Abstract
Protopanaxatriol saponins obtained with AB-8 macroporous resin mainly consisted of ginsenosides Rg1 and Re. A novel mono-ester of ginsenoside-Rh1 (ginsenoside-ORh1) was synthesized through further enzymatic hydrolysis and octanoyl chloride modifications. A 53% yield was obtained by a facile synthetic method. The structures were identified on the basis of 1D-NMR and 2D-NMR, as well as ESI-TOF-MS mass spectroscopic analyses. The isolated and synthetic compounds were applied in an anti-tumor bioassay, in which ginsenoside ORh1 showed moderate effects on Murine H22 Hepatoma Cells.
Introduction
Panax ginseng, which belongs to the Araliaceae family, has been used as a traditional medicine for thousands of years and is now a popular natural medicine used worldwide [1,2]. Ginsenosides [3,4] have been regarded as the principal components responsible for the pharmacological activities of ginseng including anti-inflammatory activity, increasing the free radical scavenging activities and reducing weight [5,6,7,8,9]. Recent studies pointed that some rare ginsenosides such as Rh1, M1 had strong anticancer activity both in vivo and in vitro [10,11]. Pharmaceutical studies [12] have shown that ginseng saponins were ingested mainly by the bacteria of the small intestine and thus converted into their final forms: Re→Rg1→F1 or Rh1→M4; Rb1→Rd→F2→M1; Rb2→M6→M2→M1; Rc→M7→M3→M1. Previous studies also showed that ginsenoside-M1 was further esterified with fatty acids and thus could be sustained longer in the body, a result that suggested that the fatty acid ester of the M1 might be the real anti-tumor active species in vivo [13]. Our laboratory previously reported the synthesis of three novel mono-esters of ginsenoside-M1 (DM1, SM1, PM1) and their bioactivity [14]. According to the previous study, we established a method for synthesizing ORh1 and evaluated its bioactivity. Ginsenoside Rh1 was obtained through enzymatic hydrolysis and the fatty ester was modified by octanoyl chloride. The structure of ginsenoside-ORh1 was identified through 1H- and 13C-NMR and ESI-TOF-MS spectroscopic analyses. The pure ginsenoside-ORh1 was used to determine the anticancer bioactivity against Murine H22 Hepatoma Cells; the results showed that ginsenoside-ORh1 had moderate effects on H22 cells. This is the first time ginsenoside-ORh1 had been synthesized and reported. In this paper, we present the synthesis method and the bioactivity evaluation of the new ginsenoside fatty acid ester ORh1.
Results and Discussion
Characterizations of compounds 1-4
Compound 1 and compound 2 have been characterized as ginsenosides Rg1 and Re, whose structures were identified by MS and NMR data analysis (not shown). After an enzymatic hydrolysis process, we obtained ginsenoside Rh1 (Compound 3). Then, ginsenoside Rh1 was modified by octanoyl chloride and formed ginsenoside fatty acid ester (compound 4). The structures of compounds 1-4 are shown in Figure 1. The HPLC analysis of compounds 3 and 4 is shown in Figure 2. From the HPLC analysis, the polarity of compound 4 was lower than that of Rh1 (compound 3). From the 13C-NMR data analysis and comparison, we found no significant chemical shifts changes for the main skeleton but an upfield shift of C’-6 of the 6-O-Glu (δ 63.5 to δ 64.8, see Table 1), indicating that the fatty acid ester subsituent was connected to that position of 6-O-Glu was observed. This assumption had been verified by HMBC (see Figure 3) which showed a cross-peak between H-6’ to the carboxyl carbon.

Table 1.
