Abstract
Background/Objectives: Neuroblastoma (NB) is a childhood cancer with heterogeneous characteristics, posing challenges to effective treatment. NBs express somatostatin receptors that facilitate the use of somatostatin analogs (SSTAs) as tumor-seeking agents for diagnosis and therapy. High-risk (HR) NBs often have gain-of-function mutations in the receptor tyrosine kinase anaplastic lymphoma kinase (ALK). Despite intensive multimodal treatment, survival rates remain below 40% for children with HR-NB. The aim of this work was to investigate the combined effect of the SSTA 177Lu-octreotide with the ALK inhibitor lorlatinib. Methods: Mice bearing human HR-NB CLB-BAR tumors were treated with lorlatinib, 177Lu-octreotide, and a combination of these pharmaceuticals or saline (control). Tumor volume was monitored and tumor samples were evaluated for cleaved caspase-3 and expression of 84 human genes involved in apoptosis. Results: Combination treatment with 177Lu-octreotide and lorlatinib demonstrated synergistic antitumor effects. An increased number of cleaved caspase 3-positive cells was observed in tumors from mice treated with 177Lu-octreotide alone and in combination with lorlatinib. Modulation of Bcl-2 family gene expression was observed only in the presence of both 177Lu-octreotide and lorlatinib, with BID down-regulated and HRK up-regulated on days 2 and 7, respectively. Conclusions: The data suggest that ALK signaling pathway inhibition may contribute to radiosensitization in radionuclide therapy with 177Lu-octreotide and could improve treatment outcomes in patients with HR-NB.
1. Introduction
Anaplastic lymphoma kinase (ALK) is a receptor tyrosine kinase (RTK), a member of the insulin RTK family, which is expressed in the developing central and peripheral nervous system [1]. ALK dimerization, in response to ALK in vivo ligand (ALKAL) binding, activates signaling pathways involved in various cellular processes, such as proliferation, transcription, growth, and survival [1]. Mutated, rearranged, or amplified ALK in a wide range of tumors acts as an oncogene. ALK overexpression has been identified in several tumor types, including ovarian cancer, breast cancer, and neuroblastoma (NB). Among newly diagnosed high-risk (HR) NB patients, ALK mutations or gene amplification occur in approximately 14% [2,3,4]. Excess of the transcription factor MYCN is associated with several tumor types, but especially NBs, and previous studies have illustrated the cooperativity of ALK and MYCN, the major oncoprotein of HR-NB [5,6,7]. Despite intensive multimodal treatment, and significant improvements, survival rates remain less than 40% for children with HR-NB [8].
Lorlatinib is a potent ALK inhibitor (ALKi) that binds to the ATP binding site of the ALK kinase domain, preventing ALK downstream signaling [9]. Lorlatinib was FDA approved in 2018 for treatment of ALK-positive metastatic non-small-cell lung cancer and has demonstrated robust antitumor effects in comparison with other ALKis [10,11,12]. Classified as a third-generation ALKi, lorlatinib demonstrates activity against several drug-resistant ALK mutations [11]. Phase I trials have been reported in patients with ALK-positive NB, both as monotherapy and in combination, with manageable toxicity profiles and antitumor effects [13,14]. Despite this, studies suggest that treatment with lorlatinib can still lead to drug resistance [15,16,17,18], strengthening the need to understand the underlying mechanisms of drug resistance and highlighting the need to identify combination therapy options.
177Lu-[DOTA°,Tyr3]octreotide (177Lu-octreotide) is a radiopharmaceutical that binds to somatostatin receptors (SSTRs) [19,20,21]. Octreotide is a synthetic somatostatin analog (SSTA) with high affinity for SSTR2 [22]. 177Lu is a beta-emitting radionuclide with a half-life of 6.7 days. The range of the emitted electrons (Eβmax = 497 keV) is <2 mm in tissue, which is well suited for disseminated tumors [23,24,25]. The closely related radiopharmaceutical, 177Lu-octreotate, is FDA and EMA approved for treatment of gastroenteropancreatic neuroendocrine tumors. Previous preclinical and clinical studies have demonstrated selective uptake and therapeutic potential from the use of radiolabeled SSTAs for SSTR-positive HR-NB; however, the modest therapy effect in some of the studies highlights the need for optimization of these treatments [26,27,28,29]. An ongoing phase II study with 177Lu-octreotate is now being conducted where an individualized treatment regimen is reviewed for children with relapsed HR-NB (neuroblastoma-LuDO-N) [30].
New treatment strategies are required to increase the cure rate in HR-NB. One proposed option for optimization of radiolabeled SSTA therapy could be to administer it in combination with another drug, in order to increase the effect on tumor tissue [31]. This increased effect could be mediated through specific drug–drug interactions or by independent, additive effects. With the treatment-resistance properties of HR-NB, combination treatments with radionuclide therapy and ALKi could be beneficial. The aim of this work was to examine the effects of combination treatment with lorlatinib and 177Lu-octreotide on growth and expression of apoptosis-related genes in the tumors of NB-bearing mice.
2. Materials and Methods
2.1. Tumor Cell Line and Animal Model
The study was performed with CLB-BAR (ALK gain of function, exon4–11 truncated ALK; MYCN amplification) NB cells obtained from The Center Leon Berard, France under MTA [32]. The CLB-BAR cell line was established from a stage 4 NB tumor at relapse for a female child, after the consent of her parents. Approval of care and use of these samples was given by the Biological Resources Center Ethical and Steering committees of Centre Léon Bérard and Institut Gustave Roussy.
CLB-BAR cells were cultured as previously described [26]. A mixture of Matrigel (354248, Corning, New York, NY, USA) and 1.5 × 106 CLB-BAR cells were injected s.c. into the flank of 5–6-week-old female BALB/c nude mice (Janvier Labs, Saint-Berthevin, France), after acclimatization for 1 week after delivery to the Experimental Biomedicine Lab at University of Gothenburg. All animal experiments were approved by the Swedish Ethical Committee on Animal Experiments in Gothenburg (ethical reference number 2779-20) and carried out following guidelines from Animal Research: Reporting of In Vivo Experiments (ARRIVE).
2.2. Pharmaceuticals
Lorlatinib (S7536, Selleckchem, Houston, TX, USA) was formulated in 2% DMSO, 30% PEG300, and double-distilled water. The solution was prepared for oral gavage with 10 mg/kg for each mouse.
177Lu-octreotide was prepared according to the manufacturer’s requirements (ITG Isotope Technologies Garching GmbH, Munich, Germany). Instant thin layer chromatography (ITLC-SG, chromatography paper 50/PK, Varian Medical Systems, Palo Alto, CA, USA), with 0.1 M of sodium citrate as the mobile phase, was implemented to determine the radiochemical purity, which was above 97%. The specific activity of 177Lu-octreotide was 66 MBq/µg, yielding approximately 0.47 µg peptide for 30 MBq 177Lu-octreotide. 177Lu activity in each syringe was measured with an ionization chamber with a 177Lu energy window setting (CRC-15R, Capintec, Inc., Florham Park, NJ, USA) before and after injection, and by subtracting the remaining activity after injection from the activity before injection, the actual administered activity to each mouse was determined.
2.3. Treatment Regimens
Tumor-bearing mice were divided into four groups (n = 10 mice/group) aiming for as similar a tumor volume distribution as possible between groups. Mean tumor volume was 480 mm3 (SEM = 30 mm3) at treatment start. Mice in the groups were treated with either (1) lorlatinib via daily oral gavage (10 mg/kg body weight) (from day 0 to day 14), (2) with a single i.v. injection with 30 MBq 177Lu-octreotide on day 1, or (3) a combination of both treatments. The fourth group acted as a control and received an i.v. injection with saline on day 1.
Mouse weight and tumor volume was measured four times per week. Tumor volume, V, was calculated based on measurements using digital calipers of the three perpendicular axes (a, b, and c) of the tumor: .
Three mice from each group were sacrificed on day 2 and three on day 7, and the remaining four mice in each group on day 14. At sacrifice, the animals were under anesthesia with pentobarbitalnatrium (vet. 60 mg/mL, Apotek Produktion & Laboratorier AB, Kungens kurva, Sweden) injected i.p. before cardiac puncture. Tumor samples from each time point (days 2, 7, and 14) were collected and divided into two parts, one placed in vials with formalin for immunohistochemical (IHC) analysis and the other freshly frozen in liquid nitrogen and stored at −80 °C for real-time reverse transcription polymerase chain reaction (qPCR) analysis.
