Abstract
We present herein the synthesis in good yields of two series of highly functionalized thiazolidinone derivatives from the reactions of various 4-phenyl-3-thio-semicarbazones with ethyl 2-bromoacetate and diethyl acetylenedicarboxylate, respectively.
1. Introduction
Thiosemicarbazones are a class of small molecules that have been evaluated over the last 50 years as antiviral [1] and as antitumoral agents [2,3,4], in addition to their antiparasitic and bacterial action against Trypanasoma cruzi [5,6,7] and Toxoplasma gondii and several bacterial strains [8]. Thiosemicarbazones have been used as intermediates for a great variety of heterocyclic products, such as thiazolidinones, thiohydantoins, thioxopyrimidinediones. It is reported that thiazolidinones exhibit antibacterial [9], antifungal [10], anticonvulsant [11], antitubercular [12], anti-inflammatory [13], antihistaminic [14,15], cardiovascular [16] and anti-HIV [17] activities. As part of our research program on new bioactive compounds [18,19,20,21,22], we report herein an efficient synthesis of some new highly functionalized thiazolidinones derived from 4-phenyl-3-thiosemicarbazones.
2. Results and Discussion
The starting materials, 4-phenyl-3-thiosemicarbazones 4a–h, were synthesized in two steps. The first step was the preparation of 4-phenyl thiosemicarbazide (2) in 86% yield from phenyl isothiocyanate (1) and hydrazine hydrate in ethanol at room temperature [23] (Scheme 1).
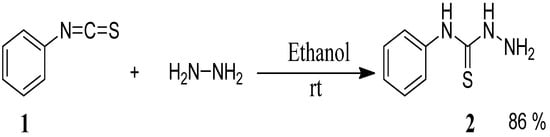
Scheme 1.
Preparation of 4-phenyl-3-thiosemicarbazide (2).
Scheme 1.
Preparation of 4-phenyl-3-thiosemicarbazide (2).
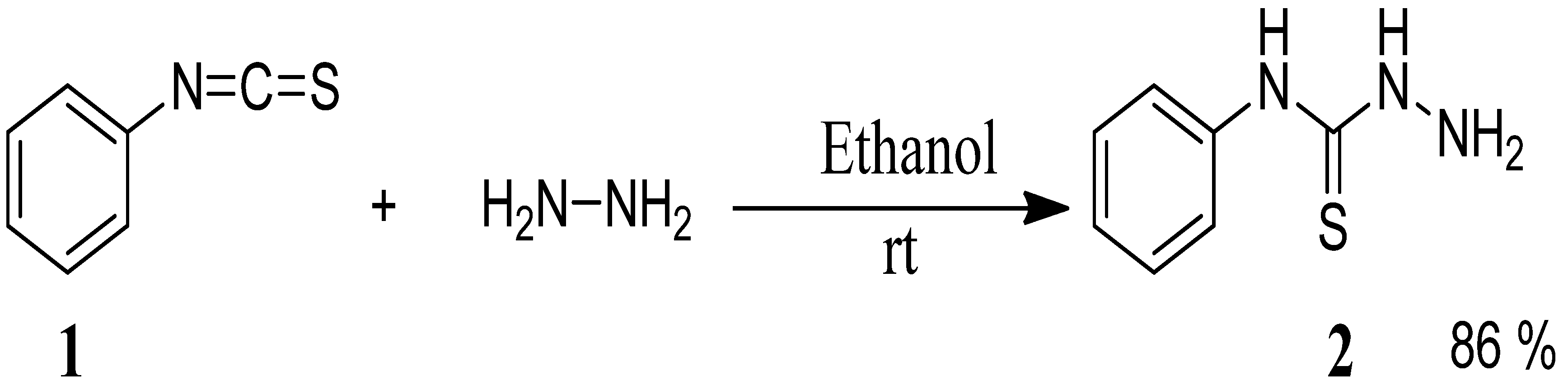
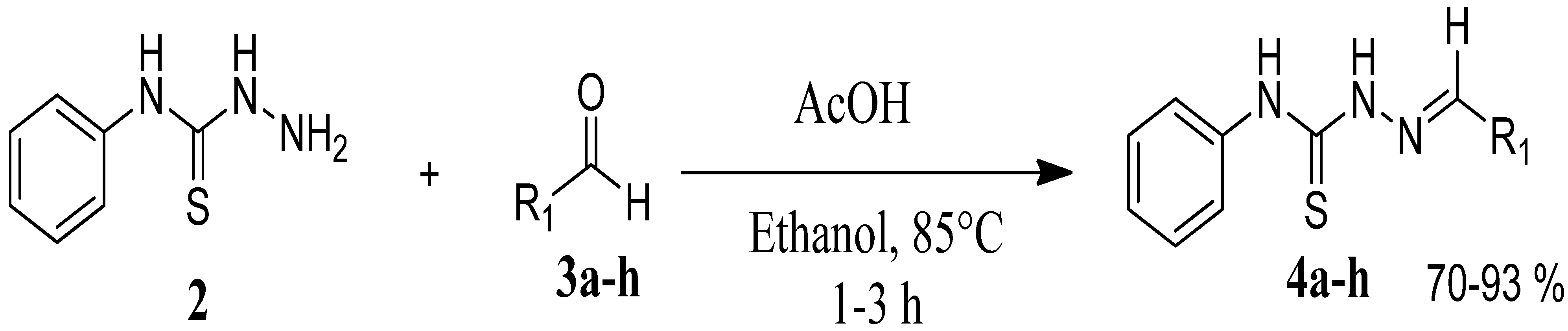
The reaction of 4-phenyl-3-thiosemicarbazide (2) with various aromatic aldehydes 3a–h in the presence of few drops of acetic acid at 85 °C for 1–3 h, led to the corresponding 4-phenyl-3-thiosemicarbazone derivatives 4a–h in good yields (70%–93%), as shown in Scheme 2 and Table 1.
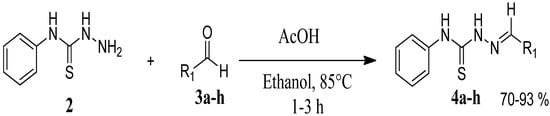
Scheme 2.
Preparation of 4-phenyl-3-thiosemicarbazones 4a–h.
Scheme 2.
Preparation of 4-phenyl-3-thiosemicarbazones 4a–h.
The most characteristic signals in the 1H-NMR spectrum of this family of thiosemicarbazones were those corresponding to the CH=N and N-H protons. 1H-NMR studies showed the CH=N protons in the 7.86–8.62 ppm range, whereas thiourea N-H protons are found in the 9.13–11.78 ppm interval for N-H adjacent to the monosubstituted phenyl ring and for the N-H adjacent to the CH=N moiety, respectively. All of the synthesized compounds were in the E-configuration, which was confirmed using 1H-NMR spectroscopy, as the signal of the NH group was in the 9–12 ppm range, in comparison to the Z-isomer, which possesses a characteristic NH signal in the 14–15 ppm range [24].

Table 1.
Reaction of 2 with various aromatic aldehydes 3a–h.
| Carbonyl compound | Product | Product number | Reaction time (h) | Yield (%) |
|---|---|---|---|---|
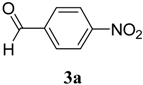 | 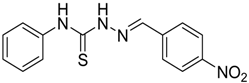 | 4a | 1 | 70 |
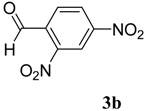 | 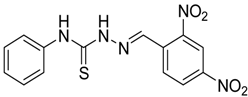 | 4b | 3 | 90 |
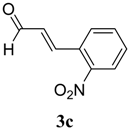 | 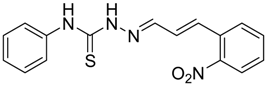 | 4c | 2 | 93 |
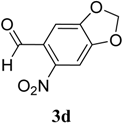 | 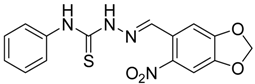 | 4d | 1 | 90 |
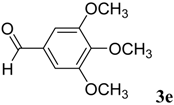 | 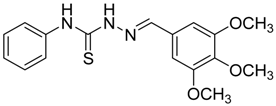 | 4e | 1 | 91 |
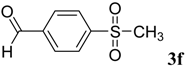 | 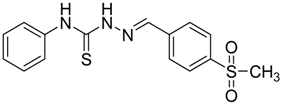 | 4f | 2 | 89 |
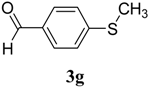 | 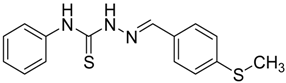 | 4g | 2 | 91 |
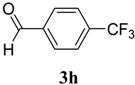 | 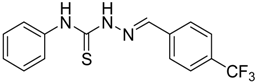 | 4h | 1 | 89 |
The reaction of various 4-phenyl thiosemicarbazones 4a–h with ethyl 2-bromoacetate (5) as cyclizing reagent in boiling absolute ethanol containing three equivalents of anhydrous sodium acetate during 1–3 h, afforded to the thiazolidin-4-ones 6a–h in good yields (68%–91%) as shown in Scheme 3 and Table 2.