13C-NMR Chemical Shifts(δ) of protopanaxatriol and compounds 3 and 4. a
| No.(C) | Rh1 | Compound 3 | Compound 4 |
|---|---|---|---|
| 1 | 39.4 | 39.2 | 39.8 |
| 2 | 27.9 | 27.2 | 27.2 |
| 3 | 78.6 | 78.7 | 78.7 |
| 4 | 40.3 | 40.2 | 39.4 |
| 5 | 61.4 | 61.4 | 60.0 |
| 6 | 78.0 | 77.7 | 80.2 |
| 7 | 45.2 | 45.4 | 44.6 |
| 8 | 41.1 | 41.3 | 41.1 |
| 9 | 50.2 | 50.4 | 50.0 |
| 10 | 39.6 | 39.9 | 39.0 |
| 11 | 32.0 | 32.2 | 31.6 |
| 12 | 71.0 | 71.2 | 70.0 |
| 13 | 48.2 | 48.4 | 49.7 |
| 14 | 51.6 | 51.8 | 50.8 |
| 15 | 31.1 | 31.4 | 29.6 |
| 16 | 27.2 | 27.2 | 26.7 |
| 17 | 54.7 | 54.9 | 50.3 |
| 18 | 17.4 | 17.2 | 17.2 |
| 19 | 17.6 | 17.8 | 17.8 |
| 20 | 78.0 | 78.3 | 73.9 |
| 21 | 26.8 | 27.0 | 27.0 |
| 22 | 25.8 | 26.0 | 34.1 |
| 23 | 28.0 | 28.1 | 22.6 |
| 24 | 126.0 | 126.5 | 125.7 |
| 25 | 130.6 | 130.9 | 130.9 |
| 26 | 25.8 | 26.0 | 25.6 |
| 27 | 17.6 | 17.5 | 17.7 |
| 28 | 31.7 | 31.8 | 31.6 |
| 29 | 16.4 | 16.5 | 15.8 |
| 30 | 16.8 | 17.0 | 16.6 |
a Compounds 3 and 4 were measured in CDCl3 and chemical shifts are expressed in ppm.
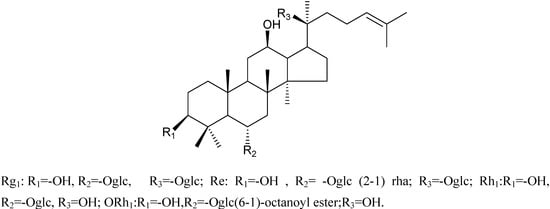
Figure 1.
Isolated, enzyme produced and synthetic ginsenosides.
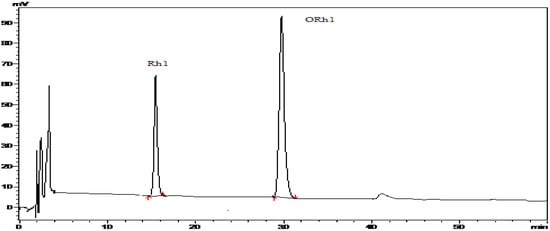
Figure 2.
HPLC spectra of Rh1 (compound 3) and the reaction product of ORh1 (compound 4) HPLC conditions: isocratic elution with 100% MeOH for 60 min.
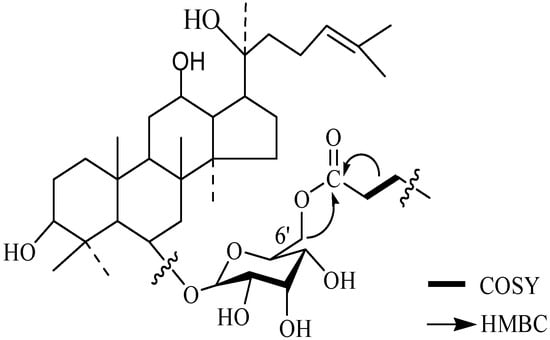
Figure 3.
Partial HMBC and COSY correlation of the synthetic compound.

Table 2.
13C-NMR data of sugar and fatty acid ester moieties of compound 3 and 4.
| No.(C) | Rh1 | Compound 3 | Compound 4 |
|---|---|---|---|
| 1′ | 105.7 | 106.2 | 104.4 |
| 2′ | 75.4 | 75.6 | 74.0 |
| 3 ′ | 80.0 | 80.2 | 80.1 |
| 4′ | 71.8 | 72.0 | 72.0 |
| 5′ | 79.5 | 79.8 | 79.2 |
| 6′ | 63.5 | 63.3 | 64.8 |
| 1′′ | 174.3 | ||
| 2′′ | 34.1 | ||
| 3′′ | 24.9 | ||
| 4′′ | 30.3 | ||
| 5′′ | 29.1 | ||
| 6′′ | 30.9 | ||
| 7′′ | 22.6 | ||
| 8′′ | 14.1 |
a Compounds 3 and 4 were measured in CDCl3 and chemical shifts are expressed in ppm.