2.4. Immunohistochemical Analysis
After fixation in formalin, tumor samples were embedded in paraffin, sectioned (thickness 4 µm), deparaffinized, rehydrated, and pretreated using the Dako PTLink system (Dako, Carpinteria, CA, USA) and processed on an automated DAKO Autostainer platform using the DAKO EnVision® FLEX High pH Link kit (pH 9) [26]. IHC staining was performed with anti-cleaved caspase-3 (CC3) (1:100, #9661, Cell Signaling Technology, Beverly, MA, USA) according to the procedure proposed by the manufacturer. Counterstaining was made with hematoxylin. Slowly growing NB tissue was used as the negative control, and irradiated NB tissue was used as the positive control. Digital images were captured with a 40 magnification using a Panoramic Scanner P250 (3DHISTECH, Budapest, Hungary) at Histocenter AB (Mölndal, Sweden), with the software CaseViewer 2.4, Slide Converter.
For a quantitative representation of the IHC data, IHC scoring was performed by a certified and experienced pathologist using the semi-quantitative Histoscore method, where
was calculated based on visual (eyeballing) assessment of the intensity of the staining (graded: 0, negative; 1, weak; 2, moderate; or 3, strong) and the percentage of positive cells within each entire section.
2.5. Gene Expression Analyses
RNA was extracted from tumor samples using a phenol-chloroform method (RNeasy Lipid Tissue Mini Kit, QIAGEN, Valencia, CA, USA). RNA purity, integrity, and concentration were assessed with a Nanodrop 1000 Spectrometer (Thermo Fisgher Scientific, Waltham, MA, USA) (260/280 > 1.8), Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA) (RIN > 8), and a Qubit 3.0 Fluorometer (Thermo Fisher Scientific, Waltham, MA, USA), respectively. cDNA was subsequently generated via reverse transcription (RT2 First Strand Kit, QIAGEN, Valencia, CA, USA) and mixed with RT2 SYBR Green Mastermix (QIAGEN, Valencia, CA, USA) before aliquoting in a 96-well RT2 Profiler PCR Array for human apoptosis (PAHS-012Z, QIAGEN, Valencia, CA, USA). In total, 84 key genes involved in the apoptosis pathway were included, where 52, 24, and 8 were classified as pro-apoptotic, apoptosis-related, and anti-apoptotic genes, respectively, classified according to the manufacturer and the Gene Ontology (GO) database.
The cycle threshold (Ct) values obtained were converted to ∆Ct values based on the gene of interest versus the geometric mean of the housekeeping genes (ACTB, B2M, GAPDH, HPRT1, and RPLP0). Thereafter, the mean relative ∆∆Ct was calculated for each treatment in relation to the mean ∆Ct of the vehicle control group. By implementing the 2–∆∆Ct method, treated vs. control, we obtained a fold change (FC) value for each gene [33]. Genes were defined as differentially expressed if |FC| > 1.5.
2.6. Statistical Analyses
All calculations and statistical analyses were made with GraphPad Prism version 9.4.1.681 (GraphPad Software, Boston, MA, USA) and Excel 2013 for Windows (Microsoft Corporation, Redmond, WA, USA). The relative tumor volume (RTV) was determined individually for each mouse and time point, and mean value and the standard error of the mean (SEM) were calculated for each group. One-way ANOVA was used for estimating the statistical differences regarding tumor volume between all groups throughout the treatment period. The Student’s t-test was applied for comparison between groups. p < 0.05 was considered statistically significant different.
A theoretical value of the level of additive effect of both monotherapies (177Lu-octreotide and lorlatinib, respectively) was calculated based on The Bliss independence model, assuming an independent effect of each monotherapy [34,35]. Firstly, the fractional response, , of each monotherapy was estimated in relation to vehicle control.
Subsequently, the theoretical additive effect was estimated.
The theoretical additive effect value was calculated for days 4 to 14 and illustrated as a dashed line in Figure 1C for each time point. An effect of combination therapy larger than the theoretical additive effect (lower ) was regarded as synergistic, and if the effect was smaller (higher ) it was regarded as antagonistic.
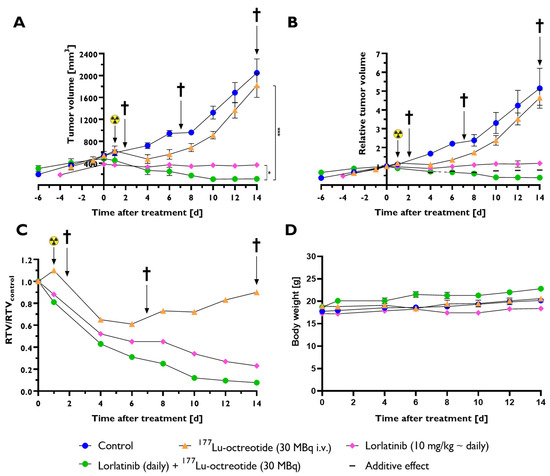
Figure 1.
Effect of treatment with either lorlatinib (pink), 177Lu-octreotide (orange) or both in combination (green) in mice bearing human CLB-BAR NB xenografts on tumor volume (A–C) and body weight (D). Mice were treated with lorlatinib (daily gavage, 10 mg/kg), and/or 177Lu-octreotide (30 MBq i.v. on day 1, symbolized by the radiation symbol) or i.v. injected with saline on day 1 (control) (n = 10 mice/group on day 0). The effect of each monotherapy and the combination therapy is shown as mean tumor volume (mm3) (A) and relative tumor volume (RTV) (B). A theoretical additive effect was calculated using the Bliss independence model (Bliss C. 1939) and presented as a dashed line in (B). (C) The RTV-ratio (RTV/RTVcontrol) for all treated groups is shown. Three mice from each group were sacrificed (†) on day 2 and day 7, respectively, and the remaining four mice in each group were sacrificed on day 14. Whole body weight of each group is presented in (D). Error bars represent SEM, not always visible because of their low values, * indicates p < 0.05 and *** p < 0.0001.
For mRNA expression analyses, Student’s t-tests were applied to compare ∆Ct-values of deregulated genes post treatment with vehicle control. p < 0.05 was considered statistically significant.
For immunohistochemical analyses, a Jarque–Bera test for normality showed that the Histoscores for CC3 staining did not have a normal distribution (p-value < 0.001), possibly due to the low number of samples. Because of this, the non-parametric Kruskal–Wallis test with Dunn’s Multiple Comparison Test was used to compare differences between groups.
3. Results
3.1. Combination Therapy Gave the Largest Tumor Volume Reduction
BALB/c nude mice with sc CLB-BAR NB xenografts were treated with either lorlatinib, 177Lu-octreotide, or both in combination over 14 days. Combined therapy with both lorlatinib and 177Lu-octreotide yielded the greatest antitumor effect with a RTV of 0.39 at day 14 (Figure 1A,B). The corresponding RTV value at day 14 for lorlatinib monotherapy was 1.2. 177Lu-octreotide as monotherapy (i.v. injection at day 1) demonstrated an initial decrease in RTV compared to control, with the lowest RTV of 1.1 on day 4, after which RTV increased to 4.6 on day 14 and displayed a similar tumor growth as the control group from day 8 to 14. RTV decreased between days 1 and 4 by 7.1%, 9.8%, and 19% for 177Lu-octreotide monotherapy, lorlatinib monotherapy, and combination therapy, respectively. The RTV in the controls increased monotonically reaching 5.2 on day 14. Combination therapy showed a more prominent antitumor effect than the theoretical value of the additive effect of both monotherapies combined from day 6 (Figure 1B).
To further illustrate the differences between each treatment versus control, we calculated the RTV ratio (RTVtherapy/RTVcontrol (RTV-ratio) over the 14 day treatment (Figure 1C). 177Lu-octreotide monotherapy resulted in a decrease in RTV ratio on day 4 that continued until day 6 and then gradually increased. In contrast, the RTV ratios of the lorlatinib and combination treatment groups decreased throughout the treatment period and reached 0.23 and 0.08 on day 14, respectively. One-way ANOVA demonstrated a significant difference between all groups from day 4 to 14, with p < 0.001 at all studied time points. No significant weight loss was observed in any group (Figure 1D).
3.2. Changes in the Expression of Genes Involved in Apoptosis
To better understand the effect of treatment on CLB-BAR xenograft growth, we harvested tumors at days 2, 7, and 14 of treatment and performed qPCR analyses for 84 selected genes, including pro-apoptotic genes (Figure 2) and anti-apoptotic and apoptosis-related genes (Figure 3). Lorlatinib treatment resulted in mRNA expression changes in 5 of the 79 detected transcripts from the 84 gene panel (|FC| > 1.5). In response to lorlatinib, all significantly regulated genes were down-regulated, two genes (TNFRSF9 and BIRC3) were found after 7 days, and four genes (CASP8, CD40, BIRC3, and BNIP3L) were found 14 days post treatment. Three pro-apoptotic genes were down-regulated in comparison with the control: CASP8, CD40, and TNFRSF9 (Figure 2). BIRC3, categorized as anti-apoptotic, was down-regulated after 7 and 14 days (Figure 3). BNIP3L, categorized as apoptosis related, was down-regulated after 14 days (Figure 3).