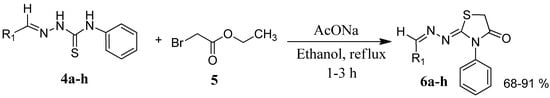
Scheme 3.
Preparation of thiazolidinones 6a–h.
Scheme 3.
Preparation of thiazolidinones 6a–h.

Table 2.
Reactions of 4a–h with ethyl 2-bromoacetate (5).
| Compound | Product | Product number | Reaction time (h) | Yield (%) |
|---|---|---|---|---|
| 4a | 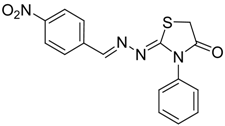 | 6a | 1 | 91 |
| 4b | 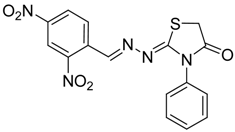 | 6b | 3 | 68 |
| 4c | 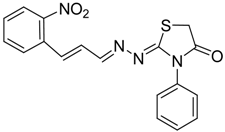 | 6c | 3 | 82 |
| 4d | 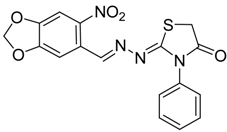 | 6d | 2 | 86 |
| 4e | 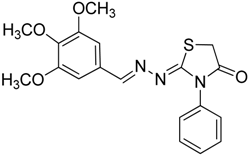 | 6e | 2 | 80 |
| 4f | 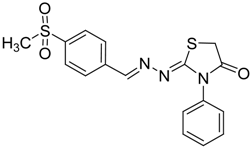 | 6f | 3 | 89 |
| 4g | 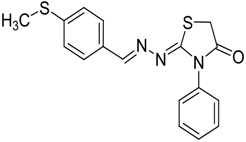 | 6g | 3 | 90 |
| 4h | 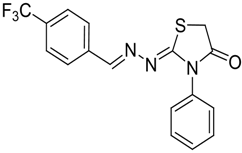 | 6h | 2 | 88 |
The structures of all new compounds 6a–h were established by analysis of their IR, 1H-NMR and 13C-NMR data. The IR spectra of the thiazolidin-4-ones 6a–h showed absorption bands at about 1,734–1,716 cm−1 characteristic of (amide group) C=O stretching vibrations. Further support was obtained from the 1H-NMR spectra, where it did not display signs of the 4-phenyl-3-thiosemicarbazone (NH) protons. On the other hand, the 1H-NMR spectra exhibited resonances assigned to the SCH2 group of the thiazolidine ring appearing as a singlet at 3.97–4.10 ppm due to the methylene protons. The CH=N protons in these structures were observed in the 7.67–8.57 ppm region. The formation of thiazolidinones 6a–h ocurred in two steps: the first step of this reaction is thought to be S-alkylation of thiosemicarbazide in its thiol form due to the sodium acetate used. Second step involved loss of ethanol to give the thiazolidin-4-one. The electronic and steric properties of the substituent at the 4-position of the thiosemicarbazones seems to be a determining factor for the formation of the thiazolidinone ring. Previous reports on these types of compounds reveal a small substituent such as phenyl or alkyl leads to a 4-thiazolidinone ring by loss of ethanol [25].
The next cyclization reaction of 4-phenyl-3-thiosemicarbazones derivatives 4a–h was conducted using diethyl acetylenedicarboxylate in methanol for 1 h [26], as shown in Scheme 4 and Table 3. In this reaction both of the sulfur group and the amino group are capable of reacting with diethyl acetylenedicarboxylate. It was found that the 4-phenyl thiosemicarbazone derivatives 4a–h reacted with diethyl acetylenedicarboxylate exclusively with the sulfur atom. In this reaction the intermediate 7 undergoes an intramolecular cyclization which leads to the compounds 8a–h.
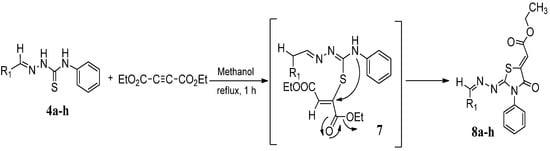
Scheme 4.
Preparation of 8a–h with 4-phenyl-3-thiosemicarbazones 4a–h and diethyl acetylenedicarboxylate.
Scheme 4.
Preparation of 8a–h with 4-phenyl-3-thiosemicarbazones 4a–h and diethyl acetylenedicarboxylate.

Table 3.
Reactions of 4a–h with diethyl acetylenedicarboxylate.
| Compound | Product | Product number | Yield (%) |
|---|---|---|---|
| 4a |  | 8a | 73 |
| 4b |  | 8b | 70 |
| 4c |  | 8c | 76 |
| 4d |  | 8d | 74 |
| 4e | 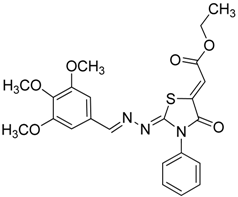 | 8e | 73 |
| 4f | 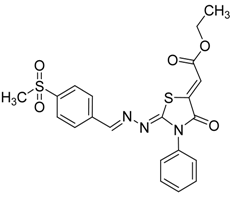 | 8f | 75 |
| 4g | 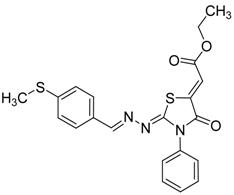 | 8g | 70 |
| 4h | 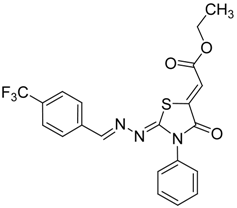 | 8h | 71 |
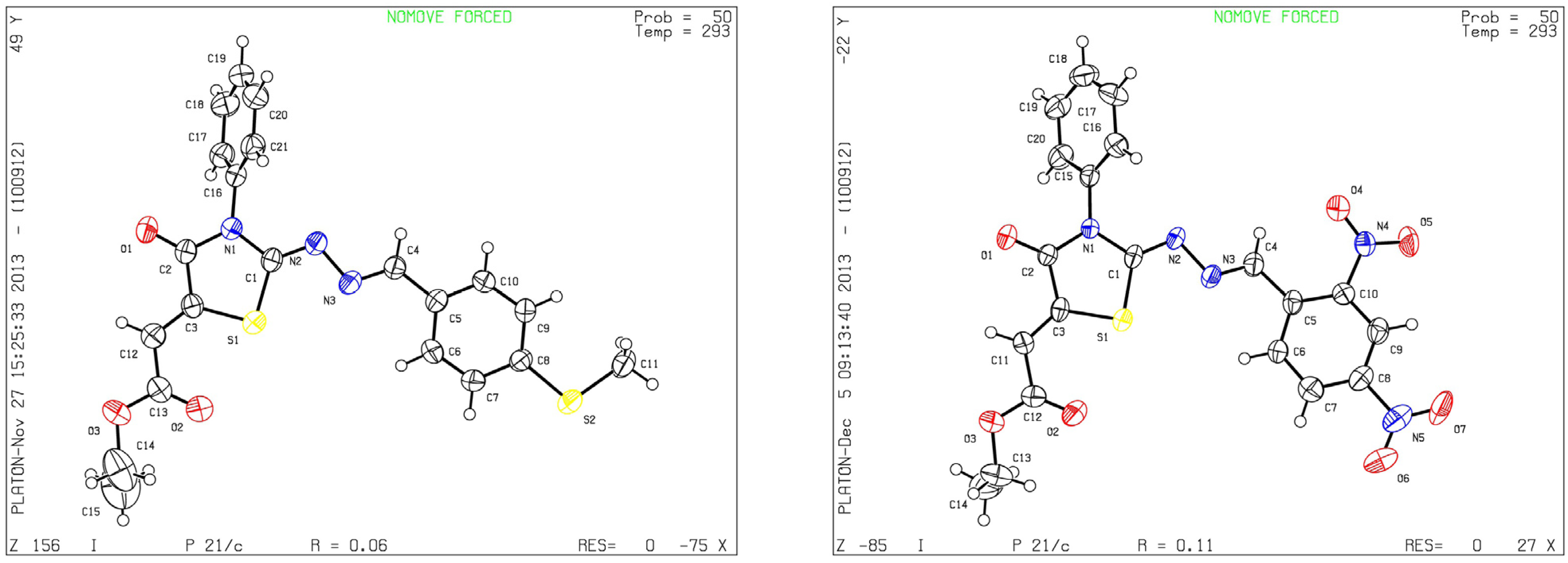
Although the two geometrical E- or Z- isomers of 8a–h could be formed in almost equal amounts from the reaction of diethyl acetylenedicarboxylate with 4a–h, 1H-NMR revealed the presence of only one singlet at 6.8 ppm (vinyl proton) indicating that only one E- or Z-isomer was formed. The structures of compound 8b and 8g obtained by X-ray structure analysis confirmed the Z-configuration for the double bond in the 5-position of the thiazolidin-4-ones (Figure 1) [27,28], probably due to the steric effect of the ester group.
