Spatial Association of Canine Rabies Outbreak and Ecological Urban Corridors, Arequipa, Peru
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Setting
2.2. Data
2.3. Statistical Analysis
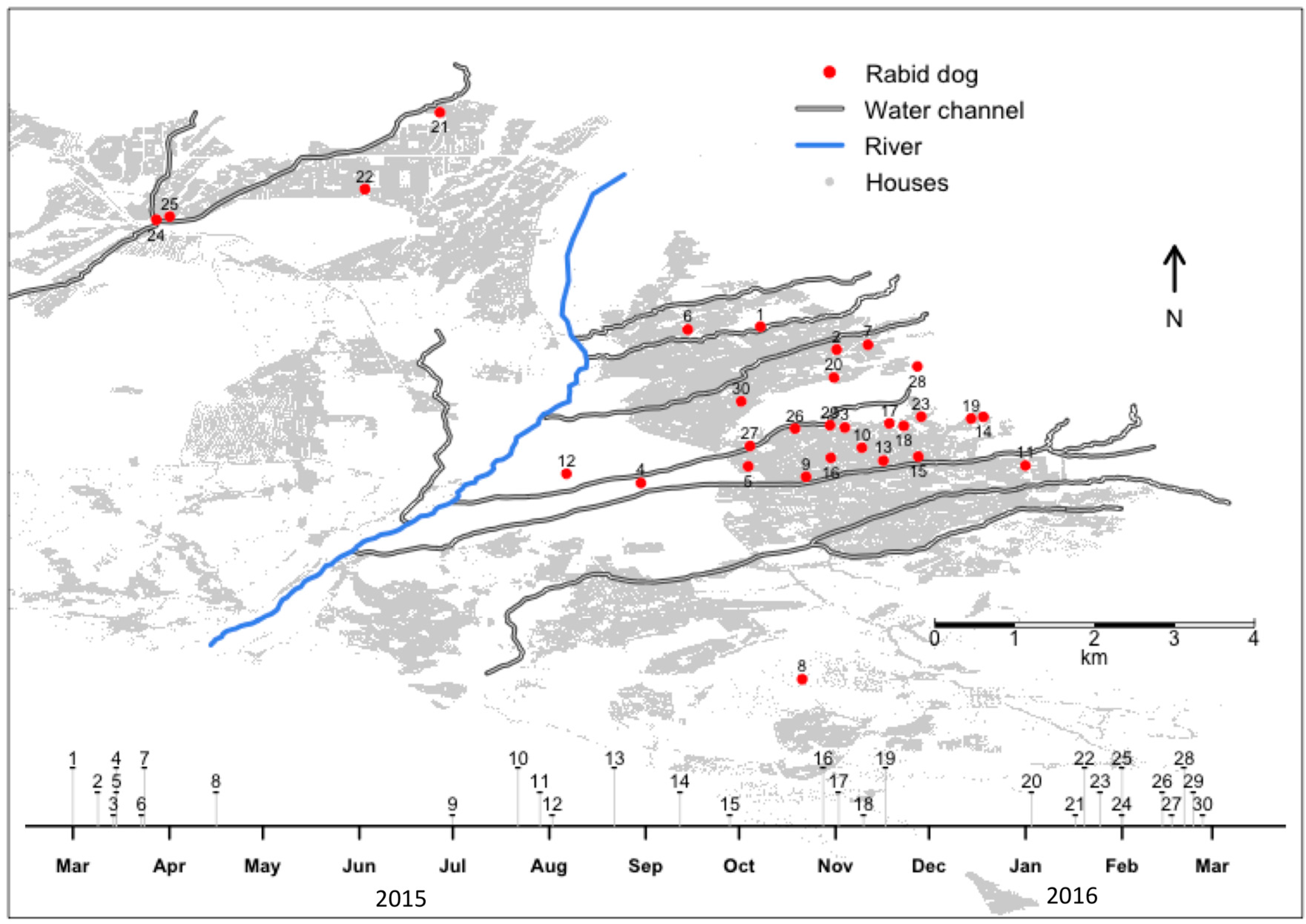
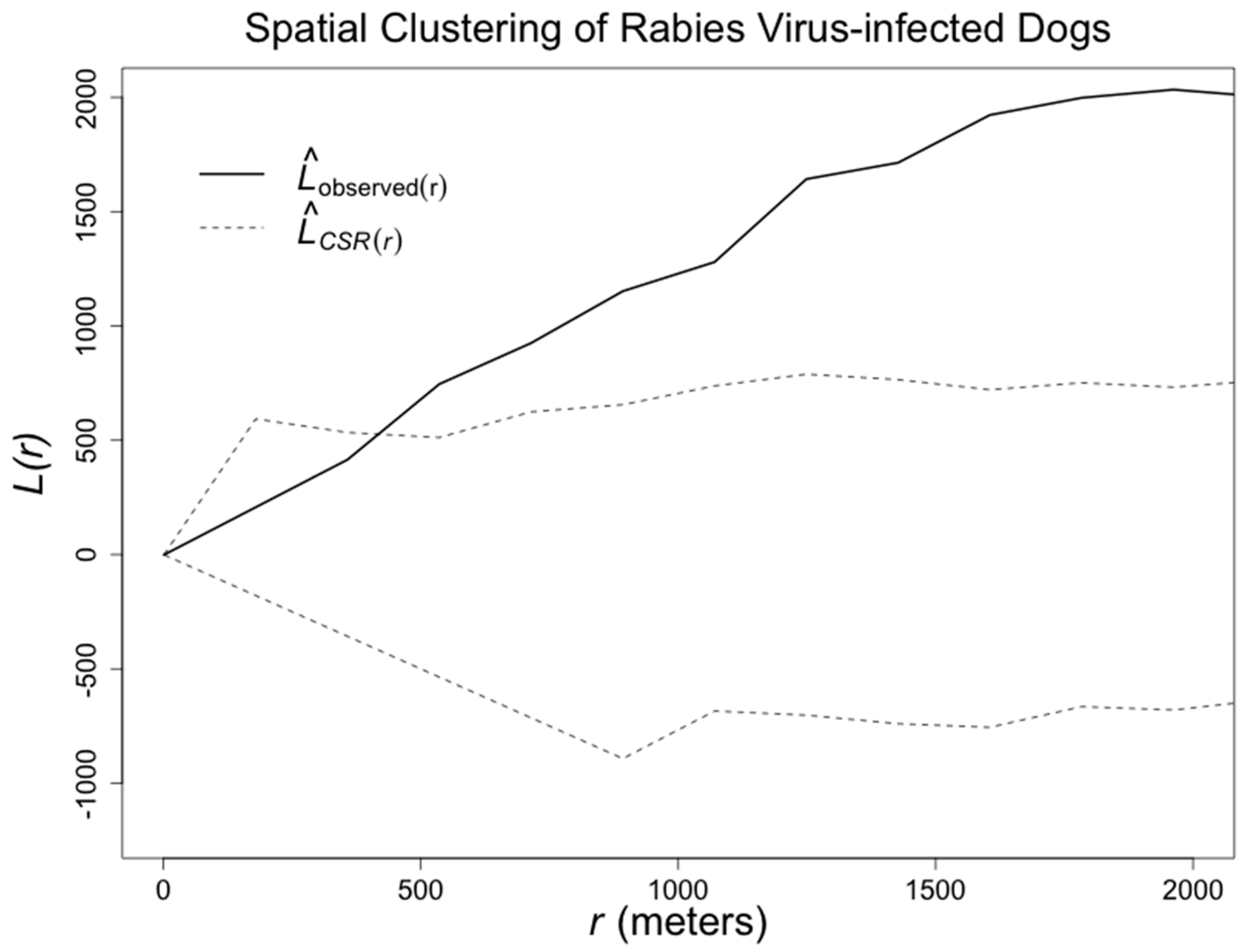
3. Results
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Clark, K.A.; Neill, S.U.; Smith, J.S.; Wilson, P.J.; Whadford, V.W.; McKirahan, G.W. Epizootic canine rabies transmitted by coyotes in south Texas. J. Am. Vet. Med. Assoc. 1994, 204, 536–540. [Google Scholar] [PubMed]
- Chang, S.-S.; Tsai, H.-J.; Chang, F.-Y.; Lee, T.-S.; Huang, K.-C.; Fang, K.-Y.; Wallace, R.M.; Inoue, S.; Fei, C.-Y. Government response to the discovery of a rabies virus reservoir species on a previously designated rabies-free Island, Taiwan, 1999–2014. Zoonoses Public Health 2016, 63, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Hu, R.; Zhang, Y.; Dong, G.; Rupprecht, C.E. Reemerging rabies and lack of systemic surveillance in People’s Republic of China. Emerg. Infect. Dis. 2009, 15, 1159–1164. [Google Scholar] [CrossRef] [PubMed]