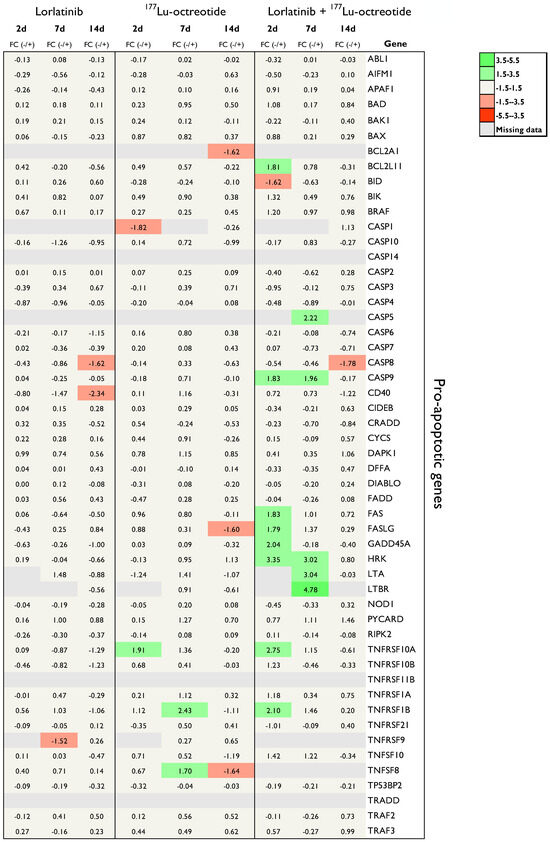
Figure 2.
mRNA expression of 52 pro-apoptotic genes in tumor tissue (CLB-BAR) from mice treated with lorlatinib and/or 177Lu-octreotide, expressed as fold change (FC) relative to controls. Tumors were analyzed from mice sacrificed on day 2 (n = 3), day 7 (n = 3), or day 14 (n = 4). Red and green colors represent down- and up-regulation, respectively, with |FC| > 1.5. Missing data are represented by gray color.
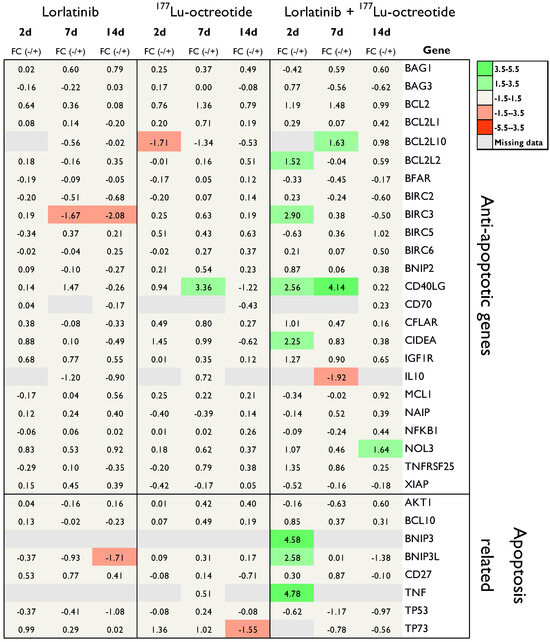
Figure 3.
mRNA expression of 24 anti-apoptotic and 8 apoptosis related genes in tumor tissue (CLB-BAR) from mice treated with lorlatinib and/or 177Lu-octreotide, expressed as fold change (FC) relative to controls. Tumors analyzed were from mice sacrificed on day 2 (n = 3), day 7 (n = 3) or day 14 (n = 4). Red and green colors represent down- and up-regulation, respectively, with |FC| > 1.5. Missing data are represented by gray color.
Treatment with 177Lu-octreotide yielded expression changes in 9 unique genes of 79 detectable mRNAs (Figure 2 and Figure 3). Six of the genes were classified as pro-apoptotic: BCL2AI, CASPI, FASLG, TNFRSF10A, TNFRSF1B, and TNFSF8. BCL2AI, CASPI, FASLG, and TNFSF8 (day 14) were down-regulated, whereas TNFRSF10A, TNFRSF1B, and TNFSF8 (day 7) were up-regulated (Figure 2). Two of the genes were classified as anti-apoptotic, BCL2L10 and CD40LG, detected as down- and up-regulated, respectively. TP73, classified as apoptosis related, was found to be down-regulated.
Combination treatment with both lorlatinib and 177Lu-octreotide resulted in differential expression of 23 unique genes of 78 detectable mRNAs (Figure 2 and Figure 3). In total, 13 pro-apoptotic genes, namely, BCL2LII, BID, CASP5, CASP8, CASP9, FAS, FASLG, GADD45A, HRK, LTA, LTBR, TNFRSF10A, and TNFRSF1B, exhibited differences in expression (Figure 2); only BID and CASP8 were down-regulated, while the remaining genes were up-regulated. CASP9 was up-regulated at both 2 and 7 days (Figure 2). Seven anti-apoptotic genes, namely, BCL2LI0, BCL2L2, BIRC3, CD40LG, CIDEA, IL10, and NOL3, were differentially expressed. Of these genes, IL10 was the only down-regulated gene, and the remaining genes were up-regulated (Figure 3). CD40LG was commonly found to be up-regulated after 2 and 7 days (Figure 3). Three apoptosis-related genes, namely, BNIP3, BNIP3L, and TNF, were down-regulated.
Figure 4 illustrates the overall transcriptional response for the statistically significant regulated genes (|FC| > 1.5, p < 0.05). The presented genes are relevant in both intrinsic (BID, BNIP3L, CASP9, and HRK) and extrinsic (CASP8, CD40, FASLG, NOL3, and TNFRSF10A) apoptosis pathways. At 2 and 7 days after treatment started, the majority of significantly regulated genes were found in the combination group, and most of them were pro-apoptotic. Most of the genes were also up-regulated compared with the control group. This pattern was not observed at 14 d.
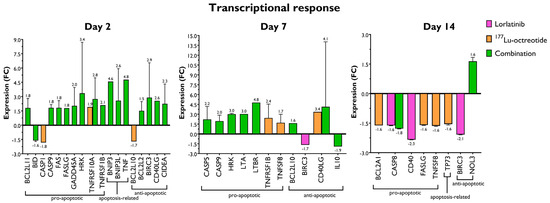
Figure 4.
mRNA expression of pro-apoptotic, anti-apoptotic, and apoptosis-related genes in human NB xenograft tumor tissue (CLB-BAR) from mice treated with lorlatinib and/or 177Lu-octreotide, expressed as fold change (FC) relative to controls, on day 2 (n = 3), day 7 (n = 3) or day 14 (n = 4). Only differentially regulated genes with |FC| > 1.5 are presented. Error bars indicate SEM.
3.3. Immunohistochemical Analyses
We next analyzed apoptosis at the protein level in treated tumors employing antibodies against cleaved caspase 3 (CC3). Immunohistochemical examination identified CC3-positive cells (with nuclear staining) at days 2 and 14 after treatment start (Figure 5). Overall, we observed an increase in CC3-positive cells (Histoscore) in tumors from treated mice, with the exception of lorlatinib on day 2, with the highest Histoscore for combination-treated tumors on day 2. However, there was a large spread within the groups, and statistical significance was only observed at day 14 in the combination group.
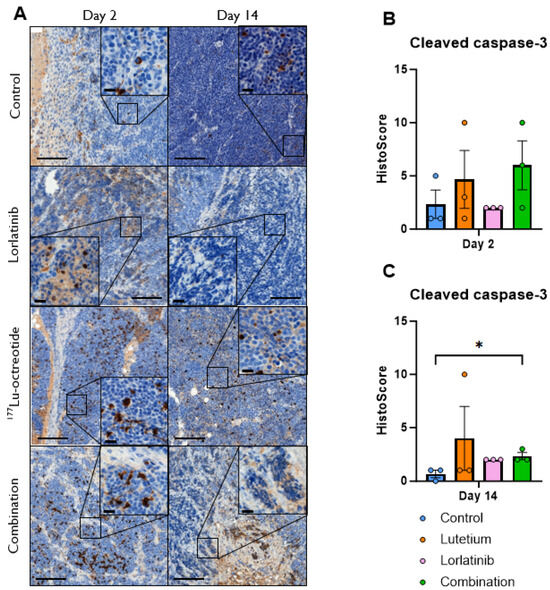
Figure 5.
(A) Immunohistochemical staining for cleaved caspase-3 (CC3) in CLB-BAR tumors, 2 and 14 days post treatment. Lorlatinib was administered daily via oral gavage from day 0, 177Lu-octreotide was administered i.v. on day 1 in both the combination group and the monotherapy and the control were i.v. injected with saline on day 1. Numerous CC3-positive cells (brown color) were found in all images (counterstained with hematoxylin), with the highest intensity (visually) in the treated groups. Scale bars equals 200 µm and 20 µm (40 magnified inserts). (B,C) Distribution of CC3 Histoscore in CLB-BAR tumors at 2 (B) and 14 days (C) after treatment start for controls (blue), 177Lu-octreotide (orange), lorlatinib (pink), and both 177Lu-octreotide and lorlatinib in combination (green). The mean Histoscore value was calculated for each group based on the intensity (graded: 0, negative; 1, weak; 2, moderate; or 3, strong) and the percentage of positive cells. Error bars represent SEM. * represents p < 0.05 calculated using Kruskal–Wallis test with Dunn’s Multiple Comparison Test.
