Abstract
Background/Objectives: Antimicrobial resistance (AMR) is a major public health threat, which is exacerbated by the lack of new antibiotics and the emergence of multidrug-resistant (MDR) superbugs. Comprehensive efforts and alternative strategies to combat AMR are urgently needed to prevent social, medical, and economic consequences. Pseudomonas aeruginosa is a pathogen responsible for a wide range of infections, from soft tissue infections to life-threatening conditions such as bacteremia and pneumonia. Bacteriophages have been considered as a potential therapeutic option to treat bacterial infections. Our aim was to isolate phages able to infect MDR P. aeruginosa strains. Methods: We isolated two lytic phages, using the conventional double layer agar technique (DLA), from samples obtained from the influent of a wastewater treatment plant in Concepción, Chile. The phages, designated as PaCCP1 and PaCCP2, were observed by electron microscopy and their host range was determined against multiple P. aeruginosa strains using DLA. Moreover, their genomes were sequenced and analyzed. Results: Phage PaCCP1 is a member of the Septimatrevirus genus and phage PaCCP2 is a member of the Pbunavirus genus. Both phages are tailed and contain dsDNA. The genome of PaCCP1 is 43,176 bp in length with a GC content of 54.4%, encoding 59 ORFs, one of them being a tRNA gene. The genome of PaCCP2 is 66,333 bp in length with a GC content of 55.6%, encoding 102 non-tRNA ORFs. PaCCP1 is capable of infecting five strains of P. aeruginosa, whereas phage PaCCP2 is capable of infecting three strains of P. aeruginosa. Both phages do not contain bacterial virulence or AMR genes and contain three and six putative Anti-CRISPR proteins. Conclusions: Phages PaCCP1 and PaCCP2 show promise as effective treatments for MDR P. aeruginosa strains, offering a potential strategy for controlling this clinically important pathogen through phage therapy.
1. Introduction
P. aeruginosa is a Gram-negative, non-glucose-fermenting bacteria that frequently causes opportunistic hospital-acquired infections. It is responsible for infections in various organs, including the skin, lungs, urinary tract, kidneys, and the gastrointestinal tract. Moreover, it is recognized as the most important cause of bacterial infection in cystic fibrosis patients, as the inflammation it triggers contributes to the progression of lung disease [1]. P. aeruginosa infects patients with burn wounds, immunodeficiency, chronic obstructive pulmonary disease, cancer, and those with severe infection requiring mechanical ventilation [2]. The symptoms of these infections include generalized inflammation and sepsis, which often require prolonged antimicrobial chemotherapy. If such colonization occurs in critical organs, the outcome can be fatal [3]. P. aeruginosa infections are particularly difficult to treat because of its intrinsic resistance to antibiotics [4], and it shows high resistance to a series of commonly used antibiotics, including β-lactams, fluoroquinolones, and aminoglycosides [2]. The number of MDR strains is increasing worldwide [5]. In addition, P. aeruginosa produces a variety of virulence factors, including rhamnolipids, pyocyanin, and biofilms [6,7]. The list of priority pathogens compiled by the World Health Organization (WHO) [8] was recently updated [9]. P. aeruginosa was reclassified from the critical priority group to the high priority group, but it remains very relevant to global public health, which implies the need to develop new antimicrobials against this pathogen.
Bacterial viruses, known as bacteriophages, have been proposed as a novel therapeutic strategy to control bacterial infections, termed “phage therapy”. Phages offer a promising solution due to their unique antibacterial properties. Although phages were first explored for human therapy shortly after their discovery over a century ago, their use was soon eclipsed by the advent of antibiotics, except in some countries in eastern Europe, like Georgia and its Eliava Institute in Tbilisi [10,11]. Unlike antibiotics, phages are highly specific, targeting only harmful bacteria while leaving beneficial bacteria unharmed. This specificity prevents collateral damage to the healthy commensal microbiota of humans and animals, in contrast to the effects of broad-spectrum antibiotics [12]. In recent years, phage therapy has experienced a resurgence, primarily driven by the increasing issue of AMR [13].
Due to the importance of P. aeruginosa, many specific bacteriophages against this pathogen have been described. Most of them are virulent (strictly lytic), tailed, have dsDNA, and belong to the Caudoviricetes class [14]. Common receptors targeted by P. aeruginosa phages include LPS, OMPs, EPS, pili, and flagella [15]. The exact receptors used can vary between phages and strains they infect. These interactions are highly specific, which allows phages to be selective to the bacterial species and strains they infect. P. aeruginosa phages have been isolated from different sources all over the world; nevertheless, it seems that sewage, including hospital and wastewater treatment plant sewage, is the best choice for their isolation [16,17]. Many of these phages have been used in cocktail applications in vitro to evaluate their potential against clinical isolates, including MDR strains in planktonic cultures or biofilms [18]. For instance, it was recently demonstrated that the phage Paride can kill dormant cells, and, in combination with carbapenem or meropenem, eradicated deep-dormant cultures in vitro and reduced a resilient bacterial infection of a tissue cage implant in mice [19]. Moreover, several works have been performed in animals [20,21] and human trials [22,23,24,25,26]. Further research on isolating phages is necessary, as using cocktails with varying host ranges in a single suspension is more effective at inhibiting bacterial infections and reduces the likelihood of bacterial resistance [27].
One of the main mechanisms by which bacteria become resistant to phages is the CRISPR-Cas system [28,29,30]. In P. aeruginosa, diverse types of CRISPR-Cas systems have been identified in >30% of the strains, with the number likely to increase as more of these gene families are discovered [31,32,33,34]. On the other hand, there is an evolutionary “arms race” between phages and bacteria. To overcome the bacterial protection afforded by CRISPR-Cas systems and promote their own replication, phages have developed Anti-CRISPR systems (Acr proteins) [35,36].
Our aim was to isolate lytic phages able to infect multiple strains of the relevant pathogen P. aeruginosa strains. In this work, we describe phages PaCCP1 and PaCCP2, which were isolated from wastewater in Concepción, Chile. These phages belong to distantly related genera in the class Caudoviricetes. They can infect and propagate in diverse MDR P. aeruginosa strains.
2. Results and Discussion
2.1. Phage Characterization
Two lytic phages able to infect P. aeruginosa strains were isolated from wastewater samples. The isolated phages, designated as PaCCP1 and PaCCP2, produced circular plaques with a diameter of 2–3 mm in DLA. Electron microscopy revealed that they are tailed-containing phages (Figure 1). Phage PaCCP1 has a capsid head of 53 nm (±4 nm) and a tail of 157 nm (±8 nm), while phage PaCCP2 has a capsid head of 46 nm (±5 nm) and a tail of 154 nm (±6 nm).

Figure 1.
Transmission electron micrographs of phages PaCCP1 (A) and PaCCP2 (B). Images show the head and tail of both phages.
The genome of the phage PaCCP1 consists of a dsDNA molecule, 43,176 bp in length, with a GC content of 54.4% (accession number PQ492277) (Figure 2). Its relative phages belong to the Septimatrevirus genus, formerly known as Septima3virus, which was created in 2015 by ICTV in proposal 2015.054a-dB. Originally, the genus included 5 species, but currently includes 21 species (ICTV proposal 2023.039B). According to the latter proposal, this genus was incorporated in the new subfamily Jondennisvirinae, which contains three genera (Septimatrevirus, Kipunavirus, Kilunavirus) and belongs to the class Caudoviricetes without belonging to a specific family (Figure 3), after the abolishment of morphology-based taxa in 2022 by ICTV [37]. To explore the taxonomy of phage PaCCP1, we downloaded all the Septimatrevirus genomes according to ICTV from GenBank in July 2024.
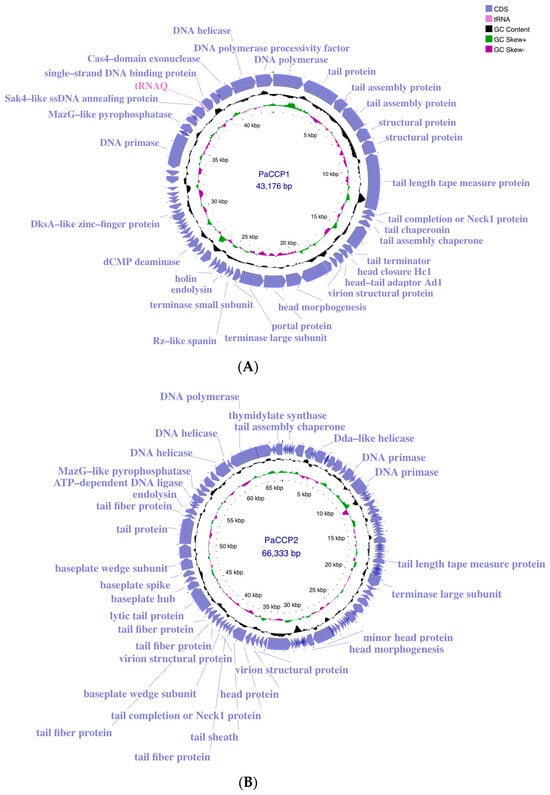
Figure 2.
Genome maps of phages PaCCP1 (A) and PaCCP2 (B) obtained using Proksee. ORFs with identified functions are shown. Moreover, GC content and GC skew are indicated.
