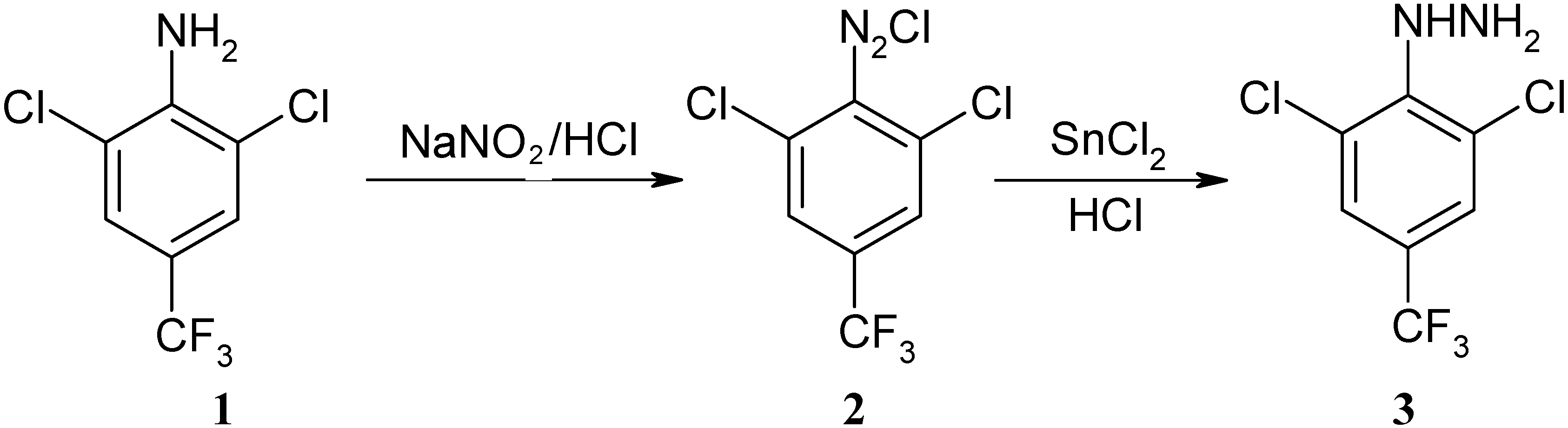
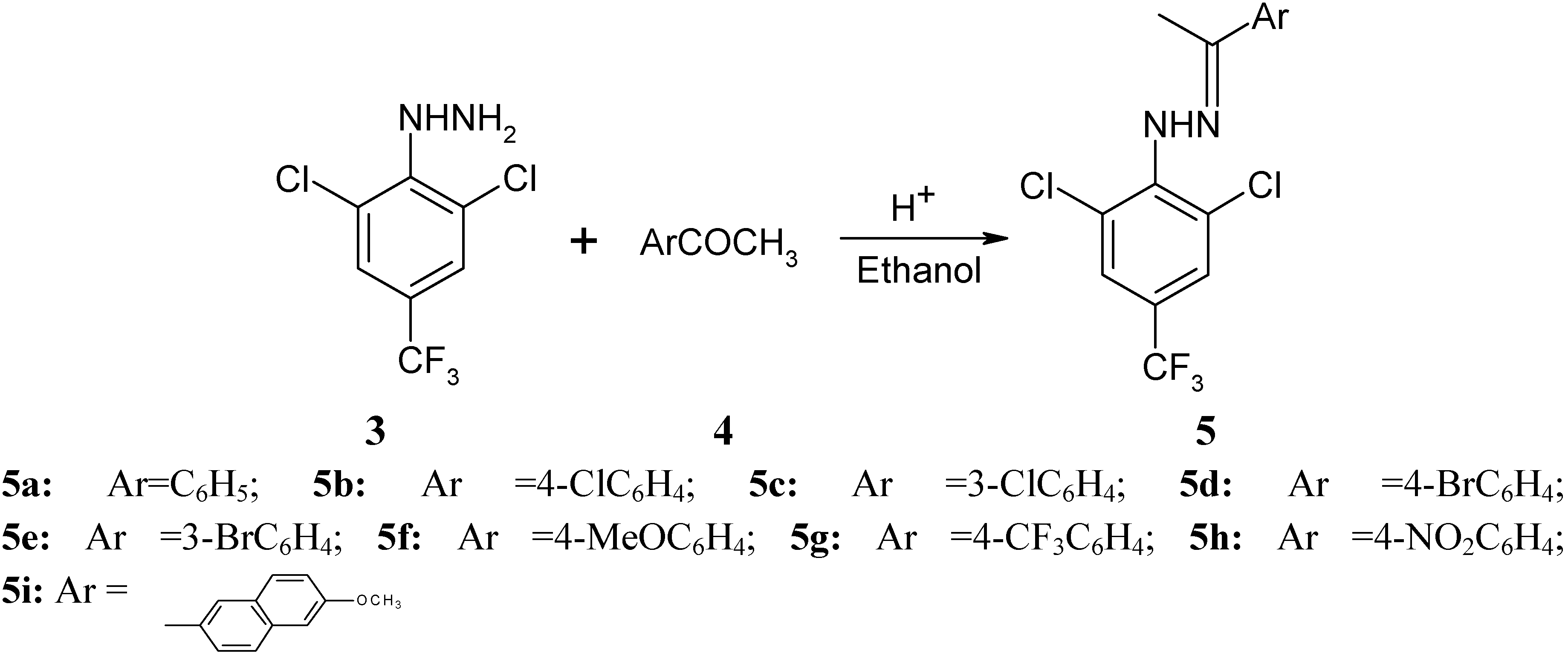
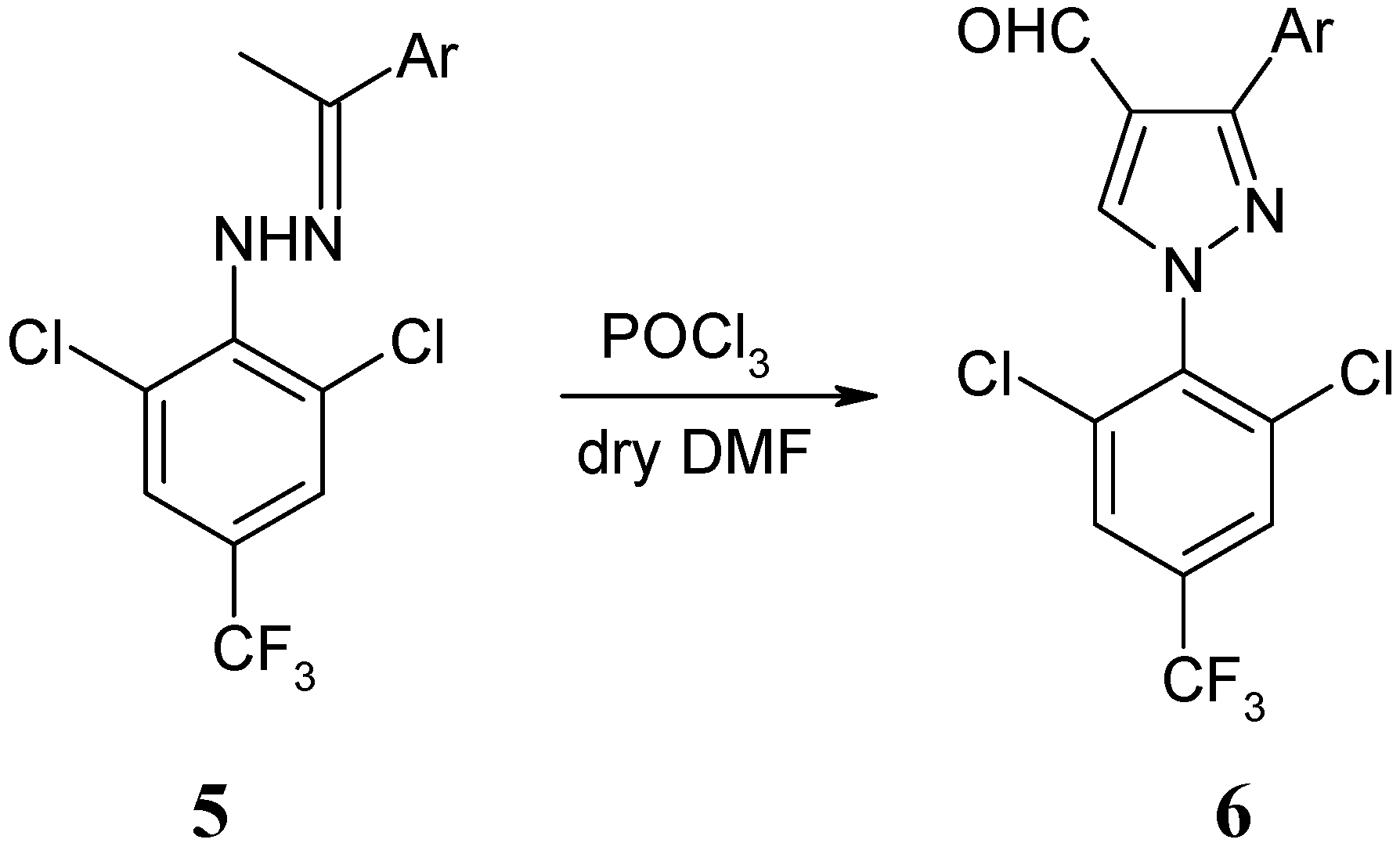
Synthesis of Novel 1-[(2,6-Dichloro-4-trifluoromethyl)phenyl]-3-aryl-1H-pyrazole-4-carbaldehydes
Abstract
:1. Introduction
2. Results and Discussion
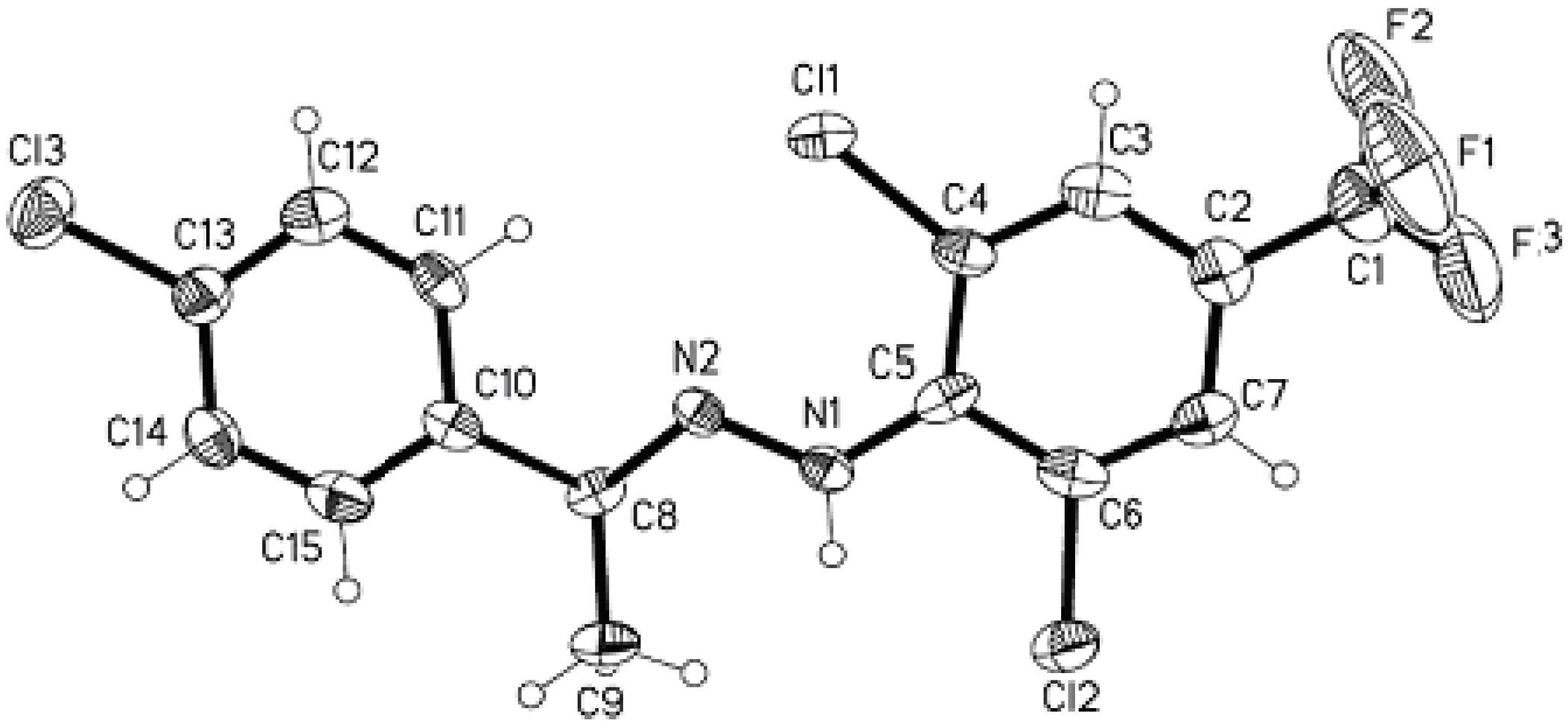
X-ray diffraction
| Compound | C15 H10 Cl3 F3 N2 |
|---|---|
| Color | Colorless |
| Formula weight | 381.60 |
| Crystal system | Monoclinic |
| Temperature,° | 25(298K) |
| Cell constants | |
| a (Å) | 16.8770(7) |
| b (Å) | 8.0054(8) |
| c (Å) | 24.119(2) |
| α (˚) | 90 |
| β (˚) | 99.654(2) |
| γ (˚) | 90 |
| Volume (Å3) | 3212.5(4) |
| Formula units | 8 |
| Calculated density (g/cm-3) | 1.578 |
| F(000) | 1536 |
| Absorption coefficient, mμ-3 | 0.599 |
| Limiting indices | -20<=h<=16, -7<=k<=9, -27<=l<=28 |
| Reflections collected / unique | 8110 / 2841 [R(int) = 0.0402] |
| Absorption correction | Semi-empirical from equivalents |
| Max. and min. transmission | 0.9370 and 0.8314 |
| Refinement method | Full-matrix least-squares on F2 |
| Data / restraints / parameters | 2841 / 0 / 208 |
| Goodness-of-fit on F2 | 1.015 |
| Final R indices | R1 = 0.1641, wR2 = 0.5152 |
| Largest diff. peak and hole (e Å-3) | 0.869 and -0.919 |
| Entry | Ar | Products(6) | Yield(%) a |
| 1 | C6H5 | 6a | 89 |
| 2 | 4-ClC6H4 | 6b | 88 |
