1. Introduction
Stress contributes to the development and maintenance of alcohol-seeking behaviors in both human (
Brady & Sonne, 1999;
K. Keyes et al., 2012) and animal (
Domi et al., 2021) models. Women experience higher rates of anxiety (
Jalnapurkar et al., 2018) and depression (
Albert, 2015) and are more likely to drink in response to acute stress (
Guinle & Sinha, 2020;
Patock-Peckham et al., 2022;
Peltier et al., 2019), contributing to faster progression to alcohol use disorder (AUD) (
K. M. Keyes et al., 2010). Depressive symptoms are more strongly linked to alcohol use in women (
Jeong et al., 2019;
Karpyak et al., 2019;
Moscato et al., 1997;
Zhan et al., 2012), whereas findings for anxiety are mixed: some studies report stronger anxiety–alcohol associations in women (
Johannessen et al., 2017;
Kendler et al., 2015), while others find stronger effects in men (
Atkinson & Finn, 2019;
Schry et al., 2014;
Torres et al., 2023;
Zale et al., 2019). Overall, evidence supports sex-specific links between stress and alcohol use, but underlying mechanisms remain unclear.
Subjective responses, i.e., individual differences in positive (e.g., stimulation and euphoria) and negative (e.g., sedation and dysphoria) alcohol effects (
A. King et al., 2021), are one candidate mechanism, as they predict AUD risk (
Morean & Corbin, 2010;
Quinn & Fromme, 2011). A key positive subjective response, stimulation, captures increases in state energy or excitement after consuming alcohol, which predicts greater alcohol craving, intake, and AUD risk (
Bujarski & Ray, 2014;
Fischer et al., 2023;
Green et al., 2019;
A. King et al., 2021;
Ray et al., 2010). A key negative subjective response, anxiety, captures reductions in state anxiety after consuming alcohol (
Wilson, 1982). Alcohol produces anxiolytic effects (i.e., reduces anxiety, tension, or nervousness) (
Bradford et al., 2017;
Hendler et al., 2011) that are stronger in those with high trait anxiety (
Stewart & Pihl, 1994), which predict craving (
Cheng et al., 2022;
McCaul et al., 2017) and AUD risk (
Schmidt et al., 2007;
Smith & Randall, 2012). Many anxious individuals drink to relieve anxiety (
Book & Randall, 2002;
McCaul et al., 2017), increasing risk for problematic use (
Anker & Kushner, 2019).
The resting-state network connectivity of the amygdala and hippocampus, regions central to emotion and stress regulation, is another candidate mechanism. Resting-state functional connectivity refers to the temporal correlation of brain activity between brain regions measured while a participant is not engaged in an explicit task. Altered amygdala connectivity and reduced hippocampal connectivity are implicated in anxiety and depression (
Gold et al., 2017;
Jacob et al., 2022;
Langhammer et al., 2025;
Tang et al., 2018). The connectivity of these regions is also linked to alcohol use: stronger amygdala–orbitofrontal connectivity predicts alcohol use (
Crane et al., 2018;
Peters et al., 2017), reduced amygdala connectivity with the dorsal anterior cingulate cortex and caudate nucleus relates to alcohol misuse and dependence (
Hu et al., 2018;
Sun et al., 2024), and reduced hippocampal–frontal connectivity relates to craving and problematic use (
Arienzo et al., 2020).
Importantly, both subjective responses and resting-state connectivity show sex effects, positioning them as strong candidates for explaining sex differences in stress-induced alcohol-seeking. Men often report stronger stimulation effects than women, which predict drinking escalation (
A. C. King et al., 2011;
Mumenthaler et al., 1999), though findings are mixed (see
Quinn & Fromme, 2011). Animal studies suggest that females show greater alcohol-related anxiety reduction than males (
D. B. Matthews & Kerr, 2024); this is untested in humans. In neural circuits, women show greater amygdala reactivity and altered amygdala–prefrontal connectivity in depression (
Almeida et al., 2011;
Lebron-Milad et al., 2012), whereas men show stronger associations between alcohol misuse and reduced amygdala–dorsal anterior cingulate connectivity (
Claus et al., 2011;
Hu et al., 2018).