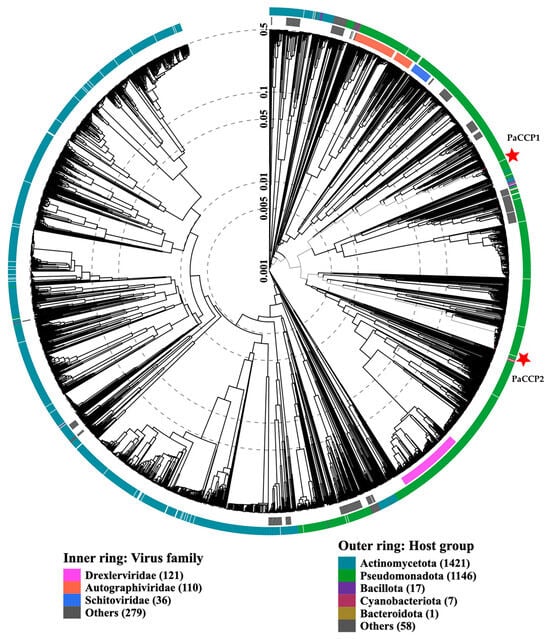
Figure 3.
ViPTree analysis of Pseudomonas phages PaCCP1 and PaCCP2. Phages are identified according to their official ICTV classification, with the outer and inner rings indicating their host group and virus family, respectively.
It is relevant to notice that some Septimatrevirus phages can infect bacteria that belong to genera other than Pseudomonas (Table 1). For instance, phage Samson and DoCa1 can infect plant-associated Xanthomonas strains. Phages DLP1 and DLP2 can infect Stenotrophomonas maltophilia strains. Interestingly, phages DLP1 and DLP2 also can infect Pseudomonas strains showing a cross-taxonomic order infectivity [38].

Table 1.
Septimatrevirus phages and their intergenomic similarity with PaCCP1. Species and genus clusters are shown.
The genome of the phage PaCCP2 consists of a dsDNA molecule, 66,333 bp in length and with a GC content of 55.6% (accession number PQ492278) (Figure 2). Their relatives belong to genus Pbunavirus, formerly known as Pb1likevirus and subsequently renamed Pbunalikevirus. The genus, created in 2009 by ICTV (proposal 2009.001a-gB), belongs to the class Caudoviricetes without belonging to a specific family or subfamily (Figure 3). Originally, the genus comprised seven phages, but it currently includes 32 species (ICTV proposal 2021.061B). Originally, the genus included six Pseudomonas phages and one Burkholderia phage (BcepF1); however, in 2020, the genus Bcepfunavirus was created with phage BcepF1 (ICTV proposal 2020.116B). Pbunavirus is one of the most rapidly growing Pseudomonas myovirus genera (ICTV proposal 2021.061B). Phage PB1 is the type species (the phages within the genus are also known as PB1-like) and it was first isolated and described almost half a century ago by Holloway et al. [47] from sewage samples. Pbunavirus is ubiquitous on Earth, and is found in the US, Europe (France, Portugal, Poland, Spain, Russia, Germany, the Netherlands, and Scotland), and Brazil in soil, freshwater, wastewater, and activated sludge samples [48,49]. Pbunavirus includes phages that can infect only P. aeruginosa strains [49]. To explore the taxonomy of phage PaCCP2, we downloaded the genomes of all the phages described as Pbunavirus according to ICTV in July 2024 (Table 2).

Table 2.
Pbunavirus phages and their intergenomic similarity with PaCCP2. Species and genus clusters are shown.
According to ICTV’s Bacterial and Archaeal Viruses Subcommittee, two phages are assigned to the same species if their genomes are more than 95% identical at the nucleotide level over their full genome length, tested reciprocally [62]. The Subcommittee also established a 70% nucleotide identity of the full genome length as the cutoff for genera, calculated in the same way as the species cutoff. These values can be calculated by several tools, such as BLASTn (% identity multiplied by % coverage) or VIRIDIC [63]. According to Simmonds et al. [64], these values only serve as an approximation of evolutionary relatedness and the relationship among viruses should be explored by phylogenetic methods that are also capable of calculating clade support, such as VICTOR [65]. We used VICTOR because it was specifically designed for prokaryotic viruses and can output classification at the species, genus, and family ranks. Moreover, we compared the VICTOR and VIRIDIC outputs.
According to VIRIDIC, PaCCP1 belongs to a unique species and, together with Guyu, Samson, DoCa1, yazdi, and kaya, forms another genus different from Septimatrevirus (Figure 4). However, according to VICTOR, phages PaCCP1, Guyu, and Kaya belong to the same species and there is not a new genus, because all the Septimatrevirus phages and PaCCP1 belong to the same genus (Figure 5). Therefore, we assume the latter classification. Interestingly, our VICTOR analysis indicated that phages DLP1, DLP2, and PX5 belong to the same species.
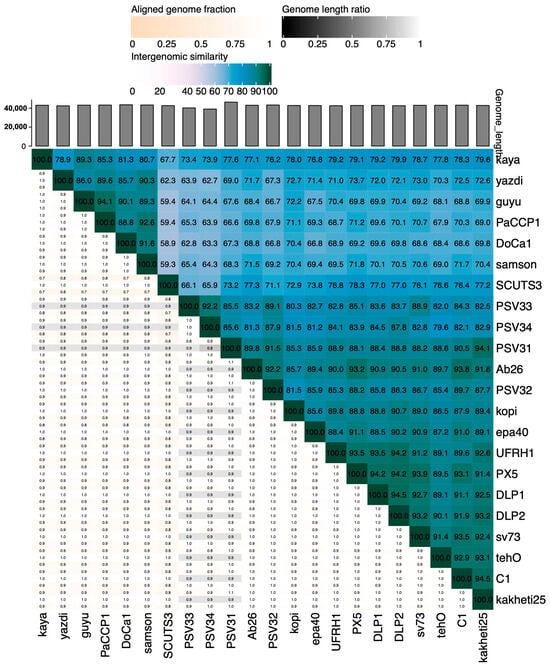
Figure 4.
Heatmap showing the intergenomic similarities of phage PaCCP1 and Septimatrevirus phages.
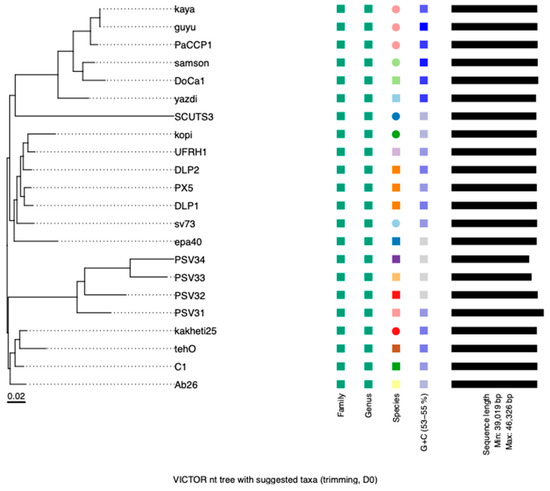
Figure 5.
Phylogenetic analysis of phage PaCCP1 and Septimatrevirus phages. The scale bar indicates the number of substitutions per site. Each species is indicated by a unique color.
The genome annotation of phage PaCCP1 demonstrated that it encodes 59 ORFs, 46 (78%) of which are encoded on the positive strand and 13 (22%) on the negative strand. One ORF encodes a tRNA (trnQ). Twenty-eight ORFs (46.7%) were identified as hypothetical proteins with unknown functions. The functional ORFs include a minor and a major capsid protein, two terminase proteins, a scaffolding protein, three tail proteins, and one exonuclease. No genes associated with bacterial virulence or AMR were identified, and the lifestyle of PaCCP1 was classified as lytic. The proteome structure is very similar to their closest relative, phage Guyu (Figure 6).
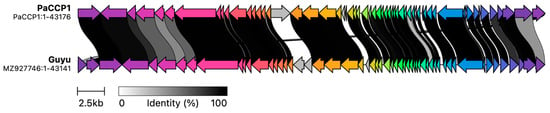
Figure 6.
Comparison of phage PaCCP1 and its closest relative, phage Guyu, using alignment of all annotated proteins. The arrow’s colors represent the gene clusters encoding similar proteins. The lines linking the arrows show gene-encoding proteins that share more than 80% sequence identity.
No further characterization of Guyu and Kaya was performed, such as host range or morphology observed by electronic microscopy. Both phages were isolated from a river in Haining, China, in 2020 using the strain P. aeruginosa PAO1 PA2072 [39]. The Guyu genome is 43,141 bp long, with a CG content of 56%, and it encodes 56 proteins, while the Kaya genome is 43,067 bp in length with a CG content of 54% and encodes 60 proteins. Phage samson, another phage related to PaCCP1, is a Xanthomonas phage that lacks phenotypic characterization. It was isolated from sewage samples from Texas, and contains a 43,314 bp genome with 58 predicted genes [40].
A phage named TR (accession number OL802211) described by Xuan et al. [66] was isolated from sewage samples in Qingdao, China, using P. aeruginosa PAO1. It is not classified as Septimatrevirus yet, but it probably will be. According to VIRIDIC, it showed an intergenomic similarity of 91,8 with PaCCP1. It was observed by electron microscopy, showing that it contains a head of approximately 50 nm with a long noncontractile tail of approximately 170 nm. Its genome is 43,354 bp long, with a CG content of 55%, and it encodes 56 proteins. It was demonstrated that the type IV pilus (TP4) acts as an adsorption receptor for phage TR. In this regard, it has been demonstrated that T4P is not only the virulence factor for some pathogens but is also the receptor for many P. aeruginosa phages [66]. Recently, Su et al. [67] described the host range of phage TR against thirteen P. aeruginosa strains using the visual assessment of plaques on the spot test. They indicated that six (46.2%) of the P. aeruginosa strains tested were lysed by phage TR.
