Photooxidation of Some Metallocenes in a Polymer Matrix
Abstract
:Introduction
Results and Discussion
Electronic absorption spectra of metallocene-doped PMMA thin films containing chloroform molecules
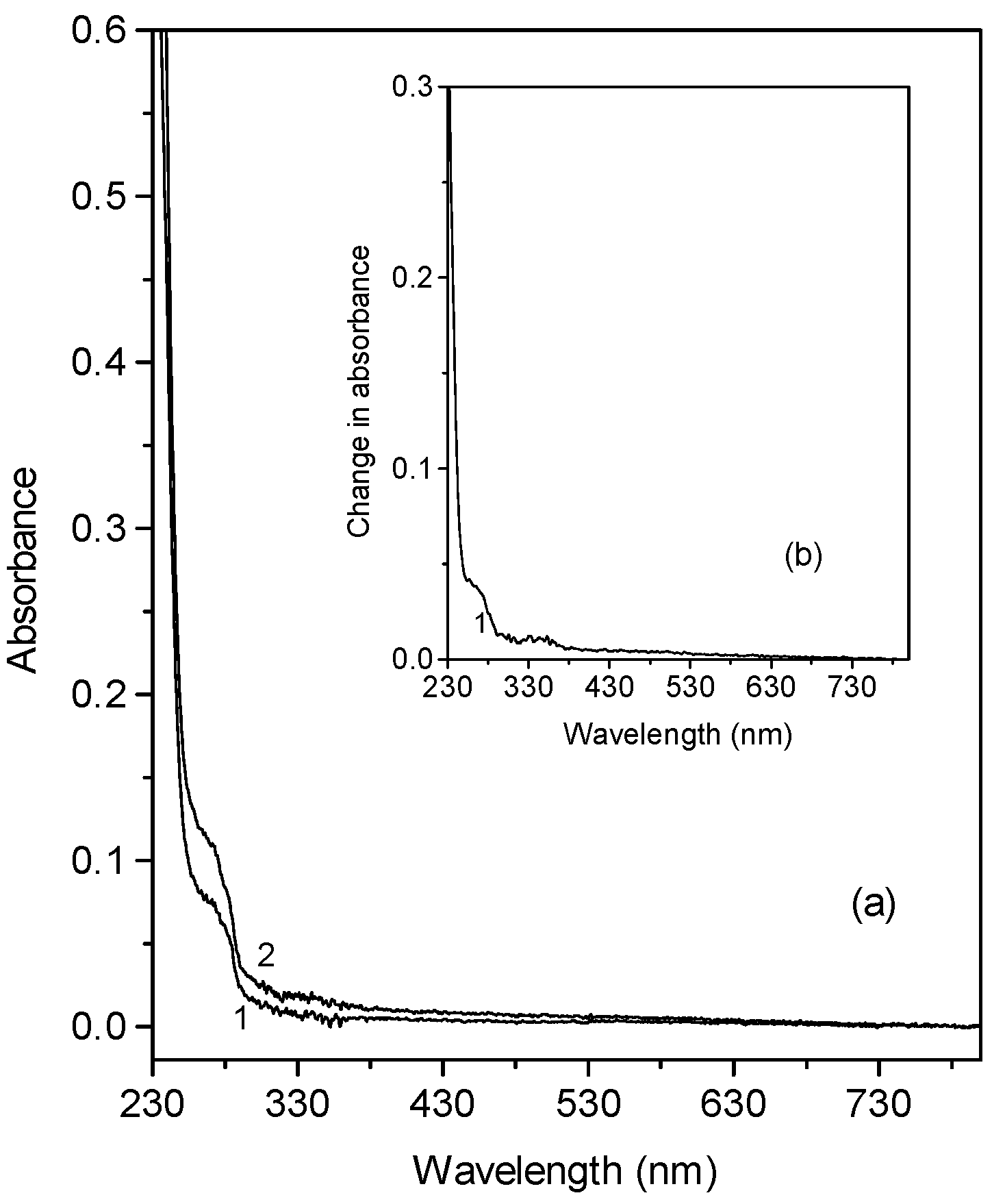
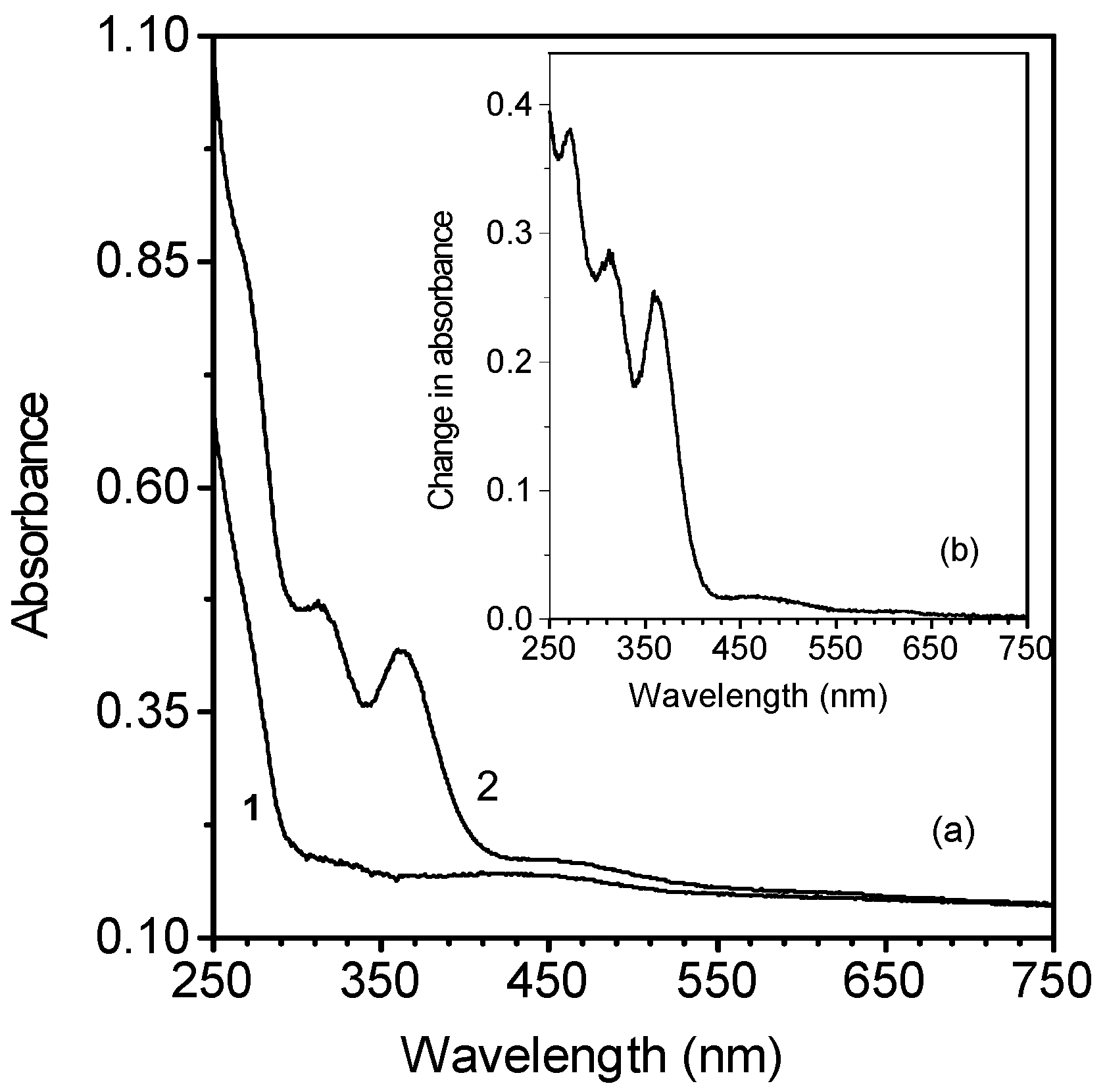
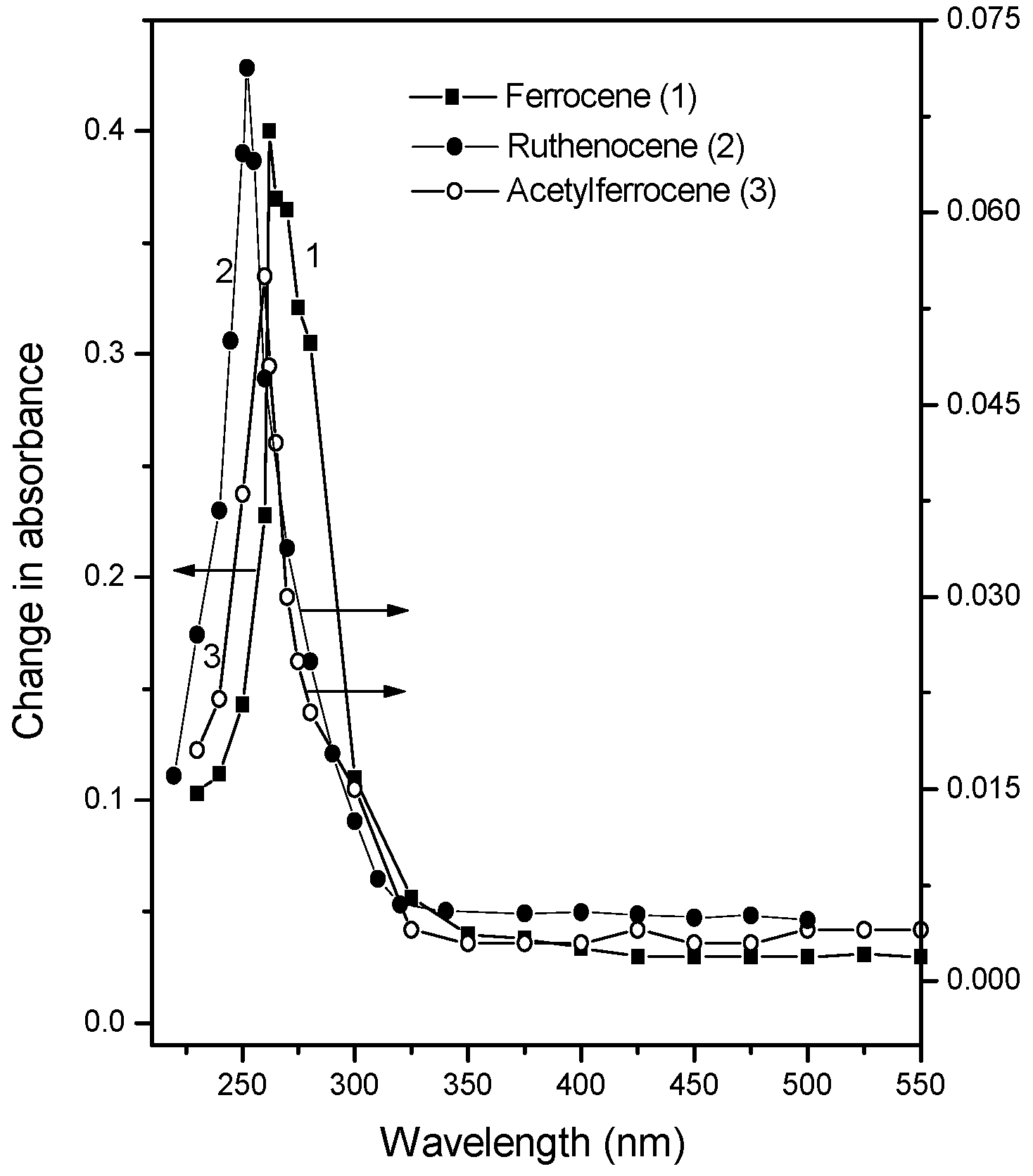
Action spectra of the photoinduced changes of metallocene -doped PMMA films
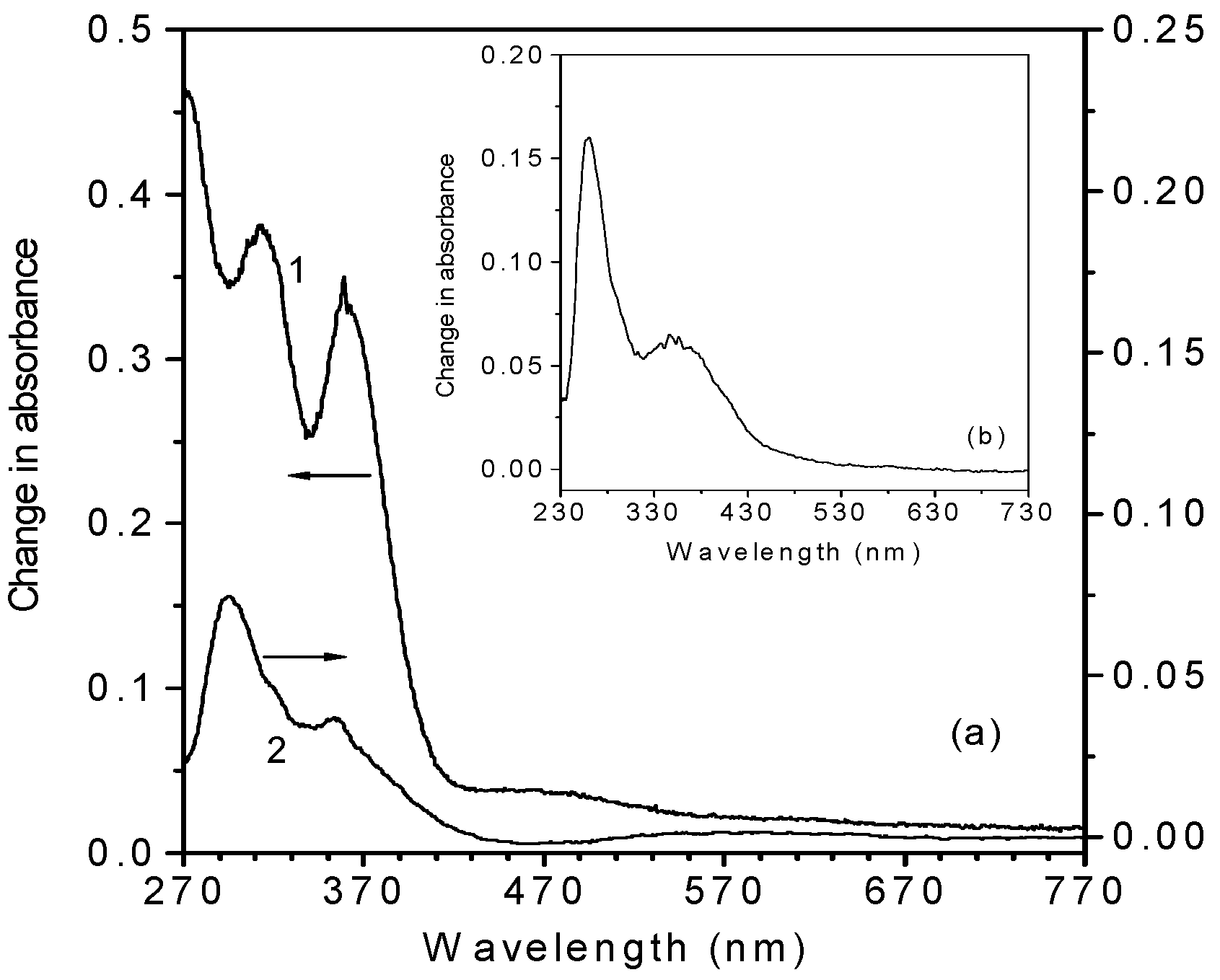
