Myocutaneous Flaps and Muscle Flaps for Management of Limbs’ Defects in Dogs and Cats: A Review
Abstract
1. Introduction
2. Materials and Methods
3. A. Myocutaneous Flaps
3.1. Forelimbs
3.1.1. Latissimus Dorsi Myocutaneous Flap
Anatomy
Vascularization
Indications
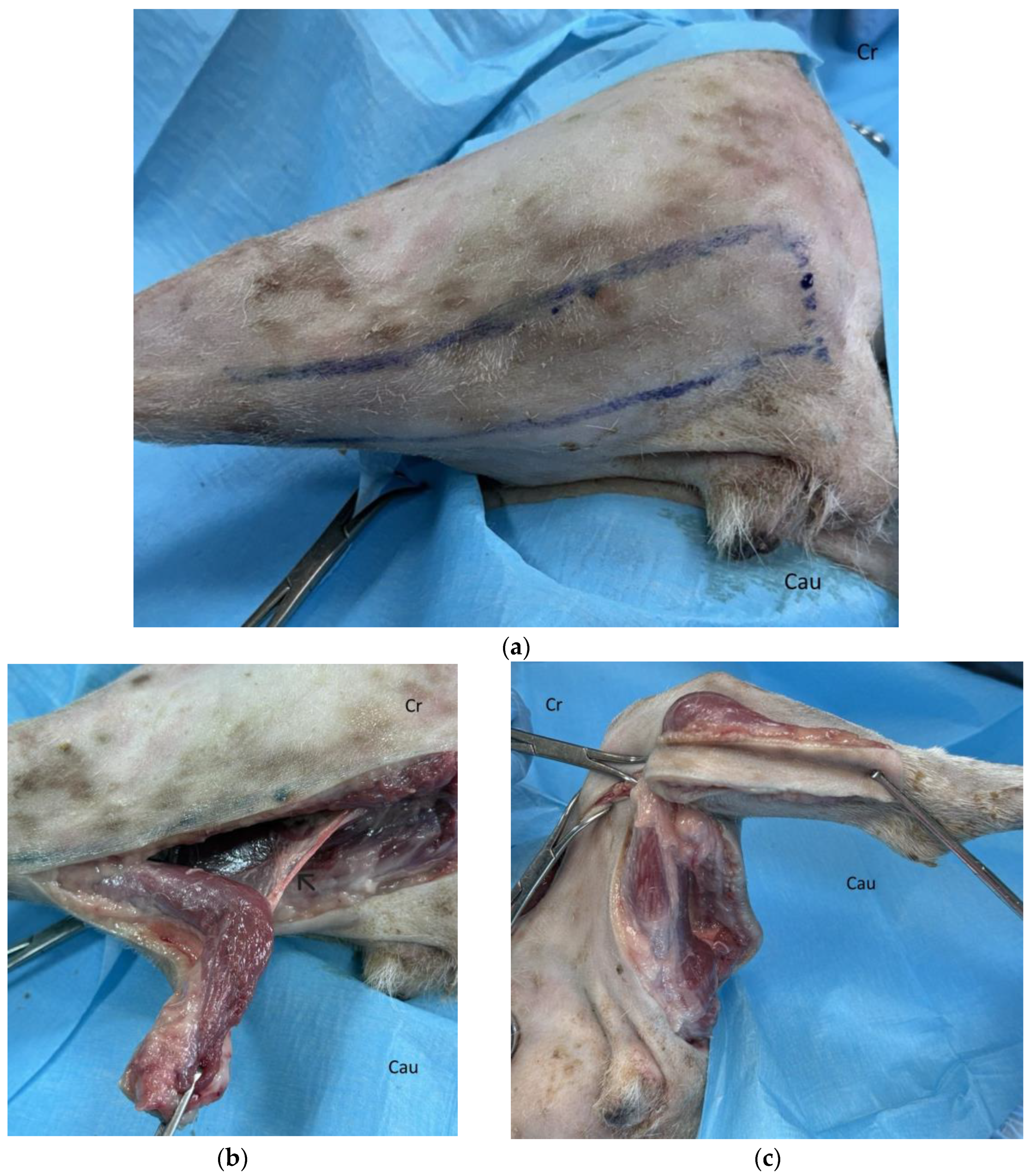
Surgical Technique
Reported Outcomes
3.1.2. Cutaneous Trunci Myocutaneous Flap
Anatomy
Vascularization
Indications
Surgical Technique
Reported Outcomes
3.1.3. Trapezius Myocutaneous Flap
Anatomy
Vascularization
Indications
Surgical Technique
Reported Outcomes
3.2. Hindlimbs
3.2.1. Semitendinosus Myocutaneous Flap
Anatomy
Vascularization
Indications
Surgical Technique
Reported Outcomes
4. B. Muscle Flaps
4.1. Forelimbs
4.1.1. Flexor Carpi Ulnaris Muscle Flap
Anatomy
Vascularization
Indications
Surgical Technique
Reported Outcomes
4.2. Hindlimbs
4.2.1. Caudal Sartorius Muscle Flap
Anatomy
Vascularization
Indications
Surgical Technique
Reported Outcomes
4.2.2. Cranial Sartorius Muscle Flap
Anatomy
Vascularization
Indications
Surgical Technique
Reported Outcomes
4.2.3. Semitendinosus Muscle Flap
Indications
Surgical Technique
Reported Outcomes
5. C. Postoperative Management of Myocutaneous and Muscle Flaps
5.1. Analgesia
5.2. Antibiotic Prophylaxis
5.3. Wound Protection and Monitoring
5.4. Activity Restriction
6. D. Complications
7. Conclusions
Future Directions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| TRAM | Transverse Rectus Abdominis Myocutaneous |
| DIEP | Deep Inferior Epigastric Artery Perforator |
| NSAIDs | nonsteroidal anti-inflammatory drugs |
References
- Swaim, S.F.; Henderson, R.A., Jr. Wounds on Limbs. In Small Animal Wound Management, 2nd ed.; Williams & Wilkins: Baltimore, MD, USA, 1997; pp. 295–370. [Google Scholar]
- Fowler, D. Distal limb and paw injuries. Vet. Clin. N. Am. Small Anim. Pract. 2006, 36, 819–845. [Google Scholar] [CrossRef] [PubMed]
- Beardsley, S.L.; Schrader, S.C. Treatment of dogs with wounds of the limbs caused by shearing forces: 98 cases (1975–1993). J. Am. Vet. Med. Assoc. 1995, 207, 1071–1075. [Google Scholar] [CrossRef] [PubMed]
- Corr, S. Intensive, extensive, expensive. Management of distal limb shearing injuries in cats. J. Feline Med. Surg. 2009, 9, 747–757. [Google Scholar] [CrossRef]
- Hosgood, G. Stages of wound healing and their clinical relevance. Vet. Clin. N. Am. Small. Anim. Pract. 2006, 36, 667–685. [Google Scholar] [CrossRef]
- Dermisiadou, E.; Panopoulos, I.; Psalla, D.; Georgiou, S.; Sideri, A.; Galatos, A.; Tsioli, V. Use of a semitendinosus myocutaneous flap for the coverage of hindlimb full-thickness skin defects in cats. J. Vet. Sci. 2023, 24, 14. [Google Scholar] [CrossRef] [PubMed]
- Mayhew, P.D. Tension-Relieving Techniques and Local Skin Flaps. In BSAVA Manual of Canine and Feline Wound Management and Reconstruction, 2nd ed.; British Small Animal Veterinary Association: Gloucester, UK, 2009; pp. 69–99. [Google Scholar]
