Synthesis and Cytotoxic Evaluation of Alkoxylated Chalcones
Abstract
:1. Introduction
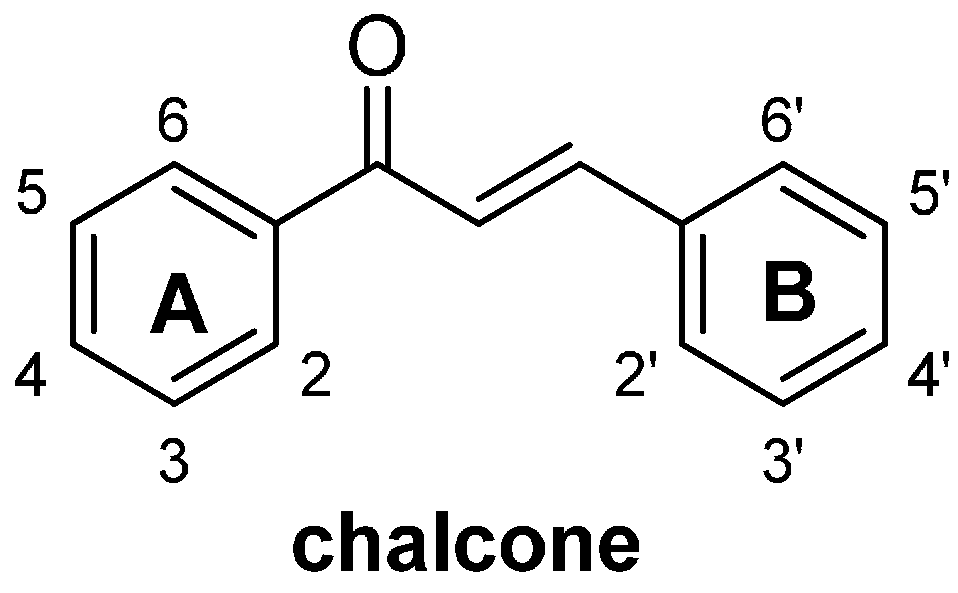
2. Results and Discussion
| Compound | R1 | R2 | R3 | R4 | R5 | Inhibition (%) ** |
|---|---|---|---|---|---|---|
| a1 | H | H | H | H | H | 42.4 ± 2.0 |
| a2 | H | H | Br | H | H | 37.6 ± 4.4 |
| a3 | F | H | Br | H | H | 41.8 ± 5.9 |
| a4 * | Br | H | F | H | H | 41.2 ± 4.6 |
| a5 * | Cl | H | Br | H | H | 9.0 ± 5.6 |
| a6 | H | H | NO2 | H | H | 9.8 ± 5.0 |
| a7 | H | H | COOH | H | H | 32.7 ± 3.0 |
| a8 | OCH3 | H | H | H | H | 61.4 ± 3.4 |
| a9 | H | OCH3 | H | H | H | 59.0 ± 2.9 |
| a10 | H | H | OCH3 | H | H | 27.7 ± 17.7 |
| a11 | H | H | OCH2CH3 | H | H | 13.1 ± 8.4 |
| a12 | OCH3 | OCH3 | H | H | H | 66.3 ± 4.2 |
| a13 | OCH3 | H | H | OCH3 | H | 77.0 ± 2.2 |
| a14 * | OCH3 | H | H | H | OCH3 | 78.1 ± 4.7 |
| a15 | H | OCH3 | OCH3 | H | H | 65.0 ± 17.6 |
| a16 | H | OCH3 | H | OCH3 | H | 70.6 ± 2.0 |
| a17 | H | OCH2Ph | OCH3 | H | H | 44.1 ± 6.2 |
| a18 | OCH3 | OCH3 | OCH3 | H | H | 31.8 ± 7.3 |
| a19 | OCH3 | H | OCH3 | OCH3 | H | 38.1 ± 10.3 |
| a20 | H | OCH3 | OCH3 | OCH3 | H | 71.1 ± 1.3 |
| Etoposide | 63.9 ± 0.1 |

| Compound | R1 | R2 | R3 | R4 | Human Cancer Cell Lines, IC50 (µM) & SI | Normal Human Cell Lines, CC50 (µM)b | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HepG2 | HeLa | MCF7 | A549 | SW1990 | HMLE | L02 | ||||||||||
| IC50 a | IC50 a | IC50 a | IC50 a | IC50 a | ||||||||||||
| SI c | SI d | SI e | SI f | SI g | SI h | SI i | SI j | SI k | SI l | |||||||
| b1 # | H | H | H | H | 20.98 ± 1.75 | >30 | 28.18 ± 0.04 | >30 | >30 | 9.47 ± 0.39 | 33.340.39e | |||||
| 0.5 | 1.6 | <0.3 | <1.1 | 0.3 | 1.2 | <0.3 | <1.1 | <0.3 | <1.1 | |||||||
| b2 | CH3 | H | H | H | 14.07 ± 1.84 | 18.40 ± 0.75 | 19.08 ± 2.11 | 25.47 ± 4.14 | >30 | 14.98 ± 0.78 | 35.87 ± 3.40 | |||||
| 1.1 | 2.5 | 0.8 | 1.9 | 0.8 | 1.9 | 0.6 | 1.4 | <0.5 | <1.2 | |||||||
| b3 | H | CH3 | H | H | 12.81 ± 0.50 | 13.28 ± 2.14 | 26.78 ± 4.26 | 20.69 ± 0.59 | 25.23 ± 2.00 | 16.20 ± 0.19 | 33.26 ± 1.61 | |||||
| 1.3 | 2.6 | 1.2 | 2.5 | 0.6 | 1.2 | 0.8 | 1.6 | 0.6 | 1.3 | |||||||
| b4 | H | H | CH3 | H | 19.51 ± 1.28 | 18.60 ± 0.36 | 25.44 ± 0.15 | 22.07 ± 0.60 | >30 | 13.77 ± 1.69 | 59.59 ± 5.56 | |||||
| 0.7 | 3.1 | 0.7 | 3.2 | 0.5 | 2.3 | 0.6 | 2.7 | <0.5 | <2.0 | |||||||
| b5 | H | CH3 | CH3 | H | 26.79 ± 0.30 | 20.33 ± 1.01 | 30.31 ± 0.29 | >30 | >30 | 15.66 ± 0.76 | 25.15 ± 2.71 | |||||
| 0.6 | 0.9 | 0.8 | 1.2 | 0.5 | 0.8 | <0.5 | <0.8 | <0.5 | <0.8 | |||||||
| b6 # | OCH3 | H | H | H | 2.05 ± 0.22 | 5.00 ± 0.03 | 5.60 ± 0.17 | 6.35 ± 0.32 | 10.02 ± 0.14 | 4.01 ± 0.05 | 8.78 ± 1.21 | |||||
| 2.0 | 4.3 | 0.8 | 1.8 | 0.7 | 1.6 | 0.6 | 1.4 | 0.4 | 0.9 | |||||||
