Evaluating Dose Titration in Semaglutide and Tirzepatide for Weight Loss: A Retrospective Academic Call Center Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Setting and Population
2.3. Data Collection
2.4. Inclusion/Exclusion Criteria
2.5. Outcome Measures
2.6. Statistical Analysis
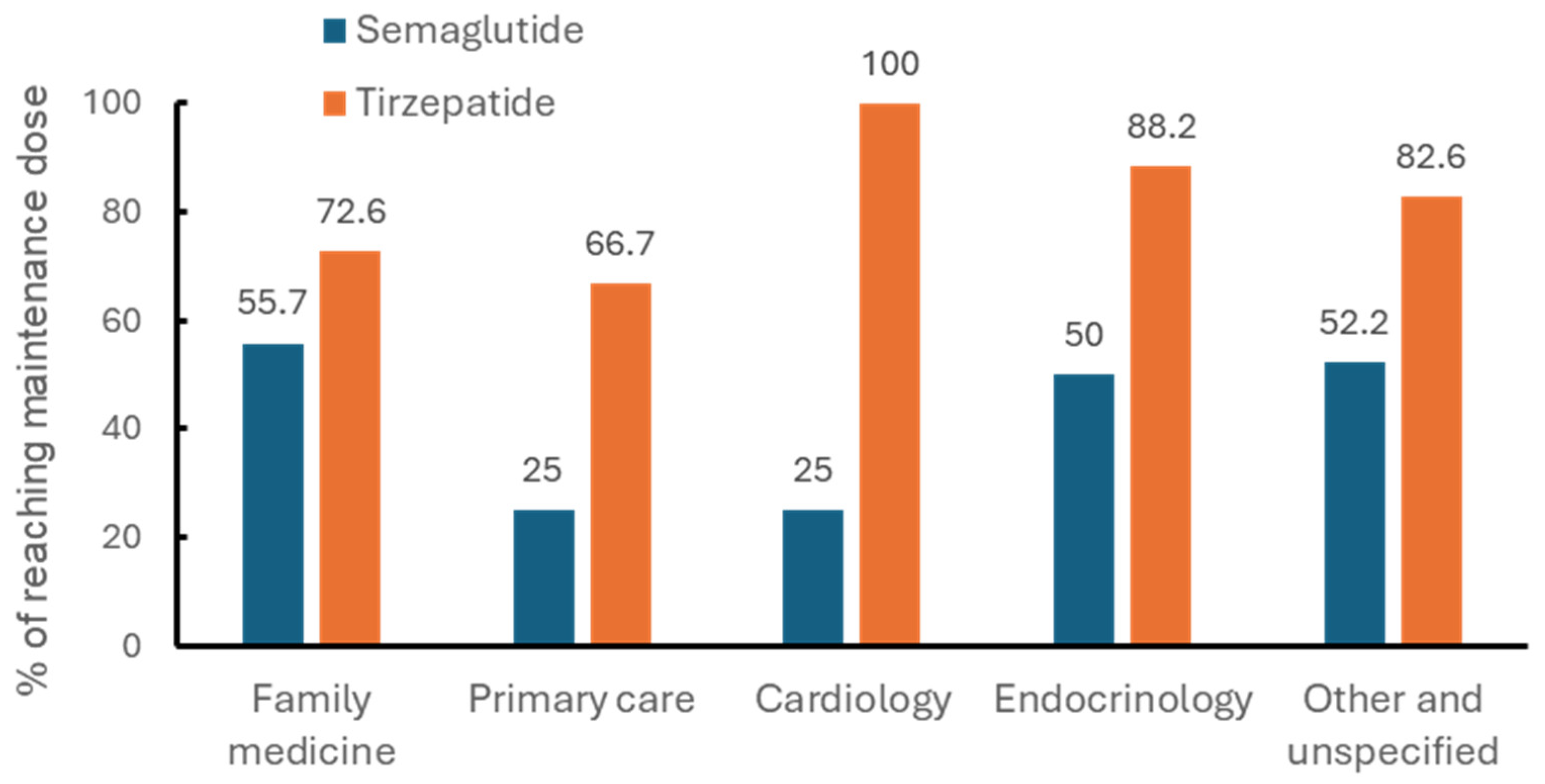
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Emmerich, S.; Fryar, C.; Stierman, B.; Ogden, C. Obesity and Severe Obesity Prevalence in Adults: United States, August 2021–August 2023; National Center for Health Statistics (U.S.): Hyattsville, Maryland, 2024. Available online: https://stacks.cdc.gov/view/cdc/159281 (accessed on 6 November 2025).