4. Discussion
Lorlatinib, a third-generation ALK inhibitor, has emerged as a treatment alternative for ALK-positive HR-NB [13]. Lorlatinib demonstrates enhanced activity and efficiency against ALK mutations compared to previous generations of ALKi [10,11,12]. Nevertheless, several studies have reported lorlatinib resistance in response to treatment, emphasizing the need for a multimodal therapy approach [15,16,17,18]. Systemic treatment with radiolabeled SSTAs, e.g., 177Lu-octreotide, is successfully used for SSTR-overexpressing neuroendocrine tumors. To our knowledge, this is the first study examining and reporting radio-sensitization and synergistic antitumor effects in an ALK-amplified and SSTR-positive HR-NB xenograft model with lorlatinib and 177Lu-octreotide.
The present study was performed on CLB-BAR cells with MYCN amplification and ALK gain-of-function that may represent high-risk neuroblastomas [32,36]. In the mice, the CLB-BAR tumor volume reduction was moderate after treatment with 177Lu-octreotide, which is consistent with results from a previous study in the same animal model, comparing effects of 177Lu-octreotide and 177Lu-octreotate [37]. Although biodistribution and biokinetics were most favorable for 177Lu-octreotate, resulting in higher absorbed dose to tumor for 177Lu-octreotate, the tumor volume reduction was somewhat better for 177Lu-octreotide.
It should be noted that we purposefully employed low doses of 177Lu-octreotide and lorlatinib in this study to enable visualization of additive or synergistic effects. The administered amounts were based on previous work using CLB-BAR xenografts [12,37,38,39], and while higher doses of each drug have been tested without significant adverse effects, the combination of drugs with different mechanisms of action might result in different or enhanced toxicities. This further warrants the choice of lower administered amounts in this study. The moderate therapeutic effects of 177Lu-octreotide monotherapy despite high uptake and retention in NB compared with other neuroendocrine tumors in similar models are unexpected, since the therapeutic effects in such studies were much more dramatic, with a possibility to eradicate the tumor totally [40,41,42,43]. CLB-BAR has a high proliferation rate and tumor growth curve compared with other neuroendocrine tumor models of, e.g., intestinal origin, which might influence the therapeutic response [42]. Monotherapy with lorlatinib resulted in a cytostatic response. Thus, the present results are difficult to interpret, and may be related to certain properties in NB. Detailed signaling pathway analyses will be needed in order to understand the radioresistance in NB, and contribute to a better understanding, needed for future optimization of therapy with radiolabeled SSTAs. It is, however, clear that inhibition of ALK with lorlatinib may act to overcome some of the radioresistance in CLB-BAR NB cell xenografts.
ALK signals via several downstream pathways, having a considerable role in, e.g., cell cycle progression, survival, DNA repair, proliferation, and angiogenesis, via Ras-extracellular signal-regulated kinase (Erk), PI3K-AKT-mTOR and Janus protein tyrosine kinase (JAK)-STAT [38,44,45]. However, how these signaling pathways are affected by irradiation, either alone or in combination with an ALKi, has not been explored. The present study demonstrated that treatment of CLB-BAR tumors with lorlatinib in combination with 177Lu-octreotide synergistically reduced tumor volume and resulted in elevated transcription of genes involved in apoptosis. Several studies suggest that inhibiting critical mediators of the DNA damage response (DDR) can enhance radiosensitivity [45,46,47,48,49]. Dolman et al. demonstrated radiosensitization of NB cells by inhibiting the DNA-dependent protein kinase catalytic subunit (DNA-PKcs), playing a central role in the repair of DNA double-strand breaks (DSBs) via non-homologous end-joining (NHEJ) [46]. Further, a combination of 177Lu-octreotate and a p53 stabilizing drug showed better therapeutic effects compared to monotherapy in NB cell spheroids and NB xenografts on mice [50,51]. Altogether, these findings indicate a possible role for DNA repair inhibition in the enhanced response to 177Lu-octreotide combined with lorlatinib. Previous studies have illustrated the radiosensitizing effects of crizotinib, the first generation of ALKi, in combination with external radiation in ALK-positive non-small cell lung cancer (NSCLC) models [52,53]. The Akt kinase is one common mediator in both DDR and ALK pathways [54,55]. DNA damage sensors, e.g., DNA-PKcs, phosphorylate Akt in the DDR pathway due to, e.g., radiation-induced DSBs [54]. Subsequently, phosphorylated Akt promotes NHEJ-mediated DSB repair and cell survival [54]. Hence, elevated levels of phosphorylated Akt is associated with malignant tumors and poor prognosis [56,57]. Lorlatinib can, like crizotinib, aggravate NHEJ-mediated DSB repair by partially inhibiting Akt, a downstream signaling effector of ALK, and act as a radiosensitizer. However, a driving force in lorlatinib resistance for HR-NB is the activation of bypass pathways, such as EGFR, ErbB4, and RAS [15,58]. Other reported mechanisms causing ALKi-resistance are structural alterations in the kinase domain, leading to a decreased binding of ALKi, as well as amplification of ALK [59]. Identifying the specific mechanisms underlying the drug-resistant properties that arise in response to ALKi treatments is essential for further treatment. In several patient case reports where relapse has occurred, multiple biopsies distributed throughout the treatment have been employed to modify the choice of ALKi to provide an optimal treatment strategy [60,61,62,63]. Of special interest is a case where an ALK-positive NSCLC gained resensitization to crizotinib after acquiring resistance to lorlatinib, highlighting the critical information provided via biopsies and the remarkable mechanisms behind ALKi resistance [64].
Given the well-documented heterogeneous properties of NB [65], it may be necessary to eradicate tumor cells via various targets, aiming to eradicate multiple sub-populations of tumor cells and thus eliminate resistance. Based on our results, radiolabeled SSTAs may be a beneficial adjunctive treatment for disseminated SSTR-positive HR-NBs. Pilot trials with 177Lu-octreotate, an alternative SSTA, have shown varied results for NB patients, highlighting the need for optimization according to each patient’s specific tumor characteristics [27,28,29]. In addition, to avoid under-treatment, the biokinetics of the radiopharmaceutical need to be investigated before the start of the treatment. In the ongoing phase II trial with HR-NB patients, LuDO-N, 68Ga-octreotide PET/CT examination will be used to determine activity levels of the two 177Lu-octreotate administrations, not exceeding mean absorbed dose levels to the organs at risk (23 and 2.4 Gy to kidneys and whole body, respectively) [30]. Combining this information with information regarding eventual gain-of-function ALK mutations to, if possible, determine an appropriate ALKi for combination therapy is a further step to individualize the treatment.
The transcriptional response of the 84 genes involved in apoptosis provided another demonstration of the synergistic antitumor effect provided by the combination therapy. In total, 40 genes were up- or down-regulated, with the strongest differential gene expression observed in the 177Lu-octreotide/lorlatinib combination therapy arm in which 26 genes were modulated, compared with 10 in response to 177Lu-octreotide treatment, and only 4 with lorlatinib. Overall, a higher number of pro-apoptotic genes were up-regulated in the combination therapy at earlier time points, although more anti-apoptotic genes were also up-regulated at the same time. After statistical analysis of the ∆Ct values (treated vs. control), only the combination treatment yielded a significant effect on the transcriptional response of genes within the Bcl-2 family (Table 1). Proteins of the Bcl-2 family are essential for apoptotic cell death in health and disease through different mechanisms [66,67,68,69]. Functionally categorized as pro-apoptotic or anti-apoptotic proteins within the Bcl-2 family, the imbalance of concentrations leads to cell survival or death [66,67]. Conversely, the tumor cells can continue their growth and progress if apoptosis can be inhibited, meaning that increased levels of anti-apoptotic proteins are associated with oncogenesis [67]. Our data demonstrated down-regulation of BID (BH3-interacting domain death agonist) on day 2, and up-regulation of HRK (Harakiri, BCL2 interacting protein) on day 7, both classified as pro-apoptotic genes. BID encodes for a protein (Bid) that connects the extrinsic apoptotic pathway with the intrinsic [70]. Cleaved Bid is translocated to the mitochondria and induces cytochrome c release, subsequently promoting downstream caspase activation [70]. Hrk is a BH3-only protein, encoded by HRK, and regulates apoptosis by displacing Bim or Bid from anti-apoptotic Bcl-xL, also leading to cytochrome c release and caspase activation [67,71]. The relatively strong up-regulation of HRK (fold change 3.02) suggests a possible mechanism for the increased apoptosis observed in Figure 5. Future studies should investigate the involvement of Bcl-xL in resistance to ALK inhibitors or radiolabeled SSTAs, and the potential use of BCL2-family inhibitors in combination with these modalities.