| b7 | H | OCH3 | H | H | 6.97 ± 0.55 | 9.98 ± 0.37 | 6.37 ± 0.05 | 14.02 ± 0.19 | 15.62 | 8.14 ± 0.53 | 18.08 ± 5.82 | |||||
| 1.2 | 2.6 | 0.8 | 1.8 | 1.3 | 2.8 | 0.6 | 1.3 | 0.5 | 1.2 | |||||||
| a16 | OCH3 | 9.20 ± 0.31 | 12.42 ± 0.34 | 23.10 ± 0.79 | 21.18 ± 0.55 | 17.80 ± 0.07 | 3.28 ± 0.31 | 8.87 ± 1.83 | ||||||||
| 0.4 | 1.0 | 0.3 | 0.7 | 0.1 | 0.4 | 0.2 | 0.4 | 0.2 | 0.5 | |||||||
| b8 | OCH2CH3 | H | H | H | 4.23 ± 0.09 | 8.72 ± 0.07 | 8.56 ± 0.17 | 7.36 ± 0.37 | 8.20 ± 0.12 | 0.83 ± 0.21 | 39.55 ± 7.53 | |||||
| 0.2 | 9.3 | 0.1 | 4.5 | 0.1 | 4.6 | 0.1 | 5.4 | 0.1 | 4.8 | |||||||
| b9 | H | OCH2CH3 | H | H | 7.71 ± 0.21 | 12.16 ± 0.25 | 11.59 ± 0.07 | 13.22 ± 0.43 | 10.68 ± 0.08 | 9.58 ± 0.44 | 17.42 ± 5.96 | |||||
| 1.2 | 2.3 | 0.8 | 1.4 | 0.8 | 1.5 | 0.7 | 1.3 | 0.9 | 1.6 | |||||||
| b10 | H | H | OCH2CH3 | H | 10.92 ± 0.46 | 14.36 ± 0.15 | 19.76 ± 0.69 | 22.72 ± 0.57 | 19.47 ± 0.23 | 13.81 ± 3.77 | 36.66 ± 3.41 | |||||
| 1.3 | 3.4 | 1.0 | 2.6 | 0.7 | 1.9 | 0.6 | 1.6 | 0.7 | 1.9 | |||||||
| b11 | OCH2Ph | H | H | H | 2.76 ± 0.04 | 3.62 ± 0.07 | 4.15 ± 0.2 | 3.63 ± 0.13 | 7.43 ± 0.56 | 20.99 ± 1.26 | 38.28 ± 5.63 | |||||
| 7.6 | 13.9 | 5.8 | 10.6 | 5.1 | 9.2 | 5.8 | 10.5 | 2.8 | 5.2 | |||||||
| b12 | H | OCH2Ph | H | H | 23.59 ± 0.84 | 25.59 ± 1.16 | 29.03 ± 0.09 | 13.25 ± 0.02 | 22.30 ± 0.07 | 31.40 ± 1.12 | >100 | |||||
| 1.3 | >4.2 | 1.2 | >3.9 | 1.1 | >3.4 | 2.4 | >7.5 | 1.4 | >7.5 | |||||||
| b13 # | H | H | OCH2Ph | H | 21.75 ± 0.62 | >30 | >30 | >30 | >30 | >100 | >100 | |||||
| >4.6 | >4.6 | - | - | - | - | - | - | - | - | |||||||
| b14 | OCH2Ph | H | OCH3 | H | >30 | >30 | >30 | >30 | >30 | >100 | >100 | |||||
| - | - | - | - | - | - | - | - | - | - | |||||||
| b15 | H | OCH3 | OCH2Ph | H | >30 | >30 | >30 | >30 | >30 | >100 | >100 | |||||
| - | - | - | - | - | - | - | - | - | - | |||||||
| b16 | H | OCH3 | OCH2CH3 | H | 4.14 ± 0.11 | 5.02 ± 0.09 | 4.79 ± 0.13 | 5.78 ± 0.11 | 7.67 ± 0.04 | 9.15 ± 1.33 | 44.70 ± 8.15 | |||||
| 2.2 | 10.8 | 1.8 | 8.9 | 1.9 | 9.3 | 1.6 | 7.7 | 1.2 | 5.8 | |||||||
| b17 | OCH2CH3 | H | OCH3 | H | 11.97 ± 0.23 | 8.54 ± 0.14 | 7.74 ± 0.10 | 9.33 ± 0.30 | 10. 80 ± 0.50 | 9.03 ± 0.80 | 10.92 ± 0.42 | |||||
| 0.8 | 0.9 | 1.1 | 1.3 | 1.2 | 1.4 | 1.0 | 1.2 | 0.8 | 1.0 | |||||||
| b18 | OCH3 | H | H | OCH3 | 1.59 ± 0.15 | 4.56 ± 0.22 | 5.60 ± 0.06 | 4.16 ± 0.72 | 9.64 ± 0.18 | 2.48 ± 0.34 | 16.97 ± 2.73 | |||||
| 1.6 | 10.7 | 0.5 | 3.7 | 0.4 | 3.0 | 0.6 | 4.1 | 0.3 | 1.8 | |||||||
| b19 # | H | OCH3 | OCH3 | H | 7.12 ± 0.67 | 9.26 ± 0.04 | 19.67 ± 0.20 | 13.62 ± 0.85 | 13.84 ± 0.06 | 10.53 ± 0.41 | 39.75 ± 6.32 | |||||
| 1.5 | 5.6 | 1.1 | 4.3 | 0.5 | 2.0 | 0.8 | 2.9 | 0.8 | 2.9 | |||||||
| b20 # | H | OCH2O | H | 17.46 ± 0.54 | 16.23 ± 0.95 | 22.54 ± 0.37 | 19.50 ± 0.90 | 25.85 ± 0.65 | 14.21 ± 1.06 | 60.90 ± 5.61 | ||||||