According to VIRIDIC, phage PaCCP2 belongs to the same species as phages LS1, E217, and PaGU11 in the Pbunavirus genus (Figure 7). However, according to VICTOR, it belongs to the same species as epa14, R16, PaGU11, KTN6, LMA2, S1, PA5, DP1, E217, crassa, Pa01, and LS (Figure 8). Some of these phages have been characterized. For instance, it has been demonstrated that phage LS1 (Pbunavirus) has high inhibition potential on the growth of P. aeruginosa biofilm formation [50].
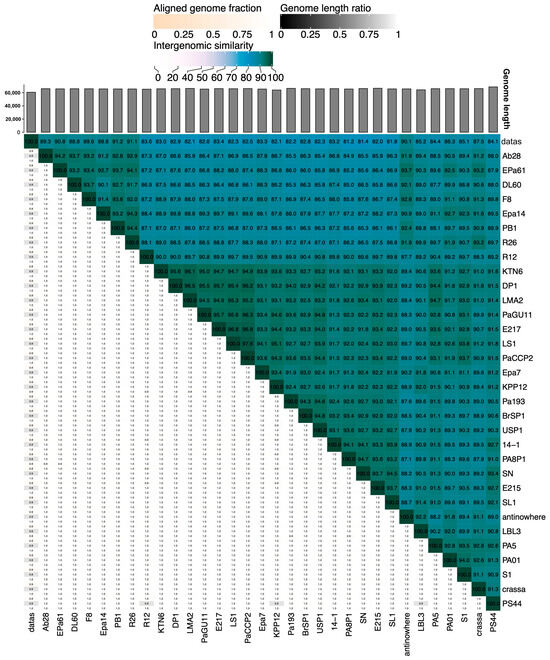
Figure 7.
Heatmap of the intergenomic similarities of phage PaCCP2 with Pbunavirus phages.
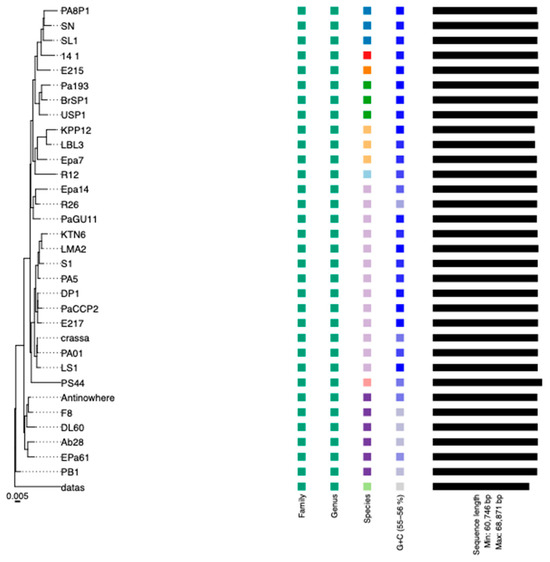
Figure 8.
Phylogenetic analysis of phage PaCCP2 and Pbunavirus phages. The scale bar indicates the number of substitutions per site. Each species is indicated by a unique color.
The genome annotation of phage PaCCP2 demonstrated that it encodes 106 ORFs, 43 (40.6%) of which were detected on the positive strand and 63 (59.4%) on the negative strand. Only 31 ORFs (29.2%) were annotated as functional proteins. The functional ORFs include, among others, two DNA helicases, a DNA polymerase, a large terminase subunit, eleven tail-related proteins, and an endolysin protein. In this regard, it has been described that virion particles of Pbunavirus are composed of at least 22 different proteins [52,68]. No genes associated with bacterial virulence or AMR were identified, and the lifestyle of PaCCP2 was categorized as lytic. The proteome structure is very similar to their closest relative, phage LS1 (Figure 9).

Figure 9.
Comparison of phage PaCCP2 and its closest relative, phage LS1, using alignment of all annotated proteins. The arrow’s colors represent the gene clusters encoding similar proteins. The lines linking the arrows show gene-encoding proteins that share more than 80% sequence identity.
Several Pbunavirus phages are part of a phage cocktail developed to eradicate P. aeruginosa infections. For instance, Pa193 was part of a phage cocktail candidate developed by Armata Pharmaceuticals [56,69]. For instance, Forti et al. [51] demonstrated that a phage cocktail composed of six phages (PYO2, DEV, E215 and E217, PAK_P1 and PAK_P4) was able to lyse P. aeruginosa both in planktonic liquid cultures and in biofilms. From these phages, E217 is a Pbunavirus.
Wannasrichan et al. [70] demonstrated that P. aeruginosa strains resistant to phage JJ01 (Pbunavirus) exhibit hypersensitivity to colistin and reduce biofilm production. This trade-off has been broadly demonstrated [71]. This is helpful for phage therapy, because if bacteria become phage-resistant, they become less virulent at the same time.
2.2. Inhibition Assay and Host Range
We demonstrated that the addition of phages PaCCP1 or PaCCP2 at MOI 10 after 6 h of incubation effectively prevented the growth of P. aeruginosa UC535 or UC550, respectively.
Our experiments, performed to evaluate the host range of phages PaCCP1 and PaCCP2, indicate that even though phage PaCCP1 was isolated with strain UC528, it can successfully infect and propagate in strains UC522, UC532, UC533, and UC536 in DLA. On the other hand, phage PaCCP2, isolated with strain UC335, successfully infects and propagates in strains UC522, UC529, and UC532 in DLA.
Even though all the strains were negative for carbapenemase production using Blue Carba, it is relevant to notice that both phages PaCC1 and PaCCP2 can infect and propagate in strain UC532 (Table 3), which is an MDR strain resistant to amikacin, ciprofloxacin, meropenem, and imipenem. Strains UC529 and UC532 can also be categorized as MDR because they showed non-susceptibility to at least one agent in three antimicrobial categories [72]. These results demonstrated the effectiveness of our isolated phages, PaCCP1 and PaCCP2, against P. aeruginosa and therefore make them a very attractive alternative option to antibiotics or for use in combination with antibiotics or other antimicrobials for improved performance.

Table 3.
Antibiotic susceptibility of P. aeruginosa strains susceptible to PaCCP1 or PaCCP2. Colors indicate the interpretation of the zone diameters: resistance (red), intermediate (yellow), or susceptibility (green). Numbers indicate the diameter in mm of the inhibition zone. The strains infected by PaCCP1 (a) and PaCCP2 (b) are indicated.
2.3. Anti-CRISPR Proteins in Phages PaCCP1 and PaCCP2
As far as we know, Acrs have not been searched for in any previous work on Septimatrevirus and Pbunavirus. Using two tools for cross validation, PaCRISP [73] and AcRanker [74], we identified several putative Acr-encoding genes in both phages PaCCP1 and PaCCP2 (Table 4). Both tools are machine learning-based programs.

Table 4.
Putative Acr-encoding genes detected in phage PaCCP1 and PaCCP2. The cutoff threshold for PaCRISPR is >0.5, and the cutoff threshold for AcRanker is >−5.0.
In phage PaCCP1, seven candidate genes were identified using PaCRISP and five using AcRanker. Merging the results of both predictors, we suggested that CDS 41, 33, and 28 could be Acr-encoding genes because these genes were identified using both tools, were ranked in the first three places in the results obtained with AcRAnker (Table 4), and encode for small proteins (69, 101, and 69, respectively), which is a typical size for Acrs, usually between 50 and 150 amino acids [35]. In addition, all of these genes encode hypothetical proteins without predicted function after functional annotation performed using PHANOTATE [75], a CDS-prediction tool specifically designed for phages, in the Pharokka [76] and PHROGs database [77]. In phage PaCCP2, seventeen candidate genes were identified using AcRanker and nineteen using PaCRISP. CDS 39, 43 73, 35, 36, and 1 were identified in both tools. From them, only CDS 73 has a putative function assigned (head and packaging), and it is the largest one.
It has been described that Acrs proteins do not usually share conserved sequences; therefore, their discovery is sometimes difficult [78]. Anti-CRISPR systems have been described in P. aeruginosa phages [35,79,80], but they have not been studied in detail and have only been investigated in a few of the newly described phages [78]. The validation of the candidate Acr-encoding genes detected in phages PaCCP1 and PaCCP2 needs protein expression, purification, and biochemical characterization, which we are considering for future experiments in new projects.
3. Materials and Methods
3.1. Bacterial Strains
We used P. aeruginosa strains provided by the clinical laboratories of tertiary hospitals in 2020, with no direct involvement of patients in the study. The isolates were preliminarily identified as Pseudomonas spp. using biochemical and physiological tests, colony morphology, and pigment production. Gram-negative bacilli with the above-mentioned characteristics were considered as P aeruginosa based on a positive oxidase test, a triple sugar iron agar reaction of alkaline over no change, growth at 42 °C, and the production of bright blue to blue green diffusible pigment on Mueller–Hinton agar (Thermo Fisher Scientific, Waltham, MA, USA).
Strain UC528 was used to isolate the phage PaCCP1. This bacterial strain was obtained from a skin wound of a patient at a hospital in the city of Concepcion (Chile). Strain UC536 was used to isolate the phage PaCCP2. This bacterial strain was obtained from a urine sample of a patient hospitalized in a hospital located in the city of Talca (Chile). Moreover, 20 clinical strains of P. aeruginosa obtained from hospitalized patients from the same hospitals described above were used to determine the host range. We included P. aeruginosa reference strains obtained from the American Type Culture Collection (ATCC) (Manassas, VA, USA): ATCC 27853, ATCC 9027, and PAO1. All bacterial strains were stored in Luria–Bertani (LB) broth (Thermo Fisher Scientific, Waltham, MA, USA) containing 20% glycerol (Thermo Fisher Scientific, Waltham, MA, USA), kept at −80 °C, and were routinely grown in LB media at 37 °C.