- Stanley, B.J. Tension-Relieving Techniques. In Veterinary Surgery: Small Animal, 1st ed.; Tobias, K.M., Johnston, S.A., Eds.; Elsevier: St. Louis, MO, USA, 2012; pp. 1221–1242. [Google Scholar]
- Fahie, M.A. Primary Wound Closure. In Veterinary Surgery: Small Animal, 1st ed.; Tobias, K.M., Johnston, S.A., Eds.; Elsevier: St. Louis, MO, USA, 2012; pp. 1197–1209. [Google Scholar]
- Pavletic, M.M. Atlas of Small Animal Wound Management and Reconstructive Surgery, 4th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2018; pp. 554–590. [Google Scholar]
- Remedios, A.M.; Bauer, M.S.; Bowen, C.V. Thoracodorsal and caudal superficial epigastric axial pattern skin flaps in cats. Vet. Surg. 1989, 18, 380–385. [Google Scholar] [CrossRef]
- Pavletic, M.M. Introduction to Myocutaneous and Muscle Flaps. Vet. Clin. N. Am. Small Anim. Pract. 1990, 20, 127–146. [Google Scholar] [CrossRef]
- Remedios, A. Axial Pattern Flaps. In Manual of Canine and Feline Wound Management and Reconstruction; Fowler, D., Williams, J., Eds.; British Small Animal Veterinary Association: Cheltenham, UK, 1999; pp. 69–81. [Google Scholar]
- Degner, D.A.; Walshaw, R.; Arnoczky, S.P.; Smith, R.J.; Patterson, J.S.; Degner, L.A.; Hamaide, A.; Rosenstein, D. Evaluation of the cranial rectus abdominus muscle pedicle flap as a blood supply for the caudal superficial epigastric skin flap in dogs. Vet. Surg. 1996, 25, 292–299. [Google Scholar] [CrossRef]
- Lemarié, R.J.; Hosgood, G.; Read, R.A.; Lewis, D.D.; Bellah, R.; Salisbury, S.K.; Goldsmid, S. Distant abdominal and thoracic pedicle skin flaps for treatment of distal limb skin defects. J. Small Anim. Pract. 1995, 36, 255–261. [Google Scholar] [CrossRef]
- Swaim, S.F.; Bradley, D.M.; Steiss, J.E.; Powers, R.D.; Buxton, D.F. Free segmental paw pad grafts in dogs. Am. J. Vet. Res. 1993, 54, 2161–2170. [Google Scholar] [CrossRef]
- Shahar, R.; Shamir, M.H.; Brehm, D.M.; Johnston, D.E. Free skin grafting for treatment of distal limb skin defects in cats. J. Small Anim. Pract. 1999, 40, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Swaim, S.F. Skin Grafts. In Textbook of Small Animal Surgery, 3rd ed.; Slatter, D., Ed.; W. B. Saunders: Philadelphia, PA, USA, 2003; pp. 321–338. [Google Scholar]
- Miller, C.W. Free skin flap transfer by microvascular anastomosis. Vet. Clin. N. Am. Small Anim. Pract. 1990, 20, 189–199. [Google Scholar] [CrossRef]
- Camps, G.; Kirpensteijn, J. Avascular and Microvascular Reconstructive Techniques. In Reconstructive Surgery and Wound Management of the Dog and Cat; Kirpensteijn, J., Haar, G., Eds.; Manson Publishing Ltd.: London, UK, 2013; pp. 77–94. [Google Scholar]
- Madison, J.; Donawick, W.; Johnston, D.; Orsini, R. The use of skin expansion to repair cosmetic defects in animals. Vet. Surg. 1989, 18, 15–21. [Google Scholar] [CrossRef]
- Johnston, D.E. Wound Healing in Skin. Vet. Clin. N. Am. Small Anim. Pract. 1990, 20, 1–25. [Google Scholar] [CrossRef]