Bioactivity evaluation
The purified and synthetic compounds were tested in an anti-tumor bioassay. Compounds 3 and 4 showed moderate cytotoxicity effects against the Murine H22 Hepatoma Cells. The results are shown in Table 3 and Figure 4. We tested the sample for four concentrations (10 μM, 20 μM, 40 μM, 80 μM). The highest inhibitory rate of ORh1 was 87.16% at the concentration of 80 μM. The IC50 of ORh1 was obtained at the concentration of 42.44 μM.

Table 3.
Anti-tumor activities of compound 3 and 4 (IC50 values in μM).
| Concentration (μM) | Inhibitory rate (%) | IC50(μM) | ||
|---|---|---|---|---|
| Ginsenoside Rh1 | ORh1 | Ginsenoside Rh1 | ORh1 | |
| 0 μM | 0 | 0 | 48.63 | 42.44 |
| 10 μM | 8.97 | 11.44 | ||
| 20 μM | 9.28 | 11.09 | ||
| 40 μM | 54.21 | 62.34 | ||
| 80 μM | 76.54 | 87.16 | ||

Figure 4.
The rate of inhibition of ORh1 on H22 cell lines after 24 h.
Conclusions
The anti-tumor effects of some ginsenosides were known. For instance, Rg3 has been shown to possess anti-tumor properties and have an effect on drug-resistant cultured cancer cells [15,16]. Rh2 can reduce the proliferation of a variety of cultured cancer cells and can influence apoptosis [17,18,19]. Most of the ginsenosides with significant anti-tumor activities belongs to protopanaxdiol-type saponins. In our study, we investigated the protopanaxtriol-type ginsenoside Rh1. The protopanaxtriol-type saponins, including Rg1 and Re, were finally transformed into ginsenoside Rh1 after intestinal metabolism. We obtained the ginsenoside Rh1 by means of enzymolysis and further chemical modification led to the synthesis of Rh1 mono-fatty acid ester. The bioactivity evaluation of fatty acid ester (ORh1) showed that this kind of compound was more active against human tumor cells. We concluded that the membrane transportation of small molecules depends on their lipophilic abilities and the fatty acid ester (ORh1) with lower polarity met this requirement. In this study, we synthesized ORh1 with fatty acid acyl-chlorides and the synthetic product had the desired lower polarity. In this study, ORh1 showed more activity and higher anti-tumor efficiency than ginseonside Rh1 on Murine H22 Hepatoma Cells, and as time increased (more than 24 hr), the anti-tumor action of Rh1 was almost eliminated, whereas ORh1 still displayed considerable anti-tumor action.
Experimental
General
The 1H- and 13C-NMR spectra were measured on a Bruker AV 400 NMR spectrometer in CDCl3, using TMS as an internal standard. Chemical shifts (δ) are expressed in parts per million (ppm). The HR-ESI-TOF mass spectra were obtained from a MDS SCIEX API QSTAR-MS instrument. Column chromatographies were carried out with silica gel 60M (200-300 mesh) and AB-8 macroporous resin and HPLC were carried out on an LC-2010 system (Shimadzu).
Chemicals and reagents
Octanoyl chloride was purchased from ABCR GmbH& Co. KG. Enzyme (snailase) was purchased from BioDee BioTech Corporation Ltd. (code: S0100, Beijing, China). Other chemicals and reagents were purchased from the Chinese Chemical Group (Beijing, China).
Extraction and isolation
Extracts of leaves from Panax ginseng (100 g) was dissolved with sufficient amount of distilled water and filtered, then the filtrate was absorbed on a AB-8 macroporous resin column (100 × 10 cm) for 8 h. Gradient elution with 25%, 30%, 80% ethanol was used to elute the column. The dry eluate was obtained from 30% ethanol fraction and weighted (45 g). The fraction consisted of the protopananxtriol-type saponins Ginsenoside Rg1 (1), a white amorphous powder, mp 194–196 oC; ESI-MS[+]: m/z=801 [M+H]+ and GinsenosideRe (2), a white amorphous powder, mp 201-203 oC; ESI-MS[+]: m/z=969.3 [M+Na]+.