| Materials | Positions of the absorption bands (nm) before photoexcitation | Peak positions of the new absorption bands (appeared after photoexcitation) in the difference spectrum (nm) |
| Ferrocene | 440, 325, 270, 220 | 620, 362, 315, 290 |
| Ferrocenemethanol | 427, 328, 266, 222 | 642, 362, 318, 300 |
| Ferrocenecarbaldehyde | 454, 370, 329, 269, 229 | 608, 317, 295 |
| Ferrocenecarboxylic acid | 441, 354, 305, 261, 221 | 590, 362, 321, 288 |
| Acetylferrocene | 450, 327, 265, 227 | 541, 352, 318, 293 |
| Ferroceneacetic acid | 467, 340, 270, 225 | No new band |
| Benzoylferrocene | 468, 356, 278, 241 | No new band |
| Ruthenocene | 320, 281, 273, 245 | 410, 369, 347, 285, 260 |
| Materials studied | Peak position of the action spectrum for the photoinduced changes (nm) | Peak position of the strong absorption band of the metallocenes around 245-270 (nm) | Position of CT band (nm) |
| Ferrocene | 261 | 261 | 315 |
| Ferrocenemethanol | 270 | 266 | 318 |
| Ferrocenecarboxylic acid | 270 | 261 | 321 |
| Acetylferrocene | 260 | 265 | 318 |
| Ferrocenecarbaldehyde | 260 | 269 | 317 |
| Ruthenocene | 250 | 245 | 281 |
Dependence of the photoinduced changes on the time (duration) of photoexcitation on metallocene-doped PMMA thin films containing chloroform molecules
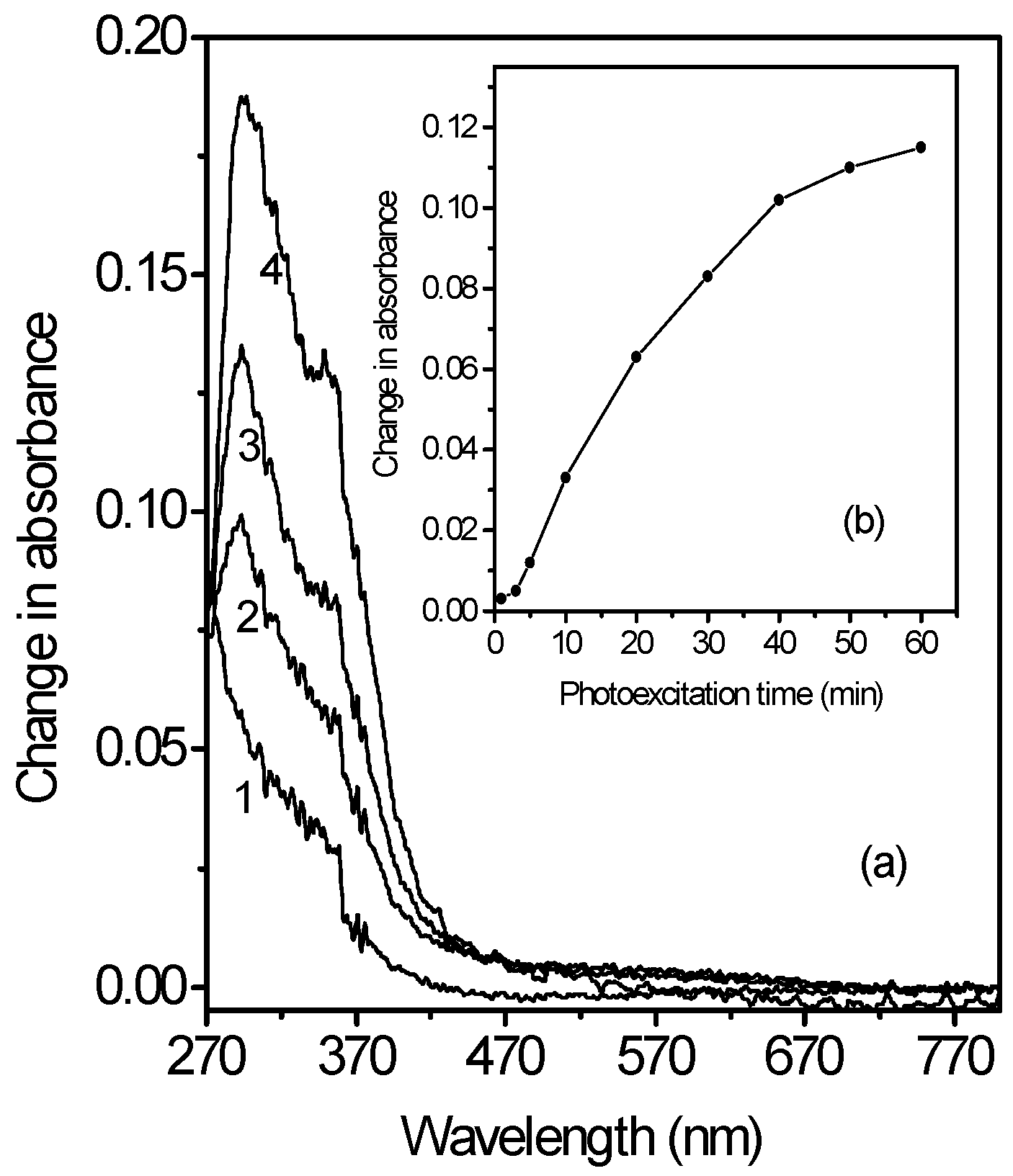
Dependence of the photoinduced changes on the amount of metallocene in the metallocene-doped PMMA thin films containing chloroform molecules

Dependence of the photoinduced changes on the amount of chloroform in the metallocene-doped PMMA thin films containing chloroform molecules

Assignment of the absorption bands that appeared due to photoexcitation of metallocene-doped PMMA thin films containing chloroform molecules
Mechanism of photoinduced changes
FcR1 + RCl → FcR1+ + Cl- + CHCl2.