Table 1.
Functional characterization of differentially regulated genes after treatment with either lorlatinib, 177Lu-octreotide, or both in combination. Differential gene expression is expressed as fold change (FC) relative to control. Only genes exhibiting |FC| > 1.5 and p < 0.05 are presented. Lilac, yellow, and brown colors represent classification as pro-apoptotic, apoptosis-related, and anti-apoptotic genes, respectively.
Other genes with their associated protein families belonged to caspase, tumor necrosis factor (TNF), and p53. The caspase family executes cell death via a cascade of activations. Various members are involved in both the extrinsic and intrinsic apoptotic pathways, and are classified as initiator or effector caspases [67,72]. CASP8, encoding the initiator caspase-8 (extrinsic pathway), was down-regulated on day 14 post treatment with lorlatinib and the combination. Conversely, CASP9, encoding for the initiator caspase-9 (intrinsic pathway), was up-regulated for the combination on days 2 and 7. TNF and TNF-receptor (TNFR) superfamilies are involved in the extrinsic signaling pathway of apoptosis and are activated by ligand binding to receptors [73]. Within the TNFR superfamily, TNFRSF10A was up-regulated on day 2 for both 177Lu-octreotide and the combination. Whereas CD40 was down-regulated on day 14 for lorlatinib, FASLG, encoding for Fas ligand belonging to the TNF superfamily, was down-regulated on day 14 for 177Lu-octreotide. The tumor-suppressor family p53 is one of the most frequently mutated genes in cancer [74,75]; within this family, TP73 was down-regulated on day 14 for 177Lu-octreotide. TP73 is involved in the regulation of, i.e., tissue development and inflammation [74,76].
The number of genes with larger spread (larger error bars) in mRNA expression were more prominent early after exposure (day 2, i.e., 1 day after injection with 177Lu-octrotide), with only a few at day 7 and none at day 14. High SEM values were in general due to one or two very high values, all values in the same regulation direction. Most of these genes were differentially expressed in the group given the combination therapy. There may be several reasons for different expression between tumors in different mice: the relatively low doses given that may result in various effects, the early time point when differences in signaling is more probable, and the heterogeneous nature of NB.
Compared with two previous studies in a human small-intestine NET GOT1 mouse model where the transcriptional response was studied after treatment with 177Lu-octreotate, some genes are commonly found. For example, BNIP3L (apoptosis-related) was up-regulated 41 days after administration with 15 MBq 177Lu-octreotate. However, in the present study, BNIP3L was down-regulated after lorlatinib treatment on days 7 and 14 [77]. Besides comparing different tumor types, the relatively large time difference between the GOT1 and the present study may suggest that the tumors were in different phases after treatment, tumor regrowth vs. tumor shrinkage, and explain differences in the transcriptional response. In another study on the GOT1 model, BIRC3 (anti-apoptotic) was up-regulated one day post administration with 30 MBq 177Lu-octreotate [78], whereas it was down-regulated after lorlatinib treatment on days 7 and 14. A common up-regulation of the death receptor genes TNFSRSF10A and TNFSRSF10B, was also observed. TNFSRSF10A was up-regulated on day 2 after the combination treatment (present study), while TNFSRSF10B was up-regulated on days 1 and 7 post treatment with 30 MBq 177Lu-octreotate in the GOT1 study.
We complemented our gene expression analyses with immunohistochemistry for CC3 to study apoptosis at the protein level, as caspase 3 has a central role in both apoptotic pathways as an executioner caspase. CC3 positivity was increased in response to treatment with the exception of lorlatinib on both day 2 and day 14. The presence of cleaved caspase 3 in cells is a clear indication of a cell undergoing apoptosis. However, since this process results in dismantling of the cellular components and eventual phagocytosis by, e.g., macrophages, the timing of the cleaved caspase 3 expression and staining positivity can be difficult, especially considering that 177Lu-octreotide is retained in the tumor cells resulting in prolonged irradiation of the cell. This means that the expression of cleaved caspase 3 will not occur simultaneously in all treated cells, and the expression will not last over a long period of time since the dying cells are removed continuously. This results in higher variability in the staining for cleaved caspase 3. However, the changes were large enough to be statistically significant in the combination group on day 14.
In future studies, it would be interesting to combine fractionated/repeated administration of 177Lu-octreotide in combination with lorlatinib. In a previous study in the same animal model, we observed that fractionated administration of 177Lu-octreotate with short intervals resulted in a more prominent antitumor effect due to higher uptake in tumor cells because of recycling of SSTRs counteracting the likely SSTR saturation effects that prevail with a single injection of higher amounts of SSTAs [37].
Treatment of HR-NB poses a challenge since most cases are diagnosed as an advanced, metastasized, and, therefore, inoperable disease. Although an intense treatment approach has been successful for certain patients, it is not always obvious which patients benefit most from them. And as the patient group is children, intensive treatments are avoided as much as possible because of the late effects. Hence, a multimodal approach could benefit two aspects: increased tumor control and mitigating side effects. Our study highlights the radiosensitizing nature of lorlatinib in NB-bearing mice. The fact that both treatments are systemic enables their combination in cases where the cancer has metastasized. Today, both lorlatinib and radiolabeled SSTAs are being studied separately in various phase I/II trials for HR-NBs, enabling the course for a possible combination for the cases where the prerequisites are met [14,30].
5. Conclusions
Synergistic antitumor effects were found when lorlatinib and 177Lu-octreotide treatment was combined in an HR-NB xenograft mouse model. Combination therapy also had a more significant impact on expression of genes involved in apoptotic processes, for example up-regulation of the Bcl-xL inhibitor HRK. This was shown to result in a significant increase in cleaved caspase 3 in tumor cells receiving combination therapy. The data suggest that inhibiting ALK signaling plays a role in the radiosensitization of radionuclide therapy with 177Lu-octreotide, implying a potential for clinical application of combining ALKi with radiolabeled SSTAs for HR-NBs overexpressing SSTRs and ALK.
Author Contributions
A.R., B.H., R.H.P., J.S., K.H. and E.F.-A. designed and conceptualized the study; D.E.L. and D.P. carried out the cell culturing; D.P. and A.R. conducted the animal experiments; H.B. and A.R. were responsible for the labeling of 177Lu. A.R., K.S. and N.R. contributed to the RNA extraction and the validation steps involved; K.S. and A.R. were responsible for the qPCR and the following data measurements; A.R., E.F.-A., N.R. and J.S. contributed to the data analysis and interpretations. All authors reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by grants from the Swedish Research Council (EFA: 2021-02636; RHP: 2019-03914; BH: 2021-01192), the Swedish Cancer Society (EFA: CAN20/1293 and 23 2975; RHP: CAN21/1459; BH: CAN21/1525), Swedish Childhood Cancer Foundation (EFA: PR2017-0057; RHP: PR2022-0029; BH: PR2021-0027), BioCARE—a National Strategic Research Program at University of Gothenburg, the Swedish state under the agreement between the Swedish government and the county councils—the ALF-agreement (ALFGBG-966074), the King Gustav V Jubilee Clinic Cancer Research Foundation, the Sahlgrenska University Hospital Research Funds, Wilhelm and Martina Lundgren Research Foundation, Assar Gabrielsson Cancer Research Foundation, Herbert & Karin Jacobsson Foundation, and Adlerbertska Research Foundation.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Swedish Ethical Committee on Animal Experiments in Gothenburg (protocol code 2779-20 and date of approval 2020-08-26).