| 0.8 | 3.5 | 0.9 | 3.8 | 0.6 | 2.7 | 0.7 | 3.1 | 0.5 | 2.4 | |||||||
| b21 | H | CH2CH2O | H | 17.34 ± 0.23 | 21.57 ± 0.43 | 22.62 ± 2.10 | 22.26 ± 0.99 | 24.37 ± 0.60 | 14.58 ± 0.81 | 57.48 ± 13.57 | ||||||
| 0.8 | 3.3 | 0.7 | 2.7 | 0.6 | 2.5 | 0.7 | 2.6 | 0.6 | 2.4 | |||||||
| b22 | H | H | Ph | H | 0.25 ± 0.03 | 0.87 ± 0.25 | 2.40 ± 0.08 | 1.75 ± 0.15 | 1.24 ± 0.08 | 2.21 ± 0.09 | 14.54 ± 6.58 | |||||
| 8.8 | 58.2 | 2.5 | 16.7 | 0.9 | 6.1 | 1.3 | 8.3 | 1.8 | 11.7 | |||||||
| b23 # | H | NH2 | H | H | 5.70 ± 0.09 | 7.23 ± 0.07 | 8.56 ± 0.75 | 7.32 ± 0.29 | 10.07 ± 0.03 | 11.41 ± 0.76 | 43.18 ± 11.74 | |||||
| 2.0 | 7.6 | 1.6 | 6.0 | 1.3 | 5.0 | 1.6 | 5.9 | 1.1 | 4.3 | |||||||
| b24 # | OH | H | H | H | 7.77 ± 0.08 | 13.38 ± 0.74 | 10.68 ± 0.38 | 8.64 ± 0.31 | 16.11 ± 0.32 | 8.96 ± 0.44 | 24.22 ± 2.71 | |||||
| 1.2 | 3.1 | 0.7 | 1.8 | 0.8 | 2.3 | 1.0 | 2.8 | 0.6 | 1.5 | |||||||
| b25 | H | OH | H | H | 7.27 ± 0.13 | 9.56 ± 0.33 | 8.92 ± 0.25 | 12.73 ± 0.28 | 21.03 ± 0.43 | 6.31 ± 0.13 | 19.64 ± 1.29 | |||||
| 0.9 | 2.7 | 0.7 | 2.1 | 0.7 | 2.2 | 0.5 | 1.5 | 0.3 | 0.9 | |||||||
| b26 # | H | H | OH | H | 14.38 ± 0.81 | 12.73 ± 0.81 | 20.24 ± 0.30 | 23.75 ± 1.56 | 20.76 ± 0.54 | 12.55 ± 2.82 | 28.16 ± 1.69 | |||||
| 0.9 | 2.0 | 1.0 | 2.2 | 0.6 | 1.4 | 0.5 | 1.2 | 0.6 | 1.4 | |||||||
| b27 | OH | H | OH | H | 17.39 ± 1.01 | 24.84 ± 0.17 | 23.08 ± 0.82 | 17.27 ± 0.76 | >30 | >100 | 35.46 ± 5.72 | |||||
| >5.8 | 2.0 | >4.0 | 1.4 | >4.3 | 1.5 | >5.8 | 2.1 | - | <1.2 | |||||||
| b28 # | OH | H | OCH3 | H | 19.52 ± 0.79 | 17.02 ± 0.65 | 28.37 ± 1.25 | 17.37 ± 0.97 | 28.30 ± 0.26 | 16.25 ± 2.48 | >100 | |||||
| 0.8 | >5.1 | 1.0 | >5.9 | 0.6 | >3.5 | 0.9 | >5.8 | 0.6 | >3.5 | |||||||
| b29 | H | OCH3 | OH | H | 0.97 ± 0.04 | 1.83 ± 0.08 | 1.79 ± 0.20 | 1.50 ± 0.07 | 3.03 ± 0.05 | 6.31 ± 0.13 | >100 | |||||
| 6.5 | >103.1 | 3.4 | >54.6 | 3.5 | >55.9 | 4.2 | >66.7 | 2.1 | >33.0 | |||||||
| b30 | H | H | CN | H | 20.45 ± 0.21 | >30 | >30 | >30 | >30 | 12.55 ± 2.82 | >100 | |||||
| 0.6 | >4.9 | <0.4 | - | <0.4 | - | <0.4 | - | <0.4 | - | |||||||
| b31 | F | H | H | H | 17.98 ± 0.88 | 23.33 ± 0.42 | 23.51 ± 0.98 | 27.44 ± 0.67 | >30 | >100 | >100 | |||||
| >5.6 | >5.6 | >4.3 | >4.3 | >4.3 | >4.3 | >3.6 | >3.6 | - | - | |||||||
| b32 | H | F | H | H | 21.56 ± 0.97 | 20.67 ± 0.78 | 25.52 ± 0.78 | 27.15 ± 1.39 | 28.38 ± 0.27 | 11.91 ± 1.28 | 72.01 ± 12.10 | |||||
| 0.6 | 3.3 | 0.6 | 3.5 | 0.5 | 2.8 | 0.4 | 2.7 | 0.4 | 2.5 | |||||||
| b33 | H | H | F | H | 24.55 ± 0.36 | 17.41 ± 0.39 | 32.22 ± 1.34 | >30 | >30 | 12.73 ± 2.35 | 63.86 ± 13.78 | |||||
| 0.5 | 2.6 | 0.7 | 3.7 | 0.4 | 2.0 | <0.4 | <2.1 | <0.4 | <2.1 | |||||||
| b34 | H | Cl | H | H | 13.45 | 16.55 | 21.97 | 16.08 | 21.44 | 9.42 ± 1.21 | 46.87 ± 13.91 | |||||
| 0.7 | 3.5 | 0.6 | 2.8 | 0.4 | 2.1 | 0.6 | 2.9 | 0.4 | 2.2 | |||||||