- Spodnick, G.J.; Pavletic, M.M.; Clark, G.N.; Schelling, S.H.; Kraus, K.H. Controlled tissue expansion in the distal extremities of dogs. Vet. Surg. 1993, 22, 436–443. [Google Scholar] [CrossRef]
- Keller, W.G.; Aron, D.N.; Rakich, P.M.; Crowe, D.T.; Marks, M.A. Rapid tissue expansion for the development of rotational skin flaps in the distal portion of the hindlimb of dogs: An experimental study. Vet. Surg. 1994, 23, 31–39. [Google Scholar] [CrossRef]
- Tsioli, V.; Papazoglou, L.G.; Papaioannou, N.; Psalla, D.; Savvas, I.; Pavlidis, L.; Karayannopoulou, M. Comparison of three skin stretching devices for closing skin defects on the limbs of dogs. J. Vet. Sci. 2015, 16, 99–106. [Google Scholar] [CrossRef] [PubMed]
- McCraw, J.B.; Dibbell, D.G.; Carraway, J.H. Clinical definition of independent myocutaneous vascular territories. Plast. Reconstr. Surg. 1977, 60, 341–352. [Google Scholar] [PubMed]
- Ariyan, S.; Cuono, C.B. Myocutaneous Flaps for Head and Neck Reconstruction. Head Neck Surg. 1980, 2, 321–345. [Google Scholar] [CrossRef] [PubMed]
- Pavletic, M.M. Textbook of Small Animal Surgery, 3rd ed.; Saunders–Elsevier: Philadelphia, PA, USA, 2003; pp. 292–321. [Google Scholar]
- Dermisiadou, E. Management of Complex Skin Defects on the Feline Distal Tibia by Using Myocutaneous Flaps. Ph.D. Thesis, University of Thessaly, Karditsa, Greece, 2022. [Google Scholar]
- Dermisiadou, E.; Panopoulos, I.; Psalla, D.; Georgiou, S.; Sideri, A.; Galatos, A.; Tsioli, V. Comparison of Two Surgical Techniques Based on the Semitendinosus Myocutaneous Flap in Cats. Vet. Sci. 2023, 11, 6. [Google Scholar] [CrossRef] [PubMed]
- Mathes, S.J.; Nahai, F. Classification of the vascular anatomy of muscles: Experimental and clinical correlation. Plast. Reconstr. Surg. 1981, 2, 177–187. [Google Scholar] [CrossRef]
- Geddes, C.R.; Morris, S.F.; Neligan, P.C. Perforator flaps: Evolution, classification, and applications. Ann. Plast. Surg. 2003, 50, 90–99. [Google Scholar] [CrossRef]
- Chen, S.L.; Chen, T.M.; Chou, T.D.; Chang, S.C.; Wang, H.J. Distally based sural fasciomusculocutaneous flap for chronic calcaneal osteomyelitis in diabetic patients. Ann. Plastic Surg. 2005, 54, 44–48. [Google Scholar] [CrossRef]
- De Mello-Filho, F.V.; Mamede, R.C.; Sader, A.A.; Velludo, M.A.; Vicente, W.V. Use of the platysma myocutaneous flap for cervical trachea reconstruction: An experimental study in dogs. Laryngoscope 1993, 103, 1161–1167. [Google Scholar] [CrossRef]
- Smith, Μ.; Shults, S.; Waldron, D.R.; Moon, M.L. Platysma myocutaneous flap for head and neck reconstruction in cats. Head Neck 1993, 15, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Halfacree, Z.J.; Baines, S.J.; Lipscomb, V.J.; Grierson, J.; Summers, B.A.; Brockman, D.J. Use of a latissimus dorsi myocutaneous flap for one-stage reconstruction of the thoracic wall after en bloc resection of primary rib chondrosarcoma in five dogs. Vet. Surg. 2007, 36, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Pavletic, M.M.; Kostolich, M.; Koblik, P.; Engler, S. A comparison of the cutaneous trunci myocutaneous flap and latissimus dorsi myocutaneous flap in the dog. Vet. Surg. 1987, 16, 283–293. [Google Scholar] [CrossRef]