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Umapathy, G.; Mendoza-Garcia, P.; Hallberg, B.; Palmer, R.H. Targeting anaplastic lymphoma kinase in neuroblastoma. APMIS 2019, 127, 288–302. [Google Scholar] [CrossRef]
- De Brouwer, S.; De Preter, K.; Kumps, C.; Zabrocki, P.; Porcu, M.; Westerhout, E.M.; Lakeman, A.; Vandesompele, J.; Hoebeeck, J.; Van Maerken, T.; et al. Meta-analysis of neuroblastomas reveals a skewed ALK mutation spectrum in tumors with MYCN amplification. Clin. Cancer Res. 2010, 16, 4353–4362. [Google Scholar] [CrossRef] [PubMed]
- Eleveld, T.F.; Oldridge, D.A.; Bernard, V.; Koster, J.; Daage, L.C.; Diskin, S.J.; Schild, L.; Bentahar, N.B.; Bellini, A.; Chicard, M.; et al. Relapsed neuroblastomas show frequent RAS-MAPK pathway mutations. Nat. Genet. 2015, 47, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Pugh, T.J.; Morozova, O.; Attiyeh, E.F.; Asgharzadeh, S.; Wei, J.S.; Auclair, D.; Carter, S.L.; Cibulskis, K.; Hanna, M.; Kiezun, A.; et al. The genetic landscape of high-risk neuroblastoma. Nat. Genet. 2013, 45, 279–284. [Google Scholar] [CrossRef]
- Berry, T.; Luther, W.; Bhatnagar, N.; Jamin, Y.; Poon, E.; Sanda, T.; Pei, D.; Sharma, B.; Vetharoy, W.R.; Hallsworth, A.; et al. The ALKF1174L mutation potentiates the oncogenic activity of MYCN in neuroblastoma. Cancer Cell 2012, 22, 117–130. [Google Scholar] [CrossRef]
- Zhu, S.; Lee, J.S.; Guo, F.; Shin, J.; Perez-Atayde, A.R.; Kutok, J.L.; Rodig, S.J.; Neuberg, D.S.; Helman, D.; Feng, H.; et al. Activated ALK collaborates with MYCN in neuroblastoma pathogenesis. Cancer Cell 2012, 21, 362–373. [Google Scholar] [CrossRef]
- Schönherr, C.; Ruuth, K.; Kamaraj, S.; Wang, C.-L.; Yang, H.-L.; Combaret, V.; Djos, A.; Martinsson, T.; Christensen, J.G.; Palmer, R.H.; et al. Anaplastic Lymphoma Kinase (ALK) regulates initiation of transcription of MYCN in neuroblastoma cells. Oncogene 2012, 31, 5193–5200. [Google Scholar] [CrossRef]
- Morgenstern, D.A.; Bagatell, R.; Cohn, S.L.; Hogarty, M.D.; Maris, J.M.; Moreno, L.; Park, J.R.; Pearson, A.D.; Schleiermacher, G.; Valteau-Couanet, D.; et al. The challenge of defining “ultra-high-risk” neuroblastoma. Pediatr. Blood Cancer 2019, 66, e27556. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.W.; Richardson, P.F.; Bailey, S.; Brooun, A.; Burke, B.J.; Collins, M.R.; Cui, J.J.; Deal, J.G.; Deng, Y.-L.; Dinh, D.; et al. Discovery of (10 R)-7-Amino-12-fluoro-2, 10, 16-trimethyl-15-oxo-10, 15, 16, 17-tetrahydro-2H-8, 4-(metheno) pyrazolo [4, 3-h][2, 5, 11]-benzoxadiazacyclotetradecine-3-carbonitrile (PF-06463922), a macrocyclic inhibitor of anaplastic lymphoma kinase (ALK) and c-ros oncogene 1 (ROS1) with preclinical brain exposure and broad-spectrum potency against ALK-resistant mutations. J. Med. Chem. 2014, 57, 4720–4744. [Google Scholar]
- Syed, Y.Y. Lorlatinib: First global approval. Drugs 2019, 79, 93–98. [Google Scholar] [CrossRef]
- Zou, H.Y.; Friboulet, L.; Kodack, D.P.; Engstrom, L.D.; Li, Q.; West, M.; Tang, R.W.; Wang, H.; Tsaparikos, K.; Wang, J.; et al. PF-06463922, an ALK/ROS1 inhibitor, overcomes resistance to first and second generation ALK inhibitors in preclinical models. Cancer Cell 2015, 28, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Tucker, E.R.; Wan, H.; Chand, D.; Danielson, L.S.; Ruuth, K.; El Wakil, A.; Witek, B.; Jamin, Y.; Umapathy, G.; et al. The ALK inhibitor PF-06463922 is effective as a single agent in neuroblastoma driven by expression of ALK and MYCN. Dis. Models Mech. 2016, 9, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Merguerian, M.D.; Rowe, S.P.; Pratilas, C.A.; Chen, A.R.; Ladle, B.H. Exceptional response to the ALK and ROS1 inhibitor lorlatinib and subsequent mechanism of resistance in relapsed ALK F1174L-mutated neuroblastoma. Mol. Case Stud. 2021, 7, a006064. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, K.C.; Park, J.R.; Kayser, K.; Malvar, J.; Chi, Y.-Y.; Groshen, S.G.; Villablanca, J.G.; Krytska, K.; Lai, L.M.; Acharya, P.T.; et al. Lorlatinib with or without chemotherapy in ALK-driven refractory/relapsed neuroblastoma: Phase 1 trial results. Nat. Med. 2023, 29, 1092–1102. [Google Scholar] [CrossRef] [PubMed]
- Redaelli, S.; Ceccon, M.; Zappa, M.; Sharma, G.G.; Mastini, C.; Mauri, M.; Nigoghossian, M.; Massimino, L.; Cordani, N.; Farina, F.; et al. Lorlatinib Treatment Elicits Multiple On- and Off-Target Mechanisms of Resistance in ALK-Driven Cancer. Cancer Res. 2018, 78, 6866–6880. [Google Scholar] [CrossRef] [PubMed]
- Mizuta, H.; Okada, K.; Araki, M.; Adachi, J.; Takemoto, A.; Kutkowska, J.; Maruyama, K.; Yanagitani, N.; Oh-Hara, T.; Watanabe, K.; et al. Gilteritinib overcomes lorlatinib resistance in ALK-rearranged cancer. Nat. Commun. 2021, 12, 1261. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Araki, M.; Sakashita, T.; Ma, B.; Kanada, R.; Yanagitani, N.; Horiike, A.; Koike, S.; Oh-Hara, T.; Watanabe, K.; et al. Prediction of ALK mutations mediating ALK-TKIs resistance and drug re-purposing to overcome the resistance. EBioMedicine 2019, 41, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Xie, B.; Qiu, Y.; Zhou, J.; Du, D.; Ma, H.; Ji, J.; Zhu, L.; Zhang, W. Establishment of an acquired lorlatinib-resistant cell line of non-small cell lung cancer and its mediated resistance mechanism. Clin. Transl. Oncol. 2022, 24, 2231–2240. [Google Scholar] [CrossRef] [PubMed]
- Lamberts, S.W.; Krenning, E.P.; Reubi, J.C. The Role of Somatostatin and Its Analogs in the Diagnosis and Treatment of Tumors. Endocr. Rev. 1991, 12, 450–482. [Google Scholar] [CrossRef]
- Baum, R.P.; Kluge, A.W.; Kulkarni, H.; Schorr-Neufing, U.; Niepsch, K.; Bitterlich, N.; van Echteld, C.J. [177Lu-DOTA] 0-D-Phe1-Tyr3-octreotide (177Lu-DOTATOC) for peptide receptor radiotherapy in patients with advanced neuroendocrine tumours: A phase-II study. Theranostics 2016, 6, 501. [Google Scholar] [CrossRef]
- Esser, J.P.; Krenning, E.P.; Teunissen, J.J.M.; Kooij, P.P.M.; Van Gameren, A.L.H.; Bakker, W.H.; Kwekkeboom, D.J. Comparison of [177Lu-DOTA0, Tyr3] octreotate and [177Lu-DOTA0, Tyr3] octreotide: Which peptide is preferable for PRRT? Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 1346–1351. [Google Scholar] [CrossRef]
- Reubi, J.C.; Schär, J.-C.; Waser, B.; Wenger, S.; Heppeler, A.; Schmitt, J.S.; Mäcke, H.R. Affinity profiles for human somatostatin receptor subtypes SST1–SST5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur. J. Nucl. Med. 2000, 27, 273–282. [Google Scholar] [CrossRef]
- Uusijärvi, H.; Bernhardt, P.; Ericsson, T.; Forssell-Aronsson, E. Dosimetric characterization of radionuclides for systemic tumor therapy: Influence of particle range, photon emission, and subcellular distribution. Med. Phys. 2006, 33, 3260–3269. [Google Scholar] [CrossRef]
- Uusijärvi, H.; Bernhardt, P.; Rösch, F.; Maecke, H.R.; Forssell-Aronsson, E. Electron- and positron-emitting radiolanthanides for therapy: Aspects of dosimetry and production. J. Nucl. Med. 2006, 47, 807–814. [Google Scholar] [PubMed]
- Swärd, C.; Bernhardt, P.; Ahlman, H.; Wängberg, B.; Forssell-Aronsson, E.; Larsson, M.; Svensson, J.; Rossi-Norrlund, R.; Kölby, L. [177Lu-DOTA0-Tyr3]-octreotate treatment in patients with disseminated gastroenteropancreatic neuroendocrine tumors: The value of measuring absorbed dose to the kidney. World J. Surg. 2010, 34, 1368–1372. [Google Scholar] [CrossRef] [PubMed]