| b35 # | H | H | Cl | H | 27.09 ± 0.88 | 18.40 ± 0.06 | 29.76 ± 0.80 | >30 | >30 | 17.71 ± 1.35 | 85.43 ± 6.86 | |||||
| 0.7 | 3.2 | 1.0 | 4.6 | 0.6 | 2.9 | <0.6 | <2.8 | <0.6 | <2.8 | |||||||
| b36 | H | Br | H | H | 11.30 ± 1.45 | 13.38 ± 0.44 | 23.33 ± 0.79 | 25.03 ± 3.17 | 17.13 ± 0.57 | 13.07 ± 1.21 | 61.24 ± 12.11 | |||||
| 1.2 | 5.4 | 1.0 | 4.6 | 0.6 | 2.6 | 0.5 | 2.4 | 0.8 | 3.6 | |||||||
| b37 | Cl | H | Cl | H | 11.73 ± 0.18 | 15.69 ± 1.09 | 30.35 ± 0.71 | >30 | 15.36 ± 0.83 | 19.82 ± 1.44 | >100 | |||||
| 1.7 | >8.5 | 1.3 | >6.4 | 0.7 | >3.3 | <0.7 | - | 1.3 | >6.5 | |||||||
| b38 | H | Cl | Cl | H | 18.58 ± 1.24 | 17.55 ± 2.01 | 29.58 ± 2.13 | 27.63 ± 0.58 | 26.16 ± 0.08 | 28.34 ± 3.74 | >100 | |||||
| 1.5 | >5.4 | 1.6 | >5.7 | 1.0 | >3.4 | 1.0 | >3.6 | 1.1 | >3.8 | |||||||
| b39 | H | NO2 | H | H | >30 | >30 | >30 | >30 | >30 | >100 | >100 | |||||
| - | - | - | - | - | - | - | - | - | - | |||||||
| b40 | H | H | NO2 | H | >30 | >30 | >30 | >30 | >30 | >100 | >100 | |||||
| - | - | - | - | - | - | - | - | - | - | |||||||
| b41 | H | CF3 | H | H | 24.37 ± 5.87 | 29.07 ± 3.06 | >30 | >30 | >30 | >100 | >100 | |||||
| >4.1 | >4.1 | >3.4 | >3.4 | - | - | - | - | - | - | |||||||
| b42 | H | CF3 | H | CF3 | >30 | >30 | >30 | >30 | >30 | >100 | >100 | |||||
| - | - | - | - | - | - | - | - | - | - | |||||||
| Etoposide | 5.11 ± 0.45 | 3.96 ± 0.17 | 20.57 ± 0.34 | 3.23 ± 0.28 | 29.32 ± 1.73 | 6.42 ± 0.39 | >100 | |||||||||
| 1.3 | >19.6 | 1.6 | >25.3 | 0.3 | >4.9 | 2.0 | >31 | 0.2 | >3.4 | |||||||
3. Experimental
3.1. Chemistry
3.1.1. General Methods
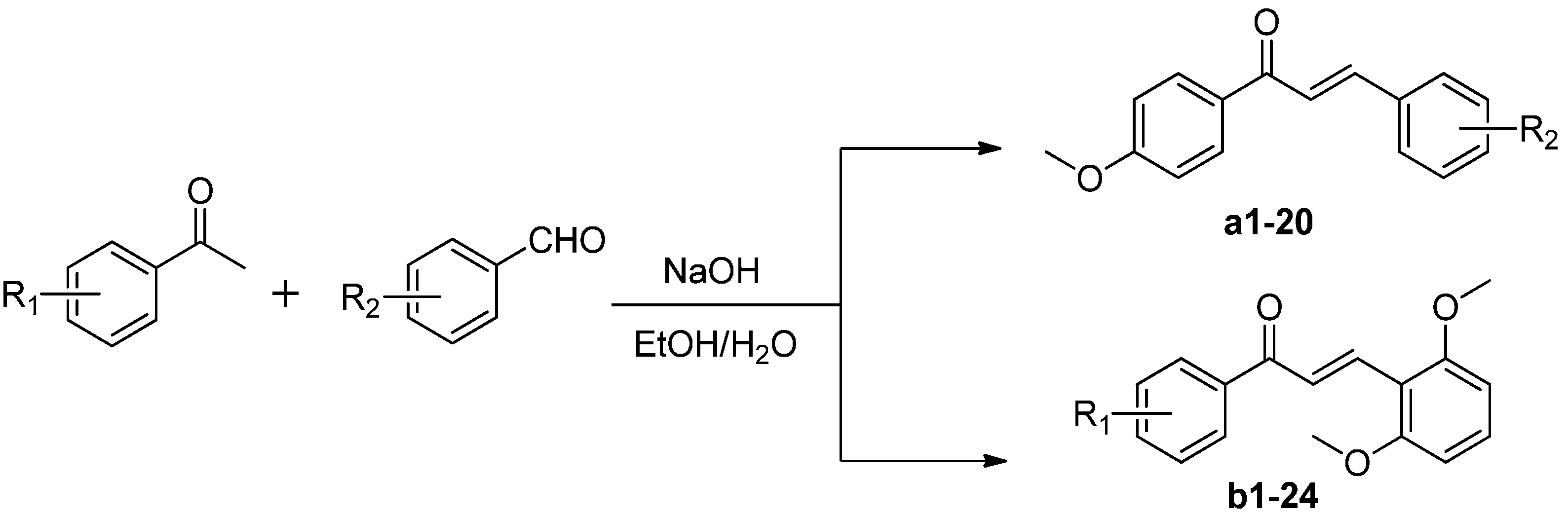
3.1.2. General Procedure for Synthesis of Chalcones
3.1.3. Characterization Data
3.2. Biology
3.2.1. Cell Culture
3.2.2. In Vitro Anti-Proliferative Assay
4. Conclusions
Supplementary Materials
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Srinivasan, B.; Johnson, T.E.; Lad, R.; Xing, C. Structure-Activity Relationship Studies of Chalcone Leading to 3-Hydroxy-4,3',4',5'-tetramethoxychalcone and Its Analogues as Potent Nuclear Factor κB Inhibitors and Their Anticancer Activities. J. Med. Chem. 2009, 52, 7228–7235. [Google Scholar]
- Lee, S.H.; Seo, G.S.; Kim, J.Y.; Jin, X.Y.; Kim, H.D.; Sohn, D.H. Heme oxygenase 1 mediates anti-inflammatory effects of 2',4',6'-tris(methoxymethoxy) chalcone. Eur. J. Pharmacol. 2006, 532, 178–186. [Google Scholar]
- Yang, H.M.; Shin, H.R.; Cho, S.H.; Bang, S.C.; Song, G.Y.; Ju, J.H.; Kim, M.K.; Lee, S.H.; Ryu, J.C.; Kim, Y.; et al. Structural requirement of chalcones for the inhibitory activity of interleukin-5. Bioorg. Med. Chem. 2007, 15, 104–111. [Google Scholar]
- Nowakowska, Z. A review of anti-infective and anti-inflammatory chalcones. Eur. J. Med. Chem. 2007, 42, 125–137. [Google Scholar]