- Doyle, C.P.; Degner, D.A. Evaluation of the Superior Labial Musculomucosal Flap in Dogs: An Angiographic Study and Case Report. Vet. Comp. Orthop. Traumatol. 2019, 32, 133–138. [Google Scholar] [PubMed]
- Nelissen, P.; White, D. Flaps and Grafts. In Feline Soft Tissue and General Surgery; Langley-Hobbs, S.J., Demetriou, J.L., Landlow, J.F., Eds.; Saunders: Philadelphia, PA, USA, 2014; pp. 195–207. [Google Scholar]
- MacPhail, C.M.; Fossum, T.W. Small Animal Surgery, 5th ed.; Elsevier: Philadelphia, PA, USA, 2018; pp. 179–265. [Google Scholar]
- Puerto, D.A.; Aronson, L.R. Use of a semitendinosus myocutaneous flap for soft-tissue reconstruction of a grade IIIB open tibial fracture in a dog. Vet. Surg. 2004, 33, 629–635. [Google Scholar] [CrossRef]
- Baltzer, W.I.; Rist, P. Achilles tendon repair in dogs using the semitendinosus muscle: Surgical technique and short-term outcome in five dogs. Vet. Surg. 2009, 38, 770–779. [Google Scholar] [CrossRef]
- Nicoll, S.A.; Fowler, J.D.; Remedios, A.M.; Clapson, J.B.; George, D. Development of a free latissimus dorsi muscle flap in cats. Vet. Surg. 1996, 25, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Mathes, S.J.; Levine, J. Muscle Flaps and Their Blood Supply. In Grabb & Smith’s Plastic Surgery, 6th ed.; Thorne, C.H., Beasley, R.W., Aston, S.J., Bartlett, S.P., Gurtner, G.C., Spear, S.L., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007. [Google Scholar]
- Hermanson, J.W. The Muscular System. In Miller’s Anatomy of the Dog, 4th ed.; Evans, H.E., de Lahunta, A., Eds.; Saunders: St. Louis, MO, USA, 2013; pp. 250–251. [Google Scholar]
- Hayden, R.E.; Kirby, S.D.; Deschler, D.G. Technical Modifications of the Latissimus Dorsi Pedicled Flap to Increase Versatility and Viability. Laryngoscope 2000, 110, 352–357. [Google Scholar] [CrossRef]
- Ong, H.S.; Ji, T.; Zhang, C.P. The pedicled latissimus dorsi myocutaneous flap in head and neck reconstruction. Oral Maxillofac. Surg. Clin. N. Am. 2014, 3, 427–434. [Google Scholar] [CrossRef]
- Motegi, S.; Tamura, A.; Abe, M.; Okada, E.; Nagai, Y.; Ishikawa, O. Reverse latissimus dorsi musculocutaneous flap for reconstruction of lumbar radiation ulcer. J. Dermatol. 2007, 34, 565–569. [Google Scholar] [CrossRef]
- Uzun, H.; Bitik, O.; Ersoy, U.S.; Bilginer, B.; Aksu, A.E. Comparison of Musculocutaneous and Fasciocutaneous Free Flaps for the Reconstruction of the Extensive Composite Scalp and Cranium Defects. J. Craniofac. Surg. 2018, 29, 1947–1951. [Google Scholar] [CrossRef]