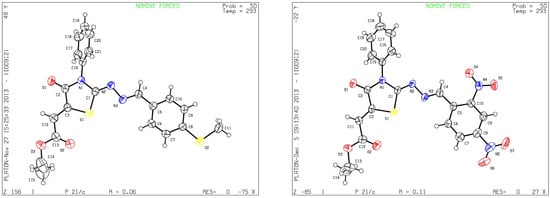
Figure 1.
ORTEP plots of 8b and 8g.
Figure 1.
ORTEP plots of 8b and 8g.
The chemical structures of the reaction products 8a–h were confirmed by their IR, 1H-NMR, 13C-NMR spectra. The IR spectrum of compound 8a, for example, showed absorptions at 1730, 1692 cm−1 due to the C=O functions of the ester and cyclic amide, respectively. Similarly, bands at 1595–1622 cm−1 are due to the C=N groups. The 1H-NMR spectrum of 8a showed a triplet at δ = 1.28 ppm and a quartet at δ = 4.28 ppm is due to the COOCH2CH3 protons. A singlet at δ = 6.80 ppm is due to C=CH. Aromatic protons appeared as a multiplet at δ = 7.38–7.60 ppm.
3. Experimental
3.1. General
Melting points were determined on Büchi B-540 apparatus and are uncorrected. IR spectra were recorded as KBr pellets on a JASCO FT/IR4200 Fourier Transform infrared spectrometer and the reported wavenumbers are given in cm−1. Elemental analyses were carried out at the Spectropole, Faculté des Sciences site Saint-Jérome. 1H-NMR (200 MHz) and 13C-NMR (50 MHz) spectra were recorded on a Bruker ARX 200 spectrometer in CDCl3 or D2O at the Service Inter-Universitaire de RMN de la Faculté de Pharmacie de Marseille. The 1H-NMR chemical shifts were reported as parts per million downfield from tetramethylsilane (Me4Si), and the 13C-NMR chemical shifts were referenced to the solvent peaks: CDCl3 (76.9 ppm) or DMSO-d6 (39.6 ppm). Silica gel 60 (Merck, 230–400 mesh) was used for column chromatography: Thin-layer chromatography was performed with silica gel Merck 60F-254 (0.25 mm layer thickness).
3.2. General Procedure for the Preparation of Compounds 4a–h
To a solution of 4-phenylthiosemicarbazide (2, 1 g, 6 mmol, 1 eq) in ethanol (33 mL) were added the benzaldehyde derivative (6.3 mmol, 1.05 eq) and acetic acid (0.50 mL). The mixture was stirred under reflux for 1–3 h and then cooled to room temperature. After, the solid separated was filtered and recrystallized from ethanol-DMF (3:1) to give compounds 4a–h.
(E)-2-(4-Nitrobenzylidene)-N-phenylhydrazinecarbothioamide (4a): yellow solid; mp: 234 °C; IR (KBr) νmax/cm−1: 3316 and 3138 (NH), 1543 (C=N), 1292 (C=S); 1H-NMR (DMSO-d6): δ 10.32 (s, 1H, NH), 8.24 (d, 2H, J = 9.2 Hz, Ar-H), 8.22 (s, 1H, CH=N), 8.17 (d, 2H, J = 9.2 Hz, Ar-H), 7.52 (d, 2H, J = 7.9 Hz, Ar-H), 7.38 (dd, 2H, J = 7.9 Hz, J = 7.2 Hz, Ar-H), 7.22 ( t, 1H J = 7.2 Hz, Ar-H); 13C-NMR (DMSO-d6): δ 177.0 (C), 148.1 (C), 141.0 (C), 140.6 (CH), 139.4 (C), 128.9 (2CH), 128.6 (2CH), 126.7 (2CH), 126.1 (CH), 124.4 (2CH); Anal. Calcd for C14H12N4O2S: C, 55.99; H, 4.03; N, 18.65; S, 10.68. Found: C, 55.98; H, 4.06; N, 18.49; S, 10.68.
(E)-2-(2,4-Dinitrobenzylidene)-N-phenylhydrazinecarbothioamide (4b): yellow solid; mp: 219 °C; IR (KBr) νmax/cm−1: 3316 and 3138 (NH), 1543 (C=N), 1292 (C=S); 1H-NMR (DMSO-d6): δ 10.40 (s, 1H, NH), 8.87 (d, 1H, J = 8.8 Hz, Ar-H), 8.75 (d, 1H, J = 2.2 Hz, Ar-H), 8.62 (s, 1H, CH=N), 8.45 (dd, 1H, J = 8.8 Hz, J = 2.2 Hz, Ar-H), 7.52 (d, 2H, J = 7.7 Hz, Ar-H), 7.39 (dd, 2H, J = 7.7 Hz, J = 7.3 Hz, Ar-H), 7.24 (t, 1H, J = 7.3 Hz, Ar-H); 13C-NMR (DMSO-d6): δ 177.2 (C), 148.3 (C), 147.5 (C), 139.2 (C), 136.4 (CH), 134.6 (C), 130.3 (CH), 128.7 (2CH), 127.4 (CH), 126.6 (2CH), 126.3 (CH), 120.8 (CH); Anal. Calcd for C14H11N5O4S: C, 48.69; H, 3.21; N, 20.28; S, 9.29. Found: C, 48.83; H, 3.22; N, 20.08; S, 9.05.
(E)-2-((E)-3-(2-Nitrophenyl)allylidene)-N-phenylhydrazinecarbothioamide (4c): brown solid; mp: 185 °C; IR (KBr) νmax/cm−1: 3226 and 3151 (NH), 1540 (C=N), 1247 (C=S); 1H-NMR (CDCl3): δ 9.67 (s, 1H, NH), 9.14 (s, 1H, NH), 8.02 (dd, 1H, J = 8.2 Hz, J = 1.2 Hz, Ar-H), 7.76-7.59 (m, 5H, Ar-H), 7.52-7.37 (m, 4H, Ar-H), 7.25–7.29 (m, 1H, Ar-H), 6.87 (dd, 1H, J = 15.9 Hz, J = 9.2 Hz, CH); 13C-NMR (CDCl3): δ 175.6 (C), 147.8 (C), 143.3 (CH), 137.7 (C), 134.8 (CH), 133.3 (CH), 131.3 (C), 129.4 (CH), 129.0 (CH), 128.8 (2CH), 128.2 (CH), 126.2 (CH), 125.0 (CH), 124.2 (2CH); Anal. Calcd for C16H14N4O2S: C, 58.88; H, 4.32; N, 17.17; S, 9.82. Found: C, 59.13; H, 4.40; N, 17.07; S, 9.78.