- Fukai, T.; Marumo, A.; Kaitou, K.; Kanda, T.; Terada, S.; Nomura, T. Antimicrobial activity of licorice flavonoids against methicillin-resistant Staphylococcus aureus. Fitoterapia 2002, 73, 536–539. [Google Scholar]
- Chiaradia, L.D.; Martins, P.G.; Cordeiro, M.N.; Guido, R.V.; Ecco, G.; Andricopulo, A.D.; Yunes, R.A.; Vernal, J.; Nunes, R.J.; Terenzi, H. Synthesis, biological evaluation, and molecular modeling of chalcone derivatives as potent inhibitors of Mycobacterium tuberculosis protein tyrosine phosphatases (PtpA and PtpB). J. Med. Chem. 2012, 55, 390–402. [Google Scholar]
- Svetaz, L.; Tapia, A.; López, S.N.; Furlán, R.L.E.; Petenatti, E.; Pioli, R.; Schmeda-Hirschmann, G.; Zacchino, S.A. Antifungal chalcones and new caffeic acid esters from Zuccagnia punctata acting against soybean infecting fungi. J. Agric. Food Chem. 2004, 52, 3297–3300. [Google Scholar]
- Lahtchev, K.L.; Batovska, D.I.; Parushev, S.P.; Ubiyvovk, V.M.; Sibirny, A.A. Antifungal activity of chalcones: A mechanistic study using various yeast strains. Eur. J. Med. Chem. 2008, 43, 2220–2228. [Google Scholar]
- Cheng, J.H.; Hung, C.F.; Yang, S.C.; Wang, J.P.; Won, S.J.; Lin, C.N. Synthesis and cytotoxic, anti-inflammatory, and anti-oxidant activities of 2',5'-dialkoxylchalcones as cancer chemopreventive agents. Bioorg. Med. Chem. 2008, 16, 7270–7276. [Google Scholar]
- Modzelewska, A.; Pettit, C.; Achanta, G.; Davidson, N.E.; Huang, P.; Khan, S.R. Anticancer activities of novel chalcone and bis-chalcone derivatives. Bioorg. Med. Chem. 2006, 14, 3491–3495. [Google Scholar]
- Kachadourian, R.; Day, B.J.; Pugazhenti, S.; Franklin, C.C.; GenouxBastide, E.; Mahaffey, G.; Gauthier, C.; di Pietro, A.; Boumendjel, A. A synthetic chalcone as a potent inducer of glutathione biosynthesis. J. Med. Chem. 2012, 55, 1382–1388. [Google Scholar]
- Achanta, G.; Modzelewska, A.; Feng, L.; Khan, S.R.; Huang, P. A boronic-chalcone derivative exhibits potent anticancer activity through inhibition of the proteasome. Mol. Pharmacol. 2006, 70, 426–433. [Google Scholar]
- Valdameri, G.; Gauthier, C.; Terreux, R.; Kachadourian, R.; Day, B.J.; Winnischofer, S.M.; Rocha, M.E.; Frachet, V.; Ronot, X.; di Pietro, A.; et al. Investigation of Chalcones as Selective Inhibitors of the Breast Cancer Resistance Protein: Critical Role of Methoxylation in both Inhibition Potency and Cytotoxicity. J. Med. Chem. 2012, 55, 3193–3200. [Google Scholar]
- Rangel, L.P.; Winter, E.; Gauthier, C.; Terreux, R.; Chiaradia-Delatorre, L.D.; Mascarello, A.; Nunes, R.J.; Yunes, R.A.; Creczynski-Pasa, T.B.; Macalou, S.; et al. New structure—Activity relationships of chalcone inhibitors of breast cancer resistance protein: Polyspecificity toward inhibition and critical substitutions against cytotoxicity. Drug Des. Dev. Ther. 2013, 7, 1043–1052. [Google Scholar]