- Romiani, A.; Spetz, J.; Shubbar, E.; Lind, D.E.; Hallberg, B.; Palmer, R.H.; Forssell-Aronsson, E. Neuroblastoma xenograft models demonstrate the therapeutic potential of 177Lu-octreotate. BMC Cancer 2021, 21, 950. [Google Scholar] [CrossRef]
- Gains, J.E.; Bomanji, J.B.; Fersht, N.L.; Sullivan, T.; D’Souza, D.; Sullivan, K.P.; Aldridge, M.; Waddington, W.; Gaze, M.N. 177Lu-DOTATATE molecular radiotherapy for childhood neuroblastoma. J. Nucl. Med. 2011, 52, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Kong, G.; Hofman, M.S.; Murray, W.K.; Wilson, S.; Wood, P.; Downie, P.; Super, L.; Hogg, A.; Eu, P.; Hicks, R.J. Initial experience with gallium-68 DOTA-octreotate PET/CT and peptide receptor radionuclide therapy for pediatric patients with refractory metastatic neuroblastoma. J. Pediatr. Hematol. Oncol. 2016, 38, 87–96. [Google Scholar] [CrossRef]
- Gains, J.E.; Moroz, V.; Aldridge, M.D.; Wan, S.; Wheatley, K.; Laidler, J.; Peet, C.; Bomanji, J.B.; Gaze, M.N. A phase IIa trial of molecular radiotherapy with 177-lutetium DOTATATE in children with primary refractory or relapsed high-risk neuroblastoma. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2348–2357. [Google Scholar] [CrossRef]
- Sundquist, F.; Georgantzi, K.; Jarvis, K.B.; Brok, J.; Koskenvuo, M.; Rascon, J.; van Noesel, M.; Grybäck, P.; Nilsson, J.; Braat, A.; et al. A Phase II Trial of a Personalized, Dose-Intense Administration Schedule of 177Lutetium-DOTATATE in Children With Primary Refractory or Relapsed High-Risk Neuroblastoma–LuDO-N. Front. Pediatr. 2022, 10, 167. [Google Scholar] [CrossRef]
- Forssell-Aronsson, E.; Spetz, J.; Ahlman, H. Radionuclide therapy via SSTR: Future aspects from experimental animal studies. Neuroendocrinology 2013, 97, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Fransson, S.; Hansson, M.; Ruuth, K.; Djos, A.; Berbegall, A.; Javanmardi, N.; Abrahamsson, J.; Palmer, R.H.; Noguera, R.; Hallberg, B.; et al. Intragenic anaplastic lymphoma kinase (ALK) rearrangements: Translocations as a novel mechanism of ALK activation in neuroblastoma tumors. Genes Chromosomes Cancer 2015, 54, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Bliss, C.I. The toxicity of poisons applied jointly 1. Ann. Appl. Biol. 1939, 26, 585–615. [Google Scholar] [CrossRef]
- Sandblom, V.; Spetz, J.; Shubbar, E.; Montelius, M.; Ståhl, I.; Swanpalmer, J.; Nilsson, O.; Forssell-Aronsson, E. Gemcitabine potentiates the anti-tumour effect of radiation on medullary thyroid cancer. PLoS ONE 2019, 14, e0225260. [Google Scholar] [CrossRef] [PubMed]
- Cazes, A.; Louis-Brennetot, C.; Mazot, P.; Dingli, F.; Lombard, B.; Boeva, V.; Daveau, R.; Cappo, J.; Combaret, V.; Schleiermacher, G.; et al. Characterization of Rearrangements Involving the ALK Gene Reveals a Novel Truncated Form Associated with Tumor Aggressiveness in Neuroblastoma. Cancer Res. 2013, 73, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Romiani, A.; Simonsson, K.; Pettersson, D.; Al-Awar, A.; Rassol, N.; Bakr, H.; Lind, D.E.; Umapathy, G.; Spetz, J.; Palmer, R.H.; et al. Comparison of 177Lu-octreotate and 177Lu-octreotide for treatment in human neuroblastoma-bearing mice. Heliyon 2024, 10, e31409. [Google Scholar] [CrossRef] [PubMed]
- Szydzik, J.; Lind, D.E.; Arefin, B.; Kurhe, Y.; Umapathy, G.; Siaw, J.T.; Claeys, A.; Gabre, J.L.; Eynden, J.V.D.; Hallberg, B.; et al. ATR inhibition enables complete tumour regression in ALK-driven NB mouse models. Nat. Commun. 2021, 12, 6813. [Google Scholar] [CrossRef] [PubMed]
- Romiani, A. Improved Radionuclide Therapy of Neuroblastoma—Preclinical Evaluation of 177Lu-Labeled Somatostatin Analogs. Ph.D. Thesis, University of Gothenburg, Gothenburg, Sweden, 2023. [Google Scholar]
- Kölby, L.; Bernhardt, P.; Johanson, V.; Schmitt, A.; Ahlman, H.; Forssell-Aronsson, E.; Mäcke, H.; Nilsson, O. Successful receptor-mediated radiation therapy of xenografted human midgut carcinoid tumour. Br. J. Cancer 2005, 93, 1144–1151. [Google Scholar] [CrossRef]
- Swärd, C.; Bernhardt, P.; Johanson, V.; Schmitt, A.; Ahlman, H.; Stridsberg, M.; Forssell-Aronsson, E.; Nilsson, O.; Kölby, L. Comparison of [177Lu-DOTA0, Tyr3]-octreotate and [177Lu-DOTA0, Tyr3]-octreotide for receptor-mediated radiation therapy of the xenografted human midgut carcinoid tumor GOT1. Cancer Biother. Radiopharm. 2008, 23, 114–120. [Google Scholar] [CrossRef]
- Elvborn, M.; Shubbar, E.; Forssell-Aronsson, E. Hyperfractionated Treatment with 177Lu-Octreotate Increases Tumor Response in Human Small-Intestine Neuroendocrine GOT1 Tumor Model. Cancers 2022, 14, 235. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, A.; Bernhardt, P.; Nilsson, O.; Ahlman, H.; Kölby, L.; Forssell-Aronsson, E. Differences in biodistribution between 99mTc-depreotide, 111In-DTPA-octreotide, and 177Lu-DOTA-Tyr3-octreotate in a small cell lung cancer animal model. Cancer Biother. Radiopharm. 2005, 20, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Ducray, S.P.; Natarajan, K.; Garland, G.D.; Turner, S.D.; Egger, G. The transcriptional roles of ALK fusion proteins in tumorigenesis. Cancers 2019, 11, 1074. [Google Scholar] [CrossRef] [PubMed]
- Borenäs, M.; Umapathy, G.; Lind, D.E.; Lai, W.-Y.; Guan, J.; Johansson, J.; Jennische, E.; Schmidt, A.; Kurhe, Y.; Gabre, J.L.; et al. ALK signaling primes the DNA damage response sensitizing ALK-driven neuroblastoma to therapeutic ATR inhibition. Proc. Natl. Acad. Sci. USA 2024, 121, e2315242121. [Google Scholar] [CrossRef]
- Dolman, M.E.M.; van der Ploeg, I.; Koster, J.; Bate-Eya, L.T.; Versteeg, R.; Caron, H.N.; Molenaar, J.J. DNA-dependent protein kinase as molecular target for radiosensitization of neuroblastoma cells. PLoS ONE 2015, 10, e0145744. [Google Scholar] [CrossRef] [PubMed]
- Toulany, M.; Kehlbach, R.; Florczak, U.; Sak, A.; Wang, S.; Chen, J.; Lobrich, M.; Rodemann, H.P. Targeting of AKT1 enhances radiation toxicity of human tumor cells by inhibiting DNA-PKcs-dependent DNA double-strand break repair. Mol. Cancer Ther. 2008, 7, 1772–1781. [Google Scholar] [CrossRef] [PubMed]
- Stronach, E.A.; Chen, M.; Maginn, E.N.; Agarwal, R.; Mills, G.B.; Wasan, H.; Gabra, H. DNA-PK mediates AKT activation and apoptosis inhibition in clinically acquired platinum resistance. Neoplasia 2011, 13, 1069-IN35. [Google Scholar] [CrossRef]
- Dong, J.; Ren, Y.; Zhang, T.; Wang, Z.; Ling, C.C.; Li, G.C.; He, F.; Wang, C.; Wen, B. Inactivation of DNA-PK by knockdown DNA-PKcs or NU7441 impairs non-homologous end-joining of radiation-induced double strand break repair. Oncol. Rep. 2018, 39, 912–920. [Google Scholar] [CrossRef]
- Lundsten, S.; Berglund, H.; Jha, P.; Krona, C.; Hariri, M.; Nelander, S.; Lane, D.P.; Nestor, M. p53-Mediated Radiosensitization of (177)Lu-DOTATATE in Neuroblastoma Tumor Spheroids. Biomolecules 2021, 11, 1695. [Google Scholar] [CrossRef]
- Berglund, H.; Salomonsson, S.L.; Mohajershojai, T.; Gago, F.J.F.; Lane, D.P.; Nestor, M. p53 stabilisation potentiates [(177)Lu]Lu-DOTATATE treatment in neuroblastoma xenografts. Eur. J. Nucl. Med. Mol. Imaging 2024, 51, 768–778. [Google Scholar] [CrossRef]