(E)-2-((6-Nitrobenzo[d][1,3]dioxol-5-yl)methylene)-N-phenylhydrazinecarbothioamide (4d): yellow solid; mp: 231 °C; IR (KBr) νmax/cm−1: 3302 and 3283 (NH), 1547 (C=N), 1268 (C=S); 1H-NMR (DMSO-d6): δ 10.21 (s, 1H, NH), 8.58 (s, 1H, Ar-H), 8.17 (s, 1H, CH=N), 7.61 (s, 1H, Ar-H), 7.50 (dd, 2H, J = 7.8 Hz , J = 1.2 Hz , Ar-H), 7.37 (dd, 2H, J = 7.8 Hz , J = 7.2 Hz, Ar-H), 7.21 (td, 1H, J = 7.2 Hz, J = 1.2 Hz, Ar-H), 6.25 (s, 2H, CH2); 13C-NMR (DMSO-d6): δ 176.9 (C), 152.2 (C), 149.2 (C), 143.9 (C), 139.4 (C), 138.5 (CH), 128.6 (2CH), 126.94 (2CH), 126.1 (CH), 125.9 (C), 106.6 (CH), 105.3 (CH), 104.1 (CH2); Anal. Calcd for C15H12N4O4S: C, 52.32; H, 3.51; N, 16.27; S, 9.31. Found: C, 52.35; H, 3.46; N, 16.01; S, 9.20.
(E)-N-Phenyl-2-(3,4,5-trimethoxybenzylidene)hydrazinecarbothioamide (4e): white solide; mp: 161 °C; IR (KBr) νmax/cm−1: 3291 and 3134 (NH), 1557 (C=N), 1262 (C=S); 1H-NMR (CDCl3): δ 10.36 (s, 1H, NH), 9.14 (s, 1H, NH), 7.93 (s, 1H, CH=N), 7.63 (d, 2H, J = 7.8 Hz, Ar-H), 7.44 (dd, 2H, J = 7.8 Hz, J = 7.2 Hz, Ar-H), 7.30 (t, 1H, J = 7.2 Hz, Ar-H ), 6.90 (s, 2H, Ar-H ), 3.90 (s, 3H, OCH3), 3.91 (s, 6H, OCH3); 13C-NMR (CDCl3): δ 175.6 (C), 153.6 (2C), 143.7 (CH), 140.5 (C), 137.7 (C), 129.0 (2CH), 128.4 (C), 126.6 (CH), 125.2 (2CH), 104.7 (2CH), 61.1 (CH3), 56.3 (2CH3); Anal. Calcd for C17H19N3O3S: C, 59.11; H, 5.54; N, 12.17; S, 9.28. Found: C, 59.25; H, 5.65; N, 12.06; S, 9.19.
(E)-2-(4-(Methylsulfonyl)benzylidene)-N-phenylhydrazinecarbothioamide (4f): yellow solid; mp: 225 °C; IR (KBr) νmax/cm−1: 3325 and 3150 (NH), 1542 (C=N), 1267 (C=S); 1H-NMR (DMSO-d6): δ 10.30 (s, 1H, NH), 8.22 (s, 1H, CH=N ), 8.19 (d, 2H, J = 8.4 Hz, Ar-H), 7.95 (d, 2H, J = 8.4 Hz, Ar-H), 7.55 (d, 2H, J = 7.60 Hz, Ar-H), 7.39 (dd, 2H, J = 7.2 Hz, J = 7.60 Hz, Ar-H), 7.23 ( t, 1H, J =7.2 Hz, Ar-H), 3.26 ( s, 3H, CH3 ); 13C-NMR (DMSO-d6): δ 177.0 (C), 141.7 (C), 141.2 (CH), 139.44 (2C), 128.7 (2CH), 128.6 (2CH), 127.7 (2CH), 126.7 (2CH), 126.10 (CH), 43.9 (CH3); Anal. Calcd for C15H15N3O2S2: C, 54.03; H, 4.53; N, 12.60; S, 19.23. Found: C, 54.11; H, 4.55; N, 12.45; S, 19.21.
(E)-2-(4-(Methylthio)benzylidene)-N-phenylhydrazinecarbothioamide (4g): yellow solid; mp: 178 °C; IR (KBr) νmax/cm−1: 3343 and 3152 (NH), 1541 (C=N), 1271 (C=S); 1H-NMR (CDCl3): δ 11.78 (s, 1H, NH), 10.08 (s, 1H, NH), 8.09 (s, 1H, CH=N), 7.82 (d, 2H, J = 8.4 Hz, Ar-H), 7.54 (d, 2H, J = 8.0 Hz, Ar-H), 7.35 (dd, 2H, J = 7.3 Hz, J = 8.0 Hz, Ar-H), 7.27 (d, J = 8.4 Hz, 2H, Ar-H), 7.18 (dd, 1H, J = 7.3 Hz, Ar-H), 13C-NMR (DMSO-d6): δ 175.4 (C), 143.1 (CH), 142.5 (C), 137.8 (C), 129.6 (C), 128.9 (2CH), 127.8 (2CH), 126.3 (CH), 125.8 (2CH), 124.8 (2CH), 15.1 (CH3); Anal. Calcd for C15H15N3O2S2: C, 59.77; H, 5.02; N, 13.94; S, 21.28. Found: C, 59.68; H, 5.04; N, 13.78; S, 21.33.
(E)-N-Phenyl-2-(4-(trifluoromethyl)benzylidene)hydrazinecarbothioamide (4h): white solid; mp: 195 °C; IR (KBr) νmax/cm−1: 3350 and 3138 (NH), 1542 (C=N), 1267 (C=S); 1H-NMR (CDCl3): δ 10.36 (s, 1H, NH), 9.18 (s, 1H, NH), 8.03 (s, 1H, CH=N), 7.85 (d, 2H, J = 8.1 Hz, Ar-H), 7.69–7.51 (m, 4H, Ar-H), 7.43 (d, 2H, J = 7.4 Hz, Ar-H), 7.29 (t, 1H, J = 7.3 Hz, Ar-H); 13C-NMR (CDCl3): δ 176.0 (C), 141.4 (CH), 137.5 (C), 133.9 (C), 131.5 (q, J = 32.9 Hz, C), 129.5 (CH), 128.9 (3CH), 127.0 (q, J = 3.6 Hz, CH), 126.6 (CH), 124.9 (2CH), 123.9 (q, J = 3.6 Hz, CH), 123.7 (q, J = 272.6 Hz, C); Anal. Calcd for C15H12F3N3S: C, 55.72; H, 3.74; N, 13.00; S, 9.92. Found: C, 55.79; H, 3.66; N, 12.86; S, 9.81.
3.3. General Procedure for the Preparation of Compounds 6a–h
A mixture of compound 4a–h (1.5 mmol, 1 eq), ethyl 2-bromoacetate (0.24 mL, 1.5 mmol) and anhydrous sodium acetate (0.37 g, 4.5 mmol, 3 eq) in ethanol (30 mL) was stirred until reflux; the mixture was stirred under the same conditions till the completion of the reaction (1–3 h). The reaction mixture was left to cool, poured into ice cold water, and the separated solid was filtered, washed with water and recrystallized from a mixture of ethanol-DMF (3:1).
2-((4-Nitrobenzylidene)hydrazono)-3-phenylthiazolidin-4-one (6a): yellow solid; mp: 258 °C; IR (KBr) νmax/cm−1: 1716 (C=O), 1662 (C=N); 1H-NMR (CDCl3): δ 8.34 (s, 1H, CH=N), 8.25 (d, 2H, J = 8.8 Hz , Ar-H), 7.88 (d, 2H, J = 8.8 Hz , Ar-H), 7.59-7.44 (m, 3H, Ar-H), 7.35 (dd, 2H, J = 7.7 Hz, J = 1.9 Hz, Ar-H), 4.00 (s, 2H, CH2); 13C-NMR (CDCl3): δ 171.6 (C), 166.9 (C), 156.2 (CH), 148.9 (C), 140.0 (C), 134.3 (C), 129.4 (2CH), 129.3 (CH), 128.6 (2CH), 127.7 (2CH), 124.0 (2CH), 32.55 (CH2); Anal. Calcd for C16H12N4O3S: C, 56.46; H, 3.55; N, 16.46; S, 9.42. Found: C, 56.44; H, 3.5; N, 16.53; S, 9.53.