3.2. Sample Collection and Processing
Samples from a municipal wastewater treatment plant (WWTP) in Concepción, Chile, were collected in January 2024. They consisted of 1 L of 24 h composite water from the influent of the WWTP, prior to any treatment. The samples were kindly provided by the Centinela Biobío-UCSC Center, a wastewater monitoring center of the Universidad Católica de la Santísima Concepción, Chile. Samples were collected in sterile 50 mL tubes (Corning, Glendale, AZ, USA) and stored at 4 °C until use within 2 days. The samples were centrifuged at 8000× g for 45 min at 4 °C, and the supernatant was collected to remove large particles and most bacterial cells. To remove the remaining bacterial debris while retaining the viral fraction, the supernatant was passed through a sterile 25 mm Whatman glass fiber membrane (MilliporeSigma, Burlington, MA, USA) with a pore size of 0.22 µm.
3.3. Disk Diffusion Method for the Determination of Antimicrobial Resistance
All strains were tested for carbapenemase production using Blue Carba [81], and antibiotic susceptibility was determined using the disk diffusion method in Mueller–Hinton agar according to the Clinical and Laboratory Standards Institute (CLSI) [82]. The zones of growth inhibition around each of the antibiotic disks were measured and related to the susceptibility of the isolate and to the diffusion rate of the drug through the agar medium (Thermo Fisher Scientific, Waltham, MA, USA). The zone diameters of each drug were interpreted using the criteria published by the CLSI. The test was performed using the following antibiotics: amikacin (AMK), ciprofloxacin (CIP), meropenem (MEM), imipenem (IPM), ceftazidime/avibactam (CZA), ceftazidime (CAZ), cefepime (FEP), and piperacillin/tazobactam (TZP) (Thermo Fisher Scientific, Waltham, MA, USA).
3.4. Phage Isolation
The phages were isolated using the classical DLA method [83]. Overnight cultures of 20 P. aeruginosa strains were prepared in LB broth and incubated at 37 °C. An aliquot of 100 µL from the overnight culture was inoculated into fresh LB broth and incubated at 37 °C until the mid-exponential phase was reached. Aliquots (100 µL) of each bacterial suspension were mixed with 100 μL of filtered environmental samples and 3 mL of melted (50 °C) soft LB agar (0.5%) supplemented with CaCl2 (final concentration 5 mM) (Thermo Fisher Scientific, Waltham, MA, USA). After overnight incubation of the plates at 37 °C, single plaques were picked and harvested in 500 µL of SM buffer (100 mM NaCl, 8 mM MgSO4, and 50 mM Tris-HCl, pH 7.5) (Thermo Fisher Scientific, Waltham, MA, USA). The isolates were then purified three times by DLA and sequential isolation.
3.5. Susceptibility to RNAse and Chloroform
The susceptibility of the isolated phages to RNAse was determined by DLA after the addition of RNAse (final concentration10 µg mL−1) to the bottom agar. The resistance of the isolates to chloroform (Thermo Fisher Scientific, Waltham, MA, USA) was determined by adding it to 200 µL of phage suspensions at a final concentration of 10 or 100 µL mL−1. The mixtures were then incubated for 60 min at room temperature and phage viability was assessed using DLA. All assays were performed in triplicate.
3.6. Propagation and Concentration of Phages
Concentrated stocks of phages were obtained using Amicon ultrafiltration membranes (100 kDa) (MilliporeSigma, Burlington, MA, USA). Phage suspensions were mixed with logarithmic growing cultures of each host strain, as described previously. Phages were then added at a multiplicity of infection (MOI) of 0.01 in 15 mL bacterial cultures. Samples were incubated overnight at 37 °C with shaking at 120 rpm. After incubation, the suspensions were centrifuged and filtered to remove bacterial debris and then transferred to Amicon and centrifuged at 3000× g for 20 min at 4 °C or until a remaining volume of less than 1 mL was achieved. The titer of the phage suspensions was determined by DLA, as described previously, and expressed as plaque-forming units per milliliter (PFU mL−1).
To obtain more concentrated stocks, precipitation with polyethylene glycol 8000 (PEG) (Merck, Rahway, NJ, USA) was performed. Briefly, phage suspensions and bacterial cultures were mixed, incubated overnight at 37 °C, filtered, and precipitated with 10% PEG and 1 M NaCl (Merck, Rahway, NJ, USA). The mixtures were then incubated at 4 °C overnight and centrifuged for 1 h at 16,000× g to obtain a phage pellet, which was resuspended in 1 mL of SM buffer. The titer of the phage suspensions was determined using DLA, as described previously.
3.7. Electron Microscopy
We visualized the morphology of the isolated phages using an aliquot of 15 µL from pure suspensions with glow-discharged 200 mesh copper-coated grids. Phages on the grids were incubated for 30 s before blotting off the liquid using a Whatman filter pmembrane (MilliporeSigma, Burlington, MA, USA). The suspensions were then fixed with 5 µL of glutaraldehyde (Merck, Rahway, NJ, USA), incubated for 10 s, and excess liquid was blotted off. The samples were stained with 3 µL of 2% uranyl acetate (Merck, Rahway, NJ, USA) and incubated for 30 s. The microscope used was a JEM 2011 (Jeol, Tokyo, Japan) at Centro de Espectroscopía y Microscopía (CESMI) at Universidad de Concepción. Images were analyzed using ImageJ software v1.54i to calculate the length of their tail and capsid using three particles for each phage.
3.8. DNase and RNase Treatment of Phage Stocks
For the effective purification of phage DNA, it was necessary to first remove bacterial DNA and RNA debris. For this, 447 µL of each purified phage suspension was mixed with 50 μL DNase buffer, 3 μL DNase (AMPD1–1KT) (Sigma-Aldrich, Burlington, MA, USA), and 3 μL RNase suspension at 5 mg/mL. The samples were then incubated for 1 h at 37 °C. DNase was inactivated by adding 10 μL DNase stop solution and 40 μL 50 mM EDTA (Thermo Fisher Scientific, Waltham, MA, USA). The titer of phage stocks was determined using DLA after appropriate dilutions.
3.9. Nucleic Acid Extraction
DNA extraction was performed from highly concentrated phage stocks (1011 PFU mL−1) using a Purelink Viral DNA/RNA mini kit (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA), according to the manufacturer’s instructions. Briefly, samples were treated with Proteinase K (50 μL at 20 mg/mL) for 1 h at 56 °C to break down viral capsid and release phage DNA. The DNA was purified using a column and washed twice with ethanol. The starting material was 0.5 mL, and the elution volume was 50 µL.
Purified DNA was visualized using 0.8% agarose gel electrophoresis. The concentration was quantified using a Qubit 1× dsDNA High Sensitivity Assay Kit (Thermo Fisher Scientific) and a Nanodrop spectrophotometer (Thermo Fisher Scientific) and was stored at −20 °C.
3.10. Sequencing
Sequencing was performed at SeqCenter Inc. (Pittsburgh, PA, USA). Illumina sequencing libraries were prepared using a tagmentation-based and PCR-based Illumina DNA Prep kit (Illumina, San Diego, CA, USA) with custom IDT 10 bp unique dual indices (UDIs) with a target insert size of 280 bp. No additional DNA fragmentation or size-selection steps were performed. Illumina sequencing was performed on an Illumina NovaSeq X Plus sequencer in one or more multiplexed shared-flow-cell runs, producing 2 × 151 bp paired-end reads.
3.11. Assembly of Genomes
Demultiplexing and adapter trimming were performed using BCL-convert v4.2.4 (Illumina). The reads were quality checked using FastQC v0.12.1 [84]. Assemblies were performed de novo using SPAdes v3.14 [85] as recommended by [86] at Phage Galaxy (Center for Phage Technology at Texas A&M University, TX, USA) [87,88]. Assemblies were evaluated using Quast v5.2.0 [89]. The first assemblies generated multiple contigs of diverse sizes with low coverage, indicating bacterial DNA contamination and the need for subsampling. Therefore, reads were randomly subsampled as recommended by Shen et al. [90] using seqtk v1.4 [91] to obtain 2% of the total reads (Table 5).

Table 5.
Random subsampling of reads used for the assemblies of phages PaCCP1 and PaCCP2.
Assemblies were repeated at the Bacterial and Viral Bioinformatics Resource Center (BV-BRC) [92] using SPAdes. The outputs were visually checked using Bandage v0.8.1 [93], error correction was performed using Pilon v1.24 [94], and Quast was used to check the assemblies. The outputs were a large contig with high coverage (>90) and several short contigs with low coverage (<2) per phage, indicating the assembly of a complete phage genome [90]. Contigs with low coverage corresponding to bacterial DNA contamination were removed manually. High-quality assemblies were further used for annotation and analysis.
3.12. Comparative Genome Analysis
The similarity of phages PaCCP1 and PaCCP2 to other described viral genomes was determined using Blastn, considering only complete viral genomes and using default settings. Similar phage sequences were downloaded in July 2024 from GenBank and used as references for further analysis. Pairwise comparisons of the nucleotide sequences were conducted using VIRIDIC [63] to determine the intergenomic similarity using the average nucleotide identity (ANI) obtained by Blastn, based in the classification guidelines described by the International Committee on Taxonomy of Viruses (ICTV) [37,95].