| 3 | 3-ClC6H4 | 6c | 83 |
| 4 | 4-BrC6H4 | 6d | 86 |
| 5 | 3-BrC6H4 | 6e | 82 |
| 6 | 4-MeOC6H4 | 6f | 83 |
| 7 | 4-CF3C6H4 | 6g | 86 |
| 8 | 4-NO2C6H4 | 6h | 85 |
| 9 | 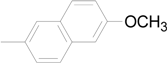 | 6i | 81 |
3. Experimental
3.1. General
3.2. Synthesis of (2,6-Dichloro-4-trifluoromethyl)phenylhydrazine (3)
3.3. General procedure for synthesis of N-(2,6-Dichloro-4-trifluoromethyl)phenyl-N’-(1-phenyl-ethylidene) hydrazines 5a-i
3.4. General procedure for synthesis of 1-[(2,6-dichloro-4-trifluoromethyl)phenyl]-3-aryl-1H-pyrazole-4- carbaldehydes 6a-i
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds 6a-i are available from the authors.
References and Notes
- Jones, G.; Stanforth, S.P. The Vilsmeier reaction of fully conjugated carbocycles and heterocycles. Org. React. 2001, 49, 1–315. [Google Scholar]
- Majo, V.J.; Perumal, P.T. Intramolecular cyclization of azides by iminium species. a novel method for the construction of nitrogen heterocycles under Vilsmeier conditions. J. Org. Chem. 1998, 63, 7136–7142. [Google Scholar] [CrossRef]
- Kira, M.A.; Abdel-Rahman, M.O.; Gadalla, K.Z. The Vilsmeier reaction-III, cyclization of hydrazones to pyrazoles. Tetrahedron Lett. 1969, 10, 109–110. [Google Scholar] [CrossRef]