- Winter, E.; Gozzi, G.J.; Chiaradia-Delatorre, L.D.; Daflon-Yunes, N.; Terreux, R.; Gauthier, C.; Mascarello, A.; Leal, P.C.; Cadena, S.M.; Yunes, R.A.; et al. Quinoxaline-substitutedchalconesasnew inhibitors of breast cancer resistance protein ABCG2: Polyspecificity at B-ring position. Drug Des. Dev. Ther. 2014, 8, 609–619. [Google Scholar]
- Kamal, A.; Shankaraiah, N.; Prabhakar, S.; Reddy, C.R.; Markandeya, N.; Reddy, K.L.; Devaiah, V. Solid-phase synthesis of new pyrrolobenzodiazepine-chalcone conjugates: DNA-binding affinity and anticancer activity. Bioorg. Med. Chem. Lett. 2008, 18, 2434–2439. [Google Scholar]
- Jin, C.; Liang, Y.J.; He, H.; Fu, L. Synthesis and antitumor activity of novel chalcone derivatives. Biomed. Pharmacother. 2013, 67, 215–217. [Google Scholar]
- Shenvi, S.; Kumar, K.; Hatti, K.S.; Rijesh, K.; Diwakar, L.; Reddy, G.C. Synthesis, anticancer and antioxidant activities of 2,4,5-trimethoxy chalcones and analogues from asaronaldehyde: Structure-activity relationship. Eur. J. Med. Chem. 2013, 62, 435–442. [Google Scholar]
- Boumendjel, A.; Boccard, J.; Carrupt, P.-A.; Nicolle, E.; Blanc, M.; Geze, A.; Choisnard, L.; Wouessidjewe, D.; Matera, E.-L.; Dumontet, C. Antimitotic and antiproliferative activities of chalcones: Forward structure-activity relationship. J. Med. Chem. 2008, 51, 2307–2310. [Google Scholar]
- Kumar, V.; Kumar, S.; Hassan, M.; Wu, H.; Thimmulappa, R.K.; Kumar, A.; Sharma, S.K.; Parmar, V.S.; Biswal, S.; Malhotra, S.V. Novel chalcone derivatives as potent Nrf2 activators in mice and human lung epithelial cells. J. Med. Chem. 2011, 54, 4147–4159. [Google Scholar]
- Hsieh, C.T.; Hsieh, T.J.; ElShazly, M.; Chuang, D.W.; Tsai, Y.H.; Yen, C.T.; Wu, S.F.; Wu, Y.C.; Chang, F.-R. Synthesis of chalcone derivatives as potential anti-diabetic agents. Bioorg. Med. Chem. Lett. 2012, 22, 3912–3915. [Google Scholar]
- Lin, A.S.; NakagawaGoto, K.; Chang, F.R.; Yu, D.; MorrisNatschke, S.L.; Wu, C.C.; Chen, S.L.; Wu, Y.C.; Lee, K.H. First total synthesis of protoapigenone and its analogues as potent cytotoxic agents. J. Med. Chem. 2007, 50, 3921–3927. [Google Scholar]
- Montes-Avila, J.; Diaz-Camacho, S.P.; Sicairos-Felix, J.; Delgado-Vargas, F.; Rivero, I.A. Solution-phase parallel synthesis of substituted chalcones and their antiparasitary activity against Giardia lamblia. Bioorg. Med. Chem. 2009, 17, 6780–6785. [Google Scholar]
- Ashtekar, K.D.; Staples, R.J.; Borhan, B. Development of a Formal Catalytic Asymmetric [4 + 2] Addition of Ethyl-2,3-butadienoate with Acyclic Enones. Org. Lett. 2011, 13, 5732–5735. [Google Scholar]
- Braun, R.U.; Ansorge, M.; Müller, T.J.J. Coupling–Isomerization Synthesis of Chalcones. Chemistry 2006, 12, 9081–9094. [Google Scholar]