- Sun, Y.; Nowak, K.A.; Zaorsky, N.G.; Winchester, C.L.; Dalal, K.; Giacalone, N.J.; Liu, N.; Werner-Wasik, M.; Wasik, M.A.; Dicker, A.P.; et al. ALK Inhibitor PF02341066 (Crizotinib) Increases Sensitivity to Radiation in Non–Small Cell Lung Cancer Expressing EML4-ALKPF02341066 Sensitizes EML4-ALK NSCLC Cells to Radiation Treatment. Mol. Cancer Ther. 2013, 12, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Wei, Q.; Schwager, C.; Moustafa, M.; Zhou, C.; Lipson, K.E.; Weichert, W.; Debus, J.; Abdollahi, A. Synergistic effects of crizotinib and radiotherapy in experimental EML4–ALK fusion positive lung cancer. Radiother. Oncol. 2015, 114, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Turner, K.M.; Alfred Yung, W.K.; Chen, K.; Zhang, W. Role of AKT signaling in DNA repair and clinical response to cancer therapy. Neuro-Oncol. 2014, 16, 1313–1323. [Google Scholar] [CrossRef] [PubMed]
- Iida, M.; Harari, P.M.; Wheeler, D.L.; Toulany, M. Targeting AKT/PKB to improve treatment outcomes for solid tumors. Mutat. Res. 2020, 819, 111690. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Zhang, Y.; Liu, S.; Lu, J.; Yang, Y.; Wen, Q.; Fan, S. Elevated expression of G3BP1 associates with YB1 and p-AKT and predicts poor prognosis in nonsmall cell lung cancer patients after surgical resection. Cancer Med. 2019, 8, 6894–6903. [Google Scholar] [CrossRef] [PubMed]
- Opel, D.; Poremba, C.; Simon, T.; Debatin, K.M.; Fulda, S. Activation of Akt predicts poor outcome in neuroblastoma. Cancer Res. 2007, 67, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Berlak, M.; Tucker, E.; Dorel, M.; Winkler, A.; McGearey, A.; Rodriguez-Fos, E.; da Costa, B.M.; Barker, K.; Fyle, E.; Calton, E.; et al. Mutations in ALK signaling pathways conferring resistance to ALK inhibitor treatment lead to collateral vulnerabilities in neuroblastoma cells. Mol. Cancer 2022, 21, 126. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.J.; Riely, G.J.; Shaw, A.T. Targeting ALK: Precision Medicine Takes on Drug Resistance. Cancer Discov. 2017, 7, 137–155. [Google Scholar] [CrossRef] [PubMed]
- Makuuchi, Y.; Hayashi, H.; Haratani, K.; Tanizaki, J.; Tanaka, K.; Takeda, M.; Sakai, K.; Shimizu, S.; Ito, A.; Nishio, K.; et al. A case of ALK-rearranged non–small cell lung cancer that responded to ceritinib after development of resistance to alectinib. Oncotarget 2018, 9, 23315. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.G.; Cortinovis, D.; Agustoni, F.; Arosio, G.; Villa, M.; Cordani, N.; Bidoli, P.; Bisson, W.H.; Pagni, F.; Piazza, R.; et al. A compound L1196M/G1202R ALK mutation in a patient with ALK-positive lung cancer with acquired resistance to brigatinib also confers primary resistance to lorlatinib. J. Thorac. Oncol. 2019, 14, e257–e259. [Google Scholar] [CrossRef]
- Takahashi, K.; Seto, Y.; Okada, K.; Uematsu, S.; Uchibori, K.; Tsukahara, M.; Oh-Hara, T.; Fujita, N.; Yanagitani, N.; Nishio, M.; et al. Overcoming resistance by ALK compound mutation (I1171S+ G1269A) after sequential treatment of multiple ALK inhibitors in non-small cell lung cancer. Thorac. Cancer 2020, 11, 581–587. [Google Scholar] [CrossRef]
- Recondo, G.; Mezquita, L.; Facchinetti, F.; Planchard, D.; Gazzah, A.; Bigot, L.; Rizvi, A.Z.; Frias, R.L.; Thiery, J.P.; Scoazec, J.Y.; et al. Diverse Resistance Mechanisms to the Third-Generation ALK Inhibitor Lorlatinib in ALK-Rearranged Lung CancerResistance to Lorlatinib in ALK-Rearranged Lung Cancer. Clin. Cancer Res. 2020, 26, 242–255. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.T.; Friboulet, L.; Leshchiner, I.; Gainor, J.F.; Bergqvist, S.; Brooun, A.; Burke, B.J.; Deng, Y.L.; Liu, W.; Dardaei, L.; et al. Resensitization to crizotinib by the lorlatinib ALK resistance mutation L1198F. New Engl. J. Med. 2016, 374, 54–61. [Google Scholar] [CrossRef]
- Gomez, R.L.; Ibragimova, S.; Ramachandran, R.; Philpott, A.; Ali, F.R. Tumoral heterogeneity in neuroblastoma. Biochim. Biophys. Acta Rev. Cancer 2022, 1877, 188805. [Google Scholar] [CrossRef]
- Youle, R.J.; Strasser, A. The BCL-2 protein family: Opposing activities that mediate cell death. Nat. Rev. Mol. Cell Biol. 2008, 9, 47–59. [Google Scholar] [CrossRef]
- Singh, R.; Letai, A.; Sarosiek, K. Regulation of apoptosis in health and disease: The balancing act of BCL-2 family proteins. Nat. Rev. Mol. Cell Biol. 2019, 20, 175–193. [Google Scholar] [CrossRef] [PubMed]
- Spetz, J.K.E.; Florido, M.H.C.; Fraser, C.S.; Qin, X.; Choiniere, J.; Yu, S.J.; Singh, R.; Friesen, M.; Rubin, L.L.; Salem, J.-E.; et al. Heightened apoptotic priming of vascular cells across tissues and life span predisposes them to cancer therapy-induced toxicities. Sci. Adv. 2022, 8, eabn6579. [Google Scholar] [CrossRef]
- Singh, R.; Yu, S.; Osman, M.; Inde, Z.; Fraser, C.; Cleveland, A.H.; Almanzar, N.; Lim, C.B.; Joshi, G.N.; Spetz, J.; et al. Radiotherapy-Induced Neurocognitive Impairment Is Driven by Heightened Apoptotic Priming in Early Life and Prevented by Blocking BAX. Cancer Res. 2023, 83, 3442–3461. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.-M. Signal transduction mediated by Bid, a pro-death Bcl-2 family proteins, connects the death receptor and mitochondria apoptosis pathways. Cell Res. 2000, 10, 161–167. [Google Scholar] [CrossRef]
- Kaya-Aksoy, E.; Cingoz, A.; Senbabaoglu, F.; Seker, F.; Sur-Erdem, I.; Kayabolen, A.; Lokumcu, T.; Sahin, G.N.; Karahuseyinoglu, S.; Bagci-Onder, T. The pro-apoptotic Bcl-2 family member Harakiri (HRK) induces cell death in glioblastoma multiforme. Cell Death Discov. 2019, 5, 64. [Google Scholar] [CrossRef]
- Fan, T.J.; Han, L.H.; Cong, R.S.; Liang, J. Caspase family proteases and apoptosis. Acta Biochim. Et Biophys. Sin. 2005, 37, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Dostert, C.; Grusdat, M.; Letellier, E.; Brenner, D. The TNF family of ligands and receptors: Communication modules in the immune system and beyond. Physiol. Rev. 2019, 99, 115–160. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.M. A role for p53 in the frequency and mechanism of mutation. Mutat. Res./Rev. Mutat. Res. 2002, 511, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Marei, H.E.; Althani, A.; Afifi, N.; Hasan, A.; Caceci, T.; Pozzoli, G.; Morrione, A.; Giordano, A.; Cenciarelli, C. p53 signaling in cancer progression and therapy. Cancer Cell Int. 2021, 21, 703. [Google Scholar] [CrossRef] [PubMed]
- Zawacka-Pankau, J.E. The Role of p53 Family in Cancer. Cancers 2022, 14, 823. [Google Scholar] [CrossRef] [PubMed]
- Spetz, J.; Rudqvist, N.; Langen, B.; Parris, T.Z.; Dalmo, J.; Schüler, E.; Wängberg, B.; Nilsson, O.; Helou, K.; Forssell-Aronsson, E. Time-dependent transcriptional response of GOT1 human small intestine neuroendocrine tumor after 177Lu [Lu]-octreotate therapy. Nucl. Med. Biol. 2018, 60, 11–18. [Google Scholar] [CrossRef]
- Rassol, N.; Andersson, C.; Pettersson, D.; Al-Awar, A.; Shubbar, E.; Kovács, A.; Åkerström, B.; Gram, M.; Helou, K.; Forssell-Aronsson, E. Co-administration with A1M does not influence apoptotic response of (177)Lu-octreotate in GOT1 neuroendocrine tumors. Sci. Rep. 2023, 13, 6417. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).