilyl protecting group. Tetrahedron Lett. 1989, 30, 6417–6420. [Google Scholar] [CrossRef]
- Hadd, M.J.; Gervay, J. Glycosyl iodides are highly efficient donors under neutral conditions. Carbohyd. Res. 1999, 320, 61–69. [Google Scholar] [CrossRef]
- Zhou, M.; O’Doherty, G.A. De novo approach to 2-deoxy-β-glycosides: Asymmetric syntheses of digoxose and digitoxin. J. Org. Chem. 2007, 72, 2485–2493. [Google Scholar] [CrossRef]
- Tanaka, H.; Yoshizawa, A.; Takahashi, T. Direct and stereoselective synthesis of β-linked 2,6-deoxyoligosaccharides. Angew. Chem., Int. Ed. 2007, 46, 2505–2507. [Google Scholar] [CrossRef]
- Morris, W.J.; Shair, M.D. Stereoselective synthesis of 2-deoxy-β-glycosides using anomeric O-alkylation/arylation. Org. Lett. 2009, 11, 9–12. [Google Scholar] [CrossRef]
- Gervay, J.; Peterson, J.M.; Oriyama, T.; Danishefsky, S.J. An unexpected sialylation: Total syntheses of ganglioside GM4 and a positional isomer. J. Org. Chem. 1993, 58, 5465–5468. [Google Scholar] [CrossRef]
- Zhang, Z.; Magnusson, G. Synthesis of double-chain bis-sulfone neoglycolipids of the 2'-, 3'- and 6'-deoxyglobotrioses. J. Org. Chem. 1995, 60, 7304–7305. [Google Scholar] [CrossRef]
- Randolph, J.T.; Danishefsky, S.J. First synthesis of a digitalis saponin. demonstration of the scope and limitations of a convergent scheme for branched oligosaccharide synthesis by the logic of glycal assembly. J. Am. Chem. Soc. 1995, 117, 5693–5700. [Google Scholar] [CrossRef]
- Blauvelt, M.L.; Khalili, M.; Jaung, W.; Paulsen, J.; Anderson, A.C.; Wilson, S.B.; Howell, A.R. α-S-Galcer: Synthesis and evaluation for iNKT cell stimulation. Bioorg. Med. Chem. Lett. 2008, 18, 6374–6376. [Google Scholar] [CrossRef]
- Burdin, N.; Brossay, L.; Degano, M.; Iijima, H.; Gui, M.; Wilson, I.A.; Kronenberg, M. Structural requirements for antigen presentation by mouse CD1. Proc. Natl. Acad. Sci. USA 2000, 97, 10156–10161. [Google Scholar] [CrossRef]
- Blomqvist, M.; Rhost, S.; Teneberg, S.; Loefbom, L.; Oesterbye, T.; Brigl, M.; Mansson, J.-E.; Cardell, S. Multiple tissue-specific isoforms of sulfatide activate CD1d-restricted type II NKT cells. Eur. J. Immunol. 2009, 39, 1726–1735. [Google Scholar] [CrossRef]
- Naidenko, O.V.; Maher, J.K.; Ernst, W.A.; Sakai, T.; Modlin, R.L.; Kronenberg, M. Binding and antigen presentation of ceramide-containing glycolipids by soluble mouse and human CD1d molecules. J. Exp. Med. 1999, 190, 1069–1079. [Google Scholar] [CrossRef]
- Giabbai, B.; Sidobre, S.; Crispin, M.D.; Sanchez-Ruiz, Y.; Bachi, A.; Kronenberg, M.; Wilson, I.A.; Degano, M. Crystal structure of mouse CD1d bound to the self ligand phosphatidylcholine: A molecular basis for NKT cell activation. J. Immunol. 2005, 175, 977–984. [Google Scholar] [CrossRef]
- Jo, S.Y.; Kim, H.C.; Woo, S.W.; Seo, M.J.; Lee, G.; Kim, H.R. Synthesis of 1-substituted-phytosphingosine: Novel protection of phytosphingosine. Bull. Korean Chem. Soc. 2003, 24, 267–268. [Google Scholar] [CrossRef]
- Sidobre, S.; Hammond, K.J.L.; Benazet-Sidobre, L.; Maltsev, S.D.; Richardson, S.K.; Ndonye, R.; Howell, A.R.; Sakai, T.; Besra, G.S.; Porcelli, S.A.; et al. The TCR expressed by Vα14i NKT has a unique mode of antigen recognition. Proc. Natl. Acad. Sci. USA 2004, 101, 12254–12259. [Google Scholar] [CrossRef]
- Tupin, E.; Kronenberg, M. Activation and natural killer T cells by glycolipids. Methods Enzymol. 2006, 417, 185–201. [Google Scholar] [CrossRef]
- Sample Availability: Samples of compound 2 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).