- Dimmock, J.R.; Jha, A.; Zello, G.A.; Quail, J.W.; Oloo, E.O.; Nienaber, K.H.; Kowalczyk, E.S.; Allen, T.M.; Santos, C.L.; de Clercq, E.; et al. Cytotoxic N-[4-(3-aryl-3-oxo-1 propenyl)phenylcarbonyl]-3,5-bis(phenylmethylene)-4-piperidones and related compounds. Eur. J. Med. Chem. 2002, 37, 961–972. [Google Scholar]
- Brown, B.R.; Cummings, W. Polymerisation of flavans. Part II. The condensation of 4[prime or minute]-methoxyflavan with phenols. J. Chem. Soc. (Resum.) 1958. [Google Scholar] [CrossRef]
- Kumar, A.; Sharma, S.; Tripathi, V.D.; Srivastava, S. Synthesis of chalcones and flavanones using Julia–Kocienski olefination. Tetrahedron 2010, 66, 9445–9449. [Google Scholar]
- Barnes, R.P.; Goodwin, T.C.; Cotten, T.W. A Study of the Direction of Enolization of p-Methoxy-p'-bromodibenzoylmethane and p-Methoxy-p'-ethoxydibenzoylmethane. J. Am. Chem. Soc. 1947, 69, 3135–3138. [Google Scholar]
- Wu, J.; Li, J.; Cai, Y.; Pan, Y.; Ye, F.; Zhang, Y.; Zhao, Y.; Yang, S.; Li, X.; Liang, G. Evaluation and Discovery of Novel Synthetic Chalcone Derivatives as Anti-Inflammatory Agents. J. Med. Chem. 2011, 54, 8110–8123. [Google Scholar]
- D’Orazio, D.; de Saizieu, A.; Raederstorff, D.; Schueler, G.; Wang-Schmidt, Y.; Wehrli, C.; Wertz, K.; Wolfram, S. Compounds for the Treatment of Non-Autoimmune Type 2 Diabetes Mellitus and/or Syndrome X. WO Patent 2,006,136,429, 28 December 2006. [Google Scholar]
- Shettigar, V.; Patil, P.S.; Naveen, S.; Dharmaprakash, S.M.; Sridhar, M.A.; Shashidhara Prasad, J. Crystal growth and characterization of new nonlinear optical chalcone derivative: 1-(4-Methoxyphenyl)-3-(3, 4-dimethoxyphenyl)-2-propen-1-one. J. Cryst. Growth 2006, 295, 44–49. [Google Scholar]
- Hwang, D.; Hyun, J.; Jo, G.; Koh, D.; Lim, Y. Synthesis and complete assignment of NMR data of 20 chalcones. Magn. Reson. Chem. 2011, 49, 41–45. [Google Scholar]
- Russell, A. The constitution of tannins. Part I. Reduction products of chalkones and the synthesis of a typical phlobatannin. J. Chem. Soc. (Resum.) 1934. [Google Scholar] [CrossRef]
- Sharma, N.; Sharma, A.; Kumar, R.; Shard, A.; Sinha, A.K. One-Pot Two-Step Oxidative Cleavage of 1,2-Arylalkenes to Aryl Ketones Instead of Arylaldehydes in an Aqueous Medium: A Complementary Approach to Ozonolysis. Eur. J. Org. Chem. 2010, 31, 6025–6032. [Google Scholar]
- Ducki, S.; Rennison, D.; Woo, M.; Kendall, A.; Chabert, J.F.D.; McGown, A.T.; Lawrence, N.J. Combretastatin-like chalcones as inhibitors of microtubule polymerization. Part 1: Synthesis and biological evaluation of antivascular activity. Bioorg. Med. Chem. 2009, 17, 7698–7710. [Google Scholar]
- Simonis, H.; Danischewski, S. Über die Anwendung der Friedel-Craftsschen Reaktion zum Aufbau der Flavone. Eur. J. Inorg. Chem. 1926, 59, 2914–2919. [Google Scholar]