[Ru(C5H5)2 ] + CHCl2 Cl → [Ru(C5H5)2 ]+ + Cl- + CHCl2.
Effects of the substituent group attached to the ferrocene unit on the photoinduced changes
| Materials studied | Relative photoesponse (%) | Half-wave potential at 298K in acetonitrile vs. SCE (mv) | Value of para Hammett Substituent constant (σp) |
| Ferrocene | 100 | 394 | 0.00 |
| Ferrocenemethanol | 52.50 | 395 | 0.00 |
| Ferrocenecarboxylic acid | 16.25 | 643 | 0.45 |
| Acetylferrocene | 13.75 | 655.5 | 0.502 |
| Ferrocenecarbaldehyde | 13.00 | 700.5 | 0.522 |
Effect of the central metal atom in the metallocene on the photoinduced changes
Photoconductivity in metallocene-doped PMMA thin films containing chloroform molecules and the role of photooxidation of metallocenes in the polymer matrix
Conclusions
Acknowledgements
Experimental
General
References
- Kealy, T. J.; Pauson, P. L. Nature (London) 1951, 168, 1039.
- Carraher, C. E.; Sheets, J. E.; Pittman, C. U. (Eds.) Advances in Organometallic and Inorganic Polymer Science; Marcel Dekker: New York, 1982.
- Turner, A. P. F.; Karube, I; Wilson, G.S. (Eds.) Biosensors; Oxford University Press: London, 1987.
- Geoffroy, G. L.; Wrighton, M. S. Organometallic Photochemistry; Academic Press: New York, 1979; pp. 242–254. [Google Scholar]
- Ozaki, J.; Watanabe, T.; Nishiyoma, Y. J. Phys. Chem. 1993, 97, 1400. [CrossRef]
- Schmitt, R. G.; Hirt, R. C. J. Appl. Polym. Sci. 1963, 7, 1565.
- Miller, J. S.; Epstein, A. J.; Reiff, W. M. Chem. Rev. 1991, 52, 1187.
- Chakraborty, A. K.; Mallik, B. Synth. Metals. 1995, 73, 239. [CrossRef]
- Mallik, B.; Chakraborty, A. K. J. Phys. Chem. 1996, 100, 2145.
- Brand, J. C. D.; Snedden, W. Trans. Faraday Soc. 1957, 53, 894. [CrossRef]
- Scott, D. R.; Becker, R. S. J. Chem. Phys. 1961, 35, 516.
- Scott, D. R.; Becker, R. S. J. Chem. Phys. 1961, 35, 2246.
- Tarr, A. M.; Wiles, D. M. Can. J. Chem. 1968, 46, 2725.
- Traverso, O; Scandola, F. Inorg. Chim. Acta 1970, 4, 493.
- Akaiyama, T; Sugimori, A.; Hermann, H. Bull. Chem. Soc. Jpn. 1973, 46, 1855.
- Augustyniak, A. J.; Wojtezak, J. Trans. Met. Chem. 1984, 9, 303. [CrossRef]
- Augustyniak, A. J.; Wojtezak, J. Trans. Met. Chem. 1984, 12, 167. [CrossRef]
- Tatistcheff, H. B.; Hancock, L. F.; Wrighton, M. S. J. Phys. Chem. 1995, 99, 7689.
- Gutmann, F.; Keyzer, H.; Lyons, L. E. Organic Semiconductors; Krieger: Malabar (FL, USA), 1983; Part B. [Google Scholar]
- Kao, K. C.; Hwang, W. Electrical Transport in Solids; Pergamon Press: Oxford, New York, 1981. [Google Scholar]
- Bhattacharjee, A.; Mallik, B. J. Phys. Chem. Solids 1989, 50, 1113. [CrossRef]
- Bhattacharjee, A.; Mallik, B. Bull. Chem. Soc. Jpn. 1991, 64, 3129. [CrossRef]
- Bhattacharjee, A.; Mallik, B. Jpn. J. Appl. Phys. 1993, 32, 1568.
- Bhattacharjee, A.; Mallik, B. J. Mat. Sci. 1992, 27, 5877.