- Selvi, S.; Perumal, P.T. Facile synthesis of [1]benzopyrano[4,3-c]pyrazoles, 1-aryl-3-(2-formamidophenyl)pyrazoles and 1-aryl-3-phenyl-4-alkylpyrazoles using Vilsmeier reagent. Indian J. Chem. 2002, 41B, 1887–1893. [Google Scholar]
- Elguero, J. Comprehensive Heterocyclic Chemistry; Katrizky, A., Ed.; Pregamon Press: Oxford, UK, 1984; p. 277. [Google Scholar]
- El-Emary, T.I.; Bakhite, E.A. Synthesis and biological screening of new 1,3-diphenylpyrazoles with different heterocyclic moieties at position-4. Pharmazie 1999, 54, 106–111. [Google Scholar]
- Bratenko, M.K.; Vovk, M.V.; Sydorchuk, I.J. Synthesis and antibacterial and antifungal activity of hydrazones and azines of 1-phenyl-3-R-4-formylpyrazole. Farm. Zh. 1999, 68–71. [Google Scholar]
- Rathelot, P.; Azas, N.; EL-Kashef, H.; Delmas, F.; Giorgio, C.D.; David, P.T.; Maldonado, V.P. 1,3-Diphenylpyrazoles: Synthesis andantiparasitic activities of azomethine derivatives. Eur. J. Med. Chem. 2002, 37, 671–679. [Google Scholar]
- Cottineau, B.; Toto, P.; Marot, C.; Pipaud, A.; Chenault, J. Synthesis and hypoglycemic evaluation of substituted pyrazole-4-carboxylic acids. Biorg. Med. Chem. Lett. 2002, 12, 2105–2108. [Google Scholar] [CrossRef]
- Welch, J.T. Advances in the preparation of biologically active organofluorine compounds. Tetrahedron 1987, 43, 3123–3197. [Google Scholar] [CrossRef]