- Johnson, I.T.; Fenwick, G.R. (Eds.) Royal Society of Chemistry; Johnson, I.T.; Fenwick, G.R. (Eds.) Royal Society of Chemistry: England, UK, 2000; Volume 255, pp. 189–192.
- Yong, K.; Lu, J.; Gu, H.; Chen, X. Process for Preparation of 4'-Hydroxy-2,6-dimethoxydihydrochalcone. CN Patent 1,01,250,098, 27 August 2008. [Google Scholar]
- Cohen, F.E.; McKerrow, J.H.; Kenyon, G.L.; Li, Z.; Chen, X.; Gong, B.; Li, R. Inhibitors of Metazoan Parasite Proteases. U.S. Patent 5,739,170 A, 14 April 1998. [Google Scholar]
- George, F.; Fellague, T. Preparation of Amino Substituted Chalcone Derivatives for Use in Cosmetic Compositions as Sunscreens and Suntanning Agents. F.R. Patent 2,839,717, Al, 21 November 2003. [Google Scholar]
- Song, L.L.; Kosmeder, J.W., II; Lee, S.K.; Gerhauser, C.; Lantvit, D.; Moon, R.C.; Moriarty, R.M.; Pezzuto, J.M. Cancer chemopreventive activity mediated by 4'-bromoflavone, a potent inducer of phase II detoxification enzymes. Cancer Res. 1999, 59, 578–585. [Google Scholar]
- Nielsen, S.F.; Christensen, S.B.; Cruciani, G.; Kharazmi, A.; Liljefors, T. Antileishmanial Chalcones: Statistical Design, Synthesis, and Three-Dimensional Quantitative Structure-Activity Relationship Analysis. J. Med. Chem. 1998, 41, 4819–4832. [Google Scholar]
- Sahu, N.K.; Bari, S.B.; Kohli, D.V. Molecular modeling studies of some substituted chalcone derivatives as cysteine protease inhibitors. Med. Chem. Res. 2012, 21, 3835–3847. [Google Scholar]
- Sun, H.X.; He, H.W.; Zhang, S.H.; Liu, T.G.; Ren, K.H.; He, Q.Y.; Shao, R.G. Suppression of N-Ras by shRNA-expressing plasmid increases sensitivity of HepG2 cells to vincristine-induced growth inhibition. Cancer Gene Ther. 2009, 16, 693–702. [Google Scholar]
- Sample Availability: Samples of the compounds a1–20 and b1–42 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bai, X.-G.; Xu, C.-L.; Zhao, S.-S.; He, H.-W.; Wang, Y.-C.; Wang, J.-X. Synthesis and Cytotoxic Evaluation of Alkoxylated Chalcones. Molecules 2014, 19, 17256-17278. https://doi.org/10.3390/molecules191117256
Bai X-G, Xu C-L, Zhao S-S, He H-W, Wang Y-C, Wang J-X. Synthesis and Cytotoxic Evaluation of Alkoxylated Chalcones. Molecules. 2014; 19(11):17256-17278. https://doi.org/10.3390/molecules191117256
Chicago/Turabian StyleBai, Xiao-Guang, Chang-Liang Xu, Shuang-Shuang Zhao, Hong-Wei He, Yu-Cheng Wang, and Ju-Xian Wang. 2014. "Synthesis and Cytotoxic Evaluation of Alkoxylated Chalcones" Molecules 19, no. 11: 17256-17278. https://doi.org/10.3390/molecules191117256
APA StyleBai, X.-G., Xu, C.-L., Zhao, S.-S., He, H.-W., Wang, Y.-C., & Wang, J.-X. (2014). Synthesis and Cytotoxic Evaluation of Alkoxylated Chalcones. Molecules, 19(11), 17256-17278. https://doi.org/10.3390/molecules191117256