- Knobel, D.L.; Lembo, T.; Morters, M.; Townsend, S.E.; Cleaveland, S.; Hampson, K. Dog rabies and Its control. In Rabies: Scientific Basis of the Disease and its Management, 3rd ed.; Elsevier: Philadelphia, PA, USA, 2013; pp. 591–615. [Google Scholar]
- Vigilato, M.A.N.; Clavijo, A.; Knobl, T.; Silva, H.M.T.; Cosivi, O.; Schneider, M.C.; Leanes, L.F.; Belotto, A.J.; Espinal, M.A. Progress towards eliminating canine rabies: Policies and perspectives from Latin America and the Caribbean. Philos. Trans. R. Soc. Lond. B 2013, 368, 20120143. [Google Scholar] [CrossRef] [PubMed]
- Dirección General. Alert for the presentation of cases of human rabies and expansion of canine rabies. In Epidemiología Epidemiologic Alert–Risk; Ministerio de Salud: Lima, Peru, 2015; pp. 1–2. [Google Scholar]
- Castillo-Neyra, R.; MacDonald, H.; Toledo Vizcarra, A.M.; Johnson, A.; Paja, W.; Buttenheim, A.M.; et al. University of Pennsylvania: Philadelphia, PA, USA, Unpublished work. 2017.