The overarching goal of this study is to examine how sex may moderate the relationship between depression and anxiety symptoms and stress-induced alcohol-seeking. It is our secondary goal to test subjective responses and resting-state connectivity as mechanisms explaining these differences. The present study integrates behavioral, subjective, and neural measures in a controlled experimental framework. Self-report of alcohol use can be biased by sex differences in social desirability and disclosure willingness (
Boniface et al., 2014;
Davis et al., 2010). Using data from a recently published intravenous (IV) alcohol self-administration trial (
Garrison et al., 2025), this study employs objective measures of alcohol-seeking. IV administration eliminates expectancy effects and standardizes alcohol exposure (
Cyders et al., 2020), reducing pharmacokinetic sex differences and allowing for precise assessment of alcohol-seeking and subjective response. Resting-state functional connectivity provides insight into the stable neural network architecture underlying mood symptoms.
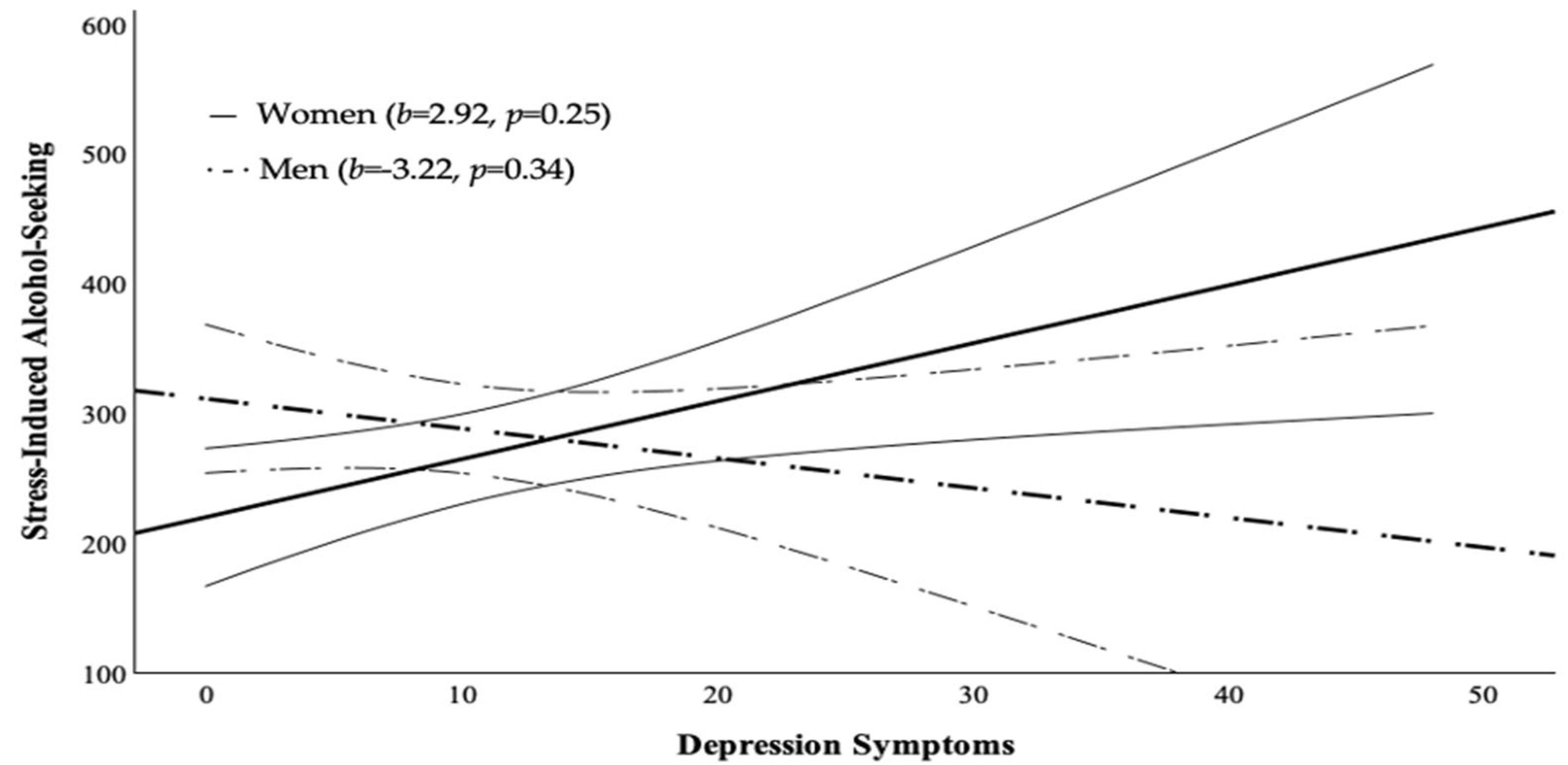
First, we tested whether sex moderates the relationship between anxiety and depression symptoms and stress-induced alcohol-seeking. We hypothesized that women would show stronger positive associations between anxiety and depression symptoms and alcohol-seeking compared to men.