To classify phages and construct phylogenetic trees, Virus Classification and the Tree Building Online Resource (VICTOR) web service was used (https://ggdc.dsmz.de/victor.php, accessed on 20 October 2024) [65]. The similarities and relationships between PaCCP1, PaCCP2, and other reported prokaryotic double-stranded DNA viruses, were analyzed using VIPTree v4.0 [96].
3.13. Genome Annotation
PhaTYP 1.0 [97] and PhageAI 1.0 [98] were used for bacteriophages’ lifestyle prediction. Open reading frames (ORFs) were predicted with PHANOTATE [75] in Pharokka v1.3.2 [76] using the PHROG database (https://phrogs.lmge.uca.fr/, accessed on 20 October 2024) [77] with MMseqs2 v 15-6f452 [99]. Moreover, the annotation of the genomes of phages PaCCP1 and PaCCP2 was performed using the Phage Commander application 1.0 [100]. This tool runs nine gene identification programs such as RAST v.1.9.5 [101], GeneMarks v.4.30 [102], Prodigal v 2.6.3 [103], and Glimmer v3.02 [104]. tRNAs were predicted using ARAGORN v2.36 [105].
Genome maps of both phages PaCCP1 and PaCCP2 were obtained using Proksee v1.0 [106]. Clinker v0.0.26 [107] was used for the visual comparison between PaCCP1 or PaCCP2 with their closest relatives.
3.14. Detection of AMR and Virulence Genes
The search of genes encoding antibiotic resistance factors was performed using Resfinder 4.0 [108] and the Resistance Gene Identifier on the Comprehensive Antibiotic Resistance Database (CARD) v1.0, which is a recent tool curated using machine learning [109]. Moreover, the web tool VirulenceFinder 2.0 [110] was used for the search of genes potentially coding for virulence factors (98% ID threshold).
3.15. Detection of Anti-CRISPR-Cas Systems
To identify putative Acrs, we used PaCRISPR v1.0 [73] and AcRanker v1.0 [74], which are both tools with a cutoff by default. These programs allow for the direct prediction of Acr-encoding genes de novo with minimal knowledge a priori.
3.16. Host Range
The host range was determined using twenty clinical strains of P. aeruginosa and the three reference strains ATCC 27853, ATCC 9027, and PAO1. For this, mixtures of overnight bacterial cultures and phage suspensions (from a series of four decimal dilutions) were incubated for 20 min and then spotted (20 μL each) onto LB agar in triplicate. The appearance of plaques in the lawn after overnight incubation at 37 °C indicated the ability of the phages to multiply in the bacterial host. When too many plaques to count appeared for the fourth decimal dilution, experiments were performed repeatedly using the next two dilutions.
3.17. Growth Inhibition Assay
Mid-exponential-phase cultures of strains UC535 or UC550 were inoculated with the phages PaCCP1 or PaCCP1, respectively, at an MOI 10 in duplicate in 250 mL flasks containing 100 mL of LB broth and incubated at 37 °C without shaking. Samples of 1 mL were taken every 30 min up to 360 min. The absorbance was measured by spectrophotometry at 600 nm using an Spectrophotometer UVISCO V-1200 (Avantor, Radnor Township, PA, USA). Controls without phages were included.
4. Conclusions
We report the characterization and the complete genome analysis of two dsDNA lytic P. aeruginosa phages. PaCCP1 is a Septimatrevirus and PaCCP2 is a Pbunavirus belonging to the class Caudoviricetes. Both phages were isolated from a wastewater sample obtained from the influent of a wastewater treatment plant in Concepción, Chile.
They have potential as biocontrol agents even against MDR strains of this major human and animal opportunistic pathogen and are responsible for a wide range of diseases, from soft tissue to life-threatening infections. More studies are needed to evaluate, for instance, the use of phage cocktails in combination with antibiotics to control bacteria, the capacity of phages to eradicate biofilms, the development of phage-resistance, and the use of Anti-CRISPR proteins.
Author Contributions
Conceptualization, B.P., A.D., A.O.-C. and G.G.-R.; methodology, B.P., M.S., C.A. and L.A.; software, B.P. and V.A.; validation, B.P., A.D., A.O.-C. and G.G.-R.; formal analysis, B.P.; investigation, B.P.; resources, B.P. and G.G.-R.; data curation, B.P.; writing—original draft preparation, B.P., A.D., A.O.-C. and G.G.-R.; writing—review and editing, B.P., A.D., A.O.-C. and G.G.-R.; visualization, B.P.; supervision, G.G.-R.; project administration, B.P.; funding acquisition, B.P. All authors have read and agreed to the published version of the manuscript.
Funding
This project received funding from the grant “VRID Postdoctorado from the Vicerrectoría de Investigación y Desarrollo, Universidad de Concepción (project CO 002200000279).
Informed Consent Statement
Not applicable.
Data Availability Statement
The sequencing data for bacteriophages PaCCP1 and PaCCP2 are available in GenBank under accession numbers PQ492277 and PQ492278, respectively.
Acknowledgments
We thank Mathias Hepp from CENTINELA group of the Universidad Católica de la Santisima Concepcion for providing the raw sewage samples. We also express our gratitude to the Centro de Espectroscopía y Microscopía (CESMI) for their invaluable assistance in providing electron microscopy images.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Pillarisetti, N.; Williamson, E.; Linnane, B.; Skoric, B.; Robertson, C.F.; Robinson, P.; Massie, J.; Hall, G.L.; Sly, P.; Stick, S.; et al. Infection, inflammation, and lung function decline in infants with cystic fibrosis. Am. J. Respir. Crit. Care Med. 2011, 184, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Pang, Z.; Raudonis, R.; Glick, B.R.; Lin, T.J.; Cheng, Z. Antibiotic resistance in Pseudomonas aeruginosa: Mechanisms and alternative therapeutic strategies. Biotechnol. Adv. 2019, 37, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Bassetti, M.; Vena, A.; Croxatto, A.; Righi, E.; Guery, B. How to manage Pseudomonas aeruginosa infections. Drugs Context 2018, 7, 212527. [Google Scholar] [CrossRef] [PubMed]
- Stover, C.K.; Pham, X.Q.; Erwin, A.L.; Mizoguchi, S.D.; Warrener, P.; Hickey, M.J.; Brinkman, F.S.L.; Hufnagle, W.O.; Kowalik, D.J.; Lagrou, M.; et al. Complete genome sequence of Pseudomonas aeruginosa PAO1, an opportunistic pathogen. Nature 2000, 406, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Moradali, M.F.; Ghods, S.; Rehm, B.H. Pseudomonas aeruginosa lifestyle: A paradigm for adaptation, survival, and persistence. Front. Cell. Infect. Microbiol. 2017, 7, 39. [Google Scholar] [CrossRef]
- Jurado-Martín, I.; Sainz-Mejías, M.; McClean, S. Pseudomonas aeruginosa: An audacious pathogen with an adaptable arsenal of virulence factors. Int. J. Mol. Sci. 2021, 22, 3128. [Google Scholar] [CrossRef]
- Thi, M.T.T.; Wibowo, D.; Rehm, B.H. Pseudomonas aeruginosa biofilms. Int. J. Mol. Sci. 2020, 21, 8671. [Google Scholar] [CrossRef]
- Murray, C.J.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- WHO. WHO Bacterial Priority Pathogens List, 2024: Bacterial Pathogens of Public Health Importance to Guide Research, Development and Strategies to Prevent and Control Antimicrobial Resistance. 2024. Available online: https://www.who.int/publications/i/item/9789240093461 (accessed on 7 July 2024).