- Vincent, A.; Hohman, M.H. Latissimus Dorsi Flap. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Tobin, G.R.; Moberg, A.W.; DuBou, R.H.; Weiner, L.J.; Bland, K.I. The split latissimus dorsi myocutaneous flap. Ann. Plast. Surg. 1981, 7, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Marshall, D.R.; Anstee, E.J.; Stapleton, M.J. Soft tissue reconstruction of the breast using an extended composite latissimus dorsi myocutaneous flap. Br. J. Plast. Surg. 1984, 37, 361–368. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Qing, L.; Wu, P.; Ketheeswaran, S.; Yu, F.; Tang, J. Variations of Extended Latissimus Dorsi Musculocutaneous Flap for Reconstruction of Large Wounds in the Extremity. Orthop. Surg. 2022, 14, 2598–2606. [Google Scholar] [CrossRef]
- Yamamoto, K.; Yokota, K.; Higaki, K. Entire pharyngoesophageal reconstruction with latissimus dorsi myocutaneous island flap. Head Neck Surg. 1985, 7, 461–464. [Google Scholar] [CrossRef]
- Ju, J.; Li, L.; Zhou, R.; Hou, R. Combined application of latissimus dorsi myocutaneous flap and iliac bone flap in the treatment of chronic osteomyelitis of the lower extremity. J. Orthop. Surg. Res. 2018, 13, 117. [Google Scholar] [CrossRef]
- Qi, J.W.; Chen, S.; Sun, B.H.; Chai, Y.T.; Huang, J.; Li, Y.; Yang, K.Y.; Sun, H.Y.; Chen, H. Clinical effects of free latissimus dorsi myocutaneous flap combined with artificial dermis and split-thickness skin graft in the treatment of degloving injury in lower extremity. Zhonghua Shao Shang Yu Chuang Mian Xiu Fu Za Zhi 2022, 38, 347–353. [Google Scholar]
- Zhou, J.; Zheng, Y.; Xiao, S.; Wei, Z.; Nie, K.; Liu, Z.; Chang, S.; Jin, W.; Chen, W.; Zhang, F. Preliminary application of foldable pedicled latissimus dorsi myocutaneous flap for repairing soft tissue defects in shoulder and back. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2024, 38, 69–73. [Google Scholar]
- Schwabegger, A.H.; Harpf, C.; Rainer, C. Muscle-sparing latissimus dorsi myocutaneous flap with maintenance of muscle innervation, function, and aesthetic appearance of the donor site. Plast. Reconstr. Surg. 2003, 111, 1407–1411. [Google Scholar] [CrossRef] [PubMed]
- Werker, P.M.; Kon, M. Cutaneous trunci myocutaneous flap: A new and reliable model for muscle free flap research in the rat. Microsurgery. 1992, 13, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Philibert, D.; Fowler, J.D.; Clapson, J.B. The anatomic basis for a trapezius muscle flap in dogs. Vet. Surg. 1992, 21, 429–434. [Google Scholar] [CrossRef]
- Kurach, L.; Smith, M.E.; Fowler, J.D. Microvascular free tissue transfer of the trapezius flap in 20 dogs and a wallaby. Vet. Surg. 2013, 42, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Panje, W.R. Myocutaneous trapezius flap. Head Neck Surg. 1980, 2, 206–212. [Google Scholar] [CrossRef]
- Philibert, D.; Fowler, J.D.; Clapson, J.B. Free microvascular transplantation of the trapezius musculocutaneous flap in dogs. Vet. Surg. 1992, 21, 435–440. [Google Scholar] [CrossRef]
- Chambers, J.N.; Rawlings, C.A. Applications of a semitendinosus muscle flap in two dogs. J. Am. Vet. Med. Assoc. 1991, 199, 84–86. [Google Scholar] [CrossRef]