- Bertrand, F.; Pevere, V.; Quiclet-Sire, B.; Zard, S.Z. A xanthate transfer radical process for the introduction of the trifluoromethyl group. Org. Lett. 2001, 3, 1069–1071. [Google Scholar] [CrossRef]
- Song, J.J.; Tan, Z.; Reeves, J.T.; Gallou, F.; Yee, N.K.; Senanayake, C.H. N-heterocyclic carbene catalyzed trifluoromethylation of carbonyl compounds. Org. Lett. 2005, 7, 2193–2195. [Google Scholar]
- Caboni, P.; Sammelson, R.E.; Casida, J.E. Phenylpyrazole insecticide photochemistry, metabolism, and GABAergic action: Ethiprole compared with fipronil. J. Agric. Food Chem. 2003, 51, 7055–7061. [Google Scholar]
- Sammelson, R.E.; Casida, J.E. Synthesis of a tritium-tabeled, fipronil-based, highly potent, photoaffinity probe for the GABA receptor. J. Org. Chem. 2003, 68, 8075–8079. [Google Scholar] [CrossRef]
- Ranatunge, R.R.; Earl, R.A.; Garvey, D.S.; Janero, D.R.; Gordon Letts, L.; Martino, A.M.; Murty, M.G.; Richardson, S.K.; Schwalb, D.J.; Young, D.V.; Zemtseva, I.S. 3-(2-Methoxytetrahydrofuran-2-yl)pyrazoles: A novel class of potent, selective cyclooxygenase-2 (COX-2) inhibitors. Bioorg. Med. Chem. Lett. 2004, 14, 6049–6052. [Google Scholar]
- Sridhar, R.; Perumal, P.T.; Etti, S.; Shanmugam, G.; Ponnuswamy, M.N.; Prabavathyc, V.R.; Mathivanan, N. Design, synthesis and anti-microbial activity of 1H-pyrazole carboxylates. Bioorg. Med. Chem. Lett. 2004, 14, 6035–6040. [Google Scholar] [CrossRef]