- Gordillo Altamirano, F.L.; Barr, J.J. Phage therapy in the postantibiotic era. Clin. Microbiol. Rev. 2019, 32, e00066-18. [Google Scholar] [CrossRef]
- Pires, D.P.; Costa, A.R.; Pinto, G.; Meneses, L.; Azeredo, J. Current challenges and future opportunities of phage therapy. FEMS Microbiol. Rev. 2020, 44, 684–700. [Google Scholar] [CrossRef]
- Principi, N.; Silvestri, E.; Esposito, S. Advantages and limitations of bacteriophages for the treatment of bacterial infections. Front. Pharmacol. 2019, 10, 457104. [Google Scholar] [CrossRef] [PubMed]
- Pirnay, J.P. Phage therapy in the year 2035. Front. Microbiol. 2020, 11, 1171. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.P.; Sillankorva, S.; Kropinski, A.M.; Lu, T.K.; Azeredo, J. Complete genome sequence of Pseudomonas aeruginosa phage vB_PaeM_CEB_DP1. Genome Announc. 2015, 3, e00918-15. [Google Scholar] [CrossRef] [PubMed]
- Bertozzi Silva, J.; Storms, Z.; Sauvageau, D. Host receptors for bacteriophage adsorption. FEMS Microbiol. Lett. 2016, 363, fnw002. [Google Scholar] [CrossRef] [PubMed]
- Aghaee, B.L.; Mirzaei, M.K.; Alikhani, M.Y.; Mojtahedi, A. Sewage and sewage-contaminated environments are the most prominent sources to isolate phages against Pseudomonas aeruginosa. BMC Microbiol. 2021, 21, 132. [Google Scholar] [CrossRef]
- Ballesté, E.; Blanch, A.R.; Muniesa, M.; García-Aljaro, C.; Rodríguez-Rubio, L.; Martín-Díaz, J.; Pascual-Benito, M.; Jofre, J. Bacteriophages in sewage: Abundance, roles, and applications. FEMS Microbes 2022, 3, xtac009. [Google Scholar] [CrossRef]
- Chegini, Z.; Khoshbayan, A.; Taati Moghadam, M.; Farahani, I.; Jazireian, P.; Shariati, A. Bacteriophage therapy against Pseudomonas aeruginosa biofilms: A review. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 45. [Google Scholar] [CrossRef]
- Maffei, E.; Woischnig, A.K.; Burkolter, M.R.; Heyer, Y.; Humolli, D.; Thürkauf, N.; Bock, T.; Schmidt, A.; Manfredi, P.; Egli, A.; et al. Phage Paride can kill dormant, antibiotic-tolerant cells of Pseudomonas aeruginosa by direct lytic replication. Nat. Commun. 2024, 15, 175. [Google Scholar] [CrossRef]
- Hawkins, C.; Harper, D.; Burch, D.; Änggård, E.; Soothill, J. Topical treatment of Pseudomonas aeruginosa otitis of dogs with a bacteriophage mixture: A before/after clinical trial. Vet. Microbiol. 2010, 146, 309–313. [Google Scholar] [CrossRef]
- Fukuda, K.; Ishida, W.; Uchiyama, J.; Rashel, M.; Kato, S.; Morita, T.; Muraoka, A.; Sumi, T.; Matsuzaki, S.; Daibata, M.; et al. Pseudomonas aeruginosa keratitis in mice: Effects of topical bacteriophage KPP12 administration. PLoS ONE 2012, 7, e47742. [Google Scholar] [CrossRef]
- Abedon, S.T.; Kuhl, S.J.; Blasdel, B.G.; Kutter, E.M. Phage treatment of human infections. Bacteriophage 2011, 1, 66–85. [Google Scholar] [CrossRef] [PubMed]
- Marza, J.S.; Soothill, J.S.; Boydell, P.; Collyns, T.A. Multiplication of therapeutically administered bacteriophages in Pseudomonas aeruginosa infected patients. Burns 2006, 32, 644–646. [Google Scholar] [CrossRef] [PubMed]
- Rhoads, D.D.; Wolcott, R.D.; Kuskowski, M.A.; Wolcott, B.M.; Ward, L.S.; Sulakvelidze, A. Bacteriophage therapy of venous leg ulcers in humans: Results of a phase I safety trial. J. Wound Care 2009, 18, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.; Hawkins, C.H.; Änggård, E.E.; Harper, D.R. A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa; a preliminary report of efficacy. Clin. Otolaryngol. 2009, 34, 349–357. [Google Scholar] [CrossRef]
- Waters, E.M.; Neill, D.R.; Kaman, B.; Sahota, J.S.; Clokie, M.R.; Winstanley, C.; Kadioglu, A. Phage therapy is highly effective against chronic lung infections with Pseudomonas aeruginosa. Thorax 2017, 72, 666–667. [Google Scholar] [CrossRef]
- Strathdee, S.A.; Hatfull, G.F.; Mutalik, V.K.; Schooley, R.T. Phage therapy: From biological mechanisms to future directions. Cell 2023, 186, 17–31. [Google Scholar] [CrossRef]
- Doudna, J.A.; Charpentier, E. The new frontier of genome engineering with CRISPR-Cas9. Science 2014, 346, 1258096. [Google Scholar] [CrossRef]
- Anzalone, A.V.; Koblan, L.W.; Liu, D.R. Genome editing with CRISPR–Cas nucleases, base editors, transposases and prime editors. Nat. Biotechnol. 2020, 38, 824–844. [Google Scholar] [CrossRef]
- Wang, J.Y.; Doudna, J.A. CRISPR technology: A decade of genome editing is only the beginning. Science 2023, 379, eadd8643. [Google Scholar] [CrossRef] [PubMed]
- van Belkum, A.; Soriaga, L.B.; LaFave, M.C.; Akella, S.; Veyrieras, J.B.; Barbu, E.M.; Shortridge, D.; Blanc, B.; Hannum, G.; Zambardi, G.; et al. Phylogenetic distribution of CRISPR-Cas systems in antibiotic-resistant Pseudomonas aeruginosa. mBio 2015, 6, e01796-15. [Google Scholar] [CrossRef]
- Cady, K.C.; Bondy-Denomy, J.; Heussler, G.E.; Davidson, A.R.; O’Toole, G.A. The CRISPR/Cas adaptive immune system of Pseudomonas aeruginosa mediates resistance to naturally occurring and engineered phages. J. Bacteriol. 2012, 194, 5728–5738. [Google Scholar] [CrossRef] [PubMed]
- Høyland-Kroghsbo, N.M.; Paczkowski, J.; Mukherjee, S.; Broniewski, J.; Westra, E.; Bondy-Denomy, J.; Bassler, B.L. Quorum sensing controls the Pseudomonas aeruginosa CRISPR-Cas adaptive immune system. Proc. Natl. Acad. Sci. USA 2017, 114, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Wheatley, R.M.; MacLean, R.C. CRISPR-Cas systems restrict horizontal gene transfer in Pseudomonas aeruginosa. ISME J. 2021, 15, 1420–1433. [Google Scholar] [CrossRef] [PubMed]
- Pawluk, A.; Bondy-Denomy, J.; Cheung, V.H.; Maxwell, K.L.; Davidson, A.R. A new group of phage anti-CRISPR genes inhibits the type IE CRISPR-Cas system of Pseudomonas aeruginosa. mBio 2014, 5, e00896. [Google Scholar] [CrossRef] [PubMed]
- Pawluk, A.; Davidson, A.R.; Maxwell, K.L. Anti-CRISPR: Discovery, mechanism and function. Nat. Rev. Microbiol. 2018, 16, 12–17. [Google Scholar] [CrossRef]
- Turner, D.; Shkoporov, A.N.; Lood, C.; Millard, A.D.; Dutilh, B.E.; Alfenas-Zerbini, P.; van Zyl, L.J.; Aziz, R.K.; Oksanen, H.M.; Poranen, M.M.; et al. Abolishment of morphology-based taxa and change to binomial species names: 2022 taxonomy update of the ICTV bacterial viruses subcommittee. Arch. Virol. 2023, 168, 74. [Google Scholar] [CrossRef]
- Peters, D.L.; Lynch, K.H.; Stothard, P.; Dennis, J.J. The isolation and characterization of two Stenotrophomonas maltophilia bacteriophages capable of cross-taxonomic order infectivity. BMC Genom. 2015, 16, 664. [Google Scholar] [CrossRef]
- Loh, B.; Wang, X.; Hua, X.; Luo, J.; Wen, T.; Zhang, L.; Ma, L.; Manohar, P.; Nachimuthu, R.; Grainge, I.; et al. Complete genome sequences of bacteriophages Kaya, Guyu, kopi, and TehO, which target clinical strains of Pseudomonas aeruginosa. Microbiol. Resour. Announc. 2021, 10, e01043-21. [Google Scholar] [CrossRef]
- Clark, S.; Le, T.; Moreland, R.; Liu, M.; Gonzalez, C.F.; Gill, J.J.; Ramsey, J. Complete genome sequence of Xanthomonas siphophage Samson. Microbiol. Resour. Announc. 2019, 8, e01097-19. [Google Scholar] [CrossRef]
- Domingo-Calap, M.L.; Bernabeu-Gimeno, M.; Aure, C.M.; Marco-Noales, E.; Domingo-Calap, P. Comparative Analysis of Novel Lytic Phages for Biological Control of Phytopathogenic Xanthomonas spp. Microbiol. Spectr. 2022, 10, e0296022. [Google Scholar] [CrossRef]
- Kwan, T.; Liu, J.; DuBow, M.; Gros, P.; Pelletier, J. Comparative genomic analysis of 18 Pseudomonas aeruginosa bacteriophages. J. Bacteriol. 2006, 188, 1184–1187. [Google Scholar] [CrossRef] [PubMed]
- Essoh, C.; Latino, L.; Midoux, C.; Blouin, Y.; Loukou, G.; Nguetta, S.P.A.; Lathro, S.; Cablanmian, A.; Kouassi, A.K.; Vergnaud, G.; et al. Investigation of a large collection of Pseudomonas aeruginosa bacteriophages collected from a single environmental source in Abidjan, Côte d’Ivoire. PLoS ONE 2015, 10, e0130548. [Google Scholar] [CrossRef] [PubMed]
- Campbell, R.A.; Farlow, J.; Freyberger, H.R.; He, Y.; Ward, A.M.; Ellison, D.W.; Getnet, D.; Swierczewski, B.E.; Nikolich, M.P.; Filippov, A.A. Genome Sequences of 17 Diverse Pseudomonas aeruginosa Phages. Microbiol. Resour. Announc. 2021, 10, e00031-21. [Google Scholar] [CrossRef] [PubMed]
- Karumidze, N.; Thomas, J.A.; Kvatadze, N.; Goderdzishvili, M.; Hakala, K.W.; Weintraub, S.T.; Alavidze, Z.; Hardies, S.C. Characterization of lytic Pseudomonas aeruginosa bacteriophages via biological properties and genomic sequences. Appl. Microbiol. Biotechnol. 2012, 94, 1609–1617. [Google Scholar] [CrossRef] [PubMed]
- Shahraki, A.H.; Vahed, M.; Mirsaeidi, M. Complete Genome Sequencing of the Novel Pseudomonas aeruginosa Phage UF_RH1. Microbiol. Resour. Announc. 2023, 12, e00139-23. [Google Scholar] [CrossRef]
- Holloway, B.W.; Egan, J.B.; Monk, M. Lysogeny in Pseudomonas aeruginosa. Aust. J. Exp. Biol. Med. Sci. 1960, 38, 321–330. [Google Scholar] [CrossRef]
- Neves, P.R.; Cerdeira, L.T.; Mitne Neto, M.; Oliveira, T.G.M.D.; Mcculloch, J.A.; Sampaio, J.L.M.; Mamizuka, E.M.; Levy, C.E.; Sato, M.I.Z.; Lincopan, N. Complete genome sequence of an F8 like ltic myovirus (φSPM-1) that infects metallo-β-lactamase-producing Pseudomonas aeruginosa. Genome Announc. 2014, 2, e00061-14. [Google Scholar] [CrossRef]