Enzymatic reaction
A saponin fraction (1 g) obtained from the former procedure was weighed out and dissolved in distilled water (400 mL), enzyme (110 mg) was added and the mixture was cultured at 37 °C for 24 h while the pH was maintained at a value of 4.5. Then the enzyme reaction mixture was subjected to silica gel column chromatography eluting with CHCl3-MeOH (9:1) to afford purified Ginsenoside Rh1 (3, 320 mg) as a white amorphous powder, mp 190–192 °C; ESI-MS[+]: m/z=639 [M+H]+; 13C-NMR data, see Table 1 and Table 2.
Synthesis of Ginsenoside ORh1 (4)
Octanoyl chloride (125 mg) and K2CO3 (80 mg) were dissolved in CHCl2 (80 mL) under ice-cooling and stirred for 20min, then ginsenoside Rh1 (100 mg) dissolved in CHCl2 (1 mL) was added slowly. The mixture was reacted under stirring and with ice-cooling state for 24 h. The reacted solution was filtered through a 0.22 μm membrane, then the filtrate was condensed and subjected to silica gel column chromatography, eluted with CHCl3-MeOH=10:1 to give pure 4 (66 mg) as a colorless oil; HR-ESI-TOF-MS [+]+: m/z=764.2533 [M+H]+; 13C-NMR data, see Table 1 and Table 2. The purity was analyzed by HPLC (100%MeOH, Hypersil C18, 35 °C, 0.5 mL/min). The synthesis procedure is shown in Scheme 1.
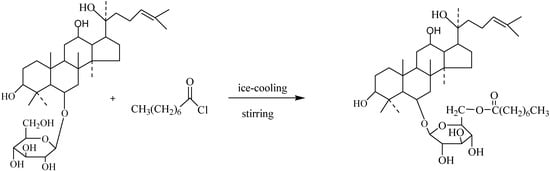
Scheme 1.
Preparation of ginsenoside ORh1(4).
In vitro anti-tumor assays
Mono-nuclear cell direct cytotoxicity assay against human tumors cell lines were carried out at the Cell Culture Laboratory, Pharmaceutical College, Jilin University, using Murine H22 Hepatoma Cells. A blank control was used in this study. Generally, 5 × 105/mL cells (190 μL) were placed in a 96-well plate and treated with obtained compound (10 μL). The normal cell was added culture medium. The plate was incubated at 37 °C for 24 h.Then, each well was added MTT (20 μL) with the concentration of 5 mg/mL and incubation at 37 °C was continued for 4 h. After that, the supernatant was removed and DMSO (150 μL) was added, the samples were agitated and the absorbance read at 490 nm.
- Sample Availability: Samples are available from the authors.
References
- Balsiger, B.M.; Murr, M.M.; Poggio, J.L.; Sarr, M.G. Bariatric surgery: Surgery for weight control in patients with morbid obesity. Med. Clin. N. Amer. 2000, 84, 477–489. [Google Scholar] [CrossRef]
- Niti, S.; Vinay, K.S.; Seo, S.Y. Screening of some medicinal plants foe anti-lipase activity. J. Ethnopharmacol. 2005, 97, 453–456. [Google Scholar] [CrossRef]
- Attele, A.S.; Wu, J.A.; Yuan, C.S. Ginseng pharmacology: Multiple constituents and multiple actions. Biochem. Pharmacol. 1999, 58, 1685–1693. [Google Scholar] [CrossRef]
- Li, T.S.C. Asian and American ginseng: A review. Hort Technol. 1995, 5, 27–34. [Google Scholar]
- Karu, N.; Reifen, R.; Kerem, Z. Weight gain reduction in mice fed panax ginseng saponin, a pancreatic lipase inhibitor. J. Agric. Food Chem. 2007, 55, 2824–2828. [Google Scholar] [CrossRef]
- Park, E.K.; Choo, M.K.; Han, M.J.; Kim, D.H. Ginsenoside Rh1 possesses antiallergic and anti-inflammatory activities. Int. Arch. Allergy Immunol. 2004, 133, 113–120. [Google Scholar] [CrossRef]