- Dirección General de Salud; Dirección de Atención Integral de Salud. Norma Técnica de Salud para la Prevención y Control de la Rabia Humana en el Perú [Technical Health Norm for the Prevention and Control of Human Rabies in Peru], 1st ed.; Ministerio de Salud: Lima, Peru, 2006; pp. 1–101.
- Aréchiga Ceballos, N.; Karunaratna, D.; Aguilar Setién, A. Control of canine rabies in developing countries: Key features and animal welfare implications. Rev. Sci. Tech. Off. Int. Epizoot. 2014, 33, 311–321. [Google Scholar] [CrossRef]
- Morters, M.K.; Restif, O.; Hampson, K.; Cleaveland, S.; Wood, J.L.N.; Conlan, A.J.K. Evidence-based control of canine rabies: A critical review of population density reduction. J. Anim. Ecol. 2013, 82, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Neyra, R.; Levy, M.Z.; Náquira, C. Effect of free-roaming dogs culling on the control of canine rabies. Rev. Peru. Med. Exp. Salud Publica 2016, 33, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Tasso, O.H.; Valdivia, A.V. Decreto supremo que declara en emergencia sanitaria por el plazo de noventa (90) días calendario, a la provincia de Arequipa. Available online: http://busquedas.elperuano.com.pe/normaslegales/decreto-supremo-que-declara-en-emergencia-sanitaria-por-el-p-decreto-supremo-n-015-2016-sa-1363166-5/ (accessed on 8 August 2017).
- Rosatte, R.C.; Power, M.J.; Donovan, D.; Davies, J.C.; Allan, M.; Bachmann, P.; Stevenson, B.; Wandeler, A.; Muldoon, F. Elimination of arctic variant rabies in red foxes, metropolitan Toronto. Emerg. Infect. Dis. 2007, 13, 25–27. [Google Scholar] [CrossRef] [PubMed]
- Tigas, L.A.; Van Vuren, D.H.; Sauvajot, R.M. Behavioral responses of bobcats and coyotes to habitat fragmentation and corridors in an urban environment. Biol. Conserv. 2002, 108, 299–306. [Google Scholar] [CrossRef]
- Freemark, K.; Bert, D.; Villard, M.-A. Patch-, landscape-, and regional-scale effects on Biota. In Applying Landscape Ecology in Biological Conservation, 1st ed.; Springer: New York, NY, USA, 2002; pp. 58–83. [Google Scholar]
- Campbell, T.; Campbell, A. Emerging disease burdens and the poor in cities of the developing world. J. Urban Health 2007, 84, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Neiderud, C.-J. How urbanization affects the epidemiology of emerging infectious diseases. Infect. Ecol. Epidemiol. 2015, 5, 27060. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.Z.; Barbu, C.M.; Castillo-Neyra, R.; Quispe Machaca, V.R.; Ancca Juárez, J.; Escalante-Mejia, P.; Borrini-Mayori, K.; Niemierko, M.; Mabud, T.S.; Behrman, J.R.; et al. Urbanization, land tenure security and vector-borne Chagas disease. Proc. Biol. Sci. 2014, 281, 20141003. [Google Scholar] [CrossRef] [PubMed]
- Duong, V.; Tarantola, A.; Ong, S.; Mey, C.; Choeung, R.; Ly, S.; Bourhyc, H.; Dussarta, P.; Buchyd, P. Laboratory diagnostics in dog-mediated rabies: An overview of performance and a proposed strategy for various settings. Int. J. Infect. Dis. 2016, 46, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Waller, L.A.; Gotway, C.A. Applied Spatial Statistics for Public Health Data; John Wiley & Sons: Hoboken, NJ, USA, 2004; pp. 1–482. [Google Scholar]
- Diggle, P.J. Statistical Analysis of Spatial Point Patterns, 2nd ed.; Hodder Education Publishers: London, UK, 2003; p. 159. [Google Scholar]