2-((2,4-Dinitrobenzylidene)hydrazono)-3-phenylthiazolidin-4-one (6b): yellow solid; mp: 275 °C; IR (KBr) νmax/cm−1: 1731 (C=O), 1592 (C=N); 1H-NMR (DMSO-d6): δ 8.74 (d, 1H, J = 2.3 Hz, Ar-H), 8.58 (dd, 1H, J = 8.4 Hz, J = 2.3 Hz , Ar-H), 8.57 (s, 1H, HC=N), 8.20 (d, 1H, J = 8.4 Hz, Ar-H), 7.54–7.45 (m, 3H, Ar-H), 7.41–7.36 (m, 2H, Ar-H), 4.14 (s, 2H, CH2); 13C-NMR (DMSO-d6): δ 172.6 (C), 170.1 (C), 152.8 (CH), 148.4 (C), 148.3 (C), 135.3 (C), 133.7 (C), 131.2 (CH), 129.4 (2CH), 129.3 (CH), 128.7 (2CH), 128.1 (CH), 120.6 (CH), 33.1 (CH2); Anal. Calcd for C16H11N5O5S: C, 49.87; H, 2.88; N, 18.17; S, 8.32.Found: C, 49.99; H, 2.87; N, 17.93; S, 8.25.
2-(3-(2-Nitrophenyl)allylidene)hydrazono)-3-phenylthiazolidin-4-one (6c): orange solid; mp: 265 °C; IR (KBr) νmax/cm−1: 1732 (C=O), 1627 (C=N); 1H-NMR (CDCl3): δ 8.13 (d, 1H, J = 9.5 Hz, CH=N), 8.0 (dd, 1H, J = 8.2 Hz, J = 1.0 Hz, Ar-H), 7.75–7.57 (m, 3H, Ar-H), 7.53–7.43 (m, 4H, Ar-H), 7.39–7.32 (m, 2H, Ar-H), 7.01 (dd, 1H, J = 15.8 Hz, J = 9.5 Hz, CH), 3.98 (s, 2H, CH2); 13C-NMR (CDCl3): δ 171.6 (C), 164.5 (C), 160.2 (CH), 148.00 (C), 135.8 (CH), 134.4 (C), 133.3 (2CH), 131.4 (C), 130.1 (CH), 129.4 (2CH), 129.1 (CH), 128.3 (CH), 127.7 (2CH), 125.0 (CH), 32.5 (CH2); Anal. Calcd for C18H14N4O3S: C, 59.01; H, 3.85; N, 15.29; S, 8.75. Found: C, 58.96; H, 3.92; N, 15.18; S, 8.50.
2-(3,4-(Methylenedioxy)-6-nitrobenzaldehyde)hydrazono)-3-phenylthiazolidin-4-one (6d): yellow solid; mp: 262 °C; IR (KBr) νmax/cm−1: 1717 (C=O), 1602 (C=N); 1H-NMR (DMSO-d6): δ 8.47 (s, 1H, Ar-H), 7.64 (s, 1H, HC=N), 7.56–7.44 (m, 3H, Ar-H), 7.35–7.40 (m, 1H, Ar-H), 7.36 (m, 2H, Ar-H), 6.27 (s, 2H , CH2), 4.10 (s, 2H, CH2); 13C-NMR (DMSO-d6): δ 172.5 (C), 167.7 (C), 154.0 (CH), 152.0 (C), 149.8 (C), 144.0 (C), 135.4 (C), 129.6 (2CH), 129.3 (CH), 128.7 (2CH), 125.4 (C), 106.6 (CH), 105.6 (CH), 104.4 (CH2), 32.9 (CH2); Anal. Calcd for C17H12N4O5S: C, 53.12; H, 3.15; N, 14.58; S, 8.34. Found: C, 53.15; H, 3.10; N, 14.67; S, 8.26.
2-((3,4,5-Trimethoxybenzylidene)hydrazono)-3-phenylthiazolidin-4-one (6e): white solid; mp: 174 °C; IR (KBr) νmax/cm−1: 1720 (C=O), 1619 (C=N); 1H-NMR (DMSO-d6): δ 8.21 (s, 1H, HC=N), 7.55–7.44 (m, 3H, Ar-H), 7.39–7.35 (m, 2H, Ar-H), 7.06 (s, 2H, Ar-H), 4.08 (s, 2H, CH2) , 3.78 (s, 6H, OCH3), 3.70 (s, 3H, OCH3); 13C-NMR (DMSO-d6): δ 172.6 (C), 165.4 (C), 158.0 (CH), 153.6 (2C), 140.3 (C), 135.5 (C), 130.0 (C), 129.5 (2CH), 129.1 (CH), 128.7 (2CH), 105.5 (2CH), 60.6 (CH3), 56.3 (2CH3), 32.7 (CH2); Anal. Calcd for C19H19N3O4S: C, 59.21; H, 4.97; N, 10.90; S, 8.32. Found: C, 59.29; H, 5.08; N, 10.64; S, 8.21.
2-(4-(Methylsulfonyl)benzylidene)hydrazono)-3-phenylthiazolidin-4-one (6f): yellow solid; mp: 277 °C; IR (KBr) νmax/cm−1: 1734 (C=O), 1615 (C=N); 1H-NMR (DMSO-d6): δ 8.43 (s, 1H, HC=N), 8.00 (d, 2H, J = 9.2 Hz, Ar-H), 7.94 (d, 2H, J = 9.2 Hz, Ar-H), 7.54–7.44 (m, 3H, Ar-H), 7.41–7.36 (m, 2H, Ar-H), 4.12 (s, 2H, CH2), 3.22 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 172.5 (C), 167.8 (C), 156.76 (CH), 142.5 (C), 139.2 (C), 135.4 (C), 129.6 (2CH), 129.2 (CH), 128.7 (4CH) , 128.0 (2CH), 43.8 (CH3), 32.9 (CH2); Anal. Calcd for C17H15N3O3S2: C, 54.67; H, 4.05; N, 11.25; S, 17.17. Found: C, 54.71; H, 4.05; N, 11.19; S, 17.14.
2-(4-(Methylthio)benzylidene)hydrazono)-3-phenylthiazolidin-4-one (6g): yellow solid; mp: 223 °C; IR (KBr) νmax/cm−1: 1732 (C=O), 1609 (C=N); 1H-NMR (CDCl3): δ 8.25 (s, 1H, HC=N), 7.64 (d, 2H, J = 8.5 Hz, Ar-H), 7.53–7.45 (m, 3H, Ar-H), 7.38–7.34 (m, 2H, Ar-H), 7.21 (d, 2H, J = 8.5 Hz, Ar-H), 3.97 (s, 2H, CH2), 2.50 (s, 3H, CH3); 13C-NMR (CDCl3): δ 171.8 (C), 163.9 (C), 158.4 (CH), 142.6 (C), 134.5 (C), 130.7 (C), 129.4 (2CH), 129.1 (CH), 128.4 (2CH) , 127.8 (2CH), 125.8 (2CH), 32.5 (CH2), 15.2 (CH3); Anal. Calcd for C17H15N3OS2: C, 59.80; H, 4.43; N, 12.31; S, 18.78. Found: C, 59.67; H, 4.43; N, 12.20; S, 18.88.