Second, we examined state stimulation and state anxiety subjective responses to alcohol as mediators of these sex-specific relationships. We hypothesized that changes in subjective responses would mediate the relationship between anxiety and depression symptoms and stress-induced alcohol-seeking. We also hypothesized that sex would moderate both the symptom-to-state subjective response and symptom-to-alcohol-seeking paths, such that higher symptoms would predict greater state stimulation and lower state anxiety and that the relationships would be stronger in women compared to men.
Finally, we investigated sex differences in resting-state network connectivity of the amygdala and hippocampus with anxiety and depression symptoms and stress-induced alcohol-seeking. We hypothesized that sex would moderate the relationship between anxiety and depression symptoms and amygdala/hippocampal connectivity with cortical resting-state brain networks. We also hypothesized that sex would moderate relationships between amygdala/hippocampal connectivity and stress-induced alcohol-seeking.
4. Conclusions
This study provides new mechanistic insight into how stress influences alcohol-seeking behavior in men and women. Using an objective model of stress-induced alcohol-seeking paired with subjective response and resting-state connectivity measures, we demonstrate sex-specific pathways linking anxiety, alcohol response, and neural circuitry to drinking behavior. Unlike prior work relying primarily on retrospective self-report, this study captured real-time alcohol-seeking in response to stress and revealed distinct pathways through which anxiety and depression symptoms may influence alcohol use, including both alcohol subjective responses and cortico-limbic networks. These findings highlight the importance of examining multiple levels of analysis—behavioral, subjective, and neural—to better characterize risk processes that differ for women and men.
As hypothesized, we found stronger associations between anxiety symptoms and stress-induced alcohol-seeking among women, which aligns with previous work showing that women are more likely to drink alcohol in response to anxiety (
Johannessen et al., 2017;
Kendler et al., 2015;
McCaul et al., 2019) but conflicts with previous work linking anxiety to alcohol problems and consumption in men (
Atkinson & Finn, 2019;
Ebbert et al., 2018;
Schry et al., 2014;
Torres et al., 2023;
Zale et al., 2019). One important explanation for these discrepant findings is differences in how anxiety has been operationalized across studies. The present study assessed generalized anxiety symptoms, whereas several prior studies, most notably
Ebbert et al. (
2018), examined anxiety sensitivity, a related but distinct construct reflecting concern about the social or physical consequences of anxiety-related sensations. These constructs have been shown to exhibit different associations with alcohol-related outcomes, including sex-specific patterns. Methodological variation in alcohol-use assessment and stress context may further contribute to discrepancies in the literature. Many prior studies have relied on self-reported alcohol use over extended periods, which can reflect averaged behavior and may be subject to recall bias, whereas the current study employed an objective measure of alcohol-seeking that directly assessed behavior in response to specific, aversive circumstances. Moreover, the present study examined alcohol-seeking following acute stress, which may differ from drinking associated with chronic stress exposure. Consistent with this distinction, prior research suggests that men may be more physiologically affected by chronic stress (
K. A. Matthews et al., 2001), whereas women demonstrate greater behavioral (
Patock-Peckham et al., 2022) and neural responses to acute stress (
Grodin et al., 2024). Overall, these findings suggest that generalized anxiety symptoms may represent particularly relevant prevention and intervention targets for reducing alcohol misuse in women, but not necessarily in men.
