Abstract
The reactions of bis(cyclopentadienyl)titanium(IV)/zirconium(IV) dichloride with a series of imine-oxime ligands (LH2), derived by condensing benzil-α-monoxime and 2-phenylenediamine, 4-phenylenediamine, 4-methyl-2-phenylenediamine, 2,6-diamino-pyridine, have been studied in anhydrous tetrahydrofuran in the presence of base and metallocycles of the [Cp2M(L)] (M=Ti or Zr) type have been isolated. Tentative structures have been proposed for the products based on elemental analysis, electrical conductance and spectral (electronic, IR and 1H-NMR) data. Proton NMR spectra indicate that on the NMR time scale there is rapid rotation of the cyclopentadienyl ring around the metal-ring axis at 25ºC. Studies were conducted to assess the growth inhibiting potential of the complexes synthesized and the ligands against various bacterial strains.
Introduction
Derivatives of the bent metallocene complexes of Group IV metals have attracted increasing attention because they are applied in a variety of asymmetric catalytic and stoichiometric reactions [1,2,3,4,5,6]. Perhaps the most important application of these complexes is in the isotactic polymerization of propylene. The introduction of modifications to the classical ligands is a very effective way of varying the physical and chemical properties of the parent metallocenes over a very wide range in order to incorporate novel reactivity and optimize existing properties [1]. In continuation of our earlier studies [7,8,9,10,11,12,13,14,15,16] on the coordination behaviour of new type of ligands towards Cp2MCl2, we report here the reactions of Cp2MCl2 (M=Ti or Zr) with imine ligands 1 derived from benzil-α-monoxime.
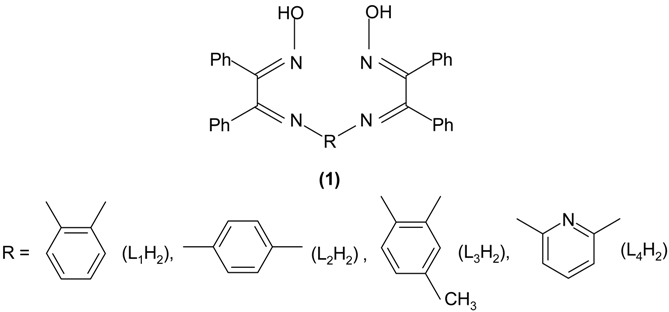
Results and Discussion
A systematic study of the reactions of bis(cyclopentadienyl)titanium(IV)/zirconium (IV) dichloride with imine-oxime ligands (molar ratio 1:1) in anhydrous THF in the presence of triethylamine may be represented by the following equation:
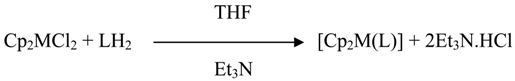
Colours, elemental analysis and molar conductivities are listed in Table 1. Complexes are soluble in dimethylformamide dimethyl sulphoxide, nitrobenzene and tetrahydrofuran. The molar conductances of the complexes in DMF are in the 5-12 ohm-1cm2mol-1 range, which are well below the range observed for univalent electrolytes in this solvent. The electronic spectra of the complexes show a broad band in at ca. 450 nm which can be assigned to a charge-transfer, a result in accord with them (n-1)d0ns0 electronic configuration. The ligands show three absorption maxima at ca. 220, 270 and 520 nm which are assigned to n → π* or π → π* transitions associated with imine and phenyl functions of the ligands. In their respective complexes, these bands shift slightly to higher frequencies.
The IR spectral bands most useful for determining the coordination mode for the ligands and complexes are listed in Table 2. Absorption bands occurring at ca. 3000 cm-1 for υ(C-H), ca. 1425 cm‑1 for υ(C-C) and ca. 1025 cm-1 for υ(C-H) in all the complexes are assigned to the cyclopenta- dienyl group and indicate that these groups are π-bonded to metal [8]. The IR spectra of ligands exhibit a broad, medium intensity band around 3200 cm-1, which is assigned [17] to υ(O-H) of the oxime group. However, in complexes this band disappears indicating the formation of bond between metal and oxygen. This is also confirmed by the appearance of band at ca. 445 – 430 cm-1, assignable [18] to υ(M-O). The strong and medium intensity bands around 1620-1610 cm-1 and 1580-1570 cm-1 in the spectra of ligands are assigned [19] to υ(C=N) and υ(C=N) (oxime), respectively, which remains almost at the same position in the complexes, indicating that C=N groups are not involved in bond formation. The ligand L4H2 and its corresponding titanium(IV)/zirconium(IV) complexes show weak bands at ca. 1560, 610 and 400 cm-1 assignable [20] to pyridine ring deformation bands.