- Kushner, R.F.; Ryan, D.H. Assessment and Lifestyle Management of Patients with Obesity: Clinical Recommendations From Systematic Reviews. JAMA 2014, 312, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Apovian, C.M.; Aronne, L.J.; Bessesen, D.H.; McDonnell, M.E.; Murad, M.H.; Pagotto, U.; Ryan, D.H.; Still, C.D. Pharmacological Management of Obesity: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2015, 100, 342–362. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, O.; Gulati, M.; Gluckman, T.J.; Kittleson, M.M.; Rikhi, R.; Saseen, J.J.; Tchang, B.G. 2025 Concise Clinical Guidance: An ACC Expert Consensus Statement on Medical Weight Management for Optimization of Cardiovascular Health. J. Am. Coll. Cardiol. 2025, 86, 536–555. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. FDA Approves First Treatment to Reduce the Risk of Serious Heart Problems Specifically in Adults with Obesity or Overweight. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-treatment-reduce-risk-serious-heart-problems-specifically-adults-obesity-or (accessed on 6 August 2025).
- U.S. Food and Drug Administration. FDA Approves New Medication for Chronic Weight Management. 2023. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-new-medication-chronic-weight-management (accessed on 6 August 2025).
- Nauck, M.A.; Meier, J.J. Incretin hormones: Their role in health and disease. Diabetes Obes. Metab. 2018, 20, 5–21. [Google Scholar] [CrossRef] [PubMed]
- Frías, J.P.; Davies, M.J.; Rosenstock, J.; Pérez Manghi, F.C.; Fernández Landó, L.; Bergman, B.K.; Liu, B.; Cui, X.; Brown, K. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N. Engl. J. Med. 2021, 385, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Jastreboff, A.M.; Aronne, L.J.; Ahmad, N.N.; Wharton, S.; Connery, L.; Alves, B.; Kiyosue, A.; Zhang, S.; Liu, B.; Bunck, M.C.; et al. Tirzepatide Once Weekly for the Treatment of Obesity. N. Engl. J. Med. 2022, 387, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Wilding, J.P.H.; Batterham, R.L.; Calanna, S.; Davies, M.; Van Gaal, L.F.; Lingvay, I.; McGowan, B.M.; Rosenstock, J.; Tran, M.T.D.; Wadden, T.A.; et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N. Engl. J. Med. 2021, 384, 989–1002. [Google Scholar] [CrossRef] [PubMed]
- Rubino, D.; Abrahamsson, N.; Davies, M.; Hesse, D.; Greenway, F.L.; Jensen, C.; Lingvay, I.; Mosenzon, O.; Rosenstock, J.; Rubio, M.A.; et al. Effect of Continued Weekly Subcutaneous Semaglutide vs. Placebo on Weight Loss Maintenance in Adults With Overweight or Obesity: The STEP 4 Randomized Clinical Trial. JAMA 2021, 325, 1414. [Google Scholar] [CrossRef] [PubMed]
- Garvey, W.T.; Frias, J.P.; Jastreboff, A.M.; Le Roux, C.W.; Sattar, N.; Aizenberg, D.; Mao, H.; Zhang, S.; Ahmad, N.N.; Bunck, M.C.; et al. Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): A double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 2023, 402, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Samuels, J.M.; Ye, F.; Irlmeier, R.; Silver, H.; Srivastava, G.; Spann, M. Real-world titration, persistence & weight loss of semaglutide and tirzepatide in an academic obesity clinic. Diabetes Obes. Metab. 2025, 27, 6200–6209. [Google Scholar] [CrossRef] [PubMed]