- Solano, M.; Purinton, P.T.; Chambers, J.N.; Munnell, J.F. Effects of vascular pedicle ligation on blood flow in canine semitendinosus muscle. Am. J. Vet. Res. 1995, 56, 731–735. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Nakagawa, T.; Nishimura, R. Evaluation of semitendinosus muscle transposition for treatment of perineal hernias in 33 small-breed dogs. Can. J. Vet. Res. 2023, 87, 282–289. [Google Scholar]
- Wysocki, R.W.; Gray, R.L.; Fernandez, J.J.; Cohen, M.S. Posterior elbow coverage using whole and split flexor carpi ulnaris flaps: A cadaveric study. J. Hand Surg. Am. 2008, 33, 1807–1812. [Google Scholar] [CrossRef]
- Franini, A.; Entani, M.G.; Colosio, E.; Melotti, L.; Patruno, M. Case report: Flexor carpi ulnaris tendinopathy in a lure-coursing dog treated with three platelet-rich plasma and platelet lysate injections. Front. Vet. Sci. 2023, 10, 1003993. [Google Scholar] [CrossRef]
- Chambers, J.N.; Purinton, P.T.; Allen, S.W.; Schneider, T.A.; Smith, J.D. Flexor carpi ulnaris (humeral head) muscle flap for reconstruction of distal forelimb injuries in two dogs. Vet. Surg. 1998, 27, 342–347. [Google Scholar] [CrossRef]
- Purinton, P.T.; Chambers, J.N.; Moore, J.L. Identification and categorization of the vascular patterns to muscles of the thoracic limb, thorax, and neck of dogs. Am. J. Vet. Res. 1992, 53, 1435–1445. [Google Scholar] [CrossRef]
- Lim, A.Y.; Kumar, V.P.; Hua, J.; Pereira, B.P.; Pho, R.W. The neuromuscular compartments of the flexor carpi ulnaris. Plast. Reconstr. Surg. 1999, 103, 1046–1051. [Google Scholar] [CrossRef]
- Lingaraj, K.; Ruben, M.; Chan, Y.H.; Das, S.D. Identification of risk factors for urinary retention following total knee arthroplasty: A Singapore hospital experience. Singapore Med. J. 2007, 48, 213–216. [Google Scholar]
- Payne, D.E.; Kaufman, A.M.; Wysocki, R.W.; Richard, M.J.; Ruch, D.S.; Leversedge, F.J. Vascular perfusion of a flexor carpi ulnaris muscle turnover pedicle flap for posterior elbow soft tissue reconstruction: A cadaveric study. J. Hand Surg. Am. 2011, 36, 246–251. [Google Scholar] [CrossRef]
- Yang, K.; Rivedal, D.; Boehm, L.; Yan, J.G.; Sanger, J.; Matloub, H. Distally Based Pedicled Flexor Carpi Ulnaris Muscle Flap: An Anatomical Study and Clinical Application. Hand 2019, 14, 121–126. [Google Scholar] [CrossRef]
- Okamoto, S.; Tada, K.; Ai, H.; Tsuchiya, H. Flexor carpi ulnaris muscle flap for soft tissue reconstruction after total elbow arthroplasty. Case Rep. Surg. 2014, 2014, 798506. [Google Scholar] [CrossRef]
- Weinstein, M.J.; Pavletic, M.M.; Boudrieau, R.J. Caudal sartorius muscle flap in the dog. Vet. Surg. 1988, 17, 203–210. [Google Scholar] [CrossRef]
- Tanaka, C.; Ide, M.R.; Junior, A.J. Anatomical contribution to the surgical construction of the sartorius muscle flap. Surg. Radiol. Anat. 2006, 28, 277–283. [Google Scholar] [CrossRef]
- Sylvestre, A.M.; Weinstein, M.J.; Popovitch, C.A.; Brockman, D.J. The sartorius muscle flap in the cat: An anatomic study and two case reports. J. Am. Anim. Hosp. Assoc. 1997, 33, 91–96. [Google Scholar] [CrossRef]