Table 1.
Characterization data of titanium(IV) and zirconium(IV) complexes
| Reactants (molar ratio) | Time (h) | Product; Yield (%) | Colour | Found (calcd.) % | |||
|---|---|---|---|---|---|---|---|
| C | H | N | M | ||||
| Cp2TiCl2+L1H2+Et3N (1 : 1 : 2) | 10 | [Cp2Ti(L1)], 68 | Yellow | 75.4 (75.6) | 4.8 (4.9) | 7.8 (8.0) | 6.8 (6.8) |
| Cp2ZrCl2+L1H2+Et3 N(1 : 1 : 2) | 12 | [Cp2Zr(L1)], 65 | Dark brown | 71.0 (71.2) | 4.4 (4.6) | 7.3 (7.5) | 12.2 (12.3) |
| Cp2TiCl2+L2H2+Et3N (1 : 1 : 2) | 10 | [Cp2Ti(L2)], 70 | Dark brown | 75.3 (75.6) | 4.6 (4.9) | 7.8 (8.0) | 6.8 (6.8) |
| Cp2ZrCl2+L2H2+Et3N (1 : 1 : 2) | 12 | [Cp2Zr(L2)], 60 | Yellowish brown | 71.0 (71.2) | 4.4 (4.6) | 7.4 (7.5) | 12.1 (12.3) |
| Cp2TiCl2+L3H2+Et3N (1 : 1 : 2) | 12 | [Cp2Ti(L3)], 68 | cream | 75.5 (75.8) | 5.0 (5.1) | 7.6 (7.9) | 6.6 (6.7) |
| Cp2ZrCl2+L3H2+Et3N (1 : 1 : 2) | 14 | [Cp2Zr(L3)], 60 | Light brown | 71.4 (71.5) | 4.8 (4.9) | 7.3 (7.4) | 12.0 (12.0) |
| Cp2TiCl2+L4H2+Et3N (1 : 1 : 2) | 15 | [Cp2Ti(L4)], 62 | Light brown | 73.6 (73.8) | 4.6 (4.7) | 9.8 (10.0) | 6.7 (6.8) |
| Cp2ZrCl2+L4H2+Et3N (1 : 1 : 2) | 15 | [Cp2Zr(L4)], 58 | Dark brown | 69.3 (69.5) | 4.4 (4.5) | 9.2 (9.4) | 12.3 (12.3) |
Where,
- L1H2 = ligand derived from benzil-α-monoxime and 2-phenylenedianine
- L2H2 = ligand derived from benzil-α-monoxime and 4-phenylenedianine
- L3H2 = ligand derived from benzil-α-monoxime and 4-methyl-2-phenylenedianine
- L4H2 = ligand derived from benzil-α-monoxime and 2,6-diaminopyridine

Table 2.