2-(4-(Trifluoromethyl)benzylidene)hydrazono)-3-phenylthiazolidin-4-one (6h): white solid; mp: 206 °C; IR (KBr) νmax/cm−1: 1732 (C=O), 1616 (C=N); 1H-NMR (CDCl3): δ 8.33 (s, 1H, HC=N), 7.84 (d, 2H, J = 8.2 Hz, Ar-H), 7.65 (d, 2H, J = 8.2 Hz, Ar-H), 7.59–7.43 (m, 3H, Ar-H), 7.39–7.32 (m, 2H, Ar-H), 3.99 (s, 2H, CH2); 13C-NMR (CDCl3): δ 171.7 (C), 165.8 (C), 157.3 (CH), 137.4 (C), 134.4 (C), 132 (q, J = 32.4 Hz, C), 129.4 (2CH), 129.2 (CH), 128.2 (2CH), 127.8 (3CH), 125.6 (q, J = 3.8 Hz, CH), 123.8 (q, J = 272.3 Hz, C), 32.5 (CH2); Anal. Calcd for C17H12F3N3OS: C, 56.19; H, 3.33; N, 11.56; S, 8.82.Found: C, 56.17; H, 3.66; N, 12.82; S, 9.96.
3.4. General Procedure for the Preparation of Compounds 8a–h
An equimolar mixture of 4a–h (1.5 mmol) and diethyl acetylenedicarboxylate (1.5 mmol) in methanol (20 mL) was refluxed for 1 h. After completion of the reaction, the reaction mixture was allowed to cool to the room temperature. The solid thus separated was collected by filtration and recrystallized using ethanol-DMF mixture.
(Z)-Ethyl-2-(2-((4-nitrobenzylidene)hydrazono)-4-oxo-3-phenylthiazolidin-5-ylidene) acetate (8a): yellow solid; mp: 206 °C; IR (KBr) νmax/cm−1: 1730 (C=O), 1692 (C=O), 1623 (C=N); 1H-NMR (DMSO-d6): δ 8.60 (s, 1H, CH=N), 8.32 (d, 2H, J = 8.8 Hz, Ar-H), 8.01 (d, 2H, J = 8.8 Hz, Ar-H), 7.58–7.48 (m, 5H, Ar-H), 6.80 (s, 1H, C=CH), 4.28 (q, 2H, J = 7.0 Hz, CH2), 1.28 (t, 3H, J = 7.0 Hz, CH3). 13C-NMR (DMSO-d6): δ 166.0 (C), 164.6 (C), 163.6 (C), 158.5 (CH), 149.2 (C), 141.7 (C), 140.0 (C), 134.5 (C), 129.6 (3CH), 129.5 (2CH), 128.6 (2CH), 124.6 (2CH), 116.2 (CH), 62.0 (CH2), 14.5 (CH3); Anal. Calcd for C20H16N4O5S: C, 56.60; H, 3.80; N, 13.20; S, 7.55. Found: C, 55.68; H, 4.59; N, 10.67; S, 5.44.
(Z)-Ethyl-2-(2-((E)-2,4-dinitrobenzylideneamino)-4-oxo-3-phenylthiazolidin-5-ylidene) acetate (8b): yellow solid; mp: 193 °C; IR (KBr) νmax/cm−1: 1727 (C=O), 1698 (C=O), 1620 (C=N); 1H-NMR (DMSO-d6): δ 8.77 (d, 1H, J = 2.2 Hz, Ar-H), 8.7 (s, 1H, CH=N), 8.64 (dd, 1H, J = 8.6 Hz, J = 2.2 Hz, Ar-H), 8.25 (d, 1H, J = 8.6 Hz, Ar-H), 7.60-7.46 (m, 5H, Ar-H), 6.83 (s, 1H, C=CH), 4.29 (q, 2H, J = 7.1 Hz, CH2), 1.28 (t, 3H, J = 7.1 Hz, CH3); 13C-NMR (DMSO-d6): δ 165.9 (C), 165.2 (C), 164.7 (C), 155.2 (CH), 148.6 (C), 141.5 (C), 134.5 (C), 133.3 (C), 131.4 (CH), 129.8 (CH), 129.7 (3CH), 128.7 (2CH), 128.3 (CH), 120.7 (CH), 116.6 (CH), 62.1 (CH2), 14.5 (CH3); Anal. Calcd for C20H15N5O7S: C, 51.17; H, 3.22; N, 14.92; S, 6.83. Found: C, 51.29; H, 3.15; N, 14.78; S, 6.77.
(Z)-Ethyl-2-(2-((E)-3-(2-nitrophenyl)allylidene)amino)-4-oxo-3-phenylthiazolidin-5-ylidene) acetate (8c): yellow solid; mp: 251 °C; IR (KBr) νmax/cm−1: 1727 (C=O), 1692 (C=O), 1618 (C=N); 1H-NMR (DMSO-d6): δ 8.27 (d, 1H, J = 9.4 Hz, CH), 8.01 (ddd, 2H, J = 7.1 Hz, J = 7.9 Hz, J = 1.1 Hz, Ar-H), 7.73 (dd, 1H, J = 7.9 Hz, J = 7.1 Hz, Ar-H), 7.63-7.42 (m, 7H, Ar-H), 7.20 (dd, 1H, J = 15.7 Hz, J = 9.4 Hz, Ar-H), 6.77 (s, 1H, C=CH), 4.27 (q, 2H, J = 7.1 Hz, CH2), 1.27 (t, 3H, J = 7.1 Hz, CH3); 13C-NMR (DMSO-d6): δ 166.0 (C), 164.6 (C), 162.1 (CH), 161.5 (C), 148.6 (C), 141.9 (C), 137.3 (CH), 134.6 (C), 134.0 (CH), 130.7 (CH), 130.4 (C), 129.8 (CH), 129.6 (3CH), 129.0 (CH), 128.6 (2CH), 125.0 (CH), 115.9 (CH), 62.0 (CH2), 14.5 (CH3). Anal. Calcd for C22H18N4O5S: C, 58.66; H, 4.03; N, 12.44; S, 7.12. Found: C, 58.47; H, 3.92; N, 12.05; S, 6.64
(Z)-Ethyl-2-(2-((E)-(4-nitrobenzo[d][1,3]dioxol-5-yl)methyleneamino)-4-oxo-3-phenylthiazolidin-5-ylidene) acetate (8d): yellow solid; mp: 251 °C; IR (KBr) νmax/cm−1: 1721 (C=O) 1702 (C=O), 1597 (C=N); 1H-NMR (DMSO-d6): δ 8.59 (s, 1H, Ar-H), 7.70 (s, 1H, Ar-H), 7.54–7.47 (m, 5H, Ar-H), 7.38 (s, 1H, Ar-H), 6.80 (s, 1H, C=CH), 6.30 (s, 2H, CH2 ), 4.27 (q, 2H, J = 7.1 Hz, CH2), 1.27 (t, 3H, J = 7.1 Hz, CH3); 13C-NMR (DMSO-d6): δ 166.0 (C), 164.7 (C), 163.0 (C), 156.5 (CH), 152.1 (C), 150.3 (C), 144.3 (C), 141.8 (C), 137.2 (C), 134.6 (C), 129.7 (2CH), 128.7 (2CH), 124.9 (CH), 116.2 (CH), 106.8 (CH), 105.8 (CH), 104.6 (CH2), 62.0 (CH2), 14.5 (CH3); Anal. Calcd for C21H16N4O7S: C, 53.84; H, 3.44; N, 11.96; S, 6.85. Found: C, 53.79; H, 3.42; N, 11.98; S, 6.75.
(Z)-Ethyl-2-(2-((E)-3,4,5-trimethoxybenzylideneamino)-4-oxo-3-phenylthiazolidin-5-ylidene) acetate (8e): yellow solid; mp: 145 °C; IR (KBr) νmax/cm−1: 1721 (C=O), 1689 (C=O), 1622 (C=N); 1H-NMR (DMSO-d6): δ 8.35 (s, 1H, CH=N), 7.58–7.47 (m, 5H, Ar-H), 7.11 (s, 2H, Ar-H), 6.77 (s, 1H, C=CH), 4.27 (q, 2H, J = 7.0 Hz, CH2), 3.80 (s, 6H, OCH3), 3.70 (s, 3H, OCH3), 1.28 (t, 3H, J = 7.0 Hz, CH3); 13C-NMR (DMSO-d6): δ 165.9 (C), 164.6 (C), 160.6 (C), 160.4 (CH), 153.6 (2C), 142.0 (C), 140.9 (C), 134.7 (C), 129.6 (3CH), 129.4 (C), 128.6 (2CH), 115.7 (CH), 105.9 (2CH), 62.0 (CH2), 60.6 (CH3), 56.3 (2CH3), 14.5 (CH3); Anal. Calcd for C23H23N3O6S: C, 58.84; H, 4.94; N, 8.95; S, 6.83. Found: C, 58.47; H, 4.93; N, 8.77; S, 6.64.