- Watkins, S.C.; Sible, E.; Putonti, C. Pseudomonas PB1-like phages: Whole genomes from metagenomes offer insight into an abundant group of bacteriophages. Viruses 2018, 10, 331. [Google Scholar] [CrossRef]
- Yuan, Y.; Qu, K.; Tan, D.; Li, X.; Wang, L.; Cong, C.; Xiu, Z.; Xu, Y. Isolation and characterization of a bacteriophage and its potential to disrupt multi-drug resistant Pseudomonas aeruginosa biofilms. Microb. Pathog. 2019, 128, 329–336. [Google Scholar] [CrossRef]
- Forti, F.; Roach, D.R.; Cafora, M.; Pasini, M.E.; Horner, D.S.; Fiscarelli, E.V.; Rossitto, M.; Cariani, L.; Briani, F.; Debarbieux, L.; et al. Design of a broad-range bacteriophage cocktail that reduces Pseudomonas aeruginosa biofilms and treats acute infections in two animal models. Antimicrob. Agents Chemother. 2018, 62, 10–1128. [Google Scholar] [CrossRef]
- Ceyssens, P.J.; Miroshnikov, K.; Mattheus, W.; Krylov, V.; Robben, J.; Noben, J.P.; Vanderschraeghe, S.; Sykilinda, N.; Kropinski, A.M.; Volckaert, G.; et al. Comparative analysis of the widespread and conserved PB1-like viruses infecting Pseudomonas aeruginosa. Environ. Microbiol. 2009, 11, 2874–2883. [Google Scholar] [CrossRef] [PubMed]
- Danis-Wlodarczyk, K.; Olszak, T.; Arabski, M.; Wasik, S.; Majkowska-Skrobek, G.; Augustyniak, D.; Gula, G.; Briers, Y.; Bin Jang, H.; Vandenheuvel, D.; et al. Characterization of the newly isolated lytic bacteriophages KTN6 and KT28 and their efficacy against Pseudomonas aeruginosa biofilm. PLoS ONE 2015, 10, e0127603. [Google Scholar]
- Oliveira, V.C.; Bim, F.L.; Monteiro, R.M.; Macedo, A.P.; Santos, E.S.; Silva-Lovato, C.H.; Paranhos, H.F.O.; Melo, L.D.R.; Santos, S.B.; Watanabe, E. Identification and characterization of new bacteriophages to control multidrug-resistant Pseudomonas aeruginosa biofilm on endotracheal tubes. Front. Microbiol. 2020, 11, 580779. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Chen, P.; Lin, Z.; Wang, T. Characterization of two Pseudomonas aeruginosa viruses vB_PaeM_SCUT-S1 and vB_PaeM_SCUT-S2. Viruses 2019, 11, 318. [Google Scholar] [CrossRef]
- Cingolani, G.; Iglesias, S.; Hou, C.F.; Lemire, S.; Soriaga, A.; Kyme, P. Cryo-EM analysis of Pseudomonas phage Pa193 structural components. Res. Sq. 2024. [Google Scholar] [CrossRef]
- Aleshkin, A.V.; Ershova, O.N.; Volozhantsev, N.V.; Svetoch, E.A.; Popova, A.V.; Rubalskii, E.O.; Borzilov, A.I.; Alёshkin, V.A.; Afanas’Ev, S.S.; Karaulov, A.; et al. Phagebiotics in treatment and prophylaxis of healthcare-associated infections. Bacteriophage 2016, 6, 40–46. [Google Scholar] [CrossRef]
- Latz, S.; Krüttgen, A.; Häfner, H.; Buhl, E.M.; Ritter, K.; Horz, H.P. Differential effect of newly isolated phages belonging to PB1-like, phiKZ-like and LUZ24-like viruses against multi-drug resistant Pseudomonas aeruginosa under varying growth conditions. Viruses 2017, 9, 315. [Google Scholar] [CrossRef]
- Fujiki, J.; Furusawa, T.; Munby, M.; Kawaguchi, C.; Matsuda, Y.; Shiokura, Y.; Nakamura, K.; Nakamura, T.; Sasaki, M.; Usui, M.; et al. Susceptibility of Pseudomonas aeruginosa veterinary isolates to Pbunavirus PB1-like phages. Microbiol. Immunol. 2020, 64, 778–782. [Google Scholar] [CrossRef]
- De Smet, J.; Wagemans, J.; Hendrix, H.; Staes, I.; Visnapuu, A.; Horemans, B.; Aertsen, A.; Lavigne, R. Bacteriophage-mediated interference of the c-di-GMP signalling pathway in Pseudomonas aeruginosa. Microb. Biotechnol. 2021, 14, 967–978. [Google Scholar] [CrossRef]
- Alves, D.R.; Perez-Esteban, P.; Kot, W.; Bean, J.E.; Arnot, T.; Hansen, L.H.; Enright, M.C.; Jenkins, A.T.A. A novel bacteriophage cocktail reduces and disperses P seudomonas aeruginosa biofilms under static and flow conditions. Microb. Biotechnol. 2016, 9, 61–74. [Google Scholar] [CrossRef]
- Turner, D.; Kropinski, A.M.; Adriaenssens, E.M. A roadmap for genome-based phage taxonomy. Viruses 2021, 13, 506. [Google Scholar] [CrossRef] [PubMed]
- Moraru, C.; Varsani, A.; Kropinski, A.M. VIRIDIC-A novel tool to calculate the intergenomic similarities of prokaryote-infecting viruses. Viruses 2020, 12, 1268. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, P.; Adriaenssens, E.M.; Zerbini, F.M.; Abrescia, N.G.; Aiewsakun, P.; Alfenas-Zerbini, P.; Bao, Y.; Barylski, J.; Drosten, C.; Duffy, S.; et al. Four principles to establish a universal virus taxonomy. PLoS Biol. 2023, 21, e3001922. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kolthoff, J.P.; Göker, M. VICTOR: Genome-based phylogeny and classification of prokaryotic viruses. Bioinformatics 2017, 33, 3396–3404. [Google Scholar] [CrossRef] [PubMed]
- Xuan, G.; Dou, Q.; Kong, J.; Lin, H.; Wang, J. Pseudomonas aeruginosa resists phage infection via eavesdropping on indole signaling. Microbiol. Spectr. 2023, 11, e03911-22. [Google Scholar] [CrossRef]
- Su, Q.; Lu, D.; Kong, J.; Lin, H.; Xuan, G.; Wang, J. PqsA mutation-mediated enhancement of phage-mediated combat against Pseudomonas aeruginosa. Front. Cell. Infect. Microbiol. 2024, 14, 1296777. [Google Scholar] [CrossRef]
- Li, F.; Hou, C.F.D.; Lokareddy, R.K.; Yang, R.; Forti, F.; Briani, F.; Cingolani, G. High-resolution cryo-EM structure of the Pseudomonas bacteriophage E217. Nat. Commun. 2023, 14, 4052. [Google Scholar] [CrossRef]
- Fong, S.A.; Drilling, A.; Morales, S.; Cornet, M.E.; Woodworth, B.A.; Fokkens, W.J.; Psaltis, A.J.; Vreugde, S.; Wormald, P.-J. Activity of bacteriophages in removing biofilms of Pseudomonas aeruginosa isolates from chronic rhinosinusitis patients. Front. Cell. Infect. Microbiol. 2017, 7, 418. [Google Scholar] [CrossRef]
- Wannasrichan, W.; Htoo, H.H.; Suwansaeng, R.; Pogliano, J.; Nonejuie, P.; Chaikeeratisak, V. Phage-resistant Pseudomonas aeruginosa against a novel lytic phage JJ01 exhibits hypersensitivity to colistin and reduces biofilm production. Front. Microbiol. 2022, 13, 1004733. [Google Scholar] [CrossRef]
- Mangalea, M.R.; Duerkop, B.A. Fitness trade-offs resulting from bacteriophage resistance potentiate synergistic antibacterial strategies. Infect. Immun. 2020, 88, e00926-19. [Google Scholar] [CrossRef]
- Magiorakos, A.P.; Srinivasan, A.; Carey, R.B.; Carmeli, Y.; Falagas, M.E.; Giske, C.G.; Harbarth, S.; Hindler, J.F.; Kahlmeter, G.; Olsson-Liljequist, B.; et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012, 18, 268–281. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Dai, W.; Li, J.; Xie, R.; Dunstan, R.A.; Stubenrauch, C.; Zhang, Y.; Lithgow, T. PaCRISPR: A server for predicting and visualizing anti-CRISPR proteins. Nucleic Acids Res. 2020, 48, W348–W357. [Google Scholar] [CrossRef] [PubMed]
- Eitzinger, S.; Asif, A.; Watters, K.E.; Iavarone, A.T.; Knott, G.J.; Doudna, J.A.; Minhas, F.U.A.A. Machine learning predicts new anti-CRISPR proteins. Nucleic Acids Res. 2020, 48, 4698–4708. [Google Scholar] [CrossRef]
- McNair, K.; Zhou, C.; Dinsdale, E.A.; Souza, B.; Edwards, R.A. PHANOTATE: A novel approach to gene identification in phage genomes. Bioinformatics 2019, 35, 4537–4542. [Google Scholar] [CrossRef] [PubMed]
- Bouras, G.; Nepal, R.; Houtak, G.; Psaltis, A.J.; Wormald, P.J.; Vreugde, S. Pharokka: A fast scalable bacteriophage annotation tool. Bioinformatics 2023, 39, btac776. [Google Scholar] [CrossRef] [PubMed]
- Terzian, P.; Olo Ndela, E.; Galiez, C.; Lossouarn, J.; Pérez Bucio, R.E.; Mom, R.; Toussaint, A.; Petit, M.-A.; Enault, F. PHROG: Families of prokaryotic virus proteins clustered using remote homology. NAR Genom. Bioinform. 2021, 3, lqab067. [Google Scholar] [CrossRef]
- Marino, N.D.; Pinilla-Redondo, R.; Csörgő, B.; Bondy-Denomy, J. Anti-CRISPR protein applications: Natural brakes for CRISPR-Cas technologies. Nat. Methods 2020, 17, 471–479. [Google Scholar] [CrossRef]
- Bondy-Denomy, J.; Pawluk, A.; Maxwell, K.L.; Davidson, A.R. Bacteriophage genes that inactivate the CRISPR/Cas bacterial immune system. Nature 2013, 493, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Yang, L.; Gao, T.; Dong, C.; Zhang, B.; Yin, P.; Feng, Y. A type IF anti-CRISPR protein inhibits the CRISPR-Cas surveillance complex by ADP-ribosylation. Mol. Cell 2020, 80, 512–524. [Google Scholar] [CrossRef]
- Pires, J.; Novais, A.; Peixe, L. Blue-carba, an easy biochemical test for detection of diverse carbapenemase producers directly from bacterial cultures. J. Clin. Microbiol. 2013, 51, 4281–4283. [Google Scholar] [CrossRef]
- Reller, L.B.; Weinstein, M.; Jorgensen, J.H.; Ferraro, M.J. Antimicrobial susceptibility testing: A review of general principles and contemporary practices. Clin. Infect. Dis. 2009, 49, 1749–1755. [Google Scholar]
- Kropinski, A.M.; Mazzocco, A.; Waddell, T.E.; Lingohr, E.; Johnson, R.P. Enumeration of bacteriophages by double agar overlay plaque assay. In Bacteriophages: Methods and protocols, Volume 1: Isolation, Characterization, and interactions; Humana: Totowa, NJ, USA, 2009; pp. 69–76. [Google Scholar]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 20 April 2024).