IR (cm-1) and 1H-NMR (δ, ppm) data of titanium(IV) and zirconium(IV) complexes
| Complex | IR | 1H NMR | Aromatic ring | |||
|---|---|---|---|---|---|---|
| η5-C5H5 | υ(C=N) | υ(N-O) | υ(M-O) | η5-C5H5 | ||
| [Cp2Ti(L1)] | 3000s, 1420m, 1025m | 1620s, 1585m | 1230m | 440m | 6.80s | 7.65-7.82m |
| [Cp2Zr(L1)] | 3000s, 1425m, 1020m | 1610s, 1580m | 1225m | 430m | 6.68s | 7.50-7.80m |
| [Cp2Ti(L2)] | 3010s, 1410m, 1020m | 1615s, 1575m | 1235m | 445m | 6.85s | 7.70-7.85m |
| [Cp2Zr(L2)] | 3015s, 1420m, 1015m | 1620s, 1570m | 1220m | 440m | 6.72s | 7.40-7.65m |
| [Cp2Ti(L3)] | 3000s, 1430m, 1025m | 1610s, 1575m | 1230m | 435m | 6.78s | 7.60-7.78m |
| [Cp2Zr(L3)] | 3010s, 1420m, 1030m | 1620s, 1570m | 1225m | 430m | 6.70s | 7.42-7.58m |
| [Cp2Ti(L4)] | 2990s, 1415m, 1025m | 1625s, 1585m | 1240m | 440m | 6.75s | 7.60-7.80m 8.20-8.35m |
| [Cp2Zr(L4)] | 3000s, 1410m, 1025m | 1610s, 1575m | 1235m | 430m | 6.65s | 7.45-7.70m 8.10-8.25m |
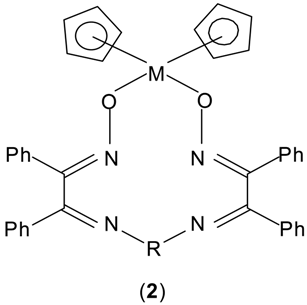
The 1H-NMR spectra of the ligands and complexes (Table 2) have been recorded in deuterated dimethyl sulphoxide. The line intensities were determined by planimetric integration. The complexes show signals at δ 6.65-6.85, which may be assigned to the cyclopentadienyl ring protons and indicate the rapid rotation of the ring about the metal-ring axis. A signal in all ligands at ca. δ 8.20-8.40 may be assigned to the proton of oxime group. In complexes this signal disappears, indicating the displacement of the oxime group hydrogen. The multiplet at ca. 7.40-7.85 may be due to the aromatic ring protons. The ligand L3H2 and its corresponding complexes show a signal at ca. δ 2.0 due to the CH3 group. Thus on the basis of elemental analysis, conductance and spectral data, the following general structure 2 is proposed for the complexes.
The antibacterial activity of the complexes together with the parent ligands has been screened against Gram positive Bacillus subtilis and Gram negative Escherichia coli by the paper disk plate method [21] at 1000 ppm conc. The inhibition zone (mm) around each disk was measured after 24h and the results of these studies are listed in Table 3. The results lead to the following conclusions:
- (a)
- The complexes are slightly more toxic than the parent ligands.
- (b)
- Then titanium complexes show better activity than zirconium complexes.
- (c)
- The best activity was recorded with bis(cyclopentadienyl)titanium (IV) derivatives with ligand L4H2 i.e. derived from 2,6-diaminopyridine.

Table 3.
– Antibacterial activity of imine – oxime ligands and their corresponding titanium(IV)/ zirconium(IV) complexes
| Compound | Diameter of inhibition zone (mm) | |
|---|---|---|
| B. Subtilis | E-coli | |
| L1H2 [Cp2Ti(L1)] [Cp2Zr(L1)] | 6 14 10 | 8 15 10 |
| L2H2 [Cp2Ti(L2)] [Cp2Zr(L2)] | 5 10 8 | 5 10 7 |
| L3H2 [Cp2Ti(L3)] [Cp2Zr(L3)] | 12 16 14 | 10 15 12 |
| L4H2 [Cp2Ti(L4)] [Cp2Zr(L4)] | 12 16 14 | 12 18 15 |
| Ampicillin | 35 | 35 |
Experimental
General
All reactions were carried out under strictly anhydrous conditions. THF was dried by heating under reflux over sodium wire. Et3N was purified by published method [22]. Bis(cyclopentadienyl)-titanium(IV)/zirconium(IV) dichloride were prepared by heating CpNa with the appropriate metal chloride in a N2 atmosphere [23]. Elemental analysis and physical measurements were made as noted earlier [7]. The ligands were prepared as reported in the literature [24].
Reactions of Cp2MCl2 (M=Ti or Zr) with ligands
To a solution of bis(cyclopentadienyl)titanium(IV)/zirconium(IV) dichloride (20 mmol) in dry THF (ca. 40 mL) was added the appropriate ligand (20 mmol). To the resulting clear solution, Et3N (40 mmol) was added and the mixture was stirred for ca. 10-15 h at room temperature. Precipitated Et3N·HCl was removed by filtration and the volume of the solution was reduced to ca. 15 mL under reduced pressure. Light petroleum (b.p 60-80ºC) (10 mL) was then added. The coloured precipitate, thus obtained, was thoroughly washed with Et2O and recrystallized from 1:1 THF-Et2O. The details and important physical characteristics of new compounds are listed in Table 1.