(Z)-Ethyl-2-(2-((E)-4-(methylsulfonyl)benzylideneamino)-4-oxo-3-phenylthiazolidin-5-ylidene) acetate (8f): yellow solid; mp: 254 °C; IR (KBr) νmax /cm−1: 1722 (C=O), 1687 (C=O), 1611 (C=N); 1H-NMR (DMSO-d6): δ 8.58 (s, 1H, CH=N), 8.02 (s, 4H, Ar-H), 7.59–7.45 (m, 5H, Ar-H), 6.81 (s, 1H, C=CH), 4.29 (q, 2H, J = 7.1 Hz, CH2), 3.24 (s, 3H, CH3), 1.28 (t, 3H, J = 7.1 Hz, CH3); 13C-NMR (DMSO-d6): δ 166.0 (C), 164.7 (C), 163.2 (C), 159.0 (CH), 143.0 (C), 141.8 (C), 138.6 (C), 134.6 (C), 129.7 (3CH), 129.1 (2CH), 128.7 (2CH), 128.1 (2CH), 116.1 (CH), 62.1 (CH2), 43.9 (CH3), 14.5 (CH3); Anal. Calcd for C21H19N3O5S2: C, 55.13; H, 4.19; N 9.18; S, 14.02. Found: C, 55.18; H, 4.19; N, 8.96; S, 14.11.
(Z)-Ethyl-2-(2-((E)-4-(methylthio)benzylideneamino)-4-oxo-3-phenylthiazolidin-5-ylidene)acetate (8g): yellow solid; mp: 210 °C; IR (KBr) νmax/cm−1: 1720 (C=O), 1696 (C=O), 1604 (C=N); 1H-NMR (DMSO-d6): δ 8.40 (s, 1H, CH=N), 8.70 (d, 2H, J = 8.4 Hz, Ar-H), 7.58–7.44 (m, 5H, Ar-H), 7.33 (d, 2H, J = 8.4 Hz, Ar-H), 6.77 (s, 1H, C=CH), 4.28 (q, 2H, J = 7.1 Hz, CH2), 3.31 (s, 3H, CH3), 1.28 (t, 3H, J = 7.1 Hz, CH3); 13C-NMR (DMSO-d6): δ 166.0 (C), 164.6 (C), 160.9 (C), 160.0 (CH), 143.5 (C), 142.1 (C), 134.7 (C), 130.3 (C), 129.6 (3CH), 128.9 (2CH), 128.7 (2CH), 126.0 (2CH), 115.6 (CH), 62.0 (CH2), 14.6 (CH3), 14.5 (CH3); Anal. Calcd for C21H19N3O3S2: C, 59.27; H, 4.50; N 9.87; S, 15.07. Found: C, 59.10; H, 4.48; N, 9.74; S, 14.86.
(Z)-Ethyl-2-(2-((E)-4-(trifluoromethyl)benzylideneamino)-4-oxo-3-phenylthiazolidin-5-ylidene) acetate (8h): yellow solid; mp: 154 °C; IR (KBr) νmax/cm−1: 1721 (C=O), 1690 (C=O), 1619 (C=N); 1H-NMR (DMSO-d6): δ 8.56 (s, 1H, CH=N), 8.00 (d, 2H, J = 8.2 Hz, Ar-H), 7.84 (d, 2H, J = 8.2 Hz, Ar-H), 7.58-7.45 (m, 5H, Ar-H), 6.80 (s, 1H, C=CH), 4.30 (q, 2H, J = 7.1 Hz, CH2), 1.28 (t, 3H, J = 7.1 Hz, CH3); 13C-NMR (DMSO-d6): δ 166.0 (C), 164.6 (C), 162.9 (C), 159.1 (CH), 141.9 (C), 137.9 (C), 134.6 (C), 131.3 (q, J = 32.2 Hz, C), 129.6 (q, J = 272.2 Hz, C), 129.5 (3CH), 129.1 (2CH), 128.7 (2CH), 126.3 (q, J = 3.2Hz, 2CH), 116.0 (CH), 62.0 (CH2), 14.5 (CH3); Anal. Calcd for C21H16F3N3O3S: C, 56.37; H, 3.60; N, 9.39; S, 7.17. Found: C, 56.33; H, 3.51; N, 9.17; S, 7.10.
4. Conclusions
In conclusion, we have prepared a series of 4-phenyl-3-thiosemicarbazone derivatives from 4-phenyl-3-thiosemicarbazide and various aromatic aldehydes substituted with different electron-donor and -withdrawing groups. In a second step, these 4-phenyl-3-thiosemicarbazone derivatives were reacted with 2-ethyl bromoacetate and diethyl acetylenedicarboxylate, respectively, to afford an original series of highly functionalized thiazolidinone derivatives in good yields. The antiparasitic and antibacterial evaluations of all synthesized compound are under investigation.
Acknowledgments
This work was supported by the CNEPRU (Ministry of Higher Education and Scientific Research Algeria). We express our thanks to Vincent Remusat for recording the 1H and 13C-NMR spectra.
Author Contributions
A.D.; T.T. and P.V. conceived and designed the study. A.B. and O.K. designed the experiments and interpreted the results. A.B.; O.K.; A.D.; T.T. and P.V. wrote the manuscript.
Conflictts of Interest
The authors declare no conflict of interest.