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.; Adriaenssens, E.M.; Tolstoy, I.; Kropinski, A.M. Phage annotation guide: Guidelines for assembly and high-quality annotation. Phage 2021, 2, 170–182. [Google Scholar] [CrossRef] [PubMed]
- Afgan, E.; Baker, D.; Batut, B.; Van Den Beek, M.; Bouvier, D.; Čech, M.; Chilton, J.; Clements, D.; Coraor, N.; Grüning, B.A.; et al. The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2018 update. Nucleic Acids Res. 2018, 46, W537–W544. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, J.; Rasche, H.; Maughmer, C.; Criscione, A.; Mijalis, E.; Liu, M.; Hu, J.C.; Young, R.; Gill, J.J. Galaxy and Apollo as a biologist-friendly interface for high-quality cooperative phage genome annotation. PLoS Comput. Biol. 2020, 16, e1008214. [Google Scholar] [CrossRef]
- Gurevich, A.; Saveliev, V.; Vyahhi, N.; Tesler, G. QUAST: Quality assessment tool for genome assemblies. Bioinformatics 2013, 29, 1072–1075. [Google Scholar] [CrossRef]
- Shen, A.; Millard, A. Phage genome annotation: Where to begin and end. Phage 2021, 2, 183–193. [Google Scholar] [CrossRef]
- Shen, W.; Le, S.; Li, Y.; Hu, F. SeqKit: A cross-platform and ultrafast toolkit for FASTA/Q file manipulation. PLoS ONE 2016, 11, e0163962. [Google Scholar] [CrossRef]
- Olson, R.D.; Assaf, R.; Brettin, T.; Conrad, N.; Cucinell, C.; Davis, J.J.; Dempsey, D.M.; Dickerman, A.; Dietrich, E.M.; Kenyon, R.W.; et al. Introducing the bacterial and viral bioinformatics resource center (BV-BRC): A resource combining PATRIC, IRD and ViPR. Nucleic Acids Res. 2023, 51, D678–D689. [Google Scholar] [CrossRef]
- Wick, R.R.; Schultz, M.B.; Zobel, J.; Holt, K.E. Bandage: Interactive visualization of de novo genome assemblies. Bioinformatics 2015, 31, 3350–3352. [Google Scholar] [CrossRef]
- Walker, B.J.; Abeel, T.; Shea, T.; Priest, M.; Abouelliel, A.; Sakthikumar, S.; Cuomo, C.A.; Zeng, Q.; Wortman, J.; Young, S.K.; et al. Pilon: An integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS ONE. 2014, 9, e112963. [Google Scholar] [CrossRef] [PubMed]
- Adriaenssens, E.M.; Krupovic, M.; Knezevic, P.; Ackermann, H.W.; Barylski, J.; Brister, J.R.; Clokie, M.R.C.; Duffy, S.; Dutilh, B.E.; Edwards, R.A.; et al. Taxonomy of prokaryotic viruses: 2016 update from the ICTV bacterial and archaeal viruses subcommittee. Arch. Virol. 2017, 162, 1153–1157. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, Y.; Yoshida, T.; Kuronishi, M.; Uehara, H.; Ogata, H.; Goto, S. ViPTree: The viral proteomic tree server. Bioinformatics 2017, 33, 2379–2380. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.; Tang, X.; Sun, Y. PhaTYP: Predicting the lifestyle for bacteriophages using BERT. Brief. Bioinform. 2023, 24, bbac487. [Google Scholar] [CrossRef] [PubMed]
- Tynecki, P.; Guziński, A.; Kazimierczak, J.; Jadczuk, M.; Dastych, J.; Onisko, A. PhageAI-bacteriophage life cycle recognition with machine learning and natural language processing. bioRxiv 2020. [Google Scholar] [CrossRef]
- Steinegger, M.; Söding, J. MMseqs2 enables sensitive protein sequence searching for the analysis of massive data sets. Nat. Biotechnol. 2017, 35, 1026–1028. [Google Scholar] [CrossRef]
- Lazeroff, M.; Ryder, G.; Harris, S.L.; Tsourkas, P.K. Phage commander, an application for rapid gene identification in bacteriophage genomes using multiple programs. Phage 2021, 2, 204–213. [Google Scholar] [CrossRef]
- Aziz, R.K.; Bartels, D.; Best, A.A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The RAST server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef]
- Besemer, J.; Lomsadze, A.; Borodovsky, M. GeneMarkS: A self-training method for prediction of gene starts in microbial genomes. Implications for finding sequence motifs in regulatory regions. Nucleic Acids Res. 2001, 29, 2607–2618. [Google Scholar] [CrossRef]
- Hyatt, D.; Chen, G.W.; LoCascio, P.F.; Land, M.L.; Larimer, F.W.; Hauser, L.J. Prodigal: Prokaryotic gene recognition and translation initiation site identification. BMC Bioinform. 2010, 11, 119. [Google Scholar] [CrossRef]
- Delcher, A.L.; Harmon, D.; Kasif, S.; White, O.; Salzberg, S.L. Improved microbial gene identification with GLIMMER. Nucleic Acid Res. 1999, 27, 4636–4641. [Google Scholar] [CrossRef] [PubMed]
- Laslett, D.; Canback, B. ARAGORN, a program to detect tRNA genes and tmRNA genes in nucleotide sequences. Nucleic Acids Res. 2004, 32, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Grant, J.R.; Enns, E.; Marinier, E.; Mandal, A.; Herman, E.K.; Chen, C.Y.; Graham, M.; Van Domselaar, G.; Stothard, P. Proksee: In-depth characterization and visualization of bacterial genomes. Nucleic Acids Res. 2023, 51, W484–W492. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, C.L.; Chooi, Y.H. Clinker & clustermap. js: Automatic generation of gene cluster comparison figures. Bioinformatics 2021, 37, 2473–2475. [Google Scholar]
- Bortolaia, V.; Kaas, R.S.; Ruppe, E.; Roberts, M.C.; Schwarz, S.; Cattoir, V.; Philippon, A.; Allesoe, R.L.; Rebelo, A.R.; Florensa, A.F.; et al. ResFinder 4.0 for predictions of phenotypes from genotypes. J. Antimicrob. Chemother. 2020, 75, 3491–3500. [Google Scholar] [CrossRef] [PubMed]
- Alcock, B.P.; Huynh, W.; Chalil, R.; Smith, K.W.; Raphenya, A.R.; Wlodarski, M.A.; Edalatmand, A.; Petkau, A.; Syed, S.A.; Tsang, K.K.; et al. CARD 2023: Expanded curation, support for machine learning, and resistome prediction at the Comprehensive Antibiotic Resistance Database. Nucleic Acids Res. 2023, 51, D690–D699. [Google Scholar] [CrossRef]
- Joensen, K.G.; Scheutz, F.; Lund, O.; Hasman, H.; Kaas, R.S.; Nielsen, E.M.; Aarestrup, F.M. Real-time whole-genome sequencing for routine typing, surveillance, and outbreak detection of verotoxigenic Escherichia coli. J. Clin. Microbiol. 2014, 52, 1501–1510. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).