- Bhattacharjee, A.; Mallik, B. J. Mat. Sci. 1994, 29, 4875.
- Chakraborty, A. K.; Bhattacharjee, A.; Mallik, B. Bull. Chem. Soc. Jpn. 1994, 67, 607. [CrossRef]
- Bera, R. N.; Mallik, B. Solid State Commn. 1998, 108, 695.
- Chakraborty, A. K.; Bera, R. N.; Bhattacharjee, A.; Mallik, B. Synthetic Met. 1998, 97, 63.
- Mallik, B.; Chakraborty, A. K. J. Chem. Soc. Faraday. Trans. 1997, 93, 3677. [CrossRef]
- Chakraborty, A. K.; Mallik, B. Solid State Commun. 1997, 104, 473.
- Bera, R. N.; Mallik, B. Synthetic Met. 1997, 87, 187.
- Inganas, O.; Lundstrom, I. Handbook of conducting polymers; Skothem, T. A., Ed.; Marcel Dekker: New York, 1986; Vol. 1, p. 525. [Google Scholar]
- Lichtenbergen, D. L.; Elkadi, Y.; Gruhn, N. E.; Hughes, R. P.; Curnow, O. J.; Zheng, X. Organometallics 1997, 16, 5209.
- Hohm, U.; Goebel, D. AIP Conf. Proc. (USA) 1998, No. 430. 522.
- Richardson, D. E.; Lang, L.; Eyler, J. R.; Kircus, S.; Zheng, X.; Morse, C. R. P.; Hughes, R. P. Organometallics 1997, 16, 149.
- Traverso, O.; Sostero, S.; Mazzocchin, G. A. Inorg Chim Acta 1974, 11, 237.
- Borrell, P.; Henderson, E. J. Chem. Soc., Dalton Trans., Part-I, 1975; 432.
- Thander, A.; Mallik, B. Solid State Commun. 1999, 111, 341.
- Thander, A.; Mallik, B. Chem. Phys. Letts. 2000, 330, 521.
- Thander, A.; Basak, D.; Mallik, B. Spectrochim. Acta Part. A 2004, 60, 2393. [CrossRef]
- Chen, E. C. M.; Wentworth, W. E. J. Chem. Phys. 1975, 63, 3183.
- Ryan, M. F.; Richardson, D. E.; Lichtenberger, D. L.; Gruhn, N. E. Organometallics 1994, 13, 1190.
- Mallik, B.; Thander, A.; Sanyal, M. K. Spectrochim. Acta, Part A 2005, 61, 485.
- Johnson, C. D. The Hammett Equation; Cambridge University Press: Cambridge, 1973. [Google Scholar]
- Little, W. F.; Reilly, C. N.; Johnson, J. D.; Sanders, A. P. J. Am. Chem. Soc. 1964, 86, 1382.
- Hansch, C.; Leo, A.; Taft, R. W. Chem. Rev. 1991, 91, 65.
- Borrell, P.; Henderson, E. Inorg. Chim. Acta 1975, 12, 215.
- Basak, D; Mallik, B. Chem. Lett. 2004, 33, 1524.
- Thander, A.; Mallik, B. Solid State Commun. 2002, 121, 159.
- Basak, D; Mallik, B. Synthetic Met. 2004, 146, 151.
- Cyr, P. W.; Tzolov, M.; Manners, I.; Sargent, E. H. Macromol. Chem. Phys. 2003, 204, 915.
- Tzolov, M.; Cyr, P. W.; Sargent, E. H.; Manners, I. J. Chem. Phys. 2004, 120, 1990.
- Sample availability: available commercially.
© 2005 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Mallik, B. Photooxidation of Some Metallocenes in a Polymer Matrix. Molecules 2005, 10, 708-727. https://doi.org/10.3390/10060708
Mallik B. Photooxidation of Some Metallocenes in a Polymer Matrix. Molecules. 2005; 10(6):708-727. https://doi.org/10.3390/10060708
Chicago/Turabian StyleMallik, B. 2005. "Photooxidation of Some Metallocenes in a Polymer Matrix" Molecules 10, no. 6: 708-727. https://doi.org/10.3390/10060708
APA StyleMallik, B. (2005). Photooxidation of Some Metallocenes in a Polymer Matrix. Molecules, 10(6), 708-727. https://doi.org/10.3390/10060708