- Prakash, O.; Pannu, K.; Naithani, R.; Kaur, H. One-pot synthesis of oxime derivatives of 1,3-diphenylpyrazole-4-carboxaldehydes from acetophenone phenylhydrazones using Vilsmeier-Haack reagent. Synth. Commun. 2006, 36, 3479–3485. [Google Scholar] [CrossRef]
- CCDC 685556 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre, 12, Union Road, Cambridge, CB2 1EZ, UK; E-Mail: deposit@ccdc.cam.ac.uk.
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hu, H.; Ge, C.; Ding, L.; Zhang, A. Synthesis of Novel 1-[(2,6-Dichloro-4-trifluoromethyl)phenyl]-3-aryl-1H-pyrazole-4-carbaldehydes. Molecules 2010, 15, 7472-7481. https://doi.org/10.3390/molecules15107472
Hu H, Ge C, Ding L, Zhang A. Synthesis of Novel 1-[(2,6-Dichloro-4-trifluoromethyl)phenyl]-3-aryl-1H-pyrazole-4-carbaldehydes. Molecules. 2010; 15(10):7472-7481. https://doi.org/10.3390/molecules15107472
Chicago/Turabian StyleHu, Huanan, Changhua Ge, Lisheng Ding, and Anjiang Zhang. 2010. "Synthesis of Novel 1-[(2,6-Dichloro-4-trifluoromethyl)phenyl]-3-aryl-1H-pyrazole-4-carbaldehydes" Molecules 15, no. 10: 7472-7481. https://doi.org/10.3390/molecules15107472
APA StyleHu, H., Ge, C., Ding, L., & Zhang, A. (2010). Synthesis of Novel 1-[(2,6-Dichloro-4-trifluoromethyl)phenyl]-3-aryl-1H-pyrazole-4-carbaldehydes. Molecules, 15(10), 7472-7481. https://doi.org/10.3390/molecules15107472