- Team, R. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; p. 3604. [Google Scholar]
- Gulachenski, A.; Ghersi, B.; Lesen, A.; Blum, M. Abandonment, ecological assembly and public health risks in counter-urbanizing cities. Sustainability 2016, 8, 491. [Google Scholar] [CrossRef]
- Vogt, P.; Riitters, K.H.; Iwanowski, M.; Estreguil, C.; Kozak, J.; Soille, P. Mapping landscape corridors. Ecol. Indic. 2007, 7, 481–488. [Google Scholar] [CrossRef]
- Rees, E.E.; Pond, B.A.; Tinline, R.R.; Bélanger, D. Understanding effects of barriers on the spread and control of rabies. Adv. Virus Res. 2011, 79, 421–447. [Google Scholar] [CrossRef] [PubMed]
- Riley, S.; Eames, K.; Isham, V.; Mollison, D.; Trapman, P. Five challenges for spatial epidemic models. Epidemics 2015, 10, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Townsend, S.E.; Lembo, T.; Cleaveland, S.; Meslin, F.X.; Miranda, M.E.; Putra, A.A.G.; Haydona, D.T.; Hampsona, K. Surveillance guidelines for disease elimination: A case study of canine rabies. Comp. Immunol. Microbiol. Infect. Dis. 2013, 36, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Hampson, K.; Dushoff, J.; Cleaveland, S.; Haydon, D.T.; Kaare, M.; Packer, C.; Dobson, A. Transmission dynamics and prospects for the elimination of canine rabies. PLoS Biol. 2009, 7, e53. [Google Scholar] [CrossRef] [PubMed]
- Coleman, P.G.; Dye, C. Immunization coverage required to prevent outbreaks of dog rabies. Vaccine 1996, 14, 185–186. [Google Scholar] [CrossRef]
- Townsend, S.E.; Sumantra, I.P.; Pudjiatmoko; Bagus, G.N.; Brum, E.; Cleaveland, S.; Crafter, S.; Dewi, A.P.M.; Dharma, D.M.N.; Dushoff, J.; et al. Designing programs for eliminating canine rabies from islands: Bali, Indonesia as a case study. PLoS Negl. Trop. Dis. 2013, 7, e2372. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Neyra, R.; Brown, J.; Borrini, K.; Arevalo, C.; Levy, M.Z.; Buttenheim, A.; Hunter, G.C.; Becerra, V.; Behrman, J.; Paz-Soldan, V.A. Barriers to dog rabies vaccination during an urban rabies outbreak: Qualitative findings from Arequipa, Peru. PLoS Negl. Trop. Dis. 2017, 11, e0005460. [Google Scholar] [CrossRef] [PubMed]
- Davlin, S.L.; Vonville, H.M. Canine rabies vaccination and domestic dog population characteristics in the developing world: A systematic review. Vaccine 2012, 30, 3492–3502. [Google Scholar] [CrossRef] [PubMed]
- Morters, M.K.; McKinley, T.J.; Restif, O.; Conlan, A.J.K.; Cleaveland, S.; Hampson, K.; Whay, H.R.; Damriyasa, I.M.; Wood, J.L.N. The demography of free-roaming dog populations and applications to disease and population control. J. Appl. Ecol. 2014, 51, 1096–1106. [Google Scholar] [CrossRef] [PubMed]
- Wera, E.; Velthuis, A.G.J.; Geong, M.; Hogeveen, H. Costs of rabies control: An economic calculation method applied to Flores Island. PLoS ONE 2013, 8, e83654. [Google Scholar] [CrossRef] [PubMed]
- Zinsstag, J.; Durr, S.; Penny, M.A.; Mindekem, R.; Roth, F.; Menendez Gonzalez, S.; Naissengard, S.; Hattendorfa, J. Transmission dynamics and economics of rabies control in dogs and humans in an African city. Proc. Natl. Acad. Sci. USA 2009, 106, 14996–15001. [Google Scholar] [CrossRef] [PubMed]