- Müllertz, A.L.O.; Sandsdal, R.M.; Jensen, S.B.K.; Torekov, S.S. Potent incretin-based therapy for obesity: A systematic review and meta-analysis of the efficacy of semaglutide and tirzepatide on body weight and waist circumference, and safety. Obes. Rev. 2024, 25, e13717. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, R.W.; Mailhac, A.; Løhde, J.B.; Pottegård, A. Real-world evidence on the utilization, clinical and comparative effectiveness, and adverse effects of newer GLP-1RA-based weight-loss therapies. Diabetes Obes. Metab. 2025, 27, 66–88. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.J.; Goodwin Cartwright, B.M.; Gratzl, S.; Brar, R.; Baker, C.; Gluckman, T.J.; Stucky, N.L. Comparative Effectiveness of Semaglutide and Tirzepatide for Weight Loss in Adults with Overweight and Obesity in the US: A Real-World Evidence Study. MedRxiv 2023. [Google Scholar] [CrossRef]
- Brown, J.D.; Buscemi, J.; Milsom, V.; Malcolm, R.; O’Neil, P.M. Effects on cardiovascular risk factors of weight losses limited to 5–10%. Transl. Behav. Med. 2016, 6, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.J.; Goodwin Cartwright, B.M.; Gratzl, S.; Brar, R.; Baker, C.; Gluckman, T.J.; Stucky, N.L. Semaglutide vs. Tirzepatide for Weight Loss in Adults With Overweight or Obesity. JAMA Intern. Med. 2024, 184, 1056–1064. [Google Scholar] [CrossRef] [PubMed]


| Sex | Patients (n =662), n (%) | Prescriptions (n = 739) | |
|---|---|---|---|
| Semaglutide (n = 386), n (%) | Tirzepatide (n = 353), n (%) | ||
| Female | 418 (63.1) | 248 (64.3) | 229 (64.9) |
| Male | 244 (36.9) | 138 (35.7) | 124 (35.1) |
| Outcomes | Semaglutide (n = 386) | Tirzepatide (n = 353) | p-Value |
|---|---|---|---|
| Achieving maintenance dose, n (%) | 204 (52.9%) | 274 (77.6%) | <0.001 |
| Days to achieving maintenance dose, median (95% CI) | 143 (119, 173) | 32 (30, 38) | <0.001 |
| Monthly Cost per Patient ($), Mean (SD) | Total Monthly Cost ($) | |
|---|---|---|
| Semaglutide (n = 182) | $1330 (28) | $242,133 |
| Tirzepatide (n = 79) | $1060 (21) | $83,705 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alvarez, G.; Veliz, L.; Michaels, S.; Pino, D.; Wu, J. Evaluating Dose Titration in Semaglutide and Tirzepatide for Weight Loss: A Retrospective Academic Call Center Study. Obesities 2025, 5, 90. https://doi.org/10.3390/obesities5040090
Alvarez G, Veliz L, Michaels S, Pino D, Wu J. Evaluating Dose Titration in Semaglutide and Tirzepatide for Weight Loss: A Retrospective Academic Call Center Study. Obesities. 2025; 5(4):90. https://doi.org/10.3390/obesities5040090
Chicago/Turabian StyleAlvarez, Goar, Lianette Veliz, Stephanie Michaels, David Pino, and Jun Wu. 2025. "Evaluating Dose Titration in Semaglutide and Tirzepatide for Weight Loss: A Retrospective Academic Call Center Study" Obesities 5, no. 4: 90. https://doi.org/10.3390/obesities5040090
APA StyleAlvarez, G., Veliz, L., Michaels, S., Pino, D., & Wu, J. (2025). Evaluating Dose Titration in Semaglutide and Tirzepatide for Weight Loss: A Retrospective Academic Call Center Study. Obesities, 5(4), 90. https://doi.org/10.3390/obesities5040090