- Yang, D.; Morris, S.F.; Sigurdson, L. The sartorius muscle: Anatomic considerations for reconstructive surgeons. Surg. Radiol. Anat. 1998, 20, 307–310. [Google Scholar] [CrossRef]
- Manjunath, K.N.; Venkatesh, M.S.; Shivaprasad, A. Distal major pedicle of sartorius muscle flap: Anatomical study and its clinical implications. Indian J. Plast. Surg. 2018, 51, 40–45. [Google Scholar] [CrossRef]
- Hong, J.P.; Lee, H.B.; Chung, Y.K.; Kim, S.W.; Tark, K.C. Coverage of difficult wounds around the knee joint with prefabricated, distally based sartorius muscle flaps. Ann. Plast. Surg. 2003, 50, 484–490. [Google Scholar] [CrossRef]
- Minami, N.; Tsukamoto, S.; Shimizu, T.; Honoki, K.; Hasegawa, H.; Masunaga, T.; Kido, A.; Errani, C.; Mavrogenis, A.F.; Tanaka, Y. Coverage of lateral soft tissue defects with sartorius muscle flap after distal femoral replacement for malignant bone tumors. SICOT J. 2024, 10, 27. [Google Scholar] [CrossRef]
- Mejia, S.; Boston, S.E.; Skinner, O.T. Sartorius muscle flap for body wall reconstruction: Surgical technique description and retrospective case series. Can. Vet. J. 2018, 59, 1187–1194. [Google Scholar]
- Weinstein, M.J.; Pavletic, M.M.; Boudrieau, R.J.; Engler, S.J. Cranial sartorius muscle flap in the dog. Vet. Surg. 1989, 18, 286–291. [Google Scholar] [CrossRef]
- Chambers, J.N.; Purinton, P.T.; Moore, J.L.; Allen, S.W. Treatment of trochanteric ulcers with cranial sartorius and rectus femoris muscle flaps. Vet. Surg. 1990, 19, 424–428. [Google Scholar] [CrossRef]
- Feng, Y.C.; Chen, K.S.; Chang, S.C. Reconstruction with latissimus dorsi, external abdominal oblique and cranial sartorius muscle flaps for a large defect of abdominal wall in a dog after surgical removal of infiltrative lipoma. J. Vet. Med. Sci. 2016, 78, 1717–1721. [Google Scholar] [CrossRef][Green Version]
- Doust, R.; Sullivan, M. Semitendinosus muscle transfer flap for external anal sphincter incompetence in a dog. J. Am. Vet. Med. Assoc. 2003, 222, 1385–1387. [Google Scholar] [CrossRef]
- Mortari, A.C.; Rahal, S.C.; Resende, L.A.; Dal-pai-silva, M.; Mamprim, M.J.; Corrêa, M.A.; Antunes, S.H. Electromyographical, ultrasonographical and morphological modifications in semitendinous muscle after transposition as ventral perineal muscle flap. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2005, 52, 359–365. [Google Scholar] [CrossRef]
- Riggs, J.; Ladlow, J.F.; Owen, L.J.; Hall, J.L. Novel application of internal obturator and semitendinosus muscle flaps for rectal wall repair or reinforcement. J. Small Anim. Pract. 2019, 60, 191–197. [Google Scholar] [CrossRef]
- Morello, E.; Martano, M.; Zabarino, S.; Piras, L.A.; Nicoli, S.; Bussadori, R.; Buracco, P. Modified semitendinosus muscle transposition to repair ventral perineal hernia in 14 dogs. J. Small Anim. Pract. 2015, 56, 370–376. [Google Scholar] [CrossRef]