- Ki, S.K.; Noriko, Y.; Kim, H.Y.; Takuya, O.; Yasuo, S.; Takako, Y. Increase in the free radical scavenging activities of American ginseng by heat processing and its safety evaluation. J. Ethnopharmacol. 2007, 113, 225–232. [Google Scholar] [CrossRef]
- Hu, C.; Kitts, D. Free radical scavenging capacity as related to antioxidant activity and ginsenoside composition of Asian and North American ginseng extracts. J. Amer. Oil Chem. Soc. 2001, 78, 249–255. [Google Scholar] [CrossRef]
- Liu, W.C.; Zheng, Y.N.; Masato, S.; Wang, H.M.; Yoshiyuki, K.; Ling, M.S.; Feng, Y.X. Saponins from stems and leaves of Panax quinquefolium prevented high fat diet-induced obesity in mice. Phytomedicine 2008, 15, 1140–1145. [Google Scholar] [CrossRef]
- Lee, Y.N.; Lee, H.Y.; Chung, H.Y.; Kim, S.I.; Lee, S.K.; Park, B.C.; Kim, K.W. In vitro induction of differentiation by ginsenosides in F9 etratocarcinoma cells. Eur. J. Cancer 1996, 32A, 1420–1428. [Google Scholar]
- Kang, J.; Lee, Y.; No, K.; Jung, E.; Sung, J.; Kim, Y.; Nam, S. Ginseng intestinal metabolite-I (GIM-I) reduces doxorubicin toxicity in the mouse testis [J]. Reprod. Toxicol. 2002, 16, 291–298. [Google Scholar] [CrossRef]
- Tawab, M.; Bahr, U.; Karas, M.; Wurglics, M. Degeneration of ginsenosides in humans after oral administration. Drug Metab. Dispos. 2003, 31, 1065–1071. [Google Scholar] [CrossRef]
- Hasegawa, H.; Lee, K.; Nagaoka, T.; Tezuka, Y.; Uchiyama, M.; Kadota, S.; Saiki, I. Pharmacokinetics of ginsenoside deglycosylated by intestinal bacteria and its transformation to biologically active fatty acid esters. Biol. Pharm. Bull. 2000, 23, 298–304. [Google Scholar] [CrossRef]
- Lei, J.; Li, X.; Gong, X.J.; Zheng, Y.N. Isolation, synthesis and structures of cytotoxic ginsenoside derivatives. Molecules 2007, 12, 2140–2150. [Google Scholar] [CrossRef]
- Keum, Y.S.; Han, S.S.; Chun, K.S.; Park, K.K.; Park, J.H.; Lee, S.K.; Surh, Y.J. Inhibitory effects of the ginsenoside Rg3 on phorbol ester-induced cyclooxygenase-2 expression, NF-kappaB activation and tumor promotion. Mutat. Res. 2003, 523–524, 75–85. [Google Scholar] [CrossRef]
- Kim, S.W.; Kwon, H.Y.; Chi, D.W.; Shim, J.H.; Park, J.D.; Lee, Y.H.; Pyo, S.; Rhee, D.K. Reversal of P-glycoprotein-mediated multidrug resistance by ginsenoside Rg (3). Biochem. Pharmacol. 2003, 65, 75–82. [Google Scholar]
- Popovich, D.; Kitts, D. Structure-function relationship exists for ginsenosides in reducing cell proliferation and inducingapoptosis in the human leukemia (THP-1) cell line. Arch. Biochem. Biophys. 2002, 406, 1–8. [Google Scholar] [CrossRef]
- Popovich, D.G.; Kitts, D.D. Ginsenosides can inhibit proliferation and induce apoptosis in cultured leukemia and intestinal cells but effects vary according to the structure of the compounds. FASEB J. 2003, 17, A762. [Google Scholar]
- Park, J.A.; Lee, K.Y.; Oh, Y.J.; Kim, K.W.; Lee, S.K. Activation of caspase-3 protease via a Bcl-2-insensitive pathway during the process of ginsenoside Rh2-induced apoptosis. Cancer Lett. 1997, 121, 73–81. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).