Acknowledgements
The authors are grateful to UGC, New Delhi, for financial assistance
References
- Quin, Y.; Huang, J.; Bala, M.D.; Lian, B.; Zhang, H.; Zhang, H. Chem. Rev. 2003, 103, 2633.
- Togni, A.; Halterman, R.L. Metallocenes: Synthesis-Reactivity-Applications; Wiley-VCH: New York, 1998. [Google Scholar]
- Long, N.J. Metallocenes; Blackwell Science: Malden, M A, 1998. [Google Scholar]
- Boffa, L.S.; Novak, B.M. Chem. Rev. 2000, 107, 1479.
- Bochmann, M. Comprehensive Organometallic Chemistry II.; Abel, E.W., Stone, F.G.A., Wilkinson, G., Eds.; Pergamon: New York, 1995; Vol 4, p. 273. [Google Scholar]
- Jubb, J.; Song, J.; Richeson, D.; Gambarotta, S. Comprehensive Organometallic Chemistry II.; Abel, E.W., Stone, F.G.A., Wilkinson, G., Eds.; Pergamon: New York, 1995; Vol 4, p. 543. [Google Scholar]
- Pandey, O.P.; Sengupta, S.K.; Mishra, M.K.; Tripathi, C.M. Bioinorg. Chem. & Appl. 2003, 1, 35.
- Sengupta, S.K.; Pandey, O.P.; Bhatt, A.; Srivastava, V.; Mishra, K.N. Indian J. Chem. 2002, 41A, 1421.
- Bhatt, A.; Sengupta, S.K.; Pandey, O.P. Indian J. Chem. 2001, 40A, 994.
- Pandey, O.P.; Sengupta, S.K.; Baranwal, B.P.; Shukla, S.K.; Bhatt, A. Z. Naturforsch. 2001, 54b, 141.
- Mala, A.; Srivastava, A.K.; Pandey, O.P.; Sengupta, S.K. Transition Met. Chem. 2000, 25, 613.
- Sengupta, S.K.; Pandey, O.P.; Srivastava, A.K.; Bhatt, A.; Mishra, K.N. Transition Met. Chem. 1999, 24, 703.
- Sengupta, S.K.; Pandey, O.P.; Srivastava, B.K.; Sharma, V.K. Transition Met. Chem. 1998, 23, 349.
- Srivastava, B.; Srivastava, S.K.; Pandey, O.P.; Sengupta, S.K. Gazz. Chim. Ital. 1997, 127, 827.
- Rai, R.; Mishra, K.D.; Pandey, O.P.; Sengupta, S.K. Polyhedron 1992, 11, 123.
- Srivastava, V.; Pandey, O.P.; Sengupta, S.K.; Tripathi, S.C. J. Organomet. Chem. 1987, 321, 27.
- Sharma, V.K.; Pandey, O.P.; Sengupta, S.K. Synth. React. Inorg. Met-Org. Chem. 1991, 21, 1587.
- Hey-Hawkins, E. Chem. Rev. 1994, 94, 1661.
- Pandey, O.P. Indian J. Chem. 1987, 26A, 978.
- Sengupta, S.K.; Pandey, O.P.; Srivastava, A.K.; Mishra, K.D. Indian J. Chem. 1999, 38A, 951.
- Carcelli, M.; Mazza, P.; Pelizzi, C.; Pelizzi, G.; Zani, F. J. Inorg. Biochem. 1995, 57, 43.
- Vogel, A.I. A Text Book of Practical Organic Chemistry; Longmans Green: London, 1948. [Google Scholar]
- Wilkinson, G.; Birmingham, J.M. J. Am. Chem. Soc. 1954, 76, 4281.
- Al- Sogair, F.M.S. Transition Met. Chem. 2002, 27, 299.
- Sample availability: Available from the authors
© 2005 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