References and Notes
- Pirrung, M.C.; Pansare, S.V.; das Sarma, K.; Keith, K.A.; Kern, E.R. Combinatorial optimization of isatin-β-thiosemicarbazones as anti-poxvirus agents. J. Med. Chem. 2005, 48, 3045–3050. [Google Scholar] [CrossRef]
- Hu, W.-X.; Zhou, W.; Xia, C.-N.; Wen, X. Synthesis and anticancer activity of thiosemicarbazones. Bioorg. Med. Chem. Lett. 2006, 16, 2213–2218. [Google Scholar] [CrossRef]
- Kolocouris, A.; Dimas, K.; Pannecouque, C.; Witvrouw, M.; Foscolos, G.B.; Stamatiou, G.; Fytas, G.; Zoidis, G.; Kolocouris, N.; Andrei, G.; et al. New 2-(1-adamantylcarbonyl) pyridine and 1-acetyladamantane thiosemicarbazones-thiocarbonohydrazones: Cell growth inhibitory, antiviral and antimicrobial activity evaluation. Bioorg. Med. Chem. Lett. 2002, 12, 723–727. [Google Scholar] [CrossRef]
- Tarasconi, P.; Capacchi, S.; Pelosi, G.; Cornia, M.; Albertini, R.; Bonati, A.; Dall’Aglio, P.P.; Lunghi, P.; Pinelli, S. Synthesis, spectroscopic characterization and biological properties of new natural aldehydes thiosemicarbazones. Bioorg. Med. Chem. 2000, 88, 157. [Google Scholar]
- Pérez-Rebolledo, A.; Teixeira, L.R.; Batista, A.A.; Mangrich, A.S.; Aguirre, G.; Cerceretto, H.; González, M.; Hernández, P.; Ferreira, A.M.; Speziali, N.L.; et al. 4-Nitroacetophenone-derived thiosemicarbazones and their copper (II) complexes with significant in vitro anti-trypanosomal activity. Eur. J. Med. Chem. 2008, 43, 939–948. [Google Scholar] [CrossRef]
- Aguirre, G.; Boiani, L.; Cerecetto, H.; Fernández, M.; González, M.; Denicola, A.; Otero, L.; Gambino, D.; Rigol, C.; Olea-Azar, C.; et al. In vitro activity and mechanism of action against the protozoan parasite Trypanosoma cruzi of 5-nitrofuryl containing thiosemicarbazones. Bioorg. Med. Chem. 2004, 12, 4885–4893. [Google Scholar] [CrossRef]
- Du, X.; Guo, C.; Hansell, E.; Doyle, P.S.; Caffrey, C.R.; Holler, T.P.; McKerrow, J.H.; Cohen, E. Synthesis and structure-activity relationship study of potent trypanocidal thio- semicarbazone inhibitors of the trypanosomal cysteine protease cruzain. J. Med. Chem. 2002, 45, 2695–2707. [Google Scholar] [CrossRef]
- De Aquino, T.M.; Liesen, A.P.; da Silva, R.E.A.; Lima, V.T.; Carvalho, C.S.; de Faria, A.R.; de Araujo, J.M.; de Lima, J.G.; Alves, A.J.; de Melo, E.J.T.; et al. Synthesis, anti-Toxoplasma-gondii and antimicrobial activities of benzaldehyde 4-phenyl-3-thiosemicarbazones and 2-[(phenymethylene)hydrazono]-4-oxo-3-phenyl-5-thiazolidine acetic acids. Bioorg. Med. Chem. 2008, 16, 446–456. [Google Scholar] [CrossRef]
- Mayekar, S.A.; Mulwad, V.V. Synthesis and antibacterial activity of 6-(5phenyl-{1,3,4} thiadiazol-2-ylimino)-benzopyran-2-ones. Ind. J. Chem. 2008, 47, 1438–1442. [Google Scholar]
- Omar, K.; Geronikaki, A.; Zoumpoulakis, P.; Camoutsis, C.; Sokovic, M.; Ciric, A.; Glamoclija, J. Novel 4- thiazolidinone derivatives as potential antifungal and antibacterial drugs. Bioorg. Med. Chem. 2010, 18, 426–432. [Google Scholar] [CrossRef]
- Bhat, M.A.; Siddiqui, N.; Khan, S.A. Synthesis, anticonvulsant and neurotoxicity screening of 2-(substituted phenyl)-3-[3-(2-oxo-2H-chromen-3-yl)-5-thioxo-1,5-dihydro-4H-1,2,4-triazol-4-yl]-1,3-thiazolidin-4-ones. Ind. J. Het. Chem. 2008, 17, 287–288. [Google Scholar]
- Babaoglu, K.; Page, M.A.; Jones, V.C.; McNeil, M.R.; Dong, C.; Naismith, J.H.; Lee, R.E. Novel inhibitors of an emerging target in mycobacterium tuberculosis; substituted thiazolidinones as inhibitors of dTDP-rhamnose synthesis. Bioorg. Med. Chem. Lett. 2003, 13, 3227–3230. [Google Scholar] [CrossRef]
- Vigorita, M.G.; Ottanà, R.; Monforte, F.; Maccari, R.; Monforte, M.T.; Trovato, A.; Taviano, M.F.; Miceli, N.; de Luca, G.; Alcaro, S.; et al. Chiral 3,3'-(1,2-ethanediyl)-bis[2-(3,4-dimethoxyphenyl)-4-thiazolidinones] with anti-inflammatory activity. Part 11: Evaluation of COX-2 selectivity and modeling. Bioorg. Med. Chem. 2003, 11, 999–1006. [Google Scholar] [CrossRef]
- Agrawal, V.K.; Sachan, S.; Khadikar, P.V. QSAR studies on antihistaminic activity of some thiazolidine-4-ones. Acta Pharm. 2000, 50, 281–290. [Google Scholar]
- Diurno, M.V.; Mazzoni, O.; Correale, G.; Monterrey, I.G.; Calignano, A.; la Rana, G.; Bolognese, A. Synthesis and structure-activity relationships of 2-(substituted phenyl)-3-[3-(N,N-dimethylamino)-propyl]-1,3-thiazolidin-4-ones acting as H1-histamine antagonists. Farmaco 1999, 54, 579–583. [Google Scholar] [CrossRef]
- Suzuki, Y.; Akima, M.; Tamura, K. Effects of CP-060S, a novel cardioprotective drug, on cardiac function and myocardial oxygen consumption. Gen. Pharmacol. 1999, 32, 57–63. [Google Scholar] [CrossRef]
- Rawal, R.K.; Prabhakar, Y.S.; Katti, S.B.; de Clercq, E. 2-(Aryl)-3-furan-2-ylmethyl-thiazolidin-4-ones as selective HIV-RT inhibitors. Bioorg. Med. Chem. 2005, 13, 6771–6776. [Google Scholar] [CrossRef]
- Crozet, M.P.; Archaimbault, G.; Vanelle, P.; Nouguier, R. Reaction SRN1 en serie heterocyclique: Reactivite des sels du dimethyl-2,2-nitro-5-dioxane-1,3. Tetrahedron Lett. 1985, 26, 5133–5134. [Google Scholar] [CrossRef]
- Amiri-Attou, O.; Terme, T.; Vanelle, P. Functionalization of 6-nitrobenzo[1,3]dioxole with carbonyl compounds via TDAE methodology. Molecules 2005, 10, 545–551. [Google Scholar] [CrossRef]
- Verhaeghe, P.; Azas, N.; Hutter, S.; Castera-Ducros, C.; Laget, M.; Dumètre, A.; Gasquet, M.; Reboul, J.P.; Rault, S.; Rathelot, P.; et al. Synthesis and in vitro antiplasmodial evaluation of 4-anilino-2-trichloromethylquinazolines. Bioorg. Med. Chem. 2009, 17, 4313–4322. [Google Scholar] [CrossRef]
- Crozet, M.D.; Botta, C.; Gasquet, M.; Curti, C.; Remusat, V.; Hutter, S.; Chapelle, O.; Azas, N.; de Méo, M.; Vanelle, P. Lowering of 5-nitroimidazole’s mutagenicity: Towards optimal antiparasitic pharmacophore. Eur. J. Med. Chem. 2009, 44, 653–659. [Google Scholar] [CrossRef]
- Kabri, Y.; Azas, N.; Dumètre, A.; Hutter, S.; Laget, M.; Verhaeghe, P.; Gellis, A.; Vanelle, P. Original quinazoline derivatives displaying antiplasmodial properties. Eur. J. Med. Chem. 2010, 45, 616–622. [Google Scholar] [CrossRef]
- Huang, H.; Chen, Q.; Ku, X.; Meng, L.; Lin, L.; Wang, X.; Zhu, C.; Wang, Y.; Chen, Z.; Li, M.; et al. A series of α-heterocyclic carboxaldehyde thiosemicarbazones inhibit topoisomerase IIα catalytic activity. J. Med. Chem. 2010, 53, 3048–3064. [Google Scholar] [CrossRef]
- Serda, M.; Malecki, J.G.; Mrozek-Wilczkiewicz, A.; Musiol, R.; Polanski, J. Microwave assisted synthesis, X-ray crystallography and DFT calculations of selected aromatic thiosemicarbazones. J. Mol. Struct. 2013, 1037, 63–72. [Google Scholar] [CrossRef]
- Küçügüzel, G.; Kocayepe, A.; de Clercq, E.; Sahin, F.; Güllüce, M. Synthesis and biological activity of 4-thiazolidinones, thiosemicarbazides derived from diflunisal hydrazide. Eur. J. Med. Chem. 2006, 41, 353–359. [Google Scholar] [CrossRef]
- Vas’kevich, R.I.; Zborovskii, Y.L.; Staninets, V.I.; Chernega, A.N. Reaction of 4-Aryl-1-(4-oxo-3,4-dihydrothieno[2,3-d]pyrimidin-2-yl)thiosemicarbazides with Dimethyl Acetylenedicarboxylate. Russ. J. Org. Chem. 2004, 40, 1047–1052. [Google Scholar] [CrossRef]
- CCDC contains the supplementary crystallographic data of compound 8b for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via http://www.ccdc.cam.ac.uk/data_request/cif.
- CCDC contains the supplementary crystallographic data of compound 8g for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via http://www.ccdc.cam.ac.uk/data_request/cif.
- Sample Availability: Samples of the compounds 4a–h, 6a–h and 8a–h are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