- Putra, A.A.G.; Hampson, K.; Girardi, J.; Hiby, E.; Knobel, D.; Mardiana, I.W.; Townsend, S.; Scott-Orr, H. Response to a rabies epidemic, Bali, Indonesia, 2008–2011. Emerg. Infect. Dis. 2013, 19, 648–651. [Google Scholar] [CrossRef] [PubMed]
- Dannenberg, A.L.; Jackson, R.J.; Frumkin, H.; Schieber, R.A.; Pratt, M.; Kochtitzky, C.; Tilson, H.H. The impact of community design and land-use choices on public health: A scientific research agenda. Am. Public Health 2003, 93, 1500–1508. [Google Scholar] [CrossRef]
- Nagarajan, T.; Mohanasubramanian, B.; Seshagiri, E.V.; Nagendrakumar, S.B.; Saseendranath, M.R.; Satyanarayana, M.L.; Thiagarajan, D.; Rangarajan, P.N.; Srinivasan, V.A. Molecular epidemiology of rabies virus isolates in India. J. Clin. Microbiol. 2006, 44, 3218–3224. [Google Scholar] [CrossRef] [PubMed]
- Duke, J.E.; Blanton, J.D.; Ivey, M.; Rupprecht, C. Modeling enzootic raccoon rabies from land use patterns-Georgia (USA) 2006-2010. F1000Research 2013, 2, 285. [Google Scholar] [CrossRef] [PubMed]
- Recuenco, S.; Eidson, M.; Cherry, B.; Kulldorff, M.; Johnson, G. Factors associated with endemic raccoon (Procyon lotor) rabies in terrestrial mammals in New York State, USA. Prev. Vet. Med. 2008, 86, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Arjo, W.M.; Fisher, C.E.; Armstrong, J.; Boyd, F.; Slate, D. Effects of natural barriers and habitat on the western spread of raccoon rabies in Alabama. J. Wildl. Manag. 2008, 72, 1725–1735. [Google Scholar] [CrossRef]
- Bourhy, H.; Kissi, B.; Audry, L.; Smreczak, M.; Sadkowska-Todys, M.; Kulonen, K.; Tordo, N.; Zmudzinski, J.F.; Holmes, E.C. Ecology and evolution of rabies virus in Europe. J. Gen. Virol. 1999, 80, 2545–2557. [Google Scholar] [CrossRef] [PubMed]
- Hampson, K.; Abela-Ridder, B.; Brunker, K.; Bucheli, S.T.M.; Carvalho, M.; Caldas, E.; Changalucha, J.; Cleaveland, S.; Dushoff, J.; Gutierrez, V.; et al. Surveillance to establish elimination of transmission and freedom from dog-mediated rabies. bioRxiv 2016, 096883. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castillo-Neyra, R.; Zegarra, E.; Monroy, Y.; Bernedo, R.F.; Cornejo-Rosello, I.; Paz-Soldan, V.A.; Levy, M.Z. Spatial Association of Canine Rabies Outbreak and Ecological Urban Corridors, Arequipa, Peru. Trop. Med. Infect. Dis. 2017, 2, 38. https://doi.org/10.3390/tropicalmed2030038
Castillo-Neyra R, Zegarra E, Monroy Y, Bernedo RF, Cornejo-Rosello I, Paz-Soldan VA, Levy MZ. Spatial Association of Canine Rabies Outbreak and Ecological Urban Corridors, Arequipa, Peru. Tropical Medicine and Infectious Disease. 2017; 2(3):38. https://doi.org/10.3390/tropicalmed2030038
Chicago/Turabian StyleCastillo-Neyra, Ricardo, Edith Zegarra, Ynes Monroy, Reyno F. Bernedo, Ismael Cornejo-Rosello, Valerie A. Paz-Soldan, and Michael Z. Levy. 2017. "Spatial Association of Canine Rabies Outbreak and Ecological Urban Corridors, Arequipa, Peru" Tropical Medicine and Infectious Disease 2, no. 3: 38. https://doi.org/10.3390/tropicalmed2030038
APA StyleCastillo-Neyra, R., Zegarra, E., Monroy, Y., Bernedo, R. F., Cornejo-Rosello, I., Paz-Soldan, V. A., & Levy, M. Z. (2017). Spatial Association of Canine Rabies Outbreak and Ecological Urban Corridors, Arequipa, Peru. Tropical Medicine and Infectious Disease, 2(3), 38. https://doi.org/10.3390/tropicalmed2030038