Contrary to our hypotheses, depression symptoms related to stress-induced alcohol-seeking similarly across men and women. This was unexpected, given prior research indicating that depression symptoms are more strongly associated with alcohol use among women than men (
Jeong et al., 2019;
Karpyak et al., 2019;
Moscato et al., 1997;
Zhan et al., 2012). However, the directions of the observed relationships align with previous research and hypotheses: a positive relationship was found in women, while no relationship was observed in men. This pattern may indicate small sex differences that could emerge in a larger, statistically better-powered study.
Contrary to our hypotheses and previous findings (
D. B. Matthews & Kerr, 2024;
Stewart & Pihl, 1994), neither anxiety symptoms nor sex was related to state anxiety change across the alcohol priming interval. Such inconsistency may reflect differences in how anxiety was measured, such as anxiety sensitivity, state anxiety, trait anxiety, and anxiety disorder symptoms. In the current work, baseline state anxiety and generalized anxiety symptoms were both significantly related to stress-induced alcohol-seeking, but in opposite directions: higher anxiety symptoms were associated with more stress-induced alcohol-seeking, whereas higher state anxiety before alcohol administration was associated with less stress-induced alcohol-seeking. These diverging relationships underscore the importance of measuring both anxiety symptoms (e.g., over days, weeks, or months) and idiosyncratic, momentary experiences of anxiety, as their effects may differ and capture distinct processes to help explain mixed findings in the literature (
Atkinson & Finn, 2019;
Johannessen et al., 2017;
Kendler et al., 2015;
Schry et al., 2014;
Torres et al., 2023;
Zale et al., 2019). Anxiety symptoms may reflect broader difficulties regulating state anxiety that increase risk for drinking, whereas acute state anxiety in the laboratory may temporarily suppress alcohol-seeking behavior. Although the novelty of the IV alcohol procedure could contribute to this effect, counterbalancing reduces this concern. While increasing momentary anxiety is not a viable intervention target, state anxiety may nonetheless help identify individuals who respond differently to stress in drinking contexts.
The state stimulation findings suggest a possible mechanism for sex differences: anxiety symptoms related to higher state stimulation increase among men, which in turn is related to less stress-induced alcohol-seeking. This pattern is consistent with theories linking low alcohol sensitivity to greater AUD risk (
Kalu et al., 2012;
Schuckit, 1994;
Schuckit et al., 1997), though evidence is mixed (
A. C. King et al., 2014,
2016,
2019;
A. King et al., 2021). Our findings extend this work by suggesting that blunted stimulation may also contribute to stress-induced alcohol use. Mediation effects for depression symptoms were small and not significant, suggesting that state anxiety and state stimulation are not meaningful mediators for the relationship between depression symptoms and stress-induced alcohol-seeking. These results highlight the need to further explore alternative mechanisms of the relationship between depression and stress-induced alcohol use, including its moderation by sex.
We identified several sex-dependent resting-state connectivity patterns involving the amygdala and hippocampal networks. For generalized anxiety symptoms, sex differences emerged in connectivity between the left amygdala and the limbic and visual networks, consistent with prior evidence of limbic dysregulation and sex-dependent amygdala lateralization (
He et al., 2016;
Kilpatrick et al., 2006;
Ocklenburg et al., 2022). For depression, sex-dependent effects involved the right hippocampus and the visual network, aligning with work implicating hippocampal circuitry (
Posener et al., 2003;
Sheline, 2011) and visual networks (
Ocklenburg et al., 2022) in depression. For stress-induced alcohol-seeking, sex-dependent associations appeared in networks involving dorsal attention, default mode, and somatomotor systems, networks previously implicated in alcohol cue reactivity, AUD-related disconnectivity, and structural abnormalities (
Chumin et al., 2019;
Radoman et al., 2024;
Rice et al., 2024;
Seo et al., 2011;
Song et al., 2021).