- Vnuk, D.; Babić, T.; Stejskal, M.; Capak, D.; Harapin, I.; Pirkić, B. Application of a semitendinosus muscle flap in the treatment of perineal hernia in a cat. Vet. Rec. 2005, 156, 182–184. [Google Scholar] [CrossRef]
- Kim, M.Y.; Nam, C.H.; Kim, J.H.; Yoon, H.Y. Application of Bifurcated Semitendinosus Muscle Transposition for Treatment of Fecal Incontinence in Two Dogs. Vet. Sci. 2023, 10, 150. [Google Scholar] [CrossRef]
- Field, E.J.; Kelly, G.; Pleuvry, D.; Demetriou, J.; Baines, S.J. Indications, outcome and complications with axial pattern skin flaps in dogs and cats: 73 cases. J. Small Anim. Pract. 2015, 56, 698–706. [Google Scholar] [CrossRef]
- Jones, C.A.; Lipscomb, V.J. Indications, complications, and outcomes associated with subdermal plexus skin flap procedures in dogs and cats: 92 cases (2000–2017). J. Am. Vet. Med. Assoc. 2019, 255, 933–938. [Google Scholar] [CrossRef]













| Flap Type | Transposition/Arc of Rotation | Indications |
|---|---|---|
| Latissimus dorsi myocutaneous | Wide arc; cranioventral rotation to thorax/forelimb | Thoracic wall defects, large forelimb wounds |
| Cutaneous trunci myocutaneous | Moderate arc; rotated to elbow/antebrachial region | Forelimb defects up to elbow |
| Trapezius myocutaneous | Rotated to cervical/thoracic regions | Neck, upper thorax, proximal forelimb defects |
| Semitendinosus myocutaneous (limb reconstruction) | Wide arc of rotation 150–180° medially, laterally or distally | Distal hindlimb wounds, tibial/metatarsal defects, Achilles tendon rupture repair, Perineal hernia, fecal incontinence, rectal wall repair |
| Flexor carpi ulnaris muscle | Distally rotated to carpus/metacarpus | Antebrachial, carpal, and metacarpal soft tissue defects |
| Caudal sartorius muscle | Distally rotated to tibia/metatarsus | Distal tibial wounds, metatarsal defects, osteomyelitis |
| Cranial sartorius muscle | Simple rotation or inversion over trochanter | Trochanteric ulcers, abdominal wall reconstruction, femoral hernia |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Markou, M.; Dermisiadou, E.; Karagianni, K.; Flouraki, E.; Tsioli, V. Myocutaneous Flaps and Muscle Flaps for Management of Limbs’ Defects in Dogs and Cats: A Review. Pets 2025, 2, 41. https://doi.org/10.3390/pets2040041
Markou M, Dermisiadou E, Karagianni K, Flouraki E, Tsioli V. Myocutaneous Flaps and Muscle Flaps for Management of Limbs’ Defects in Dogs and Cats: A Review. Pets. 2025; 2(4):41. https://doi.org/10.3390/pets2040041
Chicago/Turabian StyleMarkou, Mandalena, Eleftheria Dermisiadou, Konstantina Karagianni, Eugenia Flouraki, and Vassiliki Tsioli. 2025. "Myocutaneous Flaps and Muscle Flaps for Management of Limbs’ Defects in Dogs and Cats: A Review" Pets 2, no. 4: 41. https://doi.org/10.3390/pets2040041
APA StyleMarkou, M., Dermisiadou, E., Karagianni, K., Flouraki, E., & Tsioli, V. (2025). Myocutaneous Flaps and Muscle Flaps for Management of Limbs’ Defects in Dogs and Cats: A Review. Pets, 2(4), 41. https://doi.org/10.3390/pets2040041