Although this study was not designed as a direct test of competing theoretical models, our findings speak to ongoing debates between subjective response frameworks, including Schuckit’s low level of response to alcohol theory (
Kalu et al., 2012;
Schuckit, 1994;
Schuckit et al., 1997). The association between blunted stimulation and greater stress-induced alcohol-seeking may align with low-response accounts, suggesting that reduced positive alcohol effects increase risk for heavier use. In contrast, a lack of findings with anxiety suggests that stimulation-based pathways may play a more prominent role in this acute stress context. Rather than positioning these perspectives as mutually exclusive, our results suggest they may reflect complementary mechanisms that operate under different conditions or in different individuals. Future work explicitly comparing these models, particularly using controlled alcohol administration paradigms, will clarify when stimulation, changes in anxiety, or other subjective responses most strongly confer risk.
The current work has several implications. First, the relationships found here highlight the importance of studying sex-specific mechanisms in AUD research (
Guinle & Sinha, 2020;
Patock-Peckham et al., 2022;
Peltier et al., 2019), using objective behavioral assessments, including alcohol administration. Further research is needed to determine whether stimulation effects represent a safe and effective mechanism to reduce alcohol-related risk in men. Women may benefit more from targeting generalized anxiety symptoms directly to reduce stress-induced alcohol-seeking. Second, anxiolytic and stimulation effects are not likely to be mechanisms linking depression symptoms with stress-induced alcohol-seeking. Third, the resting-state findings implicate the amygdala and hippocampus in sex-dependent alcohol behavior, and they could serve as promising targets for future neuroimaging studies aimed at identifying biomarkers of risk, resilience, and potentially circuit-level interventions with techniques such as repetitive transcranial magnetic stimulation. Finally, the divergence between anxiety at the symptom-level and momentary state anxiety response underscores the importance of multi-level measurement approaches to better capture dynamic risk processes.
These findings should be considered in light of several limitations. First, the modest sample size may have limited power. Second, because the study recruited healthy, heavy-drinking adults for safety during alcohol infusion and imaging, the results may not generalize to lighter drinkers or individuals across the full AUD spectrum, despite evidence that coping-related drinking and its sex differences occur broadly (
Corbin et al., 2013;
Foster et al., 2014;
Gilson et al., 2017;
Holahan et al., 2001;
Park & Levenson, 2002;
Rodriguez et al., 2020). Similarly, we intentionally balanced the sex ratio to maximize power for detecting sex effects. Therefore, our sample does not reflect population AUD prevalence, although the gender gap is narrowing (
Agabio et al., 2017;
K. M. Keyes et al., 2011;
Verplaetse et al., 2025). Third, these findings may not extend to descending-limb alcohol effects or other alcohol-seeking contexts. Fourth, while IV alcohol administration reduces self-report bias and tightly controls exposure (
Cyders et al., 2020), alcohol is typically consumed orally and in the evening, and IV administration may have influenced baseline state anxiety; complementary oral-administration studies during evening hours are needed. Fifth, drinking history was not included as a covariate (due to limited sample size, the lack of significant sex differences in drinking history in this sample, and IV alcohol’s ability to ensure equal alcohol exposure within and across participants) and should be examined in future work. Finally, limited power precluded integrating resting-state connectivity into moderated mediation models; these findings should guide larger mechanistic studies.
These findings highlight the value of integrating sex-specific approaches into AUD research, particularly using objective alcohol administration paradigms. Subjective alcohol responses and amygdala and hippocampal connectivity emerged as promising pathways that differ by sex. Future work should determine the clinical utility of these mechanisms for developing targeted, sex-informed interventions to reduce alcohol-related risk. Women may benefit from interventions targeting generalized anxiety symptoms to reduce stress-induced drinking, especially at higher levels of anxiety symptoms, whereas stimulation responses may inform risk processes in men. The amygdala and hippocampus may be potential neural targets for future mechanistic and interventional research. The divergence between generalized anxiety symptoms and state anxiety underscores the need for multi-level measurement to capture dynamic risk